Abstract
B cells are differentiated to recognize antigen and respond by producing antibodies. These activities, governed by recognition of ancillary signals, defend the individual against microorganisms and the products of microorganisms and constitute the canonical function of B cells. Despite the unique differentiation (e.g. recombination and mutation of immunoglobulin gene segments) toward this canonical function, B cells can provide other, “non-canonical” functions, such as facilitating of lymphoid organogenesis and remodeling and fashioning T cell repertoires and modifying T cell responses. Some non-canonical functions are exerted by antibodies, but most are mediated by other products and/or direct actions of B cells. The diverse set of non-canonical functions makes the B cell as much as any cell a central organizer of innate and adaptive immunity. However, the diverse products and actions also confound efforts to weigh the importance of individual non-canonical B cell functions. Here we shall describe the non-canonical functions B cells and offer our perspective on how those functions converge in the development and governance of immunity, particularly immunity to transplants, and hurdles to advancing understanding of B cell functions in transplantation.
Keywords: B cells, antibody-independent B cell functions, regulatory B cells, lymphoid organogenesis, T cell repertoire
1. INTRODUCTION
For more than eighty years the fate of transplants has been connected in one way or another with allo-antibodies [1] and by implication to the cells that produce those antibodies. Allo-specific antibodies, particularly donor-specific antibodies (DSA), provide the most reliable index of risk of rejection and graft loss [2] and an invaluable marker for diagnosis of acute and chronic antibody-mediated rejection [3–5]. Yet, for almost as long as long as allo-antibodies have been known to exist, debates have raged about whether and to which extent allo-antibodies detected in blood directly injure transplants [6–11]. Today, few would question that allo-antibodies can attack organ transplants, fulfilling what we shall call the canonical function of the antibodies. Yet, few can explain why more often than not allo-antibodies cause no acute injury. Nor is there a ready explanations why mice lacking B cells and unable to produce allo-antibodies sometimes exhibit better, sometimes worse and sometimes the same outcomes of organ and tissue transplants as wild type mice. Some differences in the impact of alloantibodies merely reflects differences in direct exposure to the antibodies and complement, as vascularization with recipient blood vessels shield cell and tissue transplants from the alloantibodies and complement that directly attack organ grafts [12, 13]. Some “paradoxical” responses of transplants to alloantibodies reflects “accommodative” changes that can protect transplants from antibody-mediated injury and some reflects suppressive actions of antibodies on immune responses. We call these other functions of antibodies “non-canonical functions.” The considerable interest in development of therapeutic agents that deplete B cells and plasma cells and agents that might suppress B cell functions will make it important to whether agents that eliminate B cells or broadly suppress B cell functions have untoward impact on a transplant or on autogenous organs. This communication will examine the non-canonical functions of B cell that may impact the outcome of transplants. We focus especially on functions that have incompletely or inconsistently defined impact in transplants and the barriers to better defining that impact.
B cell functions
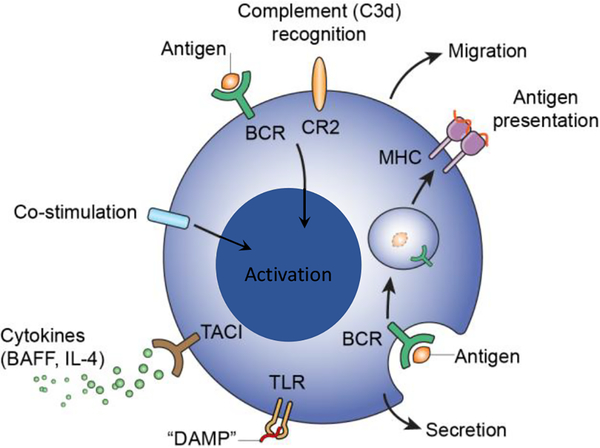
Only B cells are highly differentiated to enable the regulated production antibodies (Figure 1 and hence we refer to that as “the canonical function” of B cells. B cells clonally express B cell antigen receptors (BCR) that have the same tertiary configuration as the antigen-binding region of antibodies and allow B cells to undergo activation, anergy or apoptosis upon recognition of antigen. B cells recombine genomic DNA to encode a BCR that corresponds to the antibody the B cell produces, allowing diverse clonally distributed repertoire of BCR and immunoglobulin (Ig) to enable recognition of a vast set of different antigens. B cells undergo somatic hypermutation, preferentially targeting recombined variable regions of immunoglobulin (Ig) genes. B cells censor the diverse BCR repertoire by clonal selection and deletion based on specific interaction of BCR with antigen targets. B cells carry the “memory” of paratope binding and potentially differentiate into plasma cells that produce the Ig in quantity.
Figure 1. B cell recognition of various agonists and functional responses.
Besides the clonotypic B cell antigen receptor (BCR), B cells have co-stimulatory receptors that recognize various endogenous and exogenous agonists. These receptors include the complement receptor type 2 (CR2) that recognize C3d, and co-stimulatory receptors, including CD80, CD86, and CD40. B cells express various toll-like receptors (TLR) that recognize “danger associated molecular patterns” (DAMPS). Other B cell receptors recognize cytokines, such as IL-4, B cell activating factor (BAFF), and lymphotoxin. B cells also express major histocompatibility complex (MHC) antigens, class I and class II. Utilization and stimulation of B cell receptors, individually or in combination, promotes the diverse B cell functions, including production of antibodies, secretion of cytokines, and presentation of antigen to T cells and regulation of T cell responses. For example, at various stages of B cell development, antigens recognized by BCR can be preferentially captured, taken up by endocytosis, processed and loaded on MHCII complexes and presented to T cells (cognate presentation) in thymus, and in lymphoid follicles of secondary lymphoid tissue. The location of B cells, as determined by the stage of development, migration, etc., helps determine whether and how the B cell exerts canonical and/or non-canonical functions.
B cells also perform non-canonical cellular functions, such as migration, phagocytosis, elaboration of proteins other than Ig (e.g. cytokines, growth factors and enzymes), and expression and of MHC class II (Figure 1). These non-canonical cellular functions support lymphoid organogenesis and remodeling, regulation of B cell and T cell responses, diversification of T cell repertoires. Other cells can perform these functions, but B cells can and often do perform the functions at distinct anatomic locations, such as germinal centers, and under conditions distinct from other cells.
Central to understanding the cellular physiology of B cells must be a consideration of how the non-canonical functions are induced and regulated and what circumstances or conditions (e.g. location, diversification of immunoglobulin genes) preferentially equip B cells to perform those functions at a given site or time. Much is known about the minimal stimuli needed for activation of various non-canonical functions in vitro, less is known about the set of stimuli orchestrating non-canonical functions, as those functions are manifest in vivo.
1.1. The contribution of B cell functions to immunity
Viewed from the perspective of the whole organism and the immune system, the canonical function of B cells confers host defense. The involvement of immunoglobulin proteins in host defense has been recognized for at least 130 years (see [14] for review). The cellular source of the proteins, later called antibodies and the role that cellular source plays in overall physiology of the immune system was hotly debated for decades and not settled until three discoveries had established the cellular basis of immunity [15, 16]. First, lymphocytes and plasma cells were found to be the only cells that produces antibodies [17, 18]. Second, while lymphocytes and lymphoid organs containing clusters of lymphocytes produced antibodies, the production of antibodies by any cell or organ in chickens was found to depend on the presence of a bursa of Fabricius early in life [19], suggesting production depended on a developmental hierarchy or series of events (hence the term “B” cells). Third, bursa-dependent lymphocytes or B cells, and the antibodies B cells produce were shown to exert some facets of immunity but not others [20, 21]; the other facets were exerted by lymphocytes (T cells) that required the integrity of the thymus early in life [22].
Viewed from a current perspective, the experiments revealing that B cells and T cells mediate distinct facets of immunity ironically also revealed that most “distinct” functions of these cells are inter-dependent, that is B cell responses depend on T cells and more recently that T cell responses depend on B cells. For example, the classical experiments connecting the bursa of Fabricius to production of antibodies, tested T cell-independent responses to Salmonella typhimurium species [19, 20]. However, abolishing antibody responses to bovine albumin and Brucella abortus antigen required removal of the bursa and delivery of 650 r to newly hatched chicks, a treatment that would later prove to distinguish T cell-dependent from T cell-independent B cell responses [23]. Still more important for the present was the observation that removal of the bursa in chicks severely hindered development of delayed-type hypersensitivity responses to tetanus and diphtheria toxins [21, 24] and generation of graft versus host reactions in the newborn [21], more discerning tests of competence of cell-mediated immunity than allograft rejection. These seminal discoveries also provided the first suggestion that besides producing antibodies, B cells establish the overall structure and functionality of the immune system, as we later discuss.
1.2. Connections between B cell recognition and B cell functions
Table 1 lists various systemic functions of B cells. Besides the canonical function of B cells, i.e. production of antibodies that confer host defense or immune surveillance, are listed numerous non-canonical functions that could be viewed as antibody-dependent or antibody-independent, the later being functions manifest in a system in which B cells can express BCR but cannot produce antibody [25, 26]. Some antibody-independent functions, such as initial development of lymphoid follicles with follicular dendritic cells, appear only to be performed by B cells, but other cells express the factors needed to perform the function. Most antibody-independent functions, such as immune regulation, are performed by B cells and by other cells. As we discuss B cell functions in the sections that follow, it is instructive to consider the following: (i) why in a given setting B cells rather than other cells confer the function (e.g. since all leukocytes can produce IL-10, why does IL-10 secreted by B cells have a dominant impact in some circumstances); (ii) whether a given systemic function could reflect one or several cellular functions of B cells (e.g. antigen presentation reflects phagocytosis, processing and expression of peptide MHC complex and co-stimulatory proteins, but it also reflects B cell-mediated development and maintenance of lymphoid tissue); (iii) whether the evidence implicating one function excludes other functions; and (iv) whether experimental systems used to implicate B cells in a function, such as B cell deficiency generated by gene targeting or B cell depletion induced by administration of anti-B cell antibodies, have compensatory distorted phenotypes caused by the manipulation. For example, B cell-deficient mice and mice from which B cells are depleted are commonly used to investigate immune regulation and antigen presentation by B cells however B cell-deficiency causes profound contraction of T cell repertoires and aberrant development of lymphoid tissues and B cell-depletion can activate complement and stimulate Fc receptors, which in turn influences activation and function of residual B cells, T cells and other leukocytes.
Table 1.
Connections between cellular and systemic functions of B cells
| Cellular Function | Product | Systemic Impact | Impact on Transplant |
|---|---|---|---|
| Recombination/mutation | Ig | effector1 | + immunity |
| Ig | adaptation | − rejection | |
| Ig | enhancement | − rejection | |
| Ig | accommodation | − rejection | |
| Ig | TCR diversity | + immunity − immunity |
|
| Ig | complement reg | − rejection | |
| BCR | cognate recognition | + immunity | |
| MHC class II expression | MHC | TCR diversity | + immunity − immunity |
| MHC class II expression | MHC | Ag presentation | + immunity − immunity |
| Phagocytosis | peptides | Ag Presentation | + immunity − immunity |
| Growth factor expression | LT2 | lymphoid organogenesis3 | + immunity − immunity |
| Cytokine secretion | IL4 | + immunity − immunity |
|
| Enzyme production and secretion | proteases5 endoglycosidases6 | + immunity − immunity |
|
| Migration | tissues, lymphoid organs | Ag capture, localization of functions | + immunity − immunity |
+Abbreviation Ag, antigen; Ab, antibody; LT, lymphotoxin; IL, interleukins
Effector functions of Ab include complement activation
Lymphoid organogenesis includes lymphoid remodeling and germinal center reactions
LT (lymphotoxins) refers to various lymphotoxins, TNFα, etc. that promote lymphoid organogenesis
IL (interleukins) refers to various cytokines such as IL-10 and TNFα that facilitate or suppress immune responses
B cells secrete serine proteases and express metalloproteinases such as ADAM10 that regulate availability of surface receptors, such as TACI and BCMA, and contribute to the remodeling of lymphoid tissues
B cells secrete endoglycosidases such as heparanase that remodel lymphoid organs and sites of B cell migration.
Figure 1 depicts types of receptors that stimulate B cells and some of the diverse functions B cells exert. Whether and how B cells are stimulated to produce antibody, undergo affinity maturation, manifest memory or undergo apoptosis, as these contribute to the canonical functions, is well understood. Whether or not B cells manifest canonical functions depends in large part on the combination of receptors that is stimulated and on the location of B cells at the time of stimulation. B cells located in lymphoid tissues to which antigen is delivered may respond to that antigen by activation or anergy depending on whether antigen receptors (BCR) stimulated by polymeric or by monomeric epitopes, and whether neighboring T cells provide co-stimulation. Covalent attachment of complement end products (C3d, C4d) to antigen crosslinks and stimulates CR2 and BCR decreasing by orders of magnitude the minimum amount of antigen needed to activate B cells. Soluble agonists, such as anaphylotoxins (C3a, C5a), cytokines, agonists for TLR, and BAFF, and environmental conditions such as hypoxia, irradiation, among others, add a further dimension to B cell responses.
Far less is known about how various stimuli coordinate non-canonical B cell functions. It is useful however to consider examples of these limitations. Besides triggering antibody responses, binding of an antigen to BCR can serve as the first step in presentation of antigen to T cells. Thus, antigen bound to BCR can be taken up, processed and associated with newly synthesized MHC class II complexes (Figure 1). Lipopolysaccharide (LPS), besides triggering T cell-independent B cell responses (type 1 T cell independent B cell responses), induces B cells to express MHC class II, to process and potentially present antigen and to express co-stimulatory molecules, thus promoting the antigen presenting functions [27–29]. LPS also can trigger B cells to produce IL-10 and in doing so to suppress T cell responses [30, 31]. The action of LPS on neighboring macrophages or endothelial cells could induce production of cytokines and other bioactive metabolites [32] that potentially further modify the balance of B cell stimulation and the functions that ensue. Thus, while the characterization of B cell responses to individual agonists in vitro or in narrowly defined animal models can elucidate potential functions of B cells, experiments designed to deliver one agonist to B cells almost inevitably neglect or even misrepresent the impact of that agonist in more complex circumstances such as infection or transplantation.
In contrast to the highly specific molecular and cellular changes needed for production of antibodies, B cells are not uniquely or specifically differentiated to exert the non-canonical functions (with a few exceptions noted below). We think that understanding of non-canonical functions of B cells can be advanced by identifying: (a) the range of cells besides B cells that potentially exert the functions (e.g. since all leukocytes can produce IL-10 and many cells produce IL-35 - cytokines implicated in the regulatory function of B cells - understanding how B cells mediate immune regulation must depend on identifying why B cells rather than other cells provide these cytokines); (b) the conditions that make B cells more or less likely than other cells to exert the function (e.g. the location or migration of B cells, the timing of the process, and/or the relationship of the function to antibody production); and (c) the stimulus for B cells to provide the function of interest. Ultimately, the most important question must be how the various B cell functions, which can be protective in some settings and pathogenic in others are coordinated to promote wellbeing. We will offer a few comments to this point, especially as it might apply in transplantation, but the governing principles remain to be found.
2. THE CANONICAL SYSTEMIC FUNCTIONS OF B CELLS AND ANTIBODIES
The systemic functions of antibodies is generally understood to include neutralization of toxins, opsonization and lysis of microorganisms, and agglutination or lysis of foreign blood cells [14, 33]. We consider the production of antibodies that exert these functions to be canonical function of B cells. Antibodies, and hence the B cells producing those antibodies, can exert analogous effector functions in transplantation. Production of alloantibodies that induce hyperacute rejection, acute and chronic antibody-mediated rejection and ischemia reperfusion injury are the best-known examples. Antibodies also induce the “instant blood-mediated inflammatory reaction” observed when isolated islets of Langerhans or allogeneic or xenogeneic cells are introduced directly into the blood [13]. These canonical functions of antibodies have been reviewed in detail (see [12, 34–36] as examples) and require no further mention.
However, contrary to the canonical effector functions of B cells and antibody responses, allogeneic B cell responses and administration of alloantibodies sometimes has little or no impact and sometimes improves the outcome of certain allografts. Allogeneic B cell responses and alloantibodies have little or no impact when a cell or tissue allograft, such as a skin graft or hepatocyte graft, is vascularized by blood vessels of the recipient. The blood vessels of the recipient provide a barrier that limits contact of antibodies with the graft. The paradoxically favorable impact of alloantibodies (and hence of B cells) on allografts was discovered decades ago and must reflect processes besides the barrier posed by vascularization [11, 12, 37].
The first experiments revealing the favorable impact of antibodies on allografts suggest why that challenge remains pertinent. In these experiments, mice of various inbred strains were repeatedly immunized with lysate of tumor cells with one or more major and/or minor histocompatibility disparity and 8–10 days later living tumor cells were injected [11, 38]. In animals of some strains treated this way the allogeneic tumor grew progressively and killed the mice while in untreated controls the tumor initially grew to small size but thereafter always regressed. In mice of some strains repeatedly immunized with tumor lysate, tumor did not grow, even to small size. Subsequent experiments revealed that administration of alloantisera could increase the survival and growth of some allogeneic tumors [37, 39] and prevent or slow the rejection of transplants of non-malignant cells and organs [40, 41]. How antibodies exert such protection was then and remains incompletely understood despite growing appreciation that the protective impact of antibodies has import for optimizing host defense against infection and cancer and for improving management and outcomes to transplantation [42]. As some mechanisms are briefly discussed below, it is important to remember that experimental evidence supporting the importance of one or another mechanism, rarely if ever excluded the impact of other mechanisms. Also lacking from past and present consideration is the potential impact of B cell and plasma cell functions besides the determination of isotype, affinity and amount of antibody produced [42], such as the location (local versus systemic) and the kinetics of responses [43–46].
2.1. Protection conferred by blood vessels.
One factor that should not be confused with antibody-mediated protection of grafts is the shielding of cell and tissue grafts by blood vessels of the recipient. Cell and tissue grafts are generally less susceptible than organ grafts to antibody-mediated-injury where blood vessels of the recipient (rather than donor) feed the graft because blood vessels of the recipient provide a barrier between alloantibodies and the graft. The barrier is not absolute however and some fraction of antibodies in the blood penetrate capillary networks and venules. Nor does the barrier protect grafts from T cells and macrophages, which actively migrate through the walls of small blood vessels and might be further activated in doing so [47]. The impact of vascularization does not explain the antibody-mediated protection discussed in the example above, as both experimental and control grafts were fed by blood vessels of the recipient. However, the impact of vascularization could explain some discrepant observations on antibody-mediated rejection of tumor and skin grafts.
2.2. Adaptation.
Adaptation refers to an acquired change in a graft, particularly a change in antigen, that renders the graft less susceptible to antibody-mediated injury [48]. The term originated as one explanation for the acquired decrease in vulnerability of long-surviving grafts to alloantibodies [49, 50]. Adaptation was thought to arise over months when: (a) alloimmunity was partly suppressed, antigen disparity limited and/or grafts were placed in protected sites and (b) resistance to injury, as an alternative explanation was excluded or suitably controlled [49]. Potential mechanisms of adaption include modulation or shedding of antigen or replacement of endothelial cells of the graft with endothelial cells of the recipient. Although modulation and shedding of histocompatibility antigens has been explored using isolated cells [51], the extent to which it occurs and impacts on the outcome of clinical organ transplants is unclear. Modulation and shedding of glycolipid antigens however can occur and can have significant impact [52, 53]. Where alloantibodies induce shedding of antigen, however, the shed antigen could suppress B cell responses, making adaptation difficult to distinguish from “partial tolerance” [48, 49].
2.3. Enhancement.
Enhancement refers to more exuberant growth of tumor allografts or prolonged survival normal tissue or organ grafts associated with induction of B cell responses or transfer of antibodies specific for the source of the graft [37, 50]. Enhancement is best explained as antibody-mediated blockade of antigen recognition in the afferent limb of an alloimmune response [54]. Interest in enhancement of normal tissue transplants has waned, perhaps because more reliable and safer means of immunosuppression are available. However, enhancement does appear to have a pronounced impact in some settings, e.g. antibody-mediated blockade of tumor antigens has been associated with progression of cancer and antibody-mediated enhancement of flaviviruses with increased severity of pathologic changes and clinical manifestations [55].
2.4. Accommodation.
Accommodation refers to acquired resistance of a graft to immune or inflammatory injury. Accommodation was first described in ABO-incompatible kidney transplants and in swine-to-non-human primate cardiac xenografts [56] and is typically identified when an organ transplant functions and exhibits little or no evidence of injury despite the presence of antibodies directed against the graft in the circulation of the recipient. Accommodation probably occurs at one time or another in nearly all ABO-incompatible and in many ABO-compatible transplants [57]. Accommodation also likely protects tumors, and some tissues confronted by autoimmunity or by infection and mammalian fetuses with a haplotype distinct from the mother [57–60]. Accommodation of organ transplants appears to be induced by donor-specific anti-HLA or anti-blood group antibodies [61], although other “noxious” factors, such as cytokines, cytotoxic T cells and bacterial peptides may act in a similar fashion. Donor-specific antibodies and/or the other noxious factors induce increased expression of cytoprotective genes and expression of these genes has been considered the main explanation for accommodation [61, 62]. Recent investigation suggests that induced cyto-protection alone cannot explain accommodation and that other protective mechanisms, such as increased control of complement and regenerative modifications of tissues and vascularization, that confer more enduring protection may better explain the timing and endurance of accommodation in organ grafts [57, 63–65].
2.5. Natural antibodies.
Natural antibodies (antibodies produced without known history of exposure to foreign antigen), particularly polyreactive natural antibodies and particularly IgM, have properties distinct from the antibodies that exert effector functions [66, 67]. Natural antibodies comprise a significant fraction of Ig in serum and recognize multiple antigens (as the term polyreactive implies), including histocompatibility antigens and saccharides of various types and components of cytoplasm and nucleus and neoantigens of the cell surface [68, 69]. The antibodies provide an early defense against some pathogens and potentially a framework for generation of more specific antibodies [70, 71]. Natural antibodies are postulated to broadly regulate immune and inflammatory reactions through interaction with Fc receptors and by incompletely defined interactions with cytokines, cytokine receptors and other components of plasma and cell surfaces [72]. For example, interaction of Ig Fc with FcγRIIb on B cells and plasma cells regulates activation and survival of the cells and Ig production [73]. Whether production of natural antibodies should be considered a canonical or non-canonical function of B cells is a matter of rhetoric; we briefly discuss a few of these properties.
Natural antibodies bind neoantigens generated in ischemic tissues and in so doing activate complement, potentially leading to ischemia-reperfusion injury [74]. However, antibody binding and complement activation can also protect and/or repair tissues [67, 75–78], circumscribing inflammation, clearing apoptotic and damaged cells and regulating the function and availability of cytokines. Natural antibodies interact with cell surfaces, through the antigen combining domain and the Fc domain, modifying thrombogenicity, recruiting or regulating complement [32, 79] and suppressing or activating leukocyte functions [80–82]. What determines whether the effector or the protective properties of natural antibodies dominate in a given setting is incompletely understood but extent and type of glycation, set of specificities and isotype, among other properties have been queried [67, 83, 84]. IgM DSA in kidney transplant recipients appear to pose little risk and may even benefit graft outcomes [4] and delivery of pooled IgG, which includes polyreactive natural antibodies and some anti-HLA antibodies (but little DSA) antibodies, to transplant recipients clearly confers more benefit than harm when vascular injury is suspected [85].
A phenotypically distinct population of B cells, B1 (CD5+) B cells, produces some natural antibodies [70, 86]. The products of microorganisms and inflammatory reactions heighten production of natural antibodies and production in this setting might be viewed as providing antigen-non-specific host defense, limiting the systemic effects of tissue reactions and promoting repair of injured tissues. B1 B cells can regulate immunity through properties of the antibodies produced or by antibody-independent mechanisms [87]. The broader subject of immune regulation is discussed below.
2.6. Antibody-mediated control of complement.
Although antibodies were first characterized as the heat-insensitive components of serum that direct complement reactions to specific targets, antibodies can also modify cell surfaces in ways that change the ability of the surface to serve as a target for initiation and amplification of the complement cascade and potentially in ways that inhibit the complement cascade. The mechanisms by which Ig potentially regulates complement were elucidated in part by in vitro investigation and in part by administration of labeled cells in animals and humans (see [57, 88] for review). Direct evidence for Ig-mediated regulation of complement in transplantation is limited [89] however the mechanisms could help explain why some grafts appear unperturbed by DSA in the circulation of the recipient. Mechanisms of Ig-mediated regulation of complement include: (i) Ig-mediated activation of complement at a non-toxic level can generate catalytically inactive deposits C3d and C4d, which block sites on cell surfaces that might otherwise react with newly generated C3b and C4b [90, 91]; (ii) Ig serves as an alternative substrate for C3b and C4b, diverting convertases away from cell surfaces [72, 92]; and (iii) ionic interaction of Ig with cell surface heparan sulfate and sialic acid might impair binding of factor P, which activates complement or factor H which suppresses activation of complement.
3. THE NON-CANONICAL FUNCTIONS of B CELLS and ANTIBODIES in IMMUNITY and TRANSPLANTATION
3.1. Lymphoid Organogenesis and Maintenance
B cells may participate in the development and remodeling of the spleen, lymph nodes (LN) and the gut-associated lymphoid tissue or GALT including the Peyer’s patches) [45, 93–99]. Current understanding derives mainly from work using mice with targeted disruption of genes postulated to directly contribute to lymphoid organogenesis or the emergence of cells postulated to contribute the minimal molecular and cellular requirements and sequence of events in development of lymphoid organs (see [98, 99] for recent reviews). These requirements include signaling through the lymphotoxin-β and RORγt receptors [97], among others [100]. For spleen and LN, the minimal cellular requirements evidently include more primitive lymphoid tissue inducer or LTi cells and non-lymphoid tissue organizer cells or LTo cells (potentially lymphatic endothelial cells) [99]. Although fetal (or maternal) B cells might provide initiating signals soon after development of the spleen begins (B cells appear one day after development of spleen commences) in the mouse and during LN development, current models exclusively implicate the more primitive cells in the initial development [98].
However, B cell deficient mice have strikingly disorganized lymphoid tissues [93, 95, 101, 102]. Aberrant anatomy of lymphoid organs in B cell deficient mice in part reflects the paucity of cells available for entry into the developing organ but it also reflects in part a direct contribution of B cells to the maturation and the maintenance of the anatomy and function of mature lymphoid tissues [93, 98, 103]. For example, B cells control lymphoid organization and govern the differentiation of dendritic cells and also promote the development of T cell zones of the spleen [95, 96, 101, 104] and lymph nodes [99]. These functions depend in part by expression of lymphotoxin-α1β2 and TNFα. The distorted and deficient lymphoid organogenesis in fully immunodeficient and B cell deficient mice poses a significant challenge for immune reconstitution, especially with human hematopoietic cells [105–107].
The functions of B cells in lymphoid organogenesis are distinct from the canonical antigen-specific effector functions but require expression of the B cell antigen receptor and/or the receptor complex. Thus, lymphoid tissue organization, particularly follicular dendritic cell networks are aberrant in mice lacking recombinases [101, 106] and in mice with targeted disruption of heavy chain gene assembly (JH−/−) [102]. Since these mice also lack B cells it is not possible to discern the extent to which lymphoid tissue organization requires functions mediated by the BCR other than those associated with the development of B cells. The specificity of the B cell antigen receptor, however, appears immaterial to lymphoid organogenesis, as we found that mice expressing only clone of B cells (on a RAG-2 background) have normal lymphoid organs; yet, mice of the same genetic background that produce one clone of T cells but no B cells have markedly abnormal lymphoid tissue [108]. This observation confirms the presence of a BCR and/or associated proteins enables B cells to contribute non-canonical functions in lymphoid organ maturation and organization [101]. Clearly, the specificity of B cells, and indeed the production of antibody, is important to form germinal centers, transient structures in lymphoid tissues formed following B cell activation, which optimize B and T cell interactions and access to antigen resulting in affinity maturation and B cell selection [109].
Of import for medical immunology and therapeutics is the potential involvement of B cells in sustaining the functional anatomy of lymphoid organs and in generating tertiary lymphoid tissues. Aside from germinal center reactions (which has been the subject of intense investigation as comprehensively reviewed elsewhere [110–112]), involvement of B cells in other aspects of lymphoid tissue remodeling is not fully explored. In the course of inflammation and infection, B cell expression of lymphotoxin α1β2 induces generalized expansion of B cell content and high endothelial venule networks [113, 114], and establishes an extra-follicular, medullary niche that optimizes the production of some antibodies [115]. In our view, the potential importance of B cells in maintaining the structure and function of lymphoid tissues might be inferred from observations that depletion of B cells in mice with inducible deletion of Igα [116] or by decreases in BCR diversity [117] abolishes the structure and function of the splenic marginal zone [116]. In monkeys and humans administration of anti-CD20 profoundly distorts the anatomy of lymphoid organs and exerts prolonged decrease in the numbers of circulating B cells [118–121]. These findings suggest that B cells contribute ongoing signals for the maintenance of the lymphoid tissue.
Enzymes produced by B cells are vital for lymphoid organogenesis and remodeling. As one example, heparanase, an endoglycosidase that cleaves heparan sulfate, an acidic saccharide that binds chemokines and many cytokines, produced by newly activated B cells generates a pathway for migration in lymphoid tissue [122] and undermines the ability of nearby cells to induce tolerance [123]. A B cell sheddase, ADAM10 (A disintegrin and metalloproteinase-10), enables Notch2 signaling [124], normal development of lymphoid follicles, normal germinal center reactions and robust T cell-dependent primary and secondary B cell responses [125].
B cell expression of lymphotoxin α1β2 and TNFα, among other cytokines, also enables the generation of tertiary (ectopic) lymphoid assemblies in tumors, tissues targeted by autoimmunity and in transplants [43, 45, 126] (although other lymphoid cells, especially Th17 cells can do likewise [44, 126]). Regardless of which cell(s) initiate formation of these structures, B cells provide abundant constituents and likely drive formation of germinal centers. Whether and to which extent tertiary lymphoid tissues contribute to host defense, autoimmunity and/or rejection is not known, but treatment of mice with lymphotoxin-β receptor-Ig fusion protein: (a) blocks production of autoantibodies thought to mediate allograft vasculopathy, (b) delays rejection of cardiac allografts and (c) prevents formation of tertiary lymphoid structures in cardiac allografts [127]. If tertiary lymphoid structures were indeed found to be important in autoimmunity and chronic rejection, it might explain the impact of agents specific for B cells, such as anti-CD20 (which besides depleting B cells causes notable distortion of lymphoid structures [120]) on T cell responses and on conditions such as rheumatoid arthritis [128]. On the other hand, failure of anti-CD20 to modify the course of chronic antibody-mediated rejection in some transplant recipients [129] could be taken as evidence that tertiary lymphoid organs are a consequence and not an essential underpinning of immunologic disease or that B cells acquire resistance to anti-CD20 [130].
3.2. Development and maintenance of the T cell compartment
Among the most provocative and controversial discoveries in immunology concerned the origin of lymphocytes that produce antibodies and the origin of lymphocytes that reject tissue allografts. Identification of the bursa of Fabricius in chickens (and bursa equivalent or bone marrow in mammals) as the source of antibody producing cells [19] sparked efforts to determine whether other immune functions originated at distinct anatomic sites and led to identification of the thymus as the source of lymphocytes that reject skin allografts [22]. Surgical or chemical removal of the bursa of Fabricius in young chicks more or less abolished production of antibodies but left intact ability to reject allografts and removal of the thymus in newborn chicks or in mammals compromised the ability to reject grafts but more or less left intact ability to produce antibodies [20, 21, 24, 131]. Since antibody production was bursa-dependent and graft rejection thymus-dependent, the cells mediating these distinct functions were B cells and T cells respectively [16]. Despite the simplicity and appeal of this concept, the original experiments that distinguished the functions of T cells and B cells also revealed that removal of the thymus decreases or abolishes B cell responses to some antigens [132, 133] and removal of the bursa of Fabricius abolishes T cell responses to some antigens (e.g. albumin), the recognition of which is more challenging than recognition of alloantigens [20]. It is now apparent that B cells facilitate T cell responses in various ways. B cells present some antigens to T cells, as discussed in a later section, contribute to lymphoid organogenesis, as discussed above and effector B cell responses generate C3d, which attenuates checkpoint suppression of cell mediated immunity [134]. Here we consider how B cells influence development and selection of T cells in the thymus and survival of T cells in the periphery.
Approximately 15% of lymphocytes in the thymus are B cells. As one obvious function, B cells in the thymus (and periphery) may delete clones of T cells that otherwise could respond to tissue-specific B cell antigens. This function might be needed as B cells express the products of recombined Ig genes that cannot be anticipated through expression of AIRE in other cells. The importance of this function is suggested by the difficult barrier to adoptively constituting a B cell compartment in B cell-deficient animals. As a less obvious but equally important function, B cells promote positive selection of T cells. Thus, mice engineered to produce only one clone of T cells (monoclonal T cell mouse or MT mouse) have half as many CD4+ thymocytes as mice engineered to produce one clone of T cells and one clone of B cells (monoclonal B cell-T cell mouse or MBT mouse) [108].
B cells also promote diversification of the T cell receptor (TCR) repertoire in the thymus [135]. Mice with complete deficiency of B cells, owing to disruption of BCR (JH−/−;κ−/−) have 4-fold fewer thymocytes and a 106-fold contraction of TCR diversity of thymocytes compared with wild type mice of the same genetic background [135]. Mice with partial deficiency of B cells, such as the μMT mouse have ~50% fewer thymocytes and a 104-fold contraction of TCR diversity of thymocytes compared to wild type mice. B cells diversify the TCR repertoire of thymocytes at least in part by providing diverse peptide, as the narrow TCR repertoire of B cell deficient mice can be reversed at least in part by administration of polyclonal but not monoclonal Ig [135].
B cells also maintain the size and diversity of the peripheral T cell compartment. Mice fully deficient of B cells (JH−/− mice) have 2–5 fold fewer T cells in peripheral lymphoid organs [95, 136] than wild type mice and MT mice have ~35-fold fewer CD4+ cells than MBT mice [137]. Acute and chronic depletion of B cells in young mice by administration of anti-CD20 antibodies (one dose and repeated doses respectively) to mice reduces total naive CD4+ and CD8+ T cells by ~40–60% and memory T cells [138]. Various functions of B cells likely support the survival and hence the dimensions of the T cell compartment in the periphery, including production of cytokines and chemokines and fashioning and maintenance of the T cell zone [95]. Expression of MHC class I and MHC class II may be especially important for maintaining the size and diversity of the T cell compartment. Mice lacking MHCI and MHCII are nearly devoid of T cells [139] – MHC class II expression on thymic epithelium is more or less essential for development of CD4+ T cells [140] and B cell associated MHC class II helps maintain the number and diversity of mature CD4+ T cells [102, 141, 142]. The extent to which B cell expression of MHC is needed for peripheral T cell survival remains unclear however because under some experimental conditions unavailability of MHC is countered by homeostatic proliferation of T cells.
The capacity to mount a cell-mediated immune response has been long appreciated to depend on availability of a peripheral T cell compartment of sufficient “size,” marked by a normal frequency of T cells in peripheral blood. B cells maintain the size and hence the competence of the T cell compartment by supporting the survival of mature naive T cells by expression of MHC and proving diverse peptides.
Less certain is the extent to which immune competence depends on a diverse repertoire of T cells supported in part by BCR diversity. Although cross-reactivity and degeneracy of TCR recognition potentially allow each T cell clone to recognize many different peptides [143–145], there is little doubt that the more diverse the TCR repertoire, the more diverse the set peptides potentially recognized [146, 147] and the more timely would be primary T cell responses to rare antigens. TCR diversity facilitated by B cell diversity might help maintain latency of some viruses, peptides from which are scarce and perhaps earlier responses to tumor antigens or toxins. Mice with B cell deficiency induced early in life by treatment with anti-μ antibodies exhibit defective priming by foreign polypeptides and by allogeneic splenocytes [148]; but these mice have decreased numbers of T cells. Targeted disruption of B cell antigen receptor genes generates similar defects [149], which likewise could reflect change in the total number and/or diversity of T cells. B cell depletion with anti-CD20 does evidently impact on certain pathogenic T cell responses [128, 150], but the mechanism of that impact is not known.
However, the extent to which immune fitness absolutely depends on maintenance of TCR diversity, independent from the overall dimensions of the T cell compartment is less clear. Profound but unbiased contraction of TCR diversity in humans and mice caused by removal of the thymus early in life (but not so early to cause a decrease in total T cells), allowing restoration of T cell numbers by homeostatic proliferation, does not by itself impair primary T cell responses to many antigens, including major and minor histocompatibility antigens [102, 151]. The unfettered immune response to major histocompatibility antigens could reflect the high frequency of T cell clones that respond to the antigens, the response to minor antigens could reflect the abundance of those antigens. Unbiased contraction of TCR diversity does however have an appreciable impact on control of T cell responses [102, 151]. Accordingly, when B cell-deficient mice are used to explore antigen presentation, one should consider the possibility that deficiency of B cells also compromises immune regulation.
3.3. Antigen-presentation
B cells can present antigen to T cells, in theory [152–155]. After antigen is provided in suitable form and ancillary signals are delivered, B cells can activate naive CD4+ and CD8+ T cells, including cross-priming of CD8+ T cells, and therapeutic applications exploiting this function have been proposed [27, 29, 42, 152, 154, 156–159]. B cells also can interact in a cognate fashion with memory T cells and with T helper follicular cells. Aside from the presentation of antigen to T helper follicular cells in germinal center reactions, the relative importance of antigen presentation by B cells versus dendritic cells and distinct consequences, if any, of antigen presentation by B cells have been matters of uncertainty if not debate. This uncertainty reflects inevitable limitations of physiologic systems (especially the hurdles to identifying the antigen presenting cell that initially activated a given clone of T cells) and confounding variables associated with use of in vitro systems and mutant mice (e.g. B cell deficient mice have aberrant lymphoid organs, contracted TCR repertoires, and distorted expression of cytokines) (see [159–161] for discussion of some confounding variables).
Dendritic cells (DC) would appear better suited than B cells to present most antigens to naive T cells that will engage in cell-mediated immune responses [162, 163]. If antigen-presentation depends on the chance apposition of activated APCs expressing MHC in complex with a given peptide to the scarce (perhaps 1/105) naive CD4+ T cells capable of recognizing that peptide MHC complex, DC appear better suited than B cells. The location of DC, as residents of tissues interstices and migration to clusters of T cells in lymphoid organs make them better suited to consume macromolecules at sites of infection and communicate with random assortments of T cells. The processes of DC allow contact with numerous surrounding cells. The dramatic change in metabolism of DC upon activation and migration to lymphoid organs, leading to stable expression of MHC-peptide complexes on the cell surface [164], would seemingly favor DC over B cells as APC. B cells, in contrast, situated predominantly in lymphoid tissues and bone marrow potentially can capture abundant antigens, especially cell-associated antigens; however, to allow effective selection, B cells in germinal centers modulate MHCII turnover which is fastest in centroblasts (undergoing Ig somatic hypermutation) and slower in centrocytes which engage in cognate interactions with T cells that result in selection [165].
However, some circumstances can make B cells more effective than DC in initiating T cell responses. If the native structure of an antigen an antigen or an associated protein is recognized by BCR, the antigen or antigen complex can be taken up preferentially (103-104-fold more effectively than DC), processed and peptides from the antigen presented in complex with MHC (Figure 1) [153, 166]. BCR-mediated endocytosis thus concentrates antigen, directs the antigen to the endocytic compartment for processing and mounting on MHC and presents or cross-presents the antigen in far greater abundance than would occur on DC [29, 153]. In this way, scarce antigen can potentially activate naive T cells. Through a series of similar cognate interactions B cells also collaborate with T helper cells in lymphoid follicles to advance germinal center reactions facilitating selection needed for affinity maturation that characterizes canonical B cell responses [153, 165, 167].
Antigen presentation by B cells is also thought to generate and sustain memory CD4+ T cells and in doing so provide an enduring source of protection against viruses such as influenza virus [153, 168–170]. However, while B cells may very well be essential for T cell memory and effector functions, the fine regulation of T cell memory over time has been deduced using adoptive transfer into B cell-deficient mice or through B cell depletion. Although these manipulations alter antigen presentation, they also modify other functions (e.g. cytokine production, dysplasia of lymphoid organs and modification of TCR repertoire) that could impact on T cell memory and effector functions. These and other non-canonical actions of B cells over time, make it difficult or impossible to weigh the long-term impact of antigen presentation on T cell memory [171]. For example, activated T cells transferred into B cell-deficient mice might exert effector functions over months, but T cell effector functions are amplified and prolonged by the dearth of regulatory B cells and the limitation that TCR contraction imposes on development of regulatory T cells.
The extent to which B cells serve as APC for the immune response to transplantation or in regulation of that response has been a subject of much interest [172–178]. Antigen-presentation by B cells has been deemed essential for robust alloimmune responses leading to acute and chronic allograft rejection because mice with MHC class II-deficient or defective (DM-knockout) B cells (and unable to produce antibodies) exhibit delayed rejection of cardiac allografts and absence of chronic allograft vasculopathy [175, 176], among other evidence. However the importance of B cells as antigen presenting cells in transplantation has been challenged. B cell deficient mice (owing to targeting of membrane domain of μ heavy chain, μMT) evidence no decrement in the rate of rejection of skin grafts across minor antigen barriers [179] or cardiac grafts across major antigen barriers [180]. We also observed that mice deficient of B cells reject skin allografts as quickly as wild type mice [102].
However, we do not think the murine models provide critical insight into the relative importance of antigen presentation by B cells in clinical transplantation. As already discussed, deficiency of B cells undermines functions besides antigen presentation and antibody production and the balance of some changes is not readily predicted. B cell-deficient mice have abnormal lymphoid organs and contraction of the TCR repertoire, which might eventuate in immunological ignorance, when peptide is limiting, and hence slow or absent T cell priming or to partial activation and homeostasis-like posture with hastened responses when peptide is plentiful (as is transplantation) [102]. We have observed a modest hastening of rejection of male to female skin grafts in naive B cell-deficient mice and a more pronounced hastening of rejection in primed B cell deficient mice [102]. This observation could be interpreted to suggest that B cells play little or no essential part in T cell memory responses in transplantation; however, the observations could also reflect the absence of B cell-mediated immune regulation (as discussed in the next section), which takes weeks to be fully manifest, failure of immune regulation to develop in mice with contracted TCR repertoire, or other compensatory changes. Of course, depletion of B cells with anti-CD20 or anti-CD19 antibodies might leave enough residual B cells to initiate robust alloimmunity. Another concern however is that these models do not represent the “clinical conditions” in which antigen presentation by B cells could decide the fate of allografts. In clinical organ transplantation, immunosuppression might increase the importance of B cell-mediated antigen presentation to naive T cells and leave intact the T cell-B cell interactions that generate de novo donor-specific antibodies. Whether the importance of antigen presentation by B cells can be specifically tested in that setting remains unclear. The observation that B cell depletion impedes development of chronic vasculopathy in kidney allotransplants in mice [181] could be interpreted as potentially reflecting impairment in antigen presentation needed to generate B cell-memory effector responses, but it could also be interpreted as simply reflecting a decrease in the amount of alloantibody produced.
3.4. Regulation of immunity (regulatory B cells)
One non-canonical function of B cells that has sparked much interest and also some controversy is the regulation of immunity, particularly cellular immunity. B cells that regulate (i.e. suppress) immunity are called by regulatory B cells or “Breg.” Breg exert regulatory functions at least in part by secreting of cytokines, particularly IL-10, IL-35 and TGFβ [182–184]. B cells that secrete IL-10 and exert regulatory function can be marked by expression of a mucin-like protein, “T cell immunoglobulin and mucin domain” (TIM-1) [185]. TIM-1 and other TIM recognize phosphtidylserine and other mucin-like proteins; ligation of TIM on T cells promotes migration and effector functions [186]; ligation on B cells promotes secretion of regulatory or pro-immune cytokines, depending on which TIM molecule is targeted [187]. Regulatory cytokines, such as IL-10, suppress cellular immunity by stimulating the corresponding cytokine receptors on responder cells, antigen-presenting cells (APC) and regulatory T cells (Treg). Stimulation of certain non-antigen receptors, such as toll-like receptors (TLR), induces B cells to exert regulatory function and since most B cells have these receptors Breg do not arise as a distinct lineage.
Observations made over six decades indicate that B cells can suppress cell-mediated immunity [11, 188–191]. B cells can suppress cell-mediated immunity by production of enhancing antibodies or blocking antibodies (discussed below), by competing for antigen and by producing cytokines and metabolites that directly suppress T cell responses and/or induce suppressor or regulatory T cells (Treg) [192]. The modification of lymphoid organs and TCR diversification, described above, also may impact on cell-mediated immunity. We shall offer a critical perspective regarding the regulatory functions of B cells, especially as those functions might be manifest in transplantation and would refer the reader to recent reviews [182, 184, 193–195] for more comprehensive consideration of the subject.
Some observations in mice provide compelling evidence that Breg can influence immune mediated disease. B cell-deficient (μMT) mice develop unremitting [196] or more severe [197] manifestations of experimental allergic enchephalomyelitis (EAE) and severity and/duration of EAE and other experimental autoimmune diseases are reduced by adoptive transfer IL-10-producing Breg [182]. Depletion of B cells, particularly IL-10 producing B cells, with anti-CD20 antibodies prior to induction of EAE in mice enhances the severity of EAE [198].
Certain observations suggest Breg can determine the outcome of clinical transplants. Clinical kidney transplant recipients with stable graft function after discontinuing immunosuppression have higher frequencies of B cells in the circulation [199, 200]. In one series of six kidney transplant recipients, depletion of B cells using anti-CD20 antibodies was followed by acute cellular rejection in five (only one of 14 controls experienced rejection during period of study) [6, 201].
However, investigation of Breg in EAE and transplant models also poses challenges. Although the various antigens used to trigger EAE are well defined and the strains of mice are highly inbred, the relative importance of Breg (e.g. the dominance of immune regulation over pro-immune B cell functions, such as antigen presentation) varies considerably between laboratories and between models of EAE [202]. For example, in some models of EAE, B cell deficient mice (μMT) and wild type mice are found to have the same incidence and severity of EAE [196, 203]; in other models, μMT mice have significantly more severe EAE [197]; and in other models, μMT mice fail entirely to develop EAE [204]. These disparities may reflect subtle differences in immunogenicity of antigens used and in the contribution of antibodies to immunopathogenesis in the various strains of mice [202, 205]. The disparities also reflect the multifaceted impact of B cells on the inception and course of EAE - i.e. immune regulation, production of pathogenic antibody, and antigen presentation [198]. For example, while B cell depletion before administration of antigen makes EAE notably worse, B cell depletion after antigen has been administered makes the disease notably less severe [198]. The variation in the impact of Breg on EAE also reflects differences between inbred strains of mice in susceptibility to adjuvants, extent of complement sufficiency and differences in environmental factors and microbial populations in the gut of treated animals [205–208].
Investigation of Breg in experimental transplants is no less challenging. Transplanted tissues, such as skin, or extra-vascular cells such as hepatocytes, are relatively inured to the effector functions of antibodies [12] and therefore tissue transplants should offer a more incisive model than organ transplants (which are impacted by alloantibodies) for investigation of the contribution of Breg to the outcome of transplants. However, Breg appears to have little or no impact on the kinetics of rejection of skin transplants in unmodified recipients. For example, wild type H-2b mice, which presumably generate Breg in the course of immune responses, reject skin of H-2d mice as quickly as H-2b B cell deficient (μMT) mice, which presumably lack capacity to generate Breg [177]. The failure Breg in wild type mice to delay rejection of skin grafts might be ascribed to one or more of several confounding factors. One potential confounding factor is that μMT mice are not fully B cell deficient. However, JH−/− mice, which are fully B cell deficient, reject allografts with the same kinetics as wild type mice, suggesting the degree of B cell deficiency plays no appreciable role. Another factor potentially confounding investigation of Breg in transplantation is the possibility that other cells other than B cells may produce IL-10 when B cells are deficient or depleted. The possibility that B cell-deficient mice might exhibit a compensatory phenotype is suggested by the well-being of the mice, which, unlike IL-10-deficient mice, do not fail to thrive and do not develop lethal colitis [209]. In our view, the main obstacle to using tissue transplantation to probe the importance of Breg is that rejection of H-2 incompatible grafts proceeds too rapidly to allow the Breg functions to be fully manifest. Breg functions develop more slowly than alloreactive T cell effector functions and rejection of skin and other tissue grafts may well be complete [210] before Breg begin to suppress alloimmunity. In EAE models, in contrast, prominent differences between B cell deficient and wild type mice are first observed after 15 days [197, 205] and often much later [196]. This problem might have been bypassed by studying skin grafts performed across minor antigen barriers, however, in two reports of male-to-female skin transplants in μMT recipients, rejection occurred in wild type and B cell-deficient recipients rejected before day 20 [177, 179]. In our experience [102], both JH−/− mice, which are fully B cell-deficient, and wild type mice, with the same background, reject male-to-female skin grafts at 23 days, suggesting Breg do not delay rejection in the wild type.
Using antibodies to deplete B cells prior to transplantation avoids the confounding impact of a compensatory phenotype observed in some gene-targeted mice. Identifying whether and when antibodies impair Breg action also potentially reveals whether Breg function is constitutive or induced (Breg function would be “induced” if B cells acquire regulatory function after suitable stimulation or if constitutive Breg migrated to the site of action). Although the administration of anti-CD20 or anti-CD19 antibodies to mice does not deplete all B cells, neither does the targeting of μ-heavy chain in the μMTmodel. If anti-B cell antibodies deplete Breg and if Breg control alloimmunity, then pre-treatment with anti-CD20 or anti-CD19 should increase the frequency or severity of rejection in unmodified recipients. However, depletion of B cells from otherwise unmodified murine skin allograft recipients has not yielded the compelling evidence of Breg function observed in EAE models. For example, control and CD20-pre-treated C57BL/6 mice (H-2b) reportedly reject BALB/c (H-2d) skin grafts at 11 days and 10.7 days respectively [181]. As another example, pancreatic islets from C57BL/6 (H-2b) mice transplanted into control BALB/c (H-2d) mice are reported to function for a mean of 13 days, while islets transplanted into BALB/c mice pre-treated with anti-CD20 function for a mean of 10 days [185], consistent with the possibility that B cell functions other than immune regulation determine the rejection of islet grafts (before Breg can act).
The possibility that other canonical or non-canonical B cell functions might impair efforts to evaluate the impact of Breg in transplantation is suggested by observations connecting “innate” lymphocyte functions with ischemia-reperfusion injury. Above, in Section 2.5, we discuss the potential involvement of natural antibodies in the pathogenesis of ischemia-reperfusion injury. Other, still poorly defined contributions of B cells or combinations of B cells and T cells to persistence of early allograft injury [211–213]. Further, there is reason to suggest that sources other than B cells provide IL-10, as post-ischemic kidneys of B cell deficient mice contain more IL-10 at 10 days and 28 days (a time when Breg might provide IL-10 in a transplant) than post-ischemic kidneys of wild type or B cell-reconstituted mice [211]. Similarly, B cell deficiency in rats decreases inflammation in the early days after kidney allotransplantation; however, allografts in B cell-deficient rats nonetheless express more IL-10 [214, 215].
The manifold ways B cells might influence the fate of allografts, some promoting injury and rejection, some opposing injury and rejection make it difficult to devise experimental systems that isolate the impact of one B cell function, such a immune regulation, and exclude the impact of others, such as antigen presentation, on the outcome of allografts. Some guidance can be drawn from investigation in transplantation and in other immune conditions. The potential advantages of investigating tissue transplants (versus organ transplants) was discussed above. Work on EAE discussed above suggests that the full deployment of B cell regulatory functions takes time, at least 3 weeks, and transplant models in which rejection is delayed for a period of weeks would seem best suited for investigation of Breg. One approach might be to identify a sub-optimal dose of immunosuppression that will delay cell-mediated rejection [180] without entirely inhibiting cell-mediated immunity thus allowing the impact of Breg to be evaluated. Another approach might employ minor antigen disparities that do not typically cause rejection before 21 days and that do not depend on B cells for effective presentation [148]. Examples of such antigens might include minor H3 [216] and transgenic ovalbumin joined with transmembrane region [217], although the former can elicit an antibody response.
4. IMPACT OF B CELL RESPONSES ON THE OUTCOME OF TRANSPLANTS
B cell responses initiate pathologic processes over the course of engraftment [218–221]. These processes include ischemia-reperfusion injury, hyperacute rejection, antibody-mediated rejection and antibody-mediated chronic rejection. Each condition might be viewed as an endpoint in a linear series of pathogenic events: B cells produce DSA that enter the blood and bind to endothelium of transplants; bound DSA activates complement and/or recruits leukocytes which activate and damage endothelium inducing the histologic features of these processes [32, 222]. While useful for some purposes, linear models fail to represent the complex ways immunity impacts on graft outcome. DSA and complement not only cause injury but also initiate repair and over time increase the threshold for injury [32, 77]. B cells not only produce DSA but also suppress immunity and immune pathogenesis (e.g. antigen presentation or Breg) (Figure 1). That is why, in our view, indices of B cell immunity, such DSA in serum, may correlate with outcomes (e.g. DSA with rejection) in populations but in the absence of specific evidence of injury (e.g. graft dysfunction or biopsy) fail to predict the condition of the graft in individuals at the time the assay is performed.
Accordingly, the conditions under which an experimental model can highlight one or another function of B cells almost invariably minimize the impact of other B cell functions. Above we describe some conditions under which regulatory functions of B cells are highlighted in models of EAE and in skin transplantation. On the other hand, the clinical presentation and pathology of multiple sclerosis or AMR are probably not unalloyed stepwise consequences of autoimmunity and alloimmunity respectively but rather reflect a shift in the balance between contradictory T cell and B cell functions [12, 223]. Identifying B cell functions that govern outcome and characterizing whether and how the balance of these functions determines the outcome of an organ allograft is a daunting, but potentially instructive challenge. We shall offer a few examples.
For reasons incompletely understood kidney and heart transplants between certain strains of mice exhibit protracted function [224–226], in contrast to skin and tumor transplants between these strains that invariably undergo acute rejection. Early efforts to identify mechanisms that could explain the protracted course of kidney allografts in mice reveal a complex balance of effector and regulatory functions [227]. Thus, about 25% of recipients had cytotoxic DSA at one month but function was not significantly compromised and the frequency and concentration decreased thereafter [225], consistent with observations on accommodation in clinical transplants [57]. Also consistent with accommodation were observations that administration of sensitized serum had no impact on graft function and administration of this serum plus rabbit complement caused only transient decrease in graft function. Particularly striking however was the observation that administration of sensitized leukocytes generated no decrease or only a transient decrease in graft function [227]. Acute irreversible rejection and graft loss was only induced by administration of the combination of donor strain splenocytes, BCG, and cyclophosphamide; a regimen that depletes B cells. Exactly how antigen, adjuvant and cyclophosphamide evoked such dramatic changes in the graft was not determined, but interest centered on the possibility that antibodies (or B cells) had suppressed cell-mediated immunity in the controls, i.e. enhancement or “suppressor B cells,” as others recently had described [228, 229] and the treatment removed the source of suppression.
Kidney transplants between other strains of mice reveal what appears to be a different balance of B cell functions. Thus, kidneys from DBA/2 (H-2d) mice transplanted into unmodified C57BL/6×129/j F1 (H-2b) recipients also exhibit prolonged function (mean survival 54 days) and about 20% chronic changes over months [230]. However, in this model and in apparent contrast to the observations on EAE and kidney transplantation described above [198, 227], B cell depletion by pre-treatment of recipients with anti-CD20 or anti-CD19 (the recipients were transgenic for human CD19) failed to accelerate immune-mediated injury but instead led to improved pathology and anti-CD19 led to prolonged survival [181]. This result contrasts with observations in EAE, in which depletion of B cells before delivery of antigen caused manifestations to be worse by removing the source of immune regulation from the system whereas depletion of B cells after the manifestations of EAE were apparent significantly decreased severity of the disease possibly by decreasing production of pathogenic antibodies or antigen presentation [198]. In yet another murine allograft models, cardiac allotransplantation, depletion of B cells before transplantation does not notably influence the kinetics of rejection [181]. In our view, these apparent contradictions probably do not reflect the presence of Breg in one strain of mice and not in another but rather a shifting of the balance of multiple B cell functions, as proposed above.
For example, in one renal transplant model in which B cells were depleted by administration of anti-CD20 and anti-CD19 to C57BL/6×129/j mice before transplantation [181], all renal allograft recipients had measurable DSA in blood and deposits of IgM, IgG and C4d in the kidney and most, if not all, had cellular infiltration, yet graft survival was appreciably better in mice given anti-CD19. The linear explanation for this result is that effective depletion of B cells led to production of smaller amounts of DSA and hence to a better outcome. Against this explanation was the finding that kidney transplants in recipients treated with anti-CD20 and anti-CD19 and untreated controls had similar amounts of Ab and complement deposited. One (non-linear) explanation for this finding could be that B cell depletion selectively spared B cells that produce natural antibodies [231], and that some of the antibodies bound to grafts in anti-CD19-treated animals were protective or reparative [77, 81]. Yet another explanation might be that B cell depletion with anti-CD19 caused the greatest disruption of lymphoid structure, impairing germinal center reactions leading to production of DSA of lower affinity and the lower affinity DSA facilitated accommodation, which in turn allows more effective depletion of DSA from blood (accommodation rather than B cell depletion causing differences in DSA concentrations in blood) [57]. The balance of still other combinations of canonical and non-canonical B cell functions could be invoked to explain the observations made in this model system. Regardless of which combinations of processes actually generated the results in this relatively well-characterized model system, the results are better explained as a balance of the manifold functions of B cells than as the outcome of a linear series of events.
5. INDICES OF B CELL RESPONSES IN TRANSPLANTATION
Perhaps the greatest barrier to acquiring a more decisive perspective on the various B cell functions that impact on health and disease is the lack of indices or systems in which these functions can be assayed and the relative activity weighed. Experimental models were designed or optimized to measure the various functions discussed above but none of these models allows comparison of multiple functions.
If the outcome of organ transplants in inbred mice cannot be explained as a consequence of one dominant B cell function (e.g. production of effector antibodies), neither is the outcome of clinical transplants likely to be so explained. Although anti-donor HLA antibodies present at the time of transplantation reliably activate complement and more or less reliably induce hyperacute rejection, anti-donor blood group antibodies, which also activate complement usually do not induce hyperacute rejection [57]. Even less reliable is the propensity of DSA to induce AMR. Why DSA induce rejection in some recipients but not in others has been explored. However, such properties as the specificity, affinity, isotype, C1q binding, etc. provide only marginal insight beyond the level of risk conferred by presence of the antibodies. The focus on antibodies neglects other functions of responding B cells and other changes antibodies induce in targets. For example, as we review elsewhere [57], accommodated organs appear especially capable of absorbing and metabolizing antibodies; organs suffering injury from ischemia, cellular rejection, etc. do not so effectively take up antibody. Further, some antibodies, including natural antibodies, protect tissues from injury and promote repair of injury that does occur [67, 77, 81]. Conventional assays of DSA do not distinguish pathogenic from protective antibodies and indeed the same antibodies that injure one tissue and might protect another [77]. Finally, one must consider that if organ transplantation generates a polyclonal B cell response, the antibodies of highest affinity will selectively bind to the transplant and be depleted from the blood [232]. Unless a considerable surplus of pathogenic antibody is produced, DSA in blood may poorly represent DSA bound to the graft. For these reasons, among others, we have proposed and undertaken efforts to probe donor-specific B cells, i.e. B cell of the recipient that recognize the surface of donor cells, for clues about B cell responses to transplantation [60].
What can be said about the impact of depletion of B cells using anti-CD20 antibodies or other agents on canonical and non-canonical functions of B cells in clinical transplant recipients? As already discussed, limited investigation in normal subjects and non-human primates indicates that antibodies directed against CD20 clear B cells from blood and lower concentration of Ig in serum and disrupt the structure of lymphoid tissues [118–120] and transiently impair T cell activation [150]. In principle, these changes could compromise initiation of T cell-dependent B cell responses, somatic hypermutation, selection, isotype switching, peripheral survival of T cells, Breg-mediated suppression of T cell responses, besides the direct antibody-mediated functions. Which normal functions of human B cells are actually changed by administration of anti-CD20 might be difficult to glean from clinical experience because anti-CD20 antibodies are most often used in treating autoimmune disease and B lineage malignancy, conditions in which underlying immune functions are likely compromised. For this reason, we think the impact of B cell depletion in clinical transplantation could offer important insights.
Administration of anti-CD20 antibodies before or after transplantation decreases the levels of anti-HLA antibodies and in some cases the levels of DSA in blood [119, 233]. B cell depletion before transplantation (induction) with anti-CD20 or with other agents might be expected to prevent and/or improve the outcome of AMR; however, little evidence has emerged to support that expectation [234, 235]. Nor has clear evidence emerged that B cell depletion with anti-CD20 improves the outcome of AMR or CMR [236]. However, while these studies offer evidence regarding clinical utility, the studies provide little or no information regarding B cell functions in the pathogenesis or resolution of rejection, as eloquently put in one report [237]. Because most plasma cells do not express CD20 (and memory B cells are not effectively cleared by anti-CD20 antibodies), administration of anti-CD20 antibodies might be expected to have little direct impact on AMR [237]. However, the other functions of B cells, such as production of natural antibodies, antigen presentation, MHC class II expression with selection and survival of T cells, regulation of complement and regulation of cell-mediated immunity, are potentially modified by treatment anti-CD20.
The impact of non-canonical B cell functions has been explored in only a few clinical settings pertinent to transplantation. We mention these here hoping to spark further inquiry. A dramatic and perhaps worrisome report emerged from a trial of anti-CD20 antibodies as induction for kidney transplantation [201]. Five of six un-sensitized subjects given anti-CD20 antibodies as induction at the time of kidney transplantation developed acute CMR (versus one of seven controls). Some would question the clinical significance of the report, as other regimens including anti-CD20 do not increase the incidence of CMR [237, 238]. These reservations do not detract from the biological importance of the observation that B cell depletion in human subjects can reveal that B cells exert powerful regulatory control on T cell responses induced by transplantation [6, 194].
6. CONCLUDING REMARKS
We have presented a critical perspective on various of the B cell functions thought to have pertinence for transplantation and have lamented that assays for these functions are scarce and inaccurate. There is another important question regarding the B cell functions that has escaped resolution and it is one we think offers a considerable opportunity. The question and opportunity concern the mechanisms that balance the various B cell functions to reliably achieve adaptive endpoints (i.e. to avoid suppressing the effector immune responses needed for survival). We will suggest several working hypotheses.
One organizing principle concerns the localization of B cells. Although effector antibodies act systemically, antigen presentation to naive T cells, T cell-B cell interaction leading to production of high affinity antibodies, the production of high affinity antibodies and regulation of T cell responses by Treg and Breg are local. If localization of B cells distinguishes the non-canonical functions B cells confer from the functions provided by other cells, one would predict that the B cells that take up and present antigen should migrate to (or differentiate in) different sites in lymphoid tissues than B cells expressing IL-10. Whether the migratory properties of post-phagocytic and post-receptor-mediated endocytosis B cells and putative Breg differ is unknown, in part for want of markers predicting functions yet to be manifest.
The microenvironment in which B cell functions are manifest may provide another organizing principle [79]. Most cytokines and nearly all chemokines are secured in tissues by binding to heparan sulfate. Binding to heparan sulfate can modify the function of proteins and cluster the proteins in ways that heighten delivery of signals. Binding also protects the proteins from degradation. Binding to heparan sulfate is well known to exert profound impact on the biological properties of growth factors and enzymes but the impact on the function of chemokines and cytokines is not so well explored. Chemokines secured by heparan sulfate in lymphoid blood vessels direct the migration of dendritic cells and lymphocytes at sites of inflammation and draining lymphoid tissues [239]. IL-2 secured by heparan sulfate can have a considerable impact on the deletion of antigen-specific T cells [123, 240].
Resting B cells do not appreciably modify the microenvironment but B cells stimulated with LPS produce substantial amounts of heparanase, an endo-β-D-glucuronidase that digests heparan sulfate [122], releasing bound chemokines and other proteins and promoting local remodeling of lymphoid tissues [241]. Remodeling of lymphoid tissues could in turn determine whether and how IL-10 from Breg or other cells impacts on surrounding cells. Thus, heparan sulfate in the extracellular matrix of lymphoid organs could bind IL-10 and thus provide a secure repository. Binding to heparan sulfate may also promote some functions of IL-10 [242] and suppress others [243]. Digestion of heparan sulfate by heparanase released from activated B cells (or T cells) releases IL-10 and other cytokines and chemokines potentially to act on neighboring cells and also depletes the repository. Whether immune regulation by IL-10 reflects the action of cytokine at the time of secretion from Breg or release from stores is, of course, unknown. However, the observation that B cell depletion prior to initiation of immunity has greater impact on Breg function than depletion during the course of an immune response [244] is most consistent with the mechanism reflecting release of IL-10 from stores because IL-10 produced before initiation of immunity would diffuse away from the site or undergo metabolism before activity could be manifest.
Whether these or other organizing principles explains how disparate B cell functions contribute to the efficient development and control of immunity remains to be discovered. Nevertheless, we think knowledge of those principles would add much to understanding the ontogeny and physiology of the B cell compartment. Such knowledge also could be exploited to increase the efficacy and specificity of therapeutic strategies for manipulation B cell function. And, if organizing principles were discovered, then we could dispense with such terms as “canonical” and “non-canonical” we use here or “antibody-independent” and “paradoxical” when referring to the functions B cells as the position of B cells in immune physiology and pathophysiology would be understood, finally.
Funding Sources
The authors’ work pertinent to this communication has been supported by grants from the National Institutes of Health (AI123232), the Department of Defense (CDMRP12459925), the University of Michigan Cardiovascular Center and the Rogel Cancer Center at the University of Michigan.
Abbreviations used
- ADAM10
A disintegrin and metalloproteinase-10
- AMR
Antibody-mediated rejection
- APC
Antigen presenting cell
- AIRE
Autoimmune regulator
- BCR
B cell antigen receptor
- CMR
Cell-mediated rejection
- DC
Dendritic cell
- DSA
Donor-specific antibodies
- EAE
Experimental allergic enchephalomyelitis
- GALT
Gut-associated lymphoid tissue
- LPS
Lipopolysaccharide
- LN
Lymph node
- LTi
lymphoid tissue inducer cell
- LTo
lymphoid tissue organizer cell
- MBT mouse
Monoclonal B cell-T cell mouse
- MT mouse
Monoclonal T cell mouse MT mouse
- Breg
Regulatory B cell
- Treg
Regulatory T cell
- TCR
T cell antigen receptor
- TIM
T cell immunoglobulin and mucin domain
- TLR
Toll-like receptor
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest
None
REFERENCES CITED
- [1].Gorer P: The genetic and antigentic basis of tumor transplantation. Journal of Pathology and Bacteriology 1937;44:691. [Google Scholar]
- [2].Schinstock CA, Gandhi MJ, Stegall MD: Interpreting Anti-HLA Antibody Testing Data: A Practical Guide for Physicians. Transplantation 2016;100:1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Tait BD, Susal C, Gebel HM, Nickerson PW, Zachary AA, Claas FH et al. : Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation. Transplantation 2013;95:19. [DOI] [PubMed] [Google Scholar]
- [4].Filippone EJ, Farber JL: Humoral Immune Response and Allograft Function in Kidney Transplantation. Am J Kidney Dis 2015;66:337. [DOI] [PubMed] [Google Scholar]
- [5].Mangiola M, Marrari M, Feingold B, Zeevi A: Significance of Anti-HLA Antibodies on Adult and Pediatric Heart Allograft Outcomes. Front Immunol 2017;8:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Dijke EI, Platt JL, Blair P, Clatworthy MR, Patel JK, Kfoury AG et al. : B cells in transplantation. J Heart Lung Transplant 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Stetson CA: The role of humoral antibody in the homograft reaction. Advances in Immunology 1963;3:97. [Google Scholar]
- [8].Altman B: Tissue transplantation: circulating antibody in the homotransplantation of kidney and skin. Annals of the Royal College of Surgeons (England) 1963;33:79. [PMC free article] [PubMed] [Google Scholar]
- [9].Gorer PA: Some reactions of H-2 antibodies in vitro and in vivo. Ann N Y Acad Sci 1958;73:707. [DOI] [PubMed] [Google Scholar]
- [10].Hildemann WH, Medawar PB: Relationship between skin transplantation immunity and the formation of humoral isoantibodies in mice. Immunology 1959;2:44. [PMC free article] [PubMed] [Google Scholar]
- [11].Snell GD, Cloudman AM, Failor E, Douglass P: Inhibition and stimulation of tumor homoiotransplants by prior injections of lyophilized tumor tissue. Journal of the National Cancer Institute 1946;6:303. [PubMed] [Google Scholar]
- [12].Cascalho MI, Chen BJ, Kain M, Platt JL: The paradoxical functions of B cells and antibodies in transplantation. Journal of immunology 2013;190:875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Bennet W, Sundberg B, Groth CG, Brendel MD, Brandhorst D, Brandhorst H et al. : Incompatibility between human blood and isolated islets of Langerhans: a finding with implications for clinical intraportal islet transplantation? Diabetes 1999;48:1907. [DOI] [PubMed] [Google Scholar]
- [14].Hankin EH: Reports to the Scientific Grants Committee of the British Medical Association. Br Med J 1890;2:65. [PMC free article] [PubMed] [Google Scholar]
- [15].Burnet FM: The cellular basis of immunology. Nihon Saikingaku Zasshi 1961;16:620. [DOI] [PubMed] [Google Scholar]
- [16].Roitt IM, Greaves MF, Torrigiani G, Brostoff J, Playfair JH: The cellular basis of immunological responses. A synthesis of some current views. Lancet 1969;2:367. [DOI] [PubMed] [Google Scholar]
- [17].Ehrich WE, Harris TN: The Formation of Antibodies in the Popliteal Lymph Node in Rabbits. J Exp Med 1942;76:335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Harris TN, Grimm E, Mertens E, Ehrich WE: The Role of the Lymphocyte in Antibody Formation. J Exp Med 1945;81:73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Glick B, Chang SC, Jaap RG: The bursa of Fabricius and antibody production. Poultry Science 1956;35:224. [Google Scholar]
- [20].Warner NL, Szenberg A: THE IMMUNOLOGICAL FUNCTION OF THE BURSA OF FABRICIUS IN THE CHICKEN. Annu Rev Microbiol 1964;18:253. [DOI] [PubMed] [Google Scholar]
- [21].Cooper MD, Raymond DA, Peterson RD, South MA, Good RA: The functions of the thymus system and the bursa system in the chicken. J Exp Med 1966;123:75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Miller JF: Immunological function of the thymus. Lancet 1961;2:748. [DOI] [PubMed] [Google Scholar]
- [23].Tanay A, Strober S: Opposite effects of total lymphoid irradiation on T cell-dependent and T cell-independent antibody responses. J Immunol 1984;132:979. [PubMed] [Google Scholar]
- [24].Szenberg A, Warner NL: Dissociation of immunological responsiveness in fowls with a hormonally arrested development of lymphoid tissues. Nature 1962;194:146.13874945 [Google Scholar]
- [25].Chan OT, Hannum LG, Haberman AM, Madaio MP, Shlomchik MJ: A novel mouse with B cells but lacking serum antibody reveals an antibody-independent role for B cells in murine lupus. J Exp Med 1999;189:1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Shen P, Fillatreau S: Antibody-independent functions of B cells: a focus on cytokines. Nat Rev Immunol 2015;15:441. [DOI] [PubMed] [Google Scholar]
- [27].Constant SL: B lymphocytes as antigen-presenting cells for CD4+ T cell priming in vivo. J Immunol 1999;162:5695. [PubMed] [Google Scholar]
- [28].Wrenshall LE, Stevens RB, Cerra FB, Platt JL: Modulation of macrophage and B cell function by glycosaminoglycans. Journal of Leukocyte Biology 1999;66:391. [DOI] [PubMed] [Google Scholar]
- [29].Heit A, Huster KM, Schmitz F, Schiemann M, Busch DH, Wagner H: CpG-DNA aided cross-priming by cross-presenting B cells. J Immunol 2004;172:1501. [DOI] [PubMed] [Google Scholar]
- [30].Mauri C, Bosma A: Immune regulatory function of B cells. Annu Rev Immunol 2012;30:221. [DOI] [PubMed] [Google Scholar]
- [31].Yanaba K, Bouaziz JD, Matsushita T, Tsubata T, Tedder TF: The development and function of regulatory B cells expressing IL-10 (B10 cells) requires antigen receptor diversity and TLR signals. J Immunol 2009;182:7459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Saadi S, Wrenshall LE, Platt JL: Regional manifestations and control of the immune system. FASEB Journal 2002;16:849. [DOI] [PubMed] [Google Scholar]
- [33].Ehrlich P: On immunity with special reference to cell life. Proceedings of the Royal Society of London 1900;66:424. [Google Scholar]
- [34].Platt JL, Cascalho M: Donor specific antibodies after transplantation. Pediatric Transplantation 2011;15:686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Valenzuela NM, Reed EF: Antibody-mediated rejection across solid organ transplants: manifestations, mechanisms, and therapies. J Clin Invest 2017;127:2492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Haas M: The relationship between pathologic lesions of active and chronic antibody-mediated rejection in renal allografts. Am J Transplant 2018;18:2849. [DOI] [PubMed] [Google Scholar]
- [37].Kaliss N: Immunological enhancement of tumor homografts in mice: a review. Cancer Res 1958;18:992. [PubMed] [Google Scholar]
- [38].Snell GD, Cloudman AM, Woodworth E: Tumor immunity in mice, induced with lyophilized tissue, as influenced by tumor strain, host strain, source of tissue, and dosage. Cancer Res 1948;8:429. [PubMed] [Google Scholar]
- [39].Kaliss N, Molomut N: The effect of prior injections of tissue antiserums on the survival of cancer homoiografts in mice. Cancer Res 1952;12:110. [PubMed] [Google Scholar]
- [40].Carpenter CB, d’Apice AJ, Abbas AK: The role of antibodies in the rejection and enhancement of organ allografts. Adv Immunol 1976;22:1. [DOI] [PubMed] [Google Scholar]
- [41].Morris PJ: Suppression of rejection of organ allografts by alloantibody. Immunol Rev 1980;49:93. [DOI] [PubMed] [Google Scholar]
- [42].Cascalho M, Platt JL: Harnessing B cells in immunotherapy. Immunotherapy 2016;8:237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Germain C, Gnjatic S, Dieu-Nosjean MC: Tertiary Lymphoid Structure-Associated B Cells are Key Players in Anti-Tumor Immunity. Front Immunol 2015;6:67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Pipi E, Nayar S, Gardner DH, Colafrancesco S, Smith C, Barone F: Tertiary Lymphoid Structures: Autoimmunity Goes Local. Front Immunol 2018;9:1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Alsughayyir J, Pettigrew GJ, Motallebzadeh R: Spoiling for a Fight: B Lymphocytes As Initiator and Effector Populations within Tertiary Lymphoid Organs in Autoimmunity and Transplantation. Front Immunol 2017;8:1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Tsuji S, Stein L, Kamada N, Nunez G, Bram R, Vallance BA et al. : TACI deficiency enhances antibody avidity and clearance of an intestinal pathogen. J Clin Invest 2014;124:4857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Pober JS, Merola J, Liu R, Manes TD: Antigen Presentation by Vascular Cells. Front Immunol 2017;8:1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Platt JL, Kaufman CL, de Mattos Garcia Barbosa M, Cascalho M: Accommodation and related conditions in vascularized composite allografts. Curr Opin Organ Transplant 2017;22:470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Woodruff MF: Evidence of adaptation in homografts of normal tissue. Bull Soc Int Chir 1959;18:131. [PubMed] [Google Scholar]
- [50].Woodruff MF: Biological and Clinical Aspects of Organ Transplantation. Br Med Bull 1965;21:176. [DOI] [PubMed] [Google Scholar]
- [51].Ceppellini R, Garotta G, Malavasi F, Trucco M: Modulation of expression of HLA components at the cell surface induced by anti-beta 2m reagents. Tissue Antigens 1981;17:28. [DOI] [PubMed] [Google Scholar]
- [52].Yuzawa Y, Brentjens JR, Brett J, Caldwell PRB, Esposito C, Fukatsu A et al. : Antibody-mediated redistribution and shedding of endothelial antigens in the rabbit. Journal of Immunology 1993;150:5633. [PubMed] [Google Scholar]
- [53].Kooyman DL, Byrne GW, McClellan S, Nielsen D, Tone M, McCurry KR et al. : In vivo transfer of GPI-linked complement restriction factors from erythrocytes to the endothelium. Science 1995;269:89. [DOI] [PubMed] [Google Scholar]
- [54].Snell GD, Winn HJ, Stimpfling JH, Parker SJ: Depression by Antibody of the Immune Response to Homografts and Its Role in Immunological Enhancement. J Exp Med 1960;112:293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Dejnirattisai W, Supasa P, Wongwiwat W, Rouvinski A, Barba-Spaeth G, Duangchinda T et al. : Dengue virus sero-cross-reactivity drives antibody-dependent enhancement of infection with zika virus. Nat Immunol 2016;17:1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Platt JL, Vercellotti GM, Dalmasso AP, Matas AJ, Bolman RM, Najarian JS et al. : Transplantation of discordant xenografts: a review of progress. Immunology Today 1990;11:450. [DOI] [PubMed] [Google Scholar]
- [57].de Mattos Garcia Barbosa M, Cascalho M, Platt JL: Accommodation in ABO-incompatible organ transplants. Xenotransplantation 2018;25:e12418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Koch CA, Platt JL: Natural mechanisms for evading graft rejection: the fetus as an allograft. Springer Seminars in Immunopathology 2003;25:95. [DOI] [PubMed] [Google Scholar]
- [59].Koch CA, Khalpey ZI, Platt JL: Accommodation: preventing injury in transplantation and disease. Journal of Immunology 2004;172:5143. [DOI] [PubMed] [Google Scholar]
- [60].Lynch RJ, Silva IA, Chen BJ, Punch JD, Cascalho M, Platt JL: Cryptic B cell response to renal transplantation. Am J Transplant 2013;13:1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Salama AD, Delikouras A, Pusey CD, Cook HT, Bhangal G, Lechler RI et al. : Transplant accommodation in highly sensitized patients: a potential role for Bcl-xL and alloantibody. Am J Transplant 2001;1:260. [DOI] [PubMed] [Google Scholar]
- [62].Bach FH, Hancock WW, Ferran C: Protective genes expressed in endothelial cells: a regulatory response to injury. Immunology Today 1997;18:483. [DOI] [PubMed] [Google Scholar]
- [63].Williams JM, Holzknecht ZE, Plummer TB, Lin SS, Brunn GJ, Platt JL: Acute vascular rejection and accommodation: divergent outcomes of the humoral response to organ transplantation. Transplantation 2004;78:1471. [DOI] [PubMed] [Google Scholar]
- [64].Dalmasso AP, Benson BA, Johnson JS, Lancto C, Abrahamsen MS: Resistance against the membrane attack complex of complement induced in porcine endothelial cells with a Gal alpha(1–3)Gal binding lectin: up-regulation of CD59 expression. Journal of Immunology 2000;164:3764. [DOI] [PubMed] [Google Scholar]
- [65].Koch CA, Kanazawa A, Nishitai R, Knudsen BE, Ogata K, Plummer TB et al. : Intrinsic resistance of hepatocytes to complement-mediated injury. Journal of Immunology 2005;174:7302. [DOI] [PubMed] [Google Scholar]
- [66].Ehrenstein MR, Notley CA: The importance of natural IgM: scavenger, protector and regulator. Nat Rev Immunol 2010;10:778. [DOI] [PubMed] [Google Scholar]
- [67].Gronwall C, Silverman GJ: Natural IgM: beneficial autoantibodies for the control of inflammatory and autoimmune disease. J Clin Immunol 2014;34 Suppl 1:S12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Dimitrov JD, Planchais C, Roumenina LT, Vassilev TL, Kaveri SV, Lacroix-Desmazes S: Antibody polyreactivity in health and disease: statu variabilis. J Immunol 2013;191:993. [DOI] [PubMed] [Google Scholar]
- [69].Zorn E: New insights on innate B-cell immunity in transplantation. Xenotransplantation 2018;25:e12417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].Casali P, Notkins AL: CD5+ B lymphocytes, polyreactive antibodies and the human B-cell repertoire. Immunology Today 1989;10:364. [DOI] [PubMed] [Google Scholar]
- [71].Gunti S, Notkins AL: Polyreactive Antibodies: Function and Quantification. J Infect Dis 2015;212 Suppl 1:S42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Durandy A, Kaveri SV, Kuijpers TW, Basta M, Miescher S, Ravetch JV et al. : Intravenous immunoglobulins--understanding properties and mechanisms. Clin Exp Immunol 2009;158 Suppl 1:2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Nimmerjahn F, Ravetch JV: Fcgamma receptors: old friends and new family members. Immunity 2006;24:19. [DOI] [PubMed] [Google Scholar]
- [74].Weiser MR, Williams JP, Moore FDJ, Kobzik L, Ma M, Hechtman HB et al. : Reperfusion injury of ischemic skeletal muscle is mediated by natural antibody and complement. Journal of Experimental Medicine 1996;183:2343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Lobo PI, Bajwa A, Schlegel KH, Vengal J, Lee SJ, Huang L et al. : Natural IgM anti-leukocyte autoantibodies attenuate excess inflammation mediated by innate and adaptive immune mechanisms involving Th-17. J Immunol 2012;188:1675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Wootla B, Watzlawik JO, Denic A, Rodriguez M: The road to remyelination in demyelinating diseases: current status and prospects for clinical treatment. Expert Rev Clin Immunol 2013;9:535. [DOI] [PubMed] [Google Scholar]
- [77].Platt JL, Cascalho M: IgM in the kidney: a multiple personality disorder. Kidney Int 2015;88:439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Nus M, Tsiantoulas D, Mallat Z: Plan B (-cell) in atherosclerosis. Eur J Pharmacol 2017;816:76. [DOI] [PubMed] [Google Scholar]
- [79].Platt JL, Wrenshall LE, Johnson GB, Cascalho M: Heparan Sulfate Proteoglycan Metabolism and the Fate of Grafted Tissues. Adv Exp Med Biol 2015;865:123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Bournazos S, Ravetch JV: Diversification of IgG effector functions. Int Immunol 2017;29:303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Silverman GJ, Vas J, Gronwall C: Protective autoantibodies in the rheumatic diseases: lessons for therapy. Nat Rev Rheumatol 2013;9:291. [DOI] [PubMed] [Google Scholar]
- [82].Lobo PI, Schlegel KH, Bajwa A, Huang L, Kurmaeva E, Wang B et al. : Natural IgM Switches the Function of Lipopolysaccharide-Activated Murine Bone Marrow-Derived Dendritic Cells to a Regulatory Dendritic Cell That Suppresses Innate Inflammation. J Immunol 2015;195:5215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Anthony RM, Wermeling F, Ravetch JV: Novel roles for the IgG Fc glycan. Ann N Y Acad Sci 2012;1253:170. [DOI] [PubMed] [Google Scholar]
- [84].Burastero SE, Casali P, Wilder RL, Notkins AL: Monoreactive high affinity and polyreactive low affinity rheumatoid factors are produced by CD5+ B Cells from patients with rheumatoid arthritis. Journal of Experimental Medicine 1988;168:1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Jordan SC, Vo AA, Peng A, Toyoda M, Tyan D: Intravenous gammaglobulin (IVIG): a novel approach to improve transplant rates and outcomes in highly HLA-sensitized patients. Am J Transplant 2006;6:459. [DOI] [PubMed] [Google Scholar]
- [86].Fagarasan S, Honjo T: T-Independent immune response: new aspects of B cell biology. Science 2000;290:89. [DOI] [PubMed] [Google Scholar]
- [87].Raveche ES: Possible immunoregulatory role for CD5 + B cells. Clin Immunol Immunopathol 1990;56:135. [DOI] [PubMed] [Google Scholar]
- [88].Platt JL: C4d and the fate of organ allografts. J Am Soc Nephrol 2002;13:2417. [DOI] [PubMed] [Google Scholar]
- [89].Magee JC, Collins BH, Harland RC, Lindman BJ, Bollinger RR, Frank MM et al. : Immunoglobulin prevents complement mediated hyperacute rejection in swine-to-primate xenotransplantation. Journal of Clinical Investigation 1995;96:2404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [90].Jaffe CJ, Atkinson JP, Frank MM: The role of complement in the clearance of cold agglutinin-sensitized erythrocytes in man. Journal of Clinical Investigation 1976;58:942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Atkinson JP, Frank MM: Studies on the in vivo effects of antibody. Journal of Clinical Investigation 1974;54:339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Frank MM, Basta M, Fries LF: The effects of intravenous immune globulin on complement-dependent immune damage of cells and tissues. Clinical Immunology and Immunopathology 1992;62:s82. [DOI] [PubMed] [Google Scholar]
- [93].Gonzalez M, Mackay F, Browning JL, Kosco-Vilbois MH, Noelle RJ: The sequential role of lymphotoxin and B cells in the development of splenic follicles. J Exp Med 1998;187:997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Fu YX, Chaplin DD: Development and maturation of secondary lymphoid tissues. Annu Rev Immunol 1999;17:399. [DOI] [PubMed] [Google Scholar]
- [95].Ngo VN, Cornall RJ, Cyster JG: Splentic T zone development is B cell dependent. Journal of Experimental Medicine 2001;194:1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Cyster JG: Lymphoid organ development and cell migration. Immunol Rev 2003;195:5. [DOI] [PubMed] [Google Scholar]
- [97].Vondenhoff MF, Kraal G, Mebius RE: Lymphoid organogenesis in brief. Eur J Immunol 2007;37 Suppl 1:S46. [DOI] [PubMed] [Google Scholar]
- [98].Neely HR, Flajnik MF: Emergence and Evolution of Secondary Lymphoid Organs. Annu Rev Cell Dev Biol 2016;32:693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Onder L, Ludewig B: A Fresh View on Lymph Node Organogenesis. Trends Immunol 2018. [DOI] [PubMed] [Google Scholar]
- [100].Eberl G, Marmon S, Sunshine MJ, Rennert PD, Choi Y, Littman DR: An essential function for the nuclear receptor RORgamma(t) in the generation of fetal lymphoid tissue inducer cells. Nat Immunol 2004;5:64. [DOI] [PubMed] [Google Scholar]
- [101].Endres R, Alimzhanov MB, Plitz T, Futterer A, Kosco-Vilbois MH, Nedospasov SA et al. : Mature follicular dendritic cell networks depend on expression of lymphotoxin beta receptor by radioresistant stromal cells and of lymphotoxin beta and tumor necrosis factor by B cells. J Exp Med 1999;189:159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [102].AbuAttieh M, Rebrovich M, Wettstein PJ, Vuk-Pavlovic Z, Limper AH, Platt JL et al. : Fitness of cell-mediated immunity independent of repertoire diversity. The Journal of Immunology 2007;178:2950. [DOI] [PubMed] [Google Scholar]
- [103].Randall TD, Carragher DM, Rangel-Moreno J: Development of secondary lymphoid organs. Annu Rev Immunol 2008;26:627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [104].Fu YX, Huang G, Wang Y, Chaplin DD: B lymphocytes induce the formation of follicular dendritic cell clusters in a lymphotoxin alpha-dependent fashion. J Exp Med 1998;187:1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [105].Traggiai E, Chicha L, Mazzucchelli L, Bronz L, Piffaretti JC, Lanzavecchia A et al. : Development of a human adaptive immune system in cord blood cell-transplanted mice. Science 2004;304:104. [DOI] [PubMed] [Google Scholar]
- [106].Seymour R, Sundberg JP, Hogenesch H: Abnormal lymphoid organ development in immunodeficient mutant mice. Vet Pathol 2006;43:401. [DOI] [PubMed] [Google Scholar]
- [107].Lang J, Kelly M, Freed BM, McCarter MD, Kedl RM, Torres RM et al. : Studies of lymphocyte reconstitution in a humanized mouse model reveal a requirement of T cells for human B cell maturation. J Immunol 2013;190:2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Keshavarzi S, Rietz C, Simoes S, Shih S, Platt JL, Wong J et al. : The possibility of B-cell dependent T-cell development and function. Scandinavian Journal of Immunology 2003;57:446. [DOI] [PubMed] [Google Scholar]
- [109].Zhang Y, Meyer-Hermann M, George LA, Figge MT, Khan M, Goodall M et al. : Germinal center B cells govern their own fate via antibody feedback. J Exp Med 2013;210:457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [110].Allen CD, Okada T, Cyster JG: Germinal-center organization and cellular dynamics. Immunity 2007;27:190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Victora GD, Nussenzweig MC: Germinal centers. Annu Rev Immunol 2012;30:429. [DOI] [PubMed] [Google Scholar]
- [112].De Silva NS, Klein U: Dynamics of B cells in germinal centres. Nat Rev Immunol 2015;15:137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [113].Kuprash DV, Alimzhanov MB, Tumanov AV, Anderson AO, Pfeffer K, Nedospasov SA: TNF and lymphotoxin beta cooperate in the maintenance of secondary lymphoid tissue microarchitecture but not in the development of lymph nodes. J Immunol 1999;163:6575. [PubMed] [Google Scholar]
- [114].Kumar V, Scandella E, Danuser R, Onder L, Nitschke M, Fukui Y et al. : Global lymphoid tissue remodeling during a viral infection is orchestrated by a B cell-lymphotoxin-dependent pathway. Blood 2010;115:4725. [DOI] [PubMed] [Google Scholar]
- [115].Abe J, Ueha S, Yoneyama H, Shono Y, Kurachi M, Goto A et al. : B cells regulate antibody responses through the medullary remodeling of inflamed lymph nodes. Int Immunol 2012;24:17. [DOI] [PubMed] [Google Scholar]
- [116].Nolte MA, Arens R, Kraus M, van Oers MH, Kraal G, van Lier RA et al. : B cells are crucial for both development and maintenance of the splenic marginal zone. J Immunol 2004;172:3620. [DOI] [PubMed] [Google Scholar]
- [117].Kanayama N, Cascalho M, Ohmori H: Analysis of marginal zone B cell development in the mouse with limited B cell diversity: role of the antigen receptor signals in the recruitment of B cells to the marginal zone. J Immunol 2005;174:1438. [DOI] [PubMed] [Google Scholar]
- [118].Schroder C, Azimzadeh AM, Wu G, Price JO, Atkinson JB, Pierson RN: Anti-CD20 treatment depletes B-cells in blood and lymphatic tissue of cynomolgus monkeys. Transpl Immunol 2003;12:19. [DOI] [PubMed] [Google Scholar]
- [119].Genberg H, Hansson A, Wernerson A, Wennberg L, Tyden G: Pharmacodynamics of rituximab in kidney allotransplantation. Am J Transplant 2006;6:2418. [DOI] [PubMed] [Google Scholar]
- [120].Cioc AM, Vanderwerf SM, Peterson BA, Robu VG, Forster CL, Pambuccian SE: Rituximab-induced changes in hematolymphoid tissues found at autopsy. Am J Clin Pathol 2008;130:604. [DOI] [PubMed] [Google Scholar]
- [121].Buser A, Stern M, Arber C, Medinger M, Halter J, Rovo A et al. : Impaired B-cell reconstitution in lymphoma patients undergoing allogeneic HSCT: an effect of pretreatment with rituximab? Bone Marrow Transplant 2008;42:483. [DOI] [PubMed] [Google Scholar]
- [122].Laskov R, Michaeli R-I, Sharir H, Yefenof E, Vlodavsky I: Production of heparanse by normal and neoplastic murine B-lymphocytes. International Journal of Cancer 1991;47:92. [DOI] [PubMed] [Google Scholar]
- [123].Wrenshall LE, Platt JL: Regulation of T cell homeostasis by heparan sulfate-bound IL-2. Journal of Immunology 1999;163:3793. [PubMed] [Google Scholar]
- [124].Gibb DR, El Shikh M, Kang DJ, Rowe WJ, El Sayed R, Cichy J et al. : ADAM10 is essential for Notch2-dependent marginal zone B cell development and CD23 cleavage in vivo. J Exp Med 2010;207:623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [125].Chaimowitz NS, Martin RK, Cichy J, Gibb DR, Patil P, Kang DJ et al. : A disintegrin and metalloproteinase 10 regulates antibody production and maintenance of lymphoid architecture. J Immunol 2011;187:5114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [126].Jones GW, Hill DG, Jones SA: Understanding Immune Cells in Tertiary Lymphoid Organ Development: It Is All Starting to Come Together. Front Immunol 2016;7:401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [127].Motallebzadeh R, Rehakova S, Conlon TM, Win TS, Callaghan CJ, Goddard M et al. : Blocking lymphotoxin signaling abrogates the development of ectopic lymphoid tissue within cardiac allografts and inhibits effector antibody responses. FASEB J 2012;26:51. [DOI] [PubMed] [Google Scholar]
- [128].Eisenberg R, Looney RJ: The therapeutic potential of anti-CD20 “what do B-cells do?”. Clin Immunol 2005;117:207. [DOI] [PubMed] [Google Scholar]
- [129].Moreso F, Crespo M, Ruiz JC, Torres A, Gutierrez-Dalmau A, Osuna A et al. : Treatment of chronic antibody mediated rejection with intravenous immunoglobulins and rituximab: A multicenter, prospective, randomized, double-blind clinical trial. Am J Transplant 2018;18:927. [DOI] [PubMed] [Google Scholar]
- [130].Thaunat O, Patey N, Gautreau C, Lechaton S, Fremeaux-Bacchi V, Dieu-Nosjean MC et al. : B cell survival in intragraft tertiary lymphoid organs after rituximab therapy. Transplantation 2008;85:1648. [DOI] [PubMed] [Google Scholar]
- [131].Warner NL, Szenberg A: Effect of neonatal thymectomy on the immune response in the chicken. Nature 1962;196:784. [DOI] [PubMed] [Google Scholar]
- [132].Good RA, Dalmasso AP, Martinez C, Archer OK, Pierce JC, Papermaster BW: The role of the thymus in development of immunologic capacity in rabbits and mice. J Exp Med 1962;116:733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [133].Claman HN, Chaperon EA, Triplett RF: Thymus-marrow cell combinations. Synergism in antibody production. Proc Soc Exp Biol Med 1966;122:1167. [DOI] [PubMed] [Google Scholar]
- [134].Platt JL, Silva I, Balin SJ, Lefferts AR, Farkash E, Ross TM et al. : C3d regulates immune checkpoint blockade and enhances antitumor immunity. JCI Insight 2017;2:pii: 90201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [135].João CM, Ogle BM, Gay-Rubenstein C, Platt JL, Cascalho M: B cell-dependent TCR diversification. Journal of Immunology 2004;172:4709. [DOI] [PubMed] [Google Scholar]
- [136].Chen J, Trounstine M, Alt FW, Young F, Kurahara C, Loring JF et al. : Immunoglobulin gene rearrangement in B cell deficient mice generated by targeted deletion of the JH locus. Int Immunol 1993;5:647. [DOI] [PubMed] [Google Scholar]
- [137].Cascalho M, Platt JL: B cell-dependent T cell development. Acta Paediatrica 2004;93:52. [DOI] [PubMed] [Google Scholar]
- [138].Lykken JM, DiLillo DJ, Weimer ET, Roser-Page S, Heise MT, Grayson JM et al. : Acute and chronic B cell depletion disrupts CD4+ and CD8+ T cell homeostasis and expansion during acute viral infection in mice. J Immunol 2014;193:746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [139].Grusby MJ, Auchincloss H Jr., Lee R, Johnson RS, Spencer JP, Zijlstra M et al. : Mice lacking major histocompatibility complex class I and class II molecules. Proc Natl Acad Sci U S A 1993;90:3913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [140].Markowitz JS, Auchincloss H Jr, Grusby MJ, Glimcher LH: Class II-positive hematopoietic cells cannot mediate positive selection of CD4+ T lymphocytes in class II-deficient mice. Proc Natl Acad Sci U S A 1993;90:2779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [141].Surh CD, Lee DS, Fung-Leung WP, Karlsson L, Sprent J: Thymic selection by a single MHC/peptide ligand produces a semidiverse repertoire of CD4+ T cells. Immunity 1997;7:209. [DOI] [PubMed] [Google Scholar]
- [142].Qi Q, Liu Y, Cheng Y, Glanville J, Zhang D, Lee JY et al. : Diversity and clonal selection in the human T-cell repertoire. Proc Natl Acad Sci U S A 2014;111:13139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [143].Petrova G, Ferrante A, Gorski J: Cross-reactivity of T cells and its role in the immune system. Crit Rev Immunol 2012;32:349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [144].Nelson RW, Beisang D, Tubo NJ, Dileepan T, Wiesner DL, Nielsen K et al. : T cell receptor cross-reactivity between similar foreign and self peptides influences naive cell population size and autoimmunity. Immunity 2015;42:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [145].Mason D: A very high level of crossreactivity is an essential feature of the T-cell receptor. Immunol Today 1998;19:395. [DOI] [PubMed] [Google Scholar]
- [146].Nikolich-Zugich J, Slifka MK, Messaoudi I: The many important facets of T-cell repertoire diversity. Nat Rev Immunol 2004;4:123. [DOI] [PubMed] [Google Scholar]
- [147].Stritesky GL, Jameson SC, Hogquist KA: Selection of self-reactive T cells in the thymus. Annu Rev Immunol 2012;30:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [148].Hayglass KT, Naides SJ, Scott CF Jr., Benacerraf B, Sy MS: T cell development in B cell-deficient mice. IV. The role of B cells as antigen-presenting cells in vivo. J Immunol 1986;136:823. [PubMed] [Google Scholar]
- [149].Liu Y, Wu Y, Ramarathinam L, Guo Y, Huszar D, Trounstine M et al. : Gene-targeted B-deficient mice reveal a critical role for B cells in the CD4 T cell response. Int Immunol 1995;7:1353. [DOI] [PubMed] [Google Scholar]
- [150].Avivi I, Stroopinsky D, Katz T: Anti-CD20 monoclonal antibodies: beyond B-cells. Blood Rev 2013;27:217. [DOI] [PubMed] [Google Scholar]
- [151].Ogle BM, West LJ, Driscoll DJ, Strome SE, Razonable RR, Paya CV et al. : Effacing of the T cell compartment by cardiac transplantation in infancy. Journal of Immunology 2006;176:1962. [DOI] [PubMed] [Google Scholar]
- [152].Benacerraf B: A hypothesis to relate the specificity of T lymphocytes and the activity of I region-specific Ir genes in macrophages and B lymphocytes. J Immunol 1978;120:1809. [PubMed] [Google Scholar]
- [153].Adler LN, Jiang W, Bhamidipati K, Millican M, Macaubas C, Hung SC et al. : The Other Function: Class II-Restricted Antigen Presentation by B Cells. Front Immunol 2017;8:319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [154].Chesnut RW, Grey HM: Studies on the capacity of B cells to serve as antigen-presenting cells. J Immunol 1981;126:1075. [PubMed] [Google Scholar]
- [155].Lanzavecchia A: Antigen-specific interaction between T and B cells. Nature 1985;314:537. [DOI] [PubMed] [Google Scholar]
- [156].Constant S, Schweitzer N, West J, Ranney P, Bottomly K: B lymphocytes can be competent antigen-presenting cells for priming CD4+ T cells to protein antigens in vivo. J Immunol 1995;155:3734. [PubMed] [Google Scholar]
- [157].Colluru VT, McNeel DG: B lymphocytes as direct antigen-presenting cells for anti-tumor DNA vaccines. Oncotarget 2016;7:67901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [158].Cascalho M, Balin SJ, Platt JL: The mutable vaccine for mutable viruses. Immunotherapy 2017;9:659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [159].Hong S, Zhang Z, Liu H, Tian M, Zhu X, Zhang Z et al. : B Cells Are the Dominant Antigen-Presenting Cells that Activate Naive CD4(+) T Cells upon Immunization with a Virus-Derived Nanoparticle Antigen. Immunity 2018;49:695. [DOI] [PubMed] [Google Scholar]
- [160].Rivera A, Chen CC, Ron N, Dougherty JP, Ron Y: Role of B cells as antigen-presenting cells in vivo revisited: antigen-specific B cells are essential for T cell expansion in lymph nodes and for systemic T cell responses to low antigen concentrations. Int Immunol 2001;13:1583. [DOI] [PubMed] [Google Scholar]
- [161].Phillips JA, Romball CG, Hobbs MV, Ernst DN, Shultz L, Weigle WO: CD4+ T cell activation and tolerance induction in B cell knockout mice. J Exp Med 1996;183:1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [162].Metlay JP, Pure E, Steinman RM: Control of the immune response at the level of antigen-presenting cells: a comparison of the function of dendritic cells and B lymphocytes. Adv Immunol 1989;47:45. [DOI] [PubMed] [Google Scholar]
- [163].Banchereau J, Steinman RM: Dendritic cells and the control of immunity. Nature 1998;392:245. [DOI] [PubMed] [Google Scholar]
- [164].Kodaira Y, Nair SK, Wrenshall LE, Gilboa E, Platt JL: Phenotypic and functional maturation of dendritic cells modulated by heparan sulfate. Journal of Immunology 2000;165:1599. [DOI] [PubMed] [Google Scholar]
- [165].Bannard O, McGowan SJ, Ersching J, Ishido S, Victora GD, Shin JS et al. : Ubiquitin-mediated fluctuations in MHC class II facilitate efficient germinal center B cell responses. J Exp Med 2016;213:993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [166].Davidson HW, Reid PA, Lanzavecchia A, Watts C: Processed antigen binds to newly synthesized MHC class II molecules in antigen-specific B lymphocytes. Cell 1991;67:105. [DOI] [PubMed] [Google Scholar]
- [167].Gitlin AD, Shulman Z, Nussenzweig MC: Clonal selection in the germinal centre by regulated proliferation and hypermutation. Nature 2014;509:637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [168].Bautista BL, Devarajan P, McKinstry KK, Strutt TM, Vong AM, Jones MC et al. : Short-Lived Antigen Recognition but Not Viral Infection at a Defined Checkpoint Programs Effector CD4 T Cells To Become Protective Memory. J Immunol 2016;197:3936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [169].Whitmire JK, Murali-Krishna K, Altman J, Ahmed R: Antiviral CD4 and CD8 T-cell memory: differences in the size of the response and activation requirements. Philos Trans R Soc Lond B Biol Sci 2000;355:373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [170].Dubois Cauwelaert N, Baldwin SL, Orr MT, Desbien AL, Gage E, Hofmeyer KA et al. : Antigen presentation by B cells guides programing of memory CD4(+) T-cell responses to a TLR4-agonist containing vaccine in mice. Eur J Immunol 2016;46:2719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [171].LeBien TW, Tedder TF: B lymphocytes: how they develop and function. Blood 2008;112:1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [172].Redfield RR 3rd, Rodriguez E, Parsons R, Vivek K, Mustafa MM, Noorchashm H et al. : Essential role for B cells in transplantation tolerance. Curr Opin Immunol 2011;23:685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [173].Firl DJ, Benichou G, Kim JI, Yeh H: A Paradigm Shift on the Question of B Cells in Transplantation? Recent Insights on Regulating the Alloresponse. Front Immunol 2017;8:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [174].Karahan GE, Claas FH, Heidt S: B Cell Immunity in Solid Organ Transplantation. Front Immunol 2016;7:686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [175].Noorchashm H, Reed AJ, Rostami SY, Mozaffari R, Zekavat G, Koeberlein B et al. : B cell-mediated antigen presentation is required for the pathogenesis of acute cardiac allograft rejection. J Immunol 2006;177:7715. [DOI] [PubMed] [Google Scholar]
- [176].Zeng Q, Ng YH, Singh T, Jiang K, Sheriff KA, Ippolito R et al. : B cells mediate chronic allograft rejection independently of antibody production. J Clin Invest 2014;124:1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [177].Ng YH, Oberbarnscheidt MH, Chandramoorthy HC, Hoffman R, Chalasani G: B cells help alloreactive T cells differentiate into memory T cells. Am J Transplant 2010;10:1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [178].Shiu KY, McLaughlin L, Rebollo-Mesa I, Zhao J, Semik V, Cook HT et al. : B-lymphocytes support and regulate indirect T-cell alloreactivity in individual patients with chronic antibody-mediated rejection. Kidney Int 2015;88:560. [DOI] [PubMed] [Google Scholar]
- [179].Epstein MM, Di Rosa F, Jankovic D, Sher A, Matzinger P: Successful T cell priming in B cell-deficient mice. J Exp Med 1995;182:915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [180].Brandle D, Joergensen J, Zenke G, Burki K, Hof RP: Contribution of donor-specific antibodies to acute allograft rejection: evidence from B cell-deficient mice. Transplantation 1998;65:1489. [DOI] [PubMed] [Google Scholar]
- [181].DiLillo DJ, Griffiths R, Seshan SV, Magro CM, Ruiz P, Coffman TM et al. : B lymphocytes differentially influence acute and chronic allograft rejection in mice. J Immunol 2011;186:2643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [182].DiLillo DJ, Matsushita T, Tedder TF: B10 cells and regulatory B cells balance immune responses during inflammation, autoimmunity, and cancer. Ann N Y Acad Sci 2010;1183:38. [DOI] [PubMed] [Google Scholar]
- [183].Lund FE, Randall TD: Effector and regulatory B cells: modulators of CD4+ T cell immunity. Nat Rev Immunol 2010;10:236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [184].Mauri C, Menon M: Human regulatory B cells in health and disease: therapeutic potential. J Clin Invest 2017;127:772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [185].Ding Q, Yeung M, Camirand G, Zeng Q, Akiba H, Yagita H et al. : Regulatory B cells are identified by expression of TIM-1 and can be induced through TIM-1 ligation to promote tolerance in mice. J Clin Invest 2011;121:3645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [186].Angiari S, Constantin G: Regulation of T cell trafficking by the T cell immunoglobulin and mucin domain 1 glycoprotein. Trends Mol Med 2014;20:675. [DOI] [PubMed] [Google Scholar]
- [187].Ding Q, Mohib K, Kuchroo VK, Rothstein DM: TIM-4 Identifies IFN-gamma-Expressing Proinflammatory B Effector 1 Cells That Promote Tumor and Allograft Rejection. J Immunol 2017;199:2585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [188].Kaliss N: The survival of homografts in mice pretreated with antisera to mouse tissue. Ann N Y Acad Sci 1957;64:977. [DOI] [PubMed] [Google Scholar]
- [189].Katz SI, Parker D, Turk JL: B-cell suppression of delayed hypersensitivity reactions. Nature 1974;251:550. [DOI] [PubMed] [Google Scholar]
- [190].Koenig S, Hoffmann MK: Bacterial lipopolysaccharide activates suppressor B lymphocytes. Proc Natl Acad Sci U S A 1979;76:4608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [191].del Guercio P, Katz DH: B-cell-derived lymphokines: regulatory effects on the immune system. Crit Rev Immunol 1986;6:185. [PubMed] [Google Scholar]
- [192].Platt JL, Tsuji S, Cascalho M: Novel functions of B cells in transplantation. Current Opinion in Organ Transplantation 2011;16:61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [193].Chesneau M, Michel L, Degauque N, Brouard S: Regulatory B cells and tolerance in transplantation: from animal models to human. Front Immunol 2013;4:497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [194].Clatworthy MR: B-cell regulation and its application to transplantation. Transpl Int 2014;27:117. [DOI] [PubMed] [Google Scholar]
- [195].Rosser EC, Mauri C: Regulatory B cells: origin, phenotype, and function. Immunity 2015;42:607. [DOI] [PubMed] [Google Scholar]
- [196].Wolf SD, Dittel BN, Hardardottir F, Janeway CA Jr.: Experimental autoimmune encephalomyelitis induction in genetically B cell-deficient mice. J Exp Med 1996;184:2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [197].Fillatreau S, Sweenie CH, McGeachy MJ, Gray D, Anderton SM: B cells regulate autoimmunity by provision of IL-10. Nat Immunol 2002;3:944. [DOI] [PubMed] [Google Scholar]
- [198].Matsushita T, Yanaba K, Bouaziz JD, Fujimoto M, Tedder TF: Regulatory B cells inhibit EAE initiation in mice while other B cells promote disease progression. J Clin Invest 2008;118:3420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [199].Newell KA, Asare A, Kirk AD, Gisler TD, Bourcier K, Suthanthiran M et al. : Identification of a B cell signature associated with renal transplant tolerance in humans. J Clin Invest 2010;120:1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [200].Chesneau M, Pallier A, Braza F, Lacombe G, Le Gallou S, Baron D et al. : Unique B cell differentiation profile in tolerant kidney transplant patients. Am J Transplant 2014;14:144. [DOI] [PubMed] [Google Scholar]
- [201].Clatworthy MR, Watson CJ, Plotnek G, Bardsley V, Chaudhry AN, Bradley JA et al. : B-cell-depleting induction therapy and acute cellular rejection. N Engl J Med 2009;360:2683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [202].Robinson AP, Harp CT, Noronha A, Miller SD: The experimental autoimmune encephalomyelitis (EAE) model of MS: utility for understanding disease pathophysiology and treatment. Handb Clin Neurol 2014;122:173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [203].Hjelmstrom P, Juedes AE, Fjell J, Ruddle NH: B-cell-deficient mice develop experimental allergic encephalomyelitis with demyelination after myelin oligodendrocyte glycoprotein sensitization. J Immunol 1998;161:4480. [PubMed] [Google Scholar]
- [204].Lyons JA, San M, Happ MP, Cross AH: B cells are critical to induction of experimental allergic encephalomyelitis by protein but not by a short encephalitogenic peptide. Eur J Immunol 1999;29:3432. [DOI] [PubMed] [Google Scholar]
- [205].Svensson L, Abdul-Majid KB, Bauer J, Lassmann H, Harris RA, Holmdahl R: A comparative analysis of B cell-mediated myelin oligodendrocyte glycoprotein-experimental autoimmune encephalomyelitis pathogenesis in B cell-deficient mice reveals an effect on demyelination. Eur J Immunol 2002;32:1939. [DOI] [PubMed] [Google Scholar]
- [206].Mann MK, Ray A, Basu S, Karp CL, Dittel BN: Pathogenic and regulatory roles for B cells in experimental autoimmune encephalomyelitis. Autoimmunity 2012;45:388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [207].Ramaglia V, Hughes TR, Donev RM, Ruseva MM, Wu X, Huitinga I et al. : C3-dependent mechanism of microglial priming relevant to multiple sclerosis. Proc Natl Acad Sci U S A 2012;109:965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [208].Ochoa-Reparaz J, Kirby TO, Kasper LH: The Gut Microbiome and Multiple Sclerosis. Cold Spring Harb Perspect Med 2018;8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [209].Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W: Interleukin-10-deficient mice develop chronic enterocolitis. Cell 1993;75:263. [DOI] [PubMed] [Google Scholar]
- [210].Mayer TG, Bhan AK, Winn HJ: Immunohistochemical analyses of skin graft rejection in mice. Kinetics of lymphocyte infiltration in grafts of limited immunogenetic disparity. Transplantation 1988;46:890. [DOI] [PubMed] [Google Scholar]
- [211].Jang HR, Gandolfo MT, Ko GJ, Satpute SR, Racusen L, Rabb H: B cells limit repair after ischemic acute kidney injury. J Am Soc Nephrol 2010;21:654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [212].Burne-Taney MJ, Yokota-Ikeda N, Rabb H: Effects of combined T- and B-cell deficiency on murine ischemia reperfusion injury. Am J Transplant 2005;5:1186. [DOI] [PubMed] [Google Scholar]
- [213].Jang HR, Rabb H: Immune cells in experimental acute kidney injury. Nat Rev Nephrol 2015;11:88. [DOI] [PubMed] [Google Scholar]
- [214].Panzer SE, Wilson NA, Verhoven BM, Xiang D, Rubinstein CD, Redfield RR et al. : Complete B Cell Deficiency Reduces Allograft Inflammation and Intragraft Macrophages in a Rat Kidney Transplant Model. Transplantation 2018;102:396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [215].Platt JL, Cascalho M: B Cells in Transplantation of Rat, Mouse, and Man. Transplantation 2018;102:357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [216].Roopenian DC, Davis AP, Christianson GJ, Mobraaten LE: The functional basis of minor histocompatibility loci. J Immunol 1993;151:4595. [PubMed] [Google Scholar]
- [217].Ehst BD, Ingulli E, Jenkins MK: Development of a novel transgenic mouse for the study of interactions between CD4 and CD8 T cells during graft rejection. Am J Transplant 2003;3:1355. [DOI] [PubMed] [Google Scholar]
- [218].Parker W, Saadi S, Lin SS, Holzknecht ZE, Bustos M, Platt JL: Transplantation of discordant xenografts: a challenge revisited. Immunology Today 1996;17:373. [DOI] [PubMed] [Google Scholar]
- [219].Saadi S, Takahashi T, Holzknecht RA, Platt JL: Pathways to acute humoral rejection. American Journal of Pathology 2004;164:1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [220].Colvin R, Smith R: Antibody-mediated organ-allograft rejection. Nature Reviews Immunology 2005;5:807. [DOI] [PubMed] [Google Scholar]
- [221].Baldwin WM 3rd, Valujskikh A, Fairchild RL: Antibody-mediated rejection: emergence of animal models to answer clinical questions. Am J Transplant 2010;10:1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [222].Cascalho M, Platt JL: The immunological barrier to xenotransplantation. Immunity 2001;14:437. [DOI] [PubMed] [Google Scholar]
- [223].Staun-Ram E, Miller A: Effector and regulatory B cells in Multiple Sclerosis. Clin Immunol 2017;184:11. [DOI] [PubMed] [Google Scholar]
- [224].Corry RJ, Winn HJ, Russell PS: Primarily vascularized allografts of hearts in mice. The role of H-2D, H-2K, and non-H-2 antigens in rejection. Transplantation 1973;16:343. [DOI] [PubMed] [Google Scholar]
- [225].Russell PS, Chase CM, Colvin RB, Plate JM: Kidney transplants in mice: an analysis of the immune status of mice bearing long-term, H-2 incompatible transplants. Journal of Experimental Medicine 1978;147:1449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [226].Burdick JF, Russell PS: Antigenic requirement for induced rejection of long-surviving murine heart transplants. J Immunol 1982;128:1551. [PubMed] [Google Scholar]
- [227].Russell PS, Chase CM, Colvin RB, Plate JM: Induced immune destruction of long-surviving, H-2 incompatible kidney transplants in mice. J Exp Med 1978;147:1469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [228].Mackaness GB, Lagrange PH: Restoration of cell-mediated immunity to animals blocked by a humoral response. J Exp Med 1974;140:865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [229].Neta R, Salvin SB: Specific suppression of delayed hypersensitivity: the possible presence of a suppressor B cell in the regulation of delayed hypersensitivity. J Immunol 1974;113:1716. [PubMed] [Google Scholar]
- [230].Mannon RB, Kopp JB, Ruiz P, Griffiths R, Bustos M, Platt JL et al. : Chronic rejection of mouse kidney allografts. Kidney International 1999;55:1935. [DOI] [PubMed] [Google Scholar]
- [231].Hamaguchi Y, Uchida J, Cain DW, Venturi GM, Poe JC, Haas KM et al. : The peritoneal cavity provides a protective niche for B1 and conventional B lymphocytes during anti-CD20 immunotherapy in mice. J Immunol 2005;174:4389. [DOI] [PubMed] [Google Scholar]
- [232].Platt JL: Accommodation: how you see it, how you don’t. American Journal of Transplantation 2011;11:2007. [DOI] [PubMed] [Google Scholar]
- [233].Jackson AM, Kraus ES, Orandi BJ, Segev DL, Montgomery RA, Zachary AA: A closer look at rituximab induction on HLA antibody rebound following HLA-incompatible kidney transplantation. Kidney Int 2015;87:409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [234].Hill P, Cross NB, Barnett AN, Palmer SC, Webster AC: Polyclonal and monoclonal antibodies for induction therapy in kidney transplant recipients. Cochrane Database Syst Rev 2017;1:CD004759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [235].Wan SS, Ying TD, Wyburn K, Roberts DM, Wyld M, Chadban SJ: The Treatment of Antibody-Mediated Rejection in Kidney Transplantation: An Updated Systematic Review and Meta-Analysis. Transplantation 2018;102:557. [DOI] [PubMed] [Google Scholar]
- [236].Webster AC, Wu S, Tallapragada K, Park MY, Chapman JR, Carr SJ: Polyclonal and monoclonal antibodies for treating acute rejection episodes in kidney transplant recipients. Cochrane Database Syst Rev 2017;7:CD004756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [237].van den Hoogen MW, Kamburova EG, Baas MC, Steenbergen EJ, Florquin S, HJ MK et al. : Rituximab as induction therapy after renal transplantation: a randomized, double-blind, placebo-controlled study of efficacy and safety. Am J Transplant 2015;15:407. [DOI] [PubMed] [Google Scholar]
- [238].Tyden G, Genberg H, Tollemar J, Ekberg H, Persson NH, Tufveson G et al. : A randomized, doubleblind, placebo-controlled, study of single-dose rituximab as induction in renal transplantation. Transplantation 2009;87:1325. [DOI] [PubMed] [Google Scholar]
- [239].Bao X, Moseman EA, Saito H, Petryniak B, Thiriot A, Hatakeyama S et al. : Endothelial heparan sulfate controls chemokine presentation in recruitment of lymphocytes and dendritic cells to lymph nodes. Immunity 2010;33:817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [240].Wrenshall LE: Role of the microenvironment in immune response to transplantation. Springer Seminars in Immunopathology 2003;25:199. [DOI] [PubMed] [Google Scholar]
- [241].Lokmic Z, Lammermann T, Sixt M, Cardell S, Hallmann R, Sorokin L: The extracellular matrix of the spleen as a potential organizer of immune cell compartments. Semin Immunol 2008;20:4. [DOI] [PubMed] [Google Scholar]
- [242].Papy-Garcia D, Albanese P: Heparan sulfate proteoglycans as key regulators of the mesenchymal niche of hematopoietic stem cells. Glycoconj J 2017;34:377. [DOI] [PubMed] [Google Scholar]
- [243].Salek-Ardakani S, Arrand JR, Shaw D, Mackett M: Heparin and heparan sulfate bind interleukin-10 and modulate its activity. Blood 2000;96:1879. [PubMed] [Google Scholar]
- [244].Yanaba K, Bouaziz JD, Haas KM, Poe JC, Fujimoto M, Tedder TF: A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell-dependent inflammatory responses. Immunity 2008;28:639. [DOI] [PubMed] [Google Scholar]