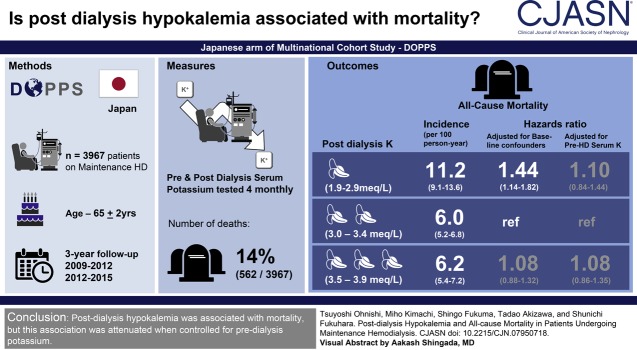
Visual Abstract
Keywords: chronic hemodialysis, hypokalemia, mortality, serum potassium, electrolytes, end-stage renal disease, Japan-Dialysis Outcomes and Practice Patterns Study, post-dialysis, renal dialysis, Potassium, Confidence Intervals, Follow-Up Studies, Dialysis Solutions, Proportional Hazards Models
Abstract
Background and objectives
Almost half of patients on dialysis demonstrate a postdialysis serum potassium ≤3.5 mEq/L. We aimed to examine the relationship between postdialysis potassium levels and all-cause mortality.
Design, setting, patients, & measurements
We conducted a cohort study of 3967 participants on maintenance hemodialysis from the Dialysis Outcomes and Practice Patterns Study in Japan (2009–2012 and 2012–2015). Postdialysis serum potassium was measured repeatedly at 4-month intervals and used as a time-varying variable. We estimated the hazard ratio of all-cause mortality rate using Cox hazard regression models, with and without adjusting for time-varying predialysis serum potassium. Models were adjusted for baseline characteristics and time-varying laboratory parameters. We also analyzed associations of combinations of pre- and postdialysis potassium with mortality.
Results
The age of participants at baseline was 65±12 years (mean±SD), 2552 (64%) were men, and 96% were treated with a dialysate potassium level of 2.0 to <2.5 mEq/L. The median follow-up period was 2.6 (interquartile range, 1.3–2.8) years. During the follow-up period, 562 (14%) of 3967 participants died, and the overall mortality rate was 6.7 per 100 person-years. Compared with postdialysis potassium of 3.0 to <3.5 mEq/L, the hazard ratios of postdialysis hypokalemia (<3.0 mEq/L) were 1.84 (95% confidence interval, 1.44 to 2.34) in the unadjusted model, 1.44 (95% confidence interval, 1.14 to 1.82) in the model without adjusting for predialysis serum potassium, and 1.10 (95% confidence interval, 0.84 to 1.44) in the model adjusted for predialysis serum potassium. The combination of pre- and postdialysis hypokalemia was associated with the highest mortality risk (hazard ratio, 1.72; 95% confidence interval, 1.35 to 2.19, reference; pre- and postdialysis nonhypokalemia).
Conclusions
Postdialysis hypokalemia was associated with mortality, but this association was not independent of predialysis potassium.
Introduction
The mortality rate of patients on hemodialysis is 13.6 per 100 person-years, which is much higher than that of the healthy population (1). The leading cause of death is sudden cardiac death, which is mainly caused by hyperkalemia, and serum potassium has been an area of intense interest in dialysis management (1). During dialysis, serum potassium levels are determined on the basis of the predialysis potassium level (predialysis K), namely, the period during which the otherwise fluctuating levels of serum potassium peak in patients on dialysis (2–5). Hence, predialysis K assessment may assist in the prevention of sudden cardiac death caused by hyperkalemia; however, hypokalemia may be underestimated.
A wide range of dialysate potassium concentrations are used in clinical practice; however, the potassium concentration in dialysate is usually lower than in the serum (2,6–9). Because of the gap between dialysate and serum potassium levels, serum potassium levels drop significantly after hemodialysis, and 45% of patients present with postdialysis hypokalemia of <3.5 mEq/L (10–12). The risk of low potassium dialysate has been reported in several studies and dialysis potassium ought to affect postdialysis serum potassium (postdialysis K) (2,5–7,13,14). Thus, postdialysis hypokalemia may be associated with a higher mortality risk.
Previous observational studies have reported that the risk of mortality was low in patients with predialysis K levels of ≥4.0 to 5.5 mEq/L (2–5). Compared with predialysis K, postdialysis K is rarely investigated, and so its acceptable range remains unknown. Postdialysis K represents the lowest levels of potassium over weekly potassium fluctuations, and we believe that its evaluation would be useful for determining the risk of hypokalemia in patients on dialysis.
We hypothesized that postdialysis K is a clinically important factor associated with mortality. The aim of this study was to examine the relationship between postdialysis K and all-cause mortality, using data from the Dialysis Outcomes and Practice Patterns Study in Japan (J-DOPPS).
Materials and Methods
Data Source
J-DOPPS is the Japanese version of a multinational cohort study, comprising adult patients on in-center hemodialysis. Patients were randomly sampled using the following two-step stratified sampling method. In the first step, facilities were randomly selected from facilities treating at least 26 adult patients on in-center maintenance hemodialysis. In the second step, patients were randomly selected from each facility. The J-DOPPS study started in 1996 and had five phases: phase 1 (1996–2002), phase 2 (2002–2005), phase 3 (2005–2009), phase 4 (2009–2012), and phase 5 (2012–2015). We used data from phases 4 and 5 because postdialysis K assessment started with phase 4. Predialysis K and postdialysis K were measured before and after one dialysis session, and data were collected at 4-month intervals in J-DOPPS. We excluded patients who underwent dialysis for <8 hours per week because serum potassium after short-duration or twice-weekly dialysis may have a different effect on mortality compared with serum potassium levels after thrice-weekly dialysis. We also excluded patients whose postdialysis K levels were not measured at study enrollment. Detailed information on the data elements, methodology, and design has been described elsewhere (15,16). Written informed consent was obtained from all patients. The study protocol was approved by the Kyoto University Institutional Review Board (approval number, R1301).
Definition of Follow-Up and Outcomes
The observational period started at study enrollment and ended at one of the following events, whichever occurred first: death, kidney transplant, loss to follow-up, or the end of the study. The primary outcome was all-cause death during the observational period. All outcomes were ascertained using reports from each patient’s facility.
Statistical Analyses
Patients were divided into groups according to baseline postdialysis K levels: a low group with <3.0 mEq/L, a medium-low group with 3.0 to <3.5 mEq/L, a medium-high group with 3.5 to <4.0 mEq/L, and a high group with ≥4.0 mEq/L. We used Kaplan–Meier curves to determine the time course of each group over the observational period.
To analyze the relationship between postdialysis K and all-cause mortality, we constructed a Cox proportional hazards regression model adjusted for the following factors: patient demographic characteristics (age, sex, and body mass index), predialysis K, dialysis-related factors (dialysis vintage, dialysis treatment time, single-pool Kt/V, type of vascular access, and dialysate bicarbonate), nutritional markers (serum albumin and normalized protein catabolic rate), an inflammatory marker (C-reactive protein), comorbidities (coronary artery disease, chronic heart failure, cerebrovascular disease, diabetes mellitus, hypertension, and cirrhosis), and medications (angiotensin II receptor blockers, angiotensin-converting enzyme inhibitors, and diuretics). We selected these factors on the basis of previous studies and clinical knowledge (10,11,17–20). We used a robust sandwich estimator in the Cox model, and Cox proportional hazard assumptions were confirmed using a log-log plot of survival.
In time-dependent Cox models, we used postdialysis K levels at 4-month intervals as a time-varying variable because the postdialysis K level changed during the observational period. We constructed an unadjusted model, a baseline confounder-adjusted model (model 1), a time-varying confounder-adjusted model without predialysis K (model 2), and a time-varying confounder-adjusted model with predialysis K (model 3). Model 1 adjusted for baseline confounders of patient demographic characteristics, comorbidities, and medications. Model 2 adjusted for baseline confounders plus time-varying covariates of dialysis-related factors, nutritional markers, and an inflammatory marker without predialysis K. Model 3 adjusted for time-varying predialysis K in addition to the confounders included in model 2. For further assessment of the independent and dependent association of postdialysis hypokalemia and mortality, we conducted additional analysis to use the combination of pre- and postdialysis hypokalemia as predictors.
We constructed categorical and nonlinear models to evaluate the effect of postdialysis K on mortality. In the nonlinear model, we used the restricted cubic spline curve to evaluate the relationship between severity of postdialysis hypokalemia and mortality. We used four knots of fifth, 35th, 65th, and 95th percentiles for the restricted cubic spline curve, and we set 3.5 mEq/L as the reference postdialysis K level (21).
In sensitivity analyses, we confirmed the robustness of outcomes, patient criteria, and statistical models. First, we evaluated cause-specific mortality by combining sudden cardiac death and death of unknown cause. Second, we analyzed patients with no exclusion on the basis of treatment duration. Third, we only analyzed patients who received thrice-weekly dialysis of at least 4 hours’ duration. Further, we constructed a logistic model to confirm the robustness of the statistical model. Finally, to confirm data correctness, we analyzed data with exclusion of postdialysis K, which was higher than predialysis K. To exclude any possibility of bias caused by time-varying confounders, we also confirmed results using generalized estimating equations. The detailed data structure and methods are described in Supplemental Appendix 1 (22).
We performed a subgroup analysis on the basis of baseline predialysis K level to assess the effect of postdialysis K on mortality in the subgroups. On the basis of baseline predialysis K level, we categorized patients into low (<4.5 mEq/L), medium (4.5 to <5.5 mEq/L), and high predialysis K (≥5.5 mEq/L) subgroups, and we defined two cutoff values of 4.5 and 5.5 mEq/L according to the preferable range of predialysis K and its distribution (25th and 75th percentiles, respectively) (2,3,5,10). We constructed a model with the interaction between baseline predialysis K and postdialysis K and tested the interaction between subgroups and postdialysis K levels.
When there were missing data, we replaced them using multiple imputations using chained equations (23). The final results from five imputation datasets were obtained according to the combination rules described by Little and Rubin (24). We performed all analyses using STATA/SE (StataCorp. 2015, Stata Statistical Software: Release 14; StataCorp. LP, College Station, TX). The level of significance was set as 0.05 for all analyses.
Results
Patient Characteristics and Dialysate Potassium
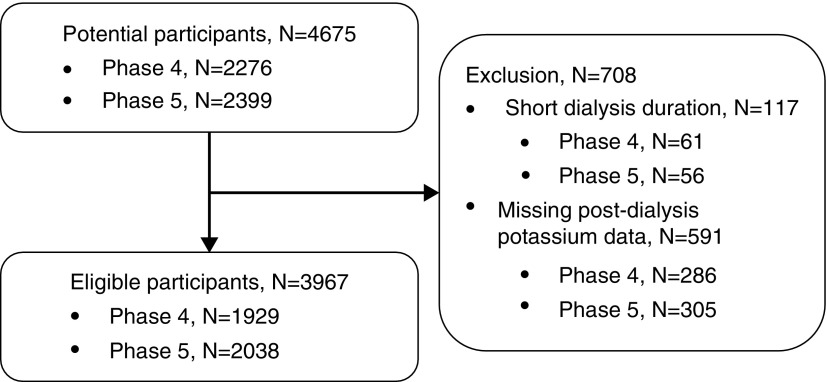
The participants had been treated at 61 facilities. Figure 1 shows the number of participants enrolled and those excluded. The number of potential participants was 4675, and 117 were excluded because of short dialysis duration and 591 were excluded because baseline postdialysis K level was not measured. Ultimately, the number of eligible participants was 3967, with 1929 in phase 4 and 2038 in phase 5.
Figure 1.
Flowchart of eligible and ineligible participants. The numbers of potential and eligible patients in each phase are shown on the left side. The reasons for ineligibility and the number of ineligible participants are shown on the right side.
There were 2552 (64%) men and 1415 (36%) women, aged 65±12 (mean±SD) years, and the median dialysis vintage was 3.9 years with an interquartile range of 0.6–9.7 years (Table 1). The number of patients in the low, medium-low, medium-high, and high postdialysis K groups was 374 (9%), 1752 (44%), 1427 (36%), and 414 (10%), respectively. The low group tended to be elderly and frail, with a high average age, low body mass index, low albumin level, high C-reactive protein level, and low normalized protein catabolic ratio. The prevalence of chronic heart failure, cancer, psychiatric disorders, and cerebrovascular disease was also highest in the low group. In all groups, >30% of patients were treated with angiotensin II receptor blockers. However, a specific trend was not observed for any medication in any group. Most of the participants (3804 out of 3967; 96%) were treated with dialysate potassium of 2.0 to <2.5 mEq/L, and this trend was consistent in all four postdialysis K groups. Only 21 participants were treated with low (<2.0 mEq/L) or high (≥2.5 mEq/L) dialysate potassium (Supplemental Table 1).
Table 1.
Baseline characteristics of 3967 participants in the Dialysis Outcomes and Practice Patterns Study in Japan
| Characteristics | Postdialysis K Range, mEq/L | ||||
|---|---|---|---|---|---|
| Low, 2.1–2.9 | Medium-Low, 3.0–3.4 | Medium-High, 3.5–3.9 | High, 4.0–6.4 | Total, 2.1–6.4 | |
| Demographic characteristics | |||||
| n (%) | 374 (9%) | 1752 (44%) | 1427 (36%) | 414 (10%) | 3967 (100%) |
| Men | 191 (51%) | 1069 (61%) | 972 (68%) | 320 (77%) | 2552 (64%) |
| Age, yr | 67±12 | 66±12 | 64±12 | 62±13 | 65±12 |
| Body mass index, kg/m2 | 20.4±3.7 | 21.2±3.4 | 21.7±3.4 | 22.3±3.9 | 21.4±3.5 |
| Laboratory measurements | |||||
| Predialysis K level, mEq/L | 3.8±0.5 | 4.6±0.6 | 5.2±0.6 | 5.6±0.7 | 4.8±0.8 |
| Albumin level, g/dl | 3.5±0.5 | 3.7±0.4 | 3.7±0.4 | 3.8±0.4 | 3.7±0.4 |
| C-reactive protein level, mg/dl | 0.16 (0.07–0.53) | 0.10 (0.06–0.39) | 0.10 (0.05–0.35) | 0.10 (0.05–0.30) | 0.10 (0.05–0.40) |
| Dialysis-related characteristics | |||||
| Dialysis vintage, yr | 3.9 (0.4–8.2) | 4.3 (0.7–10.6) | 3.7 (0.6–9.7) | 2.6 (0.5–6.5) | 3.9 (0.6–9.7) |
| Single-pool Kt/V | 1.5±0.3 | 1.4±0.3 | 1.3±0.3 | 1.2±0.3 | 1.4±0.3 |
| Normalized protein catabolic ratio, g/kg per day | 0.9±0.2 | 0.9±0.2 | 0.9±0.2 | 0.9±0.2 | 0.9±0.2 |
| Dialysis blood flow, ml/min | 209±50 | 210±47 | 205±41 | 200±35 | 207±43 |
| Weekly dialysis duration, min | 720 (677–729) | 720 (700–730) | 720 (684–727) | 720 (684–723) | 720 (690–729) |
| Comorbidities, n (%) | |||||
| Coronary artery disease | 83 (22%) | 394 (22%) | 281 (20%) | 91 (22%) | 849 (21%) |
| Chronic heart failure | 72 (19%) | 231 (13%) | 182 (13%) | 50 (12%) | 535 (13%) |
| Other heart disease | 39 (10%) | 168 (10%) | 151 (11%) | 31 (7%) | 389 (10%) |
| Cancer | 43 (11%) | 180 (10%) | 128 (9%) | 36 (9%) | 387 (10%) |
| Psychiatric disorder | 39 (10%) | 79 (5%) | 78 (5%) | 14 (3%) | 210 (5%) |
| Cerebrovascular disease | 51 (14%) | 210 (12%) | 137 (10%) | 31 (7%) | 429 (11%) |
| Diabetes mellitus | 169 (45%) | 659 (38%) | 621 (44%) | 216 (52%) | 1665 (42%) |
| Hypertension | 291 (78%) | 1364 (78%) | 1140 (80%) | 336 (81%) | 3131 (79%) |
| Dyslipidemia | 80 (21%) | 371 (21%) | 281 (20%) | 100 (24%) | 832 (21%) |
| Peripheral vascular disease | 29 (8%) | 146 (8%) | 132 (9%) | 34 (8%) | 341 (9%) |
| Medication | |||||
| Angiotensin II receptor blocker | 29 (31%) | 559 (32%) | 447 (31%) | 124 (30%) | 1245 (31%) |
| ACE inhibitor | 14 (4%) | 71 (4%) | 61 (4%) | 25 (6%) | 171 (4%) |
| Diuretics | 96 (26%) | 396 (23%) | 287 (20%) | 106 (26%) | 885 (22%) |
The numbers and/or percentages are given for dichotomized variables. The means and SD are presented for continuous variables with a normal distribution. The medium and 25–75th percentiles are given for continuous variables without a normal distribution. Postdialysis K, postdialysis serum potassium; predialysis K, predialysis serum potassium; ACE inhibitor, angiotensin-converting enzyme inhibitor.
Observational Period and Mortality
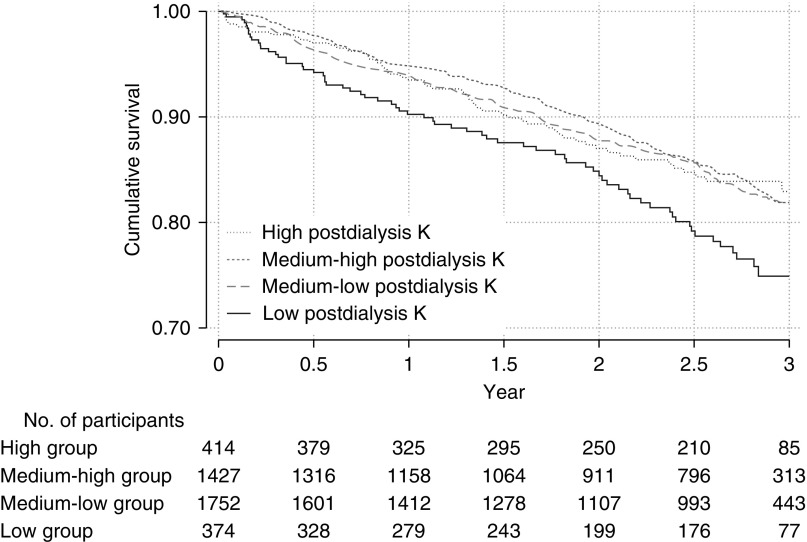
The median observational period was 2.6 years (interquartile range, 1.3–2.8 years). During this period, 562 (14%) of 3967 participants died. The number of participants who died in the low, medium-low, medium-high, and high postdialysis K groups was 96, 220, 179, and 57, respectively. The incidence rates were 11.2, 6.0, 6.2, and 7.4 per 100 person-year, respectively (Table 2). The time courses of each group are described in Figure 2. The overall incidence rate was 6.7 per 100 person-years. Sudden death and infectious disease were the most common causes of death (Table 3). The number of censored participants was 493, with 32 having undergone kidney transplantation, 458 lost to follow-up, and three converting to peritoneal dialysis.
Table 2.
Associations of postdialysis serum potassium concentration with all-cause mortality
| Postdialysis K (Range, mEq/L) | Deaths | Time at Risk, Person-Years | Incidence Rate (95% CI), per 100 Person-Years | Unadjusted Model | Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | P Value | HR (95% CI) | P Value | HR (95% CI) | P Value | HR (95% CI) | P Value | ||||
| Low (1.9–2.9) | 96 | 860 | 11.2 (9.1 to 13.6) | 1.84 (1.44 to 2.34) | <0.001 | 1.41 (0.97 to 2.05) | 0.07 | 1.44 (1.14 to 1.82) | 0.002 | 1.1 (0.84 to 1.44) | 0.49 |
| Medium-low (3.0–3.4) | 220 | 3682 | 6.0 (5.2 to 6.8) | Ref. | Ref. | Ref. | Ref. | ||||
| Medium-high (3.5–3.9) | 179 | 2869 | 6.2 (5.4 to 7.2) | 1.04 (0.86 to 1.27) | 0.69 | 1.11 (0.83 to 1.49) | 0.48 | 1.08 (0.88 to 1.32) | 0.46 | 1.08 (0.86 to 1.35) | 0.51 |
| High (4.0–6.4) | 414 | 57 | 7.4 (5.7 to 9.7) | 1.24 (0.93 to 1.66) | 0.15 | 1.13 (0.70 to 1.82) | 0.62 | 1.11 (0.81 to 1.51) | 0.52 | 1.03 (0.73 to 1.44) | 0.87 |
Categorical postdialysis K was handled as a time-varying variable in all models. Model 1 was adjusted for baseline confounders of sex, age, body mass index, comorbidities, and medications. Model 2 was adjusted for baseline confounders plus time-varying confounders of serum albumin, C-reactive protein, normalized protein catabolic rate, dialysis vintage, single-pool Kt/V, and type of vascular access. Model 3 was adjusted for confounders in model 2 plus time-varying predialysis serum potassium. Postdialysis serum potassium levels before ten deaths were missing. Postdialysis K, postdialysis serum potassium; 95% CI, 95% confidence interval; HR, hazard ratio; Ref., reference.
Figure 2.
Cumulative survival was lowest in the low postdialysis K group, and other three group showed the similar cumulative survival. The definition of each groups is as follows: low, baseline postdialysis K<3.0mEq/L; medium-low, baseline postdialysis K≥3.0 to 3.5mEq/L; medium-high, postdialysis K≥3.5 to 4.0 mEq/L; high, postdialysis K≥4.0 mEq/L.
Table 3.
Causes of death
| Cause of Death | N | Percentage |
|---|---|---|
| Sudden cardiac death/arrhythmia | 87 | 16 |
| Infectious disease | 86 | 15 |
| Malignancies | 50 | 9 |
| Coronary artery disease | 45 | 8 |
| Cerebrovascular disease | 37 | 7 |
| Gastrointestinal disease | 16 | 3 |
| Other heart disease | 14 | 3 |
| Liver disease | 11 | 2 |
| Other | 18 | 3 |
| Unknown | 198 | 35 |
| Total | 562 | 100 |
Other heart disease includes nonischemic, nonarrhythmic heart disease. Other includes traffic crash, suicide, and pulmonary and metabolic disease.
Postdialysis K and Related Factors
Over 26,193 4-month intervals, data were missing for 352 predialysis K measurements and 660 postdialysis K measurements. We observed 25,512 pairs of pre- and postdialysis K assessments during the 3-year observation period. A postdialysis K of <3.0 mEq/L was observed in 55% (1566 out of 2870) of participants in the predialysis K group with <4.0 mEq/L, in 6% (1102 out of 17,270) of those in the predialysis K group with ≥4.0 to <5.5 mEq/L, and 0.4% (20 out of 5372) of those in the predialysis K group with ≥5.5 mEq/L (Supplemental Table 2). A strong correlation was observed between predialysis K and postdialysis K (R2=0.66) (Supplemental Figure 1).
We analyzed the factors associated with postdialysis K, which were concurrently measured when measuring the postdialysis K (Supplemental Table 3). As expected, predialysis K was significantly associated with postdialysis K (coefficient 0.38; 95% confidence interval [95% CI], 0.37 to 0.38; P<0.01). Long vintage, high blood flow rate, and long dialysis duration were associated with low postdialysis K, whereas high body weight and male sex were associated with high postdialysis K. Age had little effect on postdialysis K. Dialysate potassium was not associated with postdialysis K.
Postdialysis K and Mortality
For 26,193 4-month interval assessments, we determined the postdialysis K and other covariate missing data and imputed it before survival analyses. All parameters, except for C-reactive protein level, were measured in >92% of participants (Supplemental Table 4); therefore, the missing data were not problematic.
To evaluate the nonlinear association between postdialysis K and mortality, we analyzed postdialysis K as a categorical variable and the results are shown in Table 2. Postdialysis hypokalemia (<3.0 mEq/L) was significantly associated with mortality in the unadjusted model (hazard ratio [HR], 1.84; 95% CI, 1.44 to 2.34; P<0.01). In model 1, with adjustment for baseline confounders of patient demographic characteristics, comorbidities, and medications, the HR of postdialysis hypokalemia was 1.41 (95% CI, 0.97 to 2.05; P=0.07). In model 2, with adjustment for baseline confounders plus time-varying laboratory variables without predialysis K, the HR of postdialysis hypokalemia was 1.44 (95% CI, 1.14 to 1.82; P<0.01). In model 3, with further adjustment for time-varying predialysis K, the HR of postdialysis hypokalemia was 1.10 (95% CI, 0.84 to 1.44; P=0.49). Medium-high and high postdialysis K (≥3.5 mEq/L) were not associated with mortality in either model (Table 2). The association between mortality and combination categories of pre- and postdialysis hypokalemia is shown in Table 4. The HR in participants with predialysis hypokalemia only was 1.40 (95% CI, 1.02 to 1.91; P=0.04), whereas the HR in participants with both pre- and postdialysis hypokalemia was 1.72 (95% CI, 1.35 to 2.19; P<0.01). The omnibus P value for interaction among combination categories was <0.01.
Table 4.
Associations of predialysis and postdialysis serum potassium concentrations, assessed in combination, with all-cause mortality
| Predialysis Hypokalemia | Postdialysis Hypokalemia | Participants | Deaths | Time at Risk, Person-Years | Incidence Rate (95% CI), Per 100 Person-Years | HR (95% CI) | P Value |
|---|---|---|---|---|---|---|---|
| No | No | 3318 | 407 | 6901 | 5.9 (5.4 to 6.5) | 1 (ref.) | |
| No | Yes | 139 | 17 | 361 | 4.7 (2.9 to 7.6) | 0.82 (0.51 to 1.32) | 0.42 |
| Yes | No | 271 | 46 | 412 | 11.2 (8.4 to 14.9) | 1.40 (1.02 to 1.91) | 0.04 |
| Yes | Yes | 234 | 77 | 499 | 15.4 (12.3 to 19.3) | 1.72 (1.35 to 2.19) | <0.01 |
Predialysis hypokalemia was defined as <4.0 mEq/L and postdialysis hypokalemia was defined <3.0 mEq/L. The model was adjusted for baseline confounders of sex, age, body mass index, comorbidities, and medications, and time-varying confounders of serum albumin, C-reactive protein, normalized protein catabolic rate, dialysis vintage, single-pool Kt/V, dialysate bicarbonate, and type of vascular access. P value compares the HR in the indicated group to 1 (reference). The omnibus P value for group interaction was <0.01. 95% CI, 95% confidence interval; HR, hazard ratio; ref., reference.
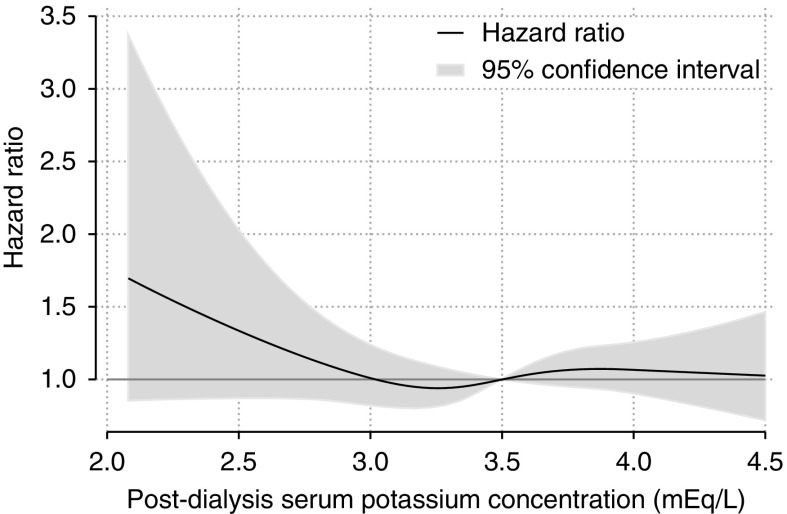
To analyze the severity of postdialysis hypokalemia and mortality, we analyzed postdialysis K as a continuous variable using a cubic spline curve. In this model, we obtained HRs and 95% CIs for each 0.1 mEq/L of postdialysis K. The results are represented graphically in Figure 3 and detailed information is shown in Supplemental Table 5. The HR increased from 1.1 (95% CI, 0.8 to 1.3) in participants with a postdialysis K level of 3.0 mEq/L to 1.7 (95% CI, 0.9 to 3.3) in those with a postdialysis K level of 2.1 mEq/L. However, the association between the severity of postdialysis hypokalemia and mortality was not statistically significant.
Figure 3.
Hazard ratio increased depending on severity of hypokalemia, but the association was not statistically significant (95% confidence interval included 1.0). The thick black line shows the HR of all-cause mortality. The reference (HR=1) is 3.5 mEq/L. The gray area shows the 95% CI. The model was adjusted for baseline confounders of sex, age, body mass index, comorbidities, and medications, and time-varying confounders of serum albumin, C-reactive protein, normalized protein catabolic rate, dialysis vintage, single-pool Kt/V, dialysate bicarbonate, type of vascular access, and predialysis K.
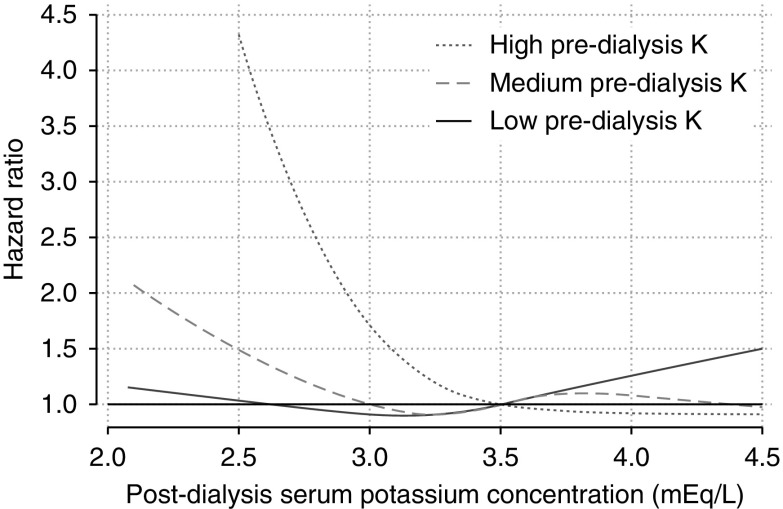
The subgroup analysis showed an L-shaped association between postdialysis K level and mortality in the medium and high baseline predialysis K subgroups (Figure 4). The interaction P value between time-varying postdialysis K and subgroups was 0.09; therefore, the subgroup difference was not statistically significant.
Figure 4.
The pattern of association of postdialysis hypokalemia with mortality different by baseline predialysis potassium level, but this effect modification was not statistically significant (interaction P=0.09). The numbers of patients in the high, medium, and low predialysis K groups were 790, 1892, and 1280, respectively. Five patients were excluded from this analysis because their predialysis K level was missing. The reference (HR=1) was 3.5mEq/L. The definition of subgroups is as follows: low, <4.5 mEq/L; medium, 4.5 to <5.5 mEq/L; and high, ≥5.5 mEq/L.
In sensitivity analyses, we analyzed cause-specific mortality, and postdialysis hypokalemia (<3.0 mEq/L) showed an L-shaped association with mortality combined sudden cardiac death and unknown death (Supplemental Figure 2, Supplemental Table 6). This result was consistent with the results from the analysis of all-cause mortality. We confirmed the robustness of treatment duration by analyses with two different exclusion criteria, one with no exclusion on the basis of treatment duration and one that limited inclusion to patients undergoing thrice-weekly dialysis of at least 4 hours’ duration. These two analyses showed L-shaped association between postdialysis K and mortality (Supplemental Figures 3 and 4). We also observed the mortality risk of postdialysis hypokalemia in a logistic model and confirmed robustness in statistical models (Supplemental Figure 5). We conducted sensitivity analysis to exclude data with higher postdialysis K than predialysis K and obtained a similar result to the original analysis. Further, the results of generalized estimating equations were consistent with those of the original analysis, and we confirmed the results in the analysis without bias caused by time-varying covariates (Supplemental Table 7).
Discussion
We investigated the association between postdialysis K and mortality in dialysis population using data from an observational study in Japan. Postdialysis hypokalemia was associated with mortality without adjustment for predialysis K. After adjusting for time-varying predialysis K, the association between postdialysis K and mortality was largely attenuated. We also found that having postdialysis hypokalemia only was not associated with mortality, whereas having both pre- and postdialysis hypokalemia was associated with the highest mortality rate, followed by having predialysis hypokalemia only.
Previous observational studies have explored the risk of low potassium dialysate (2,5–7,13,14). One case-cohort study found that patients treated with low potassium dialysate were more likely to die from sudden cardiac death (7). Another case-control study explored the risk of low potassium dialysate regardless of predialysis K level (6). Postdialysis K was not evaluated in these studies; therefore, to the best of our knowledge, this is the first study to examine the direct association between postdialysis hypokalemia and mortality.
Even after adjusting for time-varying nutritional and inflammatory status, we observed a consistent association between postdialysis K and mortality. Thus, the association of postdialysis K with mortality is independent of nutritional and inflammatory status. However, this association disappeared after adjusting for time-varying predialysis K. These results suggest that the association between postdialysis K and mortality is mainly explained by predialysis K, and the effect of postdialysis K may be negligible or nonexistent. We also found that participants with a combination of pre- and postdialysis hypokalemia had the highest mortality, and that the interaction of pre- and postdialysis K measured during a single dialysis session was significant. These results suggest that the effect of postdialysis K on mortality may be modified by predialysis K.
One strength of this study was that we could exclude the direct effect of dialysate potassium on mortality. Dialysate potassium noticeably affects postdialysis K, and its concentration may affect mortality. We observed no association between dialysate potassium and postdialysis K because dialysate potassium is not commonly adjusted in Japan and was 2.0 mEq/L in most participants. Thus, we could evaluate the association between postdialysis hypokalemia and mortality regardless of dialysate potassium. Another strength was that the postdialysis K level was measured on a 4-monthly basis, allowing us to evaluate fluctuations. We could not evaluate the daily or weekly fluctuations in postdialysis K level; however, if we were able to do so, we would have identified a clearer association between mortality and postdialysis K because ignoring daily or weekly fluctuations weakens the estimated effect of postdialysis K on mortality. Further, the generalizability of the findings was another strength, as the sampling approach of J-DOPPS makes it representative of most Japanese dialysis settings.
Aside from our inability to establish causality because of the observational study design, our study had some limitations. First, the result might include potential residual confounding factors such as malnutrition and inflammation. Although we adjusted the model for nutritional and inflammatory markers, unmeasured health conditions may remain as residual confounders. Second, the generalizability of our findings to patients treated with dialysis for extremely long or short periods is unclear. Potassium removal depends on dialysis duration. Hence, the threshold of postdialysis K may differ from the result of our study. Further, the generalizability of this study to patients treated with low (<1.5 mEq/L) or high (≥2.5 mEq/L) dialysate potassium is also unclear. Third, treatment quality in each facility might have confounded the results because we could not adjust for facilities because of the insufficient number of outcomes. In other words, participants treated at high-quality facilities might be unlikely to manifest postdialysis hypokalemia and more likely to have a better prognosis. Finally, the timing of postdialysis K measurements was a limitation. We measured postdialysis K levels only once during each dialysis session. Therefore, we could not evaluate serum potassium fluctuations after dialysis, nor distinguish persistent hypokalemia from transient hypokalemia with rebound.
In conclusion, postdialysis hypokalemia was associated with mortality, but the association was not independent of predialysis K. Having both pre- and postdialysis hypokalemia was associated with the highest mortality.
Disclosures
Dr. Akizawa reports personal fees from Bayer Health Care, personal fees from Astellas, personal fees from JT Corporation, personal fees from Torii Pharma, personal fees from Ono Pharma, personal fees from Kissei Pharma, personal fees from GlaxoSmithKline, personal fees from Fuso Pharmaceutical, personal fees from Nipro Corporation, personal fees from Chugai Pharma, outside the submitted work and acted as scientific advisors to Kyowa Hakko Kirin. Dr. Fukuhara and Dr. Fukuma acted as a scientific advisor to Kyowa Hakko Kirin. Dr. Kimachi and Dr. Ohnishi have nothing to disclose.
Supplemental Material
This article contains the following supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.07950718/-/DCSupplemental.
Supplemental Appendix 1. Data structure/methods.
Supplemental Table 1. Distribution of participants according to baseline dialysate potassium and baseline postdialysis serum potassium concentrations.
Supplemental Table 2. Participants according to the combination categories of predialysis and postdialysis serum potassium concentrations.
Supplemental Table 3. Factors related to postdialysis serum potassium concentration.
Supplemental Table 4. Missing values and multiple imputation.
Supplemental Table 5. Association of postdialysis serum potassium concentration with all-cause mortality.
Supplemental Table 6. Association of postdialysis serum potassium concentration with sudden cardiac death and unknown death.
Supplemental Table 7. Association of postdialysis serum potassium with all-cause mortality using generalized estimating equations.
Supplemental Figure 1. Correlation of predialysis serum potassium and postdialysis serum potassium.
Supplemental Figure 2. Association of postdialysis serum potassium concentration, assessed as a continuous variable, with sudden cardiac death and unknown death.
Supplemental Figure 3. Association of postdialysis serum potassium concentration with all-cause mortality in participants with long treatment durations.
Supplemental Figure 4. Association of postdialysis serum potassium concentration with all-cause mortality in participants with long treatment duration.
Supplemental Figure 5. Association of postdialysis serum potassium concentration with all-cause mortality in a logistic model.
Supplementary Material
Acknowledgments
The Dialysis Outcomes and Practice Patterns Study (DOPPS) Program was supported by Amgen, Kyowa Hakko Kirin, AbbVie Inc., Sanofi Renal, Baxter Healthcare, and Vifor Fresenius Medical Care Renal Pharma Ltd. The DOPPS.org website lists the full details. J-DOPPS was administered by the Arbor Research Collaborative for Health (Ann Arbor, MI) and supported by Kyowa Hakko Kirin Co., Ltd. All support was provided without restrictions on publication.
The authors are grateful to the study nurses, physicians, and medical directors for all of the time and attention that they devoted to our study. We appreciate the cooperation of the facilities participating in the Dialysis Outcomes and Practice Patterns Study in Japan (J-DOPPS).
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.United States Renal Data System : 2017 USRDS Annual Data Report: Epidemiology of Kidney Disease in the United States, Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2017 [Google Scholar]
- 2.Kovesdy CP, Regidor DL, Mehrotra R, Jing J, McAllister CJ, Greenland S, Kopple JD, Kalantar-Zadeh K: Serum and dialysate potassium concentrations and survival in hemodialysis patients. Clin J Am Soc Nephrol 2: 999–1007, 2007 [DOI] [PubMed] [Google Scholar]
- 3.Yusuf AA, Hu Y, Singh B, Menoyo JA, Wetmore JB: Serum potassium levels and mortality in hemodialysis patients: A retrospective cohort study. Am J Nephrol 44: 179–186, 2016 [DOI] [PubMed] [Google Scholar]
- 4.Hwang JC, Wang CT, Chen CA, Chen HC: Hypokalemia is associated with increased mortality rate in chronic hemodialysis patients. Blood Purif 32: 254–261, 2011 [DOI] [PubMed] [Google Scholar]
- 5.Lee S, Kang E, Yoo KD, Choi Y, Kim DK, Joo KW, Yang SH, Kim YL, Kang SW, Yang CW, Kim NH, Kim YS, Lee H: Lower serum potassium associated with increased mortality in dialysis patients: A nationwide prospective observational cohort study in Korea. PLoS One 12: e0171842, 2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pun PH, Lehrich RW, Honeycutt EF, Herzog CA, Middleton JP: Modifiable risk factors associated with sudden cardiac arrest within hemodialysis clinics. Kidney Int 79: 218–227, 2011 [DOI] [PubMed] [Google Scholar]
- 7.Karnik JA, Young BS, Lew NL, Herget M, Dubinsky C, Lazarus JM, Chertow GM: Cardiac arrest and sudden death in dialysis units. Kidney Int 60: 350–357, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Jadoul M, Thumma J, Fuller DS, Tentori F, Li Y, Morgenstern H, Mendelssohn D, Tomo T, Ethier J, Port F, Robinson BM: Modifiable practices associated with sudden death among hemodialysis patients in the Dialysis Outcomes and Practice Patterns Study. Clin J Am Soc Nephrol 7: 765–774, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Karaboyas A, Zee J, Brunelli SM, Usvyat LA, Weiner DE, Maddux FW, Nissenson AR, Jadoul M, Locatelli F, Winkelmayer WC, Port FK, Robinson BM, Tentori F: Dialysate potassium, serum potassium, mortality, and arrhythmia events in hemodialysis: Results from the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 69: 266–277, 2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nakai S, Suzuki K, Masakane I, Wada A, Itami N, Ogata S, Kimata N, Shigematsu T, Shinoda T, Syouji T, Taniguchi M, Tsuchida K, Nakamoto H, Nishi S, Nishi H, Hashimoto S, Hasegawa T, Hanafusa N, Hamano T, Fujii N, Marubayashi S, Morita O, Yamagata K, Wakai K, Watanabe Y, Iseki K, Tsubakihara Y: Overview of regular dialysis treatment in Japan (as of 31 December 2008). Ther Apher Dial 14: 505–540, 2010 [DOI] [PubMed] [Google Scholar]
- 11.Blumberg A, Roser HW, Zehnder C, Müller-Brand J: Plasma potassium in patients with terminal renal failure during and after haemodialysis; relationship with dialytic potassium removal and total body potassium. Nephrol Dial Transplant 12: 1629–1634, 1997 [DOI] [PubMed] [Google Scholar]
- 12.Agar BU, Culleton BF, Fluck R, Leypoldt JK: Potassium kinetics during hemodialysis. Hemodial Int 19: 23–32, 2015 [DOI] [PubMed] [Google Scholar]
- 13.Brunelli SM, Spiegel DM, Du Mond C, Oestreicher N, Winkelmayer WC, Kovesdy CP: Serum-to-dialysate potassium gradient and its association with short-term outcomes in hemodialysis patients. Nephrol Dial Transplant 33: 1207–1214, 2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ferrey A, You AS, Kovesdy CP, Nakata T, Veliz M, Nguyen DV, Kalantar-Zadeh K, Rhee CM: Dialysate potassium and mortality in a prospective hemodialysis cohort. Am J Nephrol 47: 415–423, 2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Young EW, Goodkin DA, Mapes DL, Port FK, Keen ML, Chen K, Maroni BL, Wolfe RA, Held PJ: The Dialysis Outcomes and Practice Patterns Study (DOPPS): An international hemodialysis study. Kidney Int 57: S74–S81, 2000 [Google Scholar]
- 16.Pisoni RL, Gillespie BW, Dickinson DM, Chen K, Kutner MH, Wolfe RA: The Dialysis Outcomes and Practice Patterns Study (DOPPS): Design, data elements, and methodology. Am J Kidney Dis 44[Suppl 2]: 7–15, 2004 [DOI] [PubMed] [Google Scholar]
- 17.National Kidney Foundation : KDOQI clinical practice guideline for hemodialysis adequacy: 2015 Update. Am J Kidney Dis 66: 884–930, 2015 [DOI] [PubMed] [Google Scholar]
- 18.Watanabe Y, Kawanishi H, Suzuki K, Nakai S, Tsuchida K, Tabei K, Akiba T, Masakane I, Takemoto Y, Tomo T, Itami N, Komatsu Y, Hattori M, Mineshima M, Yamashita A, Saito A, Naito H, Hirakata H, Minakuchi J; “Maintenance Hemodialysis: Hemodialysis Prescriptions” Guideline Working Group, Japanese Society for Dialysis Therapy : Japanese society for dialysis therapy clinical guideline for “Maintenance hemodialysis: Hemodialysis prescriptions”. Ther Apher Dial 19[Suppl 1]: 67–92, 2015 [DOI] [PubMed] [Google Scholar]
- 19.Heguilén RM, Sciurano C, Bellusci AD, Fried P, Mittelman G, Rosa Diez G, Bernasconi AR: The faster potassium-lowering effect of high dialysate bicarbonate concentrations in chronic haemodialysis patients. Nephrol Dial Transplant 20: 591–597, 2005 [DOI] [PubMed] [Google Scholar]
- 20.Gutzwiller JP, Schneditz D, Huber AR, Schindler C, Garbani E, Zehnder CE: Increasing blood flow increases kt/V(urea) and potassium removal but fails to improve phosphate removal. Clin Nephrol 59: 130–136, 2003 [DOI] [PubMed] [Google Scholar]
- 21.Harrell FE: Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis, New York, NY, Springer-Verlag, 2015 [Google Scholar]
- 22.Fukagawa M, Fukuma S, Onishi Y, Yamaguchi T, Hasegawa T, Akizawa T, Kurokawa K, Fukuhara S: Prescription patterns and mineral metabolism abnormalities in the cinacalcet era: Results from the MBD-5D study. Clin J Am Soc Nephrol 7: 1473–1480, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.van Buuren S, Boshuizen HC, Knook DL: Multiple imputation of missing blood pressure covariates in survival analysis. Stat Med 18: 681–694, 1999 [DOI] [PubMed] [Google Scholar]
- 24.Little RJA, Rubin DB: Statistical Analysis with Missing Data, Hoboken, N.J., Wiley, 2002 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.