Abstract
Background
Caregivers of patients with neurodegenerative diseases are at heightened risk for serious health problems, but health differences between individual caregivers abound.
Aims
To determine whether atrophy in patient brains could be used to identify caregivers at heightened risk for health problems and which patient variables mediate this relationship.
Methods
In 162 patient-caregiver dyads, we assessed patient atrophy using structural MRI, caregiver health, and patient behavior and cognitive symptoms.
Results
Patient atrophy in the right insula and medial frontal gyrus was associated with worse caregiver health; this relationship was partially mediated by patient neuropsychiatric symptoms, and assessing atrophy in these regions improved predictions of poor caregiver health above and beyond patient behavioral symptoms.
Conclusions
This study shows the value of patients’ brain data in identifying caregivers at risk for becoming sick themselves.
Keywords: Neurodegenerative disease, Caregiver health, Patient behavioral symptoms, Patient cognitive symptoms, Voxel-based morphometry
Introduction
Neurodegenerative diseases are progressive illnesses that have devastating effects on cognitive, emotional, and motor functioning. Because neurodegenerative diseases are more prevalent in late life [1], demographic shifts towards an older population mean that caregiving is becoming an increasingly common concern for many families. Caring for a loved one with a neurodegenerative disease can be a highly meaningful part of family life; however, it can have deleterious impacts on caregivers’ health. Caregivers of individuals with dementia not only have dramatically higher rates of mental health problems (e.g., depression, anxiety) than non-caregiving adults [2], they also experience greater physical health problems and mortality risks than caregivers of individuals with other illnesses (e.g., cancer, stroke, and chronic disability [3]). Importantly, there is considerable variability in these adverse health effects between individual caregivers [4]. Thus, there is a pressing need to find reliable markers that predict which caregivers are at maximal risk for illness.
An emerging consensus is that behavioral symptoms (e.g., apathy and aggression) in people with dementia play a major role in accounting for adverse caregiver outcomes, even more so than cognitive or functional symptoms [5, 6]. Different neurodegenerative diseases target different large-scale brain networks [7], and thus afflicted individuals can present with quite different symptom profiles [8]. For example, Alzheimer’s disease (AD) typically targets the default mode network, which is anchored in the posterior parietal cortex [7, 9]. Individuals with AD primarily exhibit cognitive symptoms, such as problems with memory and spatial cognition. In contrast, behavioral-variant frontotemporal dementia (bvFTD) targets the salience network, which is anchored in the right insula [7, 9]. Individuals with bvFTD primarily exhibit behavioral symptoms in the realm of socioemotional functioning, such as apathy and loss of empathy. Consistent with the importance of behavioral symptoms, results indicate that caregivers of individuals with bvFTD may fare worse than caregivers of individuals with AD [10–12].
Attempts to identify behavioral symptoms in patients with neurodegenerative disease that are linked with adverse caregiver health outcomes have been hampered by lack of agreement as to which behaviors to measure and how best to measure them [6]. Thus, some researchers have focused on psychiatric behaviors (e.g., hallucinations and delusions) measured via clinician-rated caregiver interviews (e.g., Neuropsychiatric Inventory [NPI] [13]), while others have focused on emotional behaviors (e.g., reduced visual attention to disgusting films, extraneous negative emotions in response to films, and reduced empathic accuracy [14–16]) measured using well-controlled laboratory procedures. Examining the “patient behavior to caregiver health” pathway, whether by caregiver interviews or laboratory assessments, has been fruitful in understanding individual differences in adverse caregiver health outcomes [14, 17]. However, both approaches have limitations. For example, caregiver reports can be biased [18] and laboratory assessments can be time-consuming and costly.
There can be little argument that neurodegeneration plays the primary causal role in producing behavioral symptoms in patients [8]. Moreover, measurable signs of neurodegeneration may appear well before observable changes in behavior occur [19]. Structural neuroimaging is increasingly being used as a routine part of patient assessment, thus providing a basis for characterizing patterns of neurodegeneration in individual patients. With such data being so readily available, there is a compelling case for evaluating the “patient brain to caregiver health” pathway for understanding individual differences in caregiver vulnerability to declines in health. Research evaluating this pathway have been quite rare (e.g., studies linking patient neurodegeneration or traumatic brain injury with caregiver burden [20, 21]). To our knowledge there have been no prior studies comparing the “patient brain to caregiver health” pathway with the “patient behavior to caregiver health” pathway. Comparing these pathways will inform researchers and clinicians about which patient measures may be more suitable to predict health outcomes in caregivers. If certain patient measures predict caregiver health outcomes, patient data may be further utilized to identify caregivers who may require additional support and assistance.
The present study examined whether patient neurodegeneration could be used to predict adverse health outcomes in their caregivers. We also included healthy controls (HCs) and their partners to provide a neurologically healthy reference group for assessing neurodegeneration in relationship to caregiver health. Caregiver and partner psychopathology and global health (i.e., mental and physical health) were assessed with two commonly used self-report symptom inventories. Neurodegeneration was quantified using whole-brain voxel-based morphometry (VBM) because we wanted to detect brain regions associated with both caregiver health measures. Thus, we did not employ region-of-interest analyses in order not to limit the scope of our examination. Additionally, for group level analyses, this whole-brain approach has shown greater accuracy than region-of-interest analyses in neurodegenerative disease [22]. Our primary hypothesis concerning the “patient brain to caregiver health” pathway was that neurodegeneration in areas thought to support socioemotional behavior (e.g., the insula and anterior cingulate cortex [23]) would be associated with worse caregiver health. Consistent with the literature [6, 14], we hypothesized that the relationship between neurodegeneration in patients and health in caregivers would be mediated by patient behavioral symptoms, but not by patient cognitive functioning. Finally, to begin to evaluate the relative value of the “patient brain to caregiver health” and “patient behavior to caregiver health” pathways, we conducted a set of exploratory analyses comparing the predictive power of the two pathways.
Subjects and Methods
Participants
The sample included 139 patients with different neurodegenerative diseases and 23 HCs who were evaluated through the Memory and Aging Center at the University of California, San Francisco (UCSF). UCSF research participants who received diagnostic screening and who had reliable informants were identified, and their caregivers or study partners were given the option to participate in an additional study at the University of California, Berkeley (UCB). Caregivers of patients (mostly spouses; see below) and partners of HCs were evaluated at the UCB. All participants or their caregivers, when appropriate (e.g., when patients could not consent for themselves or when caregivers were providing consent for their own participation), provided consent at each site for projects approved by their respective institutional review boards.
The sample included patients with AD (n = 35), three subtypes of FTD (bvFTD [n = 32], semantic-variant primary progressive aphasia [n = 23], and nonfluent-variant primary progressive aphasia [n = 15]), corticobasal syndrome (CBS; n = 17), and progressive supranuclear palsy (PSP; n = 17), as well as neurologically healthy controls (HCs; n = 23). CBS and PSP are predominantly characterized by changes to motor functioning [24, 25]; these groups were included to increase the neuroanatomical and symptom heterogeneity of the patient sample. Patients’ diagnoses were provided by neurologists based on consensus research criteria [24–28].
Experimental Design
Patients were referred to UCSF through self-referral, clinician referral, or referral through Alzheimer’s Disease Research Centers. HCs were volunteer participants who were recruited from the community. The patients underwent detailed clinical interviews, neurological examination, functional assessment, neuropsychological evaluation, and structural magnetic resonance imaging (MRI) at UCSF. The HCs underwent identical neurological and cognitive assessments and were screened to ensure that they had no history of neurological, psychiatric, or cognitive disorders; only HCs who passed this screening were eligible study participants.
At UCB, the caregivers of patients and partners of HCs completed questionnaires about their demographics and their mental and physical health. In all, 83% of the caregivers and partners were spouses of the patients and HCs (see Table 1 for more details).
Table 1.
Means and standard deviations (in parentheses) for participant characteristics of caregivers and patients with neurodegenerative disease (or partners and controls) by diagnosis group, unless otherwise noted
| Characteristic | AD | bvFTD | CBS | HCs | nfvPPA | PSP | svPPA |
|---|---|---|---|---|---|---|---|
| Subjects, n | 35 | 32 | 17 | 23 | 15 | 17 | 23 |
| Caregiver/partner sex, % female | 71 | 59 | 53 | 48 | 53 | 53 | 48 |
| Caregiver/partner age, years | 59.19 (8.35) | 58.56 (14.39) | 60.65 (11.09) | 64.60 (10.66) | 68.25 (15.73) | 62.94 (8.00) | 66.38 (14.80) |
| Caregiver/partner relationship, % spouses | 86 | 81 | 71 | 83 | 93 | 88 | 83 |
| Caregiver/partner ethnicity, n | |||||||
| White/Caucasian/European American | 31 | 24 | 12 | 22 | 14 | 16 | 23 |
| Latino/Chicano/Hispanic | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| Black/Black American/Afro-Caribbean | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Asian/Asian American/South Asian | 0 | 3 | 2 | 1 | 1 | 1 | 1 |
| Multi-racial/other | 4 | 4 | 1 | 0 | 0 | 0 | 0 |
| Patient Neuropsychiatric Inventory (NPI) score | 21.03 (24.22) | 41.45 (18.20) | 22.40 (23.31) | −(−) | 15.14(13.20) | 33.53 (19.30) | 36.13 (19.12) |
| Patient Clinical Dementia Rating (CDR-Box) score | 3.99 (1.88) | 6.84 (3.39) | 3.82 (2.49) | 0.00 (0.00) | 2.20 (2.33) | 5.82 (2.70) | 3.95 (2.65) |
| Patient Mini-Mental State Examination (MMSE) score | 22.23 (4.81) | 23.31 (5.51) | 22.18 (6.49) | 29.60 (0.58) | 23.40 (7.71) | 26.70 (3.12) | 23.36 (5.48) |
| Caregiver/partner Symptom Checklist 90 (SCL-90) score | 0.33 (0.23) | 0.53 (0.36) | 0.59 (0.74) | 0.24 (0.20) | 0.56 (0.44) | 0.28 (0.18) | 0.30 (0.33) |
| Caregiver/partner Short Form Health Survey (SF-36) score | 78.97 (11.82) | 75.51 (12.00) | 73.88 (19.48) | 85.20 (8.41) | 72.10 (18.16) | 77.39 (19.56) | 80.81 (11.92) |
NPI, patient neuropsychiatric (e.g., behavioral) symptoms; CDR-Box, dementia severity; MMSE, cognitive functioning; SCL-90, severity of psychopathological symptoms; SF-36, global health outcomes; AD, Alzheimer’s disease; bvFTD, behavioral-variant frontotemporal dementia; CBS, corticobasal syndrome; HCs, healthy controls; nfvPPA, nonfluent-variant primary progressive aphasia; PSP, progressive supranuclear palsy; svPPA, semantic-variant primary progressive aphasia.
In our cross-sectional observational study, we conceptualized patient neurodegeneration as the independent variable, caregiver health as the dependent variable, and patient behavioral symptoms or cognitive functioning as potential mediators for the relationship between patient neurodegeneration and caregiver health.
In order to maximize statistical power, we included all dyads (n = 162) with available MRI and caregiver/partner health data. Given an α value of 0.005, power of 0.80, and medium effect size of F2 = 0.15 for a linear regression, we computed a suggested sample size of 163. We selected α = 0.005 to match the statistical threshold in our neuroimaging analyses. Thus, our sample size was adequate to detect a relationship between patient neurodegeneration and caregiver health.
Measures
Cognitive Functioning
Cognitive functioning was assessed in patients and HCs by a neuropsychologist using the Mini-Mental State Examination (MMSE [29]). MMSE scores range from 0 to 30, with lower scores indicating greater impairment in cognitive functioning.
Disease Severity
Disease severity was assessed in patients and HCs by a clinician using the Clinical Dementia Rating (CDR [30]). The CDR Total (scores range from 0 to 3) and Sum of Boxes (CDR-Box) scores were computed for each participant (scores range from 0 to 18). On both CDR measures, higher scores indicate greater functional impairment.
Behavioral Symptoms
Behavioral symptoms were assessed in patients and HCs using the NPI [13]. The NPI assesses psychopathology in neurodegenerative diseases to evaluate 12 common neuropsychiatric symptoms: (1) delusions, (2) hallucinations, (3) agitation, (4) dysphoria, (5) anxiety, (6) apathy, (7) irritability, (8) euphoria, (9) disinhibition, (10) aberrant motor behavior, (11) night-time behavior disturbances, and (12) appetite and eating abnormalities. Clinicians administered the NPI to the patients’ caregivers and rated the severity and frequency of each neuropsychiatric symptom. A total score for all neuropsychiatric symptoms was calculated as an index of patient behavioral symptoms. Of the 139 caregivers, 135 completed this measure. Three out of 23 partners of HCs completed this measure; because of this small number, partner data for HCs were not included.
Psychopathology
Psychopathological symptoms were assessed in caregivers and partners using the Symptom Checklist 90-Revised (SCL-90-R [31]). The SCL-90-R is a 90-item questionnaire that assesses 9 domains of psychopathology: (1) somatization, (2) obsessive-compulsive disorder, (3) interpersonal sensitivity, (4) depression, (5) anxiety, (6) hostility, (7) phobic anxiety, (8) paranoid ideation, and (9) psychoticism in the past week. For each item, the caregivers rated themselves on a 5-point scale from 0 (none) to 4 (extreme). For the present study, we used the average score for all 90 items as a measure of psychopathology; higher scores indicate greater severity of psychopathological symptoms. The caregivers and partners from all 162 dyads completed this measure.
Global Health
Global health was assessed in caregivers and partners using the Medical Outcomes Study 36-Item Short Form Health Survey (SF-36 [32]). The SF-36 evaluates 8 areas of mental and physical health in the past month: (1) physical functioning, (2) role limitations due to physical health problems, (3) role limitations due to emotional problems, (4) energy and fatigue, (5) emotional well-being, (6) social functioning, (7) pain, and (8) general health. Subscale scores were calculated by summing their items, and an SF-36 composite score was computed by averaging the scores for all 8 subscales; higher scores indicate greater global health. The caregivers and partners from all 162 dyads completed this measure.
Neuroimaging
The patients and HCs underwent 1.5-T, 3-T, or 4-T research-quality structural MRI at UCSF. Structural neuroimaging analyses utilizing images collected across different modes of hardware have been shown to have robust effects [33] and are thus unlikely to cause artifacts with appropriate covariates. MR images of HCs were acquired within 12 months of their spouses having completed the questionnaires, and MR images of patients were included within 3 months of the caregivers having completed the questionnaires. The MR images were visually inspected for movement artifacts and poor scan quality. In total, 162 MR images were included in the neuroimaging analyses (139 of patients and 23 of HCs). See the Appendix for acquisition and preprocessing details.
Analytic Plan
To examine the relationship between neurodegeneration in patients and health in caregivers (n = 162), we conducted two separate whole-brain VBM analyses: one predicting caregiver/partner psychopathology and the other predicting caregiver/partner global health. For each analysis, we included caregiver/partner age, caregiver/partner sex (0 = male, 1 = female), patient diagnosis (HC = 0, patient = 1), patient disease severity, patient cognitive functioning, patient total intracranial volume, and two dummy-coded variables for MRI scanner field strength as covariates. The images were overlaid with MRIcron (http://people.cas.sc.edu/rorden/mricron/index.html) on an MNI (Montreal Neurological Institute) average brain based on the gray and white matter templates used for preprocessing. See the Appendix for more details.
Because we were linking distal variables (neurodegeneration in one person and health in another) and were utilizing a whole-brain approach with conservative covariates, results for VBM were considered significant at p < 0.005 (uncorrected) to allow for visualization of spatial overlap within brain areas related to worse caregiver health outcomes from both measures (SCL-90-R and SF-36). The convergence of the two maps, defined as voxels contained in both maps at this threshold, allowed us to compute a specific and single index of brain volume associated with worse caregiver health by extracting and combining gray matter volumes from these regions. We used this single index to reflect consistent and specific caregiver vulnerability from the patient brain in mediation analyses and comparison of predictive power analyses.
To examine what influences the relationship between patient neurodegeneration and caregiver health, we ran separate analyses to determine whether the relationships between neurodegeneration in patients and psychopathology and global health in caregivers were mediated by (a) behavioral symptoms or (b) cognitive functioning in patients. These four mediational analyses tested indirect effects using a bootstrap estimation approach with 5,000 samples and used the same covariates as the VBM analyses described above, with two exceptions: (1) the diagnosis covariate was not included, because HCs were not included, and (2) cognitive functioning was not included as a covariate when it was tested as a mediator. Given that a subset of our sample was used for these analyses (n = 135) with stringent covariates, our use of two different caregiver health outcomes, and the empirical and theoretical considerations that patient behavioral symptoms and cognitive functioning would have separate effects on caregiver health, we conducted four separate mediational analyses instead of two multiple mediation analyses.
To evaluate the relative predictive power of the “patient brain to caregiver health” pathway to the “patient behavior to caregiver health” pathway, we compared how well patient brain and patient behavior measures predicted caregiver health by calculating changes in explained variance (R2) in linear regression models. The first type of model included the covariates used in the VBM analyses described above (with the exception of diagnosis) and patient behavioral symptoms as predictors. In the second type of model, we entered the combined patient brain volumes from regions that were associated with worse caregiver health as an additional predictor.
Results
Neural Correlates of Poor Caregiver Health
Psychopathological Symptoms
Whole-brain VBM analysis revealed multiple regions in which smaller volume in patients and HCs was associated with greater severity of psychopathological symptoms in caregivers and partners (p < 0.005, uncorrected). These included several medial frontal, prefrontal, cingulate, insular, temporal, and parietal regions. Online supplementary Figure S1 (for all online suppl. material, see www.karger.com/doi/10.1159/000495345) displays the statistical maps. For a complete list of these regions, see online supplementary Table S1 for T-score and significance levels.
Global Health
Whole-brain VBM analysis revealed multiple regions in which smaller volume in patients and HCs was associated with worse global health in caregivers and partners (p < 0.005, uncorrected). These included multiple medial frontal, anterior temporal, insular, and ventrostriatal regions. Online supplementary Figure S2 displays the statistical maps. For a complete list of these regions, see online supplementary Table S2 for T-score and significance levels.
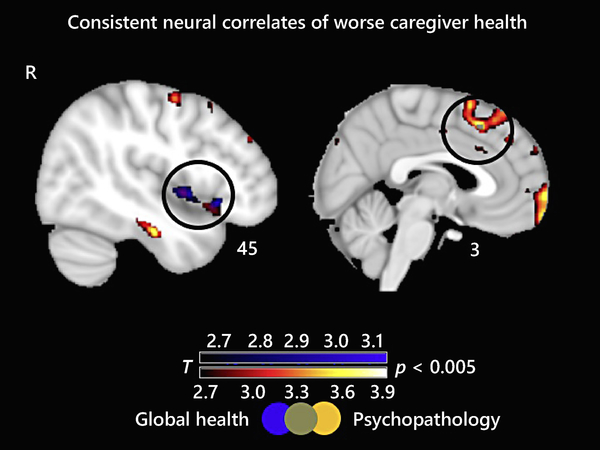
Consistent Neural Correlates of Worse Caregiver Health
The two T-score maps overlapped in two areas: the right ventral anterior insula (rvAI) and the superior medial frontal gyrus (smFG). Figure 1 displays areas of overlap. To ensure that these results were not driven by differences between patients and HCs or between individuals with FTD (where frontal and insular damage is common) and other patient groups, we conducted follow-up analyses.
Fig. 1.
Consistent neural correlates of worse caregiver health. T-score maps of brain areas for which a smaller volume in patient and healthy control brains is associated with greater severity of caregiver psychopathological symptoms and worse caregiver global health (p < 0.005, uncorrected). The T-score maps overlap in the right ventral anterior insula and in the superior medial frontal gyrus. The results for all analyses are overlaid on an MNI template brain.
Our first follow-up analysis (controlling for the same covariates) excluded HCs (n = 23). In this smaller sample (n = 123), the three T-score maps overlapped in the same two regions (rvAI and smFG; p < 0.005, uncorrected). Thus, including HCs did not change the relationship between patient neurodegeneration and caregiver health. See online supplementary Table S3 for T-score and significance levels and online supplementary Figure S3 for statistical maps.
Our second follow-up analysis (controlling for the same covariates) excluded individuals with FTD (n = 70; 32 with bvFTD, 15 with nonfluent-variant primary progressive aphasia, and 23 with semantic-variant primary progressive aphasia). In this smaller sample (n = 92; 35 with AD, 17 with CBS, 23 HCs, and 17 with PSP), the three T-score maps overlapped in the rvAI (p < 0.005, uncorrected). Thus, including individuals with FTD did not change the relationship between rvAI and caregiver health but was important for the relationship between smFG and caregiver health. See online supplementary Table S4 for T-score and significance levels and online supplementary Figure S4 for statistical maps.
Mediation by Patient Behavioral versus Cognitive Symptoms
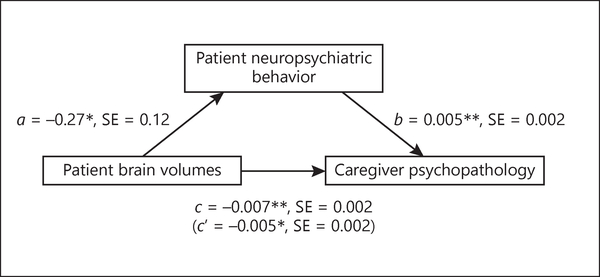
Patient behavioral symptoms partially mediated the relationship between patient brain volumes in the rvAI and smFG and caregiver psychopathology (standardized indirect effect = −0.05, 95% CI [−0.13, −0.003], accounting for 19% of the total effect)1 (Fig. 2). Patient behavioral symptoms did not mediate the relationship between patient brain volumes in the rvAI and smFG and caregiver global health (standardized indirect effect = 0.04, 95% CI [−0.005, 0.12]).2
Fig. 2.
Behavioral symptoms as a mediator. Regression coefficients and standard error (SE) for the relationship between patient brain volumes (right ventral anterior insula and superior medial frontal gyrus) and caregiver psychopathology as mediated by patient behavioral symptoms on the Neuropsychiatric Inventory. * p < 0.05, ** p < 0.01.
Patient cognitive functioning did not mediate the relationship between patient brain volumes in the rvAI and smFG and caregiver psychopathology (standardized indirect effect = 0.01, 95% CI [−0.002, 0.05]) or between patient brain volumes in the rvAI and smFG and caregiver global health (standardized indirect effect = −0.009, 95% CI [−0.05, 0.006]).
Evaluating Pathways: “Patient Brain to Caregiver Health” versus “Patient Behavior to Caregiver Health”
Compared to the model with the covariates and patient behavioral symptoms as predictors, the model that included patient brain volumes (from the rvAI and smFG) as a predictor significantly increased the explained variance of the model predicting caregiver psychopathology (F(1, 124) = 4.79, p = 0.03, R2change = 0.03). In this latter model, both patient brain volumes in the rvAI and smFG (t = −2.19, β = −0.19, p = 0.03) and patient neuropsychiatric behavior (t = 2.70, β = 0.26, p = 0.008) were significant predictors of caregiver psychopathology (Table 2).
Table 2.
Evaluating pathways: “patient brain to caregiver health” versus “patient behavior to caregiver health”
| R2 | R2 adj | R2 change | df | RSS | RSSchange | F-statistic | p value | |
|---|---|---|---|---|---|---|---|---|
| Dependent variable: caregiver psychopathology (SCL-90) | ||||||||
| Model 1: behavior only | 0.16 | 0.10 | – | 125 | 17.15 | – | 2.93 | 0.005 |
| Model 2: behavior and brain | 0.19 | 0.13 | – | 124 | 16.51 | – | 3.22 | 0.001 |
| Model Δ | – | – | 0.03 | – | – | −0.64 | 4.79 | 0.030 |
| Dependent variable: caregiver global health (SF-36) | ||||||||
| Model 1: behavior only | 0.11 | 0.05 | – | 125 | 13.72 | – | 1.95 | 0.059 |
| Model 2: behavior and brain | 0.16 | 0.10 | – | 124 | 13.38 | – | 2.64 | 0.008 |
| Model Δ | – | – | 0.05 | – | – | −0.34 | 7.40 | 0.007 |
When patient brain volumes are added as predictors to the linear regression models predicting poor caregiver health, the explained variance is increased compared to when just patient behavioral symptoms and covariates are entered as predictors. “Behavior only” model predictors: patient behavioral symptoms (NPI) + caregiver age + caregiver sex + patient diagnosis + patient dementia severity + patient cognitive functioning + patient total intracranial volume + scanner field strength. “Behavior and brain” model predictors: (same as above) + patient brain volumes extracted from the right ventral anterior insula and superior medial frontal gyrus. RSS, residual sum of squares.
Compared to the model with the covariates and patient behavioral symptoms as predictors, the model that included patient brain volumes (from the rvAI and smFG) as a predictor significantly increased the explained variance of the model predicting caregiver global health (F(1, 124) = 7.40, p = 0.007, R2change = 0.05). In this latter model, both patient brain volumes in the rvAI and smFG (t = 2.72, β = 0.24, p = 0.007) and patient neuropsychiatric behavior (t = −2.11, β = −0.21, p = 0.04) were significant predictors of caregiver global health (Table 2).
Discussion
The present study demonstrated the value of patient brain data in predicting caregiver health, supporting the viability of a “patient brain to caregiver health” pathway. Smaller volume in the rvAI and smFG in patients emerged as the most consistent predictor of greater severity of psychopathology and worse global health in caregivers. Importantly, the results remained stable with or without HCs or individuals with FTD. The mediational analyses revealed that patient behavioral symptoms (but not cognitive functioning) partially mediated the relationship between smaller volume in these two regions in patients and the severity of psychopathological symptoms in caregivers (but not global health). Lastly, we found that patient volumes in these two regions significantly increased the explained variance in caregiver health outcomes beyond that accounted for by patient behavioral symptoms.
Specific Neural Predictors of Caregiver Health
The rvAI and the smFG are particularly important for emotional and social functioning. The insula is a core hub in the salience network; it integrates multiple streams of information, including speech-language, viscero-autonomic, and socioemotional processes [34], and it has been consistently found to support bodily awareness, representation of emotional states, and detection of relevant emotional stimuli [35, 36]. The ventral anterior insula (relative to dorsal regions) is particularly associated with socioemotional and autonomic processes [37]. Individuals with ventral anterior insula damage may lose access to key bodily and social cues important for emotional responding in social interactions. In our study, the rvAI remained the single consistent predictor of worse health outcomes in caregivers and study partners when excluding the FTD group, underscoring the importance of neurodegeneration in this region across diagnoses. Patients who become emotionally “disconnected” from bodily information, with all of the attendant benefits that would normally accrue for processes such as decision-making and responsiveness to social cues [38], are likely to behave in ways that are very difficult for caregivers. For example, lack of emotional responsivity in patients is particularly distressing for caregivers [39], likely fostering reduced relationship quality and declines in caregiver health.
The smFG supports behavioral response inhibition, an important executive control function that leads to regulation of behaviors [40]. Greater activation in the medial prefrontal cortex has also been linked to use of emotion regulation strategies, including reappraisal of a situation to view it in a different way and suppression of negative affect to reduce one’s response [41, 42]. Damage to superior medial frontal regions may lead to poor response inhibition, ineffective emotion regulation, and inappropriate social behaviors (e.g., aggression and impulsivity) in patients. These behaviors can confuse and frustrate caregivers, creating additional burden and distress which negatively impact their health.
Patient Behaviors as Mediators of Caregiver Health
The present research supports our study hypothesis that neurodegeneration in brain regions critical for patient socioemotional functioning may lead to adverse health outcomes in their caregivers. Because of our cross-sectional observational design, the findings from the present study show a strong association between patient neurodegeneration, patient behavioral symptoms, and caregiver health at the between-person level. Although other mediational pathways are certainly possible, it is compelling to posit that neurodegeneration causes behavioral changes in patients and that these behavioral changes add to the burden of caregiving, leading to declines in caregiver health.
We evaluated one behavioral mediator in patients assessed by clinician-rated neuropsychiatric symptoms and one assessed by neuropsychological testing (cognitive functioning). Behavioral symptoms partially mediated the relationship between patient neurodegeneration and caregiver psychopathology, but not for global health, suggesting the importance of patient neurodegeneration and behavior in influencing caregiver mental health. Finally, patient cognitive functioning did not mediate the relationship between patient neurodegeneration and caregiver health; this is consistent with previous findings concerning the greater impact of behavioral symptoms on caregiver health [6]. Future longitudinal studies that measure patient neurodegeneration, patient behavioral symptoms, and caregiver health at multiple time points would allow for a full evaluation of these mediational pathways.
Value of the “Patient Brain to Caregiver Health” Pathway
Measuring brain volumes of the rvAI and smFG in patients significantly improved the prediction of both of our measures of caregiver health beyond patient behavioral symptoms and covariates. When considered together, brain volumes and behavioral symptoms were both significant independent predictors of caregiver psychopathology and caregiver global health. These findings suggest that the “patient brain to caregiver health” pathway can improve the prediction of adverse caregiver outcomes above and beyond the traditional “patient behavior to caregiver health” approach. Because different methodologies of measuring patient behavioral data have different advantages and disadvantages, we may never have the perfect behavioral measure. Our data suggest that including both MRI-derived measures of brain neurodegeneration and caregiver reports of problematic patient behavioral ratings may be most useful for predicting individual differences in vulnerability to the adverse health effects of caregiving.
Strengths and Limitations
Strengths of the present study include: (a) sampling a variety of neurodegenerative diseases in a relatively large sample; (b) using well-validated measures of health symptoms; (c) using whole-brain VBM instead of region-of-interest analyses; and (d) examining the additive predictive power of patient brain data beyond patient behavior data to increase the accuracy of predicting individual caregivers’ heightened risk for poor health. Limitations included: (a) use of a cross-sectional study design that is limited in determining causality without multiple measures of patient neurodegeneration, patient behavior, and caregiver health; (b) use of self-report health measures rather than more objective measures; (c) the lack of patient comparison groups, so that we cannot address whether the found effects can be generalized to other forms of neurological illness, such as stroke; (c) lack of additional variables that also undermine caregiver health, such as perceived social support and financial burden; (d) having mostly spousal caregivers, so that the results may not generalize to other familial caregivers; and (e) the lack of ethnoracial diversity in the sample, which may limit the results’ generalizability to non-European American patients and caregivers.
Conclusions
Our findings show the value of the “patient brain to caregiver health” approach, demonstrating that measuring patient behaviors and patient neurodegeneration improves predictions of poor mental and physical health in caregivers. Poor health in caregivers has enormous costs for caregivers and the patients in their care. Our recent research has shown that poor mental health in caregivers predicts shorter survival of patients, even when accounting for other key risk factors [43]. Thus, identifying predictors of poor health in caregivers is a critically important area for research and intervention that may extend the life of both the caregiver and the patient. When treatment teams encounter patients with high levels of neurodegeneration in specific regions, they may want to give particular attention to monitoring and treating health problems in their caregivers.
Supplementary Material
Acknowledgements
The research was supported by the National Institute on Aging Grants 1R01AG041762–01A1 and 2P01AG019724–11 (R.W.L. and B.L.M.). Time for this research was sponsored by the National Institutes of Mental Health (5T32MH020006–20; A.Y.H.).
Appendix: Neuroimaging Acquisition and Preprocessing Details
Ten structural MR images (6%) were acquired on a 1.5-T Siemens Magnetom VISION system (Siemens, Iselin, NJ, USA) at the San Francisco Veterans Administration Hospital, equipped with a standard quadrature head coil, using a magnetization-prepared rapid gradient-echo (MPRAGE) sequence (164 coronal slices; slice thickness, 1.5 mm; field of view [FOV], 256 × 256 mm; matrix, 256 × 256; voxel size, 1.0 × 1.5 × 1.0 mm; repetition time [TR], 10 ms; echo time [TE], 4 ms; flip angle, 15°).
Forty MR images (25%) were acquired on a 4-T Bruker MedSpec system at the San Francisco Veterans Administration Hospital with an 8-channel head coil controlled by a Siemens Trio console, using an MPRAGE sequence (192 sagittal slices; slice thickness, 1 mm; FOV, 256 × 224 mm; matrix, 256 × 224; voxel size, 1.0 × 1.0 × 1.0 mm; TR, 2,840 ms; TE, 3 ms; flip angle, 7°).
112 MR images (69%) were acquired on a 3.0-T Siemens TIM Trio scanner equipped with a 12-channel head coil located at the UCSF Neuroscience Imaging Center using volumetric MPRAGE (160 sagittal slices; slice thickness, 1.0 mm; FOV, 256 × 230 mm; matrix, 256 × 230; voxel size, 1.0 × 1.0 × 1.0 mm; TR, 2,300 ms; TE, 2.98 ms; flip angle, 9°).
For preprocessing, statistical parametric mapping version 12 default parameters were used with the light clean-up procedure in the morphological filtering step (http://www.fil.ion.ucl.ac.uk/spm/software/spm12/). Structural T1 images were corrected for bias field, segmented into gray matter, white matter, and cerebrospinal fluid, and spatially normalized into MNI space [44]. Default tissue probability priors (voxel size, 2.0 × 2.0 × 2.0 mm) of the International Consortium for Brain Mapping were used. Segmented images were visually inspected for adequate gray matter segmentation. These images were then smoothed with an 8-mm full-width-at-half-maximum gaussian kernel filter.
Footnotes
Disclosure Statement
The authors declare no conflict of interest.
Statement of Ethics
The participants (or their legal guardians) have given their written informed consent. The study protocols have been approved by the Institutional Review Boards of the UCB and the UCSF.
No specific set of behavioral symptoms (i.e., disinhibition, apathy, euphoria, etc.) individually mediated the relationship between patient gray matter volumes in the rvAI and smFG and caregiver psychopathology, suggesting that the overall degree of behavioral symptoms mediates this relationship.
To address whether patient behavioral symptoms specifically mediate the relationship between patient neurodegeneration and caregiver mental health, we computed a measure of caregiver mental health by averaging the following subscale scores on the SF-36: role limitations due to emotional problems, emotional well-being, and social functioning. Post hoc mediational analysis (using the same covariates as in the VBM analyses) revealed that patient behavioral symptoms mediated the relationship between patient gray matter volumes in the rvAI and smFG and caregiver mental health (derived from the SF-36; standardized indirect effect = 0.04, 95% CI [0.001, 0.12], accounting for 0.20 of the total effect).
References
- 1.World Health Organization. WHO | Dementia [Internet]. Geneva: WHO; 2017. [cited 2017 Aug 25]. Available from: http://www.who.int/mediacentre/factsheets/fs362/en/. [Google Scholar]
- 2.Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009; 11(2): 217–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ory MG, Hoffman RR 3rd, Yee JL, Tennstedt S, Schulz R. Prevalence and impact of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999. Apr; 39(2): 177–85. [DOI] [PubMed] [Google Scholar]
- 4.Gaugler JE, Kane RL, Newcomer R. Resilience and transitions from dementia caregiving. J Gerontol B Psychol Sci Soc Sci. 2007. January; 62(1): 38–44. [DOI] [PubMed] [Google Scholar]
- 5.Schulz R, O’Brien AT, Bookwala J, Fleissner K. Psychiatric and physical morbidity effects of dementia caregiving: prevalence, correlates, and causes. Gerontologist. 1995. December; 35(6): 771–91. [DOI] [PubMed] [Google Scholar]
- 6.Ornstein K, Gaugler JE. The problem with “problem behaviors”: a systematic review of the association between individual patient behavioral and psychological symptoms and caregiver depression and burden within the dementia patient-caregiver dyad. Int Psychogeriatr. 2012. October; 24(10): 1536–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD. Neurodegenerative diseases target large-scale human brain networks. Neuron. 2009. April; 62(1): 42–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Levenson RW, Sturm VE, Haase CM. Emotional and behavioral symptoms in neurodegenerative disease: a model for studying the neural bases of psychopathology. Annu Rev Clin Psychol. 2014; 10(1): 581–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhou J, Greicius MD, Gennatas ED, Growdon ME, Jang JY, Rabinovici GD, et al. Divergent network connectivity changes in behavioural variant frontotemporal dementia and Alzheimer’s disease. Brain. 2010. May; 133(Pt 5): 1352–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ascher EA, Sturm VE, Seider BH, Holley SR, Miller BL, Levenson RW. Relationship satisfaction and emotional language in frontotemporal dementia and Alzheimer disease patients and spousal caregivers. Alzheimer Dis Assoc Disord. 2010. Jan-Mar; 24(1): 49–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.de Vugt ME, Riedijk SR, Aalten P, Tibben A, van Swieten JC, Verhey FR. Impact of behavioural problems on spousal caregivers: a comparison between Alzheimer’s disease and frontotemporal dementia. Dement Geriatr Cogn Disord. 2006; 22(1): 35–41. [DOI] [PubMed] [Google Scholar]
- 12.Riedijk SR, De Vugt ME, Duivenvoorden HJ, Niermeijer MF, Van Swieten JC, Verhey FR, et al. Caregiver burden, health-related quality of life and coping in dementia caregivers: a comparison of frontotemporal dementia and Alzheimer’s disease. Dement Geriatr Cogn Disord. 2006; 22(5–6): 405–12. [DOI] [PubMed] [Google Scholar]
- 13.Cummings JL. The Neuropsychiatric Inventory: assessing psychopathology in dementia patients. Neurology. 1997. May; 48(5 Suppl 6):S10–6. [DOI] [PubMed] [Google Scholar]
- 14.Otero MC, Levenson RW. Lower visual avoidance in dementia patients is associated with greater psychological distress in caregivers. Dement Geriatr Cogn Disord. 2017; 43(5–6): 247–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen KH, Wells JL, Otero MC, Lwi SJ, Haase CM, Levenson RW. Greater experience of negative non-target emotions by patients with neurodegenerative diseases is related to lower emotional well-being in caregivers. Dement Geriatr Cogn Disord. 2017; 44(5–6): 245–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Brown CL, Lwi SJ, Goodkind MS, Rankin KP, Merrilees J, Miller BL, et al. Empathic accuracy deficits in patients with neurodegenerative disease: association with caregiver depression. Am J Geriatr Psychiatry. 2018. April; 26(4): 484–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Matsumoto N, Ikeda M, Fukuhara R, Shinagawa S, Ishikawa T, Mori T, et al. Caregiver burden associated with behavioral and psychological symptoms of dementia in elderly people in the local community. Dement Geriatr Cogn Disord. 2007; 23(4): 219–24. [DOI] [PubMed] [Google Scholar]
- 18.Schulz R, Cook TB, Beach SR, Lingler JH, Martire LM, Monin JK, et al. Magnitude and causes of bias among family caregivers rating Alzheimer disease patients. Am J Geriatr Psychiatry. 2013. January; 21(1): 14–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scarmeas N, Stern Y. Cognitive reserve: implications for diagnosis and prevention of Alzheimer’s disease. Curr Neurol Neurosci Rep. 2004. September; 4(5): 374–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guevara AB, Demonet JF, Polejaeva E, Knutson KM, Wassermann EM, Grafman J, et al. Association between traumatic brain injury-related brain lesions and long-term caregiver burden. J Head Trauma Rehabil. 2016. Mar-Apr; 31(2):E48–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Knutson KM, Zamboni G, Tierney MC, Grafman J. Neural correlates of caregiver burden in cortical basal syndrome and frontotemporal dementia. Dement Geriatr Cogn Disord. 2008; 26(5): 467–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Testa C, Laakso MP, Sabattoli F, Rossi R, Beltramello A, Soininen H, et al. A comparison between the accuracy of voxel-based morphometry and hippocampal volumetry in Alzheimer’s disease. J Magn Reson Imaging. 2004. March; 19(3): 274–82. [DOI] [PubMed] [Google Scholar]
- 23.Rosen HJ, Levenson RW. The emotional brain: combining insights from patients and basic science. Neurocase. 2009. June; 15(3): 173–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Armstrong MJ, Litvan I, Lang AE, Bak TH, Bhatia KP, Borroni B, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology. 2013. January; 80(5): 496–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Litvan I, Agid Y, Calne D, Campbell G, Dubois B, Duvoisin RC, et al. Clinical research criteria for the diagnosis of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome): report of the NINDS-SPSP international workshop. Neurology. 1996. July; 47(1): 1–9. [DOI] [PubMed] [Google Scholar]
- 26.Rascovsky K, Hodges JR, Knopman D, Mendez MF, Kramer JH, Neuhaus J, et al. Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain. 2011. September; 134(Pt 9): 2456–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gorno-Tempini ML, Hillis AE, Weintraub S, Kertesz A, Mendez M, Cappa SF, et al. Classification of primary progressive aphasia and its variants. Neurology. 2011. March; 76(11): 1006–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011. May; 7(3): 263–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O’Bryant SE, Humphreys JD, Smith GE, Ivnik RJ, Graff-Radford NR, Petersen RC, et al. Detecting dementia with the Mini-Mental State Examination in highly educated individuals. Arch Neurol. 2008. July; 65(7): 963–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993. November; 43(11): 2412–4. [DOI] [PubMed] [Google Scholar]
- 31.Derogatis LR, Unger R. Symptom Checklist-90-Revised [Internet]. Corsini Encycl Psychol. John Wiley & Sons; 2010. 10.1002/9780470479216.corpsy0970. [DOI] [Google Scholar]
- 32.Ware JE Jr, Sherbourne CD. The MOS 36-item Short-Form Health Survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992. June; 30(6): 473–83. [PubMed] [Google Scholar]
- 33.Abdulkadir A, Mortamet B, Vemuri P, Jack CR Jr, Krueger G, Klöppel S; Alzheimer’s Disease Neuroimaging Initiative. Effects of hardware heterogeneity on the performance of SVM Alzheimer’s disease classifier. Neuroimage. 2011. October; 58(3): 785–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Seeley WW. Anterior insula degeneration in frontotemporal dementia. Brain Struct Funct. 2010. June; 214(5–6): 465–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Craig AD. Significance of the insula for the evolution of human awareness of feelings from the body. Ann N Y Acad Sci. 2011. April; 1225(1): 72–82. [DOI] [PubMed] [Google Scholar]
- 36.Uddin LQ. Salience processing and insular cortical function and dysfunction. Nat Rev Neurosci. 2015. January; 16(1): 55–61. [DOI] [PubMed] [Google Scholar]
- 37.Chang LJ, Yarkoni T, Khaw MW, Sanfey AG. Decoding the role of the insula in human cognition: functional parcellation and large-scale reverse inference. Cereb Cortex. 2013. March; 23(3): 739–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bechara A, Damasio AR. The somatic marker hypothesis: a neural theory of economic decision. Games Econ Behav. 2005; 52(2): 336–72. [Google Scholar]
- 39.Merrilees J, Dowling GA, Hubbard E, Mastick J, Ketelle R, Miller BL. Characterization of apathy in persons with frontotemporal dementia and the impact on family caregivers. Alzheimer Dis Assoc Disord. 2013. Jan-Mar; 27(1): 62–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li CS, Huang C, Constable RT, Sinha R. Imaging response inhibition in a stop-signal task: neural correlates independent of signal monitoring and post-response processing. J Neurosci. 2006. January; 26(1): 186–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ochsner KN, Ray RD, Cooper JC, Robertson ER, Chopra S, Gabrieli JD, et al. For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion. Neuroimage. 2004. October; 23(2): 483–99. [DOI] [PubMed] [Google Scholar]
- 42.Ochsner KN, Bunge SA, Gross JJ, Gabrieli JDE. Rethinking feelings: an fMRI study of the cognitive regulation of emotion. J Cogn Neurosci. 2002. November; 14(8): 1215–29. [DOI] [PubMed] [Google Scholar]
- 43.Lwi SJ, Ford BQ, Casey JJ, Miller BL, Levenson RW. Poor caregiver mental health predicts mortality of patients with neurodegenerative disease. Proc Natl Acad Sci USA. 2017. July; 114(28): 7319–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ashburner J, Friston KJ. Unified segmentation. Neuroimage. 2005. July; 26(3): 839–51. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.