Abstract
Background
Despite of a high disease burden, mainly in Latin America, Chagas disease (CD) is underdiagnosed and undertreated. Rapid diagnostic tests (RDTs) might improve the access to diagnosis. The aim of this study is to review the accuracy of commercially available RDTs used in field conditions for the diagnosis of chronic CD in populations at risk, in endemic and non-endemic countries.
Methods/Principal findings
We undertook a comprehensive search of the following databases: PubMed, SCOPUS, LILACS (last up-date on the 01st July, 2017), without language or date limits. Non-electronic sources have been also searched. This review included clinical studies with cohort recruitment of individuals at risk of T. cruzi exposure, without age limits; adequate reference standards for the diagnosis of CD. We excluded case-control studies and those testing RDTs during acute CD. Data on test accuracies were pooled through a bivariate random-effects model. Only one index test was evaluated separately. Geographical area, commercial brand, disease prevalence, study size, and risk of bias were explored as possible source of heterogeneity. Values of sensitivity and specificity were computed to obtain summary positive/negative likelihood ratios, and summary diagnostic odds ratio. Ten studies were included on six different immunochromatographic RDTs. The pooled sensitivity and specificity of the RDTs resulted 96.6% (95% CI 91.3–98.7%) and 99.3% (95% CI 98.4–99.7%), respectively. Test accuracy was particularly good in endemic areas (98.07%/99.03% of sensitivity/specificity, respectively). One test (Stat-Pak) showed an overall sensitivity of 97% (95% CI 87.6–99.3) and specificity of 99.4% (95% CI 98.6–99.8).
Conclusions/Significance
RDTs demonstrated to be sufficiently accurate to recommend their use for screening in endemic areas, even as stand-alone tests. This approach might increase the accessibility to the diagnosis. However, an additional confirmatory test in case of positive result remains a prudent approach.
Author summary
Chagas disease (CD) is a parasitic disease that can affect seriously the health status of affected individuals. People with CD, mainly living in remote areas of Latin America, often face major barriers to the disease recognition, diagnosis and treatment. The World Health Organization recommends the combined use of two tests for diagnosis of the disease in the chronic phase, but this approach is expensive, has time-constraints, and requires well-equipped laboratories, among others. Rapid diagnostic tests (RDTs) are easy-to-use, cheaper and less time-consuming than classical techniques. Hence, their large-scale use could contribute to increase the access to diagnosis, improve treatment coverage, and reduce disease transmission. We reviewed the existing studies on the accuracy of RDTs for the diagnosis of chronic CD. The RDTs under study demonstrated sufficiently good to recommend their use for screening in endemic areas (particularly the Gran Chaco), even as stand-alone tests. On the other hand, not enough evidence has been retrieved on the use of RDTs in other settings. The use of RDTs might increase the access to the diagnosis, particularly in the Gran Chaco area of Latin America.
Introduction
Chagas disease (CD) is a parasitic disease affecting more than 8 million people and causing 806,170 DALYs lost, annually, in the endemic countries of Latin America (LA)[1]. It is caused by the protozoan parasite Trypanosoma cruzi, generally transmitted by insect vectors. Following international migration, the disease has spread also to non-endemic countries, where it can be transmitted congenitally or through organ or blood donation[2]. It has been recently estimated that in Northern America (Mexico, United States and Canada) from 1.3 to 7 million people could be affected[3].
CD has been associated to poverty as it causes relevant morbidity and mortality in working-age people and predominantly affects disadvantaged populations[1]. Moreover, the transplacental transmission[4] causes abortion, stillbirth and complications in newborns. In 30–40% adults, it evolves towards potentially fatal complications after decades of silent progression[2]. Infections in the acute phase and, to a lesser extent the chronic one, can be treated with the aim of cure and interruption of transmission, or at least a reduction in the risk of morbidity[5].
Globally, the disease is largely under-diagnosed (an estimated 90% affected people are unaware of their infection and thus at risk of transmitting it and suffering complications[6]) and under-treated (less than 1% of affected individuals have access to treatment[7]). The World Health Organization (WHO) recommends that the diagnosis of chronic CD should rely on concordant results of at least two different serological tests based on different antigens[8]. Traditionally, conventional tests based on crude antigens/parasite lysate (enzyme-linked immunosorbent assay, ELISA; immunofluorescence test, IFAT; indirect hemagglutination test, IHA) are paired with non-conventional ones (mainly ELISAs) based on recombinant antigens[9]. Blood culture and polymerase chain reaction (PCR) are not considered sufficiently sensitive for the diagnosis during the chronic phase due to the intermittent and low-level peripheral parasitemia found throughout this period[10].
Rapid diagnostic tests (RDTs) are easy-to-use and less technically and time demanding than classical serological techniques. Remarkably, many of them can be performed on serum or with a very little volume of whole blood, and they can be stored on the shelf for longer than a year. Their large-scale use could contribute to increased access to diagnosis, better treatment coverage, and a reduction of disease transmission. Yet, despite having commonly been used for field surveys, RDTs are not recommended by the WHO[11–20].
The aim of this study is to review the accuracy of RDTs in field conditions for the diagnosis of chronic CD in populations at risk living in endemic and non-endemic countries.
Methods
The protocol was registered with Prospero International prospective register of systematic reviews (record: CRD42016025990) on May 6th, 2016.
Search strategy and selection criteria
We searched PubMed, SCOPUS, LILACS on 26th November 2015 and up-dated the search on 01st July 2017, without language or date limits. Original search strategy is available in Prospero (https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=25990). Non-electronic sources have been also searched, like references listed in included studies or non-published data from expert in the field.
Inclusion criteria
a) clinical studies with cohort recruitment (phase III studies) in field conditions; b) the presence of adequate reference tests (from now called "Reference Standard", RS) for the diagnosis of CD, namely a combination of two (or more) serological tests based on different techniques (either ELISA, IFAT or IHA) and antigens according to current WHO recommendations[8], or one or more high specificity test such as radioimmunoprecipitation analysis—RIPA or immunoblot or western blot, or the use of latent class analysis (LCA)-based reference standard; c) studies conducted on individuals (adults or children) with epidemiological risk of exposure to T. cruzi such as living in endemic area for at least one month, receiving blood transfusion in an endemic country or being born to a Latin American mother. We classified studies, on the basis of the sampling method, as being consecutive or non-consecutive. Case-control studies and those testing RDTs during the acute infection phase were excluded.
Data collection
Two authors independently selected the studies, on the basis of the inclusion criteria. In case of discordant opinion, a third author was involved. Data were extracted from selected studies and risk of bias was assessed through the QUADAS-2 tool[21]. As possible sources of heterogeneity, we explored: geographical area, commercial brand of index test, type of RS, disease prevalence, study size, and risk of bias.
Statistical analysis
The values of sensitivity and specificity were automatically computed in RevMan 2014 (Version 5.312). Individual study results were graphically expressed by plotting the estimates of sensitivity and specificity and their 95% confidence intervals (CIs) through both forest plots and receiver operating characteristics (ROC) space. We assessed heterogeneity by visual inspection of forest plots of sensitivity and specificity, and through visual examination of ROC plot of the raw data. Heterogeneity was further investigated using a bivariate random-effects model[22] to obtain estimates of the between-studies variation in sensitivity and specificity and the correlation between the two. The same bivariate model was used to assess the operating point sensitivity and specificity of the diagnostic tests under scrutiny, together with likelihood ratios and summary diagnostic odds ratio (DOR), taking both heterogeneity and threshold effect into account.
All analyses were performed using all articles first, then they were repeated splitting the studies into two main subgroups: studies conducted in endemic areas (continental LA), and studies in non-endemic areas (other continents). This was considered the primary analysis. Based on the results of included studies, we further conducted a secondary analysis on datasets evaluating the RDT most frequently used, i.e. Stat-Pak. All analyses were performed using Stata IC 13.0.
Results
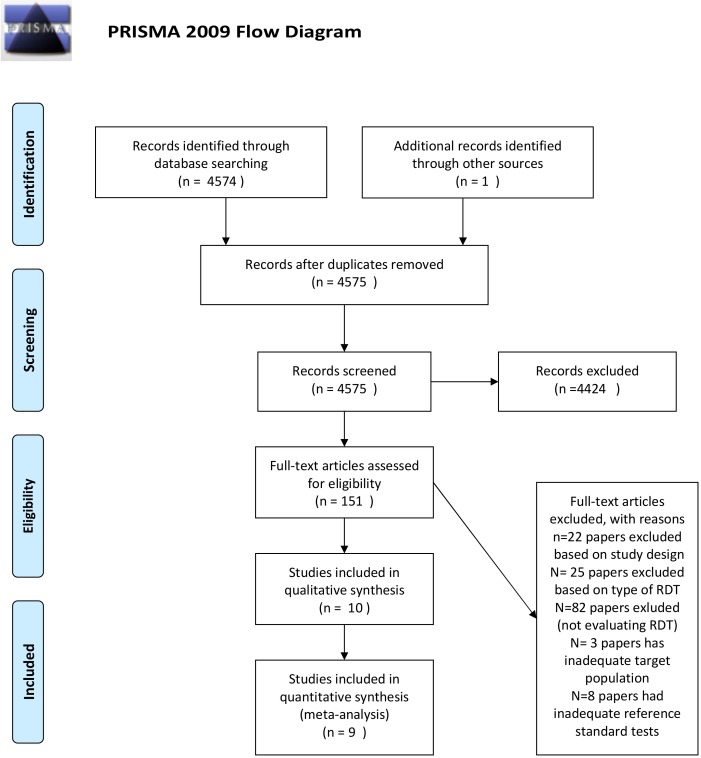
The electronic search identified 4574 records. The study flow is summarized in Fig 1.
Fig 1. Study flow chart.
Amongst the 151 articles included for full text evaluation, we excluded from the analysis studies on RDTs not based on immunochromatographic technique, because the 25 identified studies using other techniques were either evaluating old, out-of-market tests or were old studies for which we could not get access to the full text article[23–47]. Moreover, 22 papers were excluded on the base of the study design[48–69]. Eighty-two articles were excluded because they did not evaluate RDTs accuracy. Three papers were not eligible for inadequate target population (not chronic CD)[70–72], whereas 8 papers declared an inadequate reference standard for inclusion in the review[73–80].
“Eventually, 9 and 10 studies were included in the quantitative and qualitative analyses, respectively”. Two studies evaluated more than one single RDT, hence each dataset from those studies was considered separately[18,19]. One study[81] reported the results of a test conducted on a relatively small number of patients (114 children) that only had true negatives, providing no information on sensitivity. Thus, as we intended to model sensitivity and specificity jointly, we decided to exclude it from our analyses.
Finally, we included in the analysis 12 datasets, comprising a total of 6123 participants (from 101 to 1913 individuals tested in single studies). Six different RDTs were evaluated; Stat-Pak was assessed on more than half of the overall population included in the analysis (4 studies comprising 3347 participants, 53.3% of individuals included in our meta-analysis). Studies evaluating Stat-Pak were quite heterogeneous in terms of age-range of the selected population: one study included participants with more than 16 years of age; Bern et al. enrolled adults, Roddy et al. children and adolescents, and Eguez et al. individuals of all ages[11,18,19]. However, the participants resulted rather homogeneous in terms of origin, as three studies were conducted in Bolivia, and the study implemented in Europe comprised 47.4% of immigrants of Bolivian origin (474 of 999 participants)[12].
Among the other RDTs, one (Simple CHAGASWB, Operon) was evaluated by two studies with a total of 377 participants[15,20]. Both studies were conducted in Spain, and the reported prevalence of CD was 15.9% in the study by Navarro et al. (all ages, 76.4% of participants coming from Bolivia) and 5.9% in the work by Lopez-Chejade et al. (Latin American adults). Simple CHAGASWB showed a sensitivity/specificity of 88%/94.2% and 100%/96.8%, respectively, in Navarro et al. and Lopez-Chejade et al. studies.
Eguez and colleagues assessed the accuracy of a combination of two RDTs (Stat-Pak and InBios) against conventional tests (namely, IHA, lysate-antigen ELISA, and recombinant antigen ELISA)[19]: for the purpose of this review the study was split into two datasets, each one evaluating one RDT, while the combination of Stat-Pak and InBios was not included in the analysis.
Table 1 shows data and characteristics of each RDT and the reference standard for each study.
Table 1. Characteristics of studies.
| References | RDT (index test) | Reference tests | Period of study | Country of implementation | Type and Number of participants | RDT Sensitivity/Specificity |
|---|---|---|---|---|---|---|
| Angheben 2017 [14] | Chagas Quick Test | ELISA para Chagas III, (BioChile, Chile) and Bio-Elisa Chagas, (Biokit, Spain) | 2009–2015 | Italy | Migrants from Latin America, all age, 640 | 83%/99% |
| Bern 2009_a [18] | InBios—Trypanosome detect | In-house IFAT, Chagatek ELISA (BioMerieux, Lab. Lemos, Argentina), and Chagatest ELISA Recombinante (Wiener lab., Argentina) | 2006–2007 | Bolivia | Bolivian pregnant women, 519 | 91%/100% |
| Bern 2009_b [18] | Stat-Pak | 2006–2007 | Bolivia | Bolivian pregnant women, 530 | 90%/100% | |
| Brutus 2008 [17] | InBios—Trypanosome detect | IHA (Polychaco, Argentina) and Chagatest ELISA Recombinante (Wiener lab., Argentina) | 2002–2004 | Bolivia | Bolivian pregnant women, 460 | 93%/99% |
| Chappuis 2010 [12] | Stat-Pak | ELISA cruzi (bioMérieux Diagnostica, Brazil) and Bio-Elisa Chagas, (Biokit, Spain) + results of quality control of a reference lab in Brazil (performing other 4 serology tests) | 2009 | Switzerland | Migrants from Latin America, Adults, 999 | 96%/100% |
| Eguez 2017_a [19] | Stat-Pak | IHA (Polychaco, Argentina), Chagatest ELISA Recombinante (Wiener lab., Argentina), Chagatest ELISA Lisado (Wiener lab., Argentina) | 2014 | Bolivia | Bolivians from >1 years old up to 60 years old), 342 | 99%/100% |
| Eguez 2017_b [19] | InBios—CDP | 90%/100% | ||||
| Lopez-Chejade 2010 [15] | Simple Chagas WB | ELISA in house and BioELISA Chagas | Not declared | Spain | Migrants from Latin America, Adults, 148 | 100%/97% |
| Mendicino 2014 [13] | WL Check Chagas test | Chagastest ELISA, IHA, IFAT for discrepancies | Not declared | Argentina | Patients attending Primary Health Care Centers, 238 | 96%/100% |
| Navarro 2011 [20] | Simple Chagas WB | IFAT and ELISA (not specified) | 2008–2009 | Spain | Migrants from Latin America, all age, 276 | 88%/94% |
| Roddy 2008 [11] | Stat-Pak | Chagastest ELISA, Indirect hemagglutination test (HAI) (Polychaco, Argentina) | 2007 | Bolivia | Bolivians from >6 months to 17,9 years old, 1913 | 93%/99% |
| Shah 2014 [16] | InBios—CDP | Indirect hemagglutination test (HAI) (Polychaco, Argentina), IFAT, Chagatest ELISA Recombinante (Wiener lab., Argentina) or Chagatest ELISA Lisado (Wiener lab., Argentina) | 2011–2012 | Bolivia | Bolivians from >2 to 17 years old, 200 | 100%/99% |
RDT = rapid diagnostic test; ELISA = Enzyme-linked immune assay; IHA = Indirect hemagglutination test; IFAT = Immunofluorescent antibody test
Antigens composition of the RDTs according to manufacturers:
“Chagas Quick Test” is based on T.cruzi specific antigens not better specified;
“InBios—Trypanosome detect” is based on a recombinant multiepitope fusion antigen: ITC8.2;
“Stat-Pak” is based on antigens B13, 1F8 and H49/JL7;
“InBios—CDP” is based on a recombinant multiepitope fusion antigen: ITC8.2;
“Simple Chagas WB” is based on a recombinant multiepitope protein: "Pep2-TcD-TcE-SAPA;
“WL Check Chagas test” is based on T.cruzi specific antigens not better specified.
Four studies were conducted in non-endemic areas (namely, Spain, Switzerland, and Italy)[12,14,15,20]; all studies conducted in LA were carried out in Bolivia, but one that was conducted in Argentina[13]. A couple of studies were conducted in a cohort of children while the others included either adults or individuals of all ages.
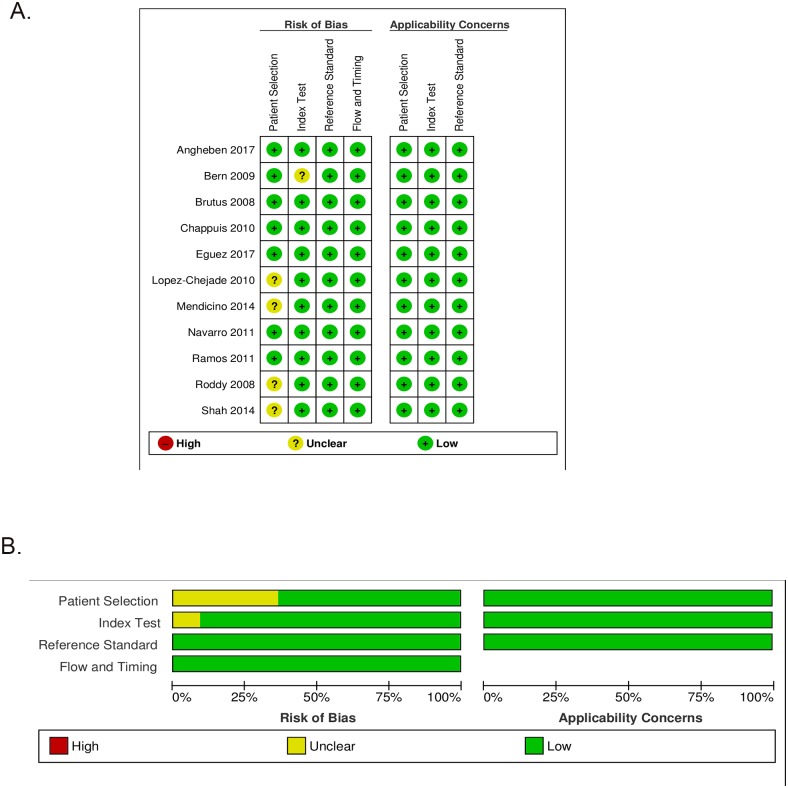
The qualitative evaluation, in terms of rating for each study finally included in the analysis, and their overall methodological quality are shown in Fig 2a and 2b, respectively.
Fig 2.
(a) Risk of bias and applicability concerns graph. (b) Risk of bias and applicability concerns summary.
In general, the risk-of-bias and applicability concerns of all studies analyzed were considered low. Patient selection was assessed as “unclear” in four cases: the main reason was that the papers did not specify methods for the enrollment of patients (consecutive recruitment or random inclusion). Moreover, one study[18] did not clearly state if the results of the index tests were interpreted without knowledge of the results of the RS, hence the risk of bias in relation to the index test was assessed as unclear[18].
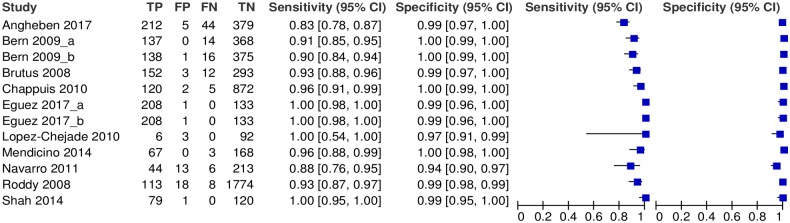
Fig 3 shows the accuracy of the RDTs according to each dataset. Notably, heterogeneity among results of different studies was low, particularly in terms of specificity. Namely, the variance of the logit of the sensitivity resulted 1·82 (95% CI: 0·55 to 5·00), whereas the variance of the logit of specificity was 1·01 (95% CI: 0·29 to 3·41). The correlation between logit of sensitivity and logit of specificity resulted 0·34 (95% CI: -0·50 to 0·86). Globally, the accuracy of all RDTs resulted in: sensitivity = 96.6% (95% CI: 91.3–98.7%) and specificity = 99.3% (95% CI: 98.4–99.7%) (Table 2).
Fig 3. Forest plot displaying the accuracy of the RDTs by study Abbreviations: TP = true positives; FP = false positives; FN = false negatives; TN = true negatives.
Table 2. Summary of findings of the review of immunochromatographic RDTs for the diagnosis of chronic CD in at risk populations.
| Interpretative criteria: Endemicity / Stat-Pak | Effect (95% CI) | # of studies | Mean Prevalencea (95% CI) | What do these results mean? |
|---|---|---|---|---|
| All areas |
Sensitivity: 96.60% (91.3–98.7); Specificity: 99.27% (98.4–99.7) |
12 | 30.33% (18.6 to 42.0) | Of the 30 out of 100 patients with CD, 1 will be missed by a single RDT (3.40% of 30). Of the other 67, not even 1 will have a false positive result of the RDT. |
| Endemic areas |
Sensitivity: 98.07% (91.6–99.6); Specificity: 99.30% (98.3–99.7) |
8 | 35.93% (20.7 to 51.1) | Of the 36 out of 100 patients with CD, not even 1 will be missed by a single RDT (1.93% of 33). Of the other 64, not even 1 will have a false positive result of the RDT. |
| Non-Endemic areas |
Sensitivity: 89.77% (79.7–95.1); Specificity: 98.52% (95.0–99.5) |
4 | 19.14% (4.3 to 42.6) | Of the 19 out of 100 patients with CD, 2 will be missed by a single RDT (10.23% of 19). Of the other 81, 1 will have a false positive result of the RDT. |
| Stat-Pak tests only |
Sensitivity: 97.02% (87.6–99.3); Specificity: 99.44% (98.6–99.8) |
4 | 26.37% (2.9 to 49.8) | Of the 26 out of 100 patients with CD, not even 1 will be missed by a single Stat-Pak test (2.98% of 26). Of the other 74, not even 1 will have a false positive result of the Stat-Pak test. |
a Estimates of true prevalences for each study were calculated as described by Rogan and Gladen (1978) [ref.]. CI: confidence interval; RDT: Rapid Diagnostic Test; CD: Chagas disease.
The RDTs showed better accuracy when used in endemic areas (Table 2): 98.1% and 99.3% respectively averaged sensitivity and specificity, whereas in non-endemic areas their sensitivity resulted lower: about 90%. The overall sensitivity of Stat-Pak was 97% (95% CI 87.6–99.3) and its specificity 99.4% (95% CI 98.6–99.8).
Discussion
Globally, the sensitivity of the RDTs examined was good (higher than 95%) and the specificity was excellent (>99%), regardless their use in endemic or non-endemic regions. The sensitivity was basically higher in endemic (namely Bolivia) than in non-endemic areas. However, it must be noted that the latter data was obtained from four studies comprising 2063 individuals (around one third of the whole study population), with a lower proportion of Bolivians (35,9%, excluding Lopez-Chejade study, where the origin of migrants is not detailed) [12,14,15,20]. A previous study comparing 11 marked-available RDTs found out 8 tests which were considered valuable for clinical purpose (performances generally >90%). However, this was a case-control study based on selected serum samples, hence the accuracy of the tests could be overestimated [66].
In our work, among all RDTs Stat-Pak could be evaluated individually thanks to the high number of individuals tested. This test showed high accuracy, and its functionality with a little volume (10 μl) of whole blood further supports its use for screening purposes, as well as in field surveillance of the disease.
On the other hand, the number of studies (and participants included) addressing the other RDTs was too low to allow a separate meta-analysis. It must also be considered that T. cruzi population is characterized by a genetic polymorphism that might account at least in part for its variability in pathogenicity and transmission. Currently, seven genetic lineages or Discrete Typing Units (DTU) have been characterized, TcI-TcVI and Tcbat[82,83]. Their distribution varies geographically. Most studies included in this work concerned nationals of Bolivia or neighbouring countries (mainly the Gran Chaco area), where the the TcV prevails[83]. Different DTUs are prevalent in other regions (i.e. Mexico and Central America), with different antigenic features, tissue tropism, and pathogenicity profile. Therefore, our results cannot be automatically transferred to other CD epidemiological contexts. Moreover, all studies, both in endemic and non-endemic countries, concerned populations with high prevalence of T. cruzi infection. In populations with lower prevalence, our findings may not be entirely applicable.
On the other hand the Reference Standards, although based on different tests, were generally similar across selected studies: for all 12 datasets at least two paired tests were used (in two studies three tests were used and case definition was based on at least two concordant results, however)[18,19]: this permitted an accurate comparison between studies. We formally assessed risk of bias through the QUADAS-2 tool, and most of the selected studies received high scores, which further contribute to the robustness of our analysis. Finally, the assessment of variation in sensitivity and of the degree of correlation between sensitivity and specificity provided evidence of limited heterogeneity among studies. The utilization of statistical techniques that consider heterogeneity and threshold effect for the estimation of summary measures, such as the bivariate model suggested by Reitsma et al.[22], allowed the achievement of exhaustive and robust estimates.
Applicability of findings
All studies included in our review were conducted under real-life conditions on populations at risk of having chronic CD. The practical implications of this statement are better summarized in Table 2. The use of a RDT would appear more appropriate for endemic than non-endemic areas, as in the latter a RDT-based screening would miss about 2 out of 19 infected subjects among 100 individuals tested. In contrast, in the endemic areas the proportion of infected subjects was higher (36 subjects with CD out of 100 tested), but less than one of the 36 would be missed by the RDT. On the other hand, in all contexts, the number of non-infected subjects erroneously found positive would be absolutely negligible, and the only significant consequence (in case a second, confirmatory test was not performed) would be to propose an unnecessary treatment. Considering only Chagas Stat-Pak test, the figures would be very similar, although in this case a comparison between endemic-non endemic areas was not possible.
While no RDT is sensitive enough to recommend its use for blood/organ donor screening, the technique appears to be sufficiently accurate for the screening of individuals at risk who could benefit from treatment. Considering that a strategy based on a single RDT would be much easier and cheaper to implement than the classical strategy based on two serological tests, it is very plausible to assume that the few cases missed (RDT false negatives) would be amply compensated by a larger population screened. This is especially valuable in rural areas of the endemic countries where access to diagnosis may be problematic. On the other hand, all positive individuals to a RDT should be submitted to a confirmatory test whenever possible, in order to avoid the side effects of an unnecessary treatment, as well as the stigma associated to the infection, for a false-positive subject. Alternatively, one of the studies included in this review[19] suggested the combined use of two RDTs as a strategy to increase the accuracy for screening purpose. The combination reached a near-perfect sensitivity (considering at least one positive out of two) and specificity (both positive).
Conclusions
The accuracy of all RDTs under study can be considered sufficiently good to recommend their use in endemic settings, particularly in the Southern Cone of LA, in order to increase access to diagnosis. The Stat-Pak test can be recommended for use in screening surveys when the expected prevalence is moderately high or high, in the setting of Southern Cone or for migrants from that area in case of non-endemic countries[12]. On the other hand, the pooled sensitivity of all RDTs studied resulted too low to recommend them as stand-alone tests for detection of CD affected individuals in a non-endemic context, as a negative result cannot rule out a T. cruzi infection with reasonable certainty.
The WHO’s principle which states the need of diagnosis confirmation through another serologic test remains a prudent approach that should be followed at least for the confirmation of positive results.
Still, further studies conducting head-to-head comparisons of different available RDTs are needed, and it would be particularly important to extend these studies to the Andean countries, the Amazon basin, Central America and Mexico. Similarly, further studies will be required to ensure the applicability of RDTs in non-endemic settings. Robust evidence from studies of high quality is also needed to advocate adequate control policies and quality assurance in endemic countries, mainly in those with lower prevalence of the disease.
Supporting information
(RTF)
Data Availability
All relevant data are within the manuscript, its Supporting Information file, and from http://dx.doi.org/10.17632/s9v89x33zd.1.
Funding Statement
This work was supported by the Italian Ministry of Health “Fondi Ricerca Corrente - Linea 3, progetto 9” to IRCCS Sacro Cuore - Don Calabria Hospital (AA, DB, FG, GG, ZB). ISGlobal Research group (JAP, JG) receives funds from the Agencia de Gestio destios Universitaris i de Recerca (grant number 2014SGR026). ISGlobal is a member of the CERCA Programme, Generalitat de Catalunya. JG and JAP belong to the Spanish RICET network (grant number RD12/0018/0010). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Lee BY, Bacon KM, Bottazzi ME, Hotez PJ (2013) Global economic burden of Chagas disease: a computational simulation model. Lancet Infect Dis 13: 342–348. 10.1016/S1473-3099(13)70002-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Perez-Molina JA, Molina I (2017) Chagas disease. Lancet. [DOI] [PubMed] [Google Scholar]
- 3.Hotez PJ, Dumonteil E, Betancourt Cravioto M, Bottazzi ME, Tapia-Conyer R, et al. (2013) An unfolding tragedy of Chagas disease in North America. PLoS Negl Trop Dis 7: e2300 10.1371/journal.pntd.0002300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Howard EJ, Xiong X, Carlier Y, Sosa-Estani S, Buekens P (2014) Frequency of the congenital transmission of Trypanosoma cruzi: a systematic review and meta-analysis. BJOG 121: 22–33. 10.1111/1471-0528.12396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rassi A Jr., Rassi A, Marcondes de Rezende J (2012) American trypanosomiasis (Chagas disease). Infect Dis Clin North Am 26: 275–291. 10.1016/j.idc.2012.03.002 [DOI] [PubMed] [Google Scholar]
- 6.Basile L, Jansa JM, Carlier Y, Salamanca DD, Angheben A, et al. (2011) Chagas disease in European countries: the challenge of a surveillance system. Euro Surveill 16. [PubMed] [Google Scholar]
- 7.Ribeiro I, Sevcsik AM, Alves F, Diap G, Don R, et al. (2009) New, improved treatments for Chagas disease: from the R&D pipeline to the patients. PLoS Negl Trop Dis 3: e484 10.1371/journal.pntd.0000484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.WHO (2007) WHO Consultation on International Biological Reference Preparations for Chagas Diagnostic Tests.
- 9.WHO (2002) WHO Expert Committee on the Control of Chagas Disease (2000: Brasilia, Brasil). Geneva.
- 10.Brasil PE, Castro R, Castro L (2016) Commercial enzyme-linked immunosorbent assay versuspolymerase chain reaction for the diagnosis of chronic Chagas disease: a systematic review and meta-analysis. Mem Inst Oswaldo Cruz 111: 1–19. 10.1590/0074-02760150296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roddy P, Goiri J, Flevaud L, Palma PP, Morote S, et al. (2008) Field evaluation of a rapid immunochromatographic assay for detection of Trypanosoma cruzi infection by use of whole blood. J Clin Microbiol 46: 2022–2027. 10.1128/JCM.02303-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chappuis F, Mauris A, Holst M, Albajar-Vinas P, Jannin J, et al. (2010) Validation of a rapid immunochromatographic assay for diagnosis of Trypanosoma cruzi infection among Latin-American Migrants in Geneva, Switzerland. J Clin Microbiol 48: 2948–2952. 10.1128/JCM.00774-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mendicino D, Stafuza M, Colussi C, Barco M, Streiger M, et al. (2014) Diagnostic reliability of an immunochromatographic test for Chagas disease screening at a primary health care centre in a rural endemic area. Mem Inst Oswaldo Cruz 109: 984–988. 10.1590/0074-0276140153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Angheben A, Staffolani S, Anselmi M, Tais S, Degani M, et al. (2017) Accuracy of a Rapid Diagnostic Test (Cypress Chagas Quick Test(R)) for the Diagnosis of Chronic Chagas Disease in a Nonendemic Area: A Retrospective Longitudinal Study. Am J Trop Med Hyg. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lopez-Chejade P, Roca C, Posada E, Pinazo MJ, Gascon J, et al. (2010) [Utility of an immunochromatographic test for Chagas disease screening in primary healthcare]. Enferm Infecc Microbiol Clin 28: 169–171. 10.1016/j.eimc.2009.04.007 [DOI] [PubMed] [Google Scholar]
- 16.Shah V, Ferrufino L, Gilman RH, Ramirez M, Saenza E, et al. (2014) Field evaluation of the InBios Chagas detect plus rapid test in serum and whole-blood specimens in Bolivia. Clin Vaccine Immunol 21: 1645–1649. 10.1128/CVI.00609-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brutus L, Schneider D, Postigo J, Romero M, Santalla J, et al. (2008) Congenital Chagas disease: diagnostic and clinical aspects in an area without vectorial transmission, Bermejo, Bolivia. Acta Trop 106: 195–199. 10.1016/j.actatropica.2008.03.009 [DOI] [PubMed] [Google Scholar]
- 18.Bern C, Verastegui M, Gilman RH, Lafuente C, Galdos-Cardenas G, et al. (2009) Congenital Trypanosoma cruzi transmission in Santa Cruz, Bolivia. Clin Infect Dis 49: 1667–1674. 10.1086/648070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Eguez KE, Alonso-Padilla J, Teran C, Chipana Z, Garcia W, et al. (2017) Rapid diagnostic tests duo as alternative to conventional serological assays for conclusive Chagas disease diagnosis. PLoS Negl Trop Dis 11: e0005501 10.1371/journal.pntd.0005501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Navarro M, Perez-Ayala A, Guionnet A, Perez-Molina JA, Navaza B, et al. (2011) Targeted screening and health education for Chagas disease tailored to at-risk migrants in Spain, 2007 to 2010. Euro Surveill 16. [DOI] [PubMed] [Google Scholar]
- 21.Whiting PF, Rutjes Aw Fau—Westwood ME, Westwood Me Fau—Mallett S, Mallett S Fau—Deeks JJ, Deeks Jj Fau—Reitsma JB, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. [DOI] [PubMed]
- 22.Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, et al. (2005) Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 58: 982–990. 10.1016/j.jclinepi.2005.02.022 [DOI] [PubMed] [Google Scholar]
- 23.Carlier Y, Breniere FS, Lemesre LJ, Carrasco R, Desjeux P, et al. (1985) The interest of immunoprecipitation tests in the immunological diagnosis of Chagas’ disease. Ann Soc Belg Med Trop 65 Suppl 1: 85–94. [PubMed] [Google Scholar]
- 24.Breniere SF, Carrasco R, Miguez H, Lemesre JL, Carlier Y (1985) Comparisons of immunological tests for serodiagnosis of Chagas disease in Bolivian patients. Tropical and Geographical Medicine 37: 231–238. [PubMed] [Google Scholar]
- 25.Camargo E, Hoshino-Shimizu S, Macedo V, Peres BA, Castro C (1977) [Serologic diagnosis of human Trypanosoma cruzi infection. Comparative study of complement fixation, immunofluorescence, hemagglutination and floculation tests in 3,624 serum samples]. Rev Inst Med Trop Sao Paulo 19: 254–260. [PubMed] [Google Scholar]
- 26.Camargo ME, Hoshino-Shimizu S, Umezawa ES (1975) Further evaluation of the "I.M.T.-Chagas flocculation test". A comparison with complement fixation, hemagglutination and immunofluorescence tests. Rev Inst Med Trop Sao Paulo 17: 230–235. [PubMed] [Google Scholar]
- 27.Houghton RL, Stevens YY, Hjerrild K, Guderian J, Okamoto M, et al. (2009) Lateral flow immunoassay for diagnosis of trypanosoma cruzi infection with high correlation to the radioimmunoprecipitation assay. Clinical and Vaccine Immunology 16: 515–520. 10.1128/CVI.00383-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Taylor AER, Smith V (1983) Micro-counterimmunoelectrophoresis: a rapid screening technique for trypanosomiasis. Transactions of the Royal Society of Tropical Medicine and Hygiene 77: 481–486. 10.1016/0035-9203(83)90119-0 [DOI] [PubMed] [Google Scholar]
- 29.Allain DS, Kagan IG (1974) An evaluation of the direct agglutination test for Chagas’ disease. J Parasitol 60: 179–184. [PubMed] [Google Scholar]
- 30.Alviarez Y, Lares M, Viettri M, Aguilar CM, Herrera L, et al. (2014) Standardization of a direct agglutination test for the immunodiagnosis of Chagas disease. Biomédica: revista del Instituto Nacional de Salud 34: 308–317. [DOI] [PubMed] [Google Scholar]
- 31.Apt W, Arribada A, Meza M, Sandoval J (1978) Direct agglutination reaction for the diagnosis of chronic Chagas disease (author’s transl). Revista medica de Chile 106: 16–18. [PubMed] [Google Scholar]
- 32.Cerisola JA, Alvarez M, Wynne de Martini GJ (1980) [Agglutination test using latex particles for the diagnosis of Chagas disease]. Medicina (B Aires) 40 Suppl 1: 132–136. [PubMed] [Google Scholar]
- 33.de Storni PD, de Bolsi FL, Yanovsky JF (1975) [Direct agglutination reaction for the diagnosis of Chagas’ disease. Systematic use of 2-mercaptoethanol for the elimination of nonspecific agglutinins]. Medicina (B Aires) 35: 67–72. [PubMed] [Google Scholar]
- 34.Enders B, Hungerer KD, Zwisler O (1975) Survey on experiences with latex-Chagas-test in various countries. Tropenmed Parasitol 26: 252–260. [PubMed] [Google Scholar]
- 35.Katzin AM, Marcipar A, Freilij H, Corral R, Yanovsky JF (1989) Rapid determination of Trypanosoma cruzi urinary antigens in human chronic chagas disease by agglutination test. Experimental Parasitology 68: 208–215. [DOI] [PubMed] [Google Scholar]
- 36.Knierim F, Eskuche G, Sandoval L, Muñoz E (1971) Latex agglutination reaction for the serodiagnosis of Chagas’ disease. Boletin Chileno de Parasitologia 26: 32–35. [PubMed] [Google Scholar]
- 37.Pellegrino J, Katz N (1971) Comparison of the latex slide agglutination and complement fixation tests for the diagnosis of Chagas’ disease. Journal of Parasitology 57: 771 [PubMed] [Google Scholar]
- 38.Peralta JM, Magalhaes TC, Abreu L, Manigot DA, Luquetti A, et al. (1981) The direct agglutination test for chronic Chagas’s disease. The effect of pre-treatment of test samples with 2-mercaptoethanol. Trans R Soc Trop Med Hyg 75: 695–698. 10.1016/0035-9203(81)90152-8 [DOI] [PubMed] [Google Scholar]
- 39.Añez N, Atencio R, Rivero Z, Bracho A, Rojas A, et al. (2011) Detección de infecciones inaparentes de la enfermedad de Chagas en individuos asintomáticos de la etnia Yukpa del occidente de Venezuela. Boletin de Malariologia y Salud Ambiental 51: 167–175. [Google Scholar]
- 40.Añez N, Crisante G, Rojas A, Carrasco H, Parada H, et al. (2001) Detection and significance of inapparent infection in Chagas disease in western Venezuela. American Journal of Tropical Medicine and Hygiene 65: 227–232. [DOI] [PubMed] [Google Scholar]
- 41.Biancardi MA, Morena MC, Torres N, Pepe C, Altcheh J, et al. (2003) Seroprevalence of Chagas disease in 17 rural communities of "monte impenetrable", chaco province. Medicina 63: 125–129. [PubMed] [Google Scholar]
- 42.De Godoy I, Meira DA (2007) Seroprevalence of Chagas infection among inhabitants of municipalities in the Botucatu region, State of São Paulo. Revista da Sociedade Brasileira de Medicina Tropical 40: 516–520. [DOI] [PubMed] [Google Scholar]
- 43.Kagan IG, Goldsmith RS, Zarate-Castaneda R, Allain DS (1979) [Evaluation of serological tests used for the study of Chagas’ disease]. Bol Oficina Sanit Panam 87: 309–318. [PubMed] [Google Scholar]
- 44.Knierim F, Rubinstein P (1970) The detection of Chagas’ disease. A rapid haemagglutination test for special use in blood banks and epidemiological studies. Vox Sang 18: 280–286. [DOI] [PubMed] [Google Scholar]
- 45.Schattschneider W, Lopes ER, De Alencar JE, Bienzle U, Feldmeier H (1992) A comparative study of four serological methods for diagnosis of acute and chronic Chagas’ disease in Brazilian patients. Tropical and Geographical Medicine 44: 210–218. [PubMed] [Google Scholar]
- 46.Streiger M, Fabbro D, Del Barco M, Beltramino R, Bovero N (1995) Congenital Chagas disease in the city of Santa Fe, Argentina. Diagnosis and treatment. Medicina 55: 125–133. [PubMed] [Google Scholar]
- 47.Szarfman A, Otatti L, Schmunis GA, Vilches AM (1973) A simple method for the detection of human congenital Chagas’ disease. J Parasitol 59: 723 [PubMed] [Google Scholar]
- 48.da Silveira JF, Umezawa ES, Luquetti AO (2001) Chagas disease: recombinant Trypanosoma cruzi antigens for serological diagnosis. Trends Parasitol 17: 286–291. [DOI] [PubMed] [Google Scholar]
- 49.Gabrielli S, Girelli G, Vaia F, Santonicola M, Fakeri A, et al. (2013) Surveillance of Chagas disease among at-risk blood donors in Italy: preliminary results from Umberto I Polyclinic in Rome. Blood Transfus 11: 558–562. 10.2450/2013.0055-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Añez N, Romero M, Crisante G, Bianchi G, Parada H (2010) Comparative evaluation of serologic diagnostic tests used to detect Chagas disease in Venezuela. Boletin de Malariologia y Salud Ambiental 50: 17–27. [Google Scholar]
- 51.Barfield CA, Barney RS, Crudder CH, Wilmoth JL, Stevens DS, et al. (2011) A highly sensitive rapid diagnostic test for Chagas disease that utilizes a recombinant Trypanosoma cruzi antigen. IEEE Transactions on Biomedical Engineering 58: 814–817. 10.1109/TBME.2010.2087334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Garcia VS, Gonzalez VD, Marcipar IS, Gugliotta LM (2014) Immunoagglutination test to diagnose Chagas disease: comparison of different latex-antigen complexes. Trop Med Int Health 19: 1346–1354. 10.1111/tmi.12379 [DOI] [PubMed] [Google Scholar]
- 53.Nagasse-Sugahara TK, Hoshino-Shimizu S, Pagliarini RC, Celeste BJ (1996) Improvement of the slide hemagglutination test for rapid Chagas’ disease screening in epidemiological surveys. Braz J Med Biol Res 29: 623–628. [PubMed] [Google Scholar]
- 54.Ponce C, Ponce E, Vinelli E, Montoya A, de Aguilar V, et al. (2005) Validation of a rapid and reliable test for diagnosis of chagas’ disease by detection of Trypanosoma cruzi-specific antibodies in blood of donors and patients in Central America. J Clin Microbiol 43: 5065–5068. 10.1128/JCM.43.10.5065-5068.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.De Marchi CR, Amato Neto V, Santos AGd (2005) Avaliação rotineira de prova de execução rápida (ELISA—ImmunoComb II Chagas Ab) para diagnóstico sorológico da doença de Chagas^ipt, An evaluation of a quick test for the serological diagnosis of Chagas disease (ELISA—ImmunoComb II Chagas Ab) in routine work^ien. Rev Soc Bras Clín Méd 3: 134–136. [Google Scholar]
- 56.Duarte LF, Florez O, Rincon G, Gonzalez CI (2014) Comparison of seven diagnostic tests to detect Trypanosoma cruzi infection in patients in chronic phase of Chagas disease. Colomb Med (Cali) 45: 61–66. [PMC free article] [PubMed] [Google Scholar]
- 57.Flores-Chavez M, Cruz I, Nieto J, Garate T, Navarro M, et al. (2012) Sensitivity and specificity of an operon immunochromatographic test in serum and whole-blood samples for the diagnosis of Trypanosoma cruzi infection in Spain, an area of nonendemicity. Clin Vaccine Immunol 19: 1353–1359. 10.1128/CVI.00227-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Flores-Chávez M, Cruz I, Rodríguez M, Nieto J, Franco E, et al. (2010) Comparison of conventional and non-conventional serological tests for the diagnosis of imported Chagas disease in Spain. Enfermedades Infecciosas y Microbiologia Clinica 28: 284–293. 10.1016/j.eimc.2009.07.005 [DOI] [PubMed] [Google Scholar]
- 59.Hoshino-Shimizu S, Camargo ME, Umezawa ES (1975) A rapid slide flocculation test for the diagnosis of American trypanosomiasis using Trypanosoma cruzi fragments preserved by lyophilization. Comparison with hemagglutination, immunofluorescence, and complement fixation tests. Am J Trop Med Hyg 24: 586–589. 10.4269/ajtmh.1975.24.586 [DOI] [PubMed] [Google Scholar]
- 60.Ji MJ, Noh JS, Cho BK, Cho YS, Kim SJ, et al. (2009) [Evaluation of SD BIOLINE Chagas Ab Rapid kit]. Korean J Lab Med 29: 48–52. 10.3343/kjlm.2009.29.1.48 [DOI] [PubMed] [Google Scholar]
- 61.Lorca M, Contreras MC, Salinas P, Guerra A, Raychaudhuri S (2008) Evaluation of a rapid diagnosis test for Trypanosoma cruzi infection in serum sample. Parasitologia Latinoamericana 63: 29–33. [Google Scholar]
- 62.Luquetti AO, Ponce C, Ponce E, Esfandiari J, Schijman A, et al. (2003) Chagas’ disease diagnosis: a multicentric evaluation of Chagas Stat-Pak, a rapid immunochromatographic assay with recombinant proteins of Trypanosoma cruzi. Diagn Microbiol Infect Dis 46: 265–271. [DOI] [PubMed] [Google Scholar]
- 63.Partel CD, Rossi CL (1998) A rapid, quantitative enzyme-linked immunosorbent assay (ELISA) for the immunodiagnosis of Chagas’ disease. Immunol Invest 27: 89–96. [DOI] [PubMed] [Google Scholar]
- 64.Peralta JM, Magalhães TCR, Abreu L, Manigot DA, Luquetti A, et al. (1981) The direct agglutination test for chronic Chagas’s disease. The effect of pre-treatment of test samples with 2-mercaptoethanol. Transactions of the Royal Society of Tropical Medicine and Hygiene 75: 695–698. 10.1016/0035-9203(81)90152-8 [DOI] [PubMed] [Google Scholar]
- 65.Reithinger R, Grijalva MJ, Chiriboga RF, de Noya BA, Torres JR, et al. (2010) Rapid detection of Trypanosoma cruzi in human serum by use of an immunochromatographic dipstick test. J Clin Microbiol 48: 3003–3007. 10.1128/JCM.02474-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sanchez-Camargo CL, Albajar-Vinas P, Wilkins PP, Nieto J, Leiby DA, et al. (2014) Comparative evaluation of 11 commercialized rapid diagnostic tests for detecting Trypanosoma cruzi antibodies in serum banks in areas of endemicity and nonendemicity. J Clin Microbiol 52: 2506–2512. 10.1128/JCM.00144-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Teixeira MdGM, Borges-Pereira J, Peralta J (1994) Avaliaçåo de testes sorológicos aplicados para a detecçåo de infecçåo pelo Trypanosoma cruzi. Rev bras patol clín 30: 133–139. [Google Scholar]
- 68.Araujo FG (1985) A method for demonstration of antibodies to Trypanosoma cruzi by using antigen-coated nitrocellulose paper strips. American Journal of Tropical Medicine and Hygiene 34: 242–245. 10.4269/ajtmh.1985.34.242 [DOI] [PubMed] [Google Scholar]
- 69.Verani JR, Seitz A, Gilman RH, LaFuente C, Galdos-Cardenas G, et al. (2009) Geographic variation in the sensitivity of recombinant antigen-based rapid tests for chronic Trypanosoma cruzi infection. Am J Trop Med Hyg 80: 410–415. [PubMed] [Google Scholar]
- 70.Cortina ME, Melli LJ, Roberti M, Mass M, Longinotti G, et al. (2016) Electrochemical magnetic microbeads-based biosensor for point-of-care serodiagnosis of infectious diseases. Biosens Bioelectron 80: 24–33. 10.1016/j.bios.2016.01.021 [DOI] [PubMed] [Google Scholar]
- 71.Bryan CF, Tegtmeier GE, Rafik N, Markham LE, Murillo D, et al. (2004) The risk for Chagas’ disease in the Midwestern United States organ donor population is low. Clinical Transplantation, 18 Supplement: 12–15. [DOI] [PubMed] [Google Scholar]
- 72.de Noya BA, Díaz-Bello Z, Colmenares C, Zavala-Jaspe R, Abate T, et al. (2012) The performance of laboratory tests in the management of a large outbreak of orally transmitted Chagas disease. Memorias do Instituto Oswaldo Cruz 107: 893–898. [DOI] [PubMed] [Google Scholar]
- 73.Roca C, Pinazo MJ, Lopez-Chejade P, Bayo J, Posada E, et al. (2011) Chagas disease among the Latin American adult population attending in a primary care center in Barcelona, Spain. PLoS Negl Trop Dis 5: e1135 10.1371/journal.pntd.0001135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Soriano Arandes A, Munoz Gutierrez J, Verges Navarro M, Castells Domenech C, Portus Vinyeta M, et al. (2009) Prevalence of Chagas disease in the Latin American immigrant population in a primary health centre in Barcelona (Spain). Acta Trop 112: 228–230. 10.1016/j.actatropica.2009.07.014 [DOI] [PubMed] [Google Scholar]
- 75.Marin C, Concha-Valdez F, Canas R, Gutierrez-Sanchez R, Sanchez-Moreno M (2014) Anti-Trypanosoma cruzi antibody detection in eastern Andalusia (Spain). Trans R Soc Trop Med Hyg 108: 165–172. 10.1093/trstmh/tru001 [DOI] [PubMed] [Google Scholar]
- 76.Chippaux JP, Postigo JR, Santalla JA, Schneider D, Brutus L (2008) Epidemiological evaluation of Chagas disease in a rural area of southern Bolivia. Trans R Soc Trop Med Hyg 102: 578–584. 10.1016/j.trstmh.2008.03.008 [DOI] [PubMed] [Google Scholar]
- 77.Chippaux JP, Santalla JA, Postigo JR, Romero M, Salas Clavijo NA, et al. (2009) Sensitivity and specificity of Chagas Stat-Pak test in Bolivia. Trop Med Int Health 14: 732–735. 10.1111/j.1365-3156.2009.02288.x [DOI] [PubMed] [Google Scholar]
- 78.Gamboa-León R, Ramirez-Gonzalez C, Pacheco-Tucuch FS, O’Shea M, Rosecrans K, et al. (2014) Seroprevalence of Trypanosoma cruzi among mothers and children in rural Mayan communities and associated reproductive outcomes. American Journal of Tropical Medicine and Hygiene 91: 348–353. 10.4269/ajtmh.13-0527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Padilla-Raygoza N, Gamboa-León R, Ramirez-Sierra MJ, Dumonteil E, Buekens P, et al. (2015) Negative studies are helpful to compute the specificity of diagnostic tests: measuring Trypanosoma cruzi seroprevalence in Guanajuato, Mexico. BMC Research Notes 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sosa-Estani S, Gamboa-Leon MR, Del Cid-Lemus J, Althabe F, Alger J, et al. (2008) Use of a rapid test on umbilical cord blood to screen for Trypanosoma cruzi infection in pregnant women in Argentina, Bolivia, Honduras, and Mexico. Am J Trop Med Hyg 79: 755–759. [PubMed] [Google Scholar]
- 81.Ramos JM, Flores-Chavez M, Fernandez-Planelles MC, Gutierrez F (2011) [Use of an immunochromatographic test for Trypanosoma cruzi infection screening in a pediatric population]. Enferm Infecc Microbiol Clin 29: 470–471. 10.1016/j.eimc.2011.01.012 [DOI] [PubMed] [Google Scholar]
- 82.Lima L, Espinosa-Alvarez O, Ortiz PA, Trejo-Varon JA, Carranza JC, et al. (2015) Genetic diversity of Trypanosoma cruzi in bats, and multilocus phylogenetic and phylogeographical analyses supporting Tcbat as an independent DTU (discrete typing unit). Acta Trop 151: 166–177. 10.1016/j.actatropica.2015.07.015 [DOI] [PubMed] [Google Scholar]
- 83.Zingales B (2017) Trypanosoma cruzi genetic diversity: Something new for something known about Chagas disease manifestations, serodiagnosis and drug sensitivity. Acta Trop. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(RTF)
Data Availability Statement
All relevant data are within the manuscript, its Supporting Information file, and from http://dx.doi.org/10.17632/s9v89x33zd.1.