Abstract
Lung cancer is one of the leading causes of cancer-related death worldwide. Non-small cell lung cancer (NSCLC) causes around 80% to 90% of deaths. The lack of an early diagnosis and inefficiency in conventional therapies causes poor prognosis and overall survival of lung cancer patients. Recent progress in nanomedicine has encouraged the development of an alternative theranostics strategy using nanotechnology. The interesting physico-chemical properties in the nanoscale have generated immense advantages for nanoparticulate systems for the early detection and active delivery of drugs for a better theranostics strategy for lung cancer. This present review provides a detailed overview of the recent progress in the theranostics application of nanoparticles including liposomes, polymeric, metal and bio-nanoparticles. Further, we summarize the advantages and disadvantages of each approach considering the improvement for the lung cancer theranostics.
Keywords: lung cancer, nanomedicine, theranostics, clinical status, cancer therapy
1. Introduction
Lung cancer is one of the prevalent malignancies and leading causes of cancer-related mortality and the most common cancer in men, accounting for an estimated 154,050 deaths in 2018 and worldwide [1,2,3]. As per the GLOBOCAN, a project of the International Agency for Research on Cancer (IARC), estimation, approximately 18.1 million new cancer cases would have been detected in 2018 [2]. This dreadful disease owes its origin mostly (85%) to long term tobacco smoking [4]. It turns out that at the time of diagnosis lung cancer in many patients has metastasized to other tissues in the body. According to the 2012 global lung cancer statistics, an estimated 1.8 million new cases were reported 58% of which occurred in less developed countries [5]. The American Cancer Society reported over 221,200 estimated new cases of lung cancer and 158,040 death cases in 2015 in the United States [2,3]. Around 85% of lung cancer patients had non-small cell lung cancer while the remaining 15% had small cell lung cancer. The survival rate in lung cancer patients depends primarily on early diagnosis and surgical resection of the tumor tissue is often the preferred therapy [6,7]. Clinically used therapeutic modalities are still associated with a poor outcome: Only <20% five-year overall survival is reported as cancer cells routinely become impervious to drugs. Among all the presently available cancer therapies, chemotherapy is the most widely used treatment strategy for lung cancer. The insufficient drug concentration in the tumor tissue is one of the major impediments retarding the clinical success of lung cancer chemotherapy. To meet this up-hill therapeutic challenge, repeated applications of anti-cancer agents at high concentrations are being used in systemic chemotherapy which is causing adverse side effects. The commonly observed adverse side effects in chemotherapy based cancer treatments mostly originate from the ability of many potent cytotoxic drugs to penetrate non-cancerous healthy body tissues in addition to tumor tissues [8]. Novel therapeutic modalities for more effective treatment of lung cancer are, therefore, urgently needed.
In recent years, enormous endeavors have been directed toward the development of new warheads and their effective carriers to reduce the probability of occurrences of multi-drug resistance, combinations of hydrophilic with hydrophobic drugs, small interfering RNA (siRNAs) with hydrophobic drugs being majorly explored among them [9]. Global efforts are also being witnessed on delivering potent anti-cancer drugs selectively to tumor tissues by encapsulating the drugs within various types of drug carriers [10,11,12]. The exo-surfaces of such drug carriers are covalently grafted with tumor-specific targeting motifs including small peptides, aptamers, proteins, and antibodies [13,14,15,16,17].
Much to our intrigue, theranostics (therapy plus diagnostic) nanomedicine has emerged as a propitious paradigm in cancer therapy. It involves advantages of the both world: highly efficacious nanocarriers to ferry cargo while loading onto them both imaging and therapeutic agents. The idea spawned a plethora of nanoparticles, well suitable for drug delivery as well as diagnosis, facilitating the advent of personalized medicine. There exists four crucial aspects to take into account while designing an efficacious nanoplatform based therapeutics: (i) Selecting a potent therapeutic, ranging from small molecule drugs to larger peptide or nucleic acid; (ii) to choose a stable carrier; (iii) to adopt a targeting and drug release strategy; and (iv) to carefully single out an imaging agent [18,19,20,21,22,23,24].
The present review is an attempt to update major advancements in lung cancer theranostics: A description of development of various nanosystems (liposomes, polymeric nanoparticles, metal nanoparticles, bio-nanoparticles, etc.) for efficient delivery of an array of theranostics in lung cancer.
2. Lung Cancer: Category, Cause, Molecular Target, and Limitations of Conventional Therapy
Non-small cell lung cancer is the most prevalent cancer and can be further categorized into two major subtypes which include small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC) based on the histological appearance. Though SCLC is more aggressive but less frequent as compared to NSCLC. NSCLC can be sub-classified into three major histological subtypes: Adenocarcinoma, squamous cell-carcinoma, and large cell-lung cancer. Again, each of the subtypes is distinct and responds differently to available therapies.
Lung carcinogenesis is influenced by the interaction of ecological factors, for example, tobacco smoke, and genetic susceptibility. The cancer susceptibility increases significantly with rare germ line mutations like p53, retinoblastoma, epidermal growth factor receptor (EGFR), etc. Moreover, reduced DNA repair efficiency may also play a critical role in lung carcinogenesis [25]. Chemicals in tobacco smoke are reported to play a major role in lung carcinogenesis. Research advances have provided clear evidence for the role of tyrosine kinases in the pathophysiology of lung cancer. Constitutive kinase activation and downstream signaling may arise due to mutation(s), overexpression, and autocrine paracrine incitement, leading to cancer. Oncogenic activation of tyrosine kinase like EGFR, PIK3CA, MET etc. are frequently observed in NSCLC and thereby offer opportunity for therapeutic targeting [25,26].
Despite the fact that progresses are made in the treatment of non-small cell lung cancer (NSCLC), the five-year survival rate for lung malignant growth has expanded by just 5% in recent years. Surgery serves an important treatment modality of early stage disease but surgery for lung cancer is complex and can have serious consequences. Early detection of lung malignant growth is another hurdle in effective treatment of lung cancer patients. Roughly seventy five percent of patients with lung disease will present symptoms, and the majority of these have an advanced stage of tumor at the time of diagnosis.
The emergence of cancer chemotherapy and radiation therapy is partially effective in the initial stage of NSCLC treatment. Due to the advent of robust sequencing techniques and initiation of large genome wide association studies it is quite clear that molecular heterogeneity exists even in the same cancer subtypes. Heterogeneity can exist among the primary tumor and the metastatic counterpart, even within the cells of a particular tumor or based on the cell of origin. The molecular diversity and the cancer cells ability to acquire adaptive resistance create a definite challenge in planning an effective therapy [27]. A new approach addressing patient specific molecular specificities and patient centered approach is needed.
3. Alternative Theranostics by Nanomedicine
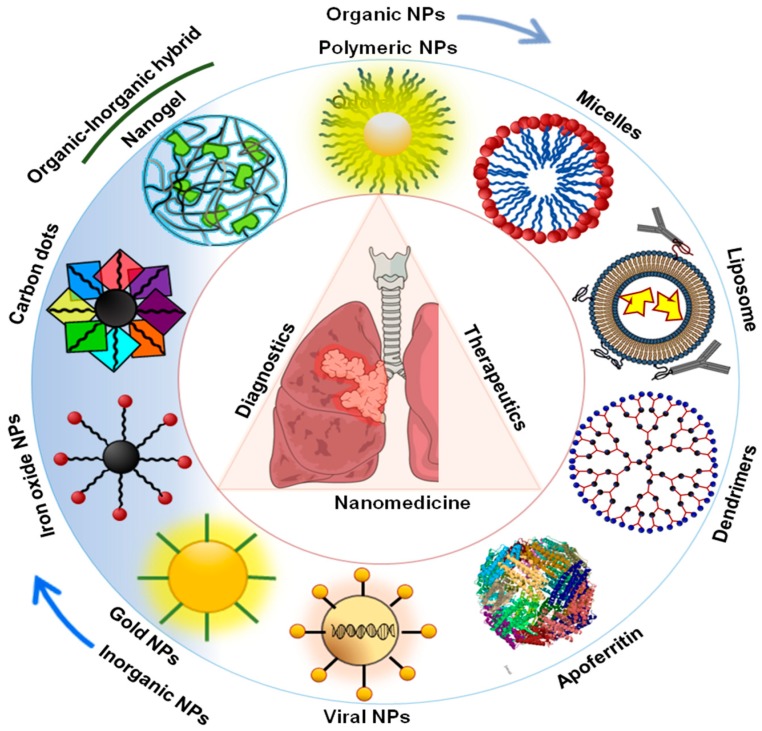
Nanotechnology is one of the fast growing fields in the area of biomedical science that has been utilized to solve different biological problems including therapeutics and diagnostics. Recently, nanotechnology has been widely utilized for the treatment of various diseases including cancer, diabetes, bacterial infections, cardiovascular diseases, etc. [28,29,30]. Due to several limitations in the conventional therapeutic strategies for lung cancers, scientists and researchers have focused on the development of the nanoscale therapeutic agents, whereas the delivery system includes liposomal nanoparticles, polymeric nanoparticles, metal nanoparticles, and bio-nano particles. The lung cancer theranostics applications of these nanoparticles has been largely effective due to their small size, that enables them to specifically accumulate in tumor cells due to an enhanced permeability and retention effect (EPR) [31]. Moreover, nanoparticles are easy to functionalize and demonstrates high drug loading due to large surface area to volume [29]. Apart from that, due to good biocompatibility and the capability of overcoming clearance by the kidney effacing long circulation nanoparticles holds edge over conventional therapeutic treatments. Moreover, several nanoparticles displayed multifunctional abilities like imaging, diagnostics, therapeutics, sensing that helps researchers to utilize these nanomaterials for multifunctional biomedical applications in lung cancer theranostics. Here in this review, we have focused on the theranostics applications of various types of nanomaterials that have shown enormous promise for the applications in lung cancer diagnosis and therapy including (a) liposomal nanoparticles, (b) polymeric nanoparticles, (c) bio-nanoparticles, and (d) metal nanoparticles (Figure 1).
Figure 1.
Schematic representation of different theranostics nanomedicine approaches in lung cancer theranostics.
4. Liposomal Nanoparticles for Lung Cancer Theranostics
Liposomes are artificially generated vesicles with a bilayer structure spontaneously formed when natural or synthetic amphiphatic lipids get dispersed in water. Ever since their inception, they have largely been explored as drug delivery vehicles because of their biocompatibility and beneficial safety profile. The bilayer structure of liposomes comprises of phosphatidylcholine, cholesterol, etc. and they are known to carry an array of small molecule and large molecule therapeutics of hydrophobic and hydrophilic origin. Their surface can be modified by grafting polyethylene glycol (PEG), which prolongs their half-life in circulation [32,33]. Doxil and Myocet, two leading Doxorubicin liposomes, received FDA approval in 1995 and 1999, respectively, followed by many of the category [34,35]. Despite availability of around sixteen liposomal drugs in the market as of now, very few formulations are designated as the treatment modality for NSCLC. Here we summarize a few recent examples of liposomes for pulmonary delivery of therapeutics.
In 2014, Cheng et al. [36] exploited EGFR binding affinity of a novel peptide GE11 in doxorubicin-loaded liposomes and characterized them in terms of size distribution, zeta potential, drug entrapment efficiency, and morphology. It turned out that optimal GE11 density was 10% in A549 cytotoxicity. Major involvement of clathrin-mediated endocytosis pathway was also determined by cellular uptake experiments. Using a near infrared (NIR) fluorescence imaging system, they found that the accumulation and retention of the GE-11 modified liposomes was 2.2-fold higher compared to unmodified liposomes [36]. For effective delivery of triptolide (TPL) to NSCLC by pulmonary administration, a dual ligand (anti-carbonic anhydrase IX (anti-CA IX) antibody and CPP33) modified triptolide-loaded liposomes (dl-TPL-lip) was designed, synthesized, and characterized by Lin et al. [37] in 2018. The cell killing ability was evaluated by an apoptosis assay. Importantly, superior tumor penetration and tumor growth inhibition efficacy of the liposomes were further demonstrated using 3D tumor spheroids. Pharmacokinetics studies in rats after endotracheal administration of the liposomal formulations exhibited a lower concentration of TPL in circulation [37]. In 2017, Song et al. [38] came up with a multifunctional targeting liposome for the treatment of NSCLC and achieved better in vivo effects. They decorated the liposome surface by Octreotide (OCT), a synthetic 8-peptide analog of somatostatin that binds to somatostatin receptors overexpressed in a variety of tumors. Two drugs were co-encapsulated in the liposome: Honokiol into the lipid bilayer to reduce tumor metastasis and inhibit vasculogenic mimicry channel formations, and epirubicin into the aqueous core as an antitumor drug. Mechanistic investigation studies revealed that these liposomes could downregulate PI3K, MMP-2, MMP-9, VE-Cadherin, and FAK and activate caspase 3 [38]. In 2013, Mukherjee et al. designed and synthesized a series of guanidinylated cationic amphiphiles and demonstrated that systemic administration of a 19 bp synthetic CDC20 siRNA encapsulated within liposomes of guanidinylated cationic amphiphile with stearyl tails inhibits B16F10 solid tumor growth and intravenous administration of the same liposomal formulation inhibits B16F10 melanoma growth on lung (metastases) in a syngeneic C57BL/6J mouse tumor model [39].
As a suitable alternative, pulmonary drug delivery strategy, i.e., local delivery via inhalation has gained substantial attention of researchers across the globe. This enables us to reach higher local drug concentration to the specific site of action. This also demands a low dose of therapeutics with reduced toxicity. We intend to provide few examples of liposomes to be inhaled as reported in the last two decades: In 2000, Anderson PM and coworkers designed a phase I study to examine the efficacy and toxicity of administering interleukin (IL)-2 liposomes by aerosol to patients with lung metastases. The liposome-aerosol was inhaled for ~20 min thrice a day. The dose chosen was based on previous studies. Nine patients were treated in three cohorts of three patients at varying IU of IL-2 thrice a day. Authors reported that inhalation of IL-2 liposomes is well tolerated with any reduced systemic toxicity [40]. In another phase I dose-escalating study performed by Wittgen et al. [41] safety, efficacy and PK of aerosolized liposomal (sustained release lipid inhalation targeting, SLIT) cis-platin were investigated in patients having lung cancer. In seventeen patients SLIT cis-platin was well tolerated. No nephrotoxicity, ototoxicity, hematologic toxicity, or neurotoxicity was revealed by safety data. Together, they concluded that the use of aerosolized liposomal cis-platin was feasible and safe [41]. Importantly, in 2010, Makale and co-workers developed a PEGylated DOX liposome with a dextran core, containing iron oxide for MRI contrast and Bodipy for fluorescence and demonstrated robust imaging ability of these nanoparticles (NPs) in an in vivo murine Lewis lung carcinoma model [42]. In 2011, Lowery et al. [43] decorated the surface of a DOX entrapped liposome with a phage displayed peptide HVGGSSV to achieve radiation guided selective drug delivery to tumors. Liposomes were labeled with Alexa Fluor 750 and the biodistribution of the labeled liposomes was studied in a murine Lewis lung carcinoma model by near infrared (NIR) imaging [43]. The liposomes are widely applied for lung cancer theranostics as they show immense promise due to their excellent biocompatibility and biodegradability [44,45]. Moreover, liposomes hold the edges over other nanoparticles as they are useful to load a high amount of therapeutic agents and can be easily controlled for sustained drug delivery [44,45]. However, liposomal nanoparticles have some disadvantages like batch to batch variability, high manufacturing cost, possible drug leakage, etc. [44]. Hence, careful fabrication strategies are required to overcome these challenges and reduction of the cost.
5. Polymeric Nanoparticles for Lung Cancer Theranostics
Polymeric NPs, on the other hand, can be prepared either by nanoprecipitation or a double emulsion method via self-assembly of biodegradable amphiphilic block-copolymers with varying hydrophobicity’s between blocks and are suitable for systemic administration. The core-shell structure of polymeric NPs facilitates encapsulation of hydrophobic drugs, extension of circulation time, and sustained drug release. Their surfaces can also be decorated for targeted drug delivery [46,47]. For instance, Genexol-PM is a formulation of paclitaxel and poly (D,L-lactide)-b-polyethylene glycol-methoxy (PLGA-mPEG), which is already marketed for metastatic breast cancer therapy in Korea and other European countries [48,49]. Here we summarize a few recent interesting examples of usage of polymeric NPs for treatment of lung cancer.
In 2015, Jiang [50] developed a nano-carrier encapsulating Crizotinib (approved for EML4-ALK fusion positive lung cancer) within polylactide tocopheryl polyethylene glycol 1000 succinate (PLA-TPGS), which showed a sustained release, induced remarkable cytotoxicity in NCIH3122 lung cancer cells, and noticeable early and late apoptosis. The polymeric nanoparticle followed an endocytosis-mediated cellular uptake [50]. Interestingly, in 2017 Hu et al. [51] reported on the efficiency of paclitaxel (PTX) loaded Polycaprolactone/ Poly (ethylene glycol)/Polycaprolactone (PCEC) nanoparticles combined with chronomodulated chemotherapy for use in lung cancer. The authors set out to map the suitable time of the day for administering drug loaded nano-carriers by making out the crucial role of circadian rhythms in cancer propagation. The combination therapy demonstrated remarkable tumor growth inhibition in vivo, while it turned out that 15HALO is optimal for chemotherapy [51]. Very recently, to circumvent the low targeting capacity of nanoparticles, Wang et al. used mesenchymal stem cells (MSC) as a carrier for drug delivery loaded with nanoparticles with docetaxel (DTX). MSC proved its superiority over fibroblasts in drug loading. Both cellular and animal experiments justified the intercellular translocation of nanoparticles from MSC to cancer cell. It also inhibited primary tumor growth in vivo [52]. Much to our intrigue, Ganesh et al. explored HA-PEI/PEG nano-carriers for CD44-targeted siRNA delivery to lung cancer cells. They undertook a detailed structure-activity study for optimal siRNA encapsulation efficiency. Importantly, the targeted HA-PEI/PEG nanosystems encapsulating SSB/PLK1 siRNA showed higher cellular uptake and sequence specific gene knockdown in vivo both in sensitive and resistant A549 primary and metastatic [53].
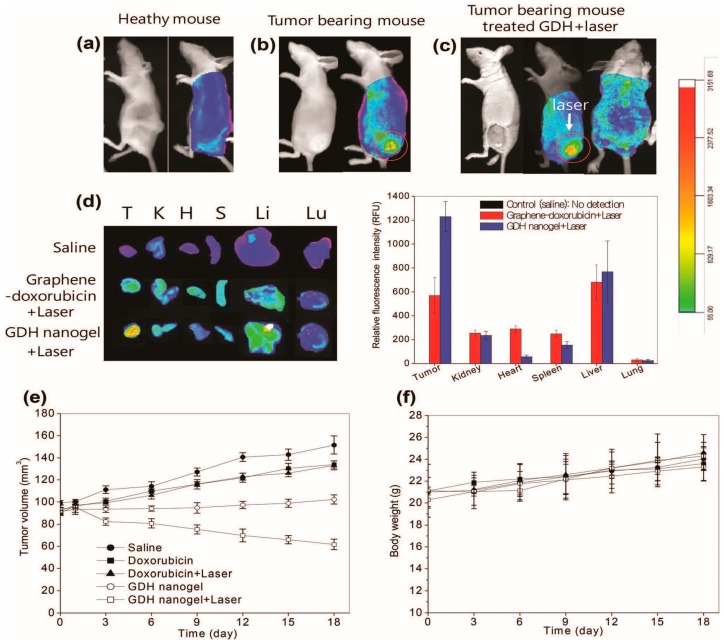
Polymer based delivery of chemotherapeutics via inhalation has also made substantial progress in recent years. Kim et al., in 2013, made an attempt to make highly porous PLGA microparticles entrapping doxorubicin where the surface of the particles was decorated with Apo2L/TRAIL (tumor necrosis factor (TNF)-related apoptosis-inducing ligand) that binds selectively to death receptors such as DR4/TRAIL-R1 expressed specifically on cancer cells. The particles got deposited in a mouse lung followed by pulmonary administration and exhibited tumor growth inhibition in nude mice with H226 metastatic lung cancer cells [54]. Furthermore, for pulmonary inhalation treatment, Feng et al. developed a two-in-one nanosystem comprising of doxorubicin and paclitaxel encapsulated into a porous PLGA microparticles and established a synergy between the two drugs at DOX:PTX 5:1 in vitro. Co-delivery remained superior over their individual counterparts in vivo as well [55]. Khatun et al. [56] demonstrated multifunctional cancer theranostics application of graphene-doxorubicin in a HA nanogel in human lung cancer cell line (A549). This nanocomposite was used as thermo and chemotherapeutic, real-time noninvasive optical imaging, and a controlled drug release [56] (Figure 2). Importantly, in a study performed in 2006, Mitra et al. [57] observed an enhanced tumor targeting the capability of N-(2-hydroxypropyl) methacrylamide (HPMA) copolymers when coupled with RGDfK and RGD4C in C57BL/6NHsd mice bearing Lewis lung carcinoma. They conjugated radionuclides with longer half-life such as 111In compounds to HPMA for scintigraphic imaging and detected contrast enhancement at the tumor site after 24h of injection [57]. In 2010, Gao and co-worker developed an MRI-visible PLA polymeric micelle whose surface was decorated by a peptide RGDLATLRQL directed towards αvβ6 integrin over-expressing human NSCLC cell H2009. DOX and super-paramagnetic iron oxide nanoparticles (SPIONs) were loaded inside the micelle core for drug delivery and MR imaging, respectively [58]. The polymeric nanoparticles are extensively applied for lung cancer theranostics due to their biocompatibility, biodegradability, high drug pay load, sustained drug release, high scalability, low batch variability, low cost production, and easy tunability [44,45].
Figure 2.
In vivo optical imaging and thermo-chemotherapy using the nanogel. (a,b) are optical images of healthy mice and tumor bearing mice, respectively. (c) Light responsive imaging. The nanogels were intravenously injected and 670 nm laser was applied to the tumors for 30 min. (d) Ex vivo imaging and fluorescence intensities of tumors and normal tissues. Organs were arranged in the following order: Tumor (T), kidney (K), heart (H), spleen (S), liver (Li), and lung (Lu). (e) Thermo-chemotherapy after treating doxorubicin and the nanogels with and without laser irradiation. (f) Body weight changes of mice after treatment with the nanogels. The data were plotted as mean ± SEM (n = 5). Reproduced from [56]. Copyright © 2015 Royal Society of Chemistry.
6. Bio-Nanoparticles for Lung Cancer Theranostics
Due to high biocompatibility, better stability and biodegradability current researchers have shifted their focal point of research towards using the bio-nanoparticles including protein nanoparticles, solid lipid nanoparticles, viral nanoparticles, aptamers, and apoferritin, wherein a bio-mimicking component is incorporated to the therapeutic nanoparticles [59,60]. In the recent past, these types of nanoparticles were successfully designed, synthesized, and utilized for cancer theranostics applications in lung cancer [59,61,62].
6.1. Viral Nanoparticles
Viral nanoparticles (VNPs) obtained from viruses and bacteriophages have gained immense interest for various biomedical applications including drug delivery, biosensing, bioimaging, and vaccine development due to their biocompatibility, flexibility in sizes and shapes, and easy surface modification [63]. Many researchers have developed a combinational approach with chemotherapy and immunotherapy for the treatment of lung cancer due to challenges of drug resistance. Veljanski et al. has published an exciting review about the use genetically modified oncolytic viruses (OVs) with conventional chemotherapies in the treatment of lung cancer. The inability of chemo-drugs to destroy the cancer stem cells is well compensated by OVs-based gene therapy [64]. The cowpea mosaic virus (CPMV) with an average size of ~28 nm has a high potential for vaccination therapy in lung cancer [65].
6.2. Protein-Based Nanoparticles
Protein nanoparticles prepared from a naturally occurring protein, such as gelatin, gliadin, albumin, and legumin have been recently used for the drug and gene delivery purposes either alone or in a mixture with biodegradable polymers in lung cancer therapy due to their excellent biocompatibility, and lack of inflammation in human bronchial cells and high cellular uptake [66,67,68]. Cationic bovine serum albumin (CBSA) has been utilized for the delivery of siRNA for the metastatic lung cancer therapy [69].
6.3. Apoferritin
Apoferritin is the hollow protein nanocage without the iron core that is composed of self-assembling 24 polypeptide subunits and has internal and external diameters of 8 nm and 12 nm, respectively. Upon removal of the iron core the apoferritin undergoes a process of assembly and disassembly with the change in pH that is extensively utilized for the synthesis of various nanoparticles for lung cancer theranostics [70,71]. Li et al. demonstrated the lung cancer diagnosis of A549 cells by using fluorescence and MR imaging of apoferritin, a ferritin-based multifunctional nanostructure [71]. The authors synthesized multifunctional hybrid nanostructures made of ferritin that showed green fluorescence and had ferrimagnetic iron oxide nanoparticles into the hollow ferritin cavity. This multifunctional apoferritin was used for the imaging αvβ3 integrin upregulated cancer cells. In another recent paper by Luo et al., demonstrated the use of hyaluronic acid (HA)-conjugated apoferritin nanocages for pH-responsive controlled intracellular prodrug release of the anticancer drug daunomycin (DN), which was encapsulated into the interior of apoferritin [72]. Moreover, the authors modified the apoferritin by HA to target and kill the cancer cells (embryonic lung MRC-5 cells and lung cancer A549 cells) upon binding to the HA-receptor CD44.
Bio-nanoparticles are widely applied for lung cancer theranostics as they demonstrate huge promise due to their good biocompatibility and biodegradability. However, the synthesis strategies can be complex at times and that can increase the cost and time of manufacturing. Hence, more research needed to manufacture these bio-nanoparticles from lab-scale to commercial industrial scale.
7. Inorganic Nanoparticles for Lung Cancer Theranostics
Inorganic nanoparticles have long been used for various biomedical applications including drug delivery, nucleic acid delivery, bio-sensing, diagnostics, imaging, and cancer therapy due to their exciting physico-chemical properties in the nanoscale range [73,74,75,76]. Among various inorganic nanoparticles, gold, silver, iron oxides, silica, rare earth oxides, carbon dots, and nanodiamonds were extensively studied in lung cancer theranostics [77,78,79]. Moreover, these nanoparticles demonstrated prominent cytotoxic effects on various lung cells in vitro and in vivo that depend largely on their size, shape, surface charge, concentration, and time of exposure. An accurate control over these physico-chemical parameters can facilitate their meaningful application in lung cancer theranostics.
7.1. Gold Nanoparticles (AuNPs)
Gold nanoparticles were extensively used for cancer theranostics applications due to easy synthesize and functionalize high biocompatibility, and multifunctional theranostics properties [80,81,82]. Several research groups utilized AuNPs for lung cancer theranostics [77]. For example, Nanospectra has developed a silica-gold nanoshells stabilized by (poly)ethylene glycol (PEG) for the photothermal therapy to the solid tumors using an NIR light source [83]. More importantly, in a recent clinical trial, AuroLase® was used for the photothermal therapy of primary or metastatic lung tumors (NCT01679470) [84]. In a recent published report by Knights et al. the authors demonstrated the size dependent effects of gold nanorods (AuNRs) on the photoacoustic (PA) imaging response and pulsed-wave photothermal therapeutic (PW-PPTT) efficacy, which is crucial for the clinical translation of AuNRs [77]. Interestingly, the PA intensity increased with the AuNR size due to the overall mass of the nanoparticles. All the different sized AuNRs showed toxicity in lung cancer cells upon laser fluence with a highest cell death in the smallest AuNR treatment, indicating the theranostics potential of AuNRs combined with PW lasers in lung cancer. In another recent report, Ramalingam et al. [85] showed the enhanced anti-cancer efficacy of doxorubicin (DOX) using polyvinylpyrrolidone functionalized AuNPs (Dox@PVP-AuNPs) in lung cancer cells. Mechanistic studies demonstrated that Dox@PVP-AuNPs treatment to lung cancer cells increases reactive oxygen species (ROS) generation, up-regulates the tumor suppressor genes, sensitize mitochondrial membrane potential and further induces apoptosis [85]. Peng et al. exhibited that novel sensors based technology using gold nanoparticles could be used for a non-invasive and inexpensive diagnostic tool for lung cancer [86].
7.2. Iron Oxides Nanoparticles (IONPs)
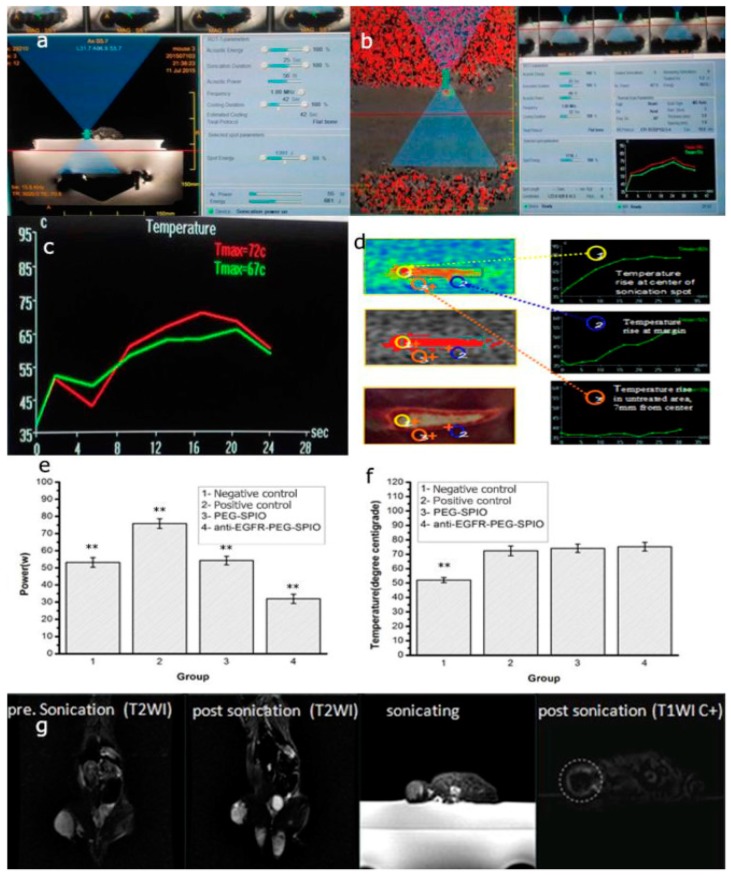
Supermagnetic iron oxide nanoparticles is extensively used as a MRI contrast agent, and also can be utilized as a delivery carrier in cancer theranostics applications. Iron oxides nanoparticles were long used for various biomedical applications including MRI imaging, drug delivery, magnetic hyperthermia, and cancer theranostics in lung cancer [87,88,89,90]. Wang et al., recently demonstrated MRI and magnetic resonance-guided focused ultrasound ablation therapy using iron oxide nanoparticles in lung cancer (Figure 3) [87]. The authors synthesized an epidermal growth factor receptor targeted PEGylated iron oxide nanoparticles (IONPs) for targeted delivery and imaging to lung cancer in an in vitro and in vivo rat xenograft model of human lung cancer (H460). In another study, Sadhuka et al. showed the usage of EGFR-targeted inhalable iron oxide nanoparticles for magnetic hyperthermia in lung cancer [89]. The authors showed that EGFR targeting enhanced the tumor retention of IONPs. Moreover, magnetic hyperthermia treatment by EGFR-targeted IONPs caused in major inhibition of lung tumor growth in in vivo orthotropic lung cancer model.
Figure 3.
(a) Sonication process images. Treatment planning software on the MRgFUS workstation. In total, five sonications are needed to measure the circumference of this tumor. (b) Tissue temperature mapping during MRgFUS ablation. (c) Real-time temperature change in MRgFUS ablation monitored by MRI. (d) Schematic illustration of therapeutic temperature map in tumor center and margin. Sonication energy (e) and therapeutic peak temperature (f) of negative control (low power, 54 W), control (76 W), PEG-SPIO (54 W), and anti-EGFR-PEG-SPIO (32 W) at 4 h post-injection of SPIO nanoparticles. (g) Anti-EGFR-PEG-SPIO group: Coronal T2WI image signal intensity of tumor before treatment and after treatment. The coronal T2WI signal intensities of tumor increased significantly after therapy compared to before therapy (Figure 6g). Enhanced axial T1WI-weighted images after injection of Gd-DTPA. Axial contrast-enhance T1WI subtraction images after injection of Gd-DTPA showed a small focal area of nonperfusion. Reproduced from [87]. Copyright © 2017. Elsevier.
7.3. Silver Nanoparticles (AgNPs)
Silver nanoparticles were long used for various biomedical applications including anti-bacterial applications, anti-cancer applications, fluorescence imaging, and biosensors [91]. Recently, He et al. demonstrated the antitumor activity of biosynthesized AgNPs against lung cancer in in vitro H1299 lung cancer cells and an in vivo xenograft immunodeficient (SCID) mouse model [92]. The potent cytotoxicity effect of AgNPs was showed by trypan blue and MTT assay. Mechanistic studies showed that AgNPs caused apoptosis (increase in caspase-3 and decrease in bcl-2) in lung cancer cells that connected well with an inhibition of NF-κB activity [92]. The cytotoxicity of AgNPs largely depends on their size, shape, morphology, and surface chemistry [93]. In another recent published report, Jeong et al. [94] explored the basic mechanism of hypoxia on AgNPs induced apoptosis that showed the upregulation of HIF-1α expression under both normoxic and hypoxic conditions. Moreover, the AgNPs treatment caused programmed cell death in lung cancer cells but not in normal cells. Notably, HIF-1α protected AgNPs-induced apoptosis by regulating autophagic flux through controlling the ATG5, p62, and LC3-II. Hence, these results suggest that hypoxia-mediated autophagy could be used to inhibit the AgNPs mediated apoptosis involving the HIF-1α as a potential target in lung cancer therapy [94].
7.4. Other Metal Nanoparticles
Among other nanoparticles rare earth, silica, and nanodiamond were used for the cancer theranostics application in lung cancer [95,96,97]. Wu et al., showed the application of a silica-polymer nanocomposite for p53 gene therapy and near infrared tumor targeted imaging of lung cancer in vitro and in vivo [95]. A nanodiamond (ND) was also utilized for the delivery of paclitaxel in lung cancer therapy. This nanodrug delivery exhibited significant tumor regression ability in immunodeficiency mice, in a lung cancer cell model. Mechanistic studies revealed that ND caused mitotic arrest and apoptosis that led to lung cancer cell death [97]. In another report, Zhang et al. developed a silica based imaging agent to detect a single miRNA in lung cancer cells that can be used for biosensing applications [96]. In another published report, Chen et al. showed the application of micromolar concentration of the neodymium oxide nanoparticles (Nd2O3) for the induction of extensive autophagy and immense vacuolization in NSCLC cells [98]. Wu et al. [99] developed a multi-functionalized, carbon dots based theranostics nanoagent that can be used for bioimaging as it emitted visible blue photoluminescence when excited at 360 nm and also can be utilized as a gene delivery vehicle for multiple siRNAs (EGFR and cyclin B1) in lung cancer. Moreover, this nanoagent was found to be accumulated in lung cancer cells by receptor mediated endocytosis in a targeted manner, resulting in improved gene silencing and anti-cancer efficacy.
Scientists and researchers are excited with metal nanoparticles in the recent past due to their small size, high surface to area, easy synthesis and scale up, low cost, multifunctional theranostics applications, exciting physico-chemical properties. However, due to the lack of enough knowledge about their long-term toxicity, pharmacokinetics, pharmacodynamics, and degradability, the clinical translation of these nanomaterials is still not widely possible compared to its other counterparts [29].
8. Clinical Status of the Nanotheranostics in Lung Cancer
Advances in nanoparticle designing and formulations are related to numerous applications in the detection and treatment of malignant growth. Ongoing advances in nanotheranostics in imaging and therapeutics of lung cancer are as follows. Porphyrins due to their favorable photophysical properties have been particularly successful for cancer imaging and photodynamic therapy (PDT). Hematoporphyrin derivative (HpD), porfimer sodium has been approved in over 120 countries since 1993 for the detection and photodynamic therapy of esophageal, lung, superficial bladder, gastric, cervical, and endobronchial cancers [100,101,102]. Though relatively non-toxic, HpD is not very effective as a primary therapy in lung cancer [103]. Laserphyrin 664 is approved for PDT in Japan for treating early centrally located lung cancer. PET (positron emission tomography) is a nuclear medicine imaging method, frequently used in combination with FDG (fluorine-18 combined with deoxy-glucose) [104]. FDG is a glucose analogue that is extensively used in cancer staging, restaging and for the analysis of tumor response to treatment. The most frequent application of theranostics is for the palliative treatment of bone metastases from lung cancer. EDTMP marked with 153Sm [105] and 99mTc-MDP an analogue of pyrophosphate similar to bone scanning agents and provides high doses of localized radiation because of its β-particle emissions.
Peptidomimetics like [68Ga]Ga-NODAGA-THERANOST™ is a 3 integrin antagonist first used in humans for lung and breast cancer diagnosis and anti-angiogenic therapy [106]. Ongoing advances in nanotheranostics for drug delivery have been mostly attempted using liposomal formulations. Irinotecan liposome injection (ONIVYDE®) is being investigated versus topotecan in patients with small cell lung cancer after platinum-based first-line therapy (Phase3; 2018–2022 study). To find the highest dose of DOTAP:Chol-fus1 liposomal formulation that can safely be given in combination with tarceva (erlotinib hydrochloride) to patients with NSCLC (Phase2; 2014–2019 study) is currently undergoing. Another trial is investigating the pain management efficiency of liposomal bupivacaine after elective thoracoscopic lobectomy (Phase3; 2018–2021 study) for non-small cell lung cancer.
An exosome is a nano-sized vesicle secreted from different cell types and has a natural ability to carry functional biomolecules, such as small RNAs, DNAs, and proteins in their lumen. This unique signature of particular cells also makes them attractive for use in drug delivery and molecular diagnosis. Moreover, exosomes can be coupled to nanoparticles and used for high precision imaging. Exosomes are considered as an important component in liquid biopsy assessments, which are useful for detecting cancers, including lung cancer. Several studies are currently underway to develop methods of exploiting exosomes for its use as efficient drug delivery vehicles and to develop novel diagnostic modalities. Srivastava et al., 2018 combined the diagnosis of CT and exosome in early lung cancer 2018–2019. Another theranostics-based use of exosome is for the dynamic monitoring circulating tumor DNA in surgical patients with lung cancer, 2017–2023 [107]. Albumin based nanoparticles are in use in different phases of clinical trials for the delivery of paclitaxel, cisplatin in NSCLC [62,108,109,110].
The development of lung cancer nanotheranostics, with an accentuation on clinical use is limited by several lacunae. The reproducible synthesis of several nanoplatforms with composite structures and its scale-up produces significant complications. Sterility can also hamper such processes. Another important aspect worth considering is nanoparticles-induced cytotoxicity, genotoxicity, and immunotoxicity related to their nanometer size. Industrial vendors and pharmaceutical companies have fewer incentives to move on with theranostics-based blockbusters because of the projected final product price as compared to conventional therapy. Though, coupling therapy with diagnosis together into a single theranostics platform should provide significant advantages but the imaging and tumor targeting components might have different pharmacokinetics and dynamics and can mount a significant manufacturing challenge.
Delay in the transition for theranostics from bench to bedside is mainly due a lack of basic-clinical scientific collaborations. Basic scientists and many clinical scientists lack the information or time to comprehend, envision, oversee cons, and manage the nuances of the multistep compliance guidelines of the clinical trial phases. Being oblivious about the obligation regarding guaranteeing consistency with the guidelines and related regulatory aspects delays the process of bringing cancer theranostics from the laboratory to clinical use. Though FDA is bringing a change in the regulatory changes to the regulatory procedures and developing tools to reduce the development time of theranostics. Weighing the potential benefits to possible adverse health risk assessment of lung cancer nanotheranostics needs to be considered based on scientifically sound, evidence-based and well-controlled clinical studies.
9. Conclusions and Future Perspectives
Theranostics or personalized cancer treatment is proof based, an individualized prescription that warrants the right treatment at the correct time, leading to significant efficacy and improvements of patient’s condition and a decrease in medicinal and services costs. Theranostics medicine is employed for delivering both therapeutic and imaging agents to the targeted area of the body, using a nanotechnology-based delivery platform. Nanomaterials have proved to be tools with tremendous benefit and are now finding application in the clinic. Nanostructures, due to their novel physical properties can often overcome solubility and stability issues through surface modification/wrappings or additional formulation. The integrated approach of combining ligands, drugs, biomolecules, and imaging agents into a functionalized nanoparticle enables targeted drug delivery and diagnostics. Another aspect that aids in the higher therapeutic payload is the high surface area because of their nanosize. Nanoparticle-based targeting specific cancer cells and specific delivery of therapeutic payloads at cancer sites via passive or active targeting can significantly reduce nonspecific toxicity. In spite of the advantages, a lot of unsolved challenges remain including scale-up problems, economical production, the pharmacokinetics of the drug, and the imaging construct. Additional issues with nanotoxicity and regulatory guidelines and hurdles need to be resolved in order to see lung cancer theranostics in the clinic. Though nanotechnology has achieved great strides but still is not used to maximal impact in lung and other malignancies.
With the advent of stronger sequencing, immunohistochemistry, and proteomic techniques, a better understanding of the mechanisms of cancer and identification of new definitive biomarkers are on the way. Greater funding for multi-center cohort studies and the advent of landmark genomic program like the Cancer Genome Atlas (TCGA) and cancer proteome studies the human protein atlas have also created new inroads in the understanding of cancer. In 2018, theranosticsimaging using PSMA (prostate specific membrane antigen) PET (positron emission tomography) and image-guided therapy using PSMA targeted radionucleotide lutetium-177 PSMA617 has shown to be a paradigm-changing practice for improving prostate cancer patient outcomes. The advances of theranostics-based radiomics will span from the current effort to the expected future of using deep learning. Development of theranosticstools for ultrasensitive and quantitative measurement of theranostics biomarkers, ability to diagnose and quantify cancer at its earliest stage with high resolution, and early prediction response to cancer therapy will also depend on how new assisting tools (e.g., artificial intelligence) are going to be harnessed. The integration of all the advances in these allied fields together as cancer theranosticsis poised to revolutionize the future of therapeutics for the ultimate eradication of cancer.
Acknowledgments
Sudip Mukherjee acknowledges Fondren Library, Rice University, Houston, TX, United States for partial financial support for the APC of this work. He also acknowledges Omid Veiseh (supervisor/mentor) for providing continuous support for his postdoctoral research. Manash Paul acknowledges Steven Dubinett and Brigitte Gomperts for providing continuous support and mentoring.
Author Contributions
Conceptualization, A.M. and S.M.; writing—original draft preparation, A.M., M.P. and S.M.; writing—review and editing, A.M., M.P. and S.M.; funding acquisition, M.P. and S.M.
Funding
This research received no external funding. The APC was partially funded by Fondren Library, Rice University, Houston, TX, USA.
Conflicts of Interest
The authors declare no conflict of interest. The authors have no other pertinent affiliations or financial connection with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
References
- 1.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2019. CA Cancer J. Clin. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 2.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Ferlay J., Soerjomataram I., Dikshit R., Eser S., Mathers C., Rebelo M., Parkin D.M., Forman D., Bray F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 4.Van der Aalst C.M., de Koning H.J. Biochemical verification of the self-reported smoking status of screened male smokers of the Dutch-Belgian randomized controlled lung cancer screening trial. Lung Cancer. 2016;94:96–101. doi: 10.1016/j.lungcan.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 5.Torre L.A., Bray F., Siegel R.L., Ferlay J., Lortet-Tieulent J., Jemal A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 6.Rudokas M., Najlah M., Alhnan M.A., Elhissi A. Liposome Delivery Systems for Inhalation: A Critical Review Highlighting Formulation Issues and Anticancer Applications. Med. Princ. Pract. 2016;25:60–72. doi: 10.1159/000445116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Miller K.D., Siegel R.L., Lin C.C., Mariotto A.B., Kramer J.L., Rowland J.H., Stein K.D., Alteri R., Jemal A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016;66:271–289. doi: 10.3322/caac.21349. [DOI] [PubMed] [Google Scholar]
- 8.Cohen Z.R., Ramishetti S., Peshes-Yaloz N., Goldsmith M., Wohl A., Zibly Z., Peer D. Localized RNAi therapeutics of chemoresistant grade IV glioma using hyaluronan-grafted lipid-based nanoparticles. ACS Nano. 2015;9:1581–1591. doi: 10.1021/nn506248s. [DOI] [PubMed] [Google Scholar]
- 9.Xiong X.B., Lavasanifar A. Traceable multifunctional micellar nanocarriers for cancer-targeted co-delivery of MDR-1 siRNA and doxorubicin. ACS Nano. 2011;5:5202–5213. doi: 10.1021/nn2013707. [DOI] [PubMed] [Google Scholar]
- 10.Shen S., Xia J.X., Wang J. Nanomedicine-mediated cancer stem cell therapy. Biomaterials. 2016;74:1–18. doi: 10.1016/j.biomaterials.2015.09.037. [DOI] [PubMed] [Google Scholar]
- 11.Tran T.H., Nguyen H.T., Pham T.T., Choi J.Y., Choi H.G., Yong C.S., Kim J.O. Development of a Graphene Oxide Nanocarrier for Dual-Drug Chemo-phototherapy to Overcome Drug Resistance in Cancer. ACS Appl. Mater. Interfaces. 2015;7:28647–28655. doi: 10.1021/acsami.5b10426. [DOI] [PubMed] [Google Scholar]
- 12.Shahin M., Soudy R., El-Sikhry H., Seubert J.M., Kaur K., Lavasanifar A. Engineered peptides for the development of actively tumor targeted liposomal carriers of doxorubicin. Cancer Lett. 2013;334:284–292. doi: 10.1016/j.canlet.2012.10.007. [DOI] [PubMed] [Google Scholar]
- 13.Zhang X.X., Eden H.S., Chen X. Peptides in cancer nanomedicine: Drug carriers, targeting ligands and protease substrates. J. Control. Release. 2012;159:2–13. doi: 10.1016/j.jconrel.2011.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu J., Wei T., Zhao J., Huang Y., Deng H., Kumar A., Wang C., Liang Z., Ma X., Liang X.J. Multifunctional aptamer-based nanoparticles for targeted drug delivery to circumvent cancer resistance. Biomaterials. 2016;91:44–56. doi: 10.1016/j.biomaterials.2016.03.013. [DOI] [PubMed] [Google Scholar]
- 15.Codony-Servat J., Garcia-Roman S., Molina-Vila M.A., Bertran-Alamillo J., Gimenez-Capitan A., Viteri S., Cardona A.F., D’Hondt E., Karachaliou N., Rosell R. Anti-Epidermal Growth Factor Vaccine Antibodies Enhance the Efficacy of Tyrosine Kinase Inhibitors and Delay the Emergence of Resistance in EGFR Mutant Lung Cancer Cells. J. Thorac. Oncol. 2018;13:1324–1337. doi: 10.1016/j.jtho.2018.04.030. [DOI] [PubMed] [Google Scholar]
- 16.Mukherjee A., Waters A.K., Babic I., Nurmemmedov E., Glassy M.C., Kesari S., Yenugonda V.M. Antibody drug conjugates: Progress, pitfalls, and promises. Hum. Antibodies. 2019;27:53–62. doi: 10.3233/HAB-180348. [DOI] [PubMed] [Google Scholar]
- 17.Lv S., Tang Z., Li M., Lin J., Song W., Liu H., Huang Y., Zhang Y., Chen X. Co-delivery of doxorubicin and paclitaxel by PEG-polypeptide nanovehicle for the treatment of non-small cell lung cancer. Biomaterials. 2014;35:6118–6129. doi: 10.1016/j.biomaterials.2014.04.034. [DOI] [PubMed] [Google Scholar]
- 18.Fernandez-Fernandez A., Manchanda R., McGoron A.J. Theranostic applications of nanomaterials in cancer: Drug delivery, image-guided therapy, and multifunctional platforms. Appl. Biochem. Biotechnol. 2011;165:1628–1651. doi: 10.1007/s12010-011-9383-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cong Y., Xiao H., Xiong H., Wang Z., Ding J., Li C., Chen X., Liang X.J., Zhou D., Huang Y. Dual Drug Backboned Shattering Polymeric Theranostic Nanomedicine for Synergistic Eradication of Patient-Derived Lung Cancer. Adv. Mater. 2018;30 doi: 10.1002/adma.201706220. [DOI] [PubMed] [Google Scholar]
- 20.Chi Y.H., Hsiao J.K., Lin M.H., Chang C., Lan C.H., Wu H.C. Lung Cancer-Targeting Peptides with Multi-subtype Indication for Combinational Drug Delivery and Molecular Imaging. Theranostics. 2017;7:1612–1632. doi: 10.7150/thno.17573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Arranja A.G., Pathak V., Lammers T., Shi Y. Tumor-targeted nanomedicines for cancer theranostics. Pharmacol. Res. 2017;115:87–95. doi: 10.1016/j.phrs.2016.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chowdhury S.R., Mukherjee S., Das S., Patra C.R., Iyer P.K. Multifunctional (3-in-1) cancer theranostics applications of hydroxyquinoline-appended polyfluorene nanoparticles. Chem. Sci. 2017;8:7566–7575. doi: 10.1039/C7SC03321D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Muthuraj B., Mukherjee S., Patra C.R., Iyer P.K. Amplified Fluorescence from Polyfluorene Nanoparticles with Dual State Emission and Aggregation Caused Red Shifted Emission for Live Cell Imaging and Cancer Theranostics. ACS Appl. Mater. Interfaces. 2016;8:32220–32229. doi: 10.1021/acsami.6b11373. [DOI] [PubMed] [Google Scholar]
- 24.Mukherjee S., Patra C.R. Biologically synthesized metal nanoparticles: Recent advancement and future perspectives in cancer theranostics. Future Sci. 2017;3:FSO203. doi: 10.4155/fsoa-2017-0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Burstein H.J., Schwartz R.S. Molecular origins of cancer. N. Engl. J. Med. 2008;358:527. doi: 10.1056/NEJMe0800065. [DOI] [PubMed] [Google Scholar]
- 26.Paul M.K., Mukhopadhyay A.K. Tyrosine kinase—Role and significance in Cancer. Int. J. Med. Sci. 2004;1:101–115. doi: 10.7150/ijms.1.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jamal-Hanjani M., Wilson G.A., McGranahan N., Birkbak N.J., Watkins T.B.K., Veeriah S., Shafi S., Johnson D.H., Mitter R., Rosenthal R., et al. Tracking the Evolution of Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017;376:2109–2121. doi: 10.1056/NEJMoa1616288. [DOI] [PubMed] [Google Scholar]
- 28.DiSanto R.M., Subramanian V., Gu Z. Recent advances in nanotechnology for diabetes treatment. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015;7:548–564. doi: 10.1002/wnan.1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mukherjee S., Patra C.R. Therapeutic application of anti-angiogenic nanomaterials in cancers. Nanoscale. 2016;8:12444–12470. doi: 10.1039/C5NR07887C. [DOI] [PubMed] [Google Scholar]
- 30.Yue X., Dai Z. Liposomal Nanotechnology for Cancer Theranostics. Curr. Med. Chem. 2018;25:1397–1408. doi: 10.2174/0929867324666170306105350. [DOI] [PubMed] [Google Scholar]
- 31.Matsumura Y., Maeda H. A New Concept for Macromolecular Therapeutics in Cancer-Chemotherapy—Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res. 1986;46:6387–6392. [PubMed] [Google Scholar]
- 32.Moghimi S.M., Szebeni J. Stealth liposomes and long circulating nanoparticles: Critical issues in pharmacokinetics, opsonization and protein-binding properties. Prog. Lipid Res. 2003;42:463–478. doi: 10.1016/S0163-7827(03)00033-X. [DOI] [PubMed] [Google Scholar]
- 33.Torchilin V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 2005;4:145–160. doi: 10.1038/nrd1632. [DOI] [PubMed] [Google Scholar]
- 34.Barenholz Y. Doxil(R)—The first FDA-approved nano-drug: Lessons learned. J. Control. Release. 2012;160:117–134. doi: 10.1016/j.jconrel.2012.03.020. [DOI] [PubMed] [Google Scholar]
- 35.Petersen G.H., Alzghari S.K., Chee W., Sankari S.S., La-Beck N.M. Meta-analysis of clinical and preclinical studies comparing the anticancer efficacy of liposomal versus conventional non-liposomal doxorubicin. J. Control. Release. 2016;232:255–264. doi: 10.1016/j.jconrel.2016.04.028. [DOI] [PubMed] [Google Scholar]
- 36.Cheng L., Huang F.Z., Cheng L.F., Zhu Y.Q., Hu Q., Li L., Wei L., Chen D.W. GE11-modified liposomes for non-small cell lung cancer targeting: Preparation, ex vitro and in vivo evaluation. Int. J. Nanomed. 2014;9:921–935. doi: 10.2147/IJN.S53310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lin C., Zhang X., Chen H., Bian Z., Zhang G., Riaz M.K., Tyagi D., Lin G., Zhang Y., Wang J., et al. Dual-ligand modified liposomes provide effective local targeted delivery of lung-cancer drug by antibody and tumor lineage-homing cell-penetrating peptide. Drug Deliv. 2018;25:256–266. doi: 10.1080/10717544.2018.1425777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Song X.L., Ju R.J., Xiao Y., Wang X., Liu S., Fu M., Liu J.J., Gu L.Y., Li X.T., Cheng L. Application of multifunctional targeting epirubicin liposomes in the treatment of non-small-cell lung cancer. Int. J. Nanomed. 2017;12:7433–7451. doi: 10.2147/IJN.S141787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mukherjee A., Bhattacharyya J., Sagar M.V., Chaudhuri A. Liposomally encapsulated CDC20 siRNA inhibits both solid melanoma tumor growth and spontaneous growth of intravenously injected melanoma cells on mouse lung. Drug Deliv. Transl. Res. 2013;3:224–234. doi: 10.1007/s13346-013-0141-3. [DOI] [PubMed] [Google Scholar]
- 40.Skubitz K.M., Anderson P.M. Inhalational interleukin-2 liposomes for pulmonary metastases: A phase I clinical trial. Anticancer Drugs. 2000;11:555–563. doi: 10.1097/00001813-200008000-00006. [DOI] [PubMed] [Google Scholar]
- 41.Wittgen B.P., Kunst P.W., van der Born K., van Wijk A.W., Perkins W., Pilkiewicz F.G., Perez-Soler R., Nicholson S., Peters G.J., Postmus P.E. Phase I study of aerosolized SLIT cisplatin in the treatment of patients with carcinoma of the lung. Clin. Cancer Res. 2007;13:2414–2421. doi: 10.1158/1078-0432.CCR-06-1480. [DOI] [PubMed] [Google Scholar]
- 42.Erten A., Wrasidlo W., Scadeng M., Esener S., Hoffman R.M., Bouvet M., Makale M. Magnetic resonance and fluorescence imaging of doxorubicin-loaded nanoparticles using a novel in vivo model. Nanomedicine. 2010;6:797–807. doi: 10.1016/j.nano.2010.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lowery A., Onishko H., Hallahan D.E., Han Z. Tumor-targeted delivery of liposome-encapsulated doxorubicin by use of a peptide that selectively binds to irradiated tumors. J. Control. Release. 2011;150:117–124. doi: 10.1016/j.jconrel.2010.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sercombe L., Veerati T., Moheimani F., Wu S.Y., Sood A.K., Hua S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015;6:286. doi: 10.3389/fphar.2015.00286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bolhassani A., Javanzad S., Saleh T., Hashemi M., Aghasadeghi M.R., Sadat S.M. Polymeric nanoparticles: Potent vectors for vaccine delivery targeting cancer and infectious diseases. Hum. Vaccin. Immunother. 2014;10:321–332. doi: 10.4161/hv.26796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Farokhzad O.C., Cheng J., Teply B.A., Sherifi I., Jon S., Kantoff P.W., Richie J.P., Langer R. Targeted nanoparticle—Aptamer bioconjugates for cancer chemotherapy in vivo. Proc. Natl. Acad. Sci. USA. 2006;103:6315–6320. doi: 10.1073/pnas.0601755103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Torchilin V.P. Micellar nanocarriers: Pharmaceutical perspectives. Pharm. Res. 2007;24:1–16. doi: 10.1007/s11095-006-9132-0. [DOI] [PubMed] [Google Scholar]
- 48.Kim T.Y., Kim D.W., Chung J.Y., Shin S.G., Kim S.C., Heo D.S., Kim N.K., Bang Y.J. Phase I and pharmacokinetic study of Genexol-PM, a cremophor-free, polymeric micelle-formulated paclitaxel, in patients with advanced malignancies. Clin. Cancer Res. 2004;10:3708–3716. doi: 10.1158/1078-0432.CCR-03-0655. [DOI] [PubMed] [Google Scholar]
- 49.Ahn H.K., Jung M., Sym S.J., Shin D.B., Kang S.M., Kyung S.Y., Park J.W., Jeong S.H., Cho E.K. A phase II trial of Cremorphor EL-free paclitaxel (Genexol-PM) and gemcitabine in patients with advanced non-small cell lung cancer. Cancer Chemother. Pharmacol. 2014;74:277–282. doi: 10.1007/s00280-014-2498-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jiang Z.M., Dai S.P., Xu Y.Q., Li T., Xie J., Li C., Zhang Z.H. Crizotinib-loaded polymeric nanoparticles in lung cancer chemotherapy. Med. Oncol. 2015;32:193. doi: 10.1007/s12032-015-0636-5. [DOI] [PubMed] [Google Scholar]
- 51.Hu J., Fu S., Peng Q., Han Y., Xie J., Zan N., Chen Y., Fan J. Paclitaxel-loaded polymeric nanoparticles combined with chronomodulated chemotherapy on lung cancer: In vitro and in vivo evaluation. Int. J. Pharm. 2017;516:313–322. doi: 10.1016/j.ijpharm.2016.11.047. [DOI] [PubMed] [Google Scholar]
- 52.Wang X.S., Chen H.Y., Zeng X.W., Guo W.P., Jin Y., Wang S., Tian R.Y., Han Y.J., Guo L., Han J.M., et al. Efficient lung cancer-targeted drug delivery via a nanoparticle/MSC system. Acta. Pharm. Sin. B. 2019;9:167–176. doi: 10.1016/j.apsb.2018.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ganesh S., Iyer A.K., Morrissey D.V., Amiji M.M. Hyaluronic acid based self-assembling nanosystems for CD44 target mediated siRNA delivery to solid tumors. Biomaterials. 2013;34:3489–3502. doi: 10.1016/j.biomaterials.2013.01.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim I., Byeon H.J., Kim T.H., Lee E.S., Oh K.T., Shin B.S., Lee K.C., Youn Y.S. Doxorubicin-loaded porous PLGA microparticles with surface attached TRAIL for the inhalation treatment of metastatic lung cancer. Biomaterials. 2013;34:6444–6453. doi: 10.1016/j.biomaterials.2013.05.018. [DOI] [PubMed] [Google Scholar]
- 55.Feng T., Tian H., Xu C., Lin L., Xie Z., Lam M.H., Liang H., Chen X. Synergistic co-delivery of doxorubicin and paclitaxel by porous PLGA microspheres for pulmonary inhalation treatment. Eur. J. Pharm. Biopharm. 2014;88:1086–1093. doi: 10.1016/j.ejpb.2014.09.012. [DOI] [PubMed] [Google Scholar]
- 56.Khatun Z., Nurunnabi M., Nafiujjaman M., Reeck G.R., Khan H.A., Cho K.J., Lee Y.K. A hyaluronic acid nanogel for photo-chemo theranostics of lung cancer with simultaneous light-responsive controlled release of doxorubicin. Nanoscale. 2015;7:10680–10689. doi: 10.1039/C5NR01075F. [DOI] [PubMed] [Google Scholar]
- 57.Mitra A., Coleman T., Borgman M., Nan A., Ghandehari H., Line B.R. Polymeric conjugates of mono- and bi-cyclic alphaVbeta3 binding peptides for tumor targeting. J. Control. Release. 2006;114:175–183. doi: 10.1016/j.jconrel.2006.06.014. [DOI] [PubMed] [Google Scholar]
- 58.Guthi J.S., Yang S.G., Huang G., Li S., Khemtong C., Kessinger C.W., Peyton M., Minna J.D., Brown K.C., Gao J. MRI-visible micellar nanomedicine for targeted drug delivery to lung cancer cells. Mol. Pharm. 2010;7:32–40. doi: 10.1021/mp9001393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sivarajakumar R., Mallukaraj D., Kadavakollu M., Neelakandan N., Chandran S., Bhojaraj S., Karri V.V.S.R. Nanoparticles for the Treatment of Lung Cancers. J. Young Pharm. 2018;10:276–281. doi: 10.5530/jyp.2018.10.62. [DOI] [Google Scholar]
- 60.Perepelyuk M., Sacko K., Thangavel K., Shoyele S.A. Evaluation of MUC1-Aptamer Functionalized Hybrid Nanoparticles for Targeted Delivery of miRNA-29b to Nonsmall Cell Lung Cancer. Mol. Pharm. 2018;15:985–993. doi: 10.1021/acs.molpharmaceut.7b00900. [DOI] [PubMed] [Google Scholar]
- 61.Rizvi N.A., Riely G.J., Azzoli C.G., Miller V.A., Ng K.K., Fiore J., Chia G., Brower M., Heelan R., Hawkins M.J., et al. Phase I/II trial of weekly intravenous 130-nm albumin-bound paclitaxel as initial chemotherapy in patients with stage IV non-small-cell lung cancer. J. Clin. Oncol. 2008;26:639–643. doi: 10.1200/JCO.2007.10.8605. [DOI] [PubMed] [Google Scholar]
- 62.Socinski M.A., Okamoto I., Hon J.K., Hirsh V., Dakhil S.R., Page R.D., Orsini J., Yamamoto N., Zhang H., Renschler M.F. Safety and efficacy analysis by histology of weekly nab-paclitaxel in combination with carboplatin as first-line therapy in patients with advanced non-small-cell lung cancer. Ann. Oncol. 2013;24:2390–2396. doi: 10.1093/annonc/mdt235. [DOI] [PubMed] [Google Scholar]
- 63.Li K., Nguyen H.G., Lu X.B., Wang Q. Viruses and their potential in bioimaging and biosensing applications. Analyst. 2010;135:21–27. doi: 10.1039/B911883G. [DOI] [PubMed] [Google Scholar]
- 64.Beljanski V., Hiscott J. The use of oncolytic viruses to overcome lung cancer drug resistance. Curr. Opin. Virol. 2012;2:629–635. doi: 10.1016/j.coviro.2012.07.006. [DOI] [PubMed] [Google Scholar]
- 65.Robertson K.L., Soto C.M., Archer M.J., Odoemene O., Liu J.L. Engineered T4 viral nanoparticles for cellular imaging and flow cytometry. Bioconjug. Chem. 2011;22:595–604. doi: 10.1021/bc100365j. [DOI] [PubMed] [Google Scholar]
- 66.Maham A., Tang Z., Wu H., Wang J., Lin Y. Protein-based nanomedicine platforms for drug delivery. Small. 2009;5:1706–1721. doi: 10.1002/smll.200801602. [DOI] [PubMed] [Google Scholar]
- 67.Wiley J.A., Richert L.E., Swain S.D., Harmsen A., Barnard D.L., Randall T.D., Jutila M., Douglas T., Broomell C., Young M., et al. Inducible bronchus-associated lymphoid tissue elicited by a protein cage nanoparticle enhances protection in mice against diverse respiratory viruses. PLoS ONE. 2009;4:e7142. doi: 10.1371/journal.pone.0007142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lohcharoenkal W., Wang L.Y., Chen Y.C., Rojanasakul Y. Protein Nanoparticles as Drug Delivery Carriers for Cancer Therapy. Biomed. Res. Int. 2014 doi: 10.1155/2014/180549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Han J.F., Wang Q., Zhang Z.R., Gong T., Sun X. Cationic Bovine Serum Albumin Based Self-Assembled Nanoparticles as siRNA Delivery Vector for Treating Lung Metastatic Cancer. Small. 2014;10:524–535. doi: 10.1002/smll.201301992. [DOI] [PubMed] [Google Scholar]
- 70.Dostalova S., Vasickova K., Hynek D., Krizkova S., Richtera L., Vaculovicova M., Eckschlager T., Stiborova M., Heger Z., Adam V. Apoferritin as an ubiquitous nanocarrier with excellent shelf life. Int. J. Nanomed. 2017;12:2265–2278. doi: 10.2147/IJN.S130267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Li K., Zhang Z.P., Luo M., Yu X., Han Y., Wei H.P., Cui Z.Q., Zhang X.E. Multifunctional ferritin cage nanostructures for fluorescence and MR imaging of tumor cells. Nanoscale. 2012;4:188–193. doi: 10.1039/C1NR11132A. [DOI] [PubMed] [Google Scholar]
- 72.Luo Y., Wang X., Du D., Lin Y. Hyaluronic acid-conjugated apoferritin nanocages for lung cancer targeted drug delivery. Biomater. Sci. 2015;3:1386–1394. doi: 10.1039/C5BM00067J. [DOI] [PubMed] [Google Scholar]
- 73.Patra C.R., Mukherjee S., Kotcherlakota R. Biosynthesized silver nanoparticles: A step forward for cancer theranostics? Nanomedicine. 2014;9:1445–1448. doi: 10.2217/nnm.14.89. [DOI] [PubMed] [Google Scholar]
- 74.Gao L., Fan K., Yan X. Iron Oxide Nanozyme: A Multifunctional Enzyme Mimetic for Biomedical Applications. Theranostics. 2017;7:3207–3227. doi: 10.7150/thno.19738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lim E.K., Kim T., Paik S., Haam S., Huh Y.M., Lee K. Nanomaterials for theranostics: Recent advances and future challenges. Chem. Rev. 2015;115:327–394. doi: 10.1021/cr300213b. [DOI] [PubMed] [Google Scholar]
- 76.Gaddam R.R., Mukherjee S., Punugupati N., Vasudevan D., Patra C.R., Narayan R., Vsn Kothapalli R. Facile synthesis of carbon dot and residual carbon nanobeads: Implications for ion sensing, medicinal and biological applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;73:643–652. doi: 10.1016/j.msec.2016.12.095. [DOI] [PubMed] [Google Scholar]
- 77.Knights O.B., McLaughlan J.R. Gold Nanorods for Light-Based Lung Cancer Theranostics. Int. J. Mol. Sci. 2018;19:3318. doi: 10.3390/ijms19113318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Silva C.O., Pinho J.O., Lopes J.M., Almeida A.J., Gaspar M.M., Reis C. Current Trends in Cancer Nanotheranostics: Metallic, Polymeric, and Lipid-Based Systems. Pharmaceutics. 2019;11:22. doi: 10.3390/pharmaceutics11010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lee H.Y., Mohammed K.A., Nasreen N. Nanoparticle-based targeted gene therapy for lung cancer. Am. J. Cancer Res. 2016;6:1118–1134. [PMC free article] [PubMed] [Google Scholar]
- 80.Liu Y., Ma W., Wang J. Theranostics of Gold Nanoparticles with an Emphasis on Photoacoustic Imaging and Photothermal Therapy. Curr. Pharm. Des. 2018;24:2719–2728. doi: 10.2174/1381612824666180604112201. [DOI] [PubMed] [Google Scholar]
- 81.Balakrishnan S., Mukherjee S., Das S., Bhat F.A., Raja Singh P., Patra C.R., Arunakaran J. Gold nanoparticles-conjugated quercetin induces apoptosis via inhibition of EGFR/PI3K/Akt-mediated pathway in breast cancer cell lines (MCF-7 and MDA-MB-231) Cell Biochem. Funct. 2017;35:217–231. doi: 10.1002/cbf.3266. [DOI] [PubMed] [Google Scholar]
- 82.Meka R.R., Mukherjee S., Patra S., Chaudhuri A. Shikimoyl-ligand decorated gold nanoparticles for use in ex vivo engineered dendritic cell based DNA vaccination. Nanoscale. 2019;11 doi: 10.1039/C8NR10293G. [DOI] [PubMed] [Google Scholar]
- 83.Anselmo A.C., Mitragotri S. Nanoparticles in the clinic. Bioeng. Transl. Med. 2016;1:10–29. doi: 10.1002/btm2.10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Singh P., Pandit S., Mokkapati V., Garg A., Ravikumar V., Mijakovic I. Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. Int. J. Mol. Sci. 2018;19:1979. doi: 10.3390/ijms19071979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ramalingam V., Varunkumar K., Ravikumar V., Rajaram R. Target delivery of doxorubicin tethered with PVP stabilized gold nanoparticles for effective treatment of lung cancer. Sci. Rep. 2018;8:3815. doi: 10.1038/s41598-018-22172-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Peng G., Tisch U., Adams O., Hakim M., Shehada N., Broza Y.Y., Billan S., Abdah-Bortnyak R., Kuten A., Haick H. Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat. Nanotechnol. 2009;4:669–673. doi: 10.1038/nnano.2009.235. [DOI] [PubMed] [Google Scholar]
- 87.Wang Z.L., Qiao R.R., Tang N., Lu Z.W., Wang H., Zhang Z.X., Xue X.D., Huang Z.Y., Zhang S.R., Zhang G.X., et al. Active targeting theranostic iron oxide nanoparticles for MRI and magnetic resonance-guided focused ultrasound ablation of lung cancer. Biomaterials. 2017;127:25–35. doi: 10.1016/j.biomaterials.2017.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ghazani A.A., Pectasides M., Sharma A., Castro C.M., Mino-Kenudson M., Lee H., Shepard J.A., Weissleder R. Molecular characterization of scant lung tumor cells using iron-oxide nanoparticles and micro-nuclear magnetic resonance. Nanomedicine. 2014;10:661–668. doi: 10.1016/j.nano.2013.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sadhukha T., Wiedmann T.S., Panyam J. Inhalable magnetic nanoparticles for targeted hyperthermia in lung cancer therapy. Biomaterials. 2013;34:5163–5171. doi: 10.1016/j.biomaterials.2013.03.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Noh M.S., Jun B.H., Kim S., Kang H., Woo M.A., Minai-Tehrani A., Kim J.E., Kim J., Park J., Lim H.T., et al. Magnetic surface-enhanced Raman spectroscopic (M-SERS) dots for the identification of bronchioalveolar stem cells in normal and lung cancer mice. Biomaterials. 2009;30:3915–3925. doi: 10.1016/j.biomaterials.2009.03.059. [DOI] [PubMed] [Google Scholar]
- 91.Mukherjee S., Chowdhury D., Kotcherlakota R., Patra S., Vinothkumar B., Bhadra M.P., Sreedhar B., Patra C.R. Potential theranostics application of bio-synthesized silver nanoparticles (4-in-1 system) Theranostics. 2014;4:316–335. doi: 10.7150/thno.7819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.He Y., Du Z.Y., Ma S.J., Liu Y., Li D.L., Huang H.R., Jiang S., Cheng S.P., Wu W.J., Zhang K., et al. Effects of green-synthesized silver nanoparticles on lung cancer cells in vitro and grown as xenograft tumors in vivo. Int. J. Nanomed. 2016;11 doi: 10.2147/IJN.S103695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Stoehr L.C., Gonzalez E., Stampfl A., Casals E., Duschl A., Puntes V., Oostingh G.J. Shape matters: Effects of silver nanospheres and wires on human alveolar epithelial cells. Part Fibre. Toxicol. 2011;8:36. doi: 10.1186/1743-8977-8-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jeong J.K., Gurunathan S., Kang M.H., Han J.W., Das J., Choi Y.J., Kwon D.N., Cho S.G., Park C., Seo H.G., et al. Hypoxia-mediated autophagic flux inhibits silver nanoparticle-triggered apoptosis in human lung cancer cells. Sci. Rep. 2016;6:21688. doi: 10.1038/srep21688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wu H., Zhao Y., Mu X., Wu H., Chen L., Liu W., Mu Y., Liu J., Wei X. A silica-polymer composite nano system for tumor-targeted imaging and p53 gene therapy of lung cancer. J. Biomater. Sci. Polym. Ed. 2015;26:384–400. doi: 10.1080/09205063.2015.1012035. [DOI] [PubMed] [Google Scholar]
- 96.Zhang J., Fu Y., Mei Y., Jiang F., Lakowicz J.R. Fluorescent metal nanoshell probe to detect single miRNA in lung cancer cell. Anal. Chem. 2010;82:4464–4471. doi: 10.1021/ac100241f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Liu K.K., Zheng W.W., Wang C.C., Chiu Y.C., Cheng C.L., Lo Y.S., Chen C., Chao J.I. Covalent linkage of nanodiamond-paclitaxel for drug delivery and cancer therapy. Nanotechnology. 2010;21:315106. doi: 10.1088/0957-4484/21/31/315106. [DOI] [PubMed] [Google Scholar]
- 98.Chen Y., Yang L., Feng C., Wen L.P. Nano neodymium oxide induces massive vacuolization and autophagic cell death in non-small cell lung cancer NCI-H460 cells. Biochem. Biophys. Res. Commun. 2005;337:52–60. doi: 10.1016/j.bbrc.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 99.Wu Y.-F., Wu H.-C., Kuan C.-H., Lin C.-J., Wang L.-W., Chang C.-W., Wang T.-W. Multi-functionalized carbon dots as theranostic nanoagent for gene delivery in lung cancer therapy. Sci. Rep. 2016;6:21170. doi: 10.1038/srep21170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Baskaran R., Lee J., Yang S.G. Clinical development of photodynamic agents and therapeutic applications. Biomater. Res. 2018;22:25. doi: 10.1186/s40824-018-0140-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Kato H., Horai T., Furuse K., Fukuoka M., Suzuki S., Hiki Y., Ito Y., Mimura S., Tenjin Y., Hisazumi H., et al. Photodynamic therapy for cancers: A clinical trial of porfimer sodium in Japan. Jpn. J. Cancer Res. 1993;84:1209–1214. doi: 10.1111/j.1349-7006.1993.tb02823.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hayata Y., Kato H., Konaka C., Ono J., Takizawa N. Hematoporphyrin derivative and laser photoradiation in the treatment of lung cancer. Chest. 1982;81:269–277. doi: 10.1378/chest.81.3.269. [DOI] [PubMed] [Google Scholar]
- 103.Hayata Y., Kato H., Konaka C., Amemiya R., Ono J., Ogawa I., Kinoshita K., Sakai H., Takahashi H. Photoradiation therapy with hematoporphyrin derivative in early and stage 1 lung cancer. Chest. 1984;86:169–177. doi: 10.1378/chest.86.2.169. [DOI] [PubMed] [Google Scholar]
- 104.Ding X.P., Zhang J., Li B.S., Li H.S., Wang Z.T., Yi Y., Sun H.F., Wang D.Q. Feasibility of shrinking field radiation therapy through 18F-FDG PET/CT after 40 Gy for stage III non-small cell lung cancers. Asian Pac. J. Cancer Prev. 2012;13:319–323. doi: 10.7314/APJCP.2012.13.1.319. [DOI] [PubMed] [Google Scholar]
- 105.Pacilio M., Ventroni G., Basile C., Ialongo P., Becci D., Mango L. Improving the dose-myelotoxicity correlation in radiometabolic therapy of bone metastases with 153Sm-EDTMP. Eur. J. Nucl. Med. Mol. Imaging. 2014;41:238–252. doi: 10.1007/s00259-013-2552-2. [DOI] [PubMed] [Google Scholar]
- 106.Baum R.P., Kulkarni H.R., Muller D., Satz S., Danthi N., Kim Y.S., Brechbiel M.W. First-In-Human Study Demonstrating Tumor-Angiogenesis by PET/CT Imaging with (68) Ga-NODAGA-THERANOST, a High-Affinity Peptidomimetic for alphavbeta3 Integrin Receptor Targeting. Cancer Biother. Radiopharm. 2015;30:152–159. doi: 10.1089/cbr.2014.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Srivastava A., Amreddy N., Razaq M., Towner R., Zhao Y., Ahmed R., Munshi A., Ramesh R. Exosomes as Theranostics for Lung Cancer. Adv. Cancer Res. 2018;139:1–33. doi: 10.1016/bs.acr.2018.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Socinski M.A., Langer C.J., Okamoto I., Hon J.K., Hirsh V., Dakhil S.R., Page R.D., Orsini J., Zhang H., Renschler M.F. Safety and efficacy of weekly nab(R)-paclitaxel in combination with carboplatin as first-line therapy in elderly patients with advanced non-small-cell lung cancer. Ann. Oncol. 2013;24:314–321. doi: 10.1093/annonc/mds461. [DOI] [PubMed] [Google Scholar]
- 109.Green M.R., Manikhas G.M., Orlov S., Afanasyev B., Makhson A.M., Bhar P., Hawkins M.J. Abraxane, a novel Cremophor-free, albumin-bound particle form of paclitaxel for the treatment of advanced non-small-cell lung cancer. Ann. Oncol. 2006;17:1263–1268. doi: 10.1093/annonc/mdl104. [DOI] [PubMed] [Google Scholar]
- 110.Zheng D., Wang J., Guo S., Zhao Z., Wang F. Formulations, Pharmacodynamic and Clinical Studies of Nanoparticles for Lung Cancer Therapy—An Overview. Curr. Drug Metab. 2018;19:759–767. doi: 10.2174/1389200219666180305145345. [DOI] [PubMed] [Google Scholar]