Abstract
The levels of several GABAA receptor subunit mRNAs increase as cerebellar granule neurons migrate to their adult positions and receive excitatory mossy fiber input. Despite the temporal similarity of these increases in transcript expression in vivo, studies in cultured granule neurons demonstrated that the subunit mRNAs are differentially regulated. To address the possibility that neuronal activity regulates transcript expression, GABAA receptor subunit mRNA levels were assessed in cultured granule neurons grown in chemically defined, serum-free medium containing either nondepolarizing (5 mM) or depolarizing (25 mm) KCl concentrations. Whereas the δ subunit mRNA was almost undetectable in cultures maintained in nondepolarizing medium, an eightfold increase occurred between days 2 and 4 in cultures grown in depolarizing medium. Furthermore, δ subunit transcript expression was reduced by 76 ± 6% when neurons in depolarizing medium were switched into nondepolarizing medium. The importance of depolarization in the initiation and maintenance of subunit transcript expression in granule neurons was selective for the GABAA receptor δ subunit. These changes in transcript expression involved calcium entry through L-type calcium channels. Nifedipine treatment (1 μm) both reduced intracellular calcium and decreased δ subunit mRNA expression by 79 ± 4%. Furthermore, inhibition of Ca2+/calmodulin-dependent protein kinases (CaM kinases) by KN-62 (1 μm) also reduced δ subunit transcript expression. These studies demonstrate that KCl-induced depolarization, a condition that mimics the effects of neuronal activity, selectively modulates GABAA receptor δ subunit mRNA expression through a pathway involving calcium entry and activation of a CaM kinase.
Keywords: GABAA receptor, receptor subunit mRNAs, rat cerebellar granule neurons, membrane depolarization, neuronal activity, calcium influx, Ca2+/calmodulin-dependent protein kinase (CaM kinase)
The importance of neuronal activity in initiating and modulating neurotransmitter receptor expression in developing and adult animals has been reported for a number of systems. In the case of the nicotinic acetylcholine receptor (nAChR) at the neuromuscular junction, levels of the γ subunit mRNA decrease and expression of the ε subunit mRNA is initiated after innervation (Witzemann et al., 1989; Hall and Sanes, 1993). Similarly, the postnatal increase in nAChR α7 subunit mRNA in rat sympathetic neurons is largely innervation-dependent (Mandelzys et al., 1994), and expression of this subunit mRNA in culture requires depolarizing conditions (De Koninck and Cooper, 1995). In addition, selective modulation of levels of a subset of nAChRs in chick parasympathetic ciliary ganglion neurons coincides with presynaptic innervation (Levey et al., 1995).
The pattern of GABAA receptor subunit mRNA expression in cerebellar granule neurons during postnatal ontogeny raises the possibility that neuronal activity also plays a role in regulating this receptor system. Granule neurons express as many as 10 receptor subunit mRNAs, and levels of 6 of these transcripts increase dramatically during the second postnatal week when granule neurons migrate to their adult positions and receive afferent inputs. Whereas α1, β2, β3, and γ2 subunit transcripts are detectable at postnatal day 6 (P6), their levels continue to increase once the neurons reach adult positions and presumably receive afferent excitatory mossy fiber innervation (Gambarana et al., 1990, 1991; Laurie et al., 1992). In contrast, the α6 and δ subunit transcripts are absent at P6, but become detectable and continue to increase once granule neurons reside in their adult positions (Shivers et al., 1989; Laurie et al., 1992;Zheng et al., 1993; Varecka et al., 1994) (L. Gault and R. Siegel, unpublished observations). Together, these findings raise the possibility that mossy fiber innervation both initiates and modulates GABAA receptor subunit mRNA expression in cerebellar granule neurons.
Although the similarity in the time course of the increase in these six GABAA receptor subunit mRNAs in vivo suggests that their expression is governed by common cues, previous studies in cultured rat cerebellar granule neurons indicate that the mRNAs are differentially regulated (Beattie and Siegel, 1993; Behringer et al., 1996). Three distinct patterns of expression were observed. First, levels of the α1 and α6 subunit mRNAs remained constant when cultures are prepared at a relatively early stage (P2) or at a later stage of cerebellar maturation (P10). Second, the mRNAs encoding the β2, β3, and γ2 subunits were constant in cultures prepared at P2, P4, or P6, but increased severalfold in cultures prepared at P8 or P10. Finally, the δ subunit mRNA exhibited a unique pattern of expression; dramatic increases in transcript levels occurred in cultures prepared at both P2 and P10.
In the studies reported here, the possibility that neuronal depolarization influences one or more of these patterns was investigated. Although it has been suggested that depolarizing conditions can modulate levels of some GABAA receptor subunit transcripts (Zheng et al., 1994), interpretation of these previous studies is complicated by the presence of serum in the growth medium. To directly determine the importance of depolarization in subunit mRNA expression, we examined levels of the α1, α6, β2, γ2, and δ subunit mRNAs in a serum-free medium containing either nondepolarizing or depolarizing concentrations of KCl. These studies demonstrate that the δ subunit transcript, but not the other GABAA receptor subunit mRNAs, requires neuronal depolarization both to initiate and maintain expression. Furthermore, an elevation in intracellular calcium (Cai) and activation of a CaM kinase pathway mediate the increase in δ subunit mRNA expression.
MATERIALS AND METHODS
Cell culture. Cerebellar granule neurons were prepared for culture from Sprague Dawley rats (Zivic Miller, Zelienople, PA) at postnatal day 10 (P10) as described previously (Beattie and Siegel, 1993; Behringer et al., 1996). The dissociated cells were plated at a density of 2 × 103cells/mm2 on 24-well tissue culture plates coated with 0.1 mg/ml poly-l-lysine and 5 μg/ml laminin. All cultured cells were maintained in a chemically defined growth medium consisting of Neurobasal medium (Gibco-Bethesda Research Labs, Grand Island, NY) supplemented with B-27 (Gibco-Bethesda Research Labs), 6.0 g/l dextrose, 2 mm glutamine, 0.1 U/ml penicillin, and 0.1 μg/ml steptomycin.
In some cases, the defined medium contained 5 mM KCl (K5 medium), a concentration in the physiological range. Cultured granule neurons maintained in K5 medium adhered to the plates, elaborated processes, and survived for at least 4 d. This finding is consistent with findings reported by other groups using mediums containing 5 mM KCl (Gallo et al., 1987; Vallano et al., 1996). In studies to examine the effects of neuronal depolarization, the defined medium contained 25 mm KCl (K25 medium). Cultured granule neurons maintained in K25 medium exhibited a morphology similar to neurons maintained in K5 medium, but survived for at least 21 d. To examine the effects of serum components, K5 or K25 media were supplemented with 10% fetal bovine serum (FBS) (Biocell, Rancho Dominguez, CA). Cultures maintained in the serum-containing media exhibited a similar morphology and survival as neurons maintained in either media alone.
In every culture condition, nearly all cells exhibited the morphology of granule neurons, a finding consistent with previous reports (Beattie and Siegel, 1993; Behringer et al., 1996). Nonneuronal cell proliferation was suppressed by the addition of 60 μm5′-fluoro-2′-deoxyuridine (Sigma, St. Louis, MO) after 2 d. The cultures were fed after 4 d by replacing half of the medium in the wells with fresh growth medium.
The signaling cascades involved in controlling GABAAreceptor subunit mRNA expression were examined using two experimental paradigms. To determine the factors that initiate gene expression, channel blockers or inhibitors were added to the K25 medium at the time of plating. Cultures maintained in these agents were harvested after 4 d in culture. To determine the involvement of a signaling cascade in the maintenance of subunit mRNA expression, cultured granule neurons were grown for 7 d in K25 medium and then switched for 2 d into the same medium containing the indicated concentrations of inhibitors or blockers. The cultures were treated with the following agents at the indicated concentrations: 1 μm tetrodotoxin (TTX) (Sigma), 1 μm nifedipine (Sigma), 45 μm veratridine (K & K Laboratories, Plainview, NY), 1 μm KN-62 (LC Laboratories, Woburn, MA), 1 μm chelerythrine (chel) (LC Laboratories, Woburn, MA), and 1 μm H-89 (LC Laboratories, Woburn, MA). The concentrations of KN-62, chel, and H-89 used in these experiments were close to or in excess of their respective pKi values of 0.9 μm, 0.66 μm, or 0.048 μm(Chijiwa et al., 1990; Herbert et al., 1990; Tokumitsu et al., 1990). Stock solutions (1 or 10 mm) of each of these agents were prepared according to the manufacturers’ instructions in either K5 medium or dimethylsulfoxide (DMSO). The final concentration of DMSO in the medium was typically 0.1% and had no effect on neuronal morphology or survival.
Measurement of intracellular calcium. Changes in free Cai were evaluated using the fluorescent dye fura-2 essentially as described by Grynkiewicz et al. (1985). For these studies, cultured granule neurons were grown on 24 mm acid-washed No. 1 glass coverslips (Fisher, Pittsburgh, PA). The coverslips were glued over a 20 mm hole in the bottom of a 35 mm tissue culture plate using SILASTIC medical adhesive (Dow Corning, Midland, MI). The dishes were UV-irradiated and prepared for culture as described above. Cultured granule neurons maintained for 2 d in K5 medium, K25 medium, or K25 medium containing 1 μm nifedipine were incubated in the same medium with 4 μm fura-2 AM (Molecular Probes, Eugene, OR) for 20 min at 37°C. The cultures were then rinsed three times with fresh medium and incubated an additional 20 min at 37°C. Individual fields were examined at 37°C by phase-contrast microscopy (Zeiss Axiovert 405M, Thornwood, NY) using an oil-immersion 100× Plan-Neofluor objective. The fluorescence intensity at 510 nm was measured with excitation at 350 nm and 380 nm at 5 sec intervals. Images were collected with Image-1 software (Universal Imaging, West Chester, PA) and stored on a Panasonic optical disk for later analysis.
For each experimental condition, images were collected at the indicated excitation wavelengths from at least four separate fields from two or more separate platings. All bipolar cells that morphologically resembled granule neurons were used for analysis, whereas cells that were obviously larger than granule neurons or irregularly shaped were excluded. Typically, only about 10% of the cell population was excluded from the analysis using these criteria. For each plate, background fluorescence was recorded for both excitation wavelengths at the same focal plane from a field that lacked cells. After background subtraction at both excitation wavelengths, the ratio of fluorescence intensity for excitation at 350 nm and 380 nm was calculated. The fluorescence ratio values were determined for the area just inside the boundaries of the cell body, and four separate readings were averaged for each neuron. Higher fluorescence ratio values represent higher levels of Cai. Because the distribution of ratio values was skewed toward high values, the nonparametric Kolmogorov-Smirnov test was used to determine statistical significance.
RNA isolation and PCR. Relative levels of GABAAreceptor subunit mRNAs expressed over time in culture were assessed using a semiquantitative RT-PCR protocol. Total cellular RNA was isolated from 2-4 culture wells by the procedure of Chomczynski and Sacchi (1987) and RT-PCR performed essentially as described previously (Beattie and Siegel, 1993; Behringer et al., 1996). A baseline measurement of GABAA receptor subunit mRNA levels was determined by harvesting some of the dissociated neurons in guanidine thiocyanate at the time of plating. For RT-PCR analysis, 0.2 μg of RNA was DNase treated and reverse transcribed. Each reaction contained 75 pg of exogenous SP64 bacterial RNA transcribed from the SP64 plasmid (Promega, Madison, WI) to control for variability between samples. PCR buffer containing [32P]dCTP was added to the reverse transcriptase reaction, layered with mineral oil and subjected to 30 cycles of 95°C for 1 min, 55°C for 30 sec, and 72°C for 45 sec in a Perkin Elmer Cetus 480 (Foster City, CA) thermocycler. The number of PCR cycles for each primer used was in the exponential phase of amplification. PCR products were separated in an 8% nondenaturing polyacrylamide gel and detected by autoradiography. The amount of32P incorporated into each PCR product was quantitated by excision and counting of the bands after gel drying. Near each PCR product band, areas of the gel without detectable bands were also excised and counted. This background count was subtracted from the count obtained for each subunit PCR product. Results for each reaction were expressed as a ratio of GABAA receptor subunit PCR product to SP64 PCR product. Similar patterns of subunit mRNA expression were observed when elongation factor 1α, an endogenous transcript, was used instead of the exogenous SP64 RNA as a control for sample variability in the RT-PCR assay (K. Behringer and R. Siegel, unpublished observations).
GABAA receptor subunit primers were generated to regions of the cDNAs where the sequence diversity is greatest among subunits. The primer sequences are listed from 5′ to 3′ with (+) being complementary to the noncoding strand and (−) being complementary to the coding strand: α1(+) = 1410–1429 and α1(−) = 1501–1520 (Khrestchatisky et al., 1989); α6(+) = 1369–1384, α6(−) = 1450–1475 (Lüddens et al., 1990); β2(+) = 1808–1831 and β2(−) = 1882–1905 (Ymer et al., 1989); γ2(+) = 1744–1768 and γ2(−) = 1807–1829; and δ(+) = 394–417 and δ(−) = 458–481 (Shivers et al., 1989). In addition, the following primers for the SP64 plasmid (Promega) were added to each reaction: SP64(+) = 234–254 and SP64(−) = 344–364. The G+C content of each primer was ∼50%. The specificity of the subunit primers was confirmed by sequence analysis of the PCR products (Beattie and Siegel, 1993; Behringer et al., 1996).
Statistical analyses. Two methodologies were used to determine the statistical significance of the data. In instances where relative subunit mRNA levels are reported as ratio values, statistical significance was determined using a paired, two-tailed Student’st test. When the data are reported as a percentage of control values, a 95% confidence interval was constructed. The result was considered significant if the interval excluded 100%.
RESULTS
The initiation of δ subunit mRNA expression has a unique requirement for depolarizing conditions
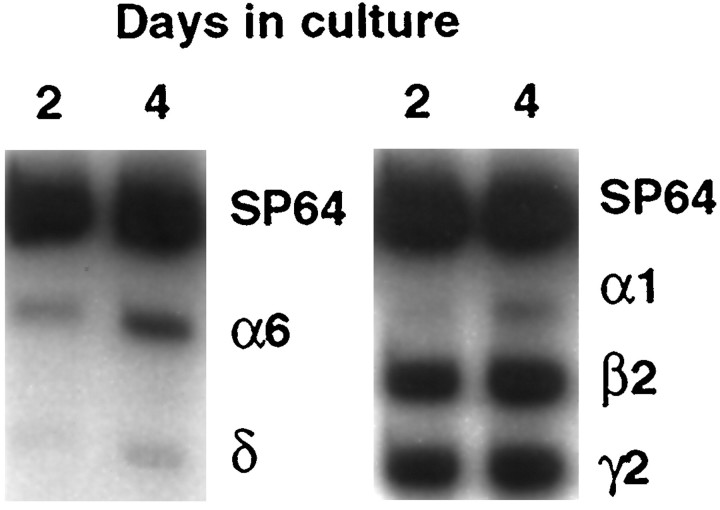
Previous studies suggested that increases in GABAAreceptor subunit mRNA expression in cerebellar granule neurons coincide with migration to the internal granule cell layer and synapse formation with afferent fibers (Gambarana et al., 1990, 1991; Laurie et al., 1992). To test the possibility that neuronal activity plays a role in initiating subunit mRNA expression, cultured granule neurons were grown in a chemically defined, serum-free medium containing either a nondepolarizing (5 mm; K5 medium) or depolarizing (25 mm; K25 medium) concentration of potassium chloride (KCl). In the nondepolarizing K5 medium, the mRNAs encoding the α1, α6, β2, and γ2 subunits were all easily detectable at 2 or 4 d in culture (Fig. 1). In contrast, the δ subunit transcript was barely detectable at these times; in fact, its level was not significantly different from background (p> 0.2). These data suggest that nondepolarizing conditions are sufficient to initiate the expression of the α1, α6, β2, and γ2 subunit mRNAs, but not the δ subunit mRNA. These findings are consistent with previous observations suggesting that expression of the δ subunit transcript is regulated differently from that of other GABAA receptor subunit mRNAs (Behringer et al., 1996).
Fig. 1.
The δ subunit mRNA is almost undetectable in nondepolarizing K5 medium. Autoradiograph of a representative RT-PCR experiment showing GABAA receptor subunit mRNA expression in cultured granule neurons grown in K5 medium for 2 or 4 d in culture. The mRNAs encoding the α1, α6, β2, and γ2 subunits are detectable, whereas the mRNA encoding the δ subunit is almost undetectable. Similar results were obtained in more than six experiments.
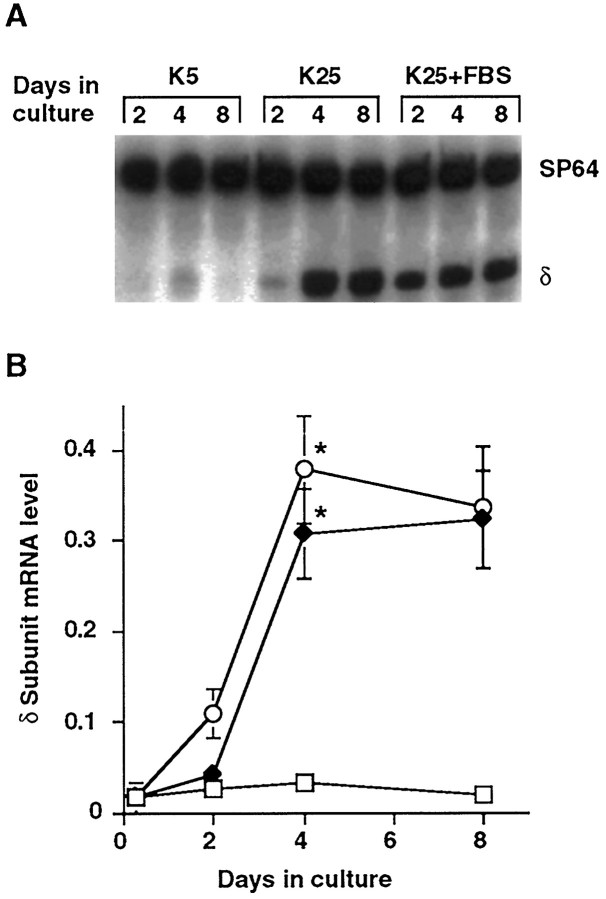
To examine whether neuronal depolarization initiates δ subunit mRNA expression, levels of this transcript were measured in cultured granule neurons maintained in the depolarizing K25 medium. Whereas δ subunit transcript expression was extremely low after 2 d in culture, its level increased more than 10-fold between 2 and 4 d in the depolarizing condition (p < 0.05; Fig.2A,B). Furthermore, the addition of another depolarizing agent, veratridine (45 μm), to K5 medium also induced an increase in δ subunit transcript expression. The time course and magnitude of the increase in the presence of veratridine were comparable to those seen in cultures maintained in K25 medium (data not shown).
Fig. 2.
δ subunit mRNA expression in K25 medium is comparable to that observed in K25 + FBS. A, Representative autoradiograph from a RT-PCR experiment showing δ subunit mRNA expression in cultured granule neurons maintained in either nondepolarizing K5 medium, depolarizing K25 medium or K25 medium containing 10% FBS. B, Quantitative analysis of δ subunit mRNA expression in cultured granule neurons maintained in either K5 medium (□), K25 medium (⧫), or K25 medium with 10% FBS (○). RNA was isolated at the designated times from cultures maintained in each condition. δ subunit mRNA levels were assessed by RT-PCR and data were plotted as a ratio of δ subunit PCR product to SP64 PCR product versus time in culture as described in Materials and Methods. Eachpoint represents the mean ± SEM of three to five separate experiments. *p < 0.05, in comparison with subunit mRNA level at 2 d in culture.
The increase in δ subunit mRNA levels in K25 medium is similar to that previously observed in cultured granule neurons maintained in medium containing 10% FBS in addition to a depolarizing KCl concentration (Zheng et al., 1994; Behringer et al., 1996). To determine whether factors present in serum could also initiate or modulate δ subunit mRNA expression, levels of this subunit mRNA were assessed in cultures maintained in K5 or K25 medium containing 10% FBS. In both K5 and K5 + FBS media, the δ subunit mRNA was barely detectable (data not shown). In addition, the time course and magnitude of δ subunit transcript expression in the K25 medium was not altered by the addition of 10% FBS (Fig. 2A,B). These results suggest that factors present in serum are neither necessary nor sufficient to initiate an increase in δ subunit mRNA expression.
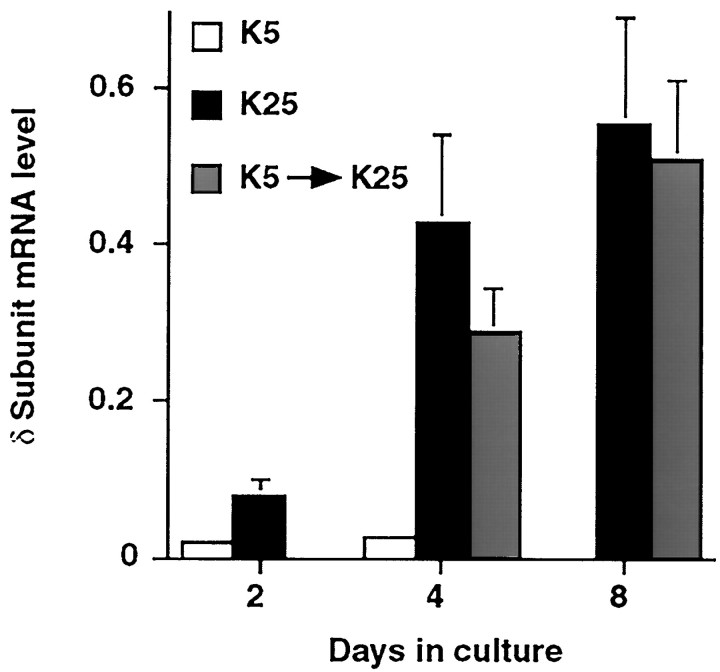
Although cultured granule neurons maintained in K5 medium are initially morphologically similar to those grown in K25 medium, neurons in the K5 medium exhibit fewer markers of differentiation and a decrease in long-term survival (Gallo et al., 1987; Balazs et al., 1988; Vallano et al., 1996). To demonstrate that the absence of δ subunit transcript expression in the nondepolarizing medium is not a result of a permanent alteration in phenotype or a nonspecific effect on neuronal viability, δ subunit mRNA expression was measured in neurons maintained for 2 d in K5 medium and switched to K25 medium for the remainder of the culture period. In these cultures, the level of the δ subunit transcript was comparable to control cultures at 3, 6, and 24 h after the change in medium (data not shown). By 2 d after the switch in medium, the level of the δ subunit transcript was sevenfold higher than in cultures maintained in K5 medium, but it had not yet attained the level expressed in cultures maintained in K25 medium (Fig.3). After 6 d, the level of this transcript was comparable to that observed in neurons maintained in depolarizing medium for the duration of the culture period. These studies suggest that neurons grown in nondepolarizing medium retain the ability to express the δ subunit mRNA. Moreover, the presence of KCl-induced depolarization alone is sufficient to initiate an increase in δ subunit transcript expression.
Fig. 3.
KCl-induced depolarization initiates an increase in δ subunit mRNA expression. δ subunit mRNA expression was analyzed in cultures maintained for 2 days in K5 medium and subsequently switched to K25 medium (hatched bars). Levels of the δ subunit mRNA were compared with those obtained in cultures maintained solely in K5 (open bars) or K25 medium (filled bars). Each point represents the mean ± SEM of three separate experiments.
Continued KCl-induced depolarization is required to maintain elevated levels of δ subunit mRNA expression
To determine if KCl-induced depolarization is also required to maintain expression of the δ subunit transcript, the level of the δ subunit mRNA was assessed in cultured granule neurons maintained for 8 d in K25 medium and subsequently switched to K5 medium for 2 d (Fig. 4A,B). The level of the δ subunit transcript in switched cultures was 24 ± 6% of that observed in control cultures maintained in K25 medium (p < 0.05). The effect of the switch in medium was specific for the δ subunit mRNA. Levels of the α1, β2, and γ2 subunit mRNAs were unaffected by the switch in medium. Although the level of the α6 subunit mRNA was 70 ± 6% (p < 0.05), the magnitude of this reduction was much smaller than that seen for the δ subunit mRNA. These results suggest that cultured granule neurons require the continued presence of depolarizing conditions to maintain expression of the δ subunit mRNA.
Fig. 4.
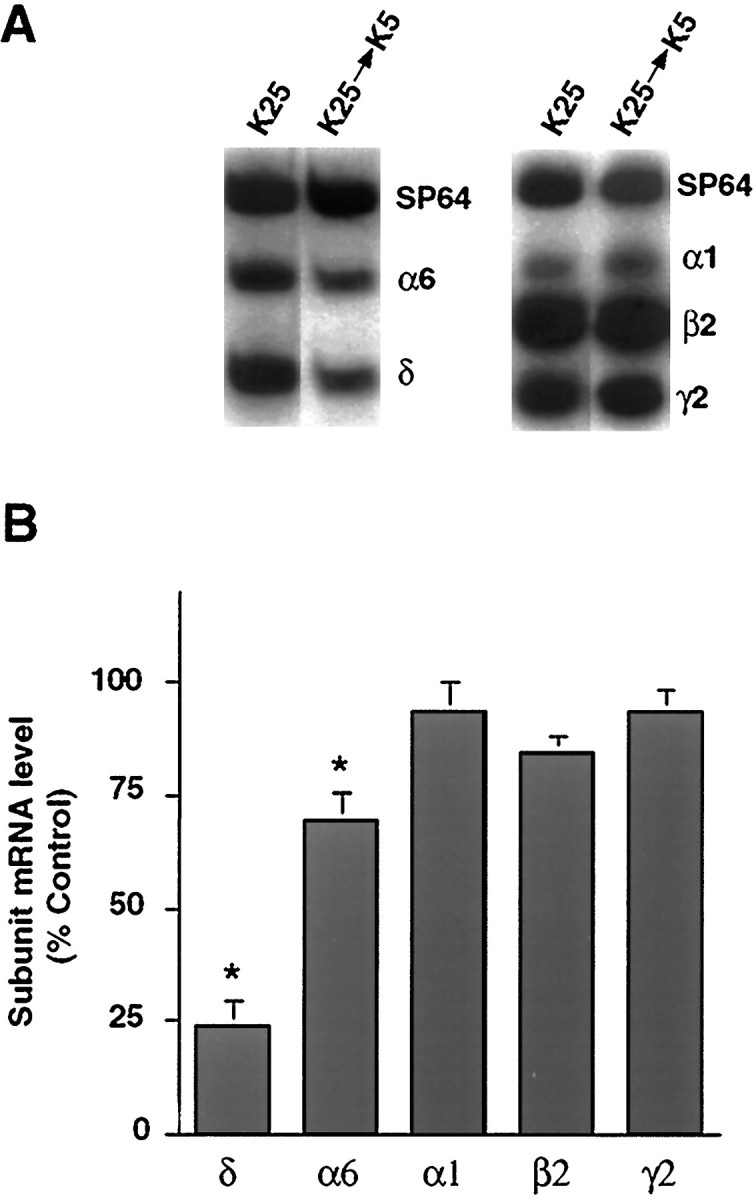
Depolarizing conditions are required to maintain δ subunit mRNA expression. A, A representative autoradiograph of subunit mRNA expression in cultures maintained for 7 d in K25 medium and then switched for 2 d to K5 medium. Levels of expression were compared with control cultures maintained in K25 medium for 9 d. The levels of the δ, and to a lesser extent the α6, subunit mRNAs decrease when cultures are switched from K25 to K5 medium, but levels of the α1, β2, and γ2 subunit mRNAs are not affected. B, Quantitative analysis of subunit mRNA levels. Levels of each transcript in the switched cultures were expressed as a percentage of the subunit mRNA levels in control cultures maintained in K25 medium. *p < 0.05. Results represent the mean ± SEM of three separate experiments.
Calcium entry is required to initiate and maintain depolarization-induced changes in δ subunit mRNA expression
Changes in δ subunit mRNA expression in depolarizing conditions may result from an alteration in ion fluxes. In particular, maintenance of neurons in depolarizing conditions has been shown to affect gene expression by altering the influx of sodium or calcium (Gallo et al., 1987; Sun et al., 1992). The possibility that sodium entry is involved in initiating δ subunit mRNA expression was examined by the addition of 1 μm TTX to K25 medium at the time of plating (Fig.5A,B). The levels of both the δ and α6 subunit mRNAs at day 4 in culture were unaffected by this treatment, suggesting that sodium entry through TTX-sensitive channels does not influence expression of these two subunit mRNAs.
Fig. 5.
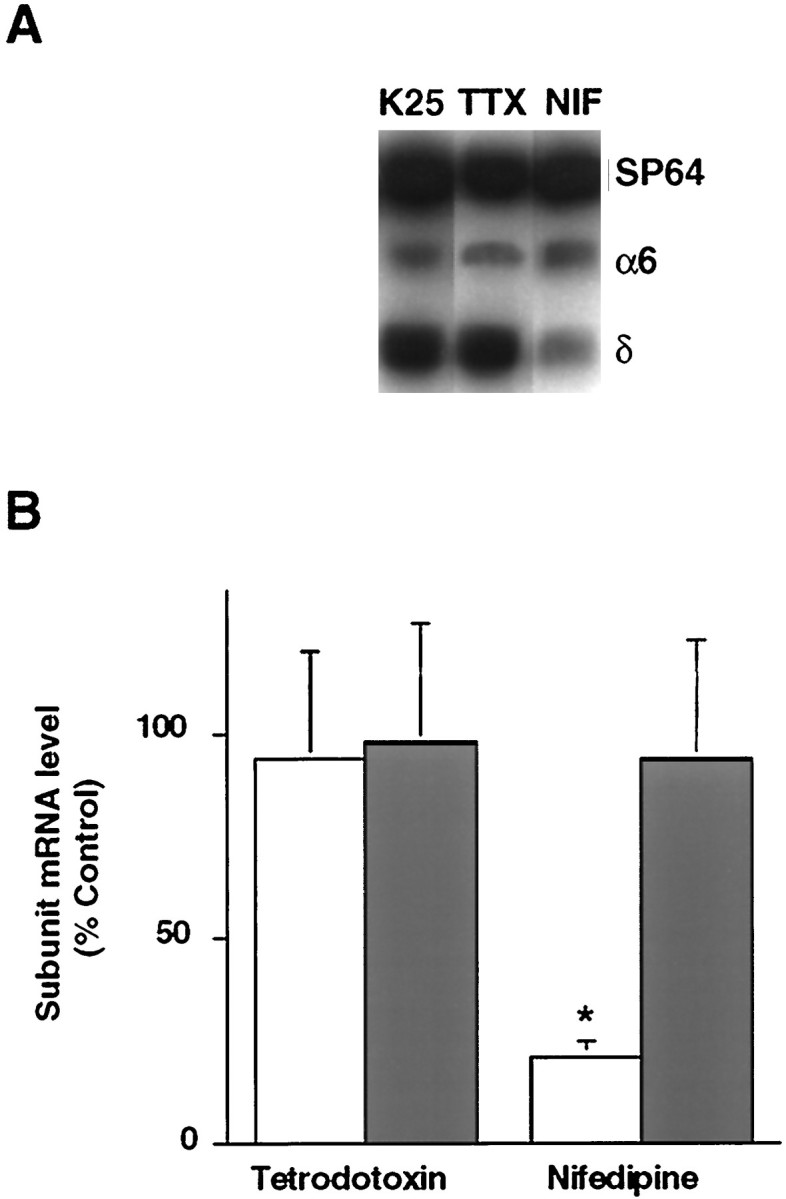
δ subunit mRNA expression depends on calcium influx through L-type calcium channels. Cultured granule neurons were maintained for 4 d in culture in K25 medium containing either 1 μm nifedipine or 1 μm TTX. A, Representative autoradiograph from a RT-PCR experiment showing δ and α6 subunit mRNA levels in cultures maintained for 4 d in either K25 medium, or K25 medium containing 1 μm nifedipine or 1 μm TTX. B, Quantitative analysis of subunit mRNA levels. Levels of the δ (open bars) and α6 (filled bars) subunit mRNAs in the nifedipine- or TTX-treated cultures were expressed as a percentage of the subunit mRNA levels in control cultures maintained in K25 medium and represent the mean ± SEM of three separate experiments. *p < 0.05.
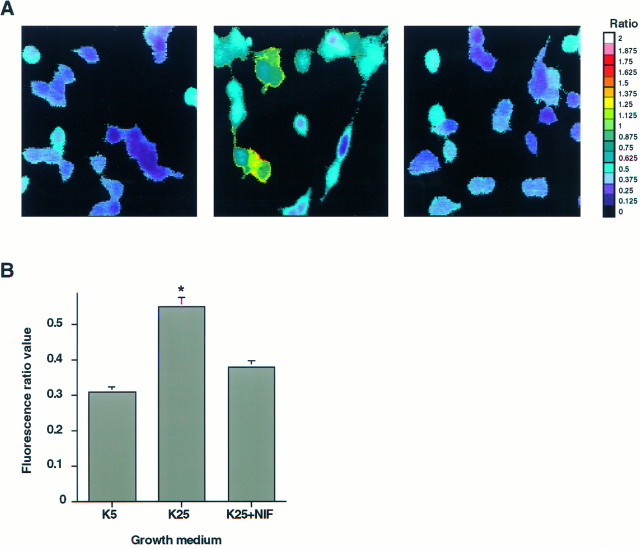
Alternatively, depolarization-induced increases in Cai may play a role in regulating δ subunit mRNA expression. To investigate this possibility, relative levels of free Cai in cultured granule neurons maintained for 2 d in depolarizing and nondepolarizing conditions were compared using the fluorescent calcium indicator, fura-2. These studies demonstrated that the fluorescence ratio value of neurons maintained in K25 medium was significantly greater (p < 0.05) than those in K5 medium (Fig. 6A,B). The average fluorescence ratio value of neurons maintained in K25 medium was 0.54 ± 0.01, as compared with 0.31 ± 0.01 for neurons maintained in K5 medium.
Fig. 6.
Cultured granule neurons maintained in depolarizing K25 medium exhibit a persistent elevation in free Cai. A, A representative field of fluorescence ratio measurements is shown for cultured granule neurons maintained for 2 d in culture in K5 medium (left), K25 medium (middle), or K25 medium (right) containing 1 μm nifedipine. Similar results were obtained in at least four other fields for each condition in two separate preparations.B, Quantitative analysis of fluorescence ratio values from neurons maintained in K5 medium (n = 257 neurons), K25 medium (n = 245 neurons), or K25 medium containing 1 μm nifedipine (n = 119 neurons). Eachpoint represents the mean ± SEM. *p < 0.05.
The elevation of Cai in cultured granule neurons maintained in depolarizing conditions may reflect calcium entry through a number of voltage-sensitive and ligand-gated channels as well as calcium release from internal stores. Calcium entry through L-type calcium channels occurs in cultured granule neurons (Amico et al., 1995;Randall and Tsien, 1995) and has been implicated in changes in gene expression (Gallo et al., 1987; Murphy et al., 1991). To test the importance of calcium entry through L-type calcium channels in regulating δ subunit mRNA expression, depolarization-induced changes in relative levels of free Cai and the δ subunit transcript were measured in the presence of the L-type calcium channel blocker, nifedipine. When 1 μm nifedipine was added to the K25 medium at the time of plating, the average fluorescence ratio value (0.38 ± 0.01) more closely resembled that of neurons maintained in K5 rather than K25 medium (Fig. 6A,B). A corresponding decrease in δ subunit mRNA levels was observed in cultured granule neurons maintained in K25 medium containing 1 μm nifedipine (Fig. 5). In neurons maintained for 4 d in this condition, the level of the δ subunit transcript was only about 21 ± 4% of that attained in neurons maintained in K25 medium alone (p < 0.05). In contrast, levels of the α6 subunit mRNA were unaffected by nifedipine.
Additional studies demonstrated that calcium influx through L-type calcium channels is required not only to initiate expression of the δ subunit mRNA, but also to maintain its expression. A 61 ± 7% reduction (p < 0.05) in δ subunit mRNA expression was noted when cultured granule neurons were maintained for 7 d in K25 medium and subsequently switched to K25 medium containing 1 μm nifedipine (data not shown). Together, these results suggest that calcium entry through L-type calcium channels during KCl-induced depolarization is largely responsible for the initiation and maintenance of δ subunit mRNA expression.
Activation of a CaM kinase pathway is required to initiate and maintain δ subunit mRNA expression
To elucidate the intracellular signaling cascades involved in mediating the increase in δ subunit mRNA expression, cells grown in depolarizing medium were treated with specific kinase inhibitors. When 1 μm KN-62, a specific inhibitor of Ca2+/calmodulin-dependent protein kinases (Tokumitsu et al., 1990) was added to K25 medium at the time of plating, a 64 ± 9% reduction (p < 0.05) in δ subunit mRNA expression was measured 4 d later (data not shown). These results suggest that activation of a CaM kinase pathway is involved in initiation of δ subunit mRNA expression.
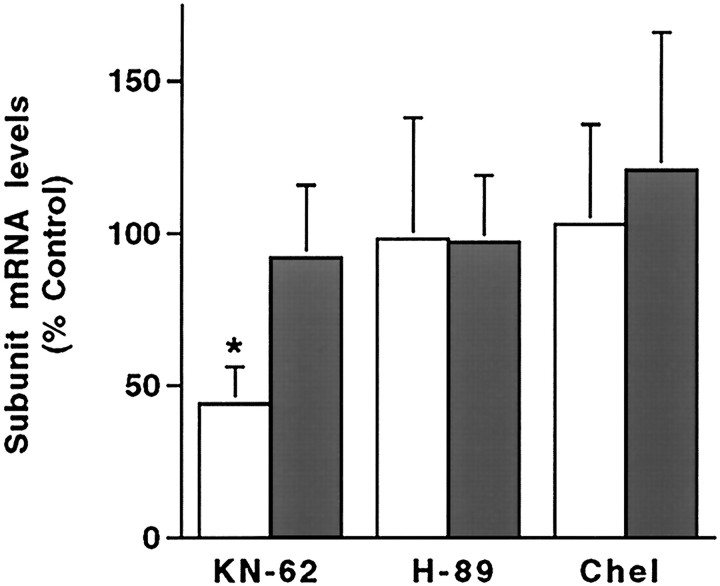
To determine if the same signaling cascade is involved in maintenance of δ subunit mRNA expression, cells grown for 7 d in K25 medium were treated with KN-62 for 2 d (Fig. 7). Inhibition of the CaM kinase pathway with 1 μm KN-62 resulted in a 55 ± 11% decrease (p < 0.05) in the amount of δ subunit transcript detected in these cultured neurons. In contrast, levels of the α6 subunit transcript were unaffected by the addition of KN-62. Furthermore, a decrease in δ subunit mRNA expression was specific for inhibition of the CaM kinase pathway. Levels of both the δ and α6 subunit mRNAs were not changed after the addition of 1 μm chel to inhibit the PKC pathway or 1 μm H-89 to inhibit the PKA pathway. Even 5- to 10-fold higher concentrations of these kinase inhibitors failed to produce changes in levels of the δ subunit mRNA (data not shown). Together, these data suggest that activation of a CaM kinase pathway by KCl-induced depolarization is required both to initiate and maintain δ subunit mRNA expression.
Fig. 7.
Maintenance of δ subunit mRNA expression depends on activation of a CaM kinase pathway. The levels of the δ (open bars) and α6 (filled bars) subunit mRNAs were determined in cultured granule neurons maintained for 7 days in K25 medium then switched for 2 d into K25 medium containing 1 μm KN-62, 1 μm H-89, or 1 μmchel. Quantitative analysis of subunit mRNA levels. Values in the treated cultures were expressed as a percentage of the subunit mRNA levels in control cultures maintained in K25 medium and represent the mean ± SEM of five separate experiments. *p < 0.05.
Although some previous studies have suggested that KN-62 blocks L-type calcium channels (Li et al., 1992), others have reported that KN-62 does not block these channels or alter calcium flux (Hack et al., 1993;De Koninck and Cooper, 1995). To determine whether KN-62 treatment alters Cai, relative levels of free Cai were measured in cultured granule neurons maintained for 2 d in K25 medium containing 1 μm KN-62. The average fluorescence ratio value was 0.47 ± 0.01 in treated cultures, a value that is not significantly different from that found in neurons maintained in K25 medium alone (p > 0.5). Consistent with this finding, Hack et al. (1993) reported that45Ca2+ influx through voltage-sensitive or NMDA-gated channels in cultured cerebellar granule neurons was not altered by KN-62. Thus, the reduction in δ subunit mRNA expression on addition of KN-62 results from an inhibition of a CaM kinase pathway rather than an alteration in free Cai.
DISCUSSION
These studies demonstrate that unique signals are required to initiate and maintain expression of the GABAA receptor δ subunit mRNA in cultured cerebellar granule neurons. Whereas expression of several other GABAA receptor subunit transcripts was initiated in nondepolarizing, serum-free medium, the δ subunit mRNA required depolarizing conditions. This selective regulation of the δ subunit mRNA is consistent with our previous report that this subunit transcript exhibited a unique pattern of expression in cultured granule neurons (Behringer et al., 1996). In that study, levels of the δ subunit mRNA increased severalfold in cultures prepared at both immature (P2) and more mature (P10) stages of cerebellar maturation. The increase in δ subunit mRNA expression in cultures prepared at P2 occurs before the observed increase in vivo, raising the possibility that expression of this subunit transcript is prevented at inappropriate times in vivo by an inhibitory cue absent in culture. Alternatively, an inductive factor may be provided in culture. Because this study demonstrates that the δ subunit mRNA was barely detectable and did not increase in K5 medium, the first possibility seems unlikely. Instead, the increase in δ subunit mRNA expression observed in culture occurs in response to a regulatory cue such as KCl-induced depolarization. Whether this observed increase in expression is caused by increased transcription or stability of the δ subunit mRNA remains to be investigated. In addition, it is not yet known whether the level of the δ subunit polypeptide changes in parallel with its mRNA.
Previous studies to examine conditions that modulate, rather than initiate, expression of GABAA receptor subunit mRNAs and other receptor subunit transcripts in the rat cerebellum have also suggested that neuronal depolarization plays a role. In one report, the levels of GABAA receptor α1 and α5 subunit mRNAs were higher in cultured granule neurons maintained in 25 mm KCl than in 12.5 mm KCl (Harris et al., 1994). Furthermore, exposure of cultured granule neurons to depolarizing conditions induced an increase in the NMDA receptor NR2A and a concomitant decrease in the NR2B subunit mRNAs (Bessho et al., 1994; Resink et al., 1995; Vallano et al., 1996). Changes in levels of these subunit mRNAs in response to depolarizing stimuli paralleled those observed during granule neuron development in situ (Akazawa et al., 1994). These findings, in conjunction with the results reported here, suggest that neuronal activity initiates and modulates expression of more than one receptor phenotype during cerebellar differentiation.
Because cultured granule neurons maintained in nondepolarizing medium appear less differentiated than those in depolarizing conditions (Balazs et al., 1988), the importance of cellular maturation in the onset of δ subunit expression was examined. Our studies show that the absence of δ subunit mRNA expression in K5 medium does not result from an irreversible effect on differentiation or a nonspecific effect on cell health. In fact, the maximal level of the δ subunit mRNA in cultures switched from K5 to K25 medium was indistinguishable from that in cultures maintained in K25 medium for the duration of the experiment. Moreover, even after the neurons have undergone extensive KCl-induced differentiation in culture, they retain the requirement for depolarization to maintain δ subunit mRNA expression. Thus, the effect of KCl-induced depolarization may be to activate specific intracellular signaling pathways that control δ subunit transcript expression rather than to promote neuronal differentiation.
Our studies suggest that one step in the pathway mediating δ subunit transcript expression results from a depolarization-induced elevation in Cai. A reduction in Cai in nifedipine-treated cultures is accompanied by a lower level of δ subunit mRNA expression, suggesting that calcium entry through L-type calcium channels plays a major role in controlling δ subunit transcript expression. Recent studies have indicated that distinct intracellular signaling cascades are activated in response to calcium influx through different channel types (Gallin and Greenberg, 1995;Ghosh and Greenberg, 1995). Although our results suggest that calcium entry through L-type calcium channels is important, it is unlikely that granule neurons possess an absolute dependence on this route of entry. In fact, the sustained, large elevation in Cai observed in depolarizing medium is probably sufficient to activate many calcium-dependent processes, eliminating the ability of the neuron to distinguish a specific pathway of calcium entry. Furthermore, because the Cai and δ subunit mRNA levels in nifedipine-treated cultures are still somewhat greater than those observed in K5 medium, the possibility that δ subunit transcript expression involves other calcium-dependent or -independent mechanisms cannot be eliminated.
Although we have identified several crucial steps involved in δ subunit mRNA expression, the exact sequence of events remains unknown. Neuronal depolarization, an elevation in free Cai and activation of a CaM kinase pathway all play a role in initiation and maintenance of δ subunit mRNA expression. The mechanism by which CaM kinase activation elevates δ subunit mRNA levels may occur through translocation to the nucleus (Jensen et al., 1991; Srinivasan et al., 1994) and activation of transcription or more indirectly by phosphorylation of transcription factors (Wegner et al., 1992). Alternatively, CaM kinase activation may affect immediate early genes (Bading et al., 1993; Enslen and Soderling, 1994; Ghosh and Greenberg, 1995) and initiate a series of events that ultimately includes transcription of the δ subunit mRNA. In support of this latter possibility, the δ subunit gene promoter contains several AP1 binding elements that may be involved in mediating this effect (Motejlek et al., 1994). In any event, because the increase in δ subunit transcript levels in depolarizing conditions requires several days, a cascade of events, possibly including de novo protein synthesis, is involved. Whether each of these crucial events occurs in series and what other regulatory molecules are involved remains to be elucidated.
Although this study has indicated that CaM kinase activation is required to initiate and maintain δ subunit mRNA expression, it has not defined which CaM kinase isoform mediates these effects. CaM KII and CaM KIV are expressed abundantly in cerebellar granule neurons and both are inhibited by the concentration of KN-62 used in these studies (Tokumitsu et al., 1990; Enslen et al., 1994). Because CaM KII is a ubiquitous kinase involved in many cellular processes (Hanson and Schulman, 1992; Schulman, 1993), it would be difficult to rule out its involvement in δ subunit mRNA expression. On the other hand, because CaM KIV is first detectable in the internal granule cell layer of the cerebellum just before the expression of the δ subunit mRNA (Ohmstede et al., 1989; Jensen et al., 1991), it may also be involved in this process. Furthermore, the spatial expression pattern of CaM KIV in the CNS (Ohmstede et al., 1989) largely overlaps that of the δ subunit mRNA (Shivers et al., 1989).
These studies have identified a crucial role of neuronal depolarization in the initiation and maintenance of δ subunit mRNA expression, but the signal(s) that regulate expression of other GABAAreceptor subunit mRNAs in cerebellar granule neurons remain largely unknown. That the α1, α6, β2, and γ2 subunit mRNAs are detectable in nondepolarizing K5 medium suggests that initiation of expression for these subunit mRNAs can occur in the absence of depolarizing conditions or serum factors. It is likely, however, that levels of these subunit mRNAs are regulated in response to local environmental cues including neurotransmitters, neuropeptides, or neurotrophins. In fact, several studies have suggested that glutamate and GABA are involved in the modulation of other GABAAreceptor subunit mRNA levels (Memo et al., 1991; Kim et al., 1993;Harris et al., 1994). The involvement of a cAMP signaling pathway in the regulation of α1 and α6 subunit polypeptide expression has also recently been reported (Thompson et al., 1996), but a physiological activator of this pathway has not yet been identified. Further studies using the defined medium system may demonstrate a role for one or more of these factors in the regulation of GABAA receptor subunit mRNA expression.
The apparent requirement of cultured granule neurons for depolarizing conditions to express the δ subunit mRNA suggests that neuronal depolarization after synaptic activity in vivo initiates transcript expression. Initiation of δ subunit mRNA expression occursin vivo by postnatal day 12, when the granule neurons have migrated to adult positions in the internal granule cell layer (Shivers et al., 1989; Laurie et al., 1992) and have presumably formed synaptic contact with afferent glutamatergic mossy fibers (Altman, 1972). Thus, activation of ionotropic glutamate receptors on the granule neurons by glutamate released from mossy fiber afferents may contribute to the elevation in Cai that is a crucial step in δ subunit mRNA expression.
In addition to the initiation of δ subunit mRNA expression after innervation, modulation of its expression may also occur in response to excitatory mossy fiber activity. Thus, neurons could integrate a pattern of ongoing synaptic activity and modify expression of this subunit mRNA. Earlier studies have suggested that the δ subunit mRNA is expressed primarily in small interneurons that limit the spread of excitatory impulses (Shivers et al., 1989) and that receptors containing the δ subunit mRNA display high affinity GABA (Benke et al., 1991) and muscimol binding (Quirk et al., 1995). A selective alteration in δ subunit mRNA and/or polypeptide levels in response to neuronal activity may serve a homeostatic function to limit the spread of excitatory impulses. Additional studies in both cultured granule neurons and the intact cerebellum are necessary to investigate the role of synaptic activity in the regulation of δ subunit mRNA expression.
Footnotes
This work was supported by grants from National Institutes of Health (NIH) (NS31266 and NS34317) to R.E.S. L.M.G. was supported in part by an NIH Medical Scientist Training Program grant (5-T32GM07250-21). We thank Drs. David Friel and Susan Burden-Gulley for assistance with calcium imaging, Dr. Hegang Chen for advice concerning the statistical analyses, and Drs. Evan Deneris, David Friel, and Bryan Roth for helpful comments on the manuscript.
Correspondence should be addressed to Ruth E. Siegel, Department of Pharmacology, Case Western Reserve University, School of Medicine, Cleveland, OH 44106-4965.
REFERENCES
- 1.Akazawa C, Shigemoto R, Bessho Y, Nakanishi S, Mizuno N. Differential expression of five N-methyl-d-aspartate receptor subunit mRNAs in the cerebellum of developing and adult rats. J Comp Neurol. 1994;347:150–160. doi: 10.1002/cne.903470112. [DOI] [PubMed] [Google Scholar]
- 2.Altman J. Postnatal development of the cerebellar cortex in the rat. III. Maturation of the components of the granule layer. J Comp Neurol. 1972;145:465–513. doi: 10.1002/cne.901450403. [DOI] [PubMed] [Google Scholar]
- 3.Amico C, Marchetti C, Nobile M, Usai C. Pharmacological types of calcium channels and their modulation by baclofen in cerebellar granules. J Neurosci. 1995;15:2839–2848. doi: 10.1523/JNEUROSCI.15-04-02839.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bading H, Ginty DD, Greenberg ME. Regulation of gene expression in hippocampal neurons by distinct calcium signaling pathways. Science. 1993;260:181–186. doi: 10.1126/science.8097060. [DOI] [PubMed] [Google Scholar]
- 5.Balazs R, Gallo V, Kingsbury A. Effect of depolarization on the maturation of cerebellar granule cells in culture. Brain Res. 1988;468:269–276. doi: 10.1016/0165-3806(88)90139-3. [DOI] [PubMed] [Google Scholar]
- 6.Beattie CE, Siegel RE. Developmental cues modulate GABAA receptor subunit mRNA expression in cultured cerebellar granule neurons. J Neurosci. 1993;13:1784–1792. doi: 10.1523/JNEUROSCI.13-04-01784.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Behringer KA, Gault LM, Siegel RE. Differential regulation of GABAA receptor subunit mRNAs in rat cerebellar granule neurons: importance of environmental cues. J Neurochem. 1996;66:1347–1353. doi: 10.1046/j.1471-4159.1996.66041347.x. [DOI] [PubMed] [Google Scholar]
- 8.Benke D, Mertens S, Trzeciak A, Gillessen D, Möhler H. Identification and immunohistochemical mapping of GABAA receptor subtypes containing the δ subunit in rat brain. FEBS Lett. 1991;1:145–149. doi: 10.1016/0014-5793(91)80573-l. [DOI] [PubMed] [Google Scholar]
- 9.Bessho Y, Nawa H, Nakanishi S. Selective up-regulation of an NMDA receptor subunit mRNA in cultured cerebellar granule cells by K+-induced depolarization and NMDA treatment. Neuron. 1994;12:87–95. doi: 10.1016/0896-6273(94)90154-6. [DOI] [PubMed] [Google Scholar]
- 10.Chijiwa T, Mishima A, Hagiwara M, Sano M, Hayashi K, Inoue T, Naito K, Toshioka T, Hidaka H. Inhibition of forskolin-induced neurite outgrowth and protein phosphorylation by a newly synthesized selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-(p-bromocinnamylamino)ethyl]-5-isoquinolinesulfonamide (H-89), of PC12D pheochromocytoma cells. J Biol Chem. 1990;265:5267–5272. [PubMed] [Google Scholar]
- 11.Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- 12.De Koninck P, Cooper E. Differential regulation of neuronal nicotinic ACh receptor subunit genes in cultured neonatal rat sympathetic neurons: specific induction of α7 by membrane depolarization through a Ca2+/calmodulin-dependent kinase pathway. J Neurosci. 1995;15:7966–7978. doi: 10.1523/JNEUROSCI.15-12-07966.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Enslen H, Soderling TR. Roles of calmodulin-dependent protein kinases and phosphatase in calcium-dependent transcription of immediate early genes. J Biol Chem. 1994;269:20872–20877. [PubMed] [Google Scholar]
- 14.Enslen H, Sun P, Brickey D, Soderling SH, Klamo E, Soderling TR. Characterization of Ca2+/calmodulin-dependent protein kinase IV: role in transcriptional regulation. J Biol Chem. 1994;269:15520–15527. [PubMed] [Google Scholar]
- 15.Gallin WJ, Greenberg ME. Calcium regulation of gene expression in neurons: the mode of entry matters. Curr Opin Neurobiol. 1995;5:367–374. doi: 10.1016/0959-4388(95)80050-6. [DOI] [PubMed] [Google Scholar]
- 16.Gallo V, Kingsbury A, Balazs R, Jorgensen OS. The role of depolarization in the survival and differentiation of cerebellar granule cells in culture. J Neurosci. 1987;7:2203–2213. doi: 10.1523/JNEUROSCI.07-07-02203.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gambarana C, Pittman R, Siegel R. Developmental expression of the GABAA receptor α1 subunit mRNA in the rat brain. J Neurobiol. 1990;21:1169–1179. doi: 10.1002/neu.480210803. [DOI] [PubMed] [Google Scholar]
- 18.Gambarana C, Beattie CE, Rodriguez ZR, Siegel RE. Region-specific expression of messenger RNAs encoding GABAA receptor subunits in the developing rat brain. Neuroscience. 1991;45:423–432. doi: 10.1016/0306-4522(91)90238-j. [DOI] [PubMed] [Google Scholar]
- 19.Ghosh A, Greenberg ME. Calcium signaling in neurons: molecular mechanisms and cellular consequences. Science. 1995;268:239–247. doi: 10.1126/science.7716515. [DOI] [PubMed] [Google Scholar]
- 20.Grynkiewicz G, Poenie M, Tsien RY. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985;260:3440–3450. [PubMed] [Google Scholar]
- 21.Hack N, Hidaka H, Wakefield MJ, Balazs R. Promotion of granule cell survival by high K+ or excitatory amino acid treatment and Ca2+/calmodulin-dependent protein kinase activity. Neuroscience. 1993;57:9–20. doi: 10.1016/0306-4522(93)90108-r. [DOI] [PubMed] [Google Scholar]
- 22.Hall ZW, Sanes JR. Synaptic structure and development: the neuromuscular junction. Cell. 1993;72:99–121. doi: 10.1016/s0092-8674(05)80031-5. [DOI] [PubMed] [Google Scholar]
- 23.Hanson PI, Schulman H. Neuronal Ca2+/calmodulin-dependent protein kinases. Annu Rev Biochem. 1992;61:559–601. doi: 10.1146/annurev.bi.61.070192.003015. [DOI] [PubMed] [Google Scholar]
- 24.Harris BT, Charlton ME, Costa E, Grayson DR. Quantitative changes in α1 and α5 γ-aminobutyric acid type A receptor subunit mRNAs and proteins after a single treatment of cerebellar granule neurons with N-methyl-d-aspartate. Mol Pharmacol. 1994;45:637–648. [PubMed] [Google Scholar]
- 25.Herbert JM, Augereau JM, Gleye J, Maffrand JP. Chelerythrine is a potent and specific inhibitor of protein kinase C. Biochem Biophys Res Commun. 1990;172:993–999. doi: 10.1016/0006-291x(90)91544-3. [DOI] [PubMed] [Google Scholar]
- 26.Jensen KF, Ohmstede CA, Fisher RS, Sahyoun N. Nuclear and axonal localization of Ca2+/calmodulin-dependent protein kinase type Gr in rat cerebellar cortex. Proc Natl Acad Sci USA. 1991;88:2850–2853. doi: 10.1073/pnas.88.7.2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim HY, Sapp DW, Olsen RW, Tobin AJ. GABA alters GABAA receptor mRNAs and increases ligand binding. J Neurochem. 1993;61:2334–2337. doi: 10.1111/j.1471-4159.1993.tb07481.x. [DOI] [PubMed] [Google Scholar]
- 28.Khrestchatisky M, MacLennan A, Chiang M-Y, Xu W, Jackson M, Brecha N, Sternini C, Olsen R, Tobin A. A novel α subunit in rat brain GABAA receptors. Neuron. 1989;3:745–753. doi: 10.1016/0896-6273(89)90243-2. [DOI] [PubMed] [Google Scholar]
- 29.Laurie D, Wisden W, Seeburg P. The distribution of thirteen GABAA receptor subunit mRNAs in the rat brain. III. Embryonic and postnatal development. J Neurosci. 1992;12:4151–4172. doi: 10.1523/JNEUROSCI.12-11-04151.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Levey MS, Brumwell CL, Dryer SE, Jacob MH. Innervation and target tissue interactions differentially regulate acetylcholine receptor subunit mRNA levels in developing neurons in situ. Neuron. 1995;14:153–162. doi: 10.1016/0896-6273(95)90249-x. [DOI] [PubMed] [Google Scholar]
- 31.Li G, Hidaka H, Wollheim CB. Inhibition of voltage-gated Ca2+ channels and insulin secretion in HIT cells by the Ca2+/calmodulin-dependent protein kinase II inhibitor KN-62: comparison with antagonists of calmodulin and L-type Ca2+ channels. Mol Pharmacol. 1992;42:489–488. [PubMed] [Google Scholar]
- 32.Lüddens H, Pritchett D, Köhler M, Killisch I, Keinanen K, Monyer H, Sprengel R, Seeburg P. Cerebellar GABAA receptor selective for a behavioural alcohol antagonist. Nature. 1990;346:648–651. doi: 10.1038/346648a0. [DOI] [PubMed] [Google Scholar]
- 33.Mandelzys A, Pie B, Deneris ES, Cooper E. The developmental increase in ACh current densities on rat sympathetic neurons correlates with changes in nicotinic ACh receptor α-subunit gene expression and occurs independent of innervation. J Neurosci. 1994;14:2357–2364. doi: 10.1523/JNEUROSCI.14-04-02357.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Memo M, Bovolin P, Costa E, Grayson DR. Regulation of γ-aminobutyric acidA receptor subunit expression by activation of N-methyl-d-aspartate-selective glutamate receptors. Mol Pharmacol. 1991;39:599–603. [PubMed] [Google Scholar]
- 35.Motejlek K, Haüselmann R, Leitgeb S, Lüscher B. BSFl, a novel brain-specific DNA-binding protein recognizing a tandemly repeated purine DNA element in the GABAA receptor δ subunit gene. J Biol Chem. 1994;269:15265–15273. [PubMed] [Google Scholar]
- 36.Murphy TH, Worley PF, Baraban JM. L-type voltage-sensitive calcium channels mediate synaptic activation of immediate early genes. Neuron. 1991;7:625–635. doi: 10.1016/0896-6273(91)90375-a. [DOI] [PubMed] [Google Scholar]
- 37.Ohmstede CA, Jensen KF, Sahyoun NE. Ca2+/calmodulin-dependent protein kinase enriched in cerebellar granule cells. Identification of a novel neuronal calmodulin-dependent protein kinase. J Biol Chem. 1989;264:5866–5875. [PubMed] [Google Scholar]
- 38.Quirk K, Whiting PJ, Ragan CI, McKernan RM. Characterisation of δ-subunit containing GABAA receptors from rat brain. Eur J Pharmacol. 1995;290:175–181. doi: 10.1016/0922-4106(95)00061-5. [DOI] [PubMed] [Google Scholar]
- 39.Randall A, Tsien RW. Pharmacological dissection of multiple types of Ca2+ channel currents in rat cerebellar granule neurons. J Neurosci. 1995;15:2995–3012. doi: 10.1523/JNEUROSCI.15-04-02995.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Resink A, Villa M, Benke D, Möhler H, Balazs R. Regulation of the expression of NMDA receptor subunits in rat cerebellar granule cells: effect of chronic K+-induced depolarization and NMDA exposure. J Neurochem. 1995;64:558–565. doi: 10.1046/j.1471-4159.1995.64020558.x. [DOI] [PubMed] [Google Scholar]
- 41.Schulman H. The multifunctional Ca2+/calmodulin-dependent protein kinases. Curr Opin Cell Biol. 1993;5:247–253. doi: 10.1016/0955-0674(93)90111-3. [DOI] [PubMed] [Google Scholar]
- 42.Shivers BD, Killisch I, Sprengel R, Sontheimer H, Köhler M, Schofield PR, Seeburg PH. Two novel GABAA receptor subunits exist in distinct neuronal subpopulations. Neuron. 1989;3:327–337. doi: 10.1016/0896-6273(89)90257-2. [DOI] [PubMed] [Google Scholar]
- 43.Srinivasan M, Edman CF, Schulman H. Alternative splicing introduces a nuclear localization signal that targets multifunctional CaM kinase to the nucleus. J Cell Biol. 1994;126:839–852. doi: 10.1083/jcb.126.4.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sun Y, Rao MS, Landis SC, Zigmond RE. Depolarization increases vasoactive intestinal peptide- and substance P-like immunoreactivities in cultured neonatal and adult sympathetic neurons. J Neurosci. 1992;12:3717–3728. doi: 10.1523/JNEUROSCI.12-10-03717.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thompson C, Pollard S, Stephenson F. Bidirectional regulation of GABAA receptor α1 and α6 subunit expression by a cyclic AMP-mediated signalling mechanism in cerebellar granule cells in primary culture. J Neurochem. 1996;67:434–437. doi: 10.1046/j.1471-4159.1996.67010434.x. [DOI] [PubMed] [Google Scholar]
- 46.Tokumitsu H, Chijiwa T, Hagiwara M, Mizutani A, Terasawa M, Hidaka H. KN-62, 1-[N,O-bis(5-isoquinolinesulfonyl)-N-methyl-L-tyrosyl]4-phenylpiperazine, a specific inhibitor of Ca2+/calmodulin-dependent protein kinase II. J Biol Chem. 1990;265:4315–4320. [PubMed] [Google Scholar]
- 47.Vallano ML, Lambolez B, Audinat E, Rossier J. Neuronal activity differentially regulates NMDA receptor subunit expression in cerebellar granule cells. J Neurosci. 1996;16:631–639. doi: 10.1523/JNEUROSCI.16-02-00631.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Varecka L, Wu C-H, Rotter A, Frostholm A. GABAA/Benzodiazepine receptor α6 subunit mRNA in granule cells of the cerebellar cortex and cochlear nuclei: expression in developing and mutant mice. J Comp Neurol. 1994;339:341–352. doi: 10.1002/cne.903390304. [DOI] [PubMed] [Google Scholar]
- 49.Wegner M, Zhaodan C, Rosenfeld M. Calcium-regulated phosphorylation within the leucine zipper of C/EBPβ. Science. 1992;256:370–373. doi: 10.1126/science.256.5055.370. [DOI] [PubMed] [Google Scholar]
- 50.Witzemann V, Barg B, Criado M, Stein E, Sakmann B. Developmental regulation of five subunit specific mRNAs encoding acetylcholine receptor subtypes in rat muscle. FEBS Lett. 1989;242:419–424. doi: 10.1016/0014-5793(89)80514-9. [DOI] [PubMed] [Google Scholar]
- 51.Ymer S, Schofield PR, Draguhn A, Werner P, Köhler M, Seeburg PH. GABAA receptor β subunit heterogeneity: functional expression of cloned cDNAs. EMBO J. 1989;8:1665–1670. doi: 10.1002/j.1460-2075.1989.tb03557.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zheng T, Santi MR, Bovolin P, Marlier LN, Grayson DR. Developmental expression of the α6 GABAA receptor subunit mRNA occurs only after cerebellar granule cell migration. Dev Brain Res. 1993;75:91–103. doi: 10.1016/0165-3806(93)90068-l. [DOI] [PubMed] [Google Scholar]
- 53.Zheng TM, Zhu WJ, Puia G, Vicini S, Grayson DR, Costa E, Caruncho HJ. Changes in γ-aminobutyrate type A receptor subunit mRNAs, translation product expression, and receptor function during neuronal maturation in vitro. Proc Natl Acad Sci USA. 1994;91:10952–10956. doi: 10.1073/pnas.91.23.10952. [DOI] [PMC free article] [PubMed] [Google Scholar]