Abstract
Introduction:
Most smokers see a physician each year, but few use any assistance when they try to quit. Text messaging programs improve smoking cessation in community and school settings; however, their efficacy in a primary care setting is unclear. The current trial assesses the feasibility and preliminary clinical outcomes of text messaging and mailed nicotine replacement therapy (NRT) among smokers in primary care.
Methods:
In this single-center pilot randomized trial, eligible smokers in primary care are offered brief advice by phone and randomly assigned to one of four interventions: (1) Brief advice only, (2) text messages targeted to primary care patients and tailored to quit readiness, (3) a 2-week supply of nicotine patches and/or lozenges (NRT), and (4) both text messaging and NRT. Randomization is stratified by practice and intention to quit. The text messages (up to 5/day) encourage those not ready to quit to practice a quit attempt, assist those with a quit date through a quit attempt, and promote NRT use. The 2-week supply of NRT is mailed to patients’ homes.
Results:
Feasibility outcomes include recruitment rates, study retention, and treatment adherence. Clinical outcomes are assessed at 1,2, 6, and 12-weeks post-enrollment. The primary outcome is ≥1 self-reported quit attempt(s). Secondary clinical outcomes include self-reported past 7- and 30-day abstinence, days not smoked, NRT adherence, and exhaled carbon monoxide.
Conclusions:
This pilot assesses text messaging plus NRT, as a proactively offered intervention for smoking cessation support in smokers receiving primary care and will inform full-scale randomized trial planning.
Trial Registration:
Keywords: Mobile health, text messaging, nicotine replacement therapy, smoking cessation, primary care
INTRODUCTION
Background
Despite large declines in smoking prevalence over the past 50 years, 37 million Americans continue to smoke.1 In the US, 70% of smokers visit a physician each year, and 52% make a quit attempt.2 Effective treatments are available to assist smokers, but just 32% use treatments in their attempt.3 One promising strategy to address this treatment gap is mobile health technology.4,5 Text messages that deliver tobacco cessation support increases abstinence among smokers interested in quitting in community- or school-based settings.6-12
Integrating text messaging programs into primary care has clear advantages. First, healthcare systems have electronic documentation of smoking status in electronic health records (EHR).13 This information enables primary care practices to identify and proactively target smokers with tailored health interventions outside of the clinic and without waiting for smokers to seek care. Furthermore, text messaging programs originating from the physician’s office may leverage the influence physicians have on smokers’ motivation to quit. Indeed, advice from a physician is a predictor of smoking cessation, and smokers cite advice from a doctor as important motivation to quit.14,15 Individuals also often look to their healthcare systems for digital health information,16 so health-promoting advice may be more salient if it originates from local healthcare providers than from public health or community entitites.
Integrating text messaging within the healthcare system provides opportunities to coordinate with pharmacotherapy. Text messaging has been used in healthcare systems to promote medication adherence for other chronic illnesses including HIV, cardiac disease, and mental illness. 17-20 For smokers, adherence to cessation medications is associated with abstinence.21-24 However, smokers have suboptimal adherence with as few as 20% ever filling a prescription for smoking cessation medications, many using lower doses than recommended, and over half of NRT users discontinuing the medication early.25-31
The few studies that have examined the feasibility or effectiveness of text messages as smoking cessation support in healthcare settings have produced mixed results. Studies examining text messaging plus varenicline among patients in primary care and HIV care have shown no effect of text messaging on smoking cessation.32, 33 One study of text messaging among smokers discharged from a hospital stay showed no effect on smoking outcomes.34 While studies comparing text messaging to usual care or a brief behavioral intervention have shown improved abstinence outcomes among treatment seeking primary care and post-myocardial infarction patients.35-37Most of these prior studies targeted motivated or treatment seeking smokers. The convenience of text messaging may have appeal to even less motivated smokers. Proactive models that offer assistance to all smokers in healthcare settings, even those not ready to quit, have shown improvements in treatment use and cessation.38-43 However, there is a need to explore whether a proactively offered text messaging intervention can effectively promote quit attempts or smoking cessation medication use in primary care patients.
METHODS
Pilot Study Aims
This pilot study aims to evaluate the feasibility of a randomized trial of text messaging for primary care patients who smoke. The original grant proposal planned a two-arm trial of mailed NRT compared to mailed NRT plus text messaging. However, studies published in the two years between grant submission and study initiation prompted changes in the design. As noted above, two published trials that compared text messaging to usual care or a brief behavioral intervention and both found an increase in sustained cessation.36,44 Two other studies compared varenicline to varenicline plus text messaging and found no effect.32,33 We therefore modified the design to add groups that are offered text messaging or a brief behavioral intervention without medications. We also maintained the original groups that are offered mailed NRT with or without text messaging to measure the effect of text messaging on medication use. The resulting four-arm trial randomizes patients to an offer of: (1) Brief advice only, (2) text messages targeted to primary care patients and tailored to quit readiness, (3) a 2-week supply of nicotine patches and/or lozenges (NRT), or (4) both text messaging and NRT. In addition to feasibility testing, this design provides preliminary estimates of smoking quit attempts, cessation outcomes, and NRT use under the four intervention conditions. Results from this study will inform the design of a full-scale pragmatic randomized controlled trial (RCT). We have the following aims:
Aim 1: To evaluate recruitment and retention, with a target of 12-months of recruitment and target response rate of ≥75% at the 12-week assessment.
Aim 2: To provide a preliminary estimate of the clinical impact of text messaging, NRT, or text messaging plus mailed NRT on self-reported quit attempts (primary outcome), 7-day self-reported point prevalent abstinence, biochemically confirmed abstinence and days not smoked compared to a group receiving brief advice from a research coordinator trained as a tobacco treatment specialist.
Aim 3: To provide a preliminary estimate of the impact of text messaging on NRT adherence among those receiving text messaging plus NRT compared to NRT alone.
Aim 4: To explain smoking outcome results using end-of-study subject interviews in anticipation of a full-scale RCT.
Study Design
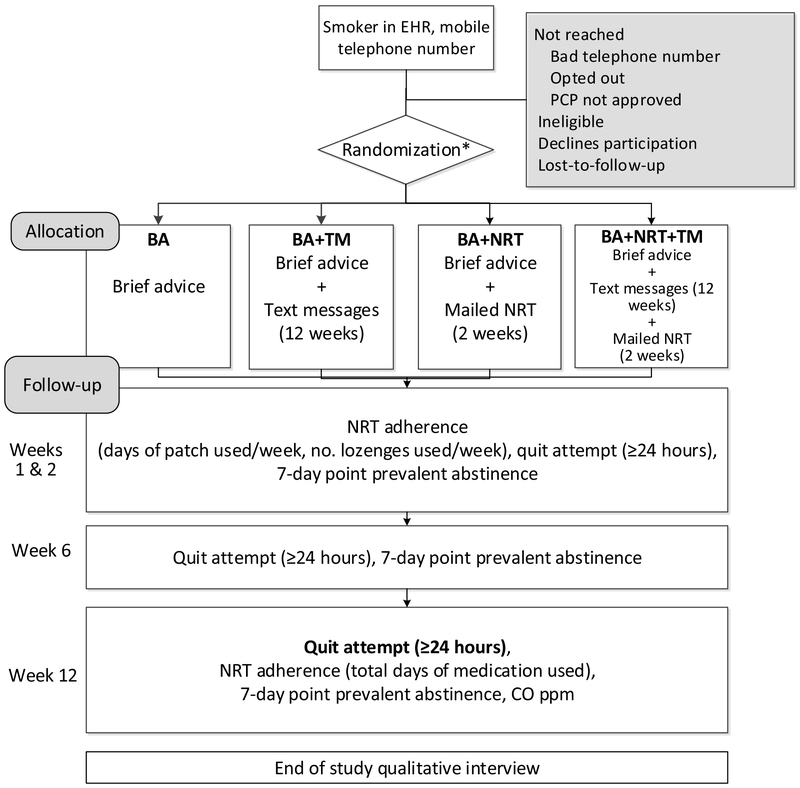
This 12-week single-center unblinded four-arm parallel pilot randomized trial (ClinicalTrials.gov Identifier: NCT03174158) aims to evaluate trial feasibility and pilot effects of a text messaging program and mailed NRT among smokers in primary care (Figure 1).
Figure 1.
Pilot Trial Schema
Abbreviations: EHR-electronic health record, PCP-primary care provider, NRT-nicotine replacement therapy, CO-carbon monoxide, ppm-parts per million.
Ethical Approval
The research methods to be used in this study were approved by the Partners Healthcare Institutional Review Board in an expedited review in July 2017.
Study Population
The study is recruiting Massachusetts General Hospital (MGH) patients who receive their primary care within the Partners Healthcare System, a large healthcare system in Massachusetts founded by MGH and Brigham and Women’s Hospital. Eligibility criteria are shown in Table 1. Smoking status was used to identify the initial sample. Smoking status in the EHR for primary care patients at MGH is not mandatory field but incentives have been used to promote structured documentation.45 In 2016, smoking status was missing for 6.27% of primary care patients.
Table 1.
Study subject eligibility criteria
| Inclusion criteria | Exclusion criteria |
|---|---|
|
|
We include adult patients only because usual care practices to address tobacco differ in pediatric practices. At this pilot stage, we are testing only an English language version, but if promising the text messaging intervention can be adapted to reach smokers who speak other languages.46,47 Trial participants must be patients at MGH because NRT prescriptions are entered into the MGH medical record and mailed from the MGH outpatient pharmacy. Exclusion criteria include contraindications or cautions to use of NRT (e.g. prior serious adverse reaction to NRT, unstable cardiac conditions or low weight) and conditions that would affect ability to participate in informed consent. Patients were excluded if the mobile phone listed in their EHR was non-working at the time of contact.
Recruitment
Patients are recruited through four methods. First, the Research Opportunities Direct to You program at MGH allows patients to consent to be contacted directly about local research studies. Patients who consent through this program and who are in MGH primary care practices are identified from a Primary Care Practice Based Research Network using an existing smoking algorithm.48 Second, using the same algorithm, lists of patients who smoke and have not consented to be contacted directly are sent to PCPs for review and approval for contact by study staff. Third, the MGH inpatient tobacco treatment counselors refer potentially eligible patients to the study research coordinator to be contacted about the study. Finally, the study is advertised on the healthcare system’s research studies website and listserv.
Patients from these sources are sent informational letters that include study objectives, activities, research subject payments and opt-out instructions before being contacted by phone. A research coordinator attempts to contact patients with up to five telephone calls including up to three voice messages. Patients who are reached are screened for eligibility by telephone script. The research coordinator administers a verbal informed consent process to eligible and interested patients. The study physician (GRK) reviews the medical charts of consented patients for contraindications to NRT, unstable cardiac disease or conditions that may affect ability to consent, seeking clarification from the primary care provider if needed. Subjects meeting eligibility criteria by telephone survey and chart review are contacted again by telephone for baseline assessment and delivery of brief behavioral advice followed by randomization.
Randomization and Blinding
We use stratified block randomization. Strata are primary care practice and intention to quit in the next 30 days. Subjects are randomized 1:1:1:1 to four groups (Figure 1). Randomization is automated using REDCap (Research Electronic Data Capture) and a locked dataset with a random treatment assignment order which automatically populates a field in REDCap. The upcoming assignment sequence is not accessible or visible to the research coordinator informing participants of their study arm assignment. After assignment, research staff and patients are not blinded to treatment assignment.
Study Interventions
Brief advice
All subjects are offered 3-5 minutes of brief advice from a research coordinator (NNS) who completed the University of Massachusetts Tobacco Treatment Specialist training. Brief advice includes discussion of treatment options and information about local tobacco cessation resources.
GetReady2Quit text messaging
Smokers randomized to the text messaging arms (TM and TM+NRT) have their mobile numbers and first names entered into a web-based text messaging platform (Mobile Commons, Upland Software, Austin, Texas). This messaging program lasts up to 12 weeks (up to 4 weeks pre-quit and 8 weeks post-quit attempt), and smokers receiving this intervention are sent up to 5 text messages per day consistent with most previously tested smoking cessation text messaging programs.49 This program combines content from the National Cancer Institute’s SmokeFreeTXT program4 tailored to the local setting, a prior pilot feasibility study50 that included motivational and practice quit content, and novel messages supporting NRT adherence Messages encouraging NRT adherence include advice for proper NRT use, education about medication safety,51,52 medication reminders, and motivational messages. These messages are based on the Information-Motivation-Behavioral Skills theoretical model of medication adherence.53 We used real-time ratings and qualitative telephone interviews with primary care patients to tailor existing content and develop novel content encouraging nicotine replacement therapy use among primary care patients.54
For smokers who have a quit date set in the next 30 days, messages include behavioral tips for cessation, motivational and encouraging messages, fact-based messages about health risks, and periodic assessments.55 Smokers can also request specific advice including messages assisting with cravings by texting keywords, e.g. “CRAVE”. Smokers are asked after their quit date if they have quit, if not they can reset their planned quit date or transfer to receive the messages for smokers who are not ready to set a quit date.
For smokers who do not have a quit date in the next 30 days, messages include motivational messages and encouragement to make a practice quit attempt. This idea of practicing quitting is based on the concept of quit induction, where pre-quit activities increase self-efficacy and motivation, eventually moving a smoker towards action and a serious quit attempt.56 We previously developed and pilot tested content promoting practice quit attempts and motivational advice.50 Smokers who do not initially have a quit date are asked again at week one and week four if they are ready to set a quit date. If they are ready to quit they are transferred to receive the messages for smokers with a quit date.
Text messages are personalized by user’s preferred first name, quit readiness and user-entered quit date. Text messages also include telephone numbers for the MGH tobacco cessation counseling service Living Tobacco-Free, which accepts self-referrals, and the Massachusetts smoker’s helpline.
Mailed NRT
Participants randomized to receive nicotine patches and/or lozenges are offered these materials in a 2-week allotment. Daily smokers with a quit date are offered patches and lozenges. Smokers not ready to quit are offered only lozenges to use when not smoking during practice quit attempts.56 We recommend patch and lozenge dosing as per package instructions. Participants have the option to request a lower dose. The hospital outpatient pharmacy mails the 2-week supply of medication directly to the subject’s residence. All participants are sent information about the Massachusetts state smoker’s helpline number and smoking cessation services available within the healthcare system. Furthermore, participants are free to participate in any other smoking cessation service or support that they wish to use.
Study Activities
Participants are asked to complete surveys by telephone or email at baseline, one week, two weeks, six weeks and 12 weeks post-enrollment. Subjects who report 7-day point-prevalent abstinence are requested to meet study staff in-person to collect an exhaled carbon monoxide. A subset of patients are also invited to participate in an end-of-study semi-structured telephone interview about their experience. We compensate participants up to $120 in the form of gift cards for the completion of each of the five surveys, the carbon monoxide measure, and the end-of-study interview. Participants assigned to text messaging arms are also reimbursed $0.25 per text message so that text messaging costs are not a barrier to participation for patients without unlimited text messaging cellular plans.
Baseline Survey
All survey data are collected using REDCap. Baseline surveys are collected by telephone after consent and chart review by the study physician. Items include the Heaviness of Smoking Index,57 previous quit attempts defined as any serious quit attempt lasting ≖24 hours,58 prior intentional quit attempts lasting less than 24 hours, prior use of smoking cessation medication (nicotine patch, lozenge, gum, inhaler or nasal spray, varenicline, or bupropion), prior use of evidence behavioral treatment for smoking cessation (clinic or class, in-person counseling, telephone counseling, prior use of text messaging, alternative therapy, self-help materials, or online communities and applications), use of other tobacco products, use of electronic nicotine delivery systems, motivation to quit, confidence to quit, perceived 2-week distress scale, readiness to quit,59 PHQ-4 screening for depression and anxiety,60 single-item alcohol61 and substance use disorder62 screening questions, and illegal use of drugs in the past 30 days.63
Follow-up Surveys
Participants complete follow-up outcome surveys at one-, two-, six- and 12-weeks post-enrollment by their choice of either email or telephone. A time-window of up to four days is allowed for completion of the one- and two-week surveys, a window of up to seven days is allowed for completion of the six-week survey and one month for completion of the 12-week survey. All calls are scripted. In addition to outcome measures, the one- and two-week surveys include items verifying receipt of text messages and/or mailed medications. At the two-week survey, we also offer to send a refill request via the EHR to the participant’s PCP so that NRT refills can be billed through their usual insurance and pharmacy for participants in the NRT or TM+NRT arms.
Exhaled carbon monoxide
We invite participants reporting past 7-day abstinence at the 12-week follow-up to provide an exhaled carbon monoxide measure in-person at a hospital-based Clinical Research Center or in their primary care clinic.
Qualitative exit interviews
Qualitative end-of-study interviews are conducted by telephone and audio-recorded. Interviews explore subjects’ reasons for joining the study, expectations and experiences with the assigned treatments, any barriers or facilitators to making a quit attempt or to successfully quitting, and any issues with the study procedures. A purposive sample of participants is selected based on 12-week survey outcomes to include representatives from all treatment arms and smoking outcomes. Interview recordings are transcribed verbatim for thematic analysis.
Outcome Measures
Process Measures
Process measures include participant recruitment rate, retention, fidelity of assigned interventions, text messaging adherence and engagement.
Recruitment: Number of subjects enrolled per week, number of recruitment calls made per enrollee, proportion of contactable patients who are randomized and proportion of patients reached and assessed who are eligible and randomized.
Retention and outcome capture: Proportion of participants at each assessment point with complete or missing data.
Treatment fidelity is measured using follow-up surveys and data from the text messaging platform. At follow-up surveys, subjects are asked to verbally confirm receipt of text messages. The text messaging platform data are stored on a secure server and used to analyze user-entered quit dates, real-time feedback requests, and time in the program before discontinuing the messages.
Text messaging program adherence is measured as the length of active time in the text messaging program. Active time is defined as number of days from first to last message sent or received.
Text messaging engagement is measured as the number of messages sent from participant to the program, which could be in response to periodic assessments that are sent every 7 days starting on the quit date and continuing until the end of the program or to request specific help with cravings, mood symptoms, or slips. We also measure the number and proportion of URL links clicked in the messages.
Delivery of the mailed nicotine replacement therapy is verbally verified at the 1-week follow-up survey. Status of mailed NRT reported as not yet received is assessed using pharmacy records and a postal tracking number. We also use a weekly audit process to reconcile prescriptions sent via fax or electronically to the pharmacy with receipt and mailing date from pharmacy.
Smoking outcome measures
One- and two-week survey items measure self-reported quit attempts, days of nicotine patch use, days and quantity of nicotine lozenge use, number of days smoked, and medication side effects. Six-week survey items measure self-reported quit attempts, days of nicotine patch use, days and quantity of nicotine lozenge use, number of days smoked in the past 30 days, past 7-day abstinence and medication side effects.
The primary clinical outcome is the proportion of participants who report having made one or more quit attempts lasting 24 hours or longer at 12-weeks post-enrollment. Other 12-week survey outcomes include the proportion of participants with self-reported quit attempts lasting less than 24 hours,64 days of nicotine patch use, days and quantity of nicotine lozenge use, receipt of text messages, number of days smoked in the past 30 days, past 7-day abstinence, medication side effects, cigarettes per day, motivation, confidence, distress, use of other evidence-based treatments,15 and use of other tobacco products.
Exhaled carbon monoxide samples are collected by study staff (SEH) with the coVita | Bedfont Micro+ Smokerlyzer carbon monoxide monitor. Exhaled carbon monoxide result of ≤9 parts per million is considered confirmation of cessation.65
Sample Size and Power Calculations
The sample size for this pilot study is selected to adequately estimate recruitment rates, retention, and to get a preliminary estimate of prevalence of clinical outcomes. We anticipate we can recruit 200 patients by proactive outreach over 12 months. With 50 patients per group and 25% loss to follow-up we will have 81% power to detect a relative risk of self-reported ≥24 hour quit attempts of 1.8 for text messaging versus brief advice groups. We base this estimate on meta-analyses of smoking cessation text messaging trials, mostly from community settings or school-based settings, report a relative risk of 1.4 for 7-day point prevalence abstinence and 1.6 for prolonged abstinence measured at six-months.66,67 With this sample size, we also have 80% power to detect an increase in days of NRT use from 9 to 12 days in NRT vs. NRT+TM.
Analysis Plan
Statistical Analysis
The process and feasibility analysis include the calculation of the proportion of potentially eligible patients who we attempt to contact, the proportion of patients we reach and screen for eligibility, and the proportion eligible and randomized. To determine our sample’s representativeness to the target population, baseline measures from the EHR are compared between the sample of smokers enrolled and potentially eligible smokers not enrolled. These measures include demographics and insurance status. To determine the success of randomization, we compare baseline measures between the four study arms. Participant retention and completion of follow-up surveys at each time point is calculated overall and across study arms to examine non-differential loss to follow-up by treatment assignment using chi-square statistics.
To examine intervention adherence, we compare days in the text messaging program and number of text messages sent and received by study arm (TM and TM+NRT) using chi-square or t-test statistics. We use linear regression to compare medication adherence between the TM+NRT group and the NRT group in terms of total days of medication used and total mg of medication over the first two weeks post-randomization. Multivariable regression models are used to identify predictors of days of medication used and mg of medication used in the NRT and TM+NRT groups.
The primary clinical outcome analysis is conducted as intention-to-treat, comparing the primary outcome using a multiple logistic regression that includes study group, practice, and any baseline factor that is significantly imbalanced by group (p < 0.05) as independent variables. The 4-arm design also allows for analysis as a factorial design. We first test for interaction between the NRT and TM interventions. If no interaction is detected, we then test the effect of text messaging using both the TM and TM+NRT arms compared to the BA and NRT arms. If an interaction is present, we compare the TM arm to the BA for our primary analysis. We secondarily compare the groups in terms of self-reported 7-day PPA, biochemically confirmed abstinence, number of days not smoked, use of any evidence-based treatments, and cigarettes per day using logistic or linear regression. Analyses of the primary outcome, 7-day PPA, treatment use, and days not smoked are stratified by readiness to quit in the next 30 days. We also use multivariable regression to examine the association between adherence and engagement with the text messaging intervention and smoking outcomes, and the association between NRT adherence and smoking outcomes.
Missing smoking outcomes are assumed to be negative, i.e. no quit attempt or currently smoking.68 We also conduct sensitivity analyses using multiple imputation to impute missing smoking outcomes. All analyses are conducted using SAS version 9.4 (Carey, NC).
Qualitative Analysis
End-of-study telephone interviews are transcribed verbatim and analyzed using NVivo version 11.55 Two coders (GRK and NNS) conduct content analysis using the constant comparative method.69 A codebook is developed by iteratively reviewing raw data to identify themes. The themes are compared between coders, and any discrepancies are resolved by discussion with a third researcher. Intercoder agreement is calculated by kappa statistic. Themes are compared by study intervention and clinical smoking outcomes to explain the pilot trial results.
Discussion
While many smokers receiving primary care attempt to quit, these attempts are not always successful. One way to increase quit attempts and improve smoking cessation successes is using mobile health technology. Text messaging programs effectively promote smoking cessation in other settings. The unique context of the healthcare system has clear advantages for the implementation of text messaging smoking cessation support including coordination with pharmacotherapy or behavioral resources and the opportunity to offer the intervention proactively to smokers identified in the electronic health record.
We believe this is also the first trial examining the feasibility of a proactively offered text messaging program on quit attempts or smoking cessation among primary care patients. Most prior trials of text messaging in primary care have recruited motivated smokers. However, there are fewer evidence-based treatments to offer smokers who are not ready to quit in the next 30 days and the time limitations of a primary care visit make it challenging for providers to deliver assistance during visits. Text messaging may be a way to meaningfully engage smokers who are not ready to quit in cessation promoting activities.
While prior trials have studied the effects of text messaging on varenicline use,32, 33 this trial is the first to our knowledge to examine the effects of theory-based medication adherence text messages on NRT use. Medication adherence is associated with smoking cessation, and text messaging has been effective at increasing adherence to other medications.70 However, few interventions aiming to improve NRT adherence have shown promising results or are readily scalable. The results of this pilot RCT should establish the feasibility of testing the effects of an intervention combining text messaging and mailed NRT to smokers in primary care and provide preliminary effect estimates of the two intervention components.
This study design is not without limitations. First, the goal of smoking cessation interventions is sustained quitting, not quit attempts, and this pilot RCT will be underpowered to detect a difference in past 7- or 30-day abstinence at the end of treatment. We feel quit attempts is a clinically meaningful and appropriate outcome for this low intensity intervention targeting both patients who are and are not ready to quit. A full-scale trial will measure sustained cessation. Our power calculation is based on comparing our TM, NRT or TM+NRT to brief advice. We do not expect to be powered to detect a difference between other intervention arms (NRT vs. TM+NRT or TM vs TM+NRT).
Second, this pilot study is not blinded. While outcome surveys completed by email are unlikely to be influenced by knowledge of treatment group, there may be bias in outcome surveys conducted by telephone by the research coordinator who is not blinded to intervention assignment. A future full-scale RCT will use a blinded outcome assessment process. Single-blind designs have been used in large-scale studies of text messaging.36, 71
A third limitation is our telephone-based recruitment method. For this pilot study, we will use proactive telephone outreach and telephone-delivered verbal consent process. Many of the patients who may be interested and benefit most from an asynchronous text messaging smoking cessation intervention may be those who are not routinely available during regular business hours. Our research staff are available to make calls outside of traditional business hours at limited times and this limited after-hours access may exclude some potential patients.
The completion of this trial will provide evidence of the feasibility of recruiting primary care patients who are documented as smokers in the electronic health record, including patients ready to quit and patients not ready to quit, to a text messaging study and collecting data about their smoking outcomes and utilization of study interventions over 12 weeks.
Acknowledgements
We are grateful to the oversight and advice of the Scientific Advisory Committee for this K award project including Drs. Joshua Metlay, Daniel Singer, Glorian Sorensen, and Vish Viswanath.
Funding and disclosures: This study was funded by National Institute on Drug Abuse #K23 5K23DA038717 and the Massachusetts General Hospital Executive Committee on Research Claflin Distinguished Scholar Award. GRK has a family financial interest in Dimagi Inc. and is a paid consultant for Click Therapeutics Inc. NAR receives royalties from UpToDate, is an unpaid consultant for Pfizer, and a paid consultant for Achieve Life Sciences. JEH is supported by K24MH114732. LA has stock in Welltok Inc. and receives royalties from the licensing of Text2Quit to Welltok Inc.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Jamal A, Phillips E, Gentzke AS, et al. Current Cigarette Smoking Among Adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(2):53–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention. Cigarette smoking among adults--United States, 2000. MMWRMorbidity and mortality weekly report. 2002;51(29):642–645. [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention. Quitting smoking among adults--United States, 2001-2010. MMWR Morb Mortal Wkly Rep. 2011;60:1513–1519. [PubMed] [Google Scholar]
- 4.National Cancer Institute. QuitNowTXT Message Library. 2011. [Google Scholar]
- 5.Abroms LC, Boal AL, Simmens SJ, et al. A randomized trial of Text2Quit: a text messaging program for smoking cessation. Am J Prev Med. 2014;47(3):242–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Whittaker R, Borland R, Bullen C, et al. Mobile phone-based interventions for smoking cessation. Cochrane Database Syst Rev. 2009(4):CD006611. [DOI] [PubMed] [Google Scholar]
- 7.Free C, Knight R, Robertson S, et al. Smoking cessation support delivered via mobile phone text messaging (txt2stop): a single-blind, randomised trial. Lancet. 2011;378(9785):49–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen YF, Madan J, Welton N, et al. Effectiveness and cost-effectiveness of computer and other electronic aids for smoking cessation: a systematic review and network meta-analysis. Health Technol Assess. 2012;16(38):1–205. [DOI] [PubMed] [Google Scholar]
- 9.Abroms LC, Ahuja M, Kodl Y, et al. Text2Quit: results from a pilot test of a personalized, interactive mobile health smoking cessation program. J Health Commun. 2012;17 Suppl 1:44–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brendryen H, Kraft P. Happy ending: a randomized controlled trial of a digital multimedia smoking cessation intervention. Addiction. 2008;103(3):478–484; discussion 485–476. [DOI] [PubMed] [Google Scholar]
- 11.Obermayer JL, Riley WT, Asif O, et al. College smoking-cessation using cell phone text messaging. J Am Coll Health. 2004;53(2):71–78. [DOI] [PubMed] [Google Scholar]
- 12.Riley W, Obermayer J, Jean-Mary J. Internet and mobile phone text messaging intervention for college smokers. J Am Coll Health. 2008;57(2):245–248. [DOI] [PubMed] [Google Scholar]
- 13.Buntin MB, Jain SH, Blumenthal D. Health information technology: laying the infrastructure for national health reform. Health Aff (Millwood). 2010;29(6):1214–1219. [DOI] [PubMed] [Google Scholar]
- 14.Cokkinides VE, Ward E, Jemal A, et al. Under-use of smoking-cessation treatments: results from the National Health Interview Survey, 2000. Am J Prev Med. 2005;28(1):119–122. [DOI] [PubMed] [Google Scholar]
- 15.Fiore MC, Jaen CR, Baker T, et al. Treating Tobacco Use and Dependence: 2008 Update Clinical Practice Guideline. U.S. Department of Health and Human Services; 2008. [Google Scholar]
- 16.Viswanath K, McCloud R, Minsky S, et al. Internet use, browsing, and the urban poor: implications for cancer control. Journal of the National Cancer Institute Monographs. 2013;2013(47):199–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Horvath T, Azman H, Kennedy GE, et al. Mobile phone text messaging for promoting adherence to antiretroviral therapy in patients with HIV infection. Cochrane Database Syst Rev. 2012;3:CD009756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Park LG, Howie-Esquivel J, Chung ML, et al. A text messaging intervention to promote medication adherence for patients with coronary heart disease: A randomized controlled trial. Patient Educ Couns. 2013. [DOI] [PubMed] [Google Scholar]
- 19.Montes JM, Medina E, Gomez-Beneyto M, et al. A short message service (SMS)-based strategy for enhancing adherence to antipsychotic medication in schizophrenia. Psychiatry Res. 2012;200(2–3):89–95. [DOI] [PubMed] [Google Scholar]
- 20.Castaño PM, Bynum JY, Andrés R, et al. Effect of daily text messages on oral contraceptive continuation: a randomized controlled trial. Obstet Gynecol. 2012; 119(1):14–20. [DOI] [PubMed] [Google Scholar]
- 21.Raupach T, Brown J, Herbec A, et al. A systematic review of studies assessing the association between adherence to smoking cessation medication and treatment success. Addiction. 2014;109(1):35–43. [DOI] [PubMed] [Google Scholar]
- 22.Shiffman S, Sweeney CT, Ferguson SG, et al. Relationship between adherence to daily nicotine patch use and treatment efficacy: secondary analysis of a 10-week randomized, double blind, placebo-controlled clinical trial simulating over-the-counter use in adult smokers. Clin Ther. 2008;30(10):1852–1858. [DOI] [PubMed] [Google Scholar]
- 23.Lam TH, Abdullah AS, Chan SS, et al. Adherence to nicotine replacement therapy versus quitting smoking among Chinese smokers: a preliminary investigation. Psychopharmacology (Berl). 2005;177(4):400–408. [DOI] [PubMed] [Google Scholar]
- 24.Catz SL, Jack LM, McClure JB, et al. Adherence to varenicline in the COMPASS smoking cessation intervention trial. Nicotine Tob Res. 2011;13(5):361–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Johnson RE, Stevens VJ, Hollis JF, et al. Nicotine chewing gum use in the outpatient care setting. J Fam Pract. 1992;34(1):61–65. [PubMed] [Google Scholar]
- 26.Bansal MA, Cummings KM, Hyland A, et al. Stop-smoking medications: who uses them, who misuses them, and who is misinformed about them? Nicotine Tob Res. 2004;6 Suppl 3:S303–310. [DOI] [PubMed] [Google Scholar]
- 27.Burns EK, Levinson AH. Discontinuation of nicotine replacement therapy among smoking-cessation attempters. Am J Prev Med. 2008;34(3):212–215. [DOI] [PubMed] [Google Scholar]
- 28.Wiggers LC, Smets EM, Oort FJ, et al. The effect of a minimal intervention strategy in addition to nicotine replacement therapy to support smoking cessation in cardiovascular outpatients: a randomized clinical trial. Eur J Cardiovasc Prev Rehabil. 2006;13(6):931–937. [DOI] [PubMed] [Google Scholar]
- 29.Ossip DJ, Abrams SM, Mahoney MC, et al. Adverse effects with use of nicotine replacement therapy among quitline clients. Nicotine Tob Res. 2009;11(4):408–417. [DOI] [PubMed] [Google Scholar]
- 30.Schmitz JM, Sayre SL, Stotts AL, et al. Medication compliance during a smoking cessation clinical trial: a brief intervention using MEMS feedback. J Behav Med. 2005;28(2):139–147. [DOI] [PubMed] [Google Scholar]
- 31.Pacek LR, McClernon FJ, Bosworth HB. Adherence to Pharmacological Smoking Cessation Interventions: A Literature Review and Synthesis of Correlates and Barriers. Nicotine Tob Res. 2018;20(10):1163–1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tseng TY, Krebs P, Schoenthaler A, et al. Combining Text Messaging and Telephone Counseling to Increase Varenicline Adherence and Smoking Abstinence Among Cigarette Smokers Living with HIV: A Randomized Controlled Study. AIDS Behav. 2017;21(7):1964–1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yingst JM, Veldheer S, Hrabovsky S, et al. Pilot Randomized Trial of an Automated Smoking Cessation Intervention via Mobile Phone Text Messages as an Adjunct to Varenicline in Primary Care. J Health Commun. 2018;23(4):370–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hammett E, Veldheer S, Hrabovsky S, et al. TXT2STAYQUIT: Pilot Randomized Trial of Brief Automated Smoking Cessation Texting Intervention for Inpatient Smokers Discharged from the Hospital. J Hosp Med. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cobos-Campos R, Apinaniz Fernandez de Larrinoa A, Saez de Lafuente Morinigo A, et al. Effectiveness of Text Messaging as an Adjuvant to Health Advice in Smoking Cessation Programs in Primary Care. A Randomized Clinical Trial. Nicotine Tob Res. 2017;19(8):901–907 [DOI] [PubMed] [Google Scholar]
- 36.Chow CK, Redfern J, Hillis GS, et al. Effect of Lifestyle-Focused Text Messaging on Risk Factor Modification in Patients With Coronary Heart Disease: A Randomized Clinical Trial. JAMA. 2015;314(12):1255–1263. [DOI] [PubMed] [Google Scholar]
- 37.Naughton F, Jamison J, Boase S, et al. Randomized controlled trial to assess the short term effectiveness of tailored web- and text-based facilitation of smoking cessation in primary care (iQuit in practice). Addiction. 2014;109(7):1184–1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fu SS, van Ryn M, Sherman SE, et al. Proactive tobacco treatment and population-level cessation: a pragmatic randomized clinical trial. JAMA Intern Med. 2014;174(5):671–677. [DOI] [PubMed] [Google Scholar]
- 39.Haas JS, Linder JA, Park ER, et al. Proactive tobacco cessation outreach to smokers of low socioeconomic status: a randomized clinical trial. JAMA Intern Med. 2015;175(2):218–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Murray RL, Coleman T, Antoniak M, et al. The effect of proactively identifying smokers and offering smoking cessation support in primary care populations: a cluster-randomized trial. Addiction. 2008;103(6):998–1006; discussion 1007–1008. [DOI] [PubMed] [Google Scholar]
- 41.Rigotti NA, Bitton A, Kelley JK, et al. Offering population-based tobacco treatment in a healthcare setting: a randomized controlled trial. Am J Prev Med. 2011;41(5):498–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rogers ES, Fu SS, Krebs P, et al. Proactive Tobacco Treatment for Smokers Using Veterans Administration Mental Health Clinics. Am J Prev Med. 2018;54(5):620–629. [DOI] [PubMed] [Google Scholar]
- 43.Vidrine DJ, Arduino RC, Lazev AB, et al. A randomized trial of a proactive cellular telephone intervention for smokers living with HIV/AIDS. AIDS. 2006;20(2):253–260. [DOI] [PubMed] [Google Scholar]
- 44.Cobos-Campos R, Apinaniz Fernandez de Larrinoa A, Saez de Lafuente Morinigo A, et al. Effectiveness of Text Messaging as an Adjuvant to Health Advice in Smoking Cessation Programs in Primary Care. A Randomized Clinical Trial. Nicotine Tob Res. 2016. [DOI] [PubMed] [Google Scholar]
- 45.Kruse GR, Chang Y, Kelley JH, et al. Healthcare system effects of pay-for-performance for smoking status documentation. Am J Manag Care. 2013;19(7):554–561. [PMC free article] [PubMed] [Google Scholar]
- 46.Colantonio LD, Pena L, Whittaker R, et al. Cross-Cultural Adaptation of a Text Message- Based Program for Smoking Cessation in Buenos Aires, Argentina. Nicotine Tob Res. 2016;18(3):314–320. [DOI] [PubMed] [Google Scholar]
- 47.McCool J, Tanielu H, Umali E, et al. Assessing the Cross-Cultural Adaptation and Translation of a Text-Based Mobile Smoking Cessation Program in Samoa (TXTTaofiTapaa): Pilot Study. JMIR mHealth and uHealth. 2018;6(8):e173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Atlas SJ, Chang Y, Lasko TA, et al. Is this “my” patient? Development and validation of a predictive model to link patients to primary care providers. J Gen Intern Med. 2006;21(9):973–978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kong G, Ells DM, Camenga DR, et al. Text messaging-based smoking cessation intervention: A narrative review. Addict Behav. 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kruse G, Kelley JH, Chase K, et al. Feasibility of a Proactive Text Messaging Intervention for Smokers in Community Health Centers: Pilot Study. JMIR Formativ Res. 2018;2(1):e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ferguson SG, Gitchell JG, Shiffman S, et al. Providing accurate safety information may increase a smoker's willingness to use nicotine replacement therapy as part of a quit attempt. Addict Behav. 2011;36(7):713–716. [DOI] [PubMed] [Google Scholar]
- 52.Shiffman S, Ferguson SG, Rohay J, et al. Perceived safety and efficacy of nicotine replacement therapies among US smokers and ex-smokers: relationship with use and compliance. Addiction. 2008;103(8):1371–1378. [DOI] [PubMed] [Google Scholar]
- 53.Fisher JD, Fisher WA, Amico KR, et al. An information-motivation-behavioral skills model of adherence to antiretroviral therapy. Health Psychol. 2006;25(4):462–473. [DOI] [PubMed] [Google Scholar]
- 54.Kruse G PE, Shahid NN, Abroms L, Haberer JE, Rigotti NA. Combining Real-Time Ratings With Qualitative Interviews to Develop a Smoking Cessation Text Messaging Program for Primary Care Patients. JMIR mHealth and uHealth. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cole-Lewis H, Augustson E, Sanders A, et al. Analysing user-reported data for enhancement of SmokefreeTXT: a national text message smoking cessation intervention. Tob Control. 2016. [DOI] [PubMed] [Google Scholar]
- 56.Carpenter MJ, Hughes JR, Gray KM, et al. Nicotine therapy sampling to induce quit attempts among smokers unmotivated to quit: a randomized clinical trial. Arch Intern Med. 2011;171(21):1901–1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Heatherton TF, Kozlowski LT, Frecker RC, et al. The Fagerstrom Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict. 1991;86(9):1119–1127. [DOI] [PubMed] [Google Scholar]
- 58.Centers for Disease Control and Prevention. Cigarette smoking among adults and trends in smoking cessation - United States, 2008. MMWRMorbidity and mortality weekly report. 2009;58(44):1227–1232. [PubMed] [Google Scholar]
- 59.Biener L, Abrams DB. The Contemplation Ladder: validation of a measure of readiness to consider smoking cessation. Health Psychol. 1991;10(5):360–365. [DOI] [PubMed] [Google Scholar]
- 60.Kroenke K, Spitzer RL, Williams JB, et al. An ultra-brief screening scale for anxiety and depression: the PHQ-4. Psychosomatics. 2009;50(6):613–621. [DOI] [PubMed] [Google Scholar]
- 61.Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single question alcohol screening test. J Gen Intern Med. 2009;24(7):783–788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170(13):1155–1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Humeniuk R, Ali R, Babor TF, et al. Validation of the Alcohol, Smoking And Substance Involvement Screening Test (ASSIST). Addiction. 2008;103(6):1039–1047. [DOI] [PubMed] [Google Scholar]
- 64.Carpenter MJ, Hughes JR. Defining quit attempts: what difference does a day make? Addiction. 2005;100(2):257–258. [DOI] [PubMed] [Google Scholar]
- 65.Verification SSoB. Biochemical verification of tobacco use and cessation. Nicotine Tob Res. 2002;4(2):149–159. [DOI] [PubMed] [Google Scholar]
- 66.Scott-Sheldon LA, Lantini R, Jennings EG, et al. Text Messaging-Based Interventions for Smoking Cessation: A Systematic Review and Meta-Analysis. JMIR mHealth and uHealth. 2016;4(2):e49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Spohr SA, Nandy R, Gandhiraj D, et al. Efficacy of SMS Text Message Interventions for Smoking Cessation: A Meta-Analysis. J Subst Abuse Treat. 2015;56:1–10. [DOI] [PubMed] [Google Scholar]
- 68.West R, Hajek P, Stead L, et al. Outcome criteria in smoking cessation trials: proposal for a common standard. Addiction. 2005;100(3):299–303. [DOI] [PubMed] [Google Scholar]
- 69.Pope C, Ziebland S, Mays N. Qualitative research in health care. Analysing qualitative data. BMJ. 2000;320(7227):114–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Thakkar J, Kurup R, Laba TL, et al. Mobile Telephone Text Messaging for Medication Adherence in Chronic Disease: A Meta-analysis. JAMA Intern Med. 2016;176(3):340–349. [DOI] [PubMed] [Google Scholar]
- 71.Free C, Knight R, Robertson S, et al. Smoking cessation support delivered via mobile phone text messaging (txt2stop): a single-blind, randomised trial. Lancet. 2011;378(9785):49–55. [DOI] [PMC free article] [PubMed] [Google Scholar]