Abstract
Ailments of the bladder are often treated via intravesical delivery—direct application of therapeutic into the bladder through a catheter. This technique is employed hundreds of thousands of times every year, but protocol development has largely been limited to empirical determination. Furthermore, the numerical analyses of intravesical delivery performed to date have been restricted to static geometries and have not accounted for bladder deformation. This study uses a finite element analysis approach with biphasic solute transport to investigate several parameters pertinent to intravesical delivery including solute concentration, solute transport properties and instillation volume. The volume of instillation was found to have a substantial impact on the exposure of solute to the deeper muscle layers of the bladder, which are typically more difficult to reach. Indeed, increasing the instillation volume from 50–100 ml raised the muscle solute exposure as a percentage of overall bladder exposure from 60–70% with higher levels achieved for larger instillation volumes. Similar increases were not seen for changes in solute concentration or solute transport properties. These results indicate the role that instillation volume may play in targeting particular layers of the bladder during an intravesical delivery.
Keywords: bladder cancer, cystitis, intravesical delivery, FEBio
1. Introduction
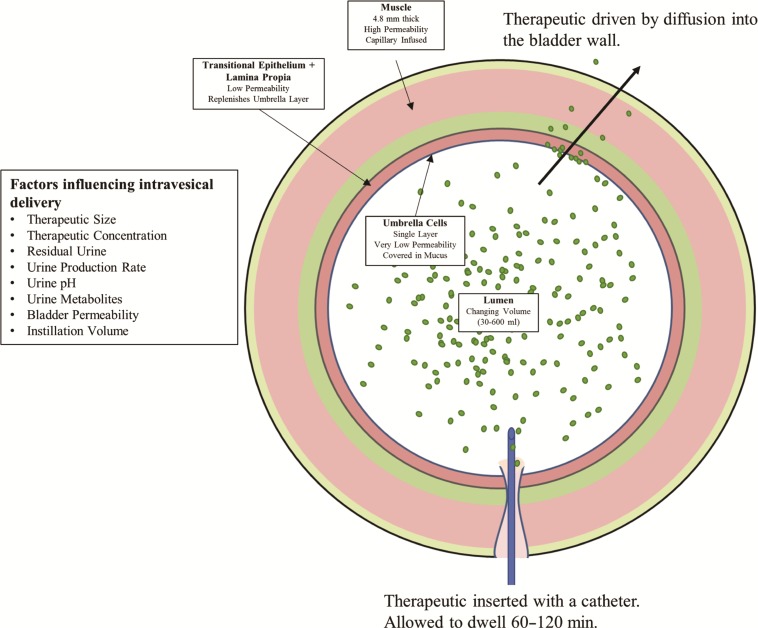
An estimated 2.7–6.5% of women in the USA suffer from interstitial cystitis or bladder pain (Konkle et al., 2012). Furthermore, in 2016, nearly 77,000 people were diagnosed with bladder cancer in the USA alone (American Cancer Society, 2016). In either case, diagnosed patients will likely undergo treatment via direct application of therapeutic into the bladder. Intravesical drug delivery (ID), i.e. direct installation of a therapeutic into the urinary bladder via catheterization (Fig. 1), is the most common mode of treatment for bladder diseases including cystitis and bladder cancer (Giannantoni et al., 2006). The instilled therapeutic moves via diffusion into the bladder wall, albeit slowly. A key advantage of this approach is that it allows for sequestration of therapeutic within the bladder, minimizing systemic side effects while maximizing exposure of the therapeutic within the bladder wall.
Fig. 1.
The bladder and factors influencing intravesical delivery.
Despite the advantage of drug localization, ID is challenging for three reasons. First, the bladder comprises the least permeable tissue in the human body because of the nearly impermeable layer of umbrella cells lining the bladder urothelium (Anderson & Italie, 2009). This often leads to inadequate absorption, especially for large molecules and deeper layers. Second, dilution and metabolism of the therapy due to urine production during 1–2-hour instillations reduce the concentration gradient throughout the procedure. Third, most of the therapies are expelled upon voiding after the instillation is completed.
Most clinical ID protocols were developed empirically, resulting in variable and often insufficient response rates. They typically consist of a 1–2-hour instillation of therapeutic given in 20–50 ml of solution. There are several controllable factors that can impact ID and that can be tailored to mitigate those hurdles. These include the following: altering the bladder permeability via the addition of absorption enhancers, changing the concentration, varying the volume of instillation, changing the duration of administration, as well as controlling residual urine, urine production rate, urine pH and urine metabolites (Au et al., 2001; Tyagi et al., 2006).Each of these factors could have substantial impacts on the effectiveness of a given intravesical administration, but their effects have largely been understudied.
Because of the large number of parameters that influence ID and the basic concepts underlying its efficacy, ID would seem to be ripe for mathematical analysis. However, there have been only two published investigations utilizing mathematical models to explore ID (Wientjes et al., 1993;Grabnar et al., 2006). The first, by Wientjes et al., used human pharmacokinetic data to form a system of equations describing mitomycin C transport for bladder cancer treatment (Wientjes et al., 1993). This simulation considered several parameters and arrived at an optimized regimen that projected to increase exposure by 8.5× over a standard protocol. This regimen consisted of efforts to maximize drug concentration by draining residual urine, lowering urine production rate by avoiding diuretics and giving a higher dose in a lower instillation volume. Based on these results, a Phase III clinical trial was designed and implemented. This trial found the optimized regimen to be more effective at enhancing recurrence-free survival as well as time to recurrence (Au et al., 2001).
The second model, by Grabnar et al., used a compartmental diffusion model. This approach allowed for the usage of diffusion coefficients instead of pharmacokinetic parameters, such as half width. The diffusion parameters in this model were based on their own data gathered through the ex vivo diffusion of pipemidic acid through pig bladders. This approach accounted for urine voiding could be extended past removal of the catheter and modelled the effects of absorption enhancers such as chitosan and polycarbophil (Grabnar et al., 2006). The major finding from this model was the promise of utilizing absorption enhancers. Adding absorption enhancers increased tissue exposure by 75–100% whereas increasing exposure time by 100 min increased exposure by 20%.
While those previous studies described the influence of several pertinent parameters on ID in a static geometry, there remains room for further investigations in a deformable geometry that considers the thinning of the bladder wall throughout the treatment. One factor especially amenable to exploration in a deformable geometry is therapeutic instillation volume ( ). We noticed throughout our own experiments using a coformulation of chitosan and interleukin-12 as an immunotherapy against murine bladder cancer that the mouse bladders were completely inflated throughout the application of the therapy (Zaharoff et al., 2009; Smith et al., 2015; Smith et al., 2016). We postulated that this inflation could impact the effectiveness of our therapeutic and represent a major difference between our preclinical model and current clinical practise. Indeed, most clinical applications call for an
). We noticed throughout our own experiments using a coformulation of chitosan and interleukin-12 as an immunotherapy against murine bladder cancer that the mouse bladders were completely inflated throughout the application of the therapy (Zaharoff et al., 2009; Smith et al., 2015; Smith et al., 2016). We postulated that this inflation could impact the effectiveness of our therapeutic and represent a major difference between our preclinical model and current clinical practise. Indeed, most clinical applications call for an  of 20–50 ml, which is hardly enough to fill or stretch a bladder whose capacity is typically near 500 ml (Au et al., 2001). Therefore, the goals of this study are 2-fold: 1) construct a finite element biphasic model of ID and 2) investigate the effects of umbrella cell transport properties, instillation concentration and instillation volume on ID.
of 20–50 ml, which is hardly enough to fill or stretch a bladder whose capacity is typically near 500 ml (Au et al., 2001). Therefore, the goals of this study are 2-fold: 1) construct a finite element biphasic model of ID and 2) investigate the effects of umbrella cell transport properties, instillation concentration and instillation volume on ID.
2. Methods
2.1 Geometry
FEBio is a free, open-source finite element analysis programme specifically tailored for solving biological problems (Ateshian et al., 2011; Jones et al., 2015). FEBio tackles some of the unique challenges of biological materials including very large deformations (hyperelastic materials), viscoelastic behaviour and coupled interactions between fluids and tissues. Because the focus of this model is transport during wall thinning and not the accurate evaluation of whole-bladder inflation we assumed the bladder to be spherical. Using this assumption, a geometry was constructed using Preview 1.19.00, FEBio’s preprocessing environment (Fig. 2). To mitigate computational costs, symmetry was used to reduce the geometry to 1/8 of a sphere with an initial inner diameter of 20 mm (corresponding to an empty volume of 33.5 ml) and a wall thickness of 5 mm (Haylen, 2006; Cruz et al., 2009). The geometry was broken into three material sections: umbrella cells, urothelium and muscle layers. Each material was assigned identical mechanical properties but different transport properties. A hexagonal mesh with a radial bias of 1.1 was applied with 25 segments in the radial direction giving thicknesses of 50  , 185
, 185  and 4.75 mm for the umbrella, urothelium and muscle layers, respectively, closely mimicking the actual anatomy (Wientjes et al., 1993). A cursory mesh analysis showed minimal effects when increasing the number of elements (Supplemental Fig. 1).
and 4.75 mm for the umbrella, urothelium and muscle layers, respectively, closely mimicking the actual anatomy (Wientjes et al., 1993). A cursory mesh analysis showed minimal effects when increasing the number of elements (Supplemental Fig. 1).
Fig. 2.

Bladder geometry and mesh used for analysis. The bladder was assumed to be sphere and reduced to 1/8 of a sphere via symmetry. Three layers were included with identical mechanical properties and varying transport properties as indicated. The baseline conditions for umbrella permeability and solubility are shown. P = Permeability; Sol = Solubility.
of a sphere via symmetry. Three layers were included with identical mechanical properties and varying transport properties as indicated. The baseline conditions for umbrella permeability and solubility are shown. P = Permeability; Sol = Solubility.
2.2 Parameter selection
The parameters for constitutive models describing the mechanical and transport properties of each bladder layer are provided in Table 1. Identifying a constitutive model for the bladder was non-trivial. While there has been significant work on bladder mechanics of multiple species, including pigs, dogs, rabbits, cows, rats, cats and toads, there is a dearth of information regarding human bladder mechanics. However, most of those species have bladders with significantly different mechanical properties than the human organ with the porcine bladder acting as the closest human analogue (Dahms et al., 1998). Thus, in choosing constitutive models and parameters for this study, strong preference was given to human data complemented by pig data when necessary.
Table 1.
Material properties of intravesical delivery model in FEBio and their sources. For transport properties, the values from top to bottom reflect the umbrella, urothelial and muscle layers, respectively. * indicates parameters that vary based on the protocol with baseline values shown.
| Material mechanical properties | |||||
|---|---|---|---|---|---|
| Parameter | Description | Value | Units | Source | Ref. |
| Material type | Holmes–Mow | FEBio handbook | Holmes, 1990 | ||
| Solid volume fraction | Fraction of material that is solid matrix | 0.08 | - | Literature value for Pig Extracellular Matrix | Cheng, 2010 |
| Material density | Density of solid matrix -used for body forces | 1.0E-09 | Tonne/m3 | Density of water | - |
| Young’s modulus | Elastic modulus | 0.005 | Mega Pascal (MPa) | Human linear model | Spirka, 2012 |
| Poisson’s ratio | Ratio of thinning | 0.43 | - | Human linear model/empirical | Spirka, 2012 |
| Beta | Exponential stiffening coefficient | 0.001 | - | Empirical - curve matching | - |
| Material Transport Properties | |||||
| Parameter | Description | Value | Units | Source | |
| Permeability | Hydraulic permeability of solvent in solid matrix | 2.94e-5* 2.94e-5, 6.32e-5 | mm4/(N*s) | From literature value for Pipemedic Acid (PPA) | Grabnar, 2006 |
| Free Diffusivity | Diffusivity of solute in solvent | 0.001 | mm2/s | Large value to simulate rapid diffusion | - |
| Diffusivity | Diffusivity of solute within solid matrix | 2.94e-5*, 2.94e-5, 6.32e-5 | mm2/s | From literature value for PPA | Grabnar, 2006 |
| Solubility | Fraction of pores that can accommodate the solute | 0.38*, 1.00, 1.00 | - | From literature value for partition coefficient of PPA | Grabnar, 2006 |
| (continued). | |||||
There have been four investigations that fit the data from the mechanical testing of human bladders to constitutive models. All of those studies utilized uniaxial testing to fit simple neo-Hookean or Moonley–Rivlin models (Dahms et al., 1998; Martins et al., 2011; Spirka et al., 2012; Chantereau et al., 2014). Unfortunately, those models are poor choices for biphasic analysis, as they are inherently incompressible. Consequently,a Holmes–Mow constitutive model has been chosen to describe the mechanical properties of the bladder (Holmes & Mow, 1990).
Table 1.
Continued.
| Prescribed values | |||||||
|---|---|---|---|---|---|---|---|
| Parameter | Description | Value | Units | Source | |||
| Intravesical solute concentration | Concentration of therapeutic in the lumen | 0.0001* | mM | Small values to limit impact of osmotic pressure. Decreases linearly to reflect urine dilution at 1 ml/min | Wientjes, 1993 | ||
| Intravesical fluid pressure | Pressure due to fluid instillation; drives expansion | 0-0.0046* | MPa | Literature pressure–volume curves of (P, V) = (0,33.5) to (0.0046,500). Values increased to reflect instillation volume/urine production | Brostrom, 2002 Damaser, 1999 | ||
| Model data | |||||||
| Parameter | Description | Value | Units | Source | |||
| Absolute temperature | Global temperature | 310.15 | K | Body temperature | - | ||
| R | Universal gas constant | 8.34E-06 | mJ/(nMol* K) | Universal constant | - | ||
Transport properties have been based almost entirely on pig data. Values of pipemedic acid (MW = 303 Da) diffusivity from data by Grabnar et al. were used to estimate permeability and diffusivity (Grabnar et al., 2003; Grabnar et al., 2006). These values should correspond to many relevant molecules as the size of pipemedic acid is similar to other bladder therapeutics including mitomycin C (334 Da), valrubicin (724 Da), docetaxel (808 Da), thiotepa (189 Da) and gemcitabine (263 Da). The umbrella layer solubility, a dimensionless value describing the fraction of pores that accommodates a solute, was assumed to correspond to the partition coefficient reported by Grabnar et al. (2006).The remaining layers were assigned a solubility of one. Two additional major assumptions concerning this model are isotropic mechanical properties and constant isotropic transport properties. Although it is certainly possible that large values of strain could affect solute transport, the experimental data involving large strains and solute transport are lacking with only a single study performed in rabbit bladders. This study showed that while the transmural electrical resistance decreased with stretch transport of fluorescein remained the same (Carattino et al., 2013).
2.3 Loads, boundary conditions, and initial conditions
Each of the edges of the geometry was constrained to induce sliding along its plane. The outer surface of the bladder was not assumed to be free-draining. The initial fluid pressure and solute concentrations within the bladder wall were set to zero. On the inner surface of the bladder, variable effective fluid pressure and effective solute concentrations were prescribed depending on the ID scenario as described in Supplementary Table 1. The instillation of therapeutic was assumed to take place over 6 min. We assumed a rate of 1 ml/min for all scenarios in accordance with literature values (Davies & Morris, 1993).
The concentrations of therapeutic used clinically vary depending on the therapeutic. As an example, a typical 40-ml instillation of 40-mg mitomycin-C has a 2–3-mM concentration. However, here the solute concentration was assumed to be arbitrarily low (100 pM) to minimize its influence on the ambient fluid pressure. This concentration decreased over time as a function of dose and urine production as
 |
(2.3.1) |
 |
(2.3.2) |
 |
(2.3.3) |
where C is the solute concentration,  is the solute concentration instilled into the lumen,
is the solute concentration instilled into the lumen,  is the installation volume, V is the total volume in the bladder at a given time point,
is the installation volume, V is the total volume in the bladder at a given time point,  is the volume of urine in the bladder,
is the volume of urine in the bladder,  is the rate of urine production and t is the duration of instillation. These equations do not account for solute transported into the wall of the bladder. However, in a previous diffusion chamber model of the bladder transport, the amount of drug transported into the wall remained below 8% of instilled therapeutic even after 120 min (Damaser, 1999; Grabnar et al., 2003; Grabnar et al., 2006).
is the rate of urine production and t is the duration of instillation. These equations do not account for solute transported into the wall of the bladder. However, in a previous diffusion chamber model of the bladder transport, the amount of drug transported into the wall remained below 8% of instilled therapeutic even after 120 min (Damaser, 1999; Grabnar et al., 2003; Grabnar et al., 2006).
2.4 Mathematical formulations and solving conditions
All analyses were performed in FEBio 2.5.1. The full mathematical formulations used by FEBio to solve solute transport problems have been described at length in other manuscripts and will not be reiterated in full here (Ateshian et al., 2011; Ateshian et al., 2013). Briefly, FEBio solves governing equations developed out of a framework of mixture theory in which there are a solid matrix (denoted by a superscript s), a solvent (denoted by a superscript w) and a solute (denoted by a superscript u). The finite element solver then seeks solutions to three partial differential equations for the three unknowns of u (displacement of the solid matrix), p (fluid pressure) and c (solute concentration). The first partial differential equation in the system is the mixture momentum balance
 |
(2.4.1) |
where T is the Cauchy stress, p is the fluid pressure and  is the stress due to strain in the solid matrix. The second equation is the mixture balance of mass described by
is the stress due to strain in the solid matrix. The second equation is the mixture balance of mass described by
 |
(2.4.2) |
where  is the velocity of the solid matrix and w is the volumetric flux of solvent with respect to the solid. The third equation is the solute mass balance described by
is the velocity of the solid matrix and w is the volumetric flux of solvent with respect to the solid. The third equation is the solute mass balance described by
 |
(2.4.3) |
where  is the mixture volume fraction of the solvent, c is the solute concentration, j is the molar flux of solute relative to the solid matrix and v
is the mixture volume fraction of the solvent, c is the solute concentration, j is the molar flux of solute relative to the solid matrix and v is the velocity of the solid matrix. Three algebraic relationships complement these partial differential equations. First, the solid mass balance
is the velocity of the solid matrix. Three algebraic relationships complement these partial differential equations. First, the solid mass balance
 |
(2.4.4) |
where  is the volume fraction of the solid matrix,
is the volume fraction of the solid matrix,  is the volume fraction of the solid matrix in the reference state and J is the determinant of the deformation gradient. Second, the solvent momentum balance which represents Darcy’s law of fluid permeation through a porous media
is the volume fraction of the solid matrix in the reference state and J is the determinant of the deformation gradient. Second, the solvent momentum balance which represents Darcy’s law of fluid permeation through a porous media
 |
(2.4.5) |
where w is the volumetric flux of the solvent relative to the solid matrix,  is the second-order permeability tensor of the solute
is the second-order permeability tensor of the solute  solvent through the solid matrix,
solvent through the solid matrix,  is the true density of the solvent, M is the molar mass of the solute, c is the concentration of the solute, d is the solute diffusivity tensor and
is the true density of the solvent, M is the molar mass of the solute, c is the concentration of the solute, d is the solute diffusivity tensor and  is the diffusivity of the solute within the solvent.
is the diffusivity of the solute within the solvent.  and
and  are the mechanochemical potentials of the solvent and solute, respectively, as defined by
are the mechanochemical potentials of the solvent and solute, respectively, as defined by
 |
(2.4.6) |
 |
(2.4.7) |
where  and
and  are the chemical potentials of the solvent and solute, respectively, at an arbitrary reference point and absolute temperature
are the chemical potentials of the solvent and solute, respectively, at an arbitrary reference point and absolute temperature  , p is the pressure of the fluid (solvent
, p is the pressure of the fluid (solvent  solute),
solute),  is the reference fluid pressure, R is the gas constant, k is solubility as defined by the number of pores accessible to a given solute and
is the reference fluid pressure, R is the gas constant, k is solubility as defined by the number of pores accessible to a given solute and  is the reference state solute concentration.
is the reference state solute concentration.
Third, the solute momentum balance which represents Fick’s law of diffusion for a solute
 |
(2.4.8) |
where j is the molar flux of the solute with respect to the solid, d is the solute diffusivity tensor, M is the molar mass of the solute,  is the volume fraction of the solvent,
is the volume fraction of the solvent,  is the diffusivity of the solute within the solvent and w is the volumetric flux of the solvent relative to the solid matrix. These equations are then solved via finite element analysis using an iterative Newton scheme (Maas et al., 2012).
is the diffusivity of the solute within the solvent and w is the volumetric flux of the solvent relative to the solid matrix. These equations are then solved via finite element analysis using an iterative Newton scheme (Maas et al., 2012).
Specific solving conditions prescribed for FEBio for this particular problem were the following: a full Newton non-linear solution method; a linear, non-symmetric solver storage matrix; and relative tolerances set to minimum values for displacement (1.0E-11), energy (1.0E-7), residual (1.0E-6), concentration (5.0E-2) and pressure (1.0E-10). The solute transport was analysed over a 3,600-s instillation with a time step of 120 s.
2.5 Postprocessing
Nodal values of displacement, effective solute concentration and effective fluid pressure were exported at must points corresponding to 120-s intervals via FEBio’s logfile output feature. For simplicity, all values of ‘effective solute concentration’ will simply be referred to as ‘solute concentration’ for the remainder of this manuscript. The output was imported into MATLAB (Mathworks, Natick, MA) using GIBBON’s importFEBIO_logfile function (Moerman, 2016). Custom functions were then used to parse the data by depth, generate average values for each depth, calculate cumulative concentrations and generate plots. Prism (GraphPad Software, Inc., La Jolla, CA) and Illustrator (Adobe Systems Incorporated, San Jose, CA) were used to compose the final figures.
3. Results
3.1 Bladder inflation and wall deformation
FEBio allows for the modelling of solute transport through a deformable geometry—an aspect not captured in existing ID models. The model was solved over a range of values for parameters that could influence ID (Table 2). In our model,  could not be directly prescribed without overprescribing the model. Therefore, a range of fluid pressures within physiological values was prescribed until the lumen volume reached 500 ml—here considered the maximum capacity of a bladder. This pressure–volume relationship (Supplementary Fig. 2) was then used to calculate fluid pressures corresponding to specific volumes as detailed in Supplementary Table 1. In all instances, the instillation was prescribed over 6 min followed by urine production at a rate of 1 ml/min.
could not be directly prescribed without overprescribing the model. Therefore, a range of fluid pressures within physiological values was prescribed until the lumen volume reached 500 ml—here considered the maximum capacity of a bladder. This pressure–volume relationship (Supplementary Fig. 2) was then used to calculate fluid pressures corresponding to specific volumes as detailed in Supplementary Table 1. In all instances, the instillation was prescribed over 6 min followed by urine production at a rate of 1 ml/min.
Table 2.
Intravesical delivery parameters investigated.
| Parameter | Symbol | Baseline | Min | Max | Units |
|---|---|---|---|---|---|
| Instillation volume | V inst | 50 | 50 | 250 | ml |
| Therapeutic concentration | C inst | 0.0001 | 0.00001 | 0.001 | mM |
| Umbrella cell solubility | U sol | 0.38 | 0.1 | 1.0 | - |
| Umbrella cell permeability | U perm | 2.96E-5 | 2.96E-6 | 2.96E-4 | mm /(N*s) /(N*s) |
A representative result of bladder displacement is shown in Fig. 3(a). To examine the effect of bladder volume on wall thickness, the model was solved for a range of pressures corresponding to nine different  (Fig. 3b). A baseline configuration of 50 ml was used for comparison across instillation volumes. There was a sharp initial drop in thickness due to therapeutic instillation followed by continued thinning due to urine production. Indeed,
(Fig. 3b). A baseline configuration of 50 ml was used for comparison across instillation volumes. There was a sharp initial drop in thickness due to therapeutic instillation followed by continued thinning due to urine production. Indeed,  had a large impact on the bladder wall thickness. Immediately after a 50-ml instillation the wall was 4.17-mm thick versus 1.67-mm thick for a 250-ml instillation. After instillation, the thinning due to urine production was more pronounced for smaller instillation volumes with the wall thinning from 4.17–2.68 mm after a 50-ml instillation and from 1.67–1.5 mm following a 250-ml instillation. Overall, this resulted in vastly different mean wall thicknesses due to
had a large impact on the bladder wall thickness. Immediately after a 50-ml instillation the wall was 4.17-mm thick versus 1.67-mm thick for a 250-ml instillation. After instillation, the thinning due to urine production was more pronounced for smaller instillation volumes with the wall thinning from 4.17–2.68 mm after a 50-ml instillation and from 1.67–1.5 mm following a 250-ml instillation. Overall, this resulted in vastly different mean wall thicknesses due to  ranging from 3.5 mm for
ranging from 3.5 mm for  –1.78 mm for
–1.78 mm for  ml.
ml.
Fig. 3.

(a) Bladder deformation at the beginning, after instillation and at the end of a 60-min intravesical treatment with  of 50 ml (top) and 100 ml (bottom). The distribution of displacement reflects the thinning bladder wall. The bladder volume over time is shown in (b) for a range of
of 50 ml (top) and 100 ml (bottom). The distribution of displacement reflects the thinning bladder wall. The bladder volume over time is shown in (b) for a range of  . The wall thickness (c) is a function of
. The wall thickness (c) is a function of  and continued urine production over time.
and continued urine production over time.  Volume of instillation;
Volume of instillation;  Volume of urine produced during treatment;
Volume of urine produced during treatment;  Final volume at the end of treatment.
Final volume at the end of treatment.
3.2 Solute transport throughout intravesical instillation
The primary purpose of this study is not a mechanical analysis of bladder deformation but rather the solute transport during that deformation. Solute transport was driven by a solute concentration that was prescribed on the inner surface of the bladder. Fig. 4(a) describes the prescribed concentration in the lumen under baseline instillation conditions (Table 2). The concentration time course of the solute specified depths within the umbrella, urothelium and muscle layers are shown in Figs 4(b–d). In this baseline scenario, the solute concentration quickly rises to 60 pM in the umbrella cells by 20 min and 30 pM in the urothelium by 30 min but requires 45 min to rise above 0.1 pM in the deeper muscle layers.
Fig. 4.

Solute concentration and exposure throughout the bladder in the baseline condition ( ml,
ml,  pg/ml,
pg/ml,  ,
,  2.94E-5 and D = 60 min). The concentration time course is shown in the lumen (a), umbrella cells (b), urothelium (c) and halfway through the muscle layer (d) with respect to time. The concentration profile (e) across the geometry after 60 min of instillation shows minimal solute penetration beyond 2.5 mm of depth. The cumulative exposure (f) across the bladder geometry demonstrates a lack of exposure at deeper layers. Bladder depth refers to the distance from the lumen in the nondeformed geometry.
2.94E-5 and D = 60 min). The concentration time course is shown in the lumen (a), umbrella cells (b), urothelium (c) and halfway through the muscle layer (d) with respect to time. The concentration profile (e) across the geometry after 60 min of instillation shows minimal solute penetration beyond 2.5 mm of depth. The cumulative exposure (f) across the bladder geometry demonstrates a lack of exposure at deeper layers. Bladder depth refers to the distance from the lumen in the nondeformed geometry.  Volume of instillation;
Volume of instillation;  Solute concentration in instillation;
Solute concentration in instillation;  Umbrella solubility;
Umbrella solubility;  Umbrella permeability; D= Duration of instillation.
Umbrella permeability; D= Duration of instillation.
Fig. 4(e) describes the final concentration profile across the geometry of the bladder. All concentration profiles are shown relative to the starting (nondeformed) geometry. Once again, there is a substantial drop in solute concentration within the muscle layer. This is also reflected in Fig. 4(f) which describes the cumulative exposure ( ) across the bladder geometry as defined by the integration of the concentration with respect to time. This ‘cumulative concentration’ (also termed
) across the bladder geometry as defined by the integration of the concentration with respect to time. This ‘cumulative concentration’ (also termed  by Schmittgen et al.) acts as a description of total exposure while considering transient solute concentrations (Schmittgen et al., 1991).
by Schmittgen et al.) acts as a description of total exposure while considering transient solute concentrations (Schmittgen et al., 1991).
3.3 Effects of umbrella layer solubility and permeability on transport
One strategy for enhancing ID is to increase the absorption rate across the umbrella cells by the administration of a permeation enhancing agent such as chitosan or dimethyl sulfoxide (Giannantoni et al., 2006). To investigate the effects of absorption enhancers on ID, two surrogate parameters were examined. The first, umbrella layer solubility ( )—a description of the fraction of pores accessible to a given solute—was selected to describe the partition coefficient generated by both the mucus layer on top of the umbrella cells as well as the umbrella cells themselves. However, as can be seen in Fig. 5(a), a 10-fold difference from
)—a description of the fraction of pores accessible to a given solute—was selected to describe the partition coefficient generated by both the mucus layer on top of the umbrella cells as well as the umbrella cells themselves. However, as can be seen in Fig. 5(a), a 10-fold difference from  to
to  incurring a relative increase in
incurring a relative increase in  of 2 to 3-fold depending on the wall depth.
of 2 to 3-fold depending on the wall depth.
Fig. 5.

Effects of the umbrella layer solubility (a), umbrella layer permeability (b) and both increasing (c) and decreasing (d) starting drug concentration on cumulative exposure across the bladder. Bladder depth refers to the distance from the lumen in the nondeformed geometry. C instilled solute concentration; U
instilled solute concentration; U Umbrella solubility; U
Umbrella solubility; U Umbrella permeability.
Umbrella permeability.
Umbrella cell permeability ( )—a description of the solvent diffusivity within the solid matrix—was also investigated as a parameter to model absorption enhancement. In all cases, the related parameter of diffusivity—the diffusivity of the solute within the solid matrix—was changed to match the value of permeability. Permeability did modulate delivery (Fig. 6b), with a 100-fold difference, from
)—a description of the solvent diffusivity within the solid matrix—was also investigated as a parameter to model absorption enhancement. In all cases, the related parameter of diffusivity—the diffusivity of the solute within the solid matrix—was changed to match the value of permeability. Permeability did modulate delivery (Fig. 6b), with a 100-fold difference, from  E-6 to
E-6 to  E-4, incurring a relative increase in
E-4, incurring a relative increase in  of 6–11-fold depending on the wall depth.
of 6–11-fold depending on the wall depth.
Fig. 6.

Impact of volume of instillation on cumulative solute exposure throughout the bladder wall both as absolute values (a, c) and relative (b, d) to the baseline condition (V ml, C
ml, C pg/ml, U
pg/ml, U , U
, U E-5 and D = 60 min). In the upper graphs (a, b) the instillation concentration was changed with respect to instillation volume to keep the dose constant (constant C
E-5 and D = 60 min). In the upper graphs (a, b) the instillation concentration was changed with respect to instillation volume to keep the dose constant (constant C ). In the lower graphs (c, d), the concentration was kept constant regardless of instillation volume, thus prescribing a changing dose. Bladder depth refers to the distance from the lumen in the nondeformed geometry. V
). In the lower graphs (c, d), the concentration was kept constant regardless of instillation volume, thus prescribing a changing dose. Bladder depth refers to the distance from the lumen in the nondeformed geometry. V Volume of instillation; C
Volume of instillation; C Solute concentration; U
Solute concentration; U Umbrella solubility; U
Umbrella solubility; U Umbrella permeability; D = Duration of instillation.
Umbrella permeability; D = Duration of instillation.
3.4 Concentration of the instillation directly impacts solute penetration
Solute penetration into the bladder wall is a diffusion driven process. Therefore, it stands to reason that altering the concentration of the instillation ( ) will impact solute distribution. Indeed, increasing (Fig. 5c) or decreasing (Fig. 5d) had a linear impact on the solute concentration and cumulative solute concentration across the entire bladder geometry with a 10-fold increase or decrease in
) will impact solute distribution. Indeed, increasing (Fig. 5c) or decreasing (Fig. 5d) had a linear impact on the solute concentration and cumulative solute concentration across the entire bladder geometry with a 10-fold increase or decrease in  resulting in a 10-fold increase or decrease in
resulting in a 10-fold increase or decrease in  . Unlike
. Unlike  and
and  , this change in
, this change in  was constant regardless of bladder depth.
was constant regardless of bladder depth.
3.5 Instillation volume impacts delivery to deeper layers
To investigate the impact of  on solute exposure,
on solute exposure,  was increased in 25-ml increments from the baseline condition of 50–250 ml (Fig. 6). This analysis was performed with both doses were held constant (Figs 6a and b) and for the starting concentration held constant resulting in a changing dose (Figs 6c and d). When a constant dose was applied, the solute exposure was reduced in both absolute (Fig. 6a) and relative (Fig. 6b) terms for layers closer to the lumen (starting depth
was increased in 25-ml increments from the baseline condition of 50–250 ml (Fig. 6). This analysis was performed with both doses were held constant (Figs 6a and b) and for the starting concentration held constant resulting in a changing dose (Figs 6c and d). When a constant dose was applied, the solute exposure was reduced in both absolute (Fig. 6a) and relative (Fig. 6b) terms for layers closer to the lumen (starting depth  mm). Conversely the exposure was increased by orders of magnitude in deeper layers (Fig. 6b). This impact on deeper layers was also maintained when the dose was changed alongside
mm). Conversely the exposure was increased by orders of magnitude in deeper layers (Fig. 6b). This impact on deeper layers was also maintained when the dose was changed alongside  to obtain the same starting concentration regardless of
to obtain the same starting concentration regardless of  (Figs 6c and d). In this instance, the exposure for the more luminal layers was also increased with respect to the baseline conditions.
(Figs 6c and d). In this instance, the exposure for the more luminal layers was also increased with respect to the baseline conditions.
Because increasing  increased exposure to the deeper layers, we were curious if this effect could be used to reduce the duration of treatment yet achieve the same level of exposure as the baseline condition. Fig. 7 shows the time to reach the baseline final exposure value (
increased exposure to the deeper layers, we were curious if this effect could be used to reduce the duration of treatment yet achieve the same level of exposure as the baseline condition. Fig. 7 shows the time to reach the baseline final exposure value ( ) for different instillation volumes with both constant dose (
) for different instillation volumes with both constant dose ( , Fig. 7a) and constant concentration (
, Fig. 7a) and constant concentration ( , Fig. 7b). In both instances, larger instillation volumes decreased the amount of time needed to reach the same values of
, Fig. 7b). In both instances, larger instillation volumes decreased the amount of time needed to reach the same values of  as the baseline conditions for deeper layers of the wall. For example, under the conditions of
as the baseline conditions for deeper layers of the wall. For example, under the conditions of  and at a depth of 2.5mm
and at a depth of 2.5mm  . This means that a 25-min instillation of 250 ml generates the same exposure as a 50-ml instillation given for 60 min at that depth.
. This means that a 25-min instillation of 250 ml generates the same exposure as a 50-ml instillation given for 60 min at that depth.
Fig. 7.

Instillation volume impacts the amount of time needed to reach the same final cumulative solute exposure at a given depth as in the baseline condition (V ml, C
ml, C pg/ml, U
pg/ml, U , U
, U E-5 and D = 60 min). Maintaining a constant dose (a) primarily impacted deeper layers (>1mm from the lumen), while (b) a constant C
E-5 and D = 60 min). Maintaining a constant dose (a) primarily impacted deeper layers (>1mm from the lumen), while (b) a constant C (or changing dose) sped the exposure to all layers. Bladder depth refers to the distance from the lumen in the nondeformed geometry. V
(or changing dose) sped the exposure to all layers. Bladder depth refers to the distance from the lumen in the nondeformed geometry. V Volume of instillation C
Volume of instillation C Solute concentration; U
Solute concentration; U Umbrella solubility; U
Umbrella solubility; U Umbrella permeability; D = Duration of instillation.
Umbrella permeability; D = Duration of instillation.
3.6 Effects on total tissue exposure
To test the amount of total therapeutic exposure within the whole bladder and specific regions of the bladder we defined the tissue exposure as
 |
(3.6.1) |
 |
(3.6.2) |
 |
(3.6.3) |
where  is the tissue exposure and
is the tissue exposure and  is the average cumulative exposure for element i with nodes i and i-1, respectively, defining locations of the outer and luminal sides of the nodes along the radial axis (R) of the elemental layer.
is the average cumulative exposure for element i with nodes i and i-1, respectively, defining locations of the outer and luminal sides of the nodes along the radial axis (R) of the elemental layer.  defines the average cumulative exposure for a given elemental layer.
defines the average cumulative exposure for a given elemental layer.  describes the volume of an elemental layer based on the starting (nondeformed) configuration. In the geometry considered here, 25 elements (n = 25) were defined along the radial axis with 26 corresponding nodes.
describes the volume of an elemental layer based on the starting (nondeformed) configuration. In the geometry considered here, 25 elements (n = 25) were defined along the radial axis with 26 corresponding nodes.
Using this tissue exposure ( ) the bladder was segmented into the urothelium + umbrella (i=2–5, maximum depth = 235
) the bladder was segmented into the urothelium + umbrella (i=2–5, maximum depth = 235  m) and muscle layers (i=6–26).
m) and muscle layers (i=6–26).  was plotted against changing;
was plotted against changing;  ,
,  ,
,  ,
,  and
and  both as absolute values (Fig. 8a) and as the percentage of total
both as absolute values (Fig. 8a) and as the percentage of total  due to exposure in the muscle layer (Fig. 8b). Each parameter effected the urothelium and muscle tissue exposure in different ways.
due to exposure in the muscle layer (Fig. 8b). Each parameter effected the urothelium and muscle tissue exposure in different ways.
Fig. 8.

Instillation volume impacts delivery to deeper bladder layers more efficiently than other parameters. The total tissue exposure (a) is a volumetric measure of cumulative exposure for the urothelium (<235  m) and the muscle layer (> 235
m) and the muscle layer (> 235  m). The percentage of total tissue exposure due to exposure within the muscle layer is shown in (b). Base conditions are the following: V
m). The percentage of total tissue exposure due to exposure within the muscle layer is shown in (b). Base conditions are the following: V ml; C
ml; C pg/ml; U
pg/ml; U ; U
; U E-5; D = 60 min; V
E-5; D = 60 min; V Volume of instillation with a constant dose; V
Volume of instillation with a constant dose; V Volume of instillation with a constant concentration; C
Volume of instillation with a constant concentration; C Instillation solute concentration; U
Instillation solute concentration; U Umbrella solubility; U
Umbrella solubility; U Umbrella permeability; D = Duration of instillation.
Umbrella permeability; D = Duration of instillation.
 had a minimal impact on exposure. An increase of
had a minimal impact on exposure. An increase of  from 0.38 to 1.00 led to 122% of baseline
from 0.38 to 1.00 led to 122% of baseline  . However, a decrease in
. However, a decrease in  from 0.38 to 0.1 resulted in 57% of baseline
from 0.38 to 0.1 resulted in 57% of baseline  .
.
Likewise,  had limited effects on bladder exposure. Decreasing
had limited effects on bladder exposure. Decreasing  had a more substantial impact than increasing
had a more substantial impact than increasing  . Indeed, 0.1× or 0.5× the baseline
. Indeed, 0.1× or 0.5× the baseline  caused a decrease in
caused a decrease in  to 30.8% and 78% of baseline values, respectively, while increasing
to 30.8% and 78% of baseline values, respectively, while increasing  by 2× or 10× produced returns of 117% or 136% of baseline values, respectively.
by 2× or 10× produced returns of 117% or 136% of baseline values, respectively.  had the greatest impact on
had the greatest impact on  reflected by a linear increase in both
reflected by a linear increase in both  and
and  . The
. The  :
:  ratio was unchanged regardless of
ratio was unchanged regardless of  .
.
Instillation volume with or without a changing dose had substantial impact on  ,
,  and
and  . Increase in
. Increase in  caused
caused  to decrease but increased
to decrease but increased  beyond baseline levels—despite lower overall exposure. Increase in
beyond baseline levels—despite lower overall exposure. Increase in  caused a non-linear increase in
caused a non-linear increase in  ,
,  and
and  . For both increasing
. For both increasing  and
and  the
the  :
:  ratio increased substantially.
ratio increased substantially.
4. Discussion
To our knowledge, this is the first study to investigate intravesical solute transport throughout ID in the context of a deformable geometry. The key finding of this investigation is that increased bladder stretch and wall thinning can lead to enhanced targeting of deeper bladder layers. Indeed, no other parameter investigated herein was nearly as effective at increasing the proportion of solute exposure in the deeper layers (Fig. 8b). These results suggest that if a therapeutic needs to reach deeper layers of the bladder, then increasing  may be a viable means of increasing exposure within those layers.
may be a viable means of increasing exposure within those layers.
This could have implications for reaching residual muscle invasive bladder cancer cells with chemotherapies, tissue-resident immune cells (which typically dwell beyond the basement membrane) with immunotherapies or treating cystitis (Abraham & Miao, 2015). One downside of ID is patient discomfort throughout the application. This increased volume may provide a trade-off between increased discomfort for a shorter dwell time and a lower level of discomfort for the standard, longer dwell time. It may also increase the amount of the solute that reaches systemic circulation—an aspect not addressed with the current model.
One surprising result was the minimal impact that umbrella cell transport properties had on bladder penetration. This could be an artefact of both the parameters chosen to model the umbrella layer as well as the values ascribed to those parameters. Nonetheless, this model suggests that directly targeting either  or
or  with an absorption enhancer will only be meaningful for very large effects on their values.
with an absorption enhancer will only be meaningful for very large effects on their values.
4.1 Limitations of this model and future directions
We acknowledge that the conclusion we have drawn above is based on computational analysis. While we believe this approach allowed for meaningful insights into ID, it also comes with its limitations that ought to be considered and built upon in future analyses. These include
(1) The parameters used to delineate material properties, both mechanical and transport, are from diverse literature sources (Table 1) and from multiple species. Mechanical and transport data for the bladder are lacking, and more accurate data could inform a better model.
(2) This model was meant to be a first pass at modelling ID in a deforming geometry, thus several simplifications were assumed that could be built upon in further iterations. Some future features may include the following: accounting for the effects of solute loss in the lumen due transport into the bladder wall and degradation, including the viscoelastic nature of the bladder wall, defining a geometry truer to the actual non-spherical bladder, including the transport of drug into systemic circulation upon contact with the capillary infused layers of the bladder via chemical uptake, prescribing a strain dependence for permeability and solubility, considering continued solute transport post-catheter removal, defining mechanical properties that differ by layer and accounting for bladder lymphatics—which could be a key consideration given the immune stimulatory nature of many bladder instillations.
(3) Finally, this model relies on numerical simulation, has not been validated experimentally and should be considered but a single component in our understanding of ID. Future ex vivo studies should be performed to provide an experimental comparison. Nonetheless, this study is useful as it relies on well-understood principles of mass transport and allows for the investigation of multiple parameters at once.
5. Conclusions
ID is commonly employed without a full understanding of the underlying mechanisms and suffers from a lack of reproducibility and inability to penetrate the deeper layers of the bladder (Giannantoni et al., 2006). While other investigations have employed mathematical analysis to ID, this is the first to do so in a deforming geometry. This study suggests that intravesical delivery with larger instillation volumes enhances delivery to deeper, blood-perfused layers due to a shorter path length through the thinner, inflated bladder wall. This characteristic could be used to decrease dwell times, target deeper layers while avoiding overexposure of agents in the urothelium or even do the opposite (limit muscle/systemic exposure). Moving forward, there remains substantial work to validate this model, incorporate additional features as defined above and define optimal parameters for intravesical delivery—parameters that are likely to differ for each therapeutic and individual patient.
Supplementary Material
Supplementary material
Supplementary material is available at http://imammb.oxfordjournals.org.
Funding
This work was supported by grants from the National Institutes of Health, National Cancer Institute (R01CA172631, R15CA176648).
References
- Abraham S. N. & Miao Y. (2015) The nature of immune responses to urinary tract infections. Nat. Rev. Immunol., 15, 655–663. doi: 10.1038/nri3887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Cancer Society (2016) Cancer Facts & Figures 2016. Atlanta: American Cancer Society. [Google Scholar]
- Anderson J. M. & Italie C. M. (2009) Physiology and Function of the Tight Junction. Cold Spring Harb. Perspect. Biol., 1, a002584. doi: 10.1101/cshperspect.a002584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ateshian G. A., Albro M. B., Maas S. & Weiss J. A. (2011) Finite element implementation of mechanochemical phenomena in neutral deformable porous media under finite deformation. J. Biomech. Eng., 133, 081005. doi:10.1115/1.4004810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ateshian G. A., Maas S. A. & Weiss J. A (2013) Multiphasic finite element framework for modeling hydrated mixtures with multiple neutral and charged solutes. J. Biomech. Eng., 135, 111001.. doi: 10.1115/1.4024823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- J Au. L., Badalament R. A., Wientjes M. G., Young D. C., Warner J. A., Venema P. L., Pollifrone D. L., Harbrecht J. D., Chin J. L. & Lerner S. P (2001) Methods to improve efficacy of intravesical mitomycin C: results of a randomized phase III trial. J. Natl. Cancer Inst., 93, 597–604. doi: 10.1093/jnci/93.8.597. [DOI] [PubMed] [Google Scholar]
- Brostrom S., Jennum P. & Lose G. (2002) Short-term reproducibility of cystometry and pressure-flow micturition studies in healthy women. Neurourol. Urodyn., 21, 457–460. doi: 10.1002/nau.10019. [DOI] [PubMed] [Google Scholar]
- Carattino M. D., Prakasam S. H., Ruiz W. G., Clayton D. R., McGuire M., Gallo L. I. & Apodaca G. (2013) Bladder filling and voiding affect umbrella cell tight junction organization and function. Am. J. Physiol. Renal. Physiol., 305, F1158–F1168. doi: 10.1152/ajprenal.00282.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chantereau P., Brieu M., Kammal M., Farthmann J., Gabriel B. & Cosson M (2014) Mechanical properties of pelvic soft tissue of young women and impact of aging. Int. Urogynecol. J., 25, 1547–1553. doi:10.1007/s00192–014-2439–1. [DOI] [PubMed] [Google Scholar]
- Cheng H. Loai Y. Beaumont M. & Farhat W.A. (2010) The acellular matrix (ACM) for bladder tissue engineering: a quantitative magnetic resonance imaging study. Magnet. Reson. Med., 64, 341–348. doi: 10.1002/mrm.22404. [DOI] [PubMed] [Google Scholar]
- Cruz F., Heesakkers J., Khullar V. & Tubaro A. (2009) Bladder wall thickness in overactive bladder: does it have a role? Eur. Urol. Suppl., 8, 769–771. doi: 10.1016/j.eursup.2009.05.002. [DOI] [Google Scholar]
- Dahms S. E., Piechota H. J., Dahiya R., Lue T. F. & Tanagho E. A. (1998) Composition and biomechanical properties of the bladder acellular matrix graft: comparative analysis in rat, pig and human. Brit. J. Urol., 82, 411–419. doi:10.1046/j.1464–410X.1998.00748.x. [DOI] [PubMed] [Google Scholar]
- Damaser M. S. (1999) Whole bladder mechanics during filling. Scand. J. Urol. and Nephrol., 33, 51–58. doi: 10.1080/003655999750169385. [DOI] [PubMed] [Google Scholar]
- Davies B. & Morris T. (1993) Physiological parameters in laboratory animals and humans. Pharm. Res., 10, 1093–1095. doi:10.1023/A:1018943613122. [DOI] [PubMed] [Google Scholar]
- Giannantoni A., Stasi S. M., Chancellor M. B., Costantini E. & Porena M. (2006) New frontiers in intravesical therapies and drug delivery. Eur. Urol., 50, 1183–1193. doi: 10.1016/j.eururo.2006.08.025. [DOI] [PubMed] [Google Scholar]
- Grabnar I., Bogataj M., Belič A., Logar V., Karba R. & Mrhar A. (2006) Kinetic model of drug distribution in the urinary bladder wall following intravesical instillation. Int. J. Pharm., 322, 52–59. doi:10.1016/S0378–5173(03)00074–7. [DOI] [PubMed] [Google Scholar]
- Grabnar I., Bogataj M. & Mrhar A. (2003) Influence of chitosan and polycarbophil on permeation of a model hydrophilic drug into the urinary bladder wall. Int. J. Pharm., 256, 167–173. doi:10.1016/S0378–5173(03)00074–7. [DOI] [PubMed] [Google Scholar]
- Haylen B. T. (2006) The empty bladder. Int. Urogynecol. J., 18, 237–239. doi:10.1007/s00192–006-0111–0. [DOI] [PubMed] [Google Scholar]
- Holmes M. H. & Mow V. C. (1990) The nonlinear characteristics of soft gels and hydrated connective tissues in ultrafiltration. J. Biomech., 23, 1145–1156. doi:10.1016/0021–9290(90)90007-P. [DOI] [PubMed] [Google Scholar]
- Jones B., Hung C. & Ateshian G (2015) Biphasic analysis of cartilage stresses in the patellofemoral joint. J. Knee Surg., 29, 92–98. doi:10.1055/s-0035–1568989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konkle K. S., Berry S. H., Elliott M. N., Hilton L., Suttorp M. J., Clauw D. J. & Clemens J. Q. (2012) Comparison of an interstitial cystitis/bladder pain syndrome clinical cohort with symptomatic community women from the RAND Interstitial Cystitis Epidemiology study. J. Urol., 187, 508–512. doi: 10.1016/j.juro.2011.10.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maas S. A., Ellis B. J., Ateshian G. A. & Weiss J. A. (2012) FEBio: finite elements for biomechanics. J. Biomech. Eng., 134, 011005. doi: 10.1115/1.4005694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins P. A., Silva Filho A. L., A. M. Fonseca Santos R. M., A., Santos L., Mascarenhas T., Jorge R. M. N. & Ferreira A. J (2011) Uniaxial mechanical behavior of the human female bladder. Int. Urogynecol. J., 22, 991–995. doi:10.1007/s00192–011-1409–0. [DOI] [PubMed] [Google Scholar]
- Moerman K. M. (2016) Gibbon (Hylobates Lar). Zenodo. doi: 10.5281/zenodo.44404. [DOI] [Google Scholar]
- Schmittgen T. D., Wientjes M. G., Badalament R. A. & Au J. L. (1991) Pharmacodynamics of mitomycin C in cultured human bladder tumors. Cancer Res., 51, 3849–3856. [PubMed] [Google Scholar]
- Smith S. G., Baltz J. L., Koppolu B. P., Ravindranathan S., Nguyen K. & Zaharoff D. A. (2016) Immunological mechanisms of intravesical chitosan/interleukin-12 immunotherapy against murine bladder cancer. OncoImmunology, 6, e1259050. doi:10.1007/s00262–015-1672-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S. G., Prasanth Koppolu B., Ravindranathan S., Kurtz S. L., Yang L., Katz M. D. & Zaharoff D. A. (2015) Intravesical chitosan/interleukin-12 immunotherapy induces tumor-specific systemic immunity against murine bladder cancer. Cancer Immunol. Immunother., 64, 689–696. doi: 10.1080/2162402X.2016.1259050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spirka T., Kenton K., Brubaker L. & Damaser M.S (2012) Effect of material properties on predicted vesical pressure during a cough in a simplified computational model of the bladder and urethra. Ann. Biomed. Eng., 41, 185–194. doi:10.1007/s10439–012-0637-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyagi P., P Wu., Chancellor M., Yoshimura N. & Huang L. (2006) Recent advances in intravesical drug/gene delivery. Mol. Pharm., 3, 369–379. doi: 10.1021/mp060001j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wientjes M. G., Badalament R. A. & Au J. L. (1993) Use of pharmacologic data and computer simulations to design an efficacy trial of intravesical mitomycin C therapy for superficial bladder cancer. Cancer Chemother. Pharmacol., 32, 255–262. doi: 10.1007/BF00686169. [DOI] [PubMed] [Google Scholar]
- Zaharoff D. A., Hoffman B. S., Hooper H. B., Benjamin Jr C. J., Khurana K. K., Hance K. W., Rogers C. J., Pinto P. A., Schlom J. & Greiner J. W (2009) Intravesical immunotherapy of superficial bladder cancer with chitosan/interleukin-12. Cancer Res. 69, 6192–6199. doi:10.1158/0008–5472.CAN-09–1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.