Abstract
Background:
Current treatment guidelines for male breast cancer are predominantly guided by female-only clinical trials. With scarce research, it is unclear if breast conserving therapy (BCT) is equivalent to mastectomy in men. We sought to compare overall survival (OS) among male breast cancer patients who underwent BCT versus mastectomy.
Methods:
We performed a retrospective analysis of 8,445 stage I-II (T1–2 N0–1 M0) male breast cancer patients from the National Cancer Database (2004–2014). Patients were grouped according to surgical and radiation therapy (RT). BCT was defined as partial mastectomy followed by RT. Multivariable and inverse probability of treatment weighted (IPTW) Cox proportional hazards models were used to compare OS between treatment groups, controlling for demographic and clinicopathologic characteristics.
Results:
Most patients underwent total mastectomy (61.2%), while 18.2% underwent BCT, 12.4% underwent total mastectomy with RT, and 8.2% underwent partial mastectomy alone. In multivariable and IPTW models, partial mastectomy alone, total mastectomy alone, and total mastectomy with RT were associated with worse OS compared to BCT (p<0.001 all). Ten-year OS was 73.8% for BCT, while 56.3%, 58.0% & 56.3% for other treatment approaches. Older age, higher T/N stage, histological grade, and triple negative receptor status were associated with poorer OS (p<0.05). Subgroup analysis by stage demonstrated similar results.
Conclusion:
In this national sample of male breast cancer patients, BCT was associated with greater survival. The underlying mechanisms of this association warrant further study, since more routine adoption of BCT in male breast cancer appears to translate into clinically meaningful improvements in survival.
Introduction
With approximately 2,400 estimated new cases in the US in 2017, male breast cancer is a rare disease comprising less than 1% of all new breast cancer diagnoses.1 As such, research investigating optimal local regional treatment for male breast cancer is sparse and current treatment guidelines for men are predominantly based on clinical trials comprised of female participants.2 For example, the landmark National Surgical Adjuvant Breast and Bowel Project (NSABP) B-06 study compared total mastectomy to breast conserving therapy (BCT, i.e. partial mastectomy followed by radiation therapy (RT)) only in women. This study still strongly influences breast cancer treatment recommendations due to the finding of equivalent overall survival (OS) for BCT and mastectomy.3 Although this recommendation is applied to men, high-level evidence supporting this recommendation is lacking. Moreover, the few retrospective studies investigating male breast cancer have largely consisted of small cohorts and/or have been descriptive in nature.4–12 One exception, Cloyd et al., performed a retrospective analysis comparing partial to total mastectomy in male breast cancer patients using the Surveillance, Epidemiology and End Results (SEER) database; however, RT, an important factor for local recurrence and survival, was not used to categorize patients and was reported at low rates, potentially secondary to underreporting.13,14
Such limitations in male breast cancer research are significant, as they impair treating clinicians’ ability to provide evidence-based recommendations to male breast cancer patients. Although the National Comprehensive Cancer Network guidelines recommend that men be treated similar to postmenopausal women,2 a multidisciplinary expert panel has advised that male breast cancer is distinct from female breast cancer with significant biologic, clinicopathologic, and prognostic differences and should be treated accordingly.15–18 For example, a recent population-based study found significant differences in tumor gene expression in male breast cancer patients compared to female patients.18 This is problematic since we are currently using female data to make assumptions about male patients. Thus, there is a need for greater research investigating the impact of different therapeutic modalities on outcomes in male breast cancer patients to optimize therapy and patient counseling. The purpose of this study was to compare OS among male breast cancer patients treated with BCT, partial mastectomy alone, and total mastectomy with and without RT using a large national database, the National Cancer Database (NCDB). We hypothesized that BCT will be equivalent to total mastectomy with and without RT, while partial mastectomy alone will be associated with worse OS.
Methods
We performed a retrospective analysis of male breast cancer patients using the NCDB from 2004–2014. The NCDB is a joint American College of Surgeons and American Cancer Society project, in which patient-level data has been collected from all cancer patients seen at Commission on Cancer (COC) sites.19,20 Although approximately 30% of US hospitals are COC sites, NCDB captures data from 70% of all newly diagnosed cancer patients in the US.
We identified 18,984 male patients with invasive breast cancer, not including sarcomas and lymphomas. We excluded patients with stage III-IIV/unknown, T3-T4, multicentric/diffuse, or Paget’s disease to replicate the NSABP B-06 cohort clinicopathologic characteristics (Supplement, Figure S1). RT was defined as postoperative external beam RT to the breast and/or chest wall +/− axilla. Patients with unknown RT including sequence/type/location; who underwent brachytherapy; or preoperative RT were excluded. Patients were excluded if survival data was missing/unknown or if they did not undergo surgery with partial or total mastectomy. The final cohort consisted of 8,445 patients, categorized into the following groups based on surgery and RT: 1,539 BCT patients (partial mastectomy with RT), 695 partial mastectomy alone patients, 5,165 total mastectomy alone patients, and 1,046 total mastectomy with RT patients. As patient information was de-identified, the study protocol was exempt from the University of California, Davis Institutional Review Board approval.
We abstracted patient demographic, clinicopathologic characteristic, and treatment information from NCDB. The Charlson-Deyo comorbidity index (CDCI) measured patient medical comorbidities. Tumors were classified by histology based on international classification of disease oncology codes (ICD-O-3; Supplement). Staging was defined by AJCC TNM pathologic stage and only by clinical stage if pathologic staging data were missing (n=581, 6.9%). Immunotherapy was categorized as ‘chemotherapy’ since Trastuzumab was classified as chemotherapy until 2013. As NCDB reports the number of lymph nodes examined and not the type of nodal surgery performed, we defined sentinel lymph node biopsy (SLNB) as examining 1–5 nodes and axillary lymph node dissection (ALND) as examining ≥6 nodes based on previously described methods21,22 and the limited clinical utility in obtaining >5 nodes in a SLNB.23 Patients’ vital status and months from diagnosis to last contact and/or death were used to determine OS.
Statistical Analysis
Patient demographics, clinicopathologic characteristics, and additional therapies were compared between treatment groups using Chi-Square, Kruskal Wallis, and ANOVA for categorical, non-normally distributed continuous, and normally distributed continuous variables respectively. We compared OS between groups using the Kaplan-Meier method, log-rank test, and Cox proportional hazards models. We performed both multivariable and inverse probability of treatment weighted (IPTW; using propensity scores) Cox regression models to account for selection bias/confounding and determine clinical factors associated with OS.24,25 Propensity scores were created by estimating the probability of selection into the four treatment groups with a multinomial logistic regression model consisting of covariates associated with treatment group differences in univariate analyses (p≤0.15). The same covariates were clinically relevant to OS and, therefore, also included in the multivariable Cox model. Model covariates included age, race, CDCI, income, insurance, facility, histology, grade, hormone receptor/HER-2 status, T/N/overall stage, axillary nodal surgery, surgical margins, chemotherapy, hormone therapy, and year of diagnosis. Evaluation of standardized differences determined appropriate balance of all covariates after IPTW. The proportional hazards assumption was not violated as assessed by Schoenfeld residuals. Missing data was incorporated into the model as ‘unknown’ for each respective covariate.
HER-2 was not reported until 2010; therefore, 45.8% of patients had missing/unknown HER-2 status (n=3,871). We performed sensitivity analyses comparing multivariable Cox models for OS of all patients and with HER-2 unknown patients excluded. As there were no significant differences between models, we are presenting analyses including all patients.
Subgroup analyses by overall and T stage were performed using multivariable Cox proportional hazards regression models controlling for the same covariates as performed on the entire cohort. Statistical analysis was performed using SAS (version 9.4, SAS Institute, Cary, NC). All tests were two sided. P-values <0.05 were considered significant.
Results
Of the 8,445 males with invasive breast cancer, most underwent total mastectomy alone (61.2%, n=5,165), while 18.2% (n=1,539) underwent BCT, 12.4% (n=1,046) underwent total mastectomy with RT, and 8.2% (n=695) underwent partial mastectomy alone. Table 1 describes key demographic and clinicopathologic characteristics by treatment approach. There were significant differences between treatment groups for age, race, CDCI, histology, tumor size, grade, T, N and overall stage, hormone receptor/HER-2 status, surgical margins, nodal surgery, chemotherapy, and hormone therapy between treatment groups (p<0.05). Hormone receptor positive/HER-2 negative disease was more common among patients who underwent total mastectomy alone and with RT compared to BCT and partial mastectomy alone patients (45.5% & 47.1% vs. 40.4% & 35.5%), while rates of triple negative receptor status were greatest among BCT patients compared to patients who underwent partial mastectomy alone, total mastectomy alone, and total mastectomy with RT (4.9% vs. 3.0%, 1.4% & 1.4%, p<0.0001). Median follow-up for the entire cohort was 52 months (IQR 30–79).
Table 1.
Patient demographics and clinicopathologic characteristics by treatment approach.
| Breast Conserving Therapy N=1,539 | Partial Mastectomy Alone N=695 | Total Mastectomy Alone N=5,165 | Total Mastectomy with Radiation N=1,046 | P-value | |||||
|---|---|---|---|---|---|---|---|---|---|
| N/Mean | %/SD | N/Mean | %/SD | N/Mean | %/SD | N/Mean | %/SD | ||
|
Age (mean, SD, years) |
61.4 | 12.0 | 66.7 | 14.7 | 66.3 | 12.6 | 63.4 | 12.5 | <0.001 |
| Race | |||||||||
| Caucasian | 1,305 | 84.8% | 586 | 84.3% | 4,495 | 87.0% | 879 | 84.0% | 0.0006 |
| African American | 179 | 11.6% | 68 | 9.8% | 501 | 9.7% | 132 | 12.6% | |
| Asian/Pacific Islander | 29 | 1.9% | 15 | 2.2% | 89 | 1.7% | 17 | 1.6% | |
| Other/Unknown | 26 | 1.7% | 26 | 3.7% | 80 | 1.6% | 18 | 1.7% | |
| Charlson-Deyo | |||||||||
| Comorbidity Index | |||||||||
| 0 | 1,326 | 86.2% | 576 | 82.9% | 3,921 | 75.9% | 846 | 80.9% | <0.0001 |
| 1 | 174 | 11.3% | 83 | 11.9% | 939 | 18.2% | 161 | 15.4% | |
| 2 | 34 | 2.2% | 25 | 3.6% | 236 | 4.6% | 30 | 2.9% | |
| ≥3 | 5 | 0.3% | 11 | 1.6% | 69 | 1.3% | 9 | 0.9% | |
| Income | |||||||||
| <$38,000 | 220 | 14.3% | 102 | 14.7% | 714 | 13.8% | 167 | 16.0% | 0.15 |
| $38,000–47,999 | 339 | 22.0% | 171 | 24.6% | 1,061 | 20.5% | 212 | 20.3% | |
| $48,000–62,999 | 413 | 26.8% | 165 | 23.7% | 1,363 | 26.4% | 279 | 26.7% | |
| ≥$63,000 | 559 | 36.3% | 248 | 35.7% | 1,984 | 38.4% | 380 | 36.3% | |
| Unknown | 8 | 0.5% | 9 | 1.3% | 43 | 0.8% | 8 | 0.8% | |
| Insurance Status | |||||||||
| Uninsured | 22 | 1.4% | 12 | 1.7% | 82 | 1.6% | 25 | 2.4% | <0.0001 |
| Private | 833 | 54.1% | 286 | 41.2% | 2,099 | 40.6% | 486 | 46.4% | |
| Government | 657 | 42.7% | 386 | 55.5% | 2,918 | 56.5% | 527 | 50.4% | |
| Unknown | 27 | 1.8% | 11 | 1.6% | 66 | 1.3% | 9 | 0.9% | |
| Facility Type | |||||||||
| Comprehensive Cancer | 730 | 47.4% | 318 | 45.8% | 2,373 | 45.9% | 519 | 49.6% | 0.0006 |
| Community Cancer | 207 | 13.5% | 102 | 14.7% | 578 | 11.2% | 122 | 11.7% | |
| Academic/Research | 385 | 25.0% | 176 | 25.3% | 1,545 | 29.9% | 278 | 26.7% | |
| Other | 217 | 14.1% | 99 | 14.2% | 669 | 13.0% | 127 | 12.1% | |
| Histology | |||||||||
| Invasive Ductal (IDC) | 1,211 | 78.7% | 555 | 79.9% | 4,387 | 84.9% | 929 | 88.8% | <0.0001 |
| Invasive Lobular (ILC) | 163 | 10.6% | 50 | 7.2% | 205 | 4.0% | 45 | 4.3% | |
| Mixed IDC/ILC | 58 | 3.8% | 21 | 3.0% | 179 | 3.5% | 33 | 3.2% | |
| Medullary | 3 | 0.2% | 2 | 0.3% | 5 | 0.1% | 2 | 0.2% | |
| Papillary | 17 | 1.1% | 21 | 3.0% | 179 | 3.5% | 8 | 0.8% | |
| Mucinous | 33 | 2.1% | 16 | 2.3% | 62 | 1.2% | 5 | 0.5% | |
| NOS | 54 | 3.5% | 30 | 4.3% | 148 | 2.9% | 24 | 2.3% | |
|
Tumor size (median, IQR, cm) |
1.4 | 0.9–2.0 | 1.5 | 1.0–2.2 | 1.8 | 1.3–2.5 | 2.3 | 1.7–3.0 | <0.0001 |
| T Stage | |||||||||
| T1 | 1,197 | 77.8% | 493 | 70.9% | 3,090 | 59.8% | 443 | 42.4% | <0.0001 |
| T2 | 342 | 22.2% | 202 | 29.1% | 2,075 | 40.2% | 603 | 57.7% | |
| N Stage | |||||||||
| N0 | 1,251 | 81.3% | 588 | 84.6% | 3,856 | 74.7% | 343 | 32.8% | <0.0001 |
| N1 | 288 | 18.7% | 107 | 15.4% | 1,309 | 25.3% | 703 | 67.2% | |
| Stage | |||||||||
| 1 | 1,040 | 67.6% | 439 | 63.2% | 2,545 | 49.3% | 160 | 15.3% | <0.0001 |
| 2 | 499 | 32.4% | 256 | 36.8% | 2,620 | 50.7% | 886 | 84.7% | |
| Grade | |||||||||
| Well differentiated | 398 | 25.9% | 163 | 23.5% | 773 | 15.0% | 95 | 9.1% | <0.0001 |
| Moderately differentiated | 656 | 42.6% | 281 | 40.4% | 2,697 | 52.2% | 507 | 48.5% | |
| Poorly differentiated/ Anaplastic |
382 | 24.8% | 196 | 27.8% | 1,460 | 28.3% | 405 | 38.7% | |
| Unknown | 103 | 6.7% | 55 | 7.9% | 235 | 4.6% | 39 | 3.7% | |
| ER Status | |||||||||
| Positive | 1,284 | 83.5% | 567 | 81.6% | 4789 | 92.7% | 985 | 94.2% | <0.0001 |
| Negative | 225 | 14.6% | 83 | 11.9% | 223 | 4.3% | 42 | 4.0% | |
| Unknown | 29 | 1.9% | 45 | 6.5% | 153 | 3.0% | 19 | 1.8% | |
| PR Status | |||||||||
| Positive | 1,162 | 75.5% | 499 | 71.8% | 4,372 | 84.6% | 892 | 85.3% | <0.0001 |
| Negative | 344 | 22.4% | 150 | 21.6% | 624 | 12.1% | 132 | 12.6% | |
| Unknown | 33 | 2.1% | 46 | 6.6% | 169 | 3.3% | 22 | 2.1% | |
| HER-2 Status | |||||||||
| Positive | 105 | 6.8% | 46 | 6.6% | 424 | 8.2% | 95 | 9.1% | <0.0001 |
| Negative | 698 | 45.4% | 268 | 38.6% | 2,429 | 47.0% | 509 | 48.7% | |
| Unknown | 736 | 47.8% | 381 | 54.8% | 2,312 | 44.8% | 442 | 42.3% | |
|
Negative Surgical Margins Nodal Surgery |
1,478 | 96.0% | 626 | 90.1% | 5,040 | 97.6% | 1,004 | 96.0% | <0.0001 |
| None | 75 | 4.9% | 174 | 25.0% | 164 | 3.2% | 21 | 2.0% | <0.0001 |
| Sentinel Lymph Node Biopsy |
1,093 | 71.0% | 361 | 51.9% | 2,500 | 48.4% | 298 | 28.5% | |
| Axillary Dissection | 354 | 23.0% | 145 | 20.9% | 2,472 | 47.9% | 720 | 68.8% | |
| Unknown | 17 | 1.1% | 15 | 2.2% | 29 | 0.6% | 7 | 0.7% | |
| Chemotherapya | 572 | 37.2% | 153 | 22.0% | 1,565 | 30.3% | 685 | 65.5% | <0.0001 |
| Hormone therapy | 1,038 | 67.5% | 220 | 31.7% | 3,074 | 59.5% | 796 | 76.1% | <0.0001 |
SD, standard deviation; NOS, not otherwise specified.
Chemotherapy includes immunotherapies (i.e. Trastuzumab).
After controlling for demographic and clinicopathologic group differences, in both the multivariable (Table 2) and IPTW models, partial mastectomy alone (multivariable: aHR 1.73, 95%CI 1.39–2.14, p<0.0001; IPTW: HR 1.60, 95%CI 1.14–2.24, p=0.006), total mastectomy alone (multivariable: aHR 1.54, 95%CI 1.29–1.83, p<0.0001; IPTW: HR 1.62, 95%CI 1.27–2.07, p<0.0001) and total mastectomy with RT (multivariable: aHR 1.44, 95%CI 1.16–1.79, p=0.001; IPTW: HR 1.58, 95%CI 1.15–2.18, p=0.005) were associated with poorer OS compared to BCT. Older age, higher CDCI scores, histological grade, T and N stage, and triple negative receptor status were associated with worse OS, while SLNB, ALND, hormone therapy, and chemotherapy were associated with improved OS (Table 2). Figures 1.A-B illustrate crude and IPTW Kaplan Meier survival curves by treatment approach. At 5- and 10-years, OS rates (with IPTW adjustment) were 86.8% & 73.8% for BCT, 84.9% & 56.3% for partial mastectomy alone, 80.6% & 58.0% for total mastectomy alone, and 81.9% & 56.3% for total mastectomy with RT patients.
Table 2.
Multivariable Model for Overall Survival of Male Breast Cancer Patients (N=8,445).
| Hazard Ratio | 95% CI | P value | |
|---|---|---|---|
| Surgery | |||
| Breast Conserving Therapy | Reference | ||
| Partial Mastectomy Alone | 1.73 | 1.39–2.14 | <0.0001 |
| Total Mastectomy Alone | 1.54 | 1.29–1.83 | <0.0001 |
| Total Mastectomy with Radiation | 1.44 | 1.16–1.79 | 0.001 |
| Age | 1.05 | 1.04–1.05 | <0.0001 |
| Race | |||
| Caucasian | Reference | ||
| African American | 1.17 | 1.00–1.38 | 0.06 |
| Asian/Pacific Islander | 0.82 | 0.52–1.31 | 0.41 |
| Charlson-Deyo Comorbidity Index | |||
| 0 | Reference | ||
| 1 | 1.69 | 1.51–1.89 | <0.0001 |
| 2 | 2.68 | 2.24–3.21 | <0.0001 |
| ≥3 | 3.53 | 2.67–4.68 | <0.0001 |
| Income | |||
| <$38,000 | Reference | ||
| $38,000–47,999 | 1.02 | 0.87–1.19 | 0.83 |
| $48,000–62,999 | 1.01 | 0.87–1.17 | 0.90 |
| ≥$63,000 | 0.83 | 0.72–0.96 | 0.02 |
| Insurance Status | |||
| Uninsured | Reference | ||
| Private | 0.69 | 0.44–1.09 | 0.11 |
| Government | 0.92 | 0.59–1.45 | 0.73 |
| Facility Type | |||
| Comprehensive Cancer | Reference | ||
| Community Cancer | 1.06 | 0.92–1.22 | 0.43 |
| Academic/Research | 0.77 | 0.69–0.87 | <0.0001 |
| Histology | |||
| Invasive Ductal (IDC) | Reference | ||
| Invasive Lobular (ILC) | 0.86 | 0.68–1.08 | 0.18 |
| Mixed IDC/ILC | 1.05 | 0.79–1.39 | 0.73 |
| Medullary | 0.37 | 0.05–2.63 | 0.32 |
| Papillary | 1.04 | 0.76–1.43 | 0.81 |
| Mucinous | 0.78 | 0.48–1.27 | 0.32 |
| Grade | |||
| Well differentiated | Reference | ||
| Moderately differentiated | 1.32 | 1.14–1.54 | 0.0003 |
| Poorly differentiated | 1.66 | 1.41–1.96 | <0.0001 |
| Hormone Receptor/HER-2 | |||
| HR+/HER-2- | Reference | ||
| HR+/HER-2+ | 1.20 | 0.94−−1.53 | 0.15 |
| HR-/HER-2+ | 1.68 | 0.79–3.58 | 0.19 |
| HR-/HER-2- | 1.87 | 1.26–2.77 | 0.002 |
| Stage II | 0.97 | 0.82–1.16 | 0.76 |
| T2 | 1.58 | 1.36–1.83 | <0.0001 |
| N1 | 1.53 | 1.34–1.74 | <0.0001 |
| Nodal Surgery | |||
| None | Reference | ||
| Sentinel Lymph Node Biopsy | 0.47 | 0.40–0.57 | <0.0001 |
| Axillary Dissection | 0.54 | 0.45–0.65 | <0.0001 |
| Positive Surgical Margins | 1.03 | 0.79–1.36 | 0.81 |
| Hormone therapy | 0.74 | 0.67–0.82 | <0.0001 |
| Chemotherapya | 0.70 | 0.61–0.80 | <0.0001 |
CI, confidence interval.
Chemotherapy includes immunotherapies (i.e. Trastuzumab).
Figure 1.
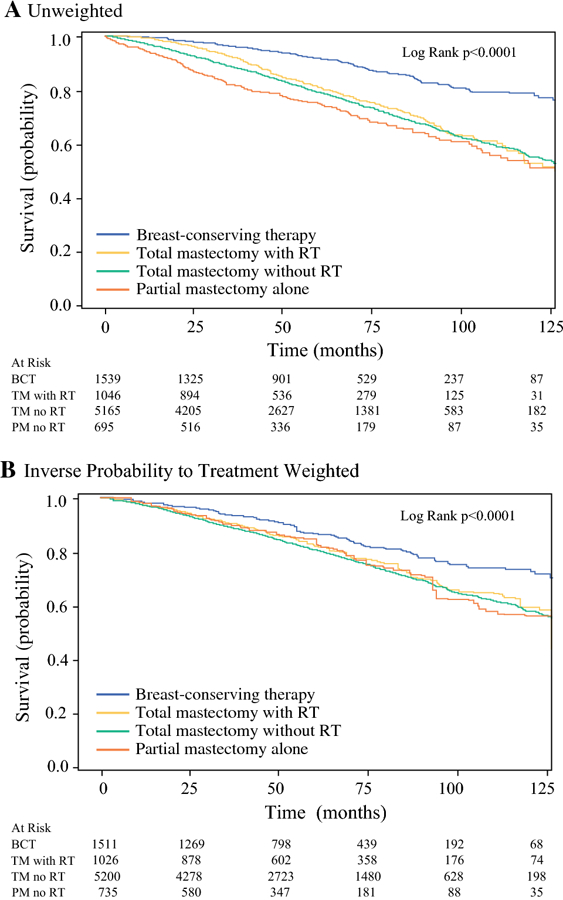
A-B. Crude (A) and inverse probability of treatment weighted (B) overall survival for male breast cancer patients with breast conserving therapy (BCT), total mastectomy with (TM with RT) and without radiation therapy (TM no RT), and partial mastectomy alone (PM no RT).
Subgroup analyses by overall and T stage are presented in Figures 2.A-B and 3.A-B and Table S1 (Supplement). For stage I and T1 disease, there were no OS differences for BCT and total mastectomy with RT patients (p>0.05, Table S1). Total and partial mastectomy alone were associated with worse OS compared to BCT (p<0.05, Table S1). For stage II and T2 disease, total mastectomy alone and with RT and partial mastectomy alone were associated with worse OS compared to BCT (p<0.05, Table S1).
Figure 2.
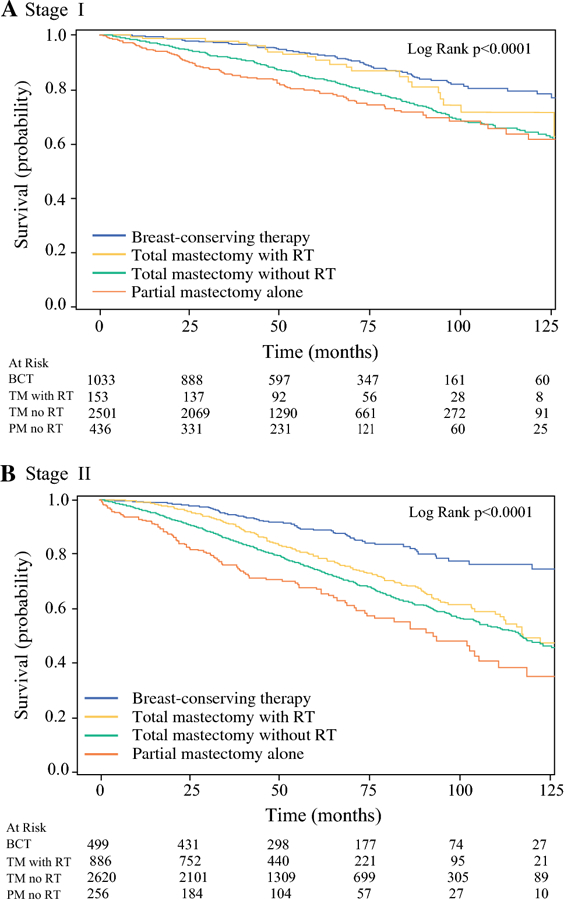
A-B. Univariate overall survival for breast conserving therapy (BCT) compared to partial mastectomy alone (PM no RT), total mastectomy alone (TM no RT) and total mastectomy with radiation therapy (TM with RT) by stage.
Figure 3.
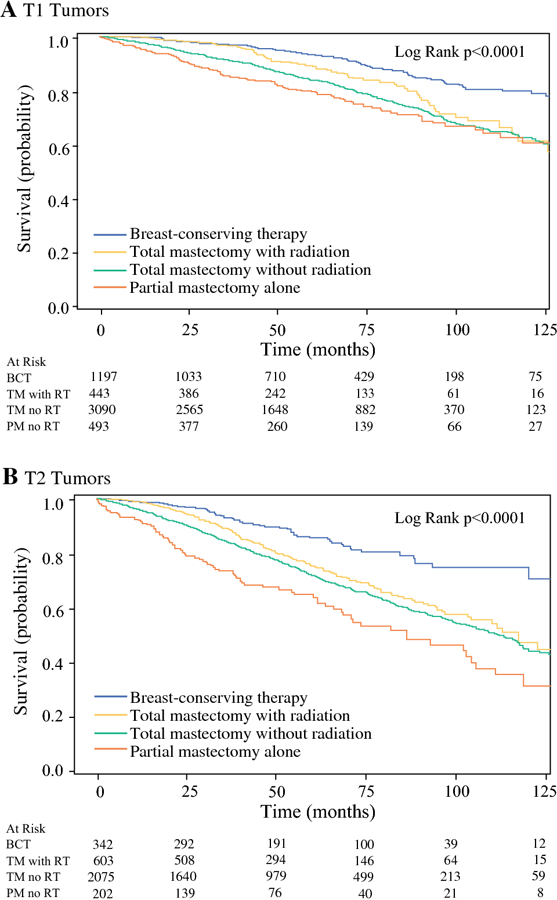
A-B. Univariate overall survival for breast conserving therapy (BCT) compared to partial mastectomy alone (PM no RT), total mastectomy alone (TM no RT) and total mastectomy with radiation therapy (TM with RT) by T stage.
Discussion
In this NCDB analysis of male breast cancer patients, despite total mastectomy being performed more commonly, BCT was associated with greater survival compared to total mastectomy with and without RT and partial mastectomy alone. These findings were surprising as current consensus guidelines based on randomized clinical trials with female patients regard BCT and total mastectomy to be oncologically equivalent.2,3,26 Therefore, these findings are particularly important as they underscore the need for greater consideration and adoption of BCT for male breast cancer patients.
This study strengthens the current limited body of research investigating oncologic outcomes among male breast cancer patients. In a retrospective SEER analysis, Cloyd et al. found equivalent disease-specific and OS among men with breast cancer who underwent partial mastectomy compared to those who underwent total mastectomy.13 Although Cloyd et. al. included RT in the multivariable analysis, they did not stratify partial mastectomy patients by receipt of RT. As shown in our analysis, there were significant differences in age, comorbidities, surgical margins, and systemic therapy for BCT and partial mastectomy alone patients, suggesting that partial mastectomy patients who did and did not undergo RT are distinct patient cohorts. Additionally, after controlling for these group differences, we found that BCT was associated with greater survival compared to partial mastectomy alone. These findings highlight the importance of RT to improve oncologic outcomes in male breast cancer patients who undergo partial mastectomy.
Although the oncologic benefit of RT in breast cancer has been widely cited in female literature,27,28 RT in male breast cancer research is predominantly limited to retrospective single institution studies (many including patients from the 1960s and 1970s).29,30 Although a number of these male-specific studies have shown an improvement in locoregional control with RT, few have observed a survival benefit.29,31,32 Therefore, the findings from the present study are noteworthy as they highlight a survival benefit associated with RT in male breast cancer. Additionally, the equivalent survival observed for stage I and T1 tumors treated with BCT or total mastectomy with RT suggests that RT may provide greater oncologic benefit than extensive surgical resection in small tumors. A recent study by Jatoi et al. suggested an ‘abscopal effect’ to explain such findings.33 The ‘abscopal effect’ refers to the potential for RT to have both a localized and systemic anti-tumor effect from immunostimulation, leading to lower incidence of distant recurrence and greater survival. Despite such oncologic benefits, multiple studies have observed lower rates of RT compliance among male breast cancer patients compared to female patients.11,12 Therefore, although our findings suggest BCT is appropriate therapy in men, BCT is only beneficial in those in which RT is feasible, as lumpectomy alone is not associated with the same survival benefit.
Although we acknowledge the potential influence of selection bias in this retrospective analysis, the similarities in tumor size, nodal disease, and surgical margins between patients who underwent BCT and mastectomy alone provides further confidence in the validity of our findings. Although the median tumor size was slightly larger for total mastectomy alone patients, the median tumor size for both groups was small (1.4–1.8cm). Additionally, most patients in both cohorts were without nodal metastases (75–81%) and had high rates of negative surgical margins (96%−98%). We acknowledge that the mastectomy with RT patients are a distinct group with larger tumors (median 2.3cm) and high rates of nodal disease (67%) and, therefore, at greater risk of confounding. However, the clinical similarities between patients who underwent BCT and mastectomy alone (in addition to stratification by stage and robust multivariable and propensity score analyses) provides credence to our findings of a survival benefit associated with BCT compared to total mastectomy alone.
Additionally, recent retrospective population-based studies comparing BCT to total mastectomy alone has observed similar results in female breast cancer patients, further validating our results.34–37 Agarwal et al. performed a retrospective analysis comparing BCT to total mastectomy in female breast cancer patients with tumors ≤4cm and ≤3 positive lymph nodes using the SEER database and found greater survival in BCT patients compared to women who underwent total mastectomy with and without RT.35 A separate SEER analysis of female patients by Bagaria et al. found similar results, with worse disease-specific survival among women who underwent total mastectomy compared to BCT in low-, intermediate-, and high-risk tumors based on hormone status and grade.34 Furthermore, in a multicenter prospective cohort study of young women with breast cancer in the United Kingdom, although BCT was associated with earlier local recurrence, BCT was associated with improved distant disease-free interval and OS compared to total mastectomy.37 Our findings complement this body of research by demonstrating similar findings in male patients, specifically that BCT is associated with improved survival compared to total mastectomy.
The present study has important implications for post-therapeutic surveillance in male breast cancer. Current guidelines for post-therapeutic surveillance in men are the same as those recommended for women, which include history and physical examination every 4–6 months for the first 5 years and annually afterwards.2,38 BCT patients should also undergo annual mammography. Future research is needed to determine if this surveillance regimen is adequate in men, as these guidelines were created based on research with predominantly female patients.
Despite this study’s strengths, it does have limitations inherent to administrative data. This study was retrospective and patients were not randomized to treatment approaches. Despite extensive multivariable analyses and model adjustment with propensity scores, our results may still have been confounded by selection bias related to nuanced clinical, pathologic, or sociodemographic features that could affect choice of therapy, including patients’ and physicians’ preferences. For example, in addition to the higher rate of triple negative receptor status among BCT patients compared to mastectomy alone patients (4.9% vs. 1.4%, which we controlled for in our IPTW and multivariable analyses), patient preferences may also potentially explain why BCT patients were more commonly treated with adjuvant systemic therapies compared to mastectomy alone patients. BCT patients may have been more amenable to additional therapies in general, including systemic therapy and RT, whereas mastectomy alone patients may have preferred to avoid additional therapies, which is why they selected total mastectomy over BCT. We acknowledge that many patients had missing HER-2 status. However, this likely had negligible influence on our results as most male breast cancer patients have been shown to be HER-2 negative and our sensitivity analyses revealed no significant survival differences when excluding HER-2 unknown patients.12 As NCDB does not provide detailed information regarding systemic therapeutics, we were not able to control for specific adjuvant regimens or assess if patients completed their entire course of adjuvant therapy. NCDB does not provide data on cause of death or recurrence, limiting our primary outcome to OS. Lastly, we lacked information regarding treatment complications and adverse side-effects, which have the potential to influence surgical and RT treatment decisions and patient quality of life.
In conclusion, in this analysis of a large, national cohort of male breast cancer patients, although total mastectomy was more commonly performed, BCT was associated with improved survival compared to total mastectomy. These findings suggest that BCT is a viable treatment approach for male breast cancer patients and should be given greater consideration by surgeons and oncologists. However, as RT compliance is an essential component in BCT, careful, personalized, shared decision-making between physicians and male breast cancer patients is necessary to ensure the selected treatment approach, whether BCT or mastectomy, is reasonable for the patient.
Supplementary Material
Synopsis:
Breast conserving therapy (BCT) in men was associated with greater survival compared to total mastectomy +/− radiation and partial mastectomy alone suggesting that BCT is a viable therapy for men and should be given greater consideration by surgical oncologists.
Acknowledgments
This study was presented at the American Society of Clinical Oncology Annual Meeting on June 2, 2018, Chicago, Illinois.
Funding: The National Center for Advancing Translational Sciences, NIH [UL1TR001860] and the Agency for Healthcare Research and Quality [T32HS022236].
Footnotes
Conflicts of interest: None.
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin January 2017;67(1):7–30. [DOI] [PubMed] [Google Scholar]
- 2.Gradishar WJ, Anderson BO, Balassanian R, et al. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer Version 22017. https://www.nccn.org/professionals/physiciangls/pdf/breast.pdf. [Google Scholar]
- 3.Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med October 17 2002;347(16):1233–1241. [DOI] [PubMed] [Google Scholar]
- 4.Aggarwal A, Liu ML, Krasnow SH. Breast cancer in male veteran population: an analysis from VA cancer registry. J Community Support Oncol August 2014;12(8):293–297. [DOI] [PubMed] [Google Scholar]
- 5.O’Malley CD, Prehn AW, Shema SJ, Glaser SL. Racial/ethnic differences in survival rates in a population-based series of men with breast carcinoma. Cancer June 1 2002;94(11):2836–2843. [DOI] [PubMed] [Google Scholar]
- 6.Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer July 1 2004;101(1):51–57. [DOI] [PubMed] [Google Scholar]
- 7.Stierer M, Rosen H, Weitensfelder W, et al. Male breast cancer: Austrian experience. World J Surg Sep-Oct 1995;19(5):687–692; discussion 692–683. [DOI] [PubMed] [Google Scholar]
- 8.Schaub NP, Maloney N, Schneider H, Feliberti E, Perry R. Changes in male breast cancer over a 30-year period. Am Surg August 2008;74(8):707–711; discussion 711–702. [PubMed] [Google Scholar]
- 9.Ribeiro GG, Swindell R, Harris M, Banarjee SS, Cramer A. A review of the management of the male breast carcinoma based on an analysis of 420 treated cases. The Breast 1996;5:141–146. [Google Scholar]
- 10.Golshan M, Rusby J, Dominguez F, Smith BL. Breast conservation for male breast carcinoma. Breast December 2007;16(6):653–656. [DOI] [PubMed] [Google Scholar]
- 11.Cloyd JM, Hernandez-Boussard T, Wapnir IL. Poor compliance with breast cancer treatment guidelines in men undergoing breast-conserving surgery. Breast Cancer Res Treat May 2013;139(1):177–182. [DOI] [PubMed] [Google Scholar]
- 12.Cardoso F, Bartlett JMS, Slaets L, et al. Characterization of male breast cancer: results of the EORTC 10085/TBCRC/BIG/NABCG International Male Breast Cancer Program. Ann Oncol February 1 2018;29(2):405–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cloyd JM, Hernandez-Boussard T, Wapnir IL. Outcomes of partial mastectomy in male breast cancer patients: analysis of SEER, 1983–2009. Ann Surg Oncol May 2013;20(5):1545–1550. [DOI] [PubMed] [Google Scholar]
- 14.Jagsi R, Abrahamse P, Hawley ST, Graff JJ, Hamilton AS, Katz SJ. Underascertainment of radiotherapy receipt in Surveillance, Epidemiology, and End Results registry data. Cancer January 15 2012;118(2):333–341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Korde LA, Zujewski JA, Kamin L, et al. Multidisciplinary meeting on male breast cancer: summary and research recommendations. J Clin Oncol April 20 2010;28(12):2114–2122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fentiman IS, Fourquet A, Hortobagyi GN. Male breast cancer. Lancet February 18 2006;367(9510):595–604. [DOI] [PubMed] [Google Scholar]
- 17.Greif JM, Pezzi CM, Klimberg VS, Bailey L, Zuraek M. Gender differences in breast cancer: analysis of 13,000 breast cancers in men from the National Cancer Data Base. Ann Surg Oncol October 2012;19(10):3199–3204. [DOI] [PubMed] [Google Scholar]
- 18.Massarweh SA, Sledge GW, Miller DP, McCullough D, Petkov VI, Shak S. Molecular Characterization and Mortality From Breast Cancer in Men. J Clin Oncol May 10 2018;36(14):1396–1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boffa DJ, Rosen JE, Mallin K, et al. Using the National Cancer Database for Outcomes Research: A Review. JAMA Oncol December 1 2017;3(12):1722–1728. [DOI] [PubMed] [Google Scholar]
- 20.Bilimoria KY, Bentrem DJ, Stewart AK, Winchester DP, Ko CY. Comparison of commission on cancer-approved and -nonapproved hospitals in the United States: implications for studies that use the National Cancer Data Base. J Clin Oncol September 1 2009;27(25):4177–4181. [DOI] [PubMed] [Google Scholar]
- 21.Schmocker RK, Caretta-Weyer H, Weiss JM, et al. Determining breast cancer axillary surgery within the surveillance epidemiology and end results-Medicare database. J Surg Oncol June 2014;109(8):756–759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lorimer PD, White RL, Walsh K, et al. Pediatric and Adolescent Melanoma: A National Cancer Data Base Update. Ann Surg Oncol November 2016;23(12):4058–4066. [DOI] [PubMed] [Google Scholar]
- 23.Yi M, Meric-Bernstam F, Ross MI, et al. How many sentinel lymph nodes are enough during sentinel lymph node dissection for breast cancer? Cancer July 1 2008;113(1):30–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garrido MM, Kelley AS, Paris J, et al. Methods for constructing and assessing propensity scores. Health Serv Res October 2014;49(5):1701–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Austin PC. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivariate Behav Res May 2011;46(3):399–424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fisher B, Jeong JH, Anderson S, Bryant J, Fisher ER, Wolmark N. Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med August 22 2002;347(8):567–575. [DOI] [PubMed] [Google Scholar]
- 27.Ebctcg, McGale P, Taylor C, et al. Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet June 21 2014;383(9935):2127–2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Early Breast Cancer Trialists’ Collaborative G, Darby S, McGale P, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet November 12 2011;378(9804):1707–1716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yu E, Suzuki H, Younus J, et al. The impact of post-mastectomy radiation therapy on male breast cancer patients--a case series. Int J Radiat Oncol Biol Phys February 1 2012;82(2):696–700. [DOI] [PubMed] [Google Scholar]
- 30.Jardel P, Vignot S, Cutuli B, et al. Should Adjuvant Radiation Therapy Be Systematically Proposed for Male Breast Cancer? A Systematic Review. Anticancer Res January 2018;38(1):23–31. [DOI] [PubMed] [Google Scholar]
- 31.Madden NA, Macdonald OK, Call JA, Schomas DA, Lee CM, Patel S. Radiotherapy and Male Breast Cancer: A Population-based Registry Analysis. Am J Clin Oncol October 2016;39(5):458–462. [DOI] [PubMed] [Google Scholar]
- 32.Abrams MJ, Koffer PP, Wazer DE, Hepel JT. Postmastectomy Radiation Therapy Is Associated With Improved Survival in Node-Positive Male Breast Cancer: A Population Analysis. Int J Radiat Oncol Biol Phys June 1 2017;98(2):384–391. [DOI] [PubMed] [Google Scholar]
- 33.Jatoi I, Benson JR, Kunkler I. Hypothesis: can the abscopal effect explain the impact of adjuvant radiotherapy on breast cancer mortality? NPJ Breast Cancer 2018;4:8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bagaria SP, Wasif N, Rawal B, McLaughlin SA, Giuliano AE. Is mastectomy undertreatment for low-risk breast cancers eligible for breast-conserving therapy? Cancer August 15 2015;121(16):2705–2712. [DOI] [PubMed] [Google Scholar]
- 35.Agarwal S, Pappas L, Neumayer L, Kokeny K, Agarwal J. Effect of breast conservation therapy vs mastectomy on disease-specific survival for early-stage breast cancer. JAMA Surg March 2014;149(3):267–274. [DOI] [PubMed] [Google Scholar]
- 36.Christiansen P, Carstensen SL, Ejlertsen B, et al. Breast conserving surgery versus mastectomy: overall and relative survival-a population based study by the Danish Breast Cancer Cooperative Group (DBCG). Acta Oncol January 2018;57(1):19–25. [DOI] [PubMed] [Google Scholar]
- 37.Maishman T, Cutress RI, Hernandez A, et al. Local Recurrence and Breast Oncological Surgery in Young Women With Breast Cancer: The POSH Observational Cohort Study. Ann Surg July 2017;266(1):165–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ferzoco RM, Ruddy KJ. Optimal delivery of male breast cancer follow-up care: improving outcomes. Breast Cancer (Dove Med Press) 2015;7:371–379. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


