Abstract
Iron impregnated activated carbon has been used as a new adsorbent for the adsorptive removal of phenol from waste water. Impregnation of iron was confirmed by Fourier transform infrared spectroscopy and scanning electron microscope and energy dispersive spectroscopy. Different parameters affecting the adsorption capacity of Iron impregnated activated carbon such as Iron impregnated activated carbon dosage, contact time, pH of solution, initial concentration of phenol and agitation speed were optimized. The residual concentration of phenol was determined by UV-Vis spectroscopy. Maximum adsorption efficiency was calculated 98.5% at optimized parameters: concentration of phenol 25 mg L−1, Iron impregnated activated carbon dose 75 mg, pH 7.0 and agitation time 90 min. The experimental data was fitted to different adsorption isotherms and adsorption capacities obtained were 20 and 15 mg g−1, respectively. Adsorption energy was found to be 1.54 kJ mol−1 which predicts that phenol was adsorbed onto the Iron impregnated activated carbon through physisorption.
Keywords: Organic chemistry, Chemical engineering, Analytical chemistry, Activated carbon, Iron impregnated, Phenol removal, Waste water treatment, Sorption, Physisorption
1. Introduction
Environmental pollution has been a real problem for the living creatures. It has increased significantly in the past few years and reached to alarming levels in terms of health effects on animals and plants. Ground water pollution has been a serious threat, caused by industrial effluents containing dissolved polycyclic aromatic hydrocarbons (PAHs) and phenols. Phenols are naturally as well as artificially existing compounds. They are found in many industrial effluents, including cooking operations, pharmaceuticals, coal processing, refineries, plastics, wood products, as well as pesticide, paper and paint industries [1, 2]. The US Environmental Protection Agency (EPA) Regulations calls for decreasing phenol content in the effluents to less than 1 mg L-1 [3]. Drinking such contaminated water results in tissue erosion, protein degeneration, and paralysis of the central nervous system and also damages the kidney, liver and pancreas of human body [4]. Water pollution damages the aesthetic nature of water, interference with the process of photosynthesis, destruction of the food web existing in water ecosystem [5].
Therefore, to get the environmental and ecological safety, it is recommended to clean the municipal and industrial wastewater from organic micro pollutants, like phenol to the level excluding their negative impact on the natural environment and surroundings.
Many techniques have been applied for the remediation of phenol from wastewater, such as electrochemical oxidation [6], adsorption by carbon fibers or activated carbon [7, 8, 9, 10], MWCNT [11, 12, 13], waste materials [14], wet air oxidation [15, 16], chemical coagulation [17], solvent extraction [18], membrane separation [19, 20], bioremediation [21, 22] and photo catalytic degradation [23, 24]. The process of bioremediation has also been used for phenol and low molecular weight (PAHs) but it has shown the limited approach for degradation of (PAHs) due to their high molecular weight19. For the minerlization/transformation of these contaminants, another alternate process used is ultraviolet radiation, but it has drawback of being expensive, particularly on large scale [25]. Many alternate techniques (physical and chemical) have been applied for the treatment of phenol contaminated water including venting, using solvents and surfactants. The disadvantage of these techniques is that high concentrations of solvent are required for getting good results [26].
Adsorption is favored by its efficiency and universally applicable [26], for the remediation of organic and inorganic compounds, even at low concentration. Adsorption has advantage of its relative ease of operation both in batch and continuous operation, regeneration and reusability of adsorbent [27]. The investigation of effluent treatment containing carbolic compounds, have revealed that uptake on activated carbon by adsorption is considered as a most potential treatment technique [28, 29, 30]. The surface properties of activated carbon, i.e. the wide range of porosity and high surface area, ease of separation, cheap operational cost and remarkable adsorption affinity make AC (activated carbon) a versatile and favored material for various applications [31, 32]. The removal of phenol using raw activated carbon is reported by many researchers, while the modified activated carbon for the phenolic compounds remediation is comparatively less reported in the literature [33, 34, 35, 36].
To enhance the efficiency of activated carbon, investigation on the modification and the reusability of AC has been carried out [37]. Therefore, we have focused in our present work on the surface modification of activated carbon to enhance its adsorption affinity towards phenol. The surface was modified by using NaOH followed by the impregnation with iron precursor to get Fe-AC.
2. Experimental
2.1. Equipments
Analysis of phenol was carried out by UV-visible spectrophotometer (Cary 60 UV-Vis, Agilent Technologies), FTIR spectra were recorded by FTIR spectrometer (Perkin Elmer Spectrum BX automatic FTIR spectrometer at 4cm−1 resolution). Shaking was carried out using (Magnetic stirrer, MR Hei – Tec). For pH measurements (Seven Compact pH S210) was used.
2.2. Reagents and solutions
All the chemicals used were of Analytical grade and double distilled water was used. AC was purchased from (Shanxi Sincere Industrial Co, LTD, China, mesh size 12), Ferric chloride (FeCl3. 6H2O) (Merck), sodium hydroxide (NaOH) (Sigma- Aldrich) were used. Standard solutions of phenol (Merck) were prepared by dissolving appropriate amount in double distilled water. The pH of solutions were maintained using 0.1 M HCl, sodium acetate buffer 3,4,5,6, 0.5 M ammonia and bicarbonate buffer were used.
2.3. Synthesis of adsorbent
The AC was heated for 1h at 100 °C in an oven to get the free moisture AC. For modification, 5g of AC were added in 0.5 M NaOH and mixture was stirred for 30 min at 200 rpm, then the mixture was filtered, washed with distilled water. The activated carbon treated in alkaline solutions shows great affinity the sorption of organic species (like phenol) from aqueous system. Alkaline treated AC was transferred to 1 M FeCl3. 6H2O, and stirred for 6 h for the uptake of iron on the surface of the AC. Finally, the mixture was filtered and washed with distilled water and dried for 6 h in an oven at 110 °C.
2.4. Adsorption procedure
The efficiency of modified AC was investigated for the removal of phenol by standard batch procedure. Batch experiments were carried out by taking adsorbent dose (10–200 mg) and concentration of phenol (10–100 mg L-1) in 20 mL solution and the pH was optimized from 2–10. The solution and adsorbent were allowed to agitate from 10–180 min at 100 rpm for the uptake of phenol ions. All the experiments were carried out at room temperature (25 °C). Removal efficiency of FAC surface was calculated using Eq. (1):
| (1) |
Where Co and Ce are the initial and equilibrium (mg L−1) of phenol respectively. The equation used to determine the phenol amount adsorbed at equilibrium per unit mass of adsorbent (phenol adsorption capacity) is given below:
| (2) |
Where qe is the phenol uptake at equilibrium or adsorption capacity (mg of phenol g−1 of the Sorbent) and V is the volume of phenol solution and m is the amount of Fe-AC.
3. Results and discussion
3.1. Characterization study
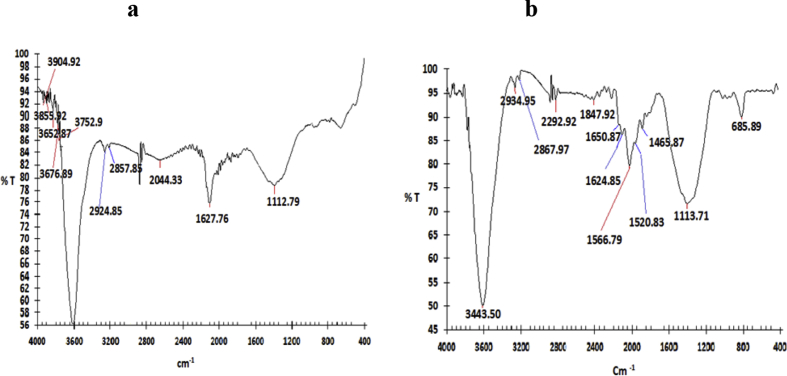
The modified AC was characterized by SEM and EDS analysis. EDS is used to determine the elements on sample surface, combined with the observation under SEM. Fig. 1 shows the presence of iron on the surface of AC by SEM and EDS analysis. Fig. 2a shows the spectrum of Fe–AC, an informative peak appeared at 1112.79 cm−1 attributed to the presence of iron on the surface.
Fig. 1.
Scanning electron microscope and energy dispersive spectroscopy images for Fe-AC.
Fig. 2.
(a) FTIR spectrum for Fe-AC (b) FTIR spectrum for phenol incorporated on Fe-AC.
Fig. 2b confirms the adsorption of phenol on the surface of Fe–AC. The peak at 1566 cm−1 indicated the C = C aromatic ring stretching vibration [38] while the peak at 685 cm−1 is attributed to OH out of the plane bending [39] and confirmed the adsorption of phenol onto the Fe–AC.
3.2. Adsorption studies
3.2.1. Effect of contact time
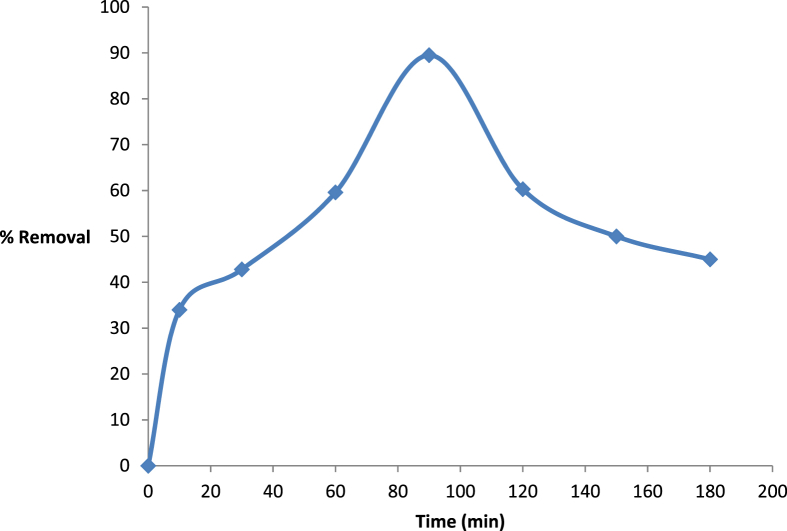
In order to achieve the maximum uptake of phenol onto Fe-AC, experiments were carried out at variable time intervals from 0 to 180 min. All other parameters were kept constant. The increase in phenol removal efficiency was observed till 90 min where maximum sorption was attained, as shown in Fig. 3. This can be explained from the equilibrium adsorption point at which the rate of adsorption was higher than desorption, at which the optimum sorption was achieved. After 90 min a clear desorption was observed due to the saturation of the active sites on surface of Fe-AC.
Fig. 3.
Effect of shaking time on removal of phenol using Fe-AC at room temperature.
The enhanced adsorption of metal with an increase in agitation time may also be due to the decrease in boundary layer resistance in the kinetic energy of the hydrate layer [40]. An increase in adsorption of phenol ions with an increase in contact time may also be due to the decrease in boundary layer resistance of the hydrate layer.
3.2.2. Effect of pH on the phenol removal
The pH of solution has a critical role in the removal of phenol from a water system, because it can affect the charge of adsorbent surface. The removal of phenol by modified AC was evaluated in the pH range from 2–10, while initial concentration of phenol ions, contact time, adsorbent amount, shaking speed were kept constant at 25 mg L-1, 90 min, 75 mg, 200 rpm respectively. The maximum removal efficiency of phenol was recorded at pH 7, as shown in Fig. 4. However, the adsorption of phenol was decreased with the pH greater than 7.
Fig. 4.
Effect of pH on the removal of phenol using Fe-AC at room temperature.
Mechanism can be elaborated on the basis of ionic chemistry of solution and surface charge of Fe-AC. Phenol adsorbs to a lesser extent at higher pH because of its week acidic nature (pKa = 10). The repulsive forces between negatively charged surface of AC and phenol may be responsible for the decrease in sorption at higher pH. While up to the pH 7, the electrostatic interaction occurs between the positive ions on the surface and phenolate ions and increases the sorption.
3.2.3. Effect of concentration
The ionic concentration of phenol was optimized from 10–100 mg L −1 to check the removal efficiency on Fe-AC, while all other parameters were kept constant. The maximum uptake of phenol observed was at lower concentration shown in Fig. 5, while removal decreased as the initial concentration increased. This can be explained as the high amount of phenol ion with limited active adsorbed sites on the Fe-AC surface, which leading to increase the concentration of phenol ion in the bulk solution and less favourable sites are involved for the uptake of phenol ions and thus decreasing the removal capacity. At certain concentration the adsorption sites of Fe-AC surface are equilibrated with the phenol ions, so further increase in the concentration, results in the decrease in uptake on the surface, because all of the sites are occupied.
Fig. 5.
Effect of conc. on the removal of phenol using Fe-AC at room temperature.
3.2.4. Effect of adsorbent dosage
The adsorbent is said to be more efficient, if its smaller amount gives maximum uptake of the desired analyte. The effect of the modified AC dose was studied from 10–200 mg for the removal of phenol from the water system at pH 7, Concentration 25 mg L-1, shaking time 90 min and at 200 rpm Fig. 6 shows that removal was increased from 10–75 mg due to the increased adsorbent surface area and availability of more vacant surfaced sites. However, slightly decrease in the adsorption was investigated after 75 mg. This reduction in per unit mass of Fe-AC surface can be elaborated on the basis of adsorbent to adsorbate ratio, for the specific solute concentration, adsorption capacity changes depending on the binding sites present on the surface.
Fig. 6.
Effect of adsorbent dose on the removal of phenol using Fe-AC at room temperature.
At 75 mg of Fe-AC the maximum number of phenol ions occupies the available sites describing the higher per unit mass adsorbent value. However, for more than 75 mg the number of active sites were increased for the same initial concentration of phenol solution, so few active sites on FAC surface remained unoccupied showing the decreased per unit weight sorbent value. Therefore 75 mg was selected optimum dose for the study.
3.2.5. Effect of shaking speed
Shaking provides proper interactions between the molecules in solutions and adsorbent binding sites. Effect of shaking speed on the removal of phenol was studied from 100–300 rpm. Fig. 7 shows that the maximum uptake of phenol ions increased up to 200 rpm, after 200 it seemed constant. This may be due to the rapid transfer of phenol ion towards modifying AC surface with the least opposing force to resist at higher shaking speed. Therefore 200 rpm was selected for subsequent study.
Fig. 7.
Effect of shaking speed on the removal of phenol using Fe-AC at room temperature.
3.2.6. Isotherm study (linear method)
The adsorption of phenol was examined by changing initial concentration of phenol ions from 10–100 mg L-1 using 75 mg Fe-AC/20 mL of phenol solution and 90 min contact time at 200 rpm. The uptake of phenol ions was maximum at 25 mg L-1. The effect of phenol ions concentration on Fe-AC was also examined in terms of following Eqs. (3), (4), and (5):
| (3) |
| (4) |
and
| Dubinin–Radushkevich Isotherms: ln Cads = KD - R – β ε2 | (5) |
where Cads is the quantity of phenol ions captured unit mass of FAC and Ce is the amount of phenol ions in the residual solution at equilibrium. Q, b, A, n, Xm, and β are the Langmuir, Freundlich and D–R constants, respectively [41]. Table 1 shows the all three isotherm constants calculated from their slopes and intercepts of linear plots. Separation factor is the important characteristic of the Langmuir isotherm, determined by using Eq. (6)
| (6) |
where Ci is the concentration of phenol ions before adsorption, monolayer sorption model can be explained by a dimensionless constant RL [42] to be irreversible (RL = should be 0), favorable (should lie within 0 and 1), linear (RL = should be greater than 1) or unfavorable (RL > 1). So, this method presents a favorable sorption for the phenol ions with the RL values 0.126–0.631. Fig. 8 shows the graphic presentation of Langmuir isotherm. The value of 1/n is less than 1, showing that the sorption capacity is only little decreased at lower equilibrium concentrations. The D-R plot was used to calculate the energy for the phenol adsorption system onto Fe-AC using Eq. (7). The energy was found in the range of 1–8 kJ mol -1, which predicts that the uptake of phenol ions onto Fe-AC was physical adsorption [43].
| (7) |
Table 1.
Sorption parameters of phenol onto Fe-AC.
| Langmuir | Freundlich | D-R | ||||||
|---|---|---|---|---|---|---|---|---|
| Q (mg/g) | b (1mol−1) | r | A (mg/g) | 1/n | r | Xm (mg/g | E (KJ/mol | r |
| 20 | 5 × 102 | 0.994 | 15 | 1.021 | 0.948 | 8.75 | 1.543 | 0.994 |
Fig. 8.
Langmuir isotherm for the phenol on Fe-AC.
3.2.7. Non-linear method
Adsorption of phenol onto Fe-AC was also investigated by applying nonlinear form of Langmuir and Freundlich isotherms to determine the sorption parameters and best fit correlation by utilizing the solver add-in with Microsoft Excel. The sorption capacity of Langmuir and Freundlich isotherms were calculated 47.8 mg g-1 and 6.23 mg g-1 respectively. Both the isotherms seemed best fit to the adsorption data with the correlation coefficient were found 0.99 and 0.97 for Langmuir and Freundlich isotherm respectively.
3.3. Kinetics of adsorption
Different kinetic equations like pseudo first order, pseudo second order and Morris–Weber were applied to study the adsorption behavior of phenol on FAC. Intra particle diffusion model was used to study the diffusion mechanism.
3.3.1. Pseudo first order kinetic model
In order to test the validation of data pseudo first order model was applied using following Eq. (8) [44]:
| (8) |
Where qe is the quantity of phenol uptake at equilibrium (mg g −1), qt is the quantity of phenol uptake at time t (mg g −1), K1 is the rate constant (min −1) for the plot of log (qe–qt) against t. By plotting the log (qe–qt) against t straight line was obtained. The slope and intercept values were used to calculate logqe and K1 for the adsorption of phenol, respectively. The calculated results are shown in Table 2.
Table 2.
Pseudo first, pseudo-second order model, Morris Weber (Intra-particle Diffusion) equation constants and correlation coefficients for adsorption of Phenol on Fe-AC at 25 °C.
| Pseudo first order | Pseudo 2nd order | Moris weber |
|---|---|---|
| K (min−1) = 0.008 | K (g/mg)min = 0.135 | Kpi (mg/g min1/2) = .551 |
| qt (mg/g) = 2.06 | qt (mg/g) = 4.7 | C = 0.920 |
| r = 0.95 | r = 0.98 | r = 0.94 |
3.3.2. Pseudo second order kinetic model
For the equilibrium adsorption pseudo second-order rate equation [45] was applied following Eq. (9):
| (9) |
Where K2 (g/mg min) is the second-order rate constant, Slope (1/qe) and intercept (1/K2qe 2) were determined from the linear plot of t/qt versus t. Fig. 9 shows best fit between the predicted and original qe values and comparable to pseudo first order model. Calculated parameters are shown in Table 2.
Fig. 9.
Pseudo 2nd order plot for the phenol on Fe-AC.
3.3.3. Moris weber equation
The graph was plotted between the adsorbed phenol concentrations, qt (mol g −1) at time t against to check best fitting of Moris Weber equation [8] in the following Eq. (10).
| (10) |
Where qt is phenol uptake at time t, kpi is the rate constant of intraparticle diffusion and C is a constant used to estimate the thickness of boundary layer. The thickness of boundary layer can be predicted from intercept value. The higher the intercept value, the greater is the thickness of boundary layer. The plotted graph between the adsorbed concentration of phenol and t results in a straight line which did not proceed through origin, so it can be predicted that intra particle diffusion was not the rate limiting step during the uptake of phenol ions, it may be governed through other mechanisms also involved in the rate determining step [46].
3.3.4. Thermodynamics of adsorption
The uptake of phenol onto the FAC was studied by changing the temperature from 25–50 °C at optimum conditions to evaluate the temperature effect using following Eqs. (11) and (12):
| (11) |
| (12) |
Where Kc is the equilibrium constant = Fe/(1−Fe), Fe is the part of phenol ion adsorbed at equilibrium, ΔH, ΔS, ΔG, and T the enthalpy, entropy, Gibbs free energy, and absolute temperature, respectively and R is the gas constant.
From the intercept and slope of plots the values of ΔH = -5.084889 kJ mol-1, ΔS = 0.00172 J mol−1K−1 and ΔG = -4.50 to -4.23 kJ mol-1 has been estimated for the phenol ions respectively. The exothermic nature of adsorption was evaluated from inverse value of ΔH, however negative value of ΔG predicted the adsorption process spontaneous and favorable [47].
3.4. Comparison with reported methods
The sorption capacity of phenol on modified activated carbon was compared with other adsorbents reported previously are given in Table 3. Adsorption capacity of modified AC is comparable with the most of the adsorbents; whereas the modification of AC is cheaper and simple.
Table 3.
Comparison of adsorption capacities of various adsorbents for the phenol.
| Adsorbent | Monolayer adsorption |
References |
|---|---|---|
| capacity (mg/g) | ||
| MWCNTs | 15.9 | [46] |
| PHEMA microbead modified Cibacron blue | 8.3 | [47] |
| PHEMA microbead modified alkali blue 6B | 13.6 | [48] |
| Samla coal | 13.3 | [49] |
| Natural coal | 18.8 | [50] |
| Activated coal | 1.48 | [51] |
| Activated carbon | 1.5106 | [52] |
| AC–TiO2 | 3.153 | [52] |
| Modified Activated Carbon | 20 | Present work |
3.5. Method validation
The developed method was applied onto the real spiked samples to check the applicability of current method. The samples were collected from industrial waste water Isparta, Turkey. The removal of phenol from industrial waste water was achieved about 98 % as shown in Table 4. These results are promising performance of Fe-AC for removing phenol from aqueous solution.
Table 4.
Removal of phenol from real environmental samples.
| Samples | Found (mg L −1) | Added (mg L −1) | Removal (%) |
|---|---|---|---|
| 1* | 45 | 0 | 97.8 |
| 2* | 45 | 10 | 98.5 |
| 3** | 0 | 20 | 98 |
Sample 1, 2*: Industrial waste water, Isparta Turkey.
Sample 3**: Tape water, Suleyman Demirel University.
4. Conclusions
Modification of AC is simple, economical and effective to treat the water samples containing phenol. Impregnation of iron enhanced the sorption capacity of AC towards the phenolic compounds. Under optimized parameters nearly quantitative adsorption is obtained for phenol on Fe-AC. Results showed that Fe-AC had a rapid rate to reach at equilibrium point with higher uptake ability for phenol. The adsorption isotherms of phenol have been well fitted by all three isotherms namely Langmuir, freundlich and D-R with the correlation coefficient values 0.99, 0.95 and 0.99 respectively. The nature of adsorption was evaluated spontaneous and exothermic reaction was predicted from negative values of Gibbs energy and enthalpy respectively. The second order kinetic model seemed best fit to the adsorption data with the correlation coefficient 0.98. Method was successfully applied for the real environmental samples and it can be concluded that Fe-AC is a new and cheap adsorbent to treat the phenol laden waste water.
Declarations
Author contribution statement
Abdul M. Channa, Sıtkı Baytak, Saima Q. Memon, Muhammad Y. Talpur: Conceived and designed the experiments; Performed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interest statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
References
- 1.Damjanović L., Rakić V., Rac V., Stošić D., Auroux A. The investigation of phenol removal from aqueous solutions by zeolites as solid adsorbents. J. Hazard Mater. 2010;184:477–484. doi: 10.1016/j.jhazmat.2010.08.059. [DOI] [PubMed] [Google Scholar]
- 2.Lin S.H., Juang R.S. Adsorption of phenol and its derivatives from water using synthetic resins and low-cost natural adsorbents: a review. J. Environ. Manag. 2009;90:1336–1349. doi: 10.1016/j.jenvman.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 3.Banat F.A., Al-Bashir B., Al-Asheh S., Hayajneh O. Adsorption of phenol by bentonite. Environ. Pollut. 2000;107:391–398. doi: 10.1016/s0269-7491(99)00173-6. [DOI] [PubMed] [Google Scholar]
- 4.Tiwari D., Kim W., Kim M., Prasad S.K., Lee S.M. Organo-modified sericite in the remediation of phenol-contaminated waters. Desalin. Wat. Treat. 2015;53:446–451. [Google Scholar]
- 5.Mittal A., Kaur D., Malviya A., Mittal J., Gupta V.K. Adsorption studies on the removal of coloring agent phenol red from wastewater using waste materials as adsorbents. J. Colloid Interface Sci. 2009;337:345–354. doi: 10.1016/j.jcis.2009.05.016. [DOI] [PubMed] [Google Scholar]
- 6.Esplugas S., Giménez J., Contreras S., Pascual E., Rodríguez M. Comparison of different advanced oxidation processes for phenol degradation. Water Res. 2002;36:1034–1042. doi: 10.1016/s0043-1354(01)00301-3. [DOI] [PubMed] [Google Scholar]
- 7.Liu Q.S., Zheng T., Wang P., Jiang J.P., Li N. Adsorption isotherm, kinetic and mechanism studies of some substituted phenols on activated carbon fibers. Chem. Eng. J. 2010;157:348–356. [Google Scholar]
- 8.Gupta V.K., Jain R., Mittal A., Saleh T.A., Nayak A., Agarwal S., Sikarwar S. Photo-catalytic degradation of toxic dye amaranth on TiO2/UV in aqueous suspensions. Mater. Sci. Eng. C. 2012;32:12–17. doi: 10.1016/j.msec.2011.08.018. [DOI] [PubMed] [Google Scholar]
- 9.Gupta V.K., Nayak A. Cadmium removal and recovery from aqueous solutions by novel adsorbents prepared from orange peel and Fe2O3 nanoparticles. Chem. Eng. J. 2012;180:81–90. [Google Scholar]
- 10.Saleh T.A., Gupta V.K. Column with CNT/magnesium oxide composite for lead (II) removal from water. Environ. Sci. Pollut. Res. Int. 2012;19 doi: 10.1007/s11356-011-0670-6. 1224-122. [DOI] [PubMed] [Google Scholar]
- 11.Fortuny A., Font J., Fabregat A. Wet air oxidation of phenol using active carbon as catalyst. Appl. Catal. B Environ. 1998;19:165–173. [Google Scholar]
- 12.Saleh T.A., Gupta V.K. Photo-catalyzed degradation of hazardous dye methyl orange by use of a composite catalyst consisting of multi-walled carbon nanotubes and titanium dioxide. J. Colloid Interface Sci. 2012;371:101–106. doi: 10.1016/j.jcis.2011.12.038. [DOI] [PubMed] [Google Scholar]
- 13.Gupta V.K., Kumar R., Nayak A., Saleh T.A., Barakat M.A. Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: a review. Adv. Colloid Interface Sci. 2013;193–194:24–34. doi: 10.1016/j.cis.2013.03.003. [DOI] [PubMed] [Google Scholar]
- 14.Mittal A., Mittal J., Malviya A., Gupta V.K. Adsorptive removal of hazardous anionic dye Congo red" from wastewater using waste materials and recovery by desorption. J. Colloid Interface Sci. 2009;340:16–26. doi: 10.1016/j.jcis.2009.08.019. [DOI] [PubMed] [Google Scholar]
- 15.Lin S.H., Wu Y.F. Catalytic wet air oxidation of phenolic wastewaters. Environ. Technol. 1996;17:175–181. [Google Scholar]
- 16.Mozia S., Tomaszewska M. Treatment of surface water using hybrid processes – adsorption on PAC and ultrafiltration. Desalination. 2004;162:23–31. [Google Scholar]
- 17.Lazarova Z., Boyadzhieva S. Treatment of phenol-containing aqueous solutions by membrane-based solvent extraction in coupled ultrafiltration modules. Chem. Eng. J. 2004;100:129–138. [Google Scholar]
- 18.Kujawski W., Warszawski A., Ratajczak W., Porębski T., Capała W., Ostrowska I. Removal of phenol from wastewater by different separation techniques. Desalination. 2004;163:287–296. [Google Scholar]
- 19.Rzeszutek K., Chow A. Extraction of phenols using polyurethane membrane. Talanta. 1998;46:507–519. doi: 10.1016/s0039-9140(97)00273-7. [DOI] [PubMed] [Google Scholar]
- 20.Alderman N.S., Guessan A.L.N., Nyman M.C. Effective treatment of PAH contaminated Superfund site soil with the peroxy-acid process. J. Hazard Mater. 2007;146:652–660. doi: 10.1016/j.jhazmat.2007.04.068. [DOI] [PubMed] [Google Scholar]
- 21.Annachhatre A.P., Gheewala S.H. Biodegradation of chlorinated phenolic compounds. Biotechnol. Adv. 1996;14:35–56. doi: 10.1016/0734-9750(96)00002-x. [DOI] [PubMed] [Google Scholar]
- 22.Sano N., Yamamoto T., Yamamoto D., Kim S.I., Eiad-Ua A., Shinomiya H., Nakaiwa M. Degradation of aqueous phenol by simultaneous use of ozone with silica-gel and zeolite. Chem. Eng. Process. 2007;46:513–519. [Google Scholar]
- 23.Guo Z., Ma R., Li G. Degradation of phenol by nanomaterial TiO2 in wastewater. Chem. Eng. J. 2006;119:55–59. [Google Scholar]
- 24.Saleh T.A., Gupta V.K. Carbonaceous adsorbent prepared from waste tires: Experimental and computational evaluations of organic dye methyl orange. J. Mol. Liq. 2014;191:85–91. [Google Scholar]
- 25.Jain A.K., Gupta V.K., Bhatnagar A., Suhas S. A comparative study of adsorbents prepared from industrial wastes for removal of dyes. Separ. Sci. Technol. 2003;38:463–481. [Google Scholar]
- 26.Kennedy L.J., Judith V.J., Kayalvizhi K., Sekaran G. Adsorption of phenol from aqueous solutions using mesoporous carbon prepared by two-stage process. Chem. Eng. J. 2007;132:279–287. [Google Scholar]
- 27.Cherifi H., Hanini S., Bentahar F. Adsorption of phenol from wastewater using vegetal cords as a new adsorbent. Desalination. 2009;244:177–187. [Google Scholar]
- 28.Tao J., Huo P., Fu Z., Zhang J., Yang Z., Zhang D. Characterization and phenol adsorption performance of activated carbon prepared from tea residue by NaOH activation. Environ. Technol. 2019;40:171–181. doi: 10.1080/09593330.2017.1384069. [DOI] [PubMed] [Google Scholar]
- 29.Yang G.X., Jiang H. Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res. 2014;48:396–405. doi: 10.1016/j.watres.2013.09.050. [DOI] [PubMed] [Google Scholar]
- 30.Huang W., Zhang Y.M., Bao S.X., Cruz R., Song S.X. Desalination by capacitive deionization process using nitric acid-modified activated carbon as the electrodes. Desalination. 2014;340:67–72. [Google Scholar]
- 31.García Blanco A.A., Alexandre de Oliveira J.C., López R., Moreno-Piraján J.C., Giraldo L., Zgrablich G., Sapag K. A study of the pore size distribution for activated carbon monoliths and their relationship with the storage of methane and hydrogen. Colloids Surf A Physicochem Eng Asp. 2010;357:74–83. [Google Scholar]
- 32.Stavropoulos G.G., Samaras P., Sakellaropoulos P.G. Effect of activated carbons modification on porosity, surface structure and phenol adsorption. J. Hazard Mater. 2008;151:414–421. doi: 10.1016/j.jhazmat.2007.06.005. [DOI] [PubMed] [Google Scholar]
- 33.Dabrowski A., Podkoscielny P., Hubicki Z., Barczak M. Adsorption of phenolic compounds by activated carbon—a critical review. Chemosphere. 2008;58:1049–1070. doi: 10.1016/j.chemosphere.2004.09.067. [DOI] [PubMed] [Google Scholar]
- 34.Villacañas F., Pereira M.F.R., Órfão J.J.M., Figueiredo J.L. Adsorption of simple aromatic compounds on activated carbons. J. Colloid Interface Sci. 2006;293:128–136. doi: 10.1016/j.jcis.2005.06.032. [DOI] [PubMed] [Google Scholar]
- 35.Zelmanov G., Semiat R. Boron removal from water and its recovery using iron (Fe+3) oxide/hydroxide-based nanoparticles (NanoFe) and NanoFe-impregnated granular activated carbon as adsorbent. Desalination. 2014;333:107–117. [Google Scholar]
- 36.Mohd-Din A.T., Hameed B.H., Ahmad A.L. Batch adsorption of phenol onto physiochemical-activated coconut shell. J. Hazard Mater. 2009;161:1522–1529. doi: 10.1016/j.jhazmat.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 37.Reusch W. 2013, May. https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/Spectrpy/InfraRed/infrared.htm
- 38.Bhattacharyya K., Gupta S. Pb (II) uptake by kaolinote and montmorillnite in aqueous medium: influence of acid activation of the clays. Colloids Surf. A Physicochem. Eng. Asp. 2007;277:191–200. [Google Scholar]
- 39.Kadirvelu K., Thamaraiselvi K., Namasivayam C. Adsorption of nickel (II) from aqueous solution onto activated carbon prepared from coirpith. Separ. Purif. Technol. 2001;24:497–505. [Google Scholar]
- 40.Megat Hanafiah M.A.K., Yahya M.Z.A., Zakaria H., Ibrahim S.C. Adsorption of Cd(II) ions from aqueous solutions by lalang (Imperata cylindrica) leaf powder: Effect of physicochemical environment. J. Appl. Sci. 2007;7:489–493. [Google Scholar]
- 41.Saeed M.M. Adsorption profile and thermodynamic parameters of the preconcentration of Eu(III) on 2-thenoyltrifluoroacetone loaded polyurethane (PUR) foam. J. Radioanal. Nucl. Chem. 2003;256:73–80. [Google Scholar]
- 42.Gupta V.K., Jain R., Nayak A., Agarwal S., Shrivastava M. Removal of the hazardous dye-Tartrazine by photodegradation on titanium dioxide surface. Mater. Sci. Eng. C. 2011;31:1062–1067. [Google Scholar]
- 43.Li J., Shi Y., Cai Y., Moua S., Jiang G. Adsorption of di-ethylphthalate from aqueous solutions with surfactant-coated nano/microsized alumina. Chem. Eng. J. 2008;140:214–220. [Google Scholar]
- 44.Koyuncu H., RızaKul A. Removal of aniline from aqueous solution by activated kaolinite: kinetic, equilibrium and thermodynamic studies. Colloids Surf A Physicochem Eng Asp. 2019;569:59–66. [Google Scholar]
- 45.Tan I.A.W., Ahmad A.L., H Hameed B. Adsorption isotherms, kinetics, thermodynamics and desorption studies of 2, 4, 6-trichlorophenol on oil palm empty fruit bunch-based activated carbon. J. Hazard Mater. 2009;164:473–482. doi: 10.1016/j.jhazmat.2008.08.025. [DOI] [PubMed] [Google Scholar]
- 46.Liao Q., Sun J., Gao L. The adsorption of resorcinol from water using multi-walled carbon nanotubes. Colloids Surf. A Physicochem. Eng. Asp. 2008;312:160–165. [Google Scholar]
- 47.Denizli A., Ozkan G., Ucar M. Removal of chlorophenols from aquatic systems with dye-affinity microbeads. Separ. Purif. Technol. 2001;24:255–262. [Google Scholar]
- 48.Denizli A., Ozkan G., Ucar M. Dye-affinity microbeads for removal of phenols and nitrophenols from aquatic systems. J. Appl. Polym. Sci. 2002;83:2411–2418. [Google Scholar]
- 49.Ahmaruzzaman M., Sharma D.K. Adsorption of phenols from wastewater. J. Colloid Interface Sci. 2005;287:14–24. doi: 10.1016/j.jcis.2005.01.075. [DOI] [PubMed] [Google Scholar]
- 50.Tarasevich Y.I. Porous structure and adsorption properties of natural porous coal. Colloids Surf. A Physicochem. Eng. Asp. 2001;176:267–272. [Google Scholar]
- 51.Vazquez I., Rodríguez-Iglesias J., Maranon E., Castrillon L., Alvarez M. Removal of residual phenols from coke wastewater by adsorption. J. Hazard Mater. 2007;147:395–400. doi: 10.1016/j.jhazmat.2007.01.019. [DOI] [PubMed] [Google Scholar]
- 52.Abussaud B., Asmaly H.A., Ihsanullah, Saleh T.A., K Gupta V., Iaoui T., Atieh M.A. Sorption of phenol from waters on activated carbon impregnated with iron oxide, aluminum oxide and titanium oxide. J. Mol. Liq. 2015;213:351–359. [Google Scholar]