Abstract
Objective
Meals high in protein induce greater intermeal satiety than meals high in fat and carbohydrates. We studied the gut hormone response and subsequent food intake after breakfasts high in protein, carbohydrate or high in fat controlled for volume, calories and appearance.
Design and Methods
Eight healthy volunteers participated in this randomized three-way crossover study. Study breakfasts were calculated to provide 20% of daily energy requirements and provided either 60% of energy from protein, fat or carbohydrate. Blood was drawn half-hourly for 4 h; energy intake at a subsequent ad libitum meal was measured.
Results
Total ghrelin decreased after food intake equally with the three breakfasts. PYY levels were highest after the high protein breakfast (P = 0.005). Indeed, PYY at 240 min was highest after the high protein breakfast compared to the high fat breakfast and to the high carbohydrate breakfast (P = 0.011 and P = 0.012, respectively). GLP-1 levels were highest after the high protein breakfast (P = 0.041) at 120 min and remained higher throughout the study. These differences in gut hormones did not translate into differences in food intake (1023 ± 390 kcal after high protein, 1016 ± 388 kcal after high fat and 1158 ± 433 kcal after high carbohydrate).
Conclusion
We conclude that a high protein meal increases circulating concentrations of the gut hormones PYY and GLP-1, but when meals are matched for volume, appearance and caloric value, these gut hormone changes do not translate into a reduction in ad libitum food intake.
Introduction
Food ingestion triggers the release of several gastrointestinal hormones including ghrelin which is secreted by the stomach and glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) which are produced primarily by the L-cells in the distal small intestine and colon (1). Recently, several studies have suggested that changes in the macronutrient composition of meals can influence gut hormone release, with differences in the time course and peak concentrations of ghrelin and PYY reported (2–4) (Table 1). However, such manipulations often also affect the energy content, energy density and volume of food delivered, which may have independent effects on gut hormone secretion and may influence nutrient absorption and satiation. We performed a randomized three-way crossover study in which volunteers were given a test breakfast in which 60% of total energy content was derived from protein/fat/carbohydrate, with 20% derived from each of the other two macronutrients (fat and carbohydrate). All three test meals were matched for volume and total energy content and effects on hunger and satiety, postprandial gut hormone levels and subsequent food intake at an ad libitum meal were measured.
Table 1. Summary of studies investigating the gut hormone response to macronutrient manipulations.
| Author | Subjects | Design | Control for | Outcomes |
|---|---|---|---|---|
| Erdmann et al. | 10 healthy volunteers | Crossover design with 6 occasions | Volume: no | Ghrelin levels increased after high protein meal |
| Regulatory Peptides 2003 | HF: 85% | Calories: no | Late ghrelin suppression less in HCHO compared to HF | |
| HP: 99% HCHO: 62% Fruits: 93% CHO 75 g glucose in 300 ml water sham feeding: gastric distension by guar Gut hormones: total ghrelin |
Appearance: no Subsequent meal: no |
|||
| Batterham et al. | 10 normal weight and 9 obese volunteers | Crossover design with three occasions | Volume: not mentioned | HP greatest increment in total plasma PYY and integrated PYY levels in normal and obese subjects. |
| Cell metabolism 2006 | HF 66 | Calories: yes, fixed | Active ghrelin, GLP-1 no differential responses to meals | |
| HP 65 HCHO 65 Gut hormones: PYY, active ghrelin, active GLP-I |
Appearance: yes Subsequent meal: no |
|||
| Foster-Schubert et al. Journal of Clinical Endocrinology and Metabolism 2008 | 16 healthy volunteers | Crossover design with 3 occasions | Volume: yes | Suppression of acyl and total ghrelin protein during HP was greater than HCHO and suppression during HCHO was greater than HF |
| HF & HP & HCHO: 80% | Calories: calculated to 20% energy requirements | |||
| Gut hormones: acyl and total grhelin | Appearance: yes | |||
| Subsequent meal: no | ||||
| Eller et al. | 10 healthy men | Crossover design with 3 occasions | Volume: not mentioned | GLP-I no differences |
| Clinical Endocrinology 2007 | HF 80% | Calories: yes, fixed (720 kcal/70 kg) | Greater des-acyl ghrelin decrease after HCHO (45%) than after HF (17%) | |
| HCHO 80% prolonged fast Gut hormones: des-acyl ghrelin, GLP-I |
Appearance: no Subsequent meal: no |
|||
| Maffeis et al. | 10 prepubertal obese boys | Crossover design with 3 occasions | Volume: no | PYY, CCK, and ghrelin concentrations not different |
| Integrative Physiology 2009 | HF 52% | Calories: Calculated to 25% of energy requirements | GLP-1 higher after HF than MF | |
| Medium fat 27% HCHO 61% Gut Hormones: PYY, CCK, total ghrelin and total GLP-I |
Appearance: no Subsequent meal: no |
|||
| Erdmann et al. | 14 healthy volunteers | Crossover design with 5 occasions | Volume: no | Intake testmeal HF 244 g, HP 293 g, HCHO 321 g |
| Journal of Clinical Endocrinology and Metabolism 2004 | HF: 86% | Calories: instructed to eat until satiation, at least 50% more provided. | After HCHO ghrelin decreased, with all the other meals ghrelin increased | |
| HP: 83% HCHO: 80% Fruits: 93% Vegetables: 75% Gut Hormones: total ghrelin |
Appearance: no Subsequent meal: yes |
|||
| Al Awar et al. | 11 healthy women | Crossover design with 3 occasions | Volume: yes | Ghrelin persisted at significantly lower levels for a longer duration after the HP meal compared to HF and balanced meal |
| Clinical Science 2005 | Balanced 45% CHO, 45% F, 10% P HP 35% HF 45% Hormones: acylated ghrelin |
Calories: Calculated to 30% energy requirements Appearance: yes Subsequent meal: no |
||
| Monteleone et al. | 14 healthy women | Crossover with 2 occasions | Volume: not mentioned | Nadir ghrelin lower after HCHO then after HF meal |
| Journal of Clinical Endocrinology and Metabolism 2003 | HCHO: 77% | Calories: yes, fixed | ||
| HF: 75% Gut hormones: plasma ghrelin |
Appearance: no Subsequent meal: no |
|||
| Lomenick et al. | 13 normal weight and 19 obese children | Crossover with 3 occasions | Volume: not mentioned | AUC for ghrelin not different between meals |
| Journal of Clinical Endocrinology and Metabolism 2009 | HCHO: 88% | Calories: yes, fixed | PYY significantly higher after HP compared to HF and HCHO in obese children not in normal weight children | |
| HP 44%: HF: 81% Gut hormones: total ghrelin, total PYY |
Appearance: no Subsequent meal: no |
HCHO: high carbohydrate, HP: high protein, HF: high fat
Methods
Eight healthy volunteers, who were weight stable, participated in this randomized three-way crossover study (five females and three males, mean age of 32 years, range 23-55 years). Exclusion criteria were use of any medication and presence of any medical illnesses or food allergies. Mean BMI of the subjects was 24.5 ± 0.9 kg m−2 (BMI range 19.8-27.5 kg m−2). Written informed consent was obtained prior to the study and approval was obtained from the Local Regional Ethics Committee in Cambridge, UK. Each study occasion was separated by at least 1 week. Subjects were fasted from 22:00 h the night before the study and were admitted to the clinical research facility at 7:00 h. An intravenous indwelling cannula was inserted and volunteers rested for ~30 min. Blood was drawn and visual analogue scores to assess hunger and fullness were completed half-hourly from 7:30 h onwards ending at 12:00 h. Isocaloric, isovolaemic test breakfasts were given at 8:00 h and subjects were instructed to finish within 25 min. The calories given per breakfast were standardized for each participant to match 20% of the individually calculated energy requirements with the Schofield formula (5).
All three breakfasts consisted of pancakes with trimmings to give 60% of the energy content as protein/fat/carbohydrate with 20% provided by the other two macronutrients in each case. Specific attention was given to ensure subjects could not tell the nature of the manipulation by avoiding foods that are well recognized to be high in particular macronutrients (e.g. sausages which are known to be high in protein). Carbohydrates made up 60% of the energy content of the high-carbohydrate breakfast, leaving 20% for protein and 20% for fat. The high-carbohydrate breakfast consisted of buckwheat pancakes served with bacon and maple syrup. The energy content of the high-protein breakfast consisted of 60% of protein, 20% of fat and 20% of carbohydrate. The pancakes were made with high-protein pancake mix (Avidlite pancake mix) and served with no-sugar maple syrup and full-fat Greek yoghurt. Fat made up 60% of the energy content of the high-fat breakfast with 20% for carbohydrate and 20% for protein. The wholemeal pancakes were served with bacon and grated cheddar cheese (Table 2). We matched the total carbohydrate to sugar ratio for the three test breakfasts. A survey after the study was completed confirmed participants did not realize the breakfasts were designed to be high in one macronutrient. Water was added to match all test meals for volume. We note that previous work has shown that water intake included in the food itself versus water intake as a beverage in a glass may have differing effects on food intake (6). An ad-libitum lunch was served at 12:15 h. The macronutrient composition of the lunch was 50% carbohydrate, 30% fat and 20% protein providing a total of 20 MJ.
Table 2. Details of the composition of the test breakfasts.
| Macronutrient composition | 60% protein/20% fat/20% carbohydrates | 60% carbohydrate/20% fat/20% protein | 60% fat/20% carbohydrate/20% protein |
|---|---|---|---|
| Ingredients | Pancakes (Avidlite pancake mix) | Pancakes (made with buckwheat, whole milk, rapeseed oil and 2 eggs) | Pancakes (Allinson whole meal flour, whole milk, rapeseed oil, sugar and 1 egg) |
| Total full fat greek yoghurt | Unsmoked bacon | Unsmoked bacon | |
| Sugar free maple syrup | Canadian maple syrup no 1 | Grated cheddar | |
| CHO:sugar ratio | 3.25 | 3.25 | 3.25 |
Percentages are energy of single macronutrient as percentage of total energy. The total energy provided was calculated to provide 20% of daily estimated energy requirements.
Blood was collected in EDTA tubes containing 100 µL of aprotinin (ghrelin, PYY and GLP-1), lithium heparin tubes (insulin) and fluoride oxalate tubes (glucose). Plasma samples were centrifuged immediately at 4° C and stored at −80°C until assays were performed. Plasma glucose was assayed on the same day by using the glucose oxidase method. Insulin was quantified using a commercially available immunoassay (AutoDELFIA Insulin Kit; Perkin Elmer, Wellesley, MA), which has an intra-assay CV of 3.5-4.5%. Plasma PYY and total GLP-1 were assessed using an established in-house radio-immunoassay (RIA) described previously (7,8). The detection limit of the PYY and GLP-1 assays was 2.5 and 7.5 pmol l−1 with an intra-assay coefficient variation of 5.8 and 5.4%, respectively. The ghrelin assay crossreacted fully with both acylated and des-acylated ghrelin and did not crossreact with any other known gastrointestinal or pancreatic hormones (9). The antiserum, SC-10368 (Santa Cruz Biotechnology, CA), was used at a final dilution of 1:50,000. 125I ghrelin was prepared using Bolton & Hunter reagent (Amersham International, UK) and purified by high-pressure liquid chromatography (HPLC). The assay detected changes of 25 pmol l−1 with 95% confidence limit. The intra-assay coefficient of variation (CV) was 5.5%.
Data are presented as mean ± standard error of the mean and analyzed using SPSS for Windows version 17.0. ANOVA analysis with repeated measures was used to test for within-subjects changes and between breakfasts differences using an interaction term for time and study breakfasts. Comparisons at specific sampling time points were made using ANOVA with post-hoc comparisons using Tukey’s best to correct for multiple testing. A P value of 0.05 was considered significant.
Results
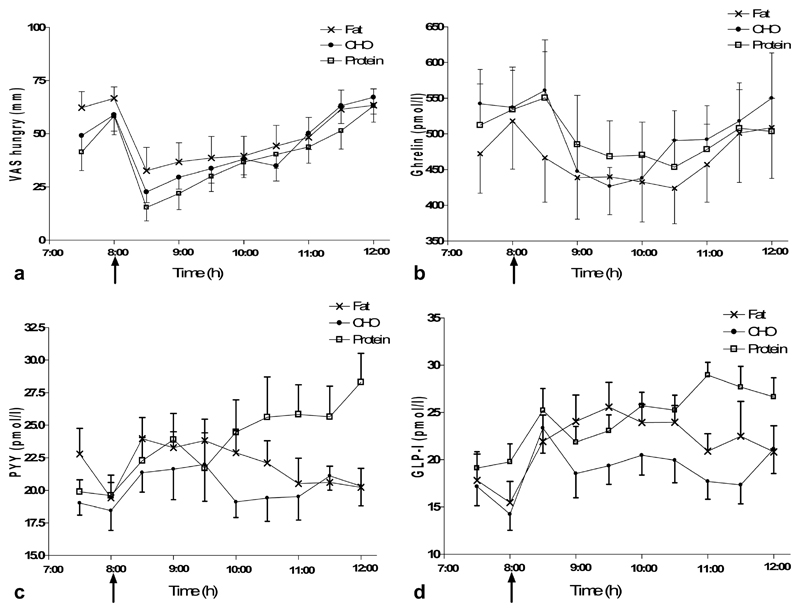
Hunger scores decreased after food intake (Figure 1a) and fullness scores increased. We did not find any differences in hunger scores (P = 0.777) or fullness scores (P = 0.888) nor in the area under the curve (AUC) (hunger scores P = 0.634 and fullness scores P = 0.461) between the different macronutrient manipulations.
Figure 1.
Changes in hunger, satiety and gut hormones after meals differing in macronutrient composition. Mean ± standard error of the mean for VAS scores (a), ghrelin (b), PYY (c), and GLP-1 (d). ANOVA analysis with repeated measures was used. PYY and GLP-1 levels were significantly higher after the high protein breakfast compared to the high carbohydrate and high fat breakfasts.
We did not find any differences in hunger scores or gut hormones at fasting between the three different study occasions. Glucose concentrations peaked after 30 min for the high protein and high fat breakfasts (5.3 ± 0.9 mmol l−1 and 5.1 ± 0.8 mmol l−1) and after 1 h for the high carbohydrate breakfast (5.2 ± 1.3 mmol l−1). The maximal plasma insulin increase was more than twofold greater after the high carbohydrate breakfast (206.5 ± 148.6 pmol l−1) than the high protein breakfast (97.7 ± 41.4 pmol l−1) and 55% higher than the high fat breakfast (163.3 ± 57.3 pmol l−1, P = 0.016).
Ghrelin levels decreased after food intake equally with the three breakfasts (Figure 1b). PYY levels were highest after the high protein breakfast (P = 0.005, Figure 1c). Indeed, after 180 min, PYY levels were highest after the high protein breakfast compared to the high fat breakfast and to the carbohydrate breakfast (P = 0.011 and P = 0.012, respectively). GLP-1 levels were also highest after the high protein breakfast (P = 0.041, Figure 1d) and remained higher throughout the study compared to the high carbohydrate and high fat breakfasts.
There were no differences in ad libitum energy intake (1023 ± 390 kcal after high protein, 1016 ± 388 kcal after high fat, and 1158 ± 433 kcal after high carbohydrate) with the three test meals. It is possible that, had the timing of the ad libitum test meal not been fixed, differences in the onset of voluntary consumption (intermeal interval) may have emerged.
Discussion
In this study, we compared the response to three isovolaemic and isocaloric test breakfasts that were high in one macronutrient in healthy volunteers. We designed our study specifically controlling for total energy content, volume and appearance as visual cues can trigger expectations of hunger and satiety and may affect ghrelin responses (10). We found that a high protein meal resulted in increased secretion of PYY and GLP-1. However, this postprandial increase did not translate into feeling less hungry or more full or a reduction in food intake. We did not standardize food intake and meal timing on the day before the manipulation of the meal with different macronutrient composition; however, baseline values for hunger, glucose, insulin, PYY, GLP-I and ghrelin did not differ between the three occasions suggesting that the participants were in comparable metabolic states when studied.
Increased circulating levels of PYY after a high protein meal have been demonstrated previously (2,4) (Summarized in Table 1). In addition, for the first time, we found that GLP-1 levels were also elevated after a high protein meal. In previous studies, there were no differences in GLP-1 response after meals or drinks high in protein or carbohydrate/fat (4,11) or when meals with 10% and 25% protein contents were compared (12). Differences in the absorption of nutrients in liquids (11) and relatively low protein concentrations used in previous studies (12) could explain these differences. The similar response in ghrelin after high protein/carbohydrate/fat found in our study has been documented previously (4).
Postprandial changes in gut hormone secretion reflect the ability of dietary proteins to directly stimulate enteroendocrine cells after being hydrolyzed to peptides and amino acids. Proposed mechanisms have all addressed the fact that nutrients trigger enteroendocrine cell excitability and hence hormone release [reviewed by (13)]. Glutamine was recently found to increase intracellular calcium and cAMP in L cells ex vivo revealing a pathway for GLP-1 secretion (14). The unravelling of further mechanisms by which various proteins or their constituent amino acids stimulate gut hormone secretion will improve understanding of the differential response to the protein/fat and carbohydrate content of meals.
In contrast to previous studies [reviewed by (15)], we did not find that a high protein meal reduced subsequent food intake. Timing, amount of protein and the volume of food ingested have been found to be key factors in studying satiation, satiety and subsequent food intake. Very few studies have tailored the amount of the meal/preload to daily energy requirements as we did; this may account for some differences in the outcome of studies (15).
Gut hormones signal satiety via their paracrine actions on vagal afferents which project to the brainstem and hypothalamus and via endocrine mechanisms directly modulating activity in brain areas associated with reward and learning (16). The lack of correlation between changes in gut hormone levels and subsequent food intake illustrates that food intake is mediated by many factors including central pathways which may modulate the response to peripheral gut-derived signals and nutrient availability.
Funding agencies
This work was supported by The Wellcome Trust, MRC Center for Obesity and Related Metabolic Disease and the National Institute of Health Research Cambridge Biomedical Research Center and the Bernard Wolfe Health Neuroscience Fund.
Footnotes
Disclosure: The authors declared no conflict of interest
References
- 1.Cummings DE, Overduin J. Gastrointestinal regulation of food intake. J Clin Invest. 2007;117:13–23. doi: 10.1172/JCI30227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lomenick JP, Melguizo MS, Mitchell SL, et al. Effects of meals high in carbohydrate, protein, and fat on ghrelin and peptide YY secretion in prepubertal children. J Clin Endocrinol Metab. 2009;94:4463–4471. doi: 10.1210/jc.2009-0949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Foster-Schubert KE, Overduin J, Prudom CE, et al. Acyl and total ghrelin are suppressed strongly by ingested proteins, weakly by lipids, and biphasically by carbohydrates. J Clin Endocrinol Metab. 2008;93:1971–1979. doi: 10.1210/jc.2007-2289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Batterham RL, Heffron H, Kapoor S, et al. Critical role for peptide YY in protein-mediated satiation and body-weight regulation. Cell Metab. 2006;4:223–233. doi: 10.1016/j.cmet.2006.08.001. [DOI] [PubMed] [Google Scholar]
- 5.Schofield WN. Predicting basal metabolic rate, new standards and review of previous work. Hum Nutr Clin Nutr. 1985;39(Suppl 1):5–41. [PubMed] [Google Scholar]
- 6.Rolls BJ, Bell EA, Thorwart ML. Water incorporated into a food but not served with a food decreases energy intake in lean women. Am J Clin Nutr. 1999;70:448–455. doi: 10.1093/ajcn/70.4.448. [DOI] [PubMed] [Google Scholar]
- 7.Adrian TE, Ferri GL, Bacarese-Hamilton AJ, Fuessl HS, Polak JM, Bloom SR. Human distribution and release of a putative new gut hormone, peptide YY. Gastroenterology. 1985;89:1070–1077. doi: 10.1016/0016-5085(85)90211-2. [DOI] [PubMed] [Google Scholar]
- 8.Kreymann B, Williams G, Ghatei MA, et al. Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet. 1987;2:1300–1304. doi: 10.1016/s0140-6736(87)91194-9. [DOI] [PubMed] [Google Scholar]
- 9.Patterson M, Murphey KG, le Roux CW, et al. Characterization of ghrelin-like immunoreactivity in human plasma. J Clin Endocrinol Metab. 2005;90:2205–2211. doi: 10.1210/jc.2004-1641. [DOI] [PubMed] [Google Scholar]
- 10.Crum AJ, Corbin WR, Brownell KD, et al. Mind over milkshakes: mindsets, not just nutrients, determine ghrelin response. Health Psychol. 2011;30:424–429. doi: 10.1037/a0023467. discussion 30–31. [DOI] [PubMed] [Google Scholar]
- 11.Carr RD, Larsen MO, Winzell MS, et al. Incretin and islet hormonal responses to fat and protein ingestion in healthy men. Am J Physiol Endocrinol Metab. 2008;295:E779–E784. doi: 10.1152/ajpendo.90233.2008. [DOI] [PubMed] [Google Scholar]
- 12.Smeets AJ, Soenen S, Luscombe-Marsh ND, et al. Energy expenditure, satiety, and plasma ghrelin, glucagon-like peptide 1, and peptide tyrosine-tyrosine concentrations following a single high-protein lunch. J Nutr. 2008;138:698–702. doi: 10.1093/jn/138.4.698. [DOI] [PubMed] [Google Scholar]
- 13.Steinert RE, Beglinger C. Nutrient sensing in the gut: interactions between chemosensory cells, visceral afferents and the secretion of satiation peptides. Physiol Behav. 2011;105:62–70. doi: 10.1016/j.physbeh.2011.02.039. [DOI] [PubMed] [Google Scholar]
- 14.Tolhurst G, Zheng Y, Parker HE, et al. Glutamine triggers and potentiates glucagon-like peptide-1 secretion by raising cytosolic Ca2+ and cAMP. Endocrinology. 2011;152:405–413. doi: 10.1210/en.2010-0956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Halton TL, Hu FB. The effects of high protein diets on thermogenesis, satiety and weight loss: a critical review. J Am Coll Nutr. 2004;23:373–385. doi: 10.1080/07315724.2004.10719381. [DOI] [PubMed] [Google Scholar]
- 16.Berthoud HR. Vagal and hormonal gut-brain communication: from satiation to satisfaction. Neurogastroenterol Motil. 2008;20(Suppl 1):64–72. doi: 10.1111/j.1365-2982.2008.01104.x. [DOI] [PMC free article] [PubMed] [Google Scholar]