Abstract
Objective
To explore the toxicity and biological activity of cobalt nanoparticles on the osteoclasts. Analyze the relationship between cobalt nanoparticles and osteolysis.
Methods
Monocyte‐macrophages (RAW 264.7) was cultured in vitro, osteoclast‐like cells were induced by lipopolysaccharides (LPS). After RAW 264.7 was induced for 24 h, Methyl Thiazolium Tetrazolium (MTT) biological toxicity test of osteoclast‐like cell was preceded using Cobalt nanoparticles (set 4 concentrations: 10, 20, 50, 100 μM) and cobalt chloride (set 4 concentrations: 10, 20, 50, 100 μM) at 2, 4, 8, 24 and 48 h respectively. The relative expression of mRNA of CA II and Cat K after RAW 264.7 induction was determined by Q‐PCR.
Results
mRNA relative expression of CA II, Cat K were reduced at multiple concentrations both cobalt nanoparticles and cobalt chloride, and was time and concentration dependent, cobalt nanoparticles are more significant than cobalt chloride group. But when the cobalt nanoparticles concentration is in 10–50 μM, the mRNA relative expression of CA II, Cat K increased.
Conclusion
Cobalt nanoparticles have biological toxicity. At multiple concentrations, the differentiation and proliferation of osteoclasts was inhibited, but when the concentration of cobalt nanoparticles is in 10–50 μM, it has been strengthened.
Keywords: Cobalt nanoparticles, Osteoclast, Osteolysis, Toxicity
Introduction
Currently, artificial arthroplasty is a mature technology, and has lifted the majority of patients' pain. But the clinical complications of artificial arthroplasty still cannot be ignored, where, aseptic loosening is one of the main reasons for revision surgery.
Studies have shown that the metal prosthesis will be acted by external forces such as friction, collision after it is implanted, which makes the inert oxide layer on the prosthesis wear faster, and makes the metal ions, nano‐sized metals released to blood and around the prosthesis1, 2. Metal ions can induce monocyte/macrophages to release bone absorption factor and cause bone around the prosthesis dissolved3, 4. It is well known that loosening is associated with osteolysis, and osteolysis is directly associated with osteoclasts. This study explored the toxicity and biological activity of cobalt nanoparticles on the osteoclasts. Analyze the relationship between cobalt nanoparticles and osteolysis.
Materials and Method
Materials
Test Cell
RAW 264.7 (ATCC Source) Bought from the cell repository of Chinese Academy of Sciences (Shanghai).
Main Reagents
Cobalt nanoparticles, produced by Shanghai Chaowei Nano Technology Company. Cobalt Chloride, produced by Shanghai National Medicine Group Reagent Company.
Primers
Primers were composited by Biomics Biotechnology Co., Ltd, as Table 1.
Table 1.
Peimers
| Name | Base sequence (5′ → 3′) | PCR product size |
|---|---|---|
| Mm‐CAII‐F | CGCTAGACGGACGACAACTT | 103 bp |
| Mm‐CAII‐R | CCTTGTGCCAGTTCTCTGGT | |
| Mm‐catk‐F | ATGTGGGTGTTCAAGTTTCTG | 109 bp |
| Mm‐catk‐R | ACTGCTTCTGGTGAGTCTTC | |
| Mm‐GAPDH‐F | GTATGACTCCACTCACGGCAAA | 101 bp |
| Mm‐GAPDH‐R | GGTCTCGCTCCTGGAAGATG |
Experimental Grouping
All cells were induced by LPS, the LPS concentration was set as 100 ng/ml. After 24 hours, grouped, each concentration of each group were three samples. Grouped as follows:
Nano Cobalt group, 4 concentrations were set: 10 μM, 20 μM, 50 μM, 100 μM.
CoCl2 group, 4 concentrations were set: 10 μM, 20 μM, 50 μM, 100 μM.
Blank control group, 0 μM.
Method
RAW264.7 Cell Was Induced by LPS to Make Osteoclast Induction Differentiation
Digested RAW264.7 cells and counted, paved cells in 6 pore plates with the density of 1 × 105 pcs/ml. DMEM + 10%FBS culture was added. After 24 h, LPS was added to culture solution, the LPS concentration was set as 100 ng/ml. TRAP staining was made separately at 24 h, 48 h and 72 h after induction. Also, microscope was adopted to observe and photographs were taken.
MTT Biological Toxicity Experiment
MTT experimental was advanced separately at 24 h, 48 h and 72 h after LPS induction, after adding different concentrations of cobalt nanoparticles, Cocl2.
Detect mRNA Relative Expression of CA II, Cat K
RNA of RAW264.7 after 24 h induction was extracted by RISO, and took appropriate RNA, using nucleic acid ultraviolet analyzer to test A260/A280 ratio and total RNA concentration(Units μg/ml, such as A260/A280 ratio is between 1.8–2.0, means has good quality total RNA). Q‐PCR system using one step method to detect the mRNA relative expression of CA II, Cat K. PCR reaction system: template 2 μl, 2× Master Mix 6 μl, 50× SYBR Green I 0.2 μl, upstream and downstream of each primer 0.2 μl, add DEPC water to 12 μl.
Statistical Analysis
All data were expressed by mean ± root‐mean‐square error, and one‐way analysis of variance was used for comparing multiple means. SNK (Student‐Newman‐Keuls) multiple comparisons were done for the items with significant variance test results. SPSS 19.0 statistical software was used to process and analyze data. P value < 0.05 was considered statistically significant.
Results
Osteoclast‐Like Cells
In this experimental, Osteoclast‐like cells were formed after mouse mononuclear cell RAW 264.7 was induced by LPS. Enzyme activity part was purple particle precipitate, after TRAT staining, distributed in all or most of the cytoplasm. Nuclei were negative (as Fig. 1). The longer the induction time was, the deeper the color would become. Cell culture is normal.
Figure 1.
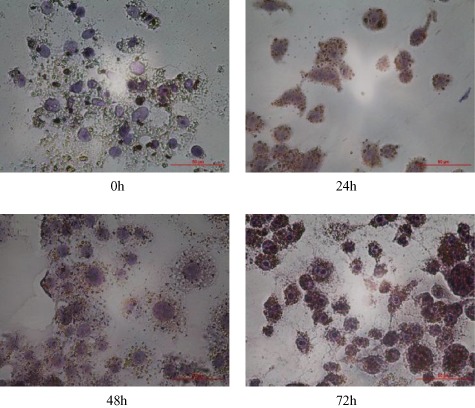
Osteoclast‐like cells induced mouse mononuclear cell RAW 264.7 by LPS.
MTT Biological Toxicity
Cobalt nanoparticles and Cobalt Chloride with different concentrations have inhibitory effects on osteoclast proliferation. Toxicity increased with time increasing, when the time > 24 h, the toxicity is very high. P < 0.01, the inhibitory effect is time and concentration dependent. Under the same concentration, proliferation and differentiation effects of Cobalt Chloride on osteoclasts were significantly lower than Cobalt nanoparticles. When the nano concentration is greater than 10, P < 0.01, it has statistically significant.
Different times (2, 4, 8, 24, 48 h), different concentrations(10, 20, 50, 100 μM), comparative study of biological activity of cobalt nanoparticles on the osteoclasts shows that, when the nano concentration is greater than 10 μM, P < 0.05, it has statistically significant. Toxicity increased with time increasing, when the time > 24 h, toxicity greatly increased, P < 0.05, compare with 2 h, it has statistically significant (as Fig. 2 shown).
Figure 2.

Proliferation inhibition rate of cobalt nanopaticles on osteoclast.
Different concentrations (10, 20, 50, 100 μM), comparative study of biological activity of Cobalt Chloride on the osteoclasts shows that, when the nano concentration is greater than 20 μM, P < 0.05, the statistical significant is significantly. Toxicity increased with time increasing, when the time > 24 h, toxicity increased greatly, P < 0.05, compare with 2 h, it has statistically significant (shown as Fig. 3).
Figure 3.

Proliferation inhibition rate of cobalt chloride on osteoclast.
mRNA Relative Expression of CA II, Cat K
Compared with blank control group, mRNA relative expression of CA II of Cobalt nanoparticles and Cobalt Chloride plurality treated groups decreased, P < 0.05, it has statistical significant. With the increase of Cobalt nanoparticles concentration, at the range of 10–50 μm, the relative expression of CA II appear increase. But for Cobalt Chloride, concentration at the range of 10–50 μm, the relative expression of CA II decrease greatly (as Fig. 4).
Figure 4.
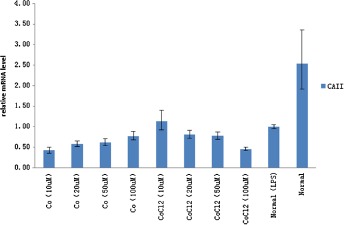
CA II relative expression of each treatment group.
Relative Expression Comparison of Cat K Under Different Concentrations of Cobalt Nanoparticles and Cobalt Chloride
Compared with blank control group, mRNA relative expression of Cat K of Cobalt nanoparticles plurality treated groups all decreased, P < 0.05, it has statistical significant. But when the Cobalt nanoparticles concentration is between 10–50 μm, the relative expression of Cat K had increasing trend (shown as Fig. 5).
Figure 5.

Cat K relative expression of each treatment group.
Discussion
It is well‐known that prosthesis loosening after artificial arthroplasty is related to bone dissolving, while bone dissolving is mainly completed by osteoclasts. Study the toxicity and biological activity of cobalt nanoparticles on the osteoclasts as well as the relationship with bone dissolving, firstly, it should make modeling for osteoclasts.
Osteoclasts is from multinuclear giant cell of hematopoietic cell with diameter of 100 μM, including 2–50 tightly packed nucleuses, mainly distributed on the bone surface and around the blood vessel channel in the bone. It is composed by multi mononuclear cells, cytoplasm is basophilic, with the aging of cell, and it becomes gradually oxyphilous. Highly expressed tartrate‐resistant acid phosphatase and tissue lysosome prolease K is the main mark of osteoclasts. It is reported that cobalt nanoparticles may cause influence on expression of some gene of osteoclasts, thus increased the activity of osteoclasts. Due to osteoclasts is terminally differentiated cell, its content is so small in the body, it can't proliferate or make passage, and the formation mechanism is disputable, so osteoclasts culture method in vitro and purification method are continuously discussed to now.
Among the known osteoclasts culture methods in vitro, the most classical is Chanbers method, ie. Mechanically separating bone cells and bone marrow cells of femur, humerus, tibia of rat to cultivate, but the number of the obtained osteoclasts is less with low purity5, 6, 7.
Considering that the well accepted method8, 9, 10 of osteoclasts culture in vitro is adopting induction agent to induce precursor cell to produce osteoclast‐like cell which is used as osteoclasts model for study.
Mouse's monocyte macrophages RAW 264.7 is an ideal osteoclasts precursor cell, its gene expression profile is close to osteoclasts, and it can form bone resorption. Mature osteoclast‐like cell can be obtained after being stimulated by induction agent, this is current only recognized lined osteoclasts precursor cell8, 10. Osteoclast‐like cell marker enzyme has four marker enzymes, including matrix metalloproteinases, matrix metalloproteinase‐9, tartrate‐resistant acid phosphatase and tissue prolease K. Latter two kinds are same as osteoclasts' main mark.
It is currently recognized that both RANKL and LPS can induce RAW264.7 cell to differentiate to osteoclasts. Shamima Islam et al. have already confirmed that LPS has good induction effect on RAW 264.7 cell by experiment11. Considering that previous research has confirmed that osteoclast‐like cell is obtained by LPS inducing RAW 264.7 cell is a good modeling method, so LPS was adopted as induction agent for this test. RAW 264.7 cell was induced to osteoclast‐like cell, thus study on toxic effect and biological effect of cobalt nanoparticles on osteoclasts can be done.
In this study, after 100 ng/ml of LPS was added to RAW 264.7 cell, cell began to enlarge in 24 h, pseudopod increased, a few multinuclear giant cells appeared, the number of multinuclear giant cells began to increase gradually in 72 h. In 24 h, individual multinuclear giant cell appeared, showing TRAP dyeing, ie cytoplasm has wine red particle precipitation. In 48 h, TRAP dyeing negative, many multinuclear giant cells and a few mononuclear cells were presented. The longer the induction time was, the deeper the color would become. It indicates that LPS can induce RAW 264.7 cell to differentiate to osteoclasts.
Cat K gene is located at Iq21.2, transcription product is 1.7 kb long, composed of 8 exons and 7 introns. It is shown that Cat K has no TATA/CAAT box by analyzing transcription initiation 5′ end flanking area, but there are two homologous Sp1 binding sites and G + C‐rich area in the promoter region, they can be used as potential transcriptional regulatory elements. Mature Cat K is a monosome with the molecular weight of 24 KD. Cat K zymogen and mature type have 3D structure with a high resolution. Cat K has 329 polypeptide residue sequences, it is a typical propolypeptide structure of papaya prolease family, including two structural domains: one contains 15 amino acid residues signal sequences, the other contains 99 leader peptide fragments of amino acid residues. There is a V‐shaped active site at the joint in these two regions12.
The experiment proved that mature Cat K molecule has 3 pairs of disulfide bonds composed of 6 cysteine residues to stabilize the structure of Cat K. In acidic environment, mature Cat K exposed the catalytic substrate degraded active site by automatically disconnecting the front‐end part, including activated cysteine, histidine and aspartic acid residues13. Researches show that in osteoclasts, IL‐α1 and RANKL or M‐CSF + RANKL stimulates Cat K gene expression by NF‐kB path.
Lysosome Cat K is key bone remodeling. Cat K dissolves organic substrate (it is mainly threadiness I type collagen). Substrate degradation mainly depends on the activity of Cat K and activity of collagenase of MMPS family, mainly, Cat K expressed in osteoclasts is a strong extracellular matrix‐degrading enzyme which plays an important role in bone resorption mediated by osteoclasts. Inhibiting Cat K through the special inhibitors or antisense nucleotide will cause undigested collagenous fiber accumulated in the lysosome of osteoclasts14. In the bone and cartilage, after Cat K chondroitin sulfate form complexes, triple‐helical structure of collagen could be dissociated in multiple loci15, 16, 17.
Carbonic anhydrase is a zinc‐containing enzyme in the 28 kD brains, the human CA II gene is located on chromosome 8: 8q22 with total length of 760 bp. It is distributed widely, and expressed in certain cytoplasm of almost all tissues or organs. It plays an important role in differentiation of osteoclasts and bone absorption by influencing the stability of internal pH and Ca2+. CA II can promote osteoclasts differentiation, which plays an important role in the process of osteoclasts bone absorption, this process need acid inner environment which is produced after CA II catalyzes carbon dioxide and makes comprehensive reaction with water. In addition, CA II expression may vary with the function of osteoclasts. In the condition of bone resorption, it is showing high expression status18.
After artificial arthroplasty, ion state and nanometer state metal particles would be produced by friction, degradation, they will be toxic to both osteoblasts and osteoclasts. Where, cobalt chloride is generally considered to be significant inhibitory effect on osteoclasts. The results show that, under the same concentration, proliferation and differentiation effects of cobalt chloride on osteoclasts were significantly lower than cobalt nanoparticles, which means that proliferation inhibition rate of cobalt nanoparticles on osteoclasts was higher than that of cobalt chloride19, 20. Generally, since cobalt nanoparticles have significant inhibitory effect on both osteoblasts and osteoclasts, osteoclasts. Osteoblast is inhibited, so its function is not strong, but osteoclasts is also inhibited, thus osteolysis should not occur. But in this test, after the relative expression quantity of isogene mRNA of two iconic indicators (CA II and Cat K) of osteoclast was determined, it is found that when the concentration range of cobalt nanoparticles is 10–50 μM, the concentration of cobalt nanoparticles is proportional to gene expression quantity of specific indicator CA II and Cat K of osteoclasts, namely the proliferation of cobalt nanoparticles to osteoclast is not inhibition, osteoclasts do not die out, but more and more active, it plays promotion and enhancing role, thus producing osteolysis. The exact mechanism is unknown.
Why the toxicity of nanometer state metal particles produced by friction and degradation to osteoclasts is stronger than that of ion state metal particles after artificial arthroplasty? This is mainly due to the nature of nanoparticles. With the decrease of nanoparticle diameter, the ratio between number of surface atoms or molecules and total number of atoms or molecules will exponentially increase, thus, the material without toxicity or less toxicity originally reaches nanoscale, then the nature and strength of biological effect of also may make qualitative changes and become high toxic. Past researches are mostly focus to ion level, cobalt nanoparticle study is less, although known cobalt nanoparticles have toxicity to related cells of human body, the study on toxic effects of cobalt nanoparticles on osteoclasts and biological effects has not been reported. This paper discussed this issue for the first time, laid a foundation to further study the relationship between cobalt nanoparticle diameter, concentration, osteoclasts proliferation and osteolysis. The mechanism of proliferation effect of the cobalt nanoparticle on osteoclasts from 10 μM to 50 μM still need further study.
Grant Sources: National Natural Science Foundation of China (No. 81171743).
Disclosure: This research was supported by grants from National Natural Science Foundation of China, No.81171743.
References
- 1. Vendittoli PA, Amzica T, Roy AG, et al Ion release with large‐diameter metal‐on‐metal hip arthroplasty. J Arthroplasty, 2011, 26: 282–288. [DOI] [PubMed] [Google Scholar]
- 2. Li G, Li Q. The influence and coumtermeasures of metal ions after metal‐on‐metal hip replacement. Zhonghua Guan Jie Wai Ke Za Zhi, 2010, 4: 666–670 (in Chinese). [Google Scholar]
- 3. Moroni A, Nocco E, Hoque M, et al Cushion bearings versus large diameter head metal‐on‐metal bearings in total hip arthroplasty: a short‐term metal ion study. Arch Orthop Trauma Surg, 2012, 132: 123–129. [DOI] [PubMed] [Google Scholar]
- 4. Kim PR, Beaulé PE, Dunbar M, et al Cobalt and chromium levels in blood and urine following hip resurfacing arthroplasty with the Conserve Plus implant. J Bone Joint Surg Am, 2011, 93 (Suppl 2): 107–117. [DOI] [PubMed] [Google Scholar]
- 5. Chamber TJ, Magnus CJ. Calcitonin alters behaviour of isolated osteoclasts. J Pathol, 1982, 137: 27–39. [DOI] [PubMed] [Google Scholar]
- 6. Kartsogiannis V, Moseley J, McKelvie B, et al Temporal expression of PTHrP during endochondral bone formation in mouse and intramembranous bone formation in an in vivo rabbit model. Bone, 1997, 21: 385–392. [DOI] [PubMed] [Google Scholar]
- 7. Tsukii K, Shima N, Mochizuki S, et al Osteoclast differentiation factor mediates an essential signal for bone resorption induced by 1 alpha,25‐dihydroxyvitamin D3, prostaglandin E2, or parathyroid hormone in the microenvironment of bone. Biochem Biophys Res Commun, 1998, 246: 337–341. [DOI] [PubMed] [Google Scholar]
- 8. Yamamoto A, Miyazaki T, Kadono Y, et al Possible involvement of IkappaB kinase 2 and MKK7 in osteoclastogenesis induced by receptor activator of nuclear factor kappaB ligand. J Bone Miner Res, 2002, 17: 612–621. [DOI] [PubMed] [Google Scholar]
- 9. Hsu H, Lacey DL, Dunstan CR, et al Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proc Natl Acad Sci U S A, 1999, 96: 3540–3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Islam S, Hassan F, Tumurkhuu G, et al Bacterial lipopolysaccharide induces osteoclast formation in RAW 264.7 macrophage cells. Biochem Biophys Res Commun, 2007, 360: 346–351. [DOI] [PubMed] [Google Scholar]
- 11. Han J, Luo M. Research process for cathepsin K. Int J Endocrinol Metab, 2006, 26: 125–127. [Google Scholar]
- 12. Lutgens SP, Cleutjens KB, Daemen MJ, Heeneman S. Cathepsin cysteine proteases in cardiovascular disease. FASEB J, 2007, 21: 3029–3041. [DOI] [PubMed] [Google Scholar]
- 13. He W, Liu K, et al Recent progress on the cathepsin k and treatment of osteoporosis disease. Chin J Osteoporos, 2008, 14: 670–673. [Google Scholar]
- 14. Lecaille F, Brömme D, Lalmanach G. Biochemical properties and regulation of cathepsin K activity. Biochimie, 2008, 90: 208–226. [DOI] [PubMed] [Google Scholar]
- 15. Li Z, Hou WS, Escalante‐Torres CR, Gelb BD, Bromme D. Collagenase activity of cathepsin K depends on complex formation with chondroitin sulfate. J Biol Chem, 2002, 277: 28669–28676. [DOI] [PubMed] [Google Scholar]
- 16. Saito S, Ishii T, Mori S, Hosaka K, Ootaki M, Tokuhashi Y. Long‐term results of metasul metal‐on‐metal total hip arthroplasty. Orthopedics, 2010, 33(8). Doi: 10.3928/01477447-20100625-11. [DOI] [PubMed] [Google Scholar]
- 17. Liu J, Hu W. Research progress for bone resorption mechanism of osteoclast. Zhongguo Jiao Xing Wai Ke Za Zhi, 2002, 9: 401–402 (in Chinese). [Google Scholar]
- 18. Teitelbaum SL. Bone resorption by osteoclasts. Science, 2000, 289: 1504–1508. [DOI] [PubMed] [Google Scholar]
- 19. Hwang do W, Lee DS, Kim S. Gene expression profiles for genotoxic effects of silica‐free and silica‐coated cobalt ferrite nanoparticles. J Nucl Med, 2012, 53: 106–112. [DOI] [PubMed] [Google Scholar]
- 20. Andrews RE, Shah KM, Wilkinson JM, Gartland A. Effects of cobalt and chromium ions at clinically equivalent concentrations after metal‐on‐metal hip replacement on human osteoblasts and osteoclasts: implications for skeletal health. Bone, 2011, 49: 717–723. [DOI] [PubMed] [Google Scholar]


