Abstract
Neurotropic herpesviruses have been associated with the onset and progression of Alzheimer’s disease, a common form of dementia that afflicts a large percentage of elderly individuals. Interestingly, among the neurotropic herpesviruses, herpes simplex virus-1, human herpesvirus-6A, and human herpesvirus-6B have been reported to infect several cell types present in the central nervous system and to dysregulate autophagy, a process required for homeostasis of cells, especially neurons. Indeed autophagosome accumulation, indicating an unbalance between autophagosome formation and autophagosome degradation, has been observed in neurons of Alzheimer’s disease patients and may play a role in the intracellular and extracellular accumulation of amyloid β and in the altered protein tau metabolism. Moreover, herpesvirus infection of central nervous system cells such as glia and microglia can increase the production of oxidant species through the alteration of mitochondrial dynamics and promote inflammation, another hallmark of Alzheimer’s disease. This evidence suggests that it is worth further investigating the role of neurotropic herpesviruses, particularly human herpesvirus-6A/B, in the etiopathogenesis of Alzheimer’s disease.
Keywords: Alzheimer's disease, autophagy, HHV-6A, HHV-6B, HSV-1, neurotropic viruses, amyloid β, tau protein, reactive oxygen species, inflammation, AD
Autophagy and Alzheimer’s Disease
Autophagy can be subdivided into three distinct degradative cellular processes, namely macroautophagy, microautophagy, and chaperone-mediated autophagy. Macroautophagy, hereafter referred to as autophagy, is articulated in several steps that go from autophagosome formation to their fusion with lysosomes where the autophagosome content is degraded and eventually recycled (Klionsky et al., 2016). A number of genes called autophagy-related genes are responsible for the execution of the entire process and the activation of molecular pathways such as phosphoinositide 3-kinase/protein kinase B/mammalian target of rapamycin and signal transducer and activator of transcription 3 is also involved in its regulation. Through autophagy, cells get rid of toxic components such as misfolded proteins (especially the long-lived ones), protein aggregates, and damaged organelles whose elimination is also mediated by selective forms of autophagy. The basal activation of autophagy contributes to the maintenance of homeostasis of cells, especially post-mitotic cells that are not able to dilute unwanted noxious cellular components through cell division. To this category belong neurons, cells that are highly dependent on autophagy, as indicated by the neurodegeneration and the enhanced neuronal cell death that occurs in the absence of autophagy (Komatsu et al., 2006). Indeed, an increased number of autophagosomes, suggesting a dysregulation of autophagy, has been observed in the neurons of patients affected by neurodegenerative pathologies including Alzheimer’s disease (AD) (Uddin et al., 2018). Likewise, aging is physiologically accompanied by a progressive reduction of autophagy and by concomitant alteration of neuronal homeostasis (Metaxakis et al., 2018). Although whether AD and brain aging are mechanistically linked remains to be clarified, it is known that AD is the most common form of dementia afflicting the elderly population. It is characterized, at the molecular level, by extracellular and intracellular accumulation of amyloid β (Aβ) peptide fragments of the amyloid precursor protein (APP) and neurofibrillary tangles, composed of the microtubule-associated hyperphosphorylated protein tau. Besides accumulating in the brain, both Aβ and tau can be detected in the cerebrospinal fluid and blood and represent AD biomarkers together with several pro-inflammatory miRNAs that are found to be increased in patients affected by this neurodegenerative disease (Femminella et al., 2015; Wu et al., 2016). It has been well demonstrated that the increased production and aggregation of Aβ monomers result in neuronal dysfunction. APP protein can be processed by enzymes called α-, β- and γ-secretases. The amyloidogenic process is initiated by the cleavage of APP mediated by the β-secretase, also known as β-site APP cleaving enzyme 1 or BACE-1 (Vassar et al., 1999). A connection between APP metabolism and tau proteostasis has been demonstrated, as the manipulation of APP metabolism through the modulation of β- and γ-secretases resulted also in changes of tau protein accumulation (Moore et al., 2015). Interestingly, autophagy strongly influences the metabolism of Aβ (Zhou et al., 2011) as well as of tau protein (Wang and Mandelkow, 2012), both components accumulating in the course of AD. In addition to APP metabolism, autophagy contributes to the degradation of BACE1, the β-secretase that cleaves APP and initiates the amyloidogenic process (Feng et al., 2017). Autophagy also plays a role in Aβ secretion and plaque formation, as indicated by an autophagy-deficient mouse model in which Aβ accumulated inside neurons, leading to neurodegeneration (Nilsson et al., 2013). In the course of AD, the intracellular accumulation of Aβ in neurons seems to precede the extracellular plaque formation. The above reported findings indicate that autophagy is required for normal neuronal physiology and that autophagy dysregulation may be involved in the onset and/or progression of AD by several means. Therefore, it is possible that the manipulation of autophagy may hold the key to help prevent or better treat this neurodegenerative condition, which in spite of considerable effort, remains an incurable disease.
Autophagy and Herpesviruses
A large number of studies have clearly documented that autophagy is often dysregulated by viruses belonging to several unrelated families. This is not surprising given the pivotal role in the innate and adaptive anti-microbial immune response (Santarelli et al., 2015). Herpesviruses may impair one or more of the several autophagic steps, and this can occur either during primary infection (Santarelli et al., 2016; Gilardini Montani et al., 2018a) or during lytic reactivation from latency (Cirone, 2018). Notably, viruses may not only inhibit autophagy, but they can also subvert this process to hijack the autophagic machinery for their own purpose. For example, gamma herpesviruses may block the last autophagic steps and use autophagosomes for intracellular transportation towards the plasma membrane, when the lytic cycle is activated (Granato et al., 2014, 2015). Other viruses belonging to the alpha herpesvirus family, such as varicella zoster virus, promote autophagy to prolong the survival of infected cells, and although the autophagic flux is not blocked, the viral particles seem to survive to the lysosome degradation (Buckingham et al., 2015; Hogestyn et al., 2018). Herpesviruses that dysregulate autophagy also include the main neurotropic viruses: herpes simplex virus-1 (HSV-1), which blocks autophagy in infected neurons by inhibiting Beclin-1 (Orvedahl et al., 2007), a protein essential for the autophagic process, and the beta herpesviruses human herpesvirus (HHV)-6A and HHV-6B (HHV-6A/B) that, as we have recently shown, modulate autophagy differently in the cells in which they replicate (Romeo et al., 2019).
Neurotropic Herpesviruses, Autophagy and AD
Investigators have searched for many years to find a possible association of neurodegenerative diseases such as multiple sclerosis and AD with viral infections (Buckingham et al., 2018). In particular, the role of the neurotropic virus HSV-1 has been extensively investigated in the etiology of AD (Piacentini et al., 2014). The virus may enter sensory neurons, reach the trigeminal ganglion and from here enter the central nervous system. Once there, it may cause acute encephalitis or remain in a latent state (Lewandowski et al., 2002; Bradshaw and Venkatesan, 2016). As an alternative route, HSV-1 may enter the central nervous system through the blood stream. HHV-6A/B are able to reach the central nervous system by entering through the olfactory pathway (Harberts et al., 2011), and like HSV-1, they may establish a latent infection in the central nervous system. However, in particular circumstances, for example in conditions in which the immune pressure is reduced, both viruses may start replicating, as indicated by the detection of viral particles in the cerebrospinal fluid. HSV-1 and HHV-6A/B can infect several cells in the central nervous system, including glial cells and neurons (Lokensgard et al., 2002; Dietrich et al., 2004; Donati et al., 2005; Reynaud and Horvat, 2013; Zerboni et al., 2013; Prusty et al., 2018). The latter are the main mediator of Aβ deposition that leads to plaque formation in the course of AD. Aβ also accumulates inside neurons and it may derive from the intracellular pool of APP or by its uptake from extracellular space. Glial cells also mediate Aβ deposition and, considering that they are highly represented in the central nervous system, their contribution to AD may be relevant (Frost and Li, 2017). Regarding the possible association of neurotropic viruses with the amyloidogenic process of AD, it has been shown that HSV-1 may alter the distribution of cellular APP, that its capsid proteins can directly interact with APP (Cheng et al., 2011), and that its infection causes neurodegeneration and AD-like phosphorylation of tau protein (Zambrano et al., 2008; Wozniak et al., 2009a). Moreover, about 60% of brains of elderly people contain latent HSV-1 DNA in the limbic system, and the frontal and temporal cortices, the central nervous system regions frequently compromised in AD (Jamieson et al., 1992; Itzhaki et al., 1997), and HSV-1 DNA can be detected within the amyloid plaques (Wozniak et al., 2009b). However, more recently, HHV-6A but not HSV-1 has been found to be highly abundant in the brain of AD patients and interestingly, HHV-6A has been linked with molecular, genetic, clinical, and neuropathological aspects of AD (Readhead et al., 2018). Moreover, it has been reported that both HHV-6A and HHV-6B infection of neurons directly induces Aβ deposition, as a host defense mechanism, although HHV-6A does so more efficiently (Eimer et al., 2018). These findings strongly suggest that further investigation of the role of HHV-6A and B in AD is warranted. Regarding autophagy dysregulation by neurotropic viruses in neuronal cells, it has been reported that HSV-1 protein ICP34.5 can block autophagy by binding and inhibiting beclin-1, and more importantly, that the neurovirulence of HSV-1 is dependent on the expression of this protein (Orvedahl et al., 2007). The impact of viral infection by HHV-6A and HHV-6B on autophagy in the central nervous system cells such as neurons and glial cells, including astrocytes and oligodendrocytes, remains to be explored. Given the pivotal role of autophagy in neuronal physiology, it is important to investigate whether it is dysregulated by neurotropic viruses and contributes to Aβ intracellular or extracellular accumulation or neurofibrillary tangle formation (Figure 1).
Figure 1.
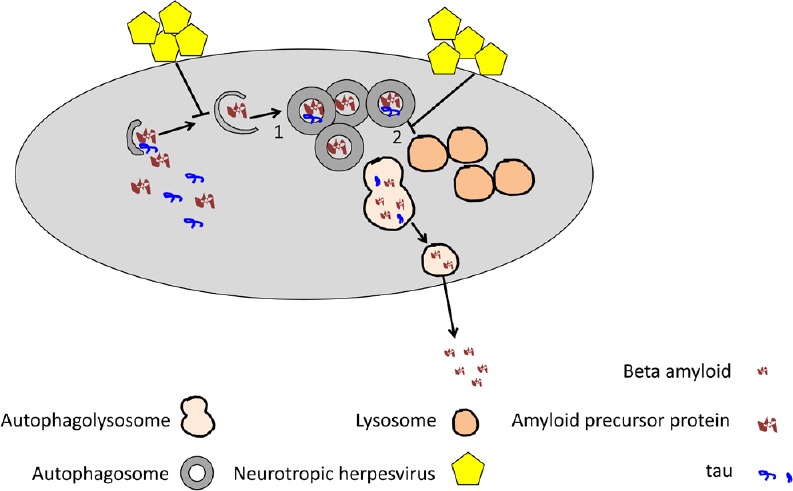
Neurotropic herpesvirus infection of neurons or glial cells may dysregulate the initial (1) or the final (2) autophagic steps and possibly, through this mechanism, alter amyloid precursor protein or tau metabolism or beta amyloid secretion.
Finally, in addition to bulk autophagy, an impairment of the selective autophagy aimed at the removal of damaged mitochondria, or mitophagy, seems to be involved in the onset/progression of AD. This is indicated by the accumulation of dysfunctional mitochondria in the neurons of patients affected by this disease (Kerr et al., 2017). Of note, damaged mitochondria increase production of ROS, and these molecules in turn induce further damage of mitochondria that are primarily responsible for the oxidative stress, another hallmark of AD (Kurihara et al., 2012). Several viruses, including herpesviruses, are able to alter mitochondrial dynamics and/or mitophagy, likely because mitochondria play multiple roles in the anti-viral immune defense and may regulate autophagy activation. Therefore, the dysregulation of mitophagy could represent another possible mechanism through which neurotropic viruses promote AD. The oxidative stress characteristic of AD may contribute to the activation of microglia (Mattson et al., 1993), which is represented by central nervous system resident cells and cells that originate from peripheral blood monocytes reaching the central nervous system by crossing the blood-brain barrier. Interestingly, HHV-6A/B can infect and activate microglia (Fotheringham et al., 2007; Reynaud and Horvat, 2013; Reynaud et al., 2014), and we have recently reported that Epstein-Barr virus activated monocytes and increased ROS production in these cells (Gilardini Montani et al., 2018b). We are currently investigating whether HHV-6 infection of monocytes may alter ROS production and/or interfere with autophagy or mitophagy. The activation of microglia also promotes inflammation, another feature of AD (Figure 2), and previous studies have indicated that HHV-6A and HHV-6B induce pro-inflammatory effects in central nervous system cells (Meeuwsen et al., 2005). Indeed, HHV-6 infection of glial or microglial cells may result in an altered release of pro-inflammatory cytokines such interleukin-6 or chemokines such as interleukin -8 and Rantes that attract leukocytes to the central nervous system, further increasing central nervous system inflammation (Reynaud and Horvat, 2013). Interestingly, besides promoting oxidative stress and inflammation, activated microglial cells do not remove Aβ, as phagocytic cells physiologically should do (Navarro et al., 2018), but instead promote Aβ fibrillogenesis (Nagele et al., 2004). These findings suggest that activated microglia may contribute to several features of AD and their infection by neurotropic viruses could alter their functions, turning microglia from cells preventing AD to cells promoting it.
Figure 2.
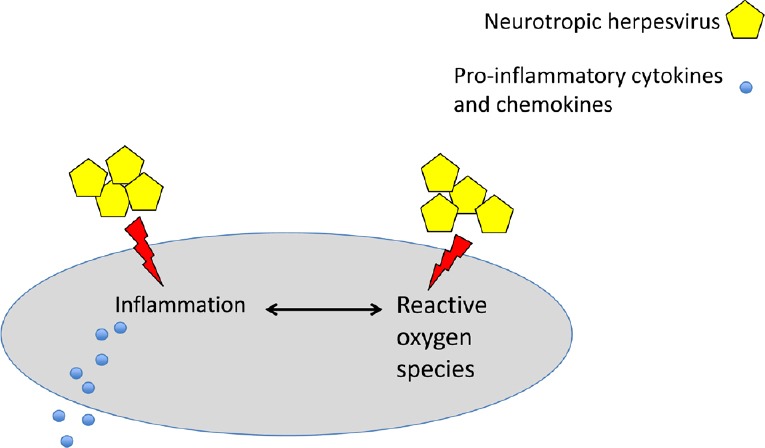
Neurotropic herpesvirus infection increases reactive oxygen species production and promotes inflammation in glial and microglial cells.
Conclusions
Although further studies are required, the above reported findings suggest that infection of central nervous system cells by neurotropic viruses such as HSV-1 and HHV-6A/B may play a role in AD. Moreover, as neurons are strongly dependent on autophagy, and autophagy is involved in the Aβ and protein tau metabolism, it could be that the dysregulation of the autophagic process in the different central nervous system cells may represent one of the mechanisms through which those viruses contribute to the onset/progression of AD. Shedding more light on the relationship between neurotropic virus infection of central nervous system cells and autophagy dysregulation, intracellular and extracellular Aβ accumulation, neurofibrillary tangle formation or mitophagy impairment, oxidative stress, and inflammation could lead to new therapeutic strategies able to prevent or restrain the progression of the neurodegenerative process that leads to AD.
Additional file: Open peer review report 1 (99.8KB, pdf) .
Footnotes
Conflicts of interest: The authors declare no conflicts of interest.
Financial support: The work was supported by Human Herpesvirus-6 Foundation and Istituto Pasteur Italia-Fondazione Cenci Bolognetti (to MC).
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewer: Emilio Varea, University of Valencia, Spain.
Funding: The work was supported by Human Herpesvirus-6 Foundation and Istituto Pasteur Italia-Fondazione Cenci Bolognetti (to MC).
P-Reviewer: Varea E; C-Editors: Zhao M, Yu J; T-Editor: Liu XL
References
- 1.Bradshaw MJ, Venkatesan A. Herpes simplex virus-1 encephalitis in adults: pathophysiology, diagnosis, and management. Neurotherapeutics. 2016;13:493–508. doi: 10.1007/s13311-016-0433-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Buckingham EM, Carpenter JE, Jackson W, Zerboni L, Arvin AM, Grose C. Autophagic flux without a block differentiates varicella-zoster virus infection from herpes simplex virus infection. Proc Natl Acad Sci U S A. 2015;112:256–261. doi: 10.1073/pnas.1417878112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Buckingham EM, Foley MA, Grose C, Syed NA, Smith ME, Margolis TP, Thurtell MJ, Kardon R. Identification of herpes zoster-associated temporal arteritis among cases of giant cell arteritis. Am J Ophthalmol. 2018;187:51–60. doi: 10.1016/j.ajo.2017.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cheng SB, Ferland P, Webster P, Bearer EL. Herpes simplex virus dances with amyloid precursor protein while exiting the cell. PLoS One. 2011;6:e17966. doi: 10.1371/journal.pone.0017966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cirone M. EBV and KSHV infection dysregulates autophagy to optimize viral replication, prevent immune recognition and promote tumorigenesis. Viruses. 2018;10:E599. doi: 10.3390/v10110599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dietrich J, Blumberg BM, Roshal M, Baker JV, Hurley SD, Mayer-Proschel M, Mock DJ. Infection with an endemic human herpesvirus disrupts critical glial precursor cell properties. J Neurosci. 2004;24:4875–4883. doi: 10.1523/JNEUROSCI.5584-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Donati D, Martinelli E, Cassiani-Ingoni R, Ahlqvist J, Hou J, Major EO, Jacobson S. Variant-specific tropism of human herpesvirus 6 in human astrocytes. J Virol. 2005;79:9439–9448. doi: 10.1128/JVI.79.15.9439-9448.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eimer WA, Vijaya Kumar DK, Navalpur Shanmugam NK, Rodriguez AS, Mitchell T, Washicosky KJ, Gyorgy B, Breakefield XO, Tanzi RE, Moir RD. Alzheimer’s disease-associated beta-amyloid is rapidly seeded by herpesviridae to protect against brain infection. Neuron. 2018;99:56–63.e3. doi: 10.1016/j.neuron.2018.06.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Femminella GD, Ferrara N, Rengo G. The emerging role of microRNAs in Alzheimer’s disease. Front Physiol. 2015;6:40. doi: 10.3389/fphys.2015.00040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Feng T, Tammineni P, Agrawal C, Jeong YY, Cai Q. Autophagy-mediated regulation of BACE1 protein trafficking and degradation. J Biol Chem. 2017;292:1679–1690. doi: 10.1074/jbc.M116.766584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fotheringham J, Akhyani N, Vortmeyer A, Donati D, Williams E, Oh U, Bishop M, Barrett J, Gea-Banacloche J, Jacobson S. Detection of active human herpesvirus-6 infection in the brain: correlation with polymerase chain reaction detection in cerebrospinal fluid. J Infect Dis. 2007;195:450–454. doi: 10.1086/510757. [DOI] [PubMed] [Google Scholar]
- 12.Frost GR, Li YM. The role of astrocytes in amyloid production and Alzheimer’s disease. Open Biol. 2017;7:170228. doi: 10.1098/rsob.170228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gilardini Montani MS, Santarelli R, Granato M, Gonnella R, Torrisi MR, Faggioni A, Cirone M. EBV reduces autophagy, intracellular ROS and mitochondria to impair monocyte survival and differentiation. Autophagy. 2018a doi: 10.1080/15548627.2018.1536530. doi: 10.1080/1554862720181536530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gilardini Montani MS, Santarelli R, Falcinelli L, Gonnella R, Granato M, Di Renzo L, Cuomo L, Vitillo M, Faggioni A, Cirone M. EBV up-regulates PD-L1 on the surface of primary monocytes by increasing ROS and activating TLR signaling and STAT3. J Leukoc Biol. 2018b;104:821–832. doi: 10.1002/JLB.2A0118-029RR. [DOI] [PubMed] [Google Scholar]
- 15.Granato M, Santarelli R, Farina A, Gonnella R, Lotti LV, Faggioni A, Cirone M. Epstein-barr virus blocks the autophagic flux and appropriates the autophagic machinery to enhance viral replication. J Virol. 2014;88:12715–12726. doi: 10.1128/JVI.02199-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Granato M, Santarelli R, Filardi M, Gonnella R, Farina A, Torrisi MR, Faggioni A, Cirone M. The activation of KSHV lytic cycle blocks autophagy in PEL cells. Autophagy. 2015;11:1978–1986. doi: 10.1080/15548627.2015.1091911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harberts E, Yao K, Wohler JE, Maric D, Ohayon J, Henkin R, Jacobson S. Human herpesvirus-6 entry into the central nervous system through the olfactory pathway. Proc Natl Acad Sci U S A. 2011;108:13734–13739. doi: 10.1073/pnas.1105143108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hogestyn JM, Mock DJ, Mayer-Proschel M. Contributions of neurotropic human herpesviruses herpes simplex virus 1 and human herpesvirus 6 to neurodegenerative disease pathology. Neural Regen Res. 2018;13:211–221. doi: 10.4103/1673-5374.226380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Itzhaki RF, Lin WR, Shang D, Wilcock GK, Faragher B, Jamieson GA. Herpes simplex virus type 1 in brain and risk of Alzheimer’s disease. Lancet. 1997;349:241–244. doi: 10.1016/S0140-6736(96)10149-5. [DOI] [PubMed] [Google Scholar]
- 20.Jamieson GA, Maitland NJ, Wilcock GK, Yates CM, Itzhaki RF. Herpes simplex virus type 1 DNA is present in specific regions of brain from aged people with and without senile dementia of the Alzheimer type. J Pathol. 1992;167:365–368. doi: 10.1002/path.1711670403. [DOI] [PubMed] [Google Scholar]
- 21.Kerr JS, Adriaanse BA, Greig NH, Mattson MP, Cader MZ, Bohr VA, Fang EF. Mitophagy and Alzheimer’s disease: cellular and molecular mechanisms. Trends Neurosci. 2017;40:151–166. doi: 10.1016/j.tins.2017.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A, Adachi H, Adams CM, Adams PD, Adeli K, Adhihetty PJ, Adler SG, Agam G, Agarwal R, Aghi MK, Agnello M, Agostinis P, Aguilar PV, Aguirre-Ghiso J, Airoldi EM, et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy. (3rd edition) 2016;12:1–222. doi: 10.1080/15548627.2015.1100356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M, Uchiyama Y, Kominami E, Tanaka K. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature. 2006;441:880–884. doi: 10.1038/nature04723. [DOI] [PubMed] [Google Scholar]
- 24.Kurihara Y, Kanki T, Aoki Y, Hirota Y, Saigusa T, Uchiumi T, Kang D. Mitophagy plays an essential role in reducing mitochondrial production of reactive oxygen species and mutation of mitochondrial DNA by maintaining mitochondrial quantity and quality in yeast. J Biol Chem. 2012;287:3265–3272. doi: 10.1074/jbc.M111.280156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lewandowski G, Zimmerman MN, Denk LL, Porter DD, Prince GA. Herpes simplex type 1 infects and establishes latency in the brain and trigeminal ganglia during primary infection of the lip in cotton rats and mice. Arch Virol. 2002;147:167–179. doi: 10.1007/s705-002-8309-9. [DOI] [PubMed] [Google Scholar]
- 26.Lokensgard JR, Cheeran MC, Hu S, Gekker G, Peterson PK. Glial cell responses to herpesvirus infections: role in defense and immunopathogenesis. J Infect Dis. 2002;186(Suppl 2):S171–179. doi: 10.1086/344272. [DOI] [PubMed] [Google Scholar]
- 27.Mattson MP, Barger SW, Cheng B, Lieberburg I, Smith-Swintosky VL, Rydel RE. beta-Amyloid precursor protein metabolites and loss of neuronal Ca2+ homeostasis in Alzheimer’s disease. Trends Neurosci. 1993;16:409–414. doi: 10.1016/0166-2236(93)90009-b. [DOI] [PubMed] [Google Scholar]
- 28.Meeuwsen S, Persoon-Deen C, Bsibsi M, Bajramovic JJ, Ravid R, De Bolle L, van Noort JM. Modulation of the cytokine network in human adult astrocytes by human herpesvirus-6A. J Neuroimmunol. 2005;164:37–47. doi: 10.1016/j.jneuroim.2005.03.013. [DOI] [PubMed] [Google Scholar]
- 29.Metaxakis A, Ploumi C, Tavernarakis N. Autophagy in age-associated neurodegeneration. Cells. 2018;7:E37. doi: 10.3390/cells7050037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Moore S, Evans LD, Andersson T, Portelius E, Smith J, Dias TB, Saurat N, McGlade A, Kirwan P, Blennow K, Hardy J, Zetterberg H, Livesey FJ. APP metabolism regulates tau proteostasis in human cerebral cortex neurons. Cell Rep. 2015;11:689–696. doi: 10.1016/j.celrep.2015.03.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nagele RG, Wegiel J, Venkataraman V, Imaki H, Wang KC, Wegiel J. Contribution of glial cells to the development of amyloid plaques in Alzheimer’s disease. Neurobiol Aging. 2004;25:663–674. doi: 10.1016/j.neurobiolaging.2004.01.007. [DOI] [PubMed] [Google Scholar]
- 32.Navarro V, Sanchez-Mejias E, Jimenez S, Munoz-Castro C, Sanchez-Varo R, Davila JC, Vizuete M, Gutierrez A, Vitorica J. Microglia in Alzheimer’s disease: activated, dysfunctional or degenerative. Front Aging Neurosci. 2018;10:140. doi: 10.3389/fnagi.2018.00140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nilsson P, Loganathan K, Sekiguchi M, Matsuba Y, Hui K, Tsubuki S, Tanaka M, Iwata N, Saito T, Saido TC. Abeta secretion and plaque formation depend on autophagy. Cell Rep. 2013;5:61–69. doi: 10.1016/j.celrep.2013.08.042. [DOI] [PubMed] [Google Scholar]
- 34.Orvedahl A, Alexander D, Talloczy Z, Sun Q, Wei Y, Zhang W, Burns D, Leib DA, Levine B. HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe. 2007;1:23–35. doi: 10.1016/j.chom.2006.12.001. [DOI] [PubMed] [Google Scholar]
- 35.Piacentini R, De Chiara G, Li Puma DD, Ripoli C, Marcocci ME, Garaci E, Palamara AT, Grassi C. HSV-1 and Alzheimer’s disease: more than a hypothesis. Front Pharmacol. 2014;5:97. doi: 10.3389/fphar.2014.00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Prusty BK, Gulve N, Govind S, Krueger GRF, Feichtinger J, Larcombe L, Aspinall R, Ablashi DV, Toro CT. Active HHV-6 infection of cerebellar purkinje cells in mood disorders. Front Microbiol. 2018;9:1955. doi: 10.3389/fmicb.2018.01955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Readhead B, Haure-Mirande JV, Funk CC, Richards MA, Shannon P, Haroutunian V, Sano M, Liang WS, Beckmann ND, Price ND, Reiman EM, Schadt EE, Ehrlich ME, Gandy S, Dudley JT. Multiscale analysis of independent Alzheimer’s cohorts finds disruption of molecular, genetic, and clinical networks by human herpesvirus. Neuron. 2018;99:64–82.e7. doi: 10.1016/j.neuron.2018.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Reynaud JM, Horvat B. Human herpesvirus 6 and neuroinflammation. ISRN Virol. 2013;2013:11. [Google Scholar]
- 39.Reynaud JM, Jegou JF, Welsch JC, Horvat B. Human herpesvirus 6A infection in CD46 transgenic mice: viral persistence in the brain and increased production of proinflammatory chemokines via Toll-like receptor 9. J Virol. 2014;88:5421–5436. doi: 10.1128/JVI.03763-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Romeo MA, Masuelli L, Gaeta A, Nazzari C, Granato M, Gilardini Montani MS, Faggioni A, Cirone M. Impact of HHV-6A and HHV-6B lytic infection on autophagy and endoplasmic reticulum stress. J Gen Virol. 2019;100:89–98. doi: 10.1099/jgv.0.001176. [DOI] [PubMed] [Google Scholar]
- 41.Santarelli R, Granato M, Faggioni A, Cirone M. Interference with the autophagic process as a viral strategy to escape from the immune control: lesson from gamma herpesviruses. J Immunol Res. 2015;2015:546063. doi: 10.1155/2015/546063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Santarelli R, Granato M, Pentassuglia G, Lacconi V, Gilardini Montani MS, Gonnella R, Tafani M, Torrisi MR, Faggioni A, Cirone M. KSHV reduces autophagy in THP-1 cells and in differentiating monocytes by decreasing CAST/calpastatin and ATG5 expression. Autophagy. 2016;12:2311–2325. doi: 10.1080/15548627.2016.1235122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Uddin MS, Stachowiak A, Mamun AA, Tzvetkov NT, Takeda S, Atanasov AG, Bergantin LB, Abdel-Daim MM, Stankiewicz AM. Autophagy and Alzheimer’s disease: from molecular mechanisms to therapeutic implications. Front Aging Neurosci. 2018;10:04. doi: 10.3389/fnagi.2018.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vassar R, Bennett BD, Babu-Khan S, Kahn S, Mendiaz EA, Denis P, Teplow DB, Ross S, Amarante P, Loeloff R, Luo Y, Fisher S, Fuller J, Edenson S, Lile J, Jarosinski MA, Biere AL, Curran E, Burgess T, Louis JC, et al. Beta-secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science. 1999;286:735–741. doi: 10.1126/science.286.5440.735. [DOI] [PubMed] [Google Scholar]
- 45.Wang Y, Mandelkow E. Degradation of tau protein by autophagy and proteasomal pathways. Biochem Soc Trans. 2012;40:644–652. doi: 10.1042/BST20120071. [DOI] [PubMed] [Google Scholar]
- 46.Wozniak MA, Frost AL, Itzhaki RF. Alzheimer’s disease-specific tau phosphorylation is induced by herpes simplex virus type 1. J Alzheimers Dis. 2009a;16:341–350. doi: 10.3233/JAD-2009-0963. [DOI] [PubMed] [Google Scholar]
- 47.Wozniak MA, Mee AP, Itzhaki RF. Herpes simplex virus type 1 DNA is located within Alzheimer’s disease amyloid plaques. J Pathol. 2009b;217:131–138. doi: 10.1002/path.2449. [DOI] [PubMed] [Google Scholar]
- 48.Wu HZ, Ong KL, Seeher K, Armstrong NJ, Thalamuthu A, Brodaty H, Sachdev P, Mather K. Circulating microRNAs as biomarkers of Alzheimer’s disease: a systematic review. J Alzheimers Dis. 2016;49:755–766. doi: 10.3233/JAD-150619. [DOI] [PubMed] [Google Scholar]
- 49.Zambrano A, Solis L, Salvadores N, Cortes M, Lerchundi R, Otth C. Neuronal cytoskeletal dynamic modification and neurodegeneration induced by infection with herpes simplex virus type 1. J Alzheimers Dis. 2008;14:259–269. doi: 10.3233/jad-2008-14301. [DOI] [PubMed] [Google Scholar]
- 50.Zerboni L, Che X, Reichelt M, Qiao Y, Gu H, Arvin A. Herpes simplex virus 1 tropism for human sensory ganglion neurons in the severe combined immunodeficiency mouse model of neuropathogenesis. J Virol. 2013;87:2791–2802. doi: 10.1128/JVI.01375-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhou F, van Laar T, Huang H, Zhang L. APP and APLP1 are degraded through autophagy in response to proteasome inhibition in neuronal cells. Protein Cell. 2011;2:377–383. doi: 10.1007/s13238-011-1047-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


