Abstract
Background
Drought is a major abiotic stress factor that influences the yield of crops. Basic leucine zipper motif (bZIP) transcription factors play an important regulatory role in plant drought stress responses. However, the functions of a number of bZIP transcription factors in rice are still unknown.
Results
In this study, a novel drought stress-related bZIP transcription factor, OsbZIP62, was identified in rice. This gene was selected from a transcriptome analysis of several typical rice varieties with different drought tolerances. OsbZIP62 expression was induced by drought, hydrogen peroxide, and abscisic acid (ABA) treatment. Overexpression of OsbZIP62-VP64 (OsbZIP62V) enhanced the drought tolerance and oxidative stress tolerance of transgenic rice, while osbzip62 mutants exhibited the opposite phenotype. OsbZIP62-GFP was localized to the nucleus, and the N-terminal sequence (amino acids 1–68) was necessary for the transcriptional activation activity of OsbZIP62. RNA-seq analysis showed that the expression of many stress-related genes (e.g., OsGL1, OsNAC10, and DSM2) was upregulated in OsbZIP62V plants. Moreover, OsbZIP62 could bind to the promoters of several putative target genes and could interact with stress/ABA-activated protein kinases (SAPKs).
Conclusions
OsbZIP62 is involved in ABA signalling pathways and positively regulates rice drought tolerance by regulating the expression of genes associated with stress, and this gene could be used for the genetic modification of crops with improved drought tolerance.
Electronic supplementary material
The online version of this article (10.1186/s12870-019-1872-1) contains supplementary material, which is available to authorized users.
Keywords: Abscisic acid, Drought tolerance, OsbZIP62, Oxidative tolerance, Rice, Transcription factor
Background
Rice is one of the most important grain crops and the most water-consuming crop (approximately 65% of agricultural water) [1, 2]. Due to environmental pollution and climate change, plants frequently encounter various abiotic stresses (such as drought, heat, and salt) that influence their growth and development during the growth process. Drought is one of the major stress factors that severely affects plant growth and results in a series of physiological and biochemical changes [3]. In response to drought stress, plants have evolved a set of very complex and effective strategies that include regulating stomatal closure to reduce plant transpiration and water loss; accumulating osmolytes (such as inorganic ions, organic solutes and sugars) to maintain the osmotic balance of cells; and increasing superoxide dismutase (SOD), catalase (CAT) and other antioxidants to maintain cell membrane stability [4]. These plant physiological and biochemical responses are achieved mainly via stress signal transduction and the transcriptional regulation of downstream stress-related genes [1].
ABA is one of the most widely studied plant hormones that modulate both plant growth and stress responses [5–7]. In recent years, studies have shown that ABA helps plants cope with abiotic stress mainly due to the participation of their receptors. The main concept is as follows: ABA combines with PYR/PYL/RCARs proteins to form a compound, which then interacts with protein phosphatase 2Cs (PP2Cs) to inhibit the activity of PP2Cs, thereby relieving the inhibition of PP2C on SnRK2s. SnRK2s subsequently activate downstream transcription factors by phosphorylation [8–10]. Among these transcription factors, bZIPs compose one of the transcription factor families that has been extensively studied. It has been found that bZIP transcription factors play various roles in plant growth and stress responses, such as seed maturation, disease resistance, and drought resistance [1, 11, 12]. There are approximately 110 bZIP transcription factors in Arabidopsis thaliana, some of which have been identified [13–15]. On the basis of their structure and function, plant bZlP transcription factors are classified into 10 subfamilies [14]. The bZIP transcription factors of different groups play various roles in the abiotic stress response. For example, many members of the group C bZIP transcription factors, known as the ABF/AREB/ABI5 subfamily, participate in ABA signal transduction and the stress response [16, 17]. ABF1 is mainly involved in ABA and low-temperature stress responses, and ABF2/AREB1 and ABF4/AREB2 are mainly involved in ABA, drought, high-salt, heat and oxidative stress responses [18–20].
There are 89 bZIP transcription factors in rice [21, 22]. Studies have shown that several members of the third subfamily, such as OsbZIP23, OsbZIP46, and OsbZIP72, function in rice stress responses [23–25]. Overexpression of OsbZIP23 in transgenic rice plants significantly improved rice drought resistance and salt tolerance and increased sensitivity to ABA [23, 26]. OsbZIP46, which encodes a transcription factor and is homologous to OsbZIP23, plays a very important role in mediating ABA signalling and drought resistance. Overexpression of a constitutively active form of OsbZIP46 significantly increased the drought resistance of rice [25, 27]. OsbZIP72 was identified by yeast one-hybrid screening. Its transcriptional activity mainly depends on the amino acid sequence of its N-terminus. Overexpression of OsbZIP72 in rice plants can also enhance the ABA sensitivity and drought resistance [24, 28].
Though the functions of several bZIP genes in rice have already been determined in terms of stress resistance, a number of bZIP genes have not been identified in rice. In this study, we identified a novel bZIP transcription factors, OsbZIP62, and the results showed that OsbZIP62 is a key regulator in rice drought and oxidative stress responses and has potential application in genetically modified crops with improved drought tolerance.
Results
OsbZIP62 encodes a bZIP protein that belongs to the third subfamily of bZIP transcription factors in rice
In our previous study, the transcriptomes of several typical rice varieties with different drought tolerances were analyzed (unpublished). Some drought tolerance candidate genes whose expression was significantly correlated with stress physiological indices (e.g., osmotic potential) were selected (data not shown). Among these genes, OsbZIP62 was selected for further study. OsbZIP62 encodes a putative bZIP transcription factor [22]. Protein sequence analysis demonstrated that OsbZIP62 contains a typical basic region and leucine zipper domains (Additional file 1: Figure S1). Phylogenetic analysis of the third subgroup members of Arabidopsis and rice bZIP transcription factors showed that OsbZIP62, OsbZIP72 (LOC_Os09g28310), OsbZIP46 (LOC_Os06g10880) and OsbZIP23 (LOC_Os02g52780) were distributed on the same branch (Additional file 1: Figure S2) and that OsbZIP62 was near OsbZIP72, whose function is related to ABA and drought [24]. These results suggested that OsbZIP62 might play a similar role in plant ABA and drought responses.
Expression pattern of OsbZIP62 under different stresses
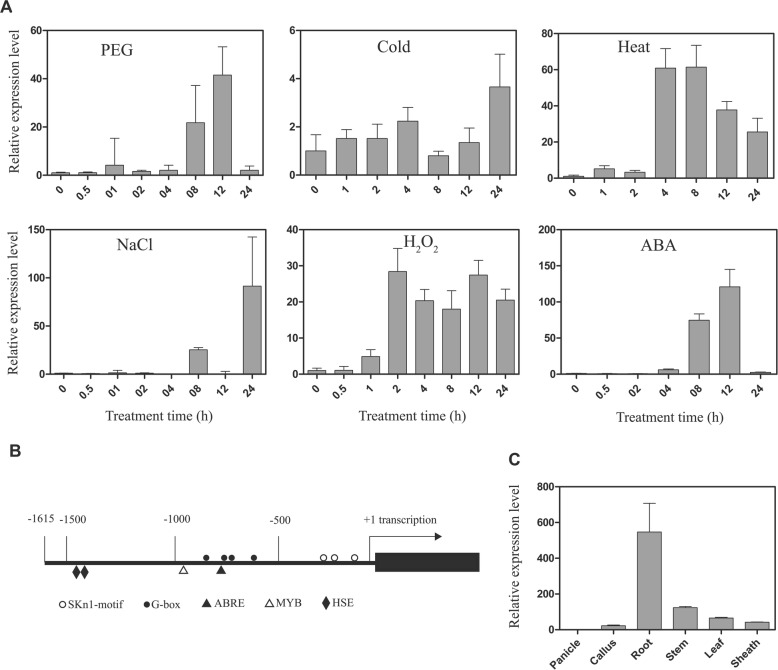
To investigate and predict the function of OsbZIP62, the expression pattern of OsbZIP62 under different abiotic stresses and phytohormone treatments was investigated by qPCR. OsbZIP62 expression was significantly induced by polyethylene glycol (PEG)-simulated drought stress, oxidative stress (induced by H2O2), heat stress, salt stress, and ABA treatment and was slightly induced by cold stress (Fig. 1a). Bioinformatic analysis showed that there were many predicted stress response-related cis-elements, such as G-box recognition site (4 hits), MYB recognition site (1 hit), ABRE element (1 hit) and HSE recognition site (2 hits), in the promoter of OsbZIP62 (Fig. 1b).
Fig. 1.
Expression pattern analysis of OsbZIP62 under abiotic stresses and hormones. a, Relative expression level of OsbZIP62 under various stresses, including PEG6000 (15%), cold (4 °C), heat (42 °C), NaCl (150 mM), H2O2 (1%) and ABA (100 μM). b, Main stress-related cis-acting elements in the OsbZIP62 promoter region. c, Relative expression level of OsbZIP62 in different tissues (roots, stems, leaves, sheaths, panicles and calli) of rice plants under normal condition. The data represent the means ± SE (n = 3). The transcription factor site recognition information resource is available (http://bioinformatics.psb.ugent.be/ebtools/plantcare/html/)
In addition, we also measured the tissue specificity of this gene in different tissues of rice by qPCR. The results showed that OsbZIP62 was expressed in various tissues, but the expression level was high in the roots and relatively low in the panicles (Fig. 1c).
Transactivation activity and subcellular localization of OsbZIP62
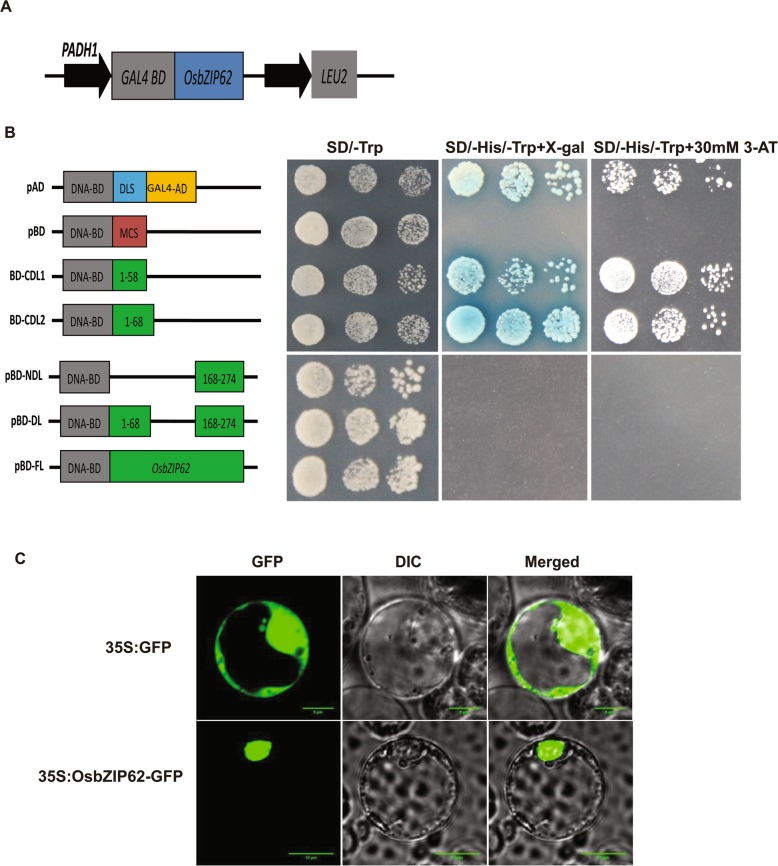
The transcriptional activation activity of the OsbZIP62 protein was determined in yeast. The full length of OsbZIP62, which was fused to the DNA-binding domain of GAL4 (pBD-FL), displayed no transactivation activity in yeast (Fig. 2a and b). Therefore, a series of OsbZIP62 deletions of part of the fragment were tested. The results showed that the expression of the reporter gene could be activated when the C-terminus (amino acids 168–274) was absent (pBD-CDL1 and pBD-CDL2), indicating that the amino acid sequences at the N-terminus (amino acids 1–68) are required for the transactivation activity of OsbZIP62 (Fig. 2b).
Fig. 2.
Transactivation assay and subcellular localization of OsbZIP62 protein. a, Transactivation activities of the different portions of OsbZIP62 were checked in yeast. BD: GAL4 DNA-binding domain; FL: full length; CDL: C-terminus deletion domain; and NDL: N-terminus deletion domain. b, Subcellular localization of OsbZIP62 in rice protoplasts. GFP and OsbZIP62-GFP under the control of the CaMV35S promoter separately transiently expressed in rice protoplasts
An OsbZIP62 protein and green fluorescent protein (GFP) under the control of the CAMV35S promoter were constructed and introduced into rice protoplasts. As shown in Fig. 2c, fluorescence of OsbZIP62-GFP was observed in the nucleus, indicating that OsbZIP62 is a nuclear-targeted protein.
Increased ABA sensitivity of OsbZIP62V transgenic plants
Because the full length of OsbZIP62 had no transactivation activity, it is possible that overexpression of OsbZIP62 is unable to lead to the presumed phenotype. Therefore, we selected OsbZIP62V plants for phenotypic investigation. In these plants, OsbZIP62 coding sequences are fused to a VP64 (tetrameric repeats of VP16) universal transcriptional activation module to maintain transcriptional activation (Additional file 1: Figure S3A). The expression level of OsbZIP62V was significantly higher in transgenic plants than in wild-type (WT) plants, as demonstrated by qPCR analysis (Additional file 1: Figure S3B).
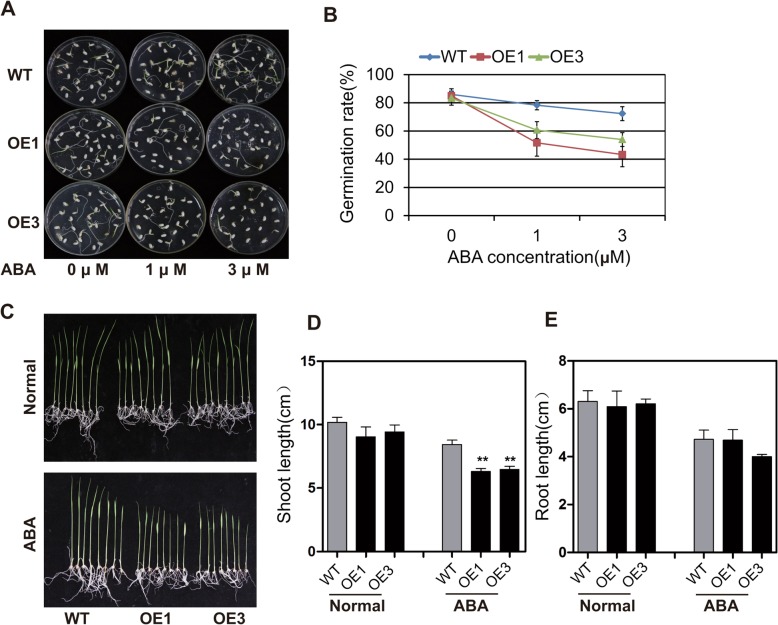
Although the germination rate of the OsbZIP62V overexpression transgenic lines (OE1 and OE3) was similar to that of the WT under 0 μM ABA, it was significantly lower than that of the WT under 1 and 3 μM ABA (Fig. 3a, b). Moreover, the shoot lengths of the OsbZIP62V transgenic rice plants were significantly shorter than those of the WT rice plants under 3 μM ABA treatment (Fig. 3c-e). These results indicated that OsbZIP62V enhanced the sensitivity of the transgenic rice plants to ABA.
Fig. 3.
Increased ABA Sensitivity of OsbZIP62V transgenic plants at the germination and seedling stages. a, Germination performance of OsbZIP62 overexpressors (OE1 and OE3) and WT seeds on 1/2-strength MS medium containing 0, 1, and 3 μM ABA at 7 days after initiation. b, Germination rate of transgenic OsbZIP62 (OE1 and OE3) and the WT transgenic seeds on 1/2-strength MS medium containing 0, 1, and 3 μM ABA. The error bars indicate SEs based on three replicates. c, Performance of two independent OsbZIP62V transgenic lines (OE1 and OE3) and the WT on 1/2-strength MS medium containing 3 μM ABA. d, Shoot length of the transgenic lines and control lines on medium without or with 3 μM ABA. E, Root length of transgenic lines and control lines on medium without or with 3 μM ABA. Statistics of the length of the shoots and the roots at 7 days after growth. The data represent the means ± SE (n ≥ 8 each), **P < 0.01 according to Student’s t test
To further determine whether knocking out OsbZIP62 could affect the sensitivity of transgenic plants to ABA, an osbzip62 mutant was generated via the CRISPR/Cas9 method (Additional file 1: Figure S4). The results showed that the germination rate of the osbzip62 mutant lines was almost the same as that of the wild-type Nipponbare plants under ABA treatment (Additional file 1: Figure S5). There was no significant difference in the length of the shoots and roots of osbzip62 mutants and WT (data not shown). These results suggested that knocking out OsbZIP62 did not significantly affect the ABA sensitivity of rice.
OsbZIP62V improved the tolerance of transgenic rice to drought and oxidative stress
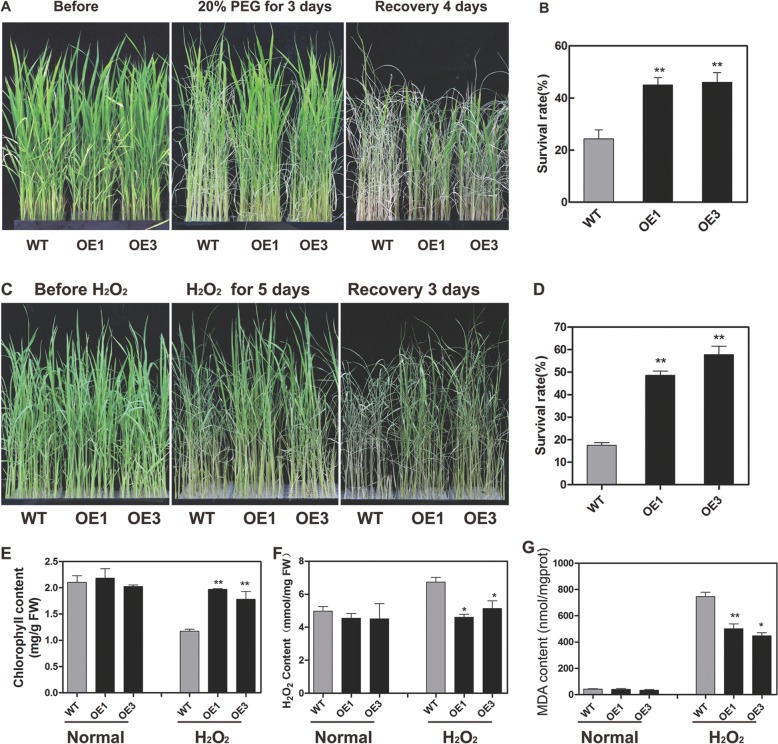
To evaluate the putative biological function of OsbZIP62, we evaluated the drought tolerance of OsbZIP62V plants at the vegetative stage. Under PEG-simulated drought stress treatment, compared with the WT plants, the OsbZIP62V plants displayed less wilting (Fig. 4a). After recovery, 45–46% of the OsbZIP62V plants recovered, while the recovery rate of the WT plants was only 24% (Fig. 4b). The results indicated that OsbZIP62 had a positive effect on rice osmotic stress tolerance.
Fig. 4.
OsbZIP62V enhanced the tolerance of rice to drought and oxidative stress. a, OsbZIP62V transgenic rice plants and WT plants were cultured in 96-well plates and treated with 20% PEG for 3 days, after which they recovered for 4 days. b, Survival rate of OsbZIP62V transgenic plants and WT plants after PEG-simulated drought stress. The error bars indicate SEs based on three replicates, **P < 0.01 according to Student’s t test. c, Phenotype of OsbZIP62V under oxidative stress. OsbZIP62V lines and WT plants were cultured in 96-well plates and treated with 60 mM H2O2 for 5 days, after which they recovered for 3 days. d, Survival rate of OsbZIP62V transgenic plants and control plants after oxidative stress. The error bars indicate SEs based on three replicates. e, Total chlorophyll content in rice leaves. f, Total H2O2 content. g, Total MDA content. The data represent the means ± SDs (n = 4 or 5), *P < 0.05, **P < 0.01 according to Student’s t test. FW: fresh weight
In addition, the seedlings of the OsbZIP62V and WT plants were also treated with 60 mM H2O2 to determine their tolerance to oxidative stress. After treatment for 4–5 d, we found that the wilting degree of the overexpression transgenic plants was lower than that of the WT plants (Fig. 4c) and that the survival rate of the OsbZIP62V (48–55%) plants was significantly greater than that of the WT plants (18%) (Fig. 4d). Physiological analysis showed that, compared with the WT plants, the transgenic rice plants accumulated less H2O2 and malonaldehyde (MDA) and had greater amounts of chlorophyll (Fig. 4e-g). These results suggested that the activated expression of OsbZIP62 improved the oxidative stress tolerance of the transgenic rice plants.
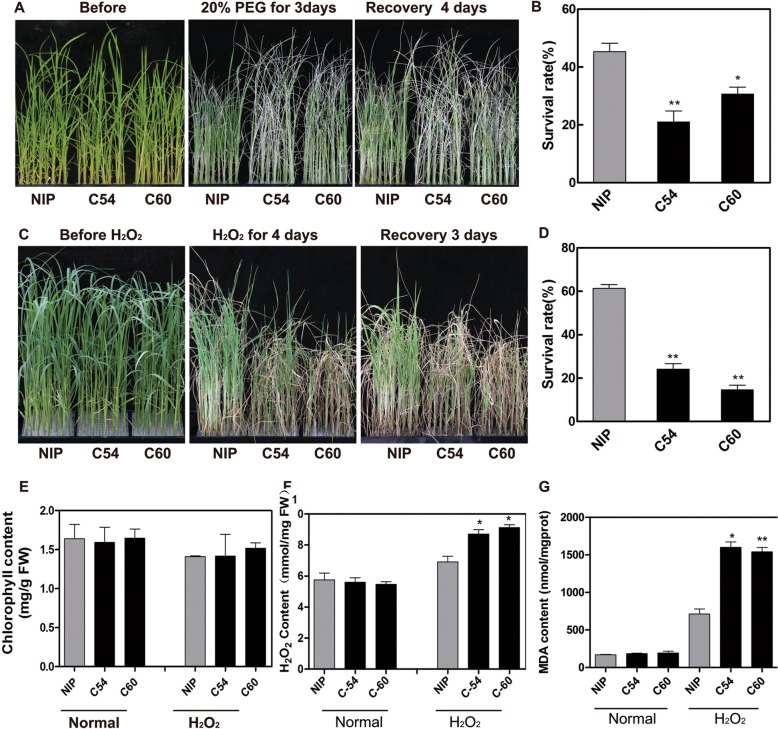
Knocking out OsbZIP62 decreased the tolerance of transgenic rice to drought and oxidative stress
To further validate the function of OsbZIP62, the tolerance of osbzip62 mutants to drought and oxidative stress was evaluated. The leaf rolling of the osbzip62-mutant was significantly worse than that of the WT plants under PEG-simulated drought stress (Fig. 5a). The osbzip62-mutant had a significantly lower recovery (20–36%) than did the WT plants (45%) (Fig. 5b). Furthermore, the degree of wilting of the osbzip62 mutants was higher than that of the WT plants under H2O2 treatment (Fig. 5c). After recovery, the survival rate of the osbzip62 mutants were only 14–24%, while that of the WT was 61% (Fig. 5d). Physiological analysis showed that the contents of H2O2 and MDA in the osbzip62 mutant were significantly greater than that in the WT (Fig. 5e-g). These results demonstrate that knocking out OsbZIP62 reduces rice tolerance to drought and oxidative stress.
Fig. 5.
Decreased tolerance of the osbzip62-mutant plants to drought and oxidative stress. a, Phenotype of the osbzip62-mutant plants under PEG-simulated drought stress. osbzip62 plants and WT plants were treated with 20% PEG for 3 days and then recovered for 4 days. b, Survival rate of osbzip62 plants and WT plants after drought stress. The error bars indicate SEs based on three replicates. c, Phenotype of the osbzip62 plants under oxidative stress. Osbzip62 plants and WT plants were treated with 60 mM H2O2 for 4 days and then recovered for 3 days. d, Survival rate of osbzip62 plants and WT plants after oxidative stress. The error bars indicate SEs based on three replicates. e, Total chlorophyll content in rice leaves. f, Total H2O2 content. g, Total MDA content. The data represent the means ± SDs (n = 4 or 5), *P < 0.05, **P < 0.01 according to Student’s t test. FW: fresh weight
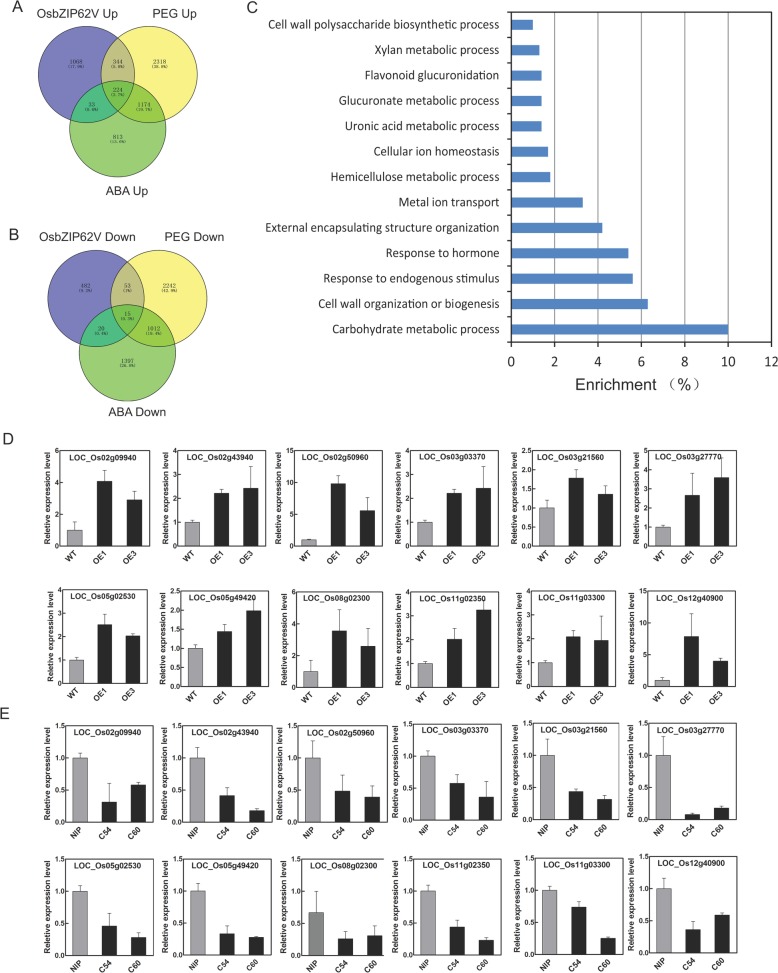
Identification of putative target genes regulated by OsbZIP62
To elucidate the molecular mechanism of OsbZIP62 in rice drought tolerance, the transcriptomes of OsbZIP62V, osbzip62 and WT plants were analyzed using high-throughput sequencing. Differentially expressed genes (DEGs) between the transgenic rice plants and WT plants were analyzed. There were 1669 upregulated genes (fold-change ≥2.0) and 570 downregulated genes (fold change ≤0.5) in the OsbZIP62V transgenic plants compared with WT plants (Additional file 2: Table S1). In the osbzip62 mutants, 663 genes are upregulated, and 480 genes were downregulated (Additional file 3: Table S2). Among the upregulated genes in the OsbZIP62V transgenic plants, 568 genes were also induced by PEG-simulated drought stress, and 257 genes were induced by ABA treatment (the RNA-seq of Nipponbare plants under PEG and ABA treatment was performed by our lab, and these data have not been published), which suggests that these genes upregulated by OsbZIP62 may participate in drought and ABA responses (Fig. 6a). The enriched upregulated genes in the OsbZIP62V plants mainly belong to the following biological process categories: carbohydrate metabolic process, response to abiotic stimuli, cell wall organization or biogenesis, response to hormones, and cellular ion homeostasis (Fig. 6c). The Expression levels of several DEGs were checked by qPCR. The results confirmed that the expression of most of the selected DEGs (e.g., LOC_Os02g43940, LOC_Os05g49420, LOC_Os11g03300 and LOC_Os03g03370) was greater in OsbZIP62V overexpression transgenic lines and lower in osbzip62 mutants than in WT plants (Fig. 6d and e). These results demonstrated that the transcription of these DEGs was affected by OsbZIP62, suggesting that these DEGs might be target genes regulated by OsbZIP62.
Fig. 6.
Transcriptome analysis of OsbZIP62V plants, osbzip62-mutant plants and WT plants. a, Venn diagram for the numbers of upregulated genes in OsbZIP62V plants, PEG-upregulated genes, and ABA-upregulated genes. b, Venn diagram of the numbers of downregulated genes in OsbZIP62V plants, PEG-downregulated genes, and ABA-downregulated genes. c, Classification of partially upregulated genes in the OsbZIP62V transgenic plants compared with the WT plants. d, Relative expression level of DEGs in OsbZIP62V and WT plants. e, Relative expression level of DEGs in osbzip62 plants and WT plants. The date represent the means ± SE (n = 3)
We also analyzed the DEGs whose expression tended to change in the opposite manner in OsbZIP62V plants and osbzip62 mutants and found 85 DEGs were upregulated in the OsbZIP62V plants but downregulated in the osbzip62 mutants (Additional file 4: Table S3). These DEGs encode different functional proteins, such as transcription factors, protease inhibitors, and antioxidant enzymes (e.g., LOC_Os07g01370, LOC_Os02g09940 and LOC_Os05g02530 encode a peroxidase; LOC_Os03g20760 and LOC_Os11g02350 encode a protease inhibitor; LOC_Os08g02300 and LOC_Os12g43110 encode a transcription factor; LOC_Os02g56920 encodes a wax synthesis protein; and LOC_Os03g21560 encodes a photosystem II repair protein).
To further identify the target genes of OsbZIP62, we first analyzed the promoters of the DEGs and identified promoters of several DEGs (e.g., LOC_Os03g03370 (DSM2), LOC_Os11g03300 (OsNAC10), and LOC_Os02g56920 (OsGL1)) containing ABREs or G-box cis-elements that may bind bZIP transcription factors (data not shown). We investigated whether the OsbZIP62 protein could bind to the promoters of these putative target genes through a yeast one-hybrid assay. A pGAD-OsbZIP62 plasmid (containing the OsbZIP62 putative DNA domain fused to the GAL4 active domain) and pHIS-cis reporter construct (~ 1 kb promoters of six presumed target genes) were co-transformed into yeast strain Y187 (Fig. 7a). As indicated by the activation of the reporter genes, OsbZIP62 could bind to the promoters of several genes (i.e., LOC_Os02g56920, LOC_Os03g21560, LOC_Os07g01370, LOC_Os11g03300, and LOC_Os03g03370) (Fig. 7b). These results indicated that OsbZIP62 exhibits DNA binding activity and might directly regulate the expression of these target genes.
Fig. 7.
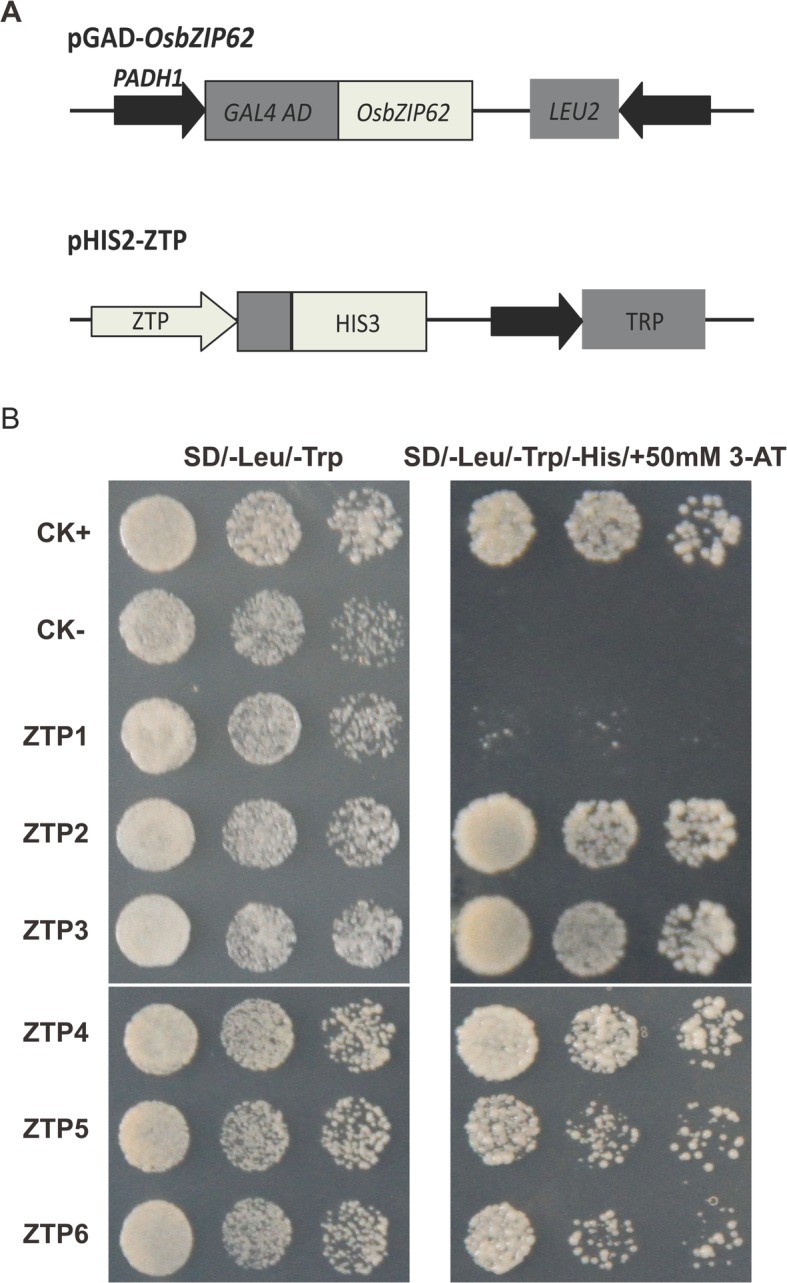
Identification of DNA binding activity of OsbZIP62. a, Schematic structure of the yeast one-hybrid expression construct pGAD-OsbZIP62 and the reporter construct pHIS2.1-ZTP (OsbZIP62 putative target gene promoter). b, Growth performance of transformants on SD/−Leu−/Trp/−His medium containing 30 mM 3-AT. ZTP1-ZTP6 indicates the pGAD-OsbZIP62 plus pHIS2.1-cis (with the promoters of LOC_Os11g02350, LOC_Os02g56920, LOC_Os03g21560, LOC_Os07g01370, LOC_Os11g03300 and LOC_Os03g03370 in pHIS2.1, respectively). ck+: positive control, ck-: negative control (pGADT7-rec2-OsbZIP62 plus p53HIS2)
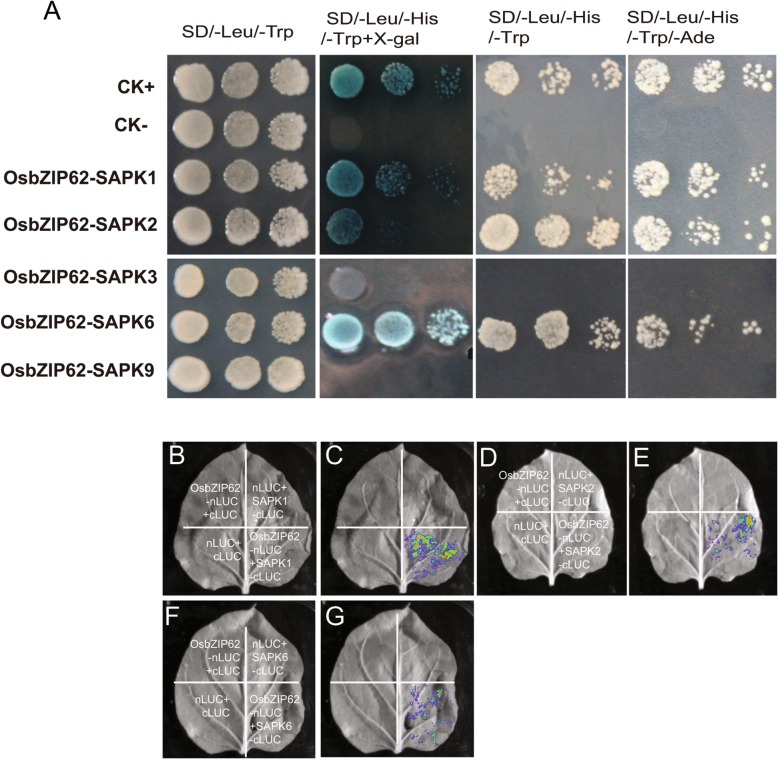
OsbZIP62 interacts with stress/ABA-activated protein kinases and might be activated by phosphorylation
The whole length of OsbZIP62 had no transcriptional activation activity, whereas the N-terminus of OsbZIP62 displayed transactivation activity, even when C-terminus (69–274 amino acids) was deleted. Therefore, we proposed that OsbZIP62 might also interact with SAPK protein kinases and be phosphorylated. To test this hypothesis, using a yeast two-hybrid assay, we first investigated the interaction between OsbZIP62 and five SAPK family members from rice. The results showed that OsbZIP62 could interact with SAPK1, SAPK2 and SAPK6 (Fig. 8a). The interaction between OsbZIP62 and the three SAPKs was further confirmed by luciferase complementation imaging assays in tobacco leaves (Fig. 8b-g). In summary, these results indicated that OsbZIP62 interacted with several SAPKs, suggesting that OsbZIP62 might be activated by the phosphorylation of these protein kinases.
Fig. 8.
OsbZIP62 interacts with SnRK2 protein kinases. a, Yeast two-hybrid assays of OsbZIP62 and SAPK members. SAPK1/SAPK2/SAPK3/SAPK6/SAPK9 were separately fused to the pGADT7 activation domain, which were then transformed into Y2H Gold cells via pGBKT7-OsbZIP62. CK+ and CK- indicate positive and negative controls, respectively. b-g, Confirmation of the in vivo interactions of OsbZIP62 with SAPK1/SAPK2/SAPK6 via Luciferase complementation imaging assays in tobacco leaves
Discussion
OsbZIP62 encodes stress-responsive bZIP transcription factors and positively regulates rice drought tolerance and oxidative tolerance
Transcription factors play an important role in regulating the stress response in plants. In Arabidopsis, many bZIP transcription factors are involved in the ABA-mediated stress response, such as ABI5 [16, 29], ABF1/3/4, and ABRE3 [20, 30–32]. However, only a few of them have been reported in rice, such as OsbZIP23, OsbZIP46, OsbZIP72 [23–25, 27, 33], and the biological functions of many other bZIP proteins remain unclear. Here, we report a novel rice bZIP transcription factor, OsbZIP62, that is involved in rice stress responses. Phylogenetic analysis showed that OsbZIP62 belongs to the third subfamily of bZIP family (Additional file 1: Figure S2). Previous studies have showed that some members of the bZIP third subfamily are also induced by abiotic stress and are involved in drought and ABA signalling pathway [24]. OsbZIP62 was obviously induced by various abiotic stresses (i.e., PEG, H2O2, salt, and heat) and hormone (i.e., ABA) treatments (Fig. 1a). In addition, there are many predicted stress-related cis-elements, including ABREs and HSEs, within the OsbZIP62 promoter region. (Fig. 1b). The current study showed that OsbZIP62V significantly enhanced the tolerance to drought and oxidative stress (Fig. 4), but compared with WT plants, the osbzip62 mutants displayed reduced drought stress tolerance (Fig. 5). In addition, the expression of hundreds of drought-responsive genes was upregulated in OsbZIP62V plants (Fig. 6 and Additional file 2: Table S1). These findings clearly demonstrated that OsbZIP62 encodes a novel bZIP transcription factor that positively regulates the drought tolerance and oxidative tolerance of rice.
OsbZIP62 might participate in the ABA response
It has been shown that bZIP transcription factors constitute the key component of ABA signalling. For example, many members of the group C bZIP transcription factors, which are known as the ABF/AREB/ABI5 subfamily, participate in ABA signal transduction of and the stress response. It is well known that SnRK2 phosphorylation of ABF/AREB is an important part of the complete ABA signalling pathway in Arabidopsis [34, 35]. In particular, TRAB1, which is homologous to OsbZIP62, has been determined to be phosphorylated by SAPKs [36, 37]. In this study, it was found that OsbZIP62 interacted with the SAPK1/2/6 ABA signalling components (Fig. 8), suggesting that OsbZIP62 may be regulated by these protein kinase phosphorylation pathways. Furthermore, we found that the full-length OsbZIP62 protein showed no transcriptional activity but that the N-terminal sequence (amino acids 1–68) exhibited transactivation activity in yeast (Fig. 2a). These results are similar to those of OsbZIP46 and suggest that the transactivation activity of OsbZIP62 might be activated through phosphorylation by these protein kinases; however, the details need to be further studied.
In this study, the activation of OsbZIP62 increased rice sensitivity to ABA (Fig. 3). Furthermore, the expression of a number of ABA-responsive genes was upregulated in OsbZIP62V plants (Fig. 6a). On the other hand, knocking out osbzip62 did not alter the sensitivity to ABA (Additional file 1: Figure S5), possibly because of the abundant bZIP proteins involved in ABA signalling. Nevertherless, all of these findings supported that the transcription and post-translation of OsbZIP62 is regulated by ABA signalling and that OsbZIP62 also partially participates in ABA signalling by regulating many ABA-responsive genes.
OsbZIP62 regulates the expression of many stress-related genes
Transcription factors and cis-elements are the core components of plant regulatory networks. Identification of direct downstream target genes is an effective strategy in terms of clarification of the function of the OsbZIP62 transcription factor. Not surprisingly, OsbZIP62 altered the expression levels of many genes. The expression of nearly more than 1000 genes was upregulated more than 2-fold in the OsbZIP62V overexpressors plants (Additional file 2: Table S1). On the other hand, the mutant of OsbZIP62 also exhibited expression changes of hundreds of genes (Additional file 3: Table S2). Among the DEGs upregulated in OsbZIP62V, more than one-third of them were drought responsive, which is approximately 14% of total drought responsive genes (Fig. 6a). Many genes that are upregulated in OsbZIP62V encode reported or predicted proteins involved in stress resistance, such as transcription factors (LOC_Os11g03300 (OsNAC10)), β-carotene hydroxylase (LOC_Os03g03370 (DSM2)), protease inhibitors (LOC_Os11g02350), and wax synthesis proteins (LOC_Os02g56920 (OsGL1)). OsGL1 has been reported to be one of the genes controlling wax synthesis; this gene is involved in wax accumulation in leaf cuticles and directly affects the drought resistance of rice [38, 39]. OsNAC10, an NAC transcription factor, enhances drought resistance and increases the yield of rice under drought conditions [40]. DSM2 enhances the resistance of rice to drought and oxidative stress by increasing the biosynthesis of lutein and ABA [41]. Yeast one-hybrid assays further confirmed that OsbZIP62 could bind to the promoters of these genes (e.g., OsGL1, OsNAC10, and DSM2) (Fig. 7). These findings suggest that the induced expression of 14% drought stress-related genes by OsbZIP62 contributes to the enhanced drought tolerance of the transgenic rice plants.
Conclusions
We screened drought tolerance candidate genes via a transcriptome analysis of several typical rice varieties with different drought tolerances and selected a novel bZIP gene, OsbZIP62, as one of the candidate genes. OsbZIP62 belongs to the third subfamily of bZIP transcription factors in rice. The expression of OsbZIP62 was obviously induced by drought, heat, salt, H2O2 and ABA treatment. By modulating many stress-related genes, OsbZIP62 is a positive regulator of ABA signalling, drought, and oxidative stress responses. The OsbZIP62 protein localized to the nucleus, and its function in activating downstream genes may depend on posttranslational modification by SAPKs. Thus, we concluded that OsbZIP62 encodes a novel stress-responsive bZIP transcription factor and positively regulates the rice drought and oxidative stress responses.
Methods
Construction of transgenic materials
The full-length cDNA of OsbZIP62 was amplified from the cDNA of Nipponbare (Oryza sativa L. japonica. cv. Nipponbare) and then cloned into a pEASY-Blunt simple cloning vector for sequencing. The seeds of Nipponbare were obtained from the germplasm bank in our lab. The seeds of transgenic japonica rice Kitaake with OsbZIP62-VP64 were acquired from Fujian Agriculture and Forestry University, and these transgenic rice plants were generated as described previously [42].
CRISPR constructs were designed according to a previously described protocol [43, 44], and the vectors were transformed into Nipponbare using the Agrobacterium-mediated method. The primers used in this study are listed in Additional file 5: Table S4. Individual T0 transformants were analyzed by sequencing their OsbZIP62 target regions, which were amplified by PCR. Mutations were detected by aligning the sequence chromatograms of these PCR products with those of the WT controls. Several individual homozygous or heterozygous plants were obtained, and two homozygous lines, C54 and C60, were selected for further experiments.
Plant materials, growth conditions and stress treatments
To determine the gene expression profiles, we planted the Nipponbare plants in culture solution in a greenhouse with a 16-h light/8-h dark cycle. Rice seedlings at the four-leaf stage were treated with hormones or subjected to non-biological stresses such as cold stress (rice seedlings were transferred to a growth chamber at 4 °C), heat stress (rice seedlings were exposed to a 42 °C growth chamber), osmotic stress (15% (m/v) PEG6000), oxidative stress (1% H2O2) and salt stress (150 mM NaCl) and then sampled at 0, 1, 2, 4, 8, 12 and 24 h. For the phytohormone ABA treatment, 0.1 mM ABA was added to the culture medium of the rice plants.
To determine the ABA sensitivity of the transgenic plants at the seedling stage, positive transgenic seeds were selected in one-half-strength MS culture medium supplemented with 50 mg/L hygromycin. WT control lines were germinated on 1/2-strength MS medium. Seedlings (12 plants each, 3 repeats) were transplanted to 1/2-strength MS culture medium or 1/2-strength MS medium containing 3 μM ABA and were grown for 7 days. To testing the ABA sensitivity of transgenic plants at the germination stage, the seeds of positive transgenic plants and wild type plants (approximately 30 seeds, three repetitions) were germinated on 1/2-strength MS medium containing a concentration gradient of ABA (0, 1, 3, and 6 μM), and the germination rate of the seeds was calculated after 7 days.
Determination of physiological indices related to stress
For all stresses, the seeds of T3 lines were germinated on 1/2-strength MS medium supplemented with 50 mg/L hygromycin, and the seeds of WT also grown on 1/2-strength MS medium. At the four-leaf stage, osmotic stress (20% PEG treatment for 3 days before rewatering) and oxidative stress (60 mM H2O2 treatment for 4–5 days) were applied, and the survival rates were measured as described previously [45, 46]. Moreover, we performed a biochemical analysis of reactive oxygen species (ROS). H2O2 was extracted from the leaves, and its contents were determined following the manufacturer’s instructions as described previously [47]. The activity of ROS-scavenging enzymes, soluble protein and MDA contents were determined according to the manufacturer’s instructions (Nanjing Institute of Bioengineering, Jiangsu, China). The chlorophyll contents were measured as described previously [48].
RNA isolation and quantification of gene expression
Total RNA was extracted from the rice leaves using TRNzol-A+ reagent (Tiangen) according to the manufacturer’s instructions. First-strand cDNA was synthesized via EasyScript One-Step gDNA Removal and cDNA Synthesis Super Mix (Transgen). Real-time quantitative PCR (qPCR) was performed on an optical 96-well plate with a Bio-Rad CFX96 Real-Time PCR Detection System using TransStart Green qPCR SuperMix (Transgen). The PCR procedure was as follows: 94 °C for 30 s, followed by 40 cycles at 94 °C for 5 s, 55 °C for 15 s and 72 °C for 10 s. The rice OsAct8 gene (No. AY212324) was used as an internal control to normalize the target gene expression, and relative expression levels were determined as described previously [49].
RNA sequencing
Three biological replicates (30 plants each) of wild-type, OsbZIP62V and osbzip62 mutant plants were sampled for RNA sequencing. The total RNA was extracted using the Trizol Reagent (Life Technologies). The qualified RNA samples were then used for library construction following the specifications of the TruSeq RNA Sample Preparation v2 Guide (Illumina), and RNA sequencing was conducted with Illumina Hiseq 2500 at Shanghai Personal Biotechnology Co., Ltd. (Shanghai, China). We used SeqPrep to strip adaptors and/or merge paired reads that overlapped into single reads and used Sickle to remove low-quality reads. The clean data were then aligned to the reference genome of rice using HISAT2 v2.1.0. FPKM (fragments per kilobase per millon mapped reads) was then calculated to estimate the expression level of the genes. DESeq2 v1.6.3 was used to analyse the differential gene expression between two samples, and genes with q<0.05 and |log2_ratio|>1 were identified as differentially expressed genes. GO (Gene Ontology; http://geneontology.org/) enrichment of DEGs was implemented by hypergeometric tests in which p-value is calculated and adjusted as q-value. GO terms with q < 0.05 were considered significantly enriched.
Subcellular localization
To study the subcellular localization of OsbZIP62 protein, the whole length of OsbZIP62 was cloned into a pCAMBIA1300GFP plant expression vector after digestion of XbaI and BamHI enzyme; thus, OsbZIP62 was fused to GFP. Rice protoplasts were subsequently prepared from rice etiolated shoots, transformed with GFP or OsbZIP62-GFP constructs, and subjected to PEG treatment. GFP fluorescence was detected under an Olympus confocal microscope 24 h after transformation.
Transactivation activity assay in yeast
The transcriptional activation activity of OsbZIP62 was studied in yeast cells. The full length or truncated fragments of OsbZIP62 were fused in frame with the receptor GAL4 DNA-binding domain in a pGBKT7 vector by recombination reactions. These constructs were separately transformed into Y2H Gold cells. The transformants were evaluated by serial dilution on SD/−Trp/−His medium containing different concentrations of 3-amino-1, 2, 4-triazole (3-AT) or X-α-Gal.
Yeast one-hybrid assays
We selected six genes (i.e., LOC_Os11g02350, LOC_Os02g56920, LOC_Os03g21560, LOC_Os07g01370, LOC_Os11g03300, and LOC_Os03g03370) that were upregulated in OsbZIP62V plants and downregulated in osbzip62 mutants. The upstream approximately 1 kb long promoter of these potential target genes was cloned from the genomic DNA of Nipponbare. For the yeast one-hybrid assays, the promoter sequences of these genes were cloned into the yeast expression vector pHIS2.1 after digestion of the SmaI and EcoRI enzymes and then was co-transformed with the pGAD-T7-OsbZIP62 into Y187 yeast cells. The DNA-protein interaction was evaluated by transformant growth assays on SD/−Leu/−Trp/−His plates supplied with 50 mM of 3-AT.
Yeast two-hybrid assays
Yeast two-hybrid assays were carried out with a Matchmaker™ Gold Yeast Two-Hybrid System (Clontech). The coding regions of OsbZIP62 and SAPKs were cloned into pGBKT7 and pGAD-T7 vectors to generate bait and prey vectors, respectively. These vectors were subsequently co-transformed into the Y2H Gold strain (Clontech). Protein interactions were evaluated by transformant growth assays on SD/−Leu/−Trp/−His, SD/−Leu/−Trp/−His with X-Gal, and SD/− Leu/−Trp/−His-Ade at 30 °C for 3–5 days.
LUC complementary imaging assays
OsbZIP62 and SAPK1/SAPK2/SAPK6 were cloned into the pCAMBIA1300-nLUC and pCAMBIA1300-cLUC firefly luciferase vectors, respectively. All of the constructs were introduced into A. tumefaciens strain EHA105, which were then injected into 3-week-old tobacco leaves to transiently transform the tobacco epidermal cells [50]. The fluorescence signal was detected 3 days after infiltration under a plant in vivo imaging system (NightSHADE LB 985, Berthold).
Additional files
Figure S1. Protein sequence alignment of OsbZIP62 and the third subfamily bZIP transcription factors in rice. Figure S2. Molecular phylogenetic tree of the third bZIP subfamily members. Figure S3. Identification of OsbZIP62-VP64 overexpression lines. Figure S4. Characterization of osbzip62 mutants. Figure S5. ABA sensitivity of osbzip62 mutants. (DOCX 2016 kb)
Table S1. DEGs in OsbZIP62-VP64 overexpressor (XLSX 177 kb)
Table S2. DEGs in osbzip62 mutant (XLSX 96 kb)
Table S3. Genes proposed to be regulated by OsbZIP62 (XLSX 14 kb)
Table S4. List of primers used in this study (XLSX 14 kb)
Acknowledgements
We thank Professor Chen-tao Lin (Fujian Agriculture and Forestry University) for providing OsbZIP62-VP64 overexpression rice seeds and Professor Yao-guang Liu (South China Agricultural University) for providing the CRISPR/CAS9 system.
Abbreviations
- ABA
abscisic acid
- bZIP
basic leucine zipper motif
- CAT
catalase
- DEGs
differentially expressed genes
- MDA
malonaldehyde
- MS
murashige and skoog
- PEG
polyethylene glycol
- SAPK
stress/ABA activated protein kinases
- SOD
superoxide dismutase
Authors’ contributions
SQY and KX performed yeast assays, subcellular localization, stress treatments, physiological analysis and wrote the manuscript; SJC carried out the gene cloning and vector construction; TFL performed the transformation of rice; HX carried out the expression analysis, LC and HYL analyzed RNA-seq data; and LJL designed the experiments, supervised this work, and assisted with editing the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by Shanghai Agriculture Applied Technology Development Program (2017-02-00-08-F00071), Shared Platform of Crop Germplasm Resources in Shanghai (18DZ2293700), Shanghai Natural Sciences Foundation (16ZR1431200), and outstanding Team Project from Shanghai Agricultural Academy of Science (2017(A-01)). The funding agencies had no role in the experimental design, data collection and analysis or preparation of the manuscript.
Availability of data and materials
The RNA-seq data supporting the results of this article have been submitted to the GEO at NCBI with the accession number GSE122887.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declared that they have no competing interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Shiqin Yang and Kai Xu contributed equally to this work.
Contributor Information
Shiqin Yang, Email: yangshiqin2008@163.com.
Kai Xu, Email: kxu@sagc.org.cn.
Shoujun Chen, Email: csj08@sagc.org.cn.
Tianfei Li, Email: ltf@sagc.org.cn.
Hui Xia, Email: hxia@sagc.org.cn.
Liang Chen, Email: cl@sagc.org.cn.
Hongyan Liu, Email: lhy@sagc.org.cn.
Lijun Luo, Phone: +86-21-62200490, Email: lijun@sagc.org.cn.
References
- 1.Rodriguez-Uribe L, O'Connell MA. A root-specific bZIP transcription factor is responsive to water deficit stress in tepary bean (Phaseolus acutifolius) and common bean (P. vulgaris) J Exp Bot. 2006;57(6):1391–1398. doi: 10.1093/jxb/erj118. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Q. Strategies for developing green super Rice. Proc Natl Acad Sci U S A. 2007;104(42):16402–16409. doi: 10.1073/pnas.0708013104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Luo LJ. Breeding for water-saving and drought-resistance rice (WDR) in China. J Exp Bot. 2010;61(13):3509–3517. doi: 10.1093/jxb/erq185. [DOI] [PubMed] [Google Scholar]
- 4.Ouyang SQ, Liu YF, Liu P, Lei G, He SJ, Ma B, Zhang WK, Zhang JS, Chen SY. Receptor-like kinase OsSIK1 improves drought and salt stress tolerance in rice (Oryza sativa) plants. Plant J. 2010;62(2):316–329. doi: 10.1111/j.1365-313X.2010.04146.x. [DOI] [PubMed] [Google Scholar]
- 5.Verma V, Ravindran P, Kumar PP. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016;16:86. doi: 10.1186/s12870-016-0771-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Seif El-Yazal SA, Seif El-Yazal MA, Dwidar EF, Rady MM. Phytohormone crosstalk research: cytokinin and its crosstalk with other phytohormones. Curr Protein Pept Sci. 2015;16(5):395–405. doi: 10.2174/1389203716666150330141159. [DOI] [PubMed] [Google Scholar]
- 7.Peleg Z, Blumwald E. Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol. 2011;14(3):290–295. doi: 10.1016/j.pbi.2011.02.001. [DOI] [PubMed] [Google Scholar]
- 8.Park E, Kim TH. Production of ABA responses requires both the nuclear and cytoplasmic functional involvement of PYR1. Biochem Biophys Res Commun. 2017;484(1):34–39. doi: 10.1016/j.bbrc.2017.01.082. [DOI] [PubMed] [Google Scholar]
- 9.Nishimura N, Sarkeshik A, Nito K, Park SY, Wang A, Carvalho PC, Lee S, Caddell DF, Cutler SR, Chory J, et al. PYR/PYL/RCAR family members are major in-vivo ABI1 protein phosphatase 2C-interacting proteins in Arabidopsis. Plant J. 2010;61(2):290–299. doi: 10.1111/j.1365-313X.2009.04054.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hubbard KE, Nishimura N, Hitomi K, Getzoff ED, Schroeder JI. Early abscisic acid signal transduction mechanisms: newly discovered components and newly emerging questions. Genes Dev. 2010;24(16):1695–1708. doi: 10.1101/gad.1953910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang L, Xia C, Zhao G, Liu J, Jia J, Kong X. A novel wheat bZIP transcription factor, TabZIP60, confers multiple abiotic stress tolerances in transgenic Arabidopsis. Physiol Plant. 2015;153(4):538–554. doi: 10.1111/ppl.12261. [DOI] [PubMed] [Google Scholar]
- 12.Kang JY, Choi HI, Im MY, Kim SY. Arabidopsis basic leucine zipper proteins that mediate stress-responsive abscisic acid signaling. Plant Cell. 2002;14(2):343–357. doi: 10.1105/tpc.010362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hossain MA, Cho JI, Han M, Ahn CH, Jeon JS, An G, Park PB. The ABRE-binding bZIP transcription factor OsABF2 is a positive regulator of abiotic stress and ABA signaling in rice. J Plant Physiol. 2010;167(17):1512–1520. doi: 10.1016/j.jplph.2010.05.008. [DOI] [PubMed] [Google Scholar]
- 14.Jakoby M, Weisshaar B, Droge-Laser W, Vicente-Carbajosa J, Tiedemann J, Kroj T, Parcy F. bZIP transcription factors in Arabidopsis. Trends Plant Sci. 2002;7(3):106–111. doi: 10.1016/S1360-1385(01)02223-3. [DOI] [PubMed] [Google Scholar]
- 15.Matiolli C. C., Tomaz J. P., Duarte G. T., Prado F. M., Del Bem L. E. V., Silveira A. B., Gauer L., Correa L. G. G., Drumond R. D., Viana A. J. C., Di Mascio P., Meyer C., Vincentz M. The Arabidopsis bZIP Gene AtbZIP63 Is a Sensitive Integrator of Transient Abscisic Acid and Glucose Signals. PLANT PHYSIOLOGY. 2011;157(2):692–705. doi: 10.1104/pp.111.181743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bensmihen S, Giraudat J, Parcy F. Characterization of three homologous basic leucine zipper transcription factors (bZIP) of the ABI5 family during Arabidopsis thaliana embryo maturation. J Exp Bot. 2005;56(412):597–603. doi: 10.1093/jxb/eri050. [DOI] [PubMed] [Google Scholar]
- 17.Wang B, Li C, Kong X, Li Y, Liu Z, Wang J, Li X, Yang Y. AtARRE, an E3 ubiquitin ligase, negatively regulates ABA signaling in Arabidopsis thaliana. Plant Cell Rep. 2018;37(9):1269–1278. doi: 10.1007/s00299-018-2311-8. [DOI] [PubMed] [Google Scholar]
- 18.Gao S, Gao J, Zhu X, Song Y, Li Z, Ren G, Zhou X, Kuai B. ABF2, ABF3, and ABF4 promote ABA-mediated chlorophyll degradation and leaf senescence by transcriptional activation of chlorophyll catabolic genes and senescence-associated genes in Arabidopsis. Mol Plant. 2016;9(9):1272–1285. doi: 10.1016/j.molp.2016.06.006. [DOI] [PubMed] [Google Scholar]
- 19.Fujita Y, Fujita M, Satoh R, Maruyama K, Parvez MM, Seki M, Hiratsu K, Ohme-Takagi M, Shinozaki K, Yamaguchi-Shinozaki K. AREB1 is a transcription activator of novel ABRE-dependent ABA signaling that enhances drought stress tolerance in Arabidopsis. Plant Cell. 2005;17(12):3470–3488. doi: 10.1105/tpc.105.035659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim S, Kang JY, Cho DI, Park JH, Kim SY. ABF2, an ABRE-binding bZIP factor, is an essential component of glucose signaling and its overexpression affects multiple stress tolerance. Plant J. 2004;40(1):75–87. doi: 10.1111/j.1365-313X.2004.02192.x. [DOI] [PubMed] [Google Scholar]
- 21.Zhao Y, Zhao S, Mao T, Qu X, Cao W, Zhang L, Zhang W, He L, Li S, Ren S, et al. The plant-specific actin binding protein SCAB1 stabilizes actin filaments and regulates stomatal movement in Arabidopsis. Plant Cell. 2011;23(6):2314–2330. doi: 10.1105/tpc.111.086546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nijhawan A, Jain M, Tyagi AK, Khurana JP. Genomic survey and gene expression analysis of the basic leucine zipper transcription factor family in rice. Plant Physiol. 2008;146(2):333–350. doi: 10.1104/pp.107.112821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xiang Y, Tang N, Du H, Ye H, Xiong L. Characterization of OsbZIP23 as a key player of the basic leucine zipper transcription factor family for conferring abscisic acid sensitivity and salinity and drought tolerance in rice. Plant Physiol. 2008;148(4):1938–1952. doi: 10.1104/pp.108.128199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lu G, Gao C, Zheng X, Han B. Identification of OsbZIP72 as a positive regulator of ABA response and drought tolerance in rice. Planta. 2009;229(3):605–615. doi: 10.1007/s00425-008-0857-3. [DOI] [PubMed] [Google Scholar]
- 25.Tang N, Zhang H, Li X, Xiao J, Xiong L. Constitutive activation of transcription factor OsbZIP46 improves drought tolerance in rice. Plant Physiol. 2012;158(4):1755–1768. doi: 10.1104/pp.111.190389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gui J, Zheng S, Liu C, Shen J, Li J, Li L. OsREM4.1 interacts with OsSERK1 to coordinate the interlinking between abscisic acid and Brassinosteroid signaling in Rice. Dev Cell. 2016;38(2):201–213. doi: 10.1016/j.devcel.2016.06.011. [DOI] [PubMed] [Google Scholar]
- 27.Tang N, Ma S, Zong W, Yang N, Lv Y, Yan C, Guo Z, Li J, Li X, Xiang Y, et al. MODD mediates deactivation and degradation of OsbZIP46 to negatively regulate ABA signaling and drought resistance in rice. Plant Cell. 2016. [DOI] [PMC free article] [PubMed]
- 28.Qiu J, Hou Y, Wang Y, Li Z, Zhao J, Tong X, Lin H, Wei X, Ao H, Zhang J. A Comprehensive Proteomic Survey of ABA-Induced Protein Phosphorylation in Rice (Oryza sativa L.) Int J Mol Sci. 2017;18:1. doi: 10.3390/ijms18122457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carles C, Bies-Etheve N, Aspart L, Leon-Kloosterziel KM, Koornneef M, Echeverria M, Delseny M. Regulation of Arabidopsis thaliana Em genes: role of ABI5. Plant J. 2002;30(3):373–383. doi: 10.1046/j.1365-313X.2002.01295.x. [DOI] [PubMed] [Google Scholar]
- 30.Uno Y, Furihata T, Abe H, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K. Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc Natl Acad Sci U S A. 2000;97(21):11632–11637. doi: 10.1073/pnas.190309197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fernando VCD, Al Khateeb W, Belmonte MF, Schroeder DF. Role of Arabidopsis ABF1/3/4 during det1 germination in salt and osmotic stress conditions. Plant Mol Biol. 2018;97(1–2):149–163. doi: 10.1007/s11103-018-0729-6. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida T, Fujita Y, Sayama H, Kidokoro S, Maruyama K, Mizoi J, Shinozaki K, Yamaguchi-Shinozaki K. AREB1, AREB2, and ABF3 are master transcription factors that cooperatively regulate ABRE-dependent ABA signaling involved in drought stress tolerance and require ABA for full activation. Plant J. 2010;61(4):672–685. doi: 10.1111/j.1365-313X.2009.04092.x. [DOI] [PubMed] [Google Scholar]
- 33.Zhang C, Li C, Liu J, Lv Y, Yu C, Li H, Zhao T, Liu B. The OsABF1 transcription factor improves drought tolerance by activating the transcription of COR413-TM1 in rice. J Exp Bot. 2017;68(16):4695–4707. doi: 10.1093/jxb/erx260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fujii H, Verslues PE, Zhu JK. Arabidopsis decuple mutant reveals the importance of SnRK2 kinases in osmotic stress responses in vivo. Proc Natl Acad Sci U S A. 2011;108(4):1717–1722. doi: 10.1073/pnas.1018367108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fujita Y, Nakashima K, Yoshida T, Katagiri T, Kidokoro S, Kanamori N, Umezawa T, Fujita M, Maruyama K, Ishiyama K, et al. Three SnRK2 protein kinases are the main positive regulators of abscisic acid signaling in response to water stress in Arabidopsis. Plant Cell Physiol. 2009;50(12):2123–2132. doi: 10.1093/pcp/pcp147. [DOI] [PubMed] [Google Scholar]
- 36.Kobayashi Y, Murata M, Minami H, Yamamoto S, Kagaya Y, Hobo T, Yamamoto A, Hattori T. Abscisic acid-activated SNRK2 protein kinases function in the gene-regulation pathway of ABA signal transduction by phosphorylating ABA response element-binding factors. Plant J. 2005;44(6):939–949. doi: 10.1111/j.1365-313X.2005.02583.x. [DOI] [PubMed] [Google Scholar]
- 37.Kagaya Y, Hobo T, Murata M, Ban A, Hattori T. Abscisic acid-induced transcription is mediated by phosphorylation of an abscisic acid response element binding factor, TRAB1. Plant Cell. 2002;14(12):3177–3189. doi: 10.1105/tpc.005272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Islam MA, Du H, Ning J, Ye H, Xiong L. Characterization of Glossy1-homologous genes in rice involved in leaf wax accumulation and drought resistance. Plant Mol Biol. 2009;70(4):443–456. doi: 10.1007/s11103-009-9483-0. [DOI] [PubMed] [Google Scholar]
- 39.Zhou L, Ni E, Yang J, Zhou H, Liang H, Li J, Jiang D, Wang Z, Liu Z, Zhuang C. Rice OsGL1-6 is involved in leaf cuticular wax accumulation and drought resistance. PLoS One. 2013;8(5):e65139. doi: 10.1371/journal.pone.0065139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jeong JS, Kim YS, Baek KH, Jung H, Ha SH, Do Choi Y, Kim M, Reuzeau C, Kim JK. Root-specific expression of OsNAC10 improves drought tolerance and grain yield in rice under field drought conditions. Plant Physiol. 2010;153(1):185–197. doi: 10.1104/pp.110.154773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Du H, Wang N, Cui F, Li X, Xiao J, Xiong L. Characterization of the beta-carotene hydroxylase gene DSM2 conferring drought and oxidative stress resistance by increasing xanthophylls and abscisic acid synthesis in rice. Plant Physiol. 2010;154(3):1304–1318. doi: 10.1104/pp.110.163741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhao T, Liu J, Li HY, Lin JZ, Bian MD, Zhang CY, Zhang YX, Peng YC, Liu B, Lin C. Using hybrid transcription factors to study gene function in rice. Sci China Life Sci. 2015;58(11):1160–1162. doi: 10.1007/s11427-015-4937-x. [DOI] [PubMed] [Google Scholar]
- 43.Ma X, Zhang Q, Zhu Q, Liu W, Chen Y, Qiu R, Wang B, Yang Z, Li H, Lin Y, et al. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol Plant. 2015;8(8):1274–1284. doi: 10.1016/j.molp.2015.04.007. [DOI] [PubMed] [Google Scholar]
- 44.Zhang H, Zhang J, Wei P, Zhang B, Gou F, Feng Z, Mao Y, Yang L, Xu N, Zhu JK. The CRISPR/Cas9 system produces specific and homozygous targeted gene editing in rice in one generation. Plant Biotechnol J. 2014;12(6):797–807. doi: 10.1111/pbi.12200. [DOI] [PubMed] [Google Scholar]
- 45.Xiao B, Huang Y, Tang N, Xiong L. Over-expression of a LEA gene in rice improves drought resistance under the field conditions. Theor Appl Genet. 2007;115(1):35–46. doi: 10.1007/s00122-007-0538-9. [DOI] [PubMed] [Google Scholar]
- 46.Yue B, Xue W, Xiong L, Yu X, Luo L, Cui K, Jin D, Xing Y, Zhang Q. Genetic basis of drought resistance at reproductive stage in rice: separation of drought tolerance from drought avoidance. Genetics. 2006;172(2):1213–1228. doi: 10.1534/genetics.105.045062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rao MV, Lee H, Creelman RA, Mullet JE, Davis KR. Jasmonic acid signaling modulates ozone-induced hypersensitive cell death. Plant Cell. 2000;12(9):1633–1646. doi: 10.1105/tpc.12.9.1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Benedetti CE, Arruda P. Altering the expression of the chlorophyllase gene ATHCOR1 in transgenic Arabidopsis caused changes in the chlorophyll-to-chlorophyllide ratio. Plant Physiol. 2002;128(4):1255–1263. doi: 10.1104/pp.010813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods. 2001;25(4):402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 50.Liu L, Zhang Y, Tang S, Zhao Q, Zhang Z, Zhang H, Dong L, Guo H, Xie Q. An efficient system to detect protein ubiquitination by agroinfiltration in Nicotiana benthamiana. Plant J. 2010;61(5):893–903. doi: 10.1111/j.1365-313X.2009.04109.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Protein sequence alignment of OsbZIP62 and the third subfamily bZIP transcription factors in rice. Figure S2. Molecular phylogenetic tree of the third bZIP subfamily members. Figure S3. Identification of OsbZIP62-VP64 overexpression lines. Figure S4. Characterization of osbzip62 mutants. Figure S5. ABA sensitivity of osbzip62 mutants. (DOCX 2016 kb)
Table S1. DEGs in OsbZIP62-VP64 overexpressor (XLSX 177 kb)
Table S2. DEGs in osbzip62 mutant (XLSX 96 kb)
Table S3. Genes proposed to be regulated by OsbZIP62 (XLSX 14 kb)
Table S4. List of primers used in this study (XLSX 14 kb)
Data Availability Statement
The RNA-seq data supporting the results of this article have been submitted to the GEO at NCBI with the accession number GSE122887.