In this cross-sectional cohort study, we analyzed maternal smoking behavior before and during pregnancy and the increased risk of SUID.
Abstract
Video Abstract
OBJECTIVES:
Maternal smoking during pregnancy is an established risk factor for sudden unexpected infant death (SUID). Here, we aim to investigate the effects of maternal prepregnancy smoking, reduction during pregnancy, and smoking during pregnancy on SUID rates.
METHODS:
We analyzed the Centers for Disease Control and Prevention Birth Cohort Linked Birth/Infant Death Data Set (2007–2011: 20 685 463 births and 19 127 SUIDs). SUID was defined as deaths at <1 year of age with International Classification of Diseases, 10th Revision codes R95 (sudden infant death syndrome), R99 (ill-defined or unknown cause), or W75 (accidental suffocation or strangulation in bed).
RESULTS:
SUID risk more than doubled (adjusted odds ratio [aOR] = 2.44; 95% confidence interval [CI] 2.31–2.57) with any maternal smoking during pregnancy and increased twofold between no smoking and smoking 1 cigarette daily throughout pregnancy. For 1 to 20 cigarettes per day, the probability of SUID increased linearly, with each additional cigarette smoked per day increasing the odds by 0.07 from 1 to 20 cigarettes; beyond 20 cigarettes, the relationship plateaued. Mothers who quit or reduced their smoking decreased their odds compared with those who continued smoking (reduced: aOR = 0.88, 95% CI 0.79–0.98; quit: aOR = 0.77, 95% CI 0.67–0.87). If we assume causality, 22% of SUIDs in the United States can be directly attributed to maternal smoking during pregnancy.
CONCLUSIONS:
These data support the need for smoking cessation before pregnancy. If no women smoked in pregnancy, SUID rates in the United States could be reduced substantially.
What’s Known on This Subject:
Approximately 3500 infants <1 year old die suddenly and unexpectedly each year in the United States. Previous research has revealed that maternal smoking during pregnancy is a known risk factor for sudden unexpected infant death (SUID).
What This Study Adds:
In this retrospective cross-sectional analysis (20 685 463 births and 19 127 SUIDs), we use advanced modeling techniques to quantitatively determine the effects of maternal smoking, smoking cessation, and smoking reduction in pregnancy on SUID rates with much higher resolution than previous studies.
In the United States, >3700 infants die annually from sudden unexpected infant death (SUID), which includes sudden infant death syndrome (SIDS), accidental suffocation and strangulation in bed, and ill-defined causes.1 Multiple epidemiologic studies have shown a strong relationship between maternal smoking and SIDS. Researchers of 1 meta-analysis reported a pooled risk associated with maternal prenatal smoking of nearly fourfold (risk ratio = 3.9; 95% confidence interval [CI] 3.8–4.1). Odds ratios (unadjusted) associated with postnatal maternal smoking range from 1.47 to 6.56.2 There are dose-dependent relationships between SIDS rates and both the number of cigarettes smoked prenatally3–5 and duration of smoke exposure postnatally.6,7 Moreover, there is compelling evidence that maternal smoking may play a causal role in SIDS deaths.2,8
Substantial work has been undertaken to understand the pathophysiology underlying this increased risk of sudden infant death. Abnormalities in major neurotransmitters, including serotonin and their receptors, have been well documented in the brainstems of SIDS infants,9–11 with experimental data supporting nicotine’s effects on respiration, autonomic regulation, chemosensitivity, sleep, and arousal.12–17 Maternal smoking has been linked to serotonergic abnormalities in important brainstem nuclei of SIDS infants.18,19 In animal models, nicotine increases serotonin release and alters the firing of serotonergic neurons in a dose-dependent manner.20,21 Serotonergic neuronal development may be disrupted by maternal smoking as early as the first trimester.18,19,22
Many conclude that maternal smoking is the strongest prenatal modifiable risk factor for SIDS in industrialized nations.23–25 Although previous research has focused on the association between pre- or postnatal smoking and sudden infant death, these studies have given limited attention to diagnostic preferences as they affect measured outcomes.26,27 Additionally, only 1 published study has provided details about prepregnancy smoking.28 Here, we use national vital statistics data29 and a logistic regression modeling approach to analyze maternal smoking behavior during pregnancy for all 2007–2011 US live births with complete smoking information (∼12 million births), using higher resolution than previous studies and expanding the analysis to the 3 major causes of SUID. Additionally, beginning in 2011, this data set recorded the number of cigarettes that mothers smoked in the 3 months prepregnancy. We thus analyzed maternal smoking and the risk of SUID by trimester by daily cigarette consumption, prepregnancy smoking levels, smoking reduction or cessation during pregnancy, and individual International Classification of Diseases, 10th Revision (ICD-10) cause of death to estimate population-attributable risk.
Methods
Study Design and Population
We conducted a retrospective, cross-sectional study to assess the relationship between SUID and self-reported maternal smoking before and during pregnancy, using data from the Centers for Disease Control and Prevention (CDC) Birth Cohort Linked Birth/Infant Death Data Set for births between 2007 and 2011.29 This data set does not include details about the frequency of autopsy or death scene investigation, although autopsy is an element of diagnostic criteria in SIDS.
We defined a SUID case as an infant (<365 days old) death with the following ICD-10 codes: R95 (SIDS), R99 (ill-defined and unknown cause), or W75 (accidental suffocation or strangulation in bed).
The analysis of the effects of maternal smoking before pregnancy was conducted by using only births in 2011 (3 134 781 total births, 2585 SUIDs; SUID rate 0.83 per 1000 live births), the first year the CDC reported on the number of daily cigarettes smoked in the 3 months before pregnancy.
We used the complete set of 20 685 463 births and 19 127 deaths and dichotomous smoking data (smoking versus no smoking) to estimate the number of deaths attributable to prenatal smoking.
Covariates
In this study, we aimed to make an inference regarding the effect of maternal prenatal smoking on SUID risk. We used regression adjustment of potential confounding variables to decrease bias and to improve the precision of estimates in the smoking-SUID association. On the basis of these calculations, we used the following covariates in all adjusted analyses: mother’s and father’s race and/or ethnicity/Hispanic origin, mother’s and father’s age, mother’s marital status, mother’s education, live birth order, number of prenatal visits, gestational length (weeks), delivery method (vaginal or cesarean), infant sex, and birth weight.
Statistical Analysis
To understand the relationship between the reported average number of cigarettes smoked per day and risk of SUID, we developed both a logistic regression model and a generalized additive model (GAM). CDC data include dichotomous data about maternal smoking (yes or no) for all births and also include daily number of cigarettes for 60% of births. To ensure that the data were consistent and that there was no bias effect, both sets of data were used to calculate adjusted odds ratios (aORs). The logistic regression model used the average number of cigarettes in the 3 trimesters as the predictor of primary interest, which was coded as a categorical variable, whereas the GAM used the same variable as the predictor of interest but coded as a continuous numerical variable. All logistic regression and GAM models were adjusted for covariates.
Using 2011 data and a logistic regression model, we assessed the increased risk from prepregnancy smoking using a variable that identified the smoking habits before and during pregnancy. We then used 3 logistic regression models to understand the effects of smoking in each trimester. The models were similar to the GAM, except that the daily number of cigarettes smoked in the 3 trimesters were modeled independently instead of averaging across all 3 trimesters.
We also examined the reduction in SUID risk when mothers quit or reduced the amount smoked compared with smokers who did not quit or reduce smoking during pregnancy. A new categorical variable was created to identify the mothers who smoked in the first trimester and then quit, reduced, or continued the number of daily cigarettes in later trimesters. If the number of cigarettes by the third trimester was 0, the mother was defined as having quit. If the number of total daily cigarettes in the second and third trimesters was less than the daily number of cigarettes in the first trimester multiplied by 2, the mother was categorized as a reduced smoker; those who continued to smoke the same amount (or more) were defined as continued smokers. In the model, we controlled for covariates and total number of cigarettes smoked during pregnancy.
To differentiate between SUID subcategories (R95, R99, and W75) and non-SUID causes of death, we developed separate logistic regression models to estimate the risk of each cause of death independently.
For estimating the proportion of SUID cases attributable to smoking, we used the same logistic regression model with a database in which all mothers were artificially set as nonsmokers.30 By assuming causation, the difference between the result of this model and actual SUID rates is the proportion of deaths that can be attributed to smoking.
Results
We investigated 20 685 463 births and 19 127 SUIDs during the years 2007–2011 (SUID rate 0.92 in 1000 live births). Of the births, 12 417 813 had complete prenatal smoking information, and of these cases, 10 737 met the SUID definition. In 2011, 11.5% of mothers smoked in the 3 months before pregnancy, and 8.9% smoked during pregnancy; 24.3% of smokers who smoked prepregnancy quit before the first trimester.
By using a dichotomous variable of smoking (yes or no), SUID risk more than doubled (aOR = 2.44; 95% CI 2.31–2.57) with any maternal smoking during pregnancy. The aOR was similar when calculated for cases in which there were data on number of cigarettes smoked during pregnancy (aOR = 2.40; 95% CI 2.23–2.59).
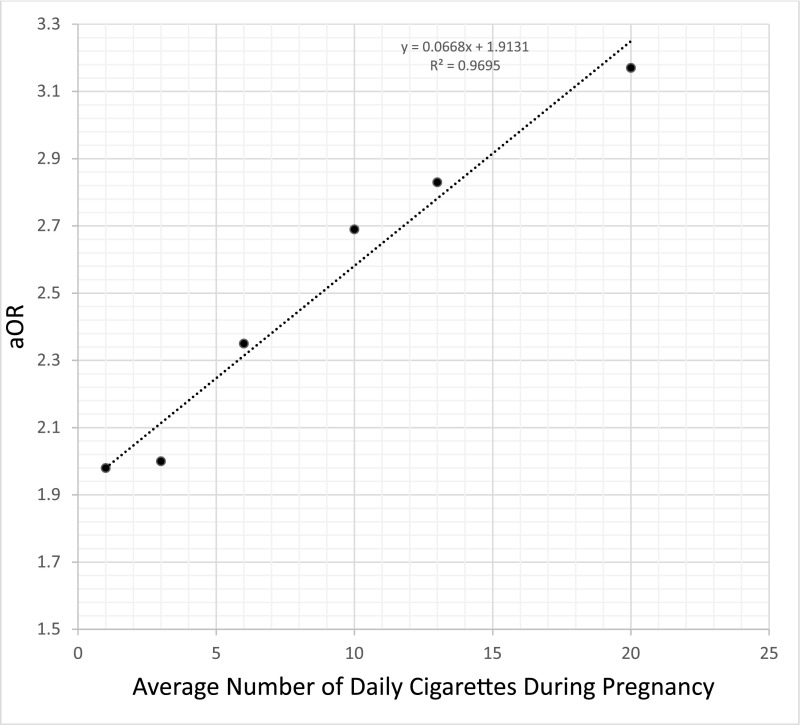
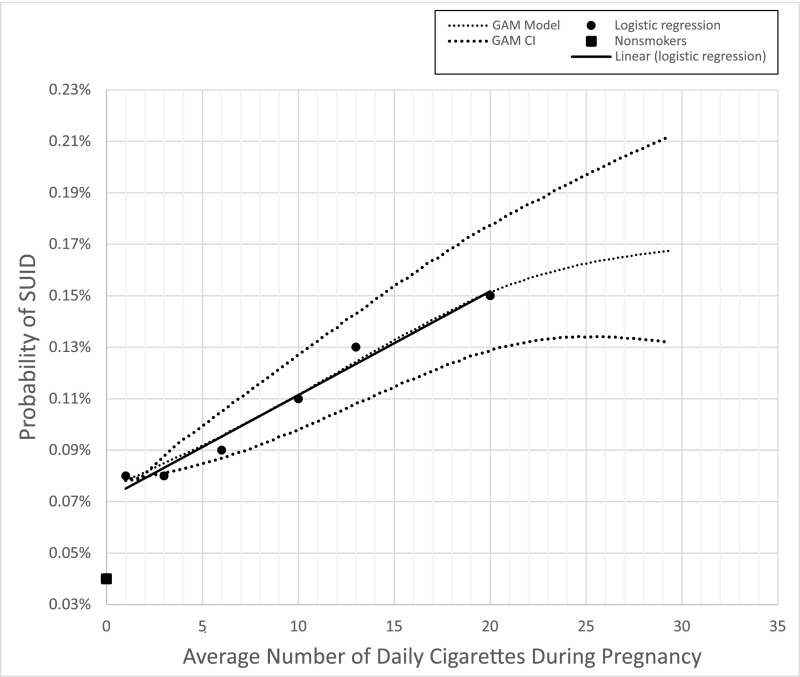
There was a positive correlation between average number of daily cigarettes during pregnancy and the risk of SUID (Table 1). This correlation was similar for each trimester when modeled independently (Fig 1), but the average number of cigarettes in the 3 trimesters together provided greater predictive power (Supplemental Fig 4). There was a twofold-increased SUID risk between no smoking and smoking 1 cigarette daily throughout pregnancy (aOR = 1.98; 95% CI, 1.73–2.28). For 1 to 20 cigarettes per day, the probability of SUID increased linearly, with each additional cigarette smoked per day increasing the odds by 0.07 (aOR = 0.07 × number of daily cigarettes + 1.91) (Supplemental Fig 4). In the GAM, we observed the same twofold increase for smoker versus nonsmoker (aOR = 1.96; 95% CI 1.72–2.23), a linear relationship in SUID risk for 1 to 20 cigarettes, and a flattening of the curve after 20+ daily cigarettes with much wider CIs because of fewer cases (Fig 2). For the population mode, between 1 and 20 cigarettes, the line fitted on the results of both logistic regression and GAM were the same (aOR = 0.07 × number of cigarettes +1.91).
TABLE 1.
aORs of SUID for 0–20 Cigarettes
| No. Daily Cigarettes | Odds Ratio | 95% CI | |
|---|---|---|---|
| Lower Bound | Upper Bound | ||
| 0 | 1 | — | — |
| 1 | 1.98 | 1.73 | 2.28 |
| 3 | 2 | 1.8 | 2.22 |
| 6 | 2.35 | 2.11 | 2.56 |
| 10 | 2.69 | 2.5 | 2.98 |
| 13 | 2.83 | 2.43 | 3.24 |
| 20 | 3.17 | 2.87 | 3.51 |
aORs of SUID by average number of cigarettes (0–20) smoked daily during pregnancy. —, not applicable.
FIGURE 1.
aORs of SUID given the average number of cigarettes (between 1 and 20) smoked daily by the mother per trimester.
FIGURE 2.
Logistic regression and GAMs. Two different computational models, logistic regression and the GAM, plot the rate of SUID given the average daily number of reported cigarettes smoked by the mother across all 3 trimesters.
Of mothers who smoked during pregnancy, 55% did not reduce smoking during pregnancy, 20% quit smoking by the beginning of the third trimester, and 24% reduced their smoking. Those who quit or reduced smoking by the third trimester decreased the amount of smoking during pregnancy by an average of 58% and 33%, respectively. Compared with continued smokers, SUID risk in the reduced group was slightly decreased (aOR = 0.88; 95% CI 0.79–0.98), whereas those who quit exhibited the largest reduction in risk (aOR = 0.77; 95% CI 0.67–0.87).
Compared with mothers who did not smoke in the 3 months before or during pregnancy, SUID risk progressively increased for those who smoked before pregnancy and quit before pregnancy (aOR = 1.47; 95% CI 1.16–1.87), those who did not smoke before but smoked during pregnancy (aOR = 2.22; 95% CI 1.15–4.29), and those who smoked before and during pregnancy (aOR = 2.52; 95% CI 2.25–2.83). For mothers who smoked prepregnancy only, the number of cigarettes smoked prepregnancy did not have a significant association with a change in SUID risk.
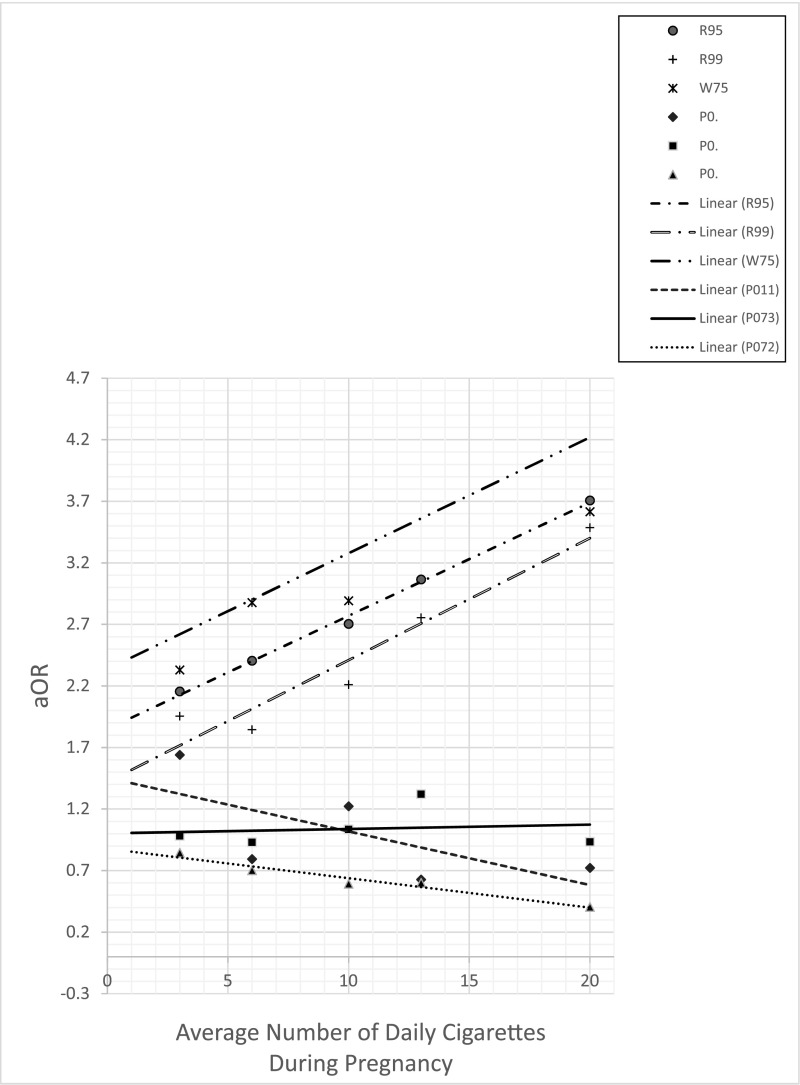
The logistic regression models plotting the correlation between maternal smoking and specific ICD-10 codes for SUID revealed a statistically significant dose-effect relationship between maternal smoking and odds of R95, R99, and W75 (Fig 3). Conversely, non-SUID causes of death, including P07.2 (extreme immaturity of the newborn), P07.3 (prematurity), and P01.1 (newborn affected by premature rupture of membranes) did not exhibit this positive dose-response relationship (Fig 3).
FIGURE 3.
aORs of specific causes of SUID and non-SUID infant death. Comparison is between aORs of specific causes of infant death, including R95 (SIDS), R99 (ill-defined or unknown cause of mortality), and W75 (accidental suffocation or strangulation in bed), and other non-SUID causes of infant death, including P07.2 (extreme immaturity of the newborn), P07.3 (prematurity), and P01.1 (newborn affected by premature rupture of membranes).
Assuming causality, an estimated ∼800 infants per year, or 22% of all SUID cases in the United States, were attributed to maternal smoking during pregnancy.
Discussion
Public health campaigns launched in the 1990s educating parents about the importance of infant sleep position and environment led to a ∼50% decrease in US SIDS rates.31 As prevalence of prone sleeping has declined, the relative contribution of prenatal maternal smoking to the risk of sudden infant death has increased.2 We found that any smoking during pregnancy was associated with a doubling in SUID risk. Additionally, if mothers quit or reduced smoking during pregnancy, the relative risk of SUID decreased compared with those who continued smoking. Although the average number of cigarettes across the 3 trimesters held greater predictive power, the increase in SUID risk due to prenatal maternal smoking was seen even when each trimester was modeled independently, suggesting that smoking during any trimester is associated with increased SUID risk. However, this phenomenon is at least partly explained by a high correlation between smoking in the first trimester and smoking in subsequent trimesters. In each model, there was a twofold risk for smokers who smoked at least 1 cigarette.
There was a linear correlation between average number of daily cigarettes smoked and increased risk for SUID. Similar dose-dependent trends have been described previously,32,33 but not with such resolution or sample size. In the GAM, the curve began to plateau after >20 cigarettes per day, suggesting that smoking cessation efforts may have greater impact on decreasing SUID rates when directed toward those who smoke fewer than 1 pack per day versus the more traditionally targeted heavy (>20 cigarettes per day) smokers.
Compared with the pregnant smokers who did not reduce their smoking during pregnancy (more than half), those who reduced the number of cigarettes smoked by the third trimester demonstrated a modest (12%) decrease in the risk of SUID, and quitting by the third trimester was associated with a greater reduction in risk (by 23%). However, there may be some selection bias because the group who reduced smoking started at a higher average number of cigarettes in the first trimester, whereas those who successfully quit smoked fewer cigarettes in the first trimester.
The largest predictor of SUID risk with maternal prenatal smoking was the average number of cigarettes smoked daily over the 3 trimesters. Thus, a woman who smoked 20 cigarettes per day in the first trimester and reduced to 10 cigarettes per day in subsequent trimesters had a similarly reduced SUID risk as a woman who averaged 13 cigarettes per day in each trimester. Public health promotion should specifically encourage women to quit before pregnancy. Furthermore, pregnant smokers seeking prenatal care in the first trimester should be strongly advised that the greatest benefit for reducing SUID risk unequivocally results from quitting but also that any reduction in the number of cigarettes smoked is associated with a small decrease in risk.
Although smoking has decreased overall in the United States in recent years, 11.6% of mothers reported smoking in the 3 months before pregnancy in 2011. Of these, only one-quarter stopped smoking for the duration of the pregnancy. The adjusted odds for SUID were slightly but significantly increased (aOR = 1.47; 95% CI 1.16–1.87) in cases wherein the mother smoked prepregnancy but quit during the pregnancy compared with those who never smoked. Part of this increase could be due to environmental tobacco exposure because it is not uncommon for those who smoke to have a partner who also smokes34; it is also likely that a proportion of women who smoked prepregnancy and quit during pregnancy restarted in the postpartum period.35 This group may have also included women who stopped smoking as soon as they knew they were pregnant and thus reported that they were nonsmokers in the first trimester, but the fetus had been exposed to maternal smoking during the period before pregnancy was diagnosed. Interestingly, the increased odds ratio was similar regardless of how many cigarettes were smoked during the 3 months prepregnancy. Although the study adjusted for many potential confounders, residual confounding, especially with socioeconomic factors, might explain this finding. There may also be other exposures (eg, women who drink alcohol during pregnancy, another potent risk factor for SUID, are more likely to smoke at moderate, high, and very high continuous levels as compared with women classified as nondrinkers and quitters).36
The relationship between smoking and rates for R95, R99, and W75 diagnoses individually revealed similar linear trends. These findings support the idea that, despite differing labels on the death certificate, there may be commonalities in intrinsic and/or extrinsic factors, and these deaths should consistently be considered together as SUID. Interestingly, specific non-SUID causes of death, including P07.2, P07.3, and P01.1, did not reveal dose-effect relationships with smoking. This was unexpected because smoking increases the risk of preterm birth, which is associated with higher mortality and morbidity.37
Researchers in various countries, including New Zealand (33%),2 Chile (33%),38 Denmark (30%–40%),4 and the United States (23%–34%),39 have attempted to estimate the percentage of SIDS and/or SUID attributable to prenatal smoking. In this study, we employed sophisticated statistical analyses in combination with high population numbers to allow for greater granularity in estimating population-attributable risk for prenatal smoke exposure. The relationship between smoking and SUID meet the criteria for a causal association,40 including (1) strength (effect size; the magnitude of the risk is strong), (2) a dose-effect relationship (a linear relationship between number of cigarettes and SUID risk), (3) temporal relationship (the risk factor [smoking] precedes the event [death]), (4) consistency of findings (smoking is identified as a risk factor in many studies), (5) biological plausibility,2,3 and (6) the reduction in risk with smoking reduction and cessation. If causality is assumed in our model, we estimate that ∼22% of all US SUID cases are directly attributable to smoking (ie, if every mother did not smoke during pregnancy, there would have been an estimated 800 fewer SUIDs in the United States in 2011 alone). This suggests that a significant reduction in SIDS incidence might occur if the prevalence of maternal smoking was reduced.
This study is limited by the likely conservative smoking estimates because our data set does not include environmental smoke exposure during pregnancy or in the postpartum period, including paternal smoking, which has an independent influence on SIDS risk.3,41 In addition, smoking rates are self-reported. Because it is widely known that smoking is an unhealthy behavior, it is likely that some women underestimated or denied their true smoking habits. Indeed, in studies documenting serum cotinine levels, maternal self-reported smoking status during pregnancy underestimated smoking prevalence by >20%.42,43 Finally, only 60% of births had data about the number of cigarettes smoked. However, the missing data were not related to maternal characteristics but instead to the adoption of the 2003 revision of the US Standard Certificate of Live Birth, and therefore had minimal effect on the estimates.
Conclusions
Educational efforts to decrease SUID risk should strongly encourage nonsmoking practices before pregnancy and smoking cessation during pregnancy. Those who are unable to quit entirely should be advised to reduce the amount smoked. We estimate that US SUID rates could be reduced by 22% if no women smoked during pregnancy.
Acknowledgments
Dr Mitchell was supported in part by the Gavin and Ann Kellaway Medical Research Fellowship. We thank Kelty Allen, Avleen Bijral, Urszula Chajewska, Ricky Johnston, Sushama Murthy, and John Thompson for statistical guidance and useful discussion, and John Kahan and Daniel Rubens for inspiring this collaboration.
Glossary
- aOR
adjusted odds ratio
- CDC
Centers for Disease Control and Prevention
- CI
confidence interval
- GAM
generalized additive model
- ICD-10
International Classification of Diseases, 10th Revision
- SIDS
sudden infant death syndrome
- SUID
sudden unexpected infant death
Footnotes
Dr Anderson provided input on the data analysis, drafted the initial manuscript, and reviewed and revised the manuscript; Mr Lavista Ferres conceptualized and designed the study, curated the data, conducted formal data analysis, and drafted the initial manuscript; Dr Ren assisted in the data analysis and critically reviewed and revised the manuscript; Drs Moon, Goldstein, and Ramirez assisted in the interpretation of the data and critically reviewed the manuscript for important intellectual content; Dr Mitchell assisted in the data analysis and the drafting of the initial manuscript and critically reviewed and revised the manuscript for important intellectual content; and all authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
FUNDING: Supported by the National Institutes of Health (grants P01HL0906654 and R01HL126523 awarded to J.M.R.), Microsoft, and the Aaron Matthew Sudden Infant Death Syndrome Research Guild. Funded by the National Institutes of Health (NIH).
POTENTIAL CONFLICT OF INTEREST: Dr Moon has served as a paid medical expert in a case of unexpected sudden infant death; the other authors have indicated they have no potential conflicts of interest to disclose.
References
- 1.Matthews TJ, MacDorman MF, Thoma ME. Infant mortality statistics from the 2013 period linked birth/infant death data set. Natl Vital Stat Rep. 2015;64(9):1–30 [PubMed] [Google Scholar]
- 2.Mitchell EA, Milerad J. Smoking and the sudden infant death syndrome. Rev Environ Health. 2006;21(2):81–103 [DOI] [PubMed] [Google Scholar]
- 3.Mitchell EA, Ford RP, Stewart AW, et al. Smoking and the sudden infant death syndrome. Pediatrics. 1993;91(5):893–896 [PubMed] [Google Scholar]
- 4.Wisborg K, Kesmodel U, Henriksen TB, Olsen SF, Secher NJ. A prospective study of smoking during pregnancy and SIDS. Arch Dis Child. 2000;83(3):203–206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haglund B, Cnattingius S. Cigarette smoking as a risk factor for sudden infant death syndrome: a population-based study. Am J Public Health. 1990;80(1):29–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Klonoff-Cohen HS, Edelstein SL, Lefkowitz ES, et al. The effect of passive smoking and tobacco exposure through breast milk on sudden infant death syndrome. JAMA. 1995;273(10):795–798 [DOI] [PubMed] [Google Scholar]
- 7.Blair PS, Fleming PJ, Bensley D, et al. ; Confidential Enquiry into Stillbirths and Deaths Regional Coordinators and Researchers . Smoking and the sudden infant death syndrome: results from 1993-5 case-control study for confidential inquiry into stillbirths and deaths in infancy. BMJ. 1996;313(7051):195–198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anderson HR, Cook DG. Passive smoking and sudden infant death syndrome: review of the epidemiological evidence. Thorax. 1997;52(11):1003–1009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kinney HC, Thach BT. The sudden infant death syndrome. N Engl J Med. 2009;361(8):795–805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duncan JR, Paterson DS, Hoffman JM, et al. Brainstem serotonergic deficiency in sudden infant death syndrome. JAMA. 2010;303(5):430–437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Machaalani R, Say M, Waters KA. Effects of cigarette smoke exposure on nicotinic acetylcholine receptor subunits α7 and β2 in the sudden infant death syndrome (SIDS) brainstem. Toxicol Appl Pharmacol. 2011;257(3):396–404 [DOI] [PubMed] [Google Scholar]
- 12.Eugenín J, Otárola M, Bravo E, et al. Prenatal to early postnatal nicotine exposure impairs central chemoreception and modifies breathing pattern in mouse neonates: a probable link to sudden infant death syndrome. J Neurosci. 2008;28(51):13907–13917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Duncan JR, Garland M, Myers MM, et al. Prenatal nicotine-exposure alters fetal autonomic activity and medullary neurotransmitter receptors: implications for sudden infant death syndrome. J Appl Physiol (1985). 2009;107(5):1579–1590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cerpa VJ, Aylwin ML, Beltrán-Castillo S, et al. The alteration of neonatal raphe neurons by prenatal-perinatal nicotine. Meaning for sudden infant death syndrome. Am J Respir Cell Mol Biol. 2015;53(4):489–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee SY, Sirieix CM, Nattie E, Li A. Pre- and early postnatal nicotine exposure exacerbates autoresuscitation failure in serotonin-deficient rat neonates. J Physiol. 2018;596(23):5977–5991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cummings KJ, Hewitt JC, Li A, Daubenspeck JA, Nattie EE. Postnatal loss of brainstem serotonin neurones compromises the ability of neonatal rats to survive episodic severe hypoxia. J Physiol. 2011;589(pt 21):5247–5256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cummings KJ, Commons KG, Hewitt JC, et al. Failed heart rate recovery at a critical age in 5-HT-deficient mice exposed to episodic anoxia: implications for SIDS. J Appl Physiol (1985). 2011;111(3):825–833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kinney HC, Randall LL, Sleeper LA, et al. Serotonergic brainstem abnormalities in Northern Plains Indians with the sudden infant death syndrome. J Neuropathol Exp Neurol. 2003;62(11):1178–1191 [DOI] [PubMed] [Google Scholar]
- 19.Duncan JR, Randall LL, Belliveau RA, et al. The effect of maternal smoking and drinking during pregnancy upon (3)H-nicotine receptor brainstem binding in infants dying of the sudden infant death syndrome: initial observations in a high risk population. Brain Pathol. 2008;18(1):21–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cimino M, Marini P, Colombo S, et al. Expression of neuronal acetylcholine nicotinic receptor alpha 4 and beta 2 subunits during postnatal development of the rat brain. J Neural Transm (Vienna). 1995;100(2):77–92 [DOI] [PubMed] [Google Scholar]
- 21.Ribeiro EB, Bettiker RL, Bogdanov M, Wurtman RJ. Effects of systemic nicotine on serotonin release in rat brain. Brain Res. 1993;621(2):311–318 [DOI] [PubMed] [Google Scholar]
- 22.Kinney HC, Belliveau RA, Trachtenberg FL, Rava LA, Paterson DS. The development of the medullary serotonergic system in early human life. Auton Neurosci. 2007;132(1–2):81–102 [DOI] [PubMed] [Google Scholar]
- 23.Friedmann I, Dahdouh EM, Kugler P, Mimran G, Balayla J. Maternal and obstetrical predictors of sudden infant death syndrome (SIDS). J Matern Fetal Neonatal Med. 2017;30(19):2315–2323 [DOI] [PubMed] [Google Scholar]
- 24.MacDorman MF, Cnattingius S, Hoffman HJ, Kramer MS, Haglund B. Sudden infant death syndrome and smoking in the United States and Sweden. Am J Epidemiol. 1997;146(3):249–257 [DOI] [PubMed] [Google Scholar]
- 25.Mitchell EA, Tuohy PG, Brunt JM, et al. Risk factors for sudden infant death syndrome following the prevention campaign in New Zealand: a prospective study. Pediatrics. 1997;100(5):835–840 [DOI] [PubMed] [Google Scholar]
- 26.Shapiro-Mendoza CK, Tomashek KM, Anderson RN, Wingo J. Recent national trends in sudden, unexpected infant deaths: more evidence supporting a change in classification or reporting. Am J Epidemiol. 2006;163(8):762–769 [DOI] [PubMed] [Google Scholar]
- 27.Goldstein RD, Trachtenberg FL, Sens MA, Harty BJ, Kinney HC. Overall postneonatal mortality and rates of SIDS. Pediatrics. 2016;137(1) [DOI] [PubMed] [Google Scholar]
- 28.Alm B, Milerad J, Wennergren G, et al. A case-control study of smoking and sudden infant death syndrome in the Scandinavian countries, 1992 to 1995. The Nordic Epidemiological SIDS Study. Arch Dis Child. 1998;78(4):329–334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Centers for Disease Control and Prevention, National Center for Health Statistics Vital statistics online data portal: cohort linked birth-infant death data files. Available at: www.cdc.gov/nchs/data_access/Vitalstatsonline.htm. Accessed August 2018
- 30.Benichou J. A review of adjusted estimators of attributable risk. Stat Methods Med Res. 2001;10(3):195–216 [DOI] [PubMed] [Google Scholar]
- 31.Hauck FR, Tanabe KO. Beyond "back to sleep": ways to further reduce the risk of sudden infant death syndrome. Pediatr Ann. 2017;46(8):e284–e290 [DOI] [PubMed] [Google Scholar]
- 32.Golding J. Sudden infant death syndrome and parental smoking--a literature review. Paediatr Perinat Epidemiol. 1997;11(1):67–77 [DOI] [PubMed] [Google Scholar]
- 33.Zhang K, Wang X. Maternal smoking and increased risk of sudden infant death syndrome: a meta-analysis. Leg Med (Tokyo). 2013;15(3):115–121 [DOI] [PubMed] [Google Scholar]
- 34.Homish GG, Leonard KE. Spousal influence on smoking behaviors in a US community sample of newly married couples. Soc Sci Med. 2005;61(12):2557–2567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jones M, Lewis S, Parrott S, Wormall S, Coleman T. Re-starting smoking in the postpartum period after receiving a smoking cessation intervention: a systematic review. Addiction. 2016;111(6):981–990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dukes K, Tripp T, Willinger M, et al. Drinking and smoking patterns during pregnancy: Development of group-based trajectories in the Safe Passage Study. Alcohol. 2017;62:49–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Abel EL. Smoking during pregnancy: a review of effects on growth and development of offspring. Hum Biol. 1980;52(4):593–625 [PubMed] [Google Scholar]
- 38.Cerda J, Bambs C, Vera C. Infant morbidity and mortality attributable to prenatal smoking in Chile. Rev Panam Salud Publica. 2017;41:e106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dietz PM, England LJ, Shapiro-Mendoza CK, Tong VT, Farr SL, Callaghan WM. Infant morbidity and mortality attributable to prenatal smoking in the U.S. Am J Prev Med. 2010;39(1):45–52 [DOI] [PubMed] [Google Scholar]
- 40.Hill AB. The environment and disease: association or causation? Proc R Soc Med. 1965;58:295–300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mitchell EA, Taylor BJ, Ford RP, et al. Dummies and the sudden infant death syndrome. Arch Dis Child. 1993;68(4):501–504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dietz PM, Homa D, England LJ, et al. Estimates of nondisclosure of cigarette smoking among pregnant and nonpregnant women of reproductive age in the United States. Am J Epidemiol. 2011;173(3):355–359 [DOI] [PubMed] [Google Scholar]
- 43.Tong VT, Dietz PM, England LJ. Reliance on self-reporting underestimates pregnancy smoking rates in Scotland, with more than 2400 pregnant smokers estimated to be missed each year. Evid Based Med. 2010;15(3):94–95 [DOI] [PubMed] [Google Scholar]