Abstract
Geraniol, an active constituent of rose and palmarosa essential oils, possesses several pharmacological properties, including antioxidant, antibacterial and antiulcer activity. Geraniol was therefore investigated for its antiulcer and anti-Helicobacter pylori activity in rats. Ulcers were induced by injecting acetic acid into the sub-serosal layer of the stomach followed by orogastric inoculation of H. pylori for 7 days. Geraniol (15 and 30 mg/kg), vehicle and a standard drug combination (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg) were administered twice daily for 14 days. All the parameters were measured at the end of treatment. The ulcer index was significantly (P < 0.05) lowered in geraniol and standard drug-treated rats as compared to the H. pylori control group (4.13 ± 0.43). Treatment with geraniol (30 mg/kg) significantly (P < 0.01) increased the gastric pH along with a reduction in total acidity and gastric juice volume. Geraniol significantly (P < 0.05) attenuated the myeloperoxidase activity and augmented the total glutathione level in gastric mucosa. The extent of damage in the stomach was measured using a histopathological score. The score in H. pylori control, geraniol (30 mg/kg) and standard drugs was 9, 3.5 and 2.0 respectively. In the rapid urease test, treatment with geraniol (30 mg/kg) and the standard drugs produced a 33% and 67% cure respectively from H. pylori infection. Further, the reduction in bacterial load in the gastric mucosa was confirmed using modified Giemsa staining. Geraniol was observed to exhibit significant antiulcer and anti-H. pylori activity in a rodent model.
Keywords: Antiulcer, Geraniol, Helicobacter pylori, Myeloperoxidase, Glutathione
Graphical abstract
Abbreviations
- ANOVA
Analysis of variance
- ATCC
American type culture collection
- b.i.d
“bis in die” (twice a day)
- CLO
Campylobacter-like organism
- DTNB
5, 5-dithiobis-2-nitrobenzoic acid
- E. coli
Escherichia coli
- GSH
Glutathione
- H&E
Haematoxylin and eosin
- H. pylori
Helicobacter pylori
- MHB
Mueller-Hinton broth
- MPO
Myeloperoxidase
- NSAIDs
Nonsteroidal anti-inflammatory drugs
- PPIs
Proton pump inhibitors
- PUD
Peptic ulcer disease
- ROS
Reactive oxygen species
- RUT
Rapid urease test
- S. aureus
Staphylococcus aureus
- SPSS
Statistical packages for Social Science
- TRPV
Transient receptor potential cation channel subfamily V
- UI
Ulcer index
1. Introduction
Peptic ulcer is a common gastrointestinal disease and is considered as a global health problem.1 The etiology of the disease is still not completely understood but it is generally accepted that an imbalance between aggressive (stress, pepsin, acid) and protective (such as bicarbonate, mucin, mucosal blood flow, prostaglandin) factors, leads to the development of gastric ulcer.2 NSAIDs, Helicobacter pylori (H. pylori) infection, and hereditary predisposition are some of the risk factors that increase the incidence of gastric ulcer.3,4 H. pylori infection, which is spread through faecal-oral and oral-oral routes, is one of the prevalent factors causing peptic ulcer disease (PUD). More than 90% of duodenal and 60% of gastric ulcer patients were diagnosed with H. pylori infection.5 Currently, drugs, including antacids, anticholinergics, proton pump inhibitors and H2-receptor antagonists are available for the treatment of gastric ulcers. However, they possess several adverse effects6 and are non-curative.7 Current standard first-line therapy for H. pylori-positive PUD consists of proton pump inhibitors (PPIs) combined with at least two antibiotics, these being clarithromycin and amoxicillin or metronidazole/tinidazole.8 Unfortunately, antibiotic resistance in H. pylori is increasing and approximately 10–20% of H. pylori infections persist despite antibiotic treatment.9 Meanwhile, H. pylori resistance rate for clarithromycin has increased significantly from 10% during 1996–1999 to 17.9% during 2005–2007,10 whereas H. pylori resistant towards amoxicillin was found to be 8.5% in 2016.11 Metronidazole shows the highest resistance rate, with around 40% in developed countries and >90% in developing countries.12 It has been challenging to find an antiulcer agent that could both address the multifactorial etiologies of gastric ulcers and cure the disease, rather than simply producing an anti-secretory action.13 Hence, the search for an effective anti-ulcer agent having multiple mechanisms of gastroprotective and antibacterial activity is essential to combat the disease.
Plant-derived essential oils are widely used as medicines, insecticides, and perfumes.14 Geraniol (3,7-dimethyl-2,6-octadien-1-ol) is an acyclic monoterpene alcohol found in the essential oils of lemongrass, rose, palmarosa, ginger, orange, lavender, citronella, nutmeg and other plants.15,16 It is commercially used for its fragrance and as a flavour in food industry.17 Geraniol exhibited good antimicrobial activity against a wide spectrum of bacteria and fungi18 and inhibited the growth of H. pylori with a MIC of 0.53 mg/L.19 We recently found a significant antibacterial activity of geraniol against S. aureus, E. coli and H. pylori with MIC values of 11.2 mg/ml, 5.6 mg/ml and 7.33 mg/ml respectively. (Unpublished data).
Besides its antibacterial activity, geraniol also exhibited anti-oxidant, anti-ulcer, anti-inflammatory and anti-apoptotic effects.20 Recent studies showed oral administration of geraniol to protect against gastric ulcers induced by either ethanol or by ischemia-reperfusion, and duodenal ulcers induced by cysteamine in rats.16 However, the effect of geraniol against H. pylori-induced ulceration has not been studied. The present study investigates geraniol for its putative antiulcer and anti-H. pylori activity in a rodent model.
2. Materials and methods
2.1. Drugs and chemicals
Geraniol (98%) was purchased from Sigma-Aldrich Chemical Co., USA. The anaesthetic agents (ketamine, xylazine and zoletil) were obtained from the animal house of University Kebangsaan Malaysia, Kuala Lumpur, Malaysia. Amoxicillin, clarithromycin, and omeprazole were procured from PI Chemicals, Shanghai, China. All the other chemicals and reagents used in the experiments were of analytical grade.
2.2. Microorganisms
H. pylori (ATCC 43504) was purchased from Choice Care, Kuala Lumpur, Malaysia. It was subcultured in Mueller-Hinton broth (MHB) supplemented with 10% fetal bovine serum (FBS) and incubated at 37 °C in an anaerobic jar containing microaerophilic gas generator pack (Campy Gen™ 2.5 L, Thermo Scientific, Oxoid Ltd, UK) for five days to obtain substantial growth of H. pylori.
2.3. Animal grouping
Adult male Sprague-Dawley rats (200–250 gm) were procured and maintained in the animal house facility of International Medical University, Bukit Jalil, Kuala Lumpur, Malaysia. The rats were housed randomly in polypropylene cages under standard condition of dark and light cycle (12/12 h), humidity (50–60%) and temperature (20–24 °C). During the acclimatisation and experimental period, animals had access to food and water ad libitum. The experimental protocol was presented to Institutional ethics committee of International Medical University, Kuala Lumpur, Malaysia. The research protocol was duly approved (BP I-01/12 (46) 2015) before initiation of the study. All the animals used in this study were received humane care and the experiments were performed according to the criteria outlined in the “Guide for the care and use of Laboratory Animals” prepared by the National Academy of Sciences and published by the National Institute of Health, USA.
The rats were divided into six groups with six animals in each group. The grouping was as follows. Group 1: normal control (saline, 5 ml/kg, b. i.d); Group 2: ulcer control (acetic acid) (corn oil, 5 ml/kg, b. i.d); Group 3: ulcer control with H. pylori (acetic acid and H. pylori) (corn oil, 5 ml/kg, b. i.d); Group 4: geraniol (15 mg/kg, b. i.d.); Group 5: geraniol (30 mg/kg, b. i.d) and Group 6: standard drugs (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg; omeprazole, 20 mg/kg).
2.4. Ulcer induction and treatment protocol
Rats were ulcerated with acetic acid according to the method described by Takagi et al. (1969).21 Under anaesthesia, laparotomy was performed through a midline epigastric incision; the stomach was exposed and 20% acetic acid (0.03 mL) was injected into the sub-serosal layer of the glandular portion, using a micro syringe (0.05 mL). After suturing the abdominal incision, the animals were returned to their home cages with daily access to food, restricted to the time periods of 9–10 a.m. and 5–6 p.m. This allows adequate fasting for the administration of H. pylori inoculum, geraniol and standard drugs.21
Twenty 4 h after acetic acid-induced ulceration, the animals were inoculated intragastrically by gavage with 1 mL of a confirmed pathogenic strain of H. pylori suspended in MHB22; this was undertaken twice daily for 7 days. Control rats and those receiving acetic acid without H. pylori infection received MHB orally for the same time period. . Geraniol, vehicle and standard drugs were administered twice daily for 14 consecutive days, starting from the third day after ulcer induction with acetic acid.22 After treatment, the animals were sacrificed by cervical dislocation. The stomach was removed and evaluated for gastric lesions.
2.5. Measurement of gastric ulcer index
The ulcerated area (mm2) and the healing rate (%) were determined according to the method of Takagi and Okabe (1968). The ulcerated area and diameter (mm2) was measured with a ruler and the ulcer score was calculated based on the severity of gastric ulcer lesion scoring system.23
| Ulcer score | Gastric Lesions |
|---|---|
| 0 | No lesion |
| 1 | Mucosal oedema and petechiae |
| 2 | One to five small lesions (1–2 mm), |
| 3 | More than five small lesions or one intermediate lesion (3–4 mm) |
| 4 | Two to more intermediate lesions or one gross lesion (>4 mm) |
| 5 | Perforated ulcers |
The ulcer index (UI) and percentage of curative ratio was calculated using the formula below:
| UI = total ulcer score/number of animals ulcerated |
| Curative ratio (%) = (UI control − UI treated)/UI control × 100 |
2.6. Measurement of gastric juice volume, pH and total acidity
Gastric content was secured by tying the pyloric and cardiac end of the stomach with a thread. A small incision was made in the greater curvature and the gastric content was collected into a centrifuge tube. Distilled water (5 mL) was added to the tube and centrifuged at 2000×g for 10 min. The pH of the supernatant was measured using a pH meter. Total acidity was determined by titrating with 0.01 N sodium hydroxide using phenolphthalein as an indicator and the results were expressed as mEq/L/100 g.24
2.7. Determination of myeloperoxidase (MPO)
The gastric tissue (100 mg) was homogenized using a small tissue homogeniser in 40 mM phosphate buffer at pH 7.4. The homogenates were centrifuged at 13,000×g for 10 min at 4 °C. MPO activity in the tissue homogenate was estimated as per the protocol described in the commercial MPO kit (BioVision Incorporated, USA). In brief, supernatants were allowed to react with 5, 5-dithiobis-2-nitrobenzoic acid (DTNB) at 25 °C for 120 min and the reaction was terminated by adding the stop mixture. The change in absorbance was measured at 412 nm using a spectrophotometer.
2.8. Determination of total glutathione (GSH)
Gastric tissue (100 mg) was washed with phosphate buffer solution (pH 7.4) and the size was reduced appropriately by chopping with scissors. The tissue was homogenized in ice-cold 5% metaphosphoric acid (MPA) (∼1 ml/100 mg tissue using a small tissue homogeniser). The homogenate was centrifuged at 10,000×g for 15 min at 4 °C. GSH level was measured as per the protocol described in the commercial GSH assay kit (Cell Biolabs Inc., San Diego, CA, USA).
2.9. Rapid urease test (RUT)
Gastric mucosa (approximately 2 mm2) from the antral area was removed and the presence of H. pylori was detected using the Campylobacter-like organism (CLO) - test kits (Kimberly-Clark, USA). The yellow urea gel turned into a bright magenta colour in the presence of H. pylori, with the production of ammonia (alkaline reaction).25
2.10. Histopathological evaluation
The collected stomach fragments were preserved in 10% buffered formalin. All the specimens were subjected to haematoxylin and eosin (H&E) and modified Giemsa staining. Giemsa-stained slides were examined by a pathologist for H. pylori detection and pathogen load in the gastric mucosa. H&E stained slides were observed and interpreted under a microscope (80i, Nikon, USA) for histopathological changes and graded according to the lesion scoring system by a pathologist who was blind to the treatment based on the criteria.24 The scorings were allotted as 1. Epithelial cell loss (Score: 0–3); 2. Oedema in the submucosa (Score: 0–4); 3. Haemorrhagic damage (Score: 0–4) and 4. Presence of inflammatory cells (Score: 0–3). The maximum total score was thus 14.
2.11. Statistical analysis
The results are expressed as mean ± standard error mean (S.E.M). Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Tukey-Kramer post-test. Histopathological data were analysed using non-parametric statistics using Kruskal-Wallis test followed by Mann-Whitney U test as post-hoc analysis between two groups. Percentage inhibition in CLO test was statistically compared using z-test. The value of P < 0.05 is considered statistically significant. All statistical analyses were done using the SPSS (Statistical packages for Social Science) version 16.0 Software.
3. Results
3.1. Gross evaluation
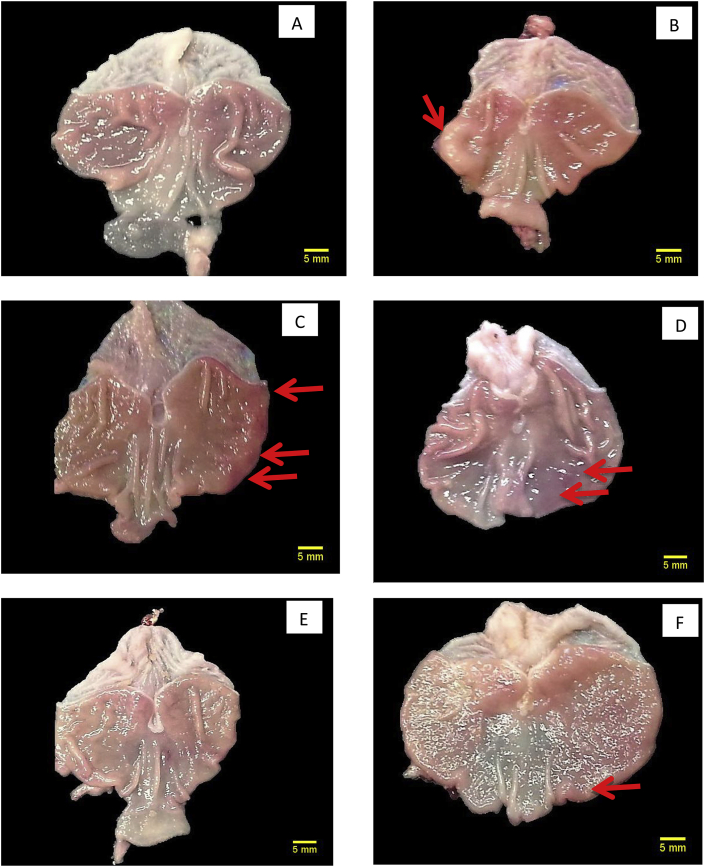
Treatment with geraniol or standard drugs considerably reduced the ulcer area and also the number of ulcer patches when compared to both ulcer control groups. The ulcer control without H. pylori showed large ulcers with haemorrhage and necrosis, whereas several ulcerogenic lesion areas (3–6 mm) with hyperemia and haemorrhagic streaks were observed in the ulcer control with H. pylori (Fig. 1). Treatment with geraniol and standard drugs resulted in fewer dilated blood vessels and haemorrhagic streaks on the mucosal surface (Fig. 1). Geraniol in both doses and the standard drugs produced a significant reduction in the UI compared with the ulcer control with H. pylori group (Table 1). Geraniol at doses of 15 and 30 mg/kg exhibited a percentage curative ratio of 42% and 52% respectively when compared to ulcer control with H. pylori, whereas the standard drugs treatment produced a maximum curative ratio of 64%.
Fig. 1.
Gross morphological observation of stomach in H. pylori and acetic acid-induced ulcer in rats (magnification = 1.3×).
A: Normal control group having intact stomach with normal morphology of the stomach; B: Ulcer control group without H. pylori showing extensive damage to gastric mucosa with sever ulceration and haemorrhage (red arrow) condition; C: Ulcer control group with H. pylori showed extensive mucosal damage and many ulcerated lesions with haemorrhage; D: Geraniol (15 mg/kg, b. i.d.) treated rats showed moderate ulcer lesions with less haemorrhage; E: Geraniol (30 mg/kg, b. i.d.) treated rats showed mild injuries and ulcer lesions without haemorrhage; F: Standard drug (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg, b. i.d.) treated rats showed mild injuries and ulcer lesions without haemorrhage.
Table 1.
Effect of geraniol on gastric juice volume, pH and total acidity in H.pylori and acetic acid - induced ulcer model.
| Groups | Gastric juice volume (mL) | pH | Total acidity (mEq/L/100 g) | Gastric ulcer index (UI) |
|---|---|---|---|---|
| NC | 1.63 ± 0.41 | 4.1 ± 0.3 | 105 ± 19.83 | 0.00 |
| UC | 2.43 ± 1.20 | 2.6 ± 0.28$ | 140 ± 15.06 | 2.80 ± 0.68$$ |
| UC with H. pylori | 6.38 ± 0.83$$ | 2.2 ± 0.06$$ | 315 ± 35.24$$ | 4.13 ± 0.43$$ |
| Ger_15 mg/kg | 3.12 ± 1.03* | 3.4 ± 0.26** | 134 ± 25.42* | 2.20 ± 0.34* |
| Ger_30 mg/kg | 1.79 ± 0.91** | 4.1 ± 0.26** | 124 ± 17.78** | 2.00 ± 0.55* |
| Standard Drugs | 3.50 ± 1.46 | 4.0 ± 0.44** | 130 ± 26.77** | 1.50 ± 0.29** |
Data are expressed as mean ± S.E.M; n = 6 per group; $ P < 0.05 vs normal control; $$ P < 0.01 vs normal control; *P < 0.05 vs ulcer control with H. pylori; **P < 0.01 vs ulcer control with H. pylori.
NC: normal control; UC: ulcer control without H. pylori; UC with H. pylori: Ulcer control with H. pylori; Ger_15 mg/kg: Geraniol (15 mg/kg); Ger_30 mg/kg: Geraniol (30 mg/kg); Standard Drugs: amoxicillin (50 mg/kg), clarithromycin (25 mg/kg) and omeprazole (20 mg/kg).
3.2. Gastric juice volume, pH and total acidity
Gastric juice volume was significantly (P < 0.01) increased in the ulcer control group with H. pylori as compared to the normal group. Treatment with geraniol (15 mg/kg, 30 mg/kg) but not with standard drugs, produced a significant reduction in gastric juice volume (P < 0.05) (Table 1). The pH of gastric juice in the ulcer control groups was significantly (P < 0.05) reduced in comparison to the normal control. Treatment with geraniol (15 mg/kg and 30 mg/kg) or the standard drugs produced a significant (P < 0.05) increase in pH compared with the ulcer with H. pylori control group. There was a marked increase in total acidity in the ulcer control group with H. pylori; this was significantly decreased by treatment with geraniol at both doses or with the standard drugs (P < 0.05) (Table 1).
3.3. Rapid urease test (RUT)
Geraniol treatment at 15 mg/kg and 30 mg/kg showed 17% and 33% reduction in H. pylori positive antral samples respectively, whereas treatment with the standard drugs showed a 67% (P < 0.05) reduction (Table 2).
Table 2.
Effect of geraniol pre-treatment on H. pylori infection in rat stomach using CLO-test.
| Groups | H. pylori Positive | H. pylori Negative | Percentage of inhibition (%) |
|---|---|---|---|
| UC with H. pylori | 6 (6) | 0 (6) | 0 |
| Ger_15 mg/kg | 5 (6) | 1 (6) | 17 |
| Ger_30 mg/kg | 4 (6) | 2 (6) | 33 |
| Standard Drugs | 2 (6) | 4 (6) | 67* |
Value in parenthesis indicates the total number of rat per group. *P < 0.05 vs ulcer control with H. pylori control group.
UC with H. pylori: Ulcer control with H. pylori; Ger_15 mg/kg: Geraniol (15 mg/kg); Ger_30 mg/kg: Geraniol (30 mg/kg); Standard Drugs: amoxicillin (50 mg/kg), clarithromycin (25 mg/kg) and omeprazole (20 mg/kg).
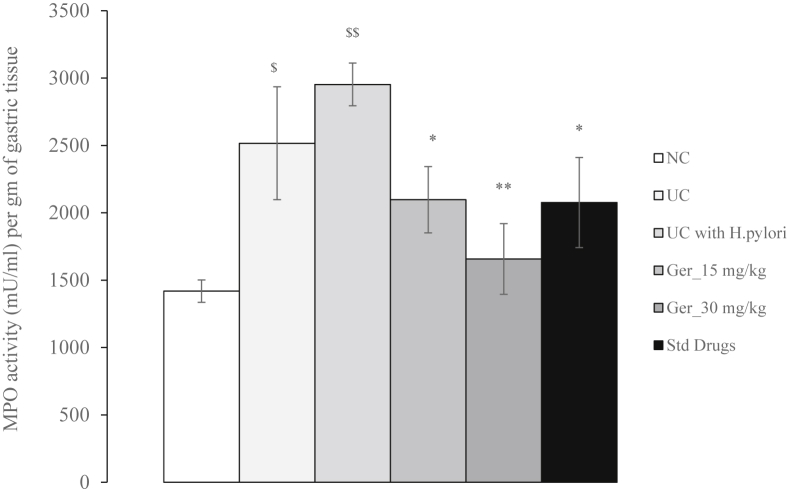
3.4. Determination of myeloperoxidase (MPO)
MPO activity was significantly (P < 0.05) increased in both ulcer control groups in comparison to normal controls. Geraniol (15 mg/kg and 30 mg/kg) significantly (P < 0.05 and P < 0.01 vs. ulcer control with H. pylori respectively) reduced the MPO levels in the stomach. Treatment with the standard drugs also reduced MPO levels (P < 0.05 vs. ulcer control with H. pylori (Fig. 2)).
Fig. 2.
Effect of geraniol on myeloperoxidase activity (mU/ml) per gm of gastric tissue of H. pylori and acetic acid-induced ulcerated rats.
$P < 0.05 vs normal control; $$P < 0.01 vs normal control; *P < 0.05 vs ulcer control with H. pylori; **P < 0.01 vs ulcer control with H. pylori.
NC: Normal control group; UC: Ulcer control group without H. pylori; UC with H. pylori: Ulcer control group with H. pylori; Ger_15 mg/kg: Geraniol (15 mg/kg, b. i.d.); Ger_30 mg/kg: Geraniol (30 mg/kg, b. i.d.); Std. Drugs: Standard Drug (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg, b. i.d).
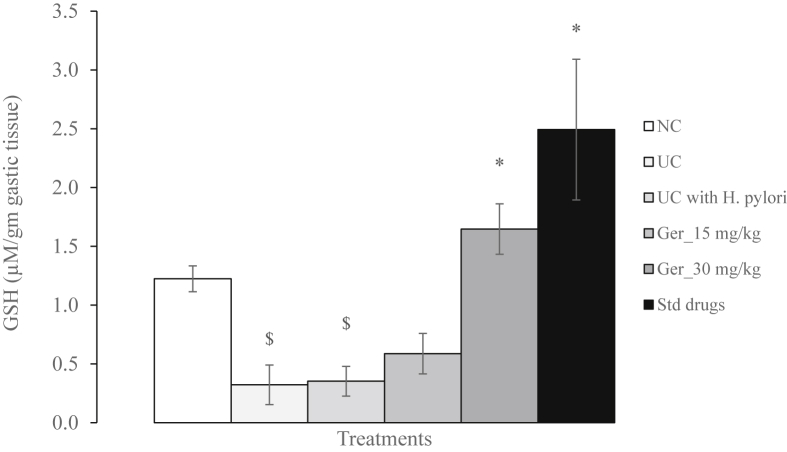
3.5. Determination of total glutathione (GSH)
The GSH content in stomach samples of both ulcer control groups was significantly (P < 0.05) reduced as compared to normal controls. Treatment with geraniol (30 mg/kg) or with the standard drugs significantly (P < 0.05) augmented the GSH content in the gastric tissue (Fig. 3).
Fig. 3.
Effect of geraniol on total glutathione (μM) per gm of gastric tissue in H. pylori and acetic acid- induced ulcerated rats.
$P < 0.05 vs normal control; *P < 0.05 vs ulcer control with H. pylori.
NC: Normal control group; UC: Ulcer control group without H. pylori; UC with H. pylori: Ulcer control group with H. pylori; Ger_15 mg/kg: Geraniol (15 mg/kg, b. i.d.); Ger_30 mg/kg: Geraniol (30 mg/kg, b. i.d.); Std. Drugs: Standard Drug (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg, b. i.d.).
3.6. Histopathological evaluation
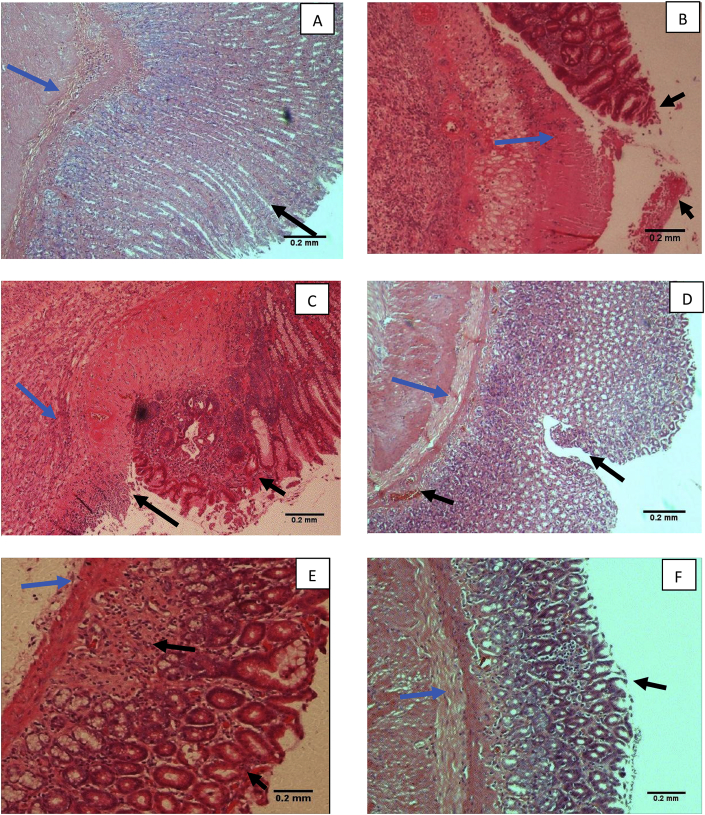
H&E staining of the gastric mucosa showed extensive lesions in both ulcer control, groups, with oedema in the sub-mucosal layer and leucocyte infiltration, along with haemorrhage and epithelial cell loss (Fig. 4; Table 3). Treatment with geraniol (30 mg/kg) or with the standard drugs significantly reduced inflammation, oedema, leucocyte infiltration and haemorrhagic damage in comparison to the ulcer control with H. pylori, with a significant reduction in lesion scores (Table 3).
Fig. 4.
Histological evaluation of gastric tissues using H&E staining (magnification = 100×).
A: Normal control group having an intact stomach. Black arrow indicates a normal morphology of the stomach with normal mucosal epithelial cells. Blue arrow indicates the normal sub-mucosal and muscular layer of the stomach; B: Ulcer control group without H. pylori. Black arrow indicates the extensive damage and loss of mucosal epithelial layer which is further extended to damage in the sub-mucosal layer. Blue arrow indicates the haemorrhages, inflammation and oedema of the sub-mucosal layer. C: Ulcer control group with H. pylori. Black arrow showed extensive damage and loss of mucosal epithelial layer. Blue arrow showed oedema, inflammation and haemorrhages in the sub-mucosal layer; D: Geraniol (15 mg/kg, b. i.d.). Black arrow showed moderate epithelial cell loss and haemorrhages. Blue arrow indicates the normal sub-mucosal layer; E: Geraniol (30 mg/kg, b. i.d.). Black arrow indicates a mild epithelial cell loss, oedema and neutrophil infiltration in mucosal layer. Blue arrow indicates the normal sub-mucosal layer; F: Standard Drugs (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg, b. i.d.). Black arrow indicates mild mucosal epithelial cell loss. Blue arrow showed the normal sub-mucosal layer.
Table 3.
Histological score of rat stomach pre-treated with geraniol in acetic acid and H. pylori induced ulcer.
| Groups | Hemorrhagic damage (score 0–4) | Oedema (score 0–4) | Epithelial cell loss (score 0–3) | Inflammatory cells (score 0–3) | Total (score 14) |
|---|---|---|---|---|---|
| NC | 0 | 0 | 0 | 0 | 0 |
| UC | 1.0$ | 1.0$ | 3.0$ | 2.0$ | 7.0$ |
| UC with H. pylori | 1.5$ | 1.5$ | 3.0$ | 3.0$ | 9.0$ |
| Ger_15 mg/kg | 0* | 1 | 1.5* | 3.0 | 5.5* |
| Ger_30 mg/kg | 0* | 1 | 0.5* | 2.0 | 3.5* |
| Standard Drugs | 0* | 1 | 0** | 1.0** | 2.0* |
n = 6 per group; $P < 0.05 vs normal control; *P < 0.05 vs ulcer control with H. pylori control group; **P < 0.01 vs ulcer control with H. pylori.
NC: normal control; UC: ulcer control without H. pylori; UC with H. pylori: Ulcer control with H. pylori; Ger_15 mg/kg: Geraniol (15 mg/kg); Ger_30 mg/kg: Geraniol (30 mg/kg); Standard Drugs: amoxicillin (50 mg/kg), clarithromycin (25 mg/kg) and omeprazole (20 mg/kg).
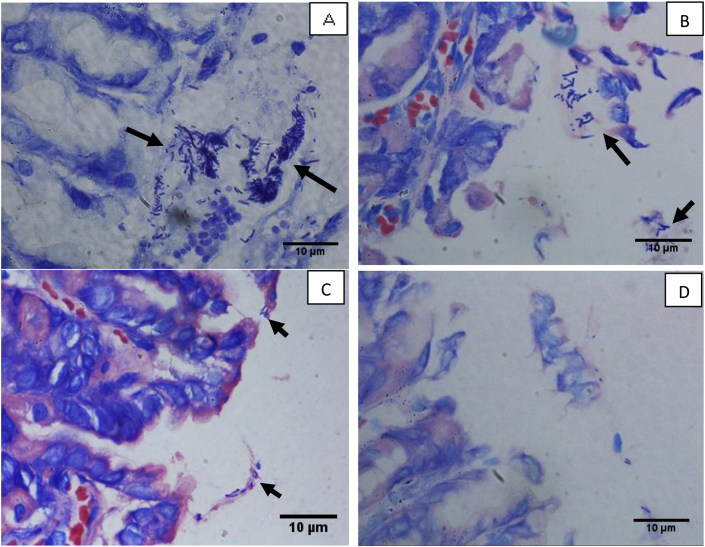
The presence of H. pylori in the gastric mucosa was verified using the modified Giemsa staining technique. Geraniol (15 and 30 mg/kg)-treated animals showed decreased H. pylori load in the gastric mucosa in comparison to ulcer control with H. pylori group. Standard drug therapy significantly reduced the pathogen load, with an absence of detectable H. pylori in the gastric mucosa (Fig. 5).
Fig. 5.
Histological evaluation of gastric tissues for H. pylori using Giemsa staining (magnification = 1000×).
A: Ulcer control group with H. pylori. Black arrow showed extensive accumulation of H. pylori cells in between gastric mucosa and mucus layer; B: Geraniol (15 mg/kg, b. i.d.) treated rats. Black arrow showed the presence of moderate number of H. pylori cells in between gastric mucosa and mucus layer; C: Geraniol (30 mg/kg, b. i.d.) treated rats. Black arrow showed fewer H. pylori cells in between gastric mucosa and mucus layer; D: Standard Drug (amoxicillin, 50 mg/kg; clarithromycin, 25 mg/kg and omeprazole, 20 mg/kg, b. i.d.) treated rats. Absence of H. pylori cells in between gastric mucosa and mucus layer was observed.
4. Discussion
H. pylori is considered as the major causative factors in the pathogenesis of peptic ulcer disease in association with predisposing factors.26 While the anti-ulcerogenic action of geraniol has been demonstrated previously in a rat model in which peptic ulceration was induced by ethanol, ischemia-reperfusion or cysteamine,16 the present study is the first investigation of the protective action of this agent using a model in which gastric ulceration was induced by a combination of acetic acid and H. pylori infection. Although it is not easy to completely mimic the basic characteristics of human H. pylori infections (an intense active chronic gastritis, either antral or diffuse), and the associated complications (mucosa atrophy and intestinal metaplasia) and pathologies (peptic ulcer, gastric adenocarcinoma and lymphoma), this is a well-validated model.27 Ulceration using acetic acid spontaneously healed. However, the introduction of H pylori, as used in the present study, leads to significant delay in ulcer healing and results in the establishment of chronic ulceration.28 Inflammation, increased apoptosis, overexpression of inflammatory markers and reduced gastric microcirculation are responsible for delayed ulcer healing.28 Further, MPO activity in the ulcerated tissue are much higher than normal gastric tissue.29 The characteristics of these ulcers are quite similar to the human peptic ulcers in regard with its pathological characteristics and healing process.30 Previous results showed that rats31 or mice22 with pre-existing gastric ulcers induced by acetic acid injection, developed active ulcers when exposed to H. pylori. In the present study geraniol clearly showed a significant reduction in ulceration, as evidenced by the reduction in ulcer index, and the improvement in the gross morphology of the mucosa, including reduced leucocyte infiltration and the absence of oedema and epithelial cell loss. The reduced inflammatory response was supported by the marked reduction in the ulcer-induced elevation in MPO seen in geraniol treated rats. MPO is an important indicator of inflammation seen in ulcer lesions and relates to extensive neutrophil infiltration/aggregation in gastric tissue.32, 33, 34 It was previously reported that H. pylori administration in Mongolian gerbil showed a significant increased MPO activity in gastric mucosa.35 Drugs those attenuate the release of proinflammatory cytokines, neutrophil adhesion/infiltration and inflammation have shown a significant effect against H. pylori infected gastric cell damage.35,36
Reactive oxygen species (ROS) are essential for defence in response to certain external stimuli.37 ROS are normally neutralised immediately by endogenous antioxidants such as GSH and superoxide dismutase. Oxidative stress develops when cellular antioxidant levels are insufficient to counter the excess ROS,38 resulting in lipid peroxidation, cellular death and tissue damage.39 The role of ROS in the etiology and pathophysiology of human gastric ulcer development is well established.40 ROS production and oxidative stress remain a major cause for the induction and aggravation of an ulcer.41 Total GSH is an essential component of the endogenous defence system; it scavenges toxic oxygen free radicals and peroxides and low GSH levels in the gastric mucosa facilitate gastric tissue damage.42 A significant reduction in total GSH was seen in both ulcer controls, and the higher dose of geraniol, as well as the standard drugs, significantly increased total GSH levels. The increased level of GSH after geraniol treatment may be a factor in its gastro-protective effects. Our observations concur with the previous findings which showed that the antiulcer effect of geraniol in an acute ulcer model was associated with increased GSH and decreased MPO activity, as well as stimulation of the nitric oxide pathway, increased prostaglandin levels, and increased mucus production.16 In the present study, the biochemical changes in gastric mucosa due to geraniol were further corroborated by the histopathological evaluation of gastric tissues. The observations from gross morphology, histopathology and biochemical estimations strongly suggest the anti-ulcer potential of geraniol in a chronic ulcer model. Oxygenated monoterpenes like geraniol, carvacrol, α-terpineol, isopulegol have shown potent anti-ulcer activity against acute ulcer models induced by many agents.16,43, 44, 45
H. pylori infection leads to an alteration of the motor or sensory function of the stomach, causing a delayed stomach emptying and increased gastric juice volume.46 We found geraniol to significantly reduce the volume of gastric juice, as well as to lower the total acidity in the stomach; these effects may indicate a primary anti-secretory effect or that the secretion was reduced secondary to ulcer healing. de Carvalho et al. suggested that the antiulcer activity of geraniol in their acute ulcer model was not related to its anti-secretory action.16 The reduced gastric acid secretion and gastric juice volume produced by geraniol in the present study may be secondary to ulcer healing, an anti-H. pylori effect or an effect of geraniol on autoregulation of gastrin release.
H. pylori colonise and move chemotactically towards the gastric mucosa layer, even in the acidic gastric environment, by using urease to convert urea into ammonia.47 The ammonia neutralises the acid around the bacteria, thus allowing their survival in an acidic environment.48 Ammonia also damages the gastric epithelial cell, as well as reducing the thickness of the mucus gel layer, resulting in chronic colonisation of H. pylori and ulcer formation.47,49 Detection of urease using the RUT is a minimally invasive test with 100% sensitivity and 89.5% specificity for H. pylori.50 However, false positive results can be obtained due to interference by other bacterial species like Klebsiella pneumonia, Enterobacter cloacae, Citrobacter freundii, and Proteus mirabilis that also produce urease.51 We found both geraniol and the standard drugs to reduce urease production, suggesting a reduction in H. pylori, although the standard drug treatment was the most effective, statistical significance being achieved only with this treatment. However, the urease data were supported by the histopathological findings, with histopathology having 97% specificity and 98% sensitivity.50,52 The ulcer control rats showed a significant pathogen load and colonies in between gastric mucosa and mucus layer of the stomach. The pathogen load in the gastric mucosal epithelium was reduced by geraniol as well as by the standard drugs.
The combination therapy including two antibiotics and an acid suppressant is effective against the eradication of H. pylori and healing of gastritis, gastric/duodenal ulcers. Amoxicillin inhibits the synthesis of glycopeptides of the bacterial cell wall. Whereas, Clarithromycin inhibits the bacterial protein synthesis. The MIC value of amoxicillin and clarithromycin is decreases in higher gastric pH. Proton pump inhibitors like pantoprazole which increase the gastric pH are co-administered to augment the antibacterial effects of amoxicillin and clarithromycin as well as improve the ulcer condition.53 Geraniol has attenuated the gastric acid secretion and increase the gastric pH in this study. In another study, we have observed anti-H. pylori effect of geraniol.54 Thus, geraniol possesses both anti-ulcer and anti-H. pylori effect which is complementing each other for delivering a better efficacy. Thus our findings suggest a considerable in vivo anti-H. pylori activity, supporting the in vitro findings by our group and other researchers.19
5. Conclusion
The antiulcer effect and anti-H. pylori action of geraniol in the present study was associated with increased GSH levels and decreased MPO activity, along with reduced gastric secretion. Thus, geraniol has the potential to be developed as a treatment for H. pylori associated PUD. The mechanism(s) underlying the anti-ulcer effect remains to be established but may involve direct gastro-protective effects due to free radical scavenging, as well as an antibacterial action against H. pylori.
Conflicts of interest
All authors declare no conflict of interest to disclose.
Acknowledgements
This work was supported by the International Medical University, Kuala Lumpur, Malaysia [Grant no. BP I-01/12 (46) 2015]. Authors are also grateful to Dr Barani Karikalan, Department of Pathology, School of Medicine, International Medical University for the histopathological slide observation and grading of stomach samples. Authors are thankful to Prof Brian Furman, University of Strathclyde, Glasgow, Scotland for thoroughly reviewing the manuscript and improving the manuscript quality.
Footnotes
Peer review under responsibility of The Center for Food and Biomolecules, National Taiwan University.
References
- 1.Klopell F.C., Lemos M., Sousa J.P.B. Nerolidol, an antiulcer constituent from the essential oil of Baccharis dracunculifolia DC (Asteraceae) Z Naturforsch C Biosci. 2007;62(7–8):537–542. doi: 10.1515/znc-2007-7-812. [DOI] [PubMed] [Google Scholar]
- 2.Lima Z.P., Severi J.A., Pellizzon C.H. Can the aqueous decoction of mango flowers be used as antiulcer agent? J Ethnopharmacol. 2006;106:29–37. doi: 10.1016/j.jep.2005.11.032. [DOI] [PubMed] [Google Scholar]
- 3.Barros M.P., Lemos M., Maistro E.L. Evaluation of antiulcer activity of the main phenolic acids found in Brazilian Green Propolis. J Ethnopharmacol. 2008;120:372–377. doi: 10.1016/j.jep.2008.09.015. [DOI] [PubMed] [Google Scholar]
- 4.Belaiche J., Burette A., De Vos M., Louis E., Huybrechts M., Deltenre M. Observational survey of NSAID-related upper gastro-intestinal adverse events in Belgium. Acta Gastroenterol Belg. 2002;65:65–73. [PubMed] [Google Scholar]
- 5.Ej K., Jc T., Hp F. The prevalence of Helicobacter pylori in peptic ulcer disease. Aliment Pharmacol Ther. 1994;9(Suppl. 2):59–69. [PubMed] [Google Scholar]
- 6.Santin J.R., Lemos M., Junior L.C.K., Niero R., Andrade S.F. Antiulcer effects of Achyrocline satureoides (Lam.) DC (Asteraceae) (Marcela), a folk medicine plant, in different experimental models. J Ethnopharmacol. 2010;130:334–339. doi: 10.1016/j.jep.2010.05.014. [DOI] [PubMed] [Google Scholar]
- 7.Chung C.S., Chiang T.H., Lee Y.C. A systematic approach for the diagnosis and treatment of idiopathic peptic ulcers. Korean J Intern Med (Engl Ed) 2015;30(5):559–570. doi: 10.3904/kjim.2015.30.5.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marcus E.A., Sachs G., Scott D.R. Eradication of Helicobacter pylori infection. Curr Gastroenterol Rep. 2016;18(7):33. doi: 10.1007/s11894-016-0509-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Coticchia J.M., Sugawa C., Tran V.R., Gurrola J., Kowalski E., Carron M.A. Presence and density of Helicobacter pylori biofilms in human gastric mucosa in patients with peptic ulcer disease. J Gastrointest Surg. 2006;10(6):883–889. doi: 10.1016/j.gassur.2005.12.009. [DOI] [PubMed] [Google Scholar]
- 10.Boyanova L., Gergova G., Nikolov R. Prevalence and evolution of Helicobacter pylori resistance to 6 antibacterial agents over 12 years and correlation between susceptibility testing methods. Diagn Microbiol Infect Dis. 2008;60(4):409–415. doi: 10.1016/j.diagmicrobio.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 11.Alarcón T., Urruzuno P., Martínez M.J. Antimicrobial susceptibility of 6 antimicrobial agents in Helicobacter pylori clinical isolates by using EUCAST breakpoints compared with previously used breakpoints. Enfermedades Infecc Microbiol Clínica. 2017;35(5):278–282. doi: 10.1016/j.eimc.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 12.Obonyo M., Zhang L., Thamphiwatana S., Pornpattananangkul D., Fu V., Zhang L. Antibacterial activities of liposomal linolenic acids against antibiotic-resistant Helicobacter pylori. Mol Pharm. 2012;9(9):2677–2685. doi: 10.1021/mp300243w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garner A. Future opportunities for drug therapy in peptic ulcer disease. Scand J Gastroenterol. 1986;125:203–210. doi: 10.3109/00365528609093838. [DOI] [PubMed] [Google Scholar]
- 14.Bakkali F., Averbeck S., Averbeck D., Idaomar M. Biological effects of essential oils - a review. Food Chem Toxicol. 2008;46(2):446–475. doi: 10.1016/j.fct.2007.09.106. [DOI] [PubMed] [Google Scholar]
- 15.Jeon J.H., Lee C.H., Lee H.S. Food protective effect of geraniol and its congeners against stored food mites. J Food Protect. 2009;72(7):1468–1471. doi: 10.4315/0362-028x-72.7.1468. [DOI] [PubMed] [Google Scholar]
- 16.de Carvalho K.I., Bonamin F., Dos Santos R.C. Geraniol- a flavoring agent with multifunctional effects in protecting the gastric and duodenal mucosa. Naunyn-Schmiedeberg's Arch Pharmacol. 2014;387(4):355–365. doi: 10.1007/s00210-013-0947-z. [DOI] [PubMed] [Google Scholar]
- 17.Sawada S., Okano J., Imamoto R. Preventive Effect of geraniol on diethylnitrosamine-induced hepatocarcinogenesis in rats. Yonago Acta Med. 2016;59(1):37–43. [PMC free article] [PubMed] [Google Scholar]
- 18.Arputha M., Selvamani P., Latha S. In-Vitro antimicrobial evaluation of extracts, oil and fractionated geraniol of Cymbopogan citratus-an aromatic grass. Int J Environ Sci. 2012;3(1):583–590. [Google Scholar]
- 19.Boyanova L., Neshev G. Inhibitory effect of rose oil products on Helicobacter pylori growth in vitro: preliminary report. J Med Microbiol. 1999;48(7):705–706. doi: 10.1099/00222615-48-7-705. [DOI] [PubMed] [Google Scholar]
- 20.Vinothkumar V., Manoharan S., Sindhu G., Nirmal M.R., Vetrichelvi V. Geraniol modulates cell proliferation, apoptosis, inflammation, and angiogenesis during 7, 12-dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Mol Cell Biochem. 2012;369:17–25. doi: 10.1007/s11010-012-1364-1. [DOI] [PubMed] [Google Scholar]
- 21.Takagi K., Okabe S., Saziki R. A new method for the production of chronic gastric ulcer in rats and the effect of several drugs on its healing. Jpn J Pharmacol. 1969;19:418–426. doi: 10.1254/jjp.19.418. [DOI] [PubMed] [Google Scholar]
- 22.Konturek P.C., Brzozowski T., Konturek S.J. Mouse model of Helicobacter pylori infection: studies of gastric function and ulcer healing. Aliment Pharmacol Ther. 1999;13:333–346. doi: 10.1046/j.1365-2036.1999.00476.x. [DOI] [PubMed] [Google Scholar]
- 23.Takagi K., Okabe S. The effects of drugs on the production and recovery processes of the stress ulcer. Jpn J Pharmacol. 1968;18(1):9–11. doi: 10.1254/jjp.18.9. [DOI] [PubMed] [Google Scholar]
- 24.Sidahmed H.M.A., Hashim N.M., Abdulla M.A. Antisecretory, gastroprotective, antioxidant and anti-Helicobcter pylori activity of zerumbone from Zingiber zerumbet (L.) Smith. PLoS One. 2015;10(3):1–21. doi: 10.1371/journal.pone.0121060. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 25.Souza C. In vitro and in vivo anti-Helicobacter pylori activity of natural products. In: Chai J., editor. Peptic Ulcer Disease. InTech; Shanghai, China: 2011. pp. 427–436. [Google Scholar]
- 26.Atherton J.C. The pathogenesis of Helicobacter pylori–induced gastro-duodenal diseases. Annu Rev Pathol. 2006;1(1):63–96. doi: 10.1146/annurev.pathol.1.110304.100125. [DOI] [PubMed] [Google Scholar]
- 27.Bui H.X., del Rosario A., Sonbati H., Lee C.Y., George M., Ross J.S. Helicobacter pylori affects the quality of experimental gastric ulcer in a new animal model. Exp Mol Pathol. 1999;55:261–268. doi: 10.1016/0014-4800(91)90006-j. [DOI] [PubMed] [Google Scholar]
- 28.Li H., Kalies I., Mellgard B., Helander H.F. A rat model of chronic Helicobacter pylori infection. Studies of epithelial cell turnover and gastric ulcer healing. Scand J Gastroenterol. 1998;33:370–378. doi: 10.1080/00365529850170991. [DOI] [PubMed] [Google Scholar]
- 29.Keto Y., Ebata M., Tomita K., Okabe S. Influence of Helicobacter pylori infection on healing and relapse of acetic acid ulcers in Mongolian gerbils. Dig Dis Sci. 2002;47(4):837–849. doi: 10.1023/a:1014760504955. [DOI] [PubMed] [Google Scholar]
- 30.Okabe S., Pfeiffer C.J. Chronicity of acetic acid ulcer in the rat stomach. Am J Dig Dis. 1972;14:619–629. doi: 10.1007/BF02231748. [DOI] [PubMed] [Google Scholar]
- 31.Souza M.C., Beserra A.M., Martins D.C. In vitro and in vivo anti-Helicobacter pylori activity of Calophyllum brasiliense Camb. J Ethnopharmacol. 2009;123(3):452–458. doi: 10.1016/j.jep.2009.03.030. [DOI] [PubMed] [Google Scholar]
- 32.Naito Y., Yoshikawa T., Matsuyama K. Neutrophils, lipid peroxidation, and nitric oxide in gastric reperfusion injury in rats. Free Radic Biol Med. 1998;24:494–502. doi: 10.1016/s0891-5849(97)00323-7. [DOI] [PubMed] [Google Scholar]
- 33.Geetha A., Saranya P., Selvamathy S. A biochemical study on the gastroprotective effect of andrographolide in rats induced with gastric ulcer. Indian J Pharmaceut Sci. 2011;73(5):550. doi: 10.4103/0250-474X.99012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guha P., Dey A., Chatterjee A., Chattopadhyay S., Bandyopadhyay S. Pro-ulcer effects of resveratrol in mice with indomethacin-induced gastric ulcers are reversed by L-arginine. Br J Pharmacol. 2010;159(3):726–734. doi: 10.1111/j.1476-5381.2009.00572.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Takahashi S., Keto Y., Fujita T., Uchiyama T., Yamamoto A. FR167653, a p38 mitogen-activated protein kinase inhibitor, prevents Helicobacter pylori-induced gastritis in Mongolian gerbils. J Pharmacol Exp Therapeut. 2001;296(1):48–56. [PubMed] [Google Scholar]
- 36.Kim C.D., Kim H.H., Hong K.W. Inhibitory effect of rebamipide on the neutrophil adherence stimulated by conditioned media from Helicobacter pylori-infected gastric epithelial cells. J Pharmacol Exp Therapeut. 1999;288(1):133–138. [PubMed] [Google Scholar]
- 37.Aruoma O.I., Hayashi Y., Marotta F., Mantello P., Rachmilewitz E., Montagnier L. Applications and bioefficacy of the functional food supplement fermented papaya preparation. Toxicology. 2010;278(1):6–16. doi: 10.1016/j.tox.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 38.Sasaki M., Joh T. Oxidative stress and ischemia-reperfusion injury in gastrointestinal tract and antioxidant, protective agents. J Clin Biochem Nutr. 2007;40(1):1. doi: 10.3164/jcbn.40.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Adibhatla R.M., Hatcher J.F. Lipid oxidation and peroxidation in CNS health and disease: from molecular mechanisms to therapeutic opportunities. Antioxidants Redox Signal. 2010;12(1):125–169. doi: 10.1089/ars.2009.2668. [DOI] [PubMed] [Google Scholar]
- 40.Repetto M.G., Llesuy S.F. Antioxidant properties of natural compounds used in popular medicine for gastric ulcers. Braz J Med Biol Res. 2002;35:523–534. doi: 10.1590/s0100-879x2002000500003. [DOI] [PubMed] [Google Scholar]
- 41.Jainu M., Devi C.S. Antiulcerogenic and ulcer healing effects of Solanum nigrum (L.) on experimental ulcer models: possible mechanism for the inhibition of acid formation. J Ethnopharmacol. 2006;104(1–2):156–163. doi: 10.1016/j.jep.2005.08.064. [DOI] [PubMed] [Google Scholar]
- 42.La Casa C., Villegas I., Alarcón de la Lastra C., Motilva V., Martín Calero M.J. Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol induced gastric lesions. J Ethnopharmacol. 2000;71(1–2):45–53. doi: 10.1016/s0378-8741(99)00174-9. [DOI] [PubMed] [Google Scholar]
- 43.Oliveira I.S., da Silva F.V., Viana A.F. Gastroprotective activity of carvacrol on experimentally induced gastric lesions in rodents. Naunyn-Schmiedeberg's Arch Pharmacol. 2012;385:899–908. doi: 10.1007/s00210-012-0771-x. [DOI] [PubMed] [Google Scholar]
- 44.Souza R., Cardoso M., Menezes C., Silva J., De Sousa D., Batista J. Gastroprotective activity of α-terpineol in two experimental models of gastric ulcer in rats. Daru. 2011;19:277–281. [PMC free article] [PubMed] [Google Scholar]
- 45.Silva M.I.G., Moura B.A., Neto M.R.A. Gastroprotective activity of isopulegol on experimentally induced gastric lesions in mice: investigation of possible mechanisms of action. Naunyn-Schmiedeberg's Arch Pharmacol. 2009;380:233–245. doi: 10.1007/s00210-009-0429-5. [DOI] [PubMed] [Google Scholar]
- 46.Saslow S.B., Thumshirn M., Camilleri M. Influence of H. pylori infection on gastric motor and sensory function in asymptomatic volunteers. Dig Dis Sci. 1998;43(2):258–264. doi: 10.1023/a:1018833701109. [DOI] [PubMed] [Google Scholar]
- 47.Kusters J.G., Vliet A.H.M. van, Kuipers E.J. Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev. 2006;19(3):449–490. doi: 10.1128/CMR.00054-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ladeira M.S.P., Salvadori D.M.F., Rodrigues M.A.M. Biopatologia do Helicobacter pylori. J Bras Patol Med Lab. 2003;39(4):335–342. [Google Scholar]
- 49.Wang Y.C. Medicinal plant activity on Helicobacter pylori related diseases. World J Gastroenterol. 2014;20(30):10368. doi: 10.3748/wjg.v20.i30.10368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ogata S.K., Kawakami E., Reis F.P.S. Evaluation of invasive methods to diagnosis Helicobacter pylori infection in children and adolescents with dyspepsia invasive methods to diagnose Hp infection. Medicina, Ribeirão Preto. 2002;35:24–29. [Google Scholar]
- 51.Osaki T., Mabe K., Hanawa T., Kamiya S. Urease-positive bacteria in the stomach induce a false-positive reaction in a urea breath test for diagnosis of Helicobacter pylori infection. J Med Microbiol. 2008;57(Pt 7):814–919. doi: 10.1099/jmm.0.47768-0. [DOI] [PubMed] [Google Scholar]
- 52.Lin S.Y., Jeng Y.S., Wang C.K. Polymerase chain reaction diagnosis of Helicobacter pylori in gastroduodenal diseases: comparison with culture and histopathological examinations. J Gastroenterol Hepatol. 1996;11(3):286–289. doi: 10.1111/j.1440-1746.1996.tb00077.x. [DOI] [PubMed] [Google Scholar]
- 53.Labenz J. Current role of acid suppressants in Helicobacter pylori eradication therapy. Best Pract Res Clin Gastroenterol. 2001;15(3):413–431. doi: 10.1053/bega.2001.0188. [DOI] [PubMed] [Google Scholar]
- 54.Bhattamisra S.K., Hui Kuean C., Boon Chieh L. Antibacterial activity of geraniol in combination with standard antibiotics against Staphylococcus aureus, Escherichia coli and Helicobacter pylori. Nat Prod Commun. 2018;13(7):791–793. [Google Scholar]