Abstract
Drought, cold, and high salinity are three major abiotic stresses effecting apple tree growth and fruit production. Understanding the genetic mechanisms of crosstalk between stress responses signalling networks and identifying the genes involved in apple has potential importance for crop improvement and breeding strategies. Here, the transcriptome profiling analysis of in vitro-grown apple plants subjected to drought, cold and high salinity stress, showed a total of 377 upregulated and 211 downregulated common differentially expressed genes (DEGs) to all 3 stress treatments compared with the control. Gene Ontology (GO) analysis indicated that these common DEGs were enriched in ‘metabolic process’ under the ‘biological process’ category, as well as in ‘binding’ and ‘catalytic activity’ under the ‘molecular function’ category. Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis showed that common DEGs were mainly belong to the ‘biological functions’ category and 17 DEGs were identified in ‘environmental information processing’ sub-category which may act as signal transduction components in response crosstalk regulation. Overexpression of 5 upregulated genes individually, out of these 17 common DEGs in apple calli promoted the consistent upregulation of DREB6, CBF1 and ZAT10 and increased the mass weight and antioxidase ability, implying these five common DEGs involved in multiple pathways and improved comprehensive resistance to stress.
Subject terms: Abiotic, Transcriptomics
Introduction
Plants are frequently exposed to various biotic and abiotic stresses, of which drought, cold and high-salinity stresses are the principal causes of declines in crop productivity worldwide1. Stress-inducing factors can occur simultaneously or sequentially and cause osmotic stress, water deficits, ionic imbalances, peroxidation damage and, ultimately, growth inhibition, as well as effects associated with similar physiological processes2,3. In response to these stresses, plants have developed diverse pathways that coordinate to combat and tolerate stress.
Many studies have revealed details of the signal transduction pathways that are activated by individual stresses4. For example, abscisic acid (ABA)-dependent and -independent pathways are activated by drought stress, the ICE-CBF-COR signalling pathway is activated by cold resistance, and the SOS pathway is induced by salt stress5–7. Moreover, similar regulatory components involving shared gene expression patterns, physiological indicators and phenotypic characteristics have been shown to be involved in drought, cold and high-salinity stresses. In some plant species, the induction of cold resistance also promotes drought resistance and high-salinity tolerance, which is consistent with an increase in the levels of osmo-regulatory compounds and antioxidant enzyme activities8. In other studies, an overlap between the expression patterns of stress-responsive genes in Citrus9, grape (Vitis vinifera)10, poplar (Populus)11, tea tree (Camellia sinensis)12, Arabidopsis thaliana13, maize (Zea mays)14, and other plant species was observed after drought, cold and high-salinity stress induction.
Overexpression of stress resistance genes involved in one type of stress can enhance resistance to other stresses, further suggesting complex cross-regulation of different stress signalling pathways. For example, the overexpression of DREB2a, which has been identified as a drought resistance gene, in transgenic A. thaliana and Lotus corniculatus forage plants resulted in enhanced tolerance to both drought and salt stress. Under stress conditions, the transgenic plants were taller and had longer roots, elevated levels of soluble sugars and a lower content of malondialdehyde compared with the control15. Antioxidase GPX3, commonly considered a vital scavenger of reactive oxygen species (ROS), also plays an important role in ABA-mediated stomatal closure under drought stress due to the oxidation of ABI1 and ABI2 by GPX316,17. Previous studies have revealed genes and pathways that are involved in diverse abiotic stresses and that are potentially useful candidates for genetic engineering to improve multiple stress tolerance.
Transcriptome analysis has been widely applied to explore and identify differentially expressed genes (DEGs) involved in plant growth, fruit development and stress hormone regulation18–20. The availability of the apple draft genome sequence provides an opportunity for detailed analysis of stress resistance genes and their transcription; however, in contrast to A. thaliana21,22, maize23, and rice (Oryza sativa)24, little has been reported about transcriptome changes in apple in response to drought, cold and high salinity. Here, we describe RNA-seq analyses of apple plants grown under drought, cold or acute high-salinity stress to identify shared regulatory pathways, key functional genes or signal transduction components involved in the three stresses. This integrated study provides insights into the molecular mechanisms of the cross-regulation of abiotic stress responses in apples.
Results
Apple gene expression profiles in response to three abiotic stresses
After quality assessment and data filtering, an average of 8.16 billion reads with a Q30 >93.5% were retained as high-quality reads for each library (Supplementary Table 1). These clean reads were then combined in a de novo assembly, which resulted in 43,752 unigenes, of which 20,913 were longer than 1 kb (Supplementary Table 2).
To analyse variation in gene expression under drought, cold and high-salinity stresses, DEGs with FC > 2.0 and FDR < 0.01 between the treatments and controls (drought vs. control, cold vs. control, and high salinity vs. control) were identified (Fig. 1a). Generally, more DEGs were detected, including both upregulated and downregulated DEGs, in the cold and high-salinity treatment comparisons than in the drought vs. control comparison. Notably, there were 3552 upregulated DEGs under cold stress treatment, more than the number of upregulated DEGs in response to drought and high-salinity stress, with 1379 and 2312 members, respectively. Of the genes that were differentially expressed as a result of all three stresses (common DEGs), 377 were consistently upregulated and 211 were consistently downregulated (Fig. 1b,c).
Figure 1.
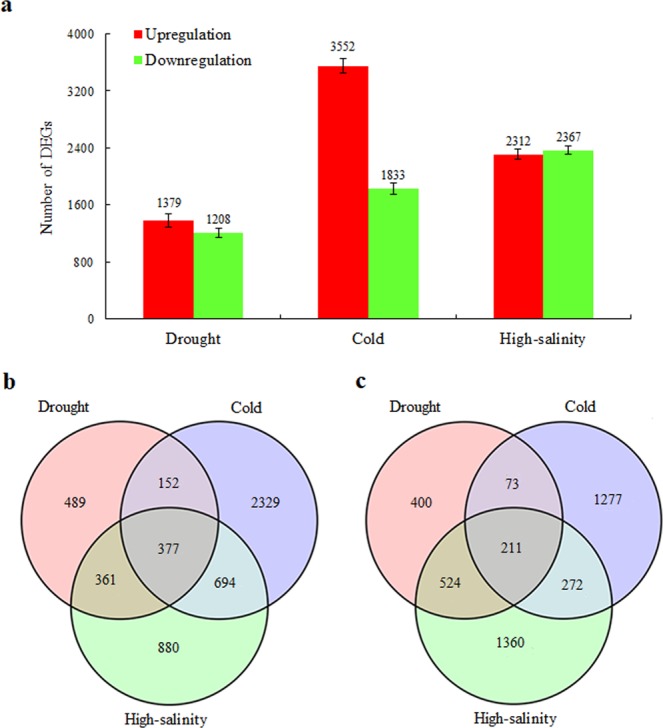
Apple gene expression profiles in response to drought, cold and high-salinity stress. (a) Total number of upregulated and downregulated genes. (b) Venn diagram of upregulated genes in the three samples vs. control. (c) Venn diagram of downregulated genes in the three samples vs. control.
GO classification of common DEGs
To understand the functional categories of commonly expressed genes, Gene Ontology (GO) classification was performed on 377 commonly upregulated and 211 commonly downregulated genes (Supplementary Fig. 1). The most frequently assigned GO terms in the ‘biological process’ category were ‘metabolic process’ (GO: 0008152) and ‘cellular process’ (GO: 0009987); the most frequently assigned GO terms in the ‘cellular component’ category were ‘cell’ (GO: 0005623) and ‘cell part’ (GO: 0044464); and the most frequently assigned GO terms in the ‘molecular function’ category were ‘binding’ (GO: 0005488) and ‘catalytic activity’ (GO: 0003824) (Table 1).
Table 1.
Gene Ontology (GO) classification of common differentially expressed genes (DEGs) in response to drought, cold and high-salinity stress.
| Gene Ontology term | Number of DEGs | |
|---|---|---|
| Upregulated | Downregulated | |
| ▶ Biological process | ||
| biological regulation (GO:0065007) | 43 | 16 |
| cellular component organization or biogenesis (GO:0071840) | 5 | 11 |
| cellular process (GO:0009987) | 59 | 39 |
| developmental process (GO:0032502) | 7 | 6 |
| growth (GO:0040007) | 1 | 2 |
| localization (GO:0051179) | 20 | 7 |
| metabolic process (GO:0008152) | 88 | 49 |
| multi-organism process (GO:0051704) | 1 | — |
| multicellular organismal process (GO:0032501) | 3 | 4 |
| reproduction (GO:0000003) | 1 | 1 |
| reproductive process (GO:0022414) | 5 | 3 |
| response to stimulus (GO:0050896) | 37 | 11 |
| signalling (GO:0023052) | 7 | 1 |
| single-organism process (GO:0044699) | 56 | 32 |
| ▶ Cellular component | ||
| cell (GO:0005623) | 38 | 24 |
| cell junction (GO:0030054) | 1 | — |
| cell part (GO:0044464) | 38 | 25 |
| extracellular region (GO:0005576) | 3 | 6 |
| macromolecular complex (GO:0032991) | 2 | 4 |
| membrane (GO:0016020) | 20 | 20 |
| membrane part (GO:0044425) | 11 | 12 |
| membrane-enclosed lumen (GO:0031974) | 1 | — |
| nucleoid (GO:0009295) | 1 | — |
| organelle (GO:0043226) | 32 | 13 |
| organelle part (GO:0044422) | 12 | 4 |
| ▶ Molecular function | ||
| antioxidant activity (GO:0016209) | 2 | 2 |
| binding (GO:0005488) | 83 | 32 |
| catalytic activity (GO:0003824) | 81 | 43 |
| electron carrier activity (GO:0009055) | 8 | — |
| enzyme regulator activity (GO:0030234) | 1 | 5 |
| metallochaperone activity (GO:0016530) | 1 | — |
| molecular transducer activity (GO:0060089) | 1 | — |
| nucleic acid binding transcription factor activity (GO:0001071) | 17 | 6 |
| transporter activity (GO:0005215) | 8 | 3 |
Pathway analysis of common DEGs
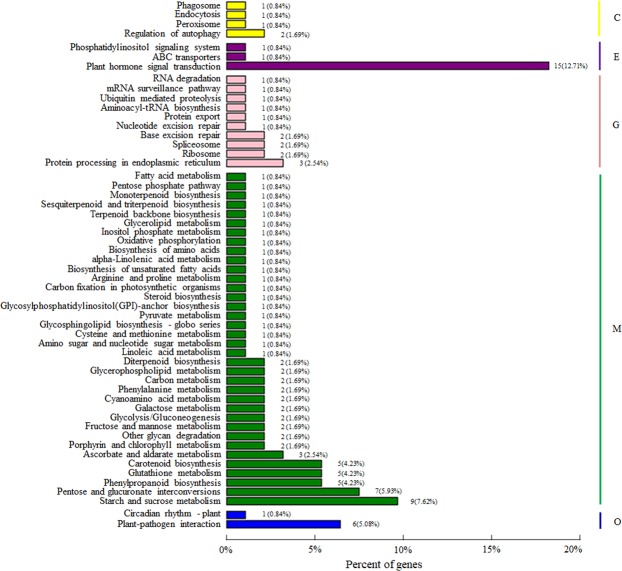
To identify the DEGs putatively involved in crosstalk between drought, cold and high-salinity stress regulation, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis was performed, and the biological functions of the common DEGs were further categorized (Fig. 2). The DEGs were mainly classified into ‘cellular processes’, ‘environmental information processing’, ‘genetic information processing’, ‘metabolism’, and ‘organismal systems’. Focusing on signal transduction components in pre-response crosstalk regulation, 17 DEGs in ‘environmental information processing’ were identified, including genes in the ‘phosphatidylinositol signalling system’, ‘ABC transporters’ and ‘plant hormone signal transduction’ pathways. ‘Plant hormone’ was the largest functional pathway, comprising a total of 15 hormone-related DEGs. Among the 17 DEGs, 15 were upregulated and 2 were downregulated by all three stresses, and some of these DEGs were classified as involved in the ABA, auxin, ethylene, and brassinolide (BR) hormone pathways. Seven of the 17 DEGs were members of the protein phosphatase 2 C family, which is predicted to play a role in the ABA regulation pathway (Table 2).
Figure 2.
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis based on the common differentially expressed genes (DEGs) involved in crosstalk between drought, cold and high-salinity stress regulation. C, cellular processes; E, environmental information processing; G, genetic information processing; M, metabolism; O, organismal systems.
Table 2.
Characters of shared differentially expressed genes (DEGs) between drought, cold and high-salinity stress responses classified in the ‘environmental information processing’ pathway.
| No. | Accession number | Gene name | Function | Protein length (amino acids) | Common regulation |
|---|---|---|---|---|---|
| 1 | MD06G1200300 | DGK1 | diacylglycerol kinase | 565 | up |
| 2 | MD15G1010400 | ABCB13 | ABC transporter | 479 | down |
| 3 | MD01G1139200 | PP2C-37a | protein phosphatase 2C | 430 | up |
| 4 | MD07G1203700 | PP2C-37b | protein phosphatase 2C | 423 | up |
| 5 | MD07G1291000 | PP2C-51a | protein phosphatase 2C | 388 | up |
| 6 | MD01G1220800 | PP2C-51b | protein phosphatase 2C | 354 | up |
| 7 | MD15G1195800 | PP2C-56 | protein phosphatase 2C | 443 | up |
| 8 | MD02G1084600 | PP2C-77a | protein phosphatase 2C | 418 | up |
| 9 | MD15G1212000 | PP2C-77b | protein phosphatase 2C | 446 | up |
| 10 | MD07G1297400 | SAUR32 | auxin-responsive protein | 134 | up |
| 11 | MD12G1113400 | SAUR40 | auxin-responsive protein | 149 | up |
| 12 | MD05G1082000 | ABI5-5a | ABA insensitive protein | 431 | up |
| 13 | MD15G1081800 | ABI5-5b | ABA insensitive protein | 419 | up |
| 14 | MD13G1213100 | ERF1b | ethylene-responsive transcription factor | 229 | up |
| 15 | MD06G1046300 | SAPK3 | serine/threonine-protein kinase | 341 | up |
| 16 | MD08G1102600 | BKI1 | BR receptor kinase inhibitor | 326 | down |
| 17 | MD11G1293900 | HPt3a | histidine-containing phosphotransfer | 152 | up |
ABA, abscisic acid; BR, brassinosteroids.
Validation and expression correlation of common DEGs
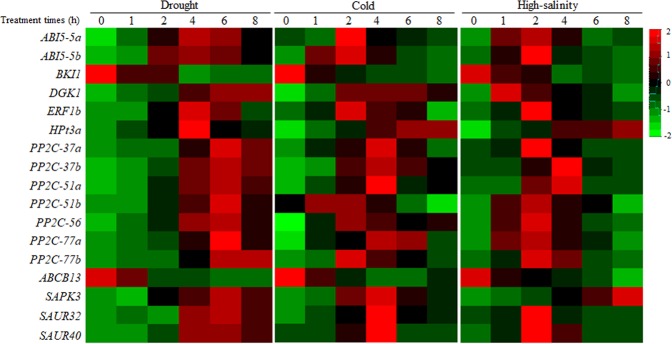
To validate the expression data obtained by RNA-seq, we performed qRT-PCR analysis of the expression of the 17 DEGs following the drought, cold and high-salinity treatments (primers are listed in Supplementary Table 3). In general, the data sets were congruent (Fig. 3). The expression of two downregulated genes, BKI1 and ABCB13, was strongly inhibited at 4 h by all three stresses, while the 15 other genes were upregulated. Based on these results, five upregulated genes, PP2C-37b, PP2C-77a, ABI5-5b, SAPK3, and HPt3a, were selected for further analysis.
Figure 3.
Expression patterns of common differentially expressed genes (DEGs) from drought-, cold- and high-salinity-stressed samples.
Effect of overexpression of five common DEGs on stress resistance genes
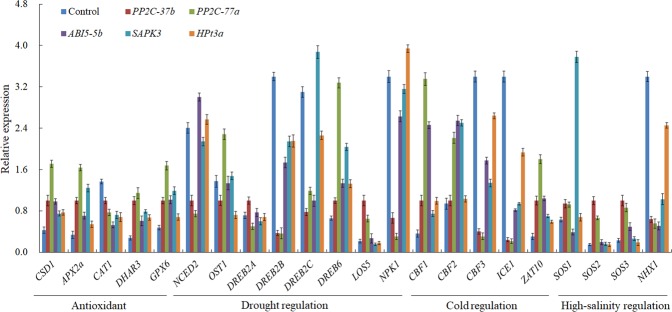
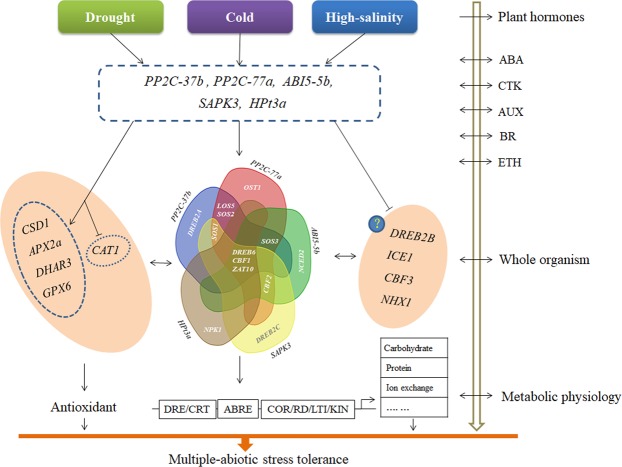
To explore potential crosstalk between the regulatory pathways of the common DEGs, five upregulated genes were individually transformed into apple calli, resulting in a 6- to 15-fold increase in expression, as determined by qRT-PCR (Supplementary Fig. 2). A total of 22 genes in four response pathways (antioxidant, drought, cold and high-salinity response pathways) were used to reflect regulation by the common DEGs (primers are listed in Supplementary Table 4). The elevated expression of PP2C-37b coincided with the upregulation of four antioxidant enzymatic genes (CSD1, APX2a, DHAR3, and GPX6), three drought-related genes (DREB2A, DREB6, LOS5), two cold-related genes (CBF1, ZAT10), and three high-salinity-related genes (SOS1, SOS2 and SOS3) (Fig. 4). Overexpression of PP2C-77a caused the upregulation of the above four antioxidant enzymatic genes, as well as three drought-related genes (OST1, DREB6, LOS5), three cold-related genes (CBF1, CBF2, ZAT10), and three high-salinity-related genes (SOS1, SOS2 and SOS3). In comparison, overexpression of ABI5-5b promoted the expression of the CSD1, APX2a, DHAR3 and GPX6 antioxidant enzyme genes, NCED2 and DREB6, which are related to drought stress, CBF1, CBF2, and ZAT10, which are related to cold stress, and SOS3, which is related to high-salinity stress. Furthermore, overexpression of SAPK3 induced the transcription of the CSD1, APX2a, DHAR3, and GPX6 antioxidant enzyme genes, DREB2C and DREB6, which are related to drought stress, CBF1, CBF2, and ZAT10, which are related to cold stress, and SOS1, which is related to high-salinity stress. Finally, overexpression of HPt3a caused the upregulation of CSD1, APX2a, DHAR3, GPX6, DREB6, NPK1, CBF1 and ZAT10, while CAT1, DREB2B, CBF3, ICE1 and NHX1 were all downregulated in response to the overexpression of all five genes.
Figure 4.
Effect of PP2C-37b, PP2C-77a, ABI5-5b, SAPK3, and HPt3a overexpression on regulatory genes involved in abiotic stress resistance. GenBank accession numbers: PP2C-37b (XM008373834.2); PP2C-77a (XM008390079.2); ABI5-5b (XM008379818.2); SAPK3 (NM001320015.1); HPt3a (XM008361181.2).
Overall, the regulation of common DEGs involved in drought, cold and high-salinity stress responses involved multiple pathways and complex regulation patterns within gene families, e.g., PP2C-77a promoted CBF1 and CBF2 expression but downregulated CBF3, while SOS1 expression decreased and expression increased SOS2 and SOS3 in ABI5-5b-overexpressing calli. Of the 22 genes involved in stress resistance, the expression of three genes (DREB6, CBF1 and ZAT10) consistently increased in response to the overexpression of all five common DEGs, implying cross-regulation downstream of those stress responses.
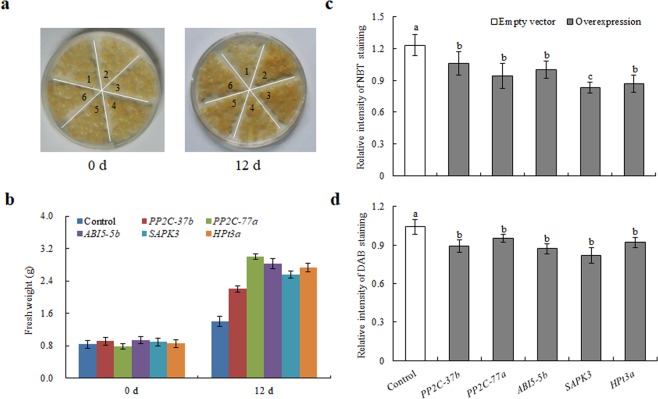
Effect of the overexpression of five common DEGs on the stress resistance of apple calli
To further examine the stress tolerance characteristics of these five common DEGs, transgenic apple calli were grown under 16 °C with drought and high-salinity stress simultaneously. After 12 days of cultivation, transgenic calli became dark yellow and gained more mass weight than the control (Fig. 5a,b). The PP2C-77a and ABI5-5b transgenic calli showed a better growth status than the other transgenic calli, indicating superior drought, cold and high-salinity stress resistance. In view of the upregulation of antioxidant enzyme genes, the transgenic calli with these five common DEGs were stained by nitroblue tetrazolium (NBT, for the detection of superoxides) and 3′,3′-diaminobenzidine (DAB, for the detection of H2O2) separately, and it was found that the overexpression of the five common DEGs promoted superoxide enzyme activities and decreased the H2O2 concentration (Fig. 5c,d). These results suggested that the elevation of antioxidant activity could be one pathway by which these five common DEGs confer comprehensive resistance to drought, cold and high-salinity stress.
Figure 5.
Effect of PP2C-37b, PP2C-77a, ABI5-5b, SAPK3, and HPt3a overexpression on the abiotic stress resistance of apple calli. (a) Apple calli transfected with an empty vector (1), PP2C-37b-overexpression calli (2), PP2C-77a-overexpression calli (3), ABI5-5b-overexpression calli (4), SAPK3-overexpression calli (5), and HPt3a-overexpression calli (6) were grown under 16 °C with 40% PEG8000 and 100 mM NaCl simultaneously. (b) Measurement of calli weight. Transfected apple calli were weighed before treatment (0 d) and after 12 d of treatment. (c) Relative intensity of NBT staining for transfected apple calli after treatment. (d) Relative intensity of DAB staining for transfected apple calli after treatment. Samples with different letters are significantly different P < 0.05.
Discussion
In addition to horticultural attributes such as tree vigour, earlier flowering, fruit quality and yield, the ability of appropriate rootstocks to survive abiotic stresses, including drought, cold or salinization, in order to influence scion cultivars in apple cultivation has been widely recognized25. The apple dwarfing rootstock ‘SH6’, which was used in this paper, was bred by Ralls × Wuxiang crabapple in Shanxi province, China, in the 1980s. After decades of field evaluations, SH6 was retained not only because of its comprehensive agronomic dwarf traits, fruit sugar-to-acid ratio and stable production but also for, in particular, its multiple abiotic stress tolerance to drought, cold, high-temperature, and other stresses26–28, which greatly expanded apple cultivation areas and provided suitable research material for abiotic stress crosstalk regulation mechanisms.
Transcriptome analysis has been widely used in studies of apples, including investigations of flower development29,30, fruit ripening31,32, postharvest storage33, and biotic and abiotic stresses such as drought or low-temperature stress34–37. However, most studies have only investigated transcription profiles or mechanisms under a single stress. In this study, we focused on the expression genes involved in the physiological responses of apple seedlings to drought, cold and high-salinity stress and identified 1,379, 3,552 and 2,312 DEGs that were upregulated and 1,208, 1,833 and 2,367 DEGs that were downregulated by these stresses, respectively. Cold stress resulted in more DEGs than drought and high-salinity stress, which has also been reported for maize23 and wheat38 but was distinct from findings in A. thaliana, where cold resulted in less DEGs than drought and high-salinity stresses19. In addition, more DEGs were induced by drought than cold in cassava (Manihot esculenta)39, while fewer DEGs were induced by drought than cold in Dianthus spiculifolius40, and more DEGs were upregulated in response to drought than salinity stress in poplar11. The transcription response difference in the numbers of down- and upregulated genes among plant species might be related to the resistance levels and complexity of stress response pathways in these plants.
We found evidence of crosstalk between drought, cold and high-salinity stress signalling, with 377 commonly upregulated and 211 commonly downregulated genes. GO classification of these genes revealed that most commonly regulated DEGs were associated with the ‘biological process’ category (Supplementary Fig. 1) and that the three stresses primarily affected genes assigned ‘metabolic processes’ and ‘stress responses’ terms. Phytohormones have been proposed to play key roles in stress responses and/or adaptation41. In a KEGG pathway analysis, we found that the largest portion of the common DEGs were from the ‘plant hormone signal transduction’ pathway (15 members), with only two other response pathways with one DEG each in the ‘environmental information processing’ category (17 members) (Fig. 2). Among the 15 DEGs, ABA-, auxin-, ethylene-, and BR-related functions were observed in the early responses to drought, cold and high-salinity stress.
ABA has been reported to control the expression of drought, cold and high-salinity stress responsive genes, and most of the transcripts for genes encoding enzymes involved in ABA biosynthesis displayed enhanced expression under drought, oxidative, and temperature stresses42. Our results suggest crosstalk pathways or potentially genes involved in the responses to all three stresses involving ABA signalling, including the PP2C and ABI5 families. In plants, members of the PP2C family have been implicated as positive regulators within ABA-mediated signalling networks activated by diverse environmental stresses or developmental signalling cascades43. Moreover, the bZIP transcription factor ABI5 has been shown to bind to the promoter regions of many stress-responsive genes, thereby controlling the expression of ABA-inducible genes17. With 9 out of 15 common DEGs identified in this study being related to ABA signalling, our results add to the information on signalling cross-regulation in response to abiotic stress.
Five out of these 15 DEGs that were upregulated in response to all three stresses (protein phosphatases PP2C-37b and PP2C-77a, ABA insensitive transcription factor ABI5-5b, serine/threonine-protein kinase SAPK3, and histidine-containing phosphotransfer HPt3a) were overexpressed in apple calli. We found that the elevated expression of these common DEGs modulated the expression of multiple genes known to be involved in stress tolerance (Fig. 4). The common DEGs were predominantly associated with antioxidant-, drought-, cold- or high-salinity- stress regulation pathways: PP2C-77a overexpression caused increased expression of the CSD1, APX2a, DHAR3, and GPX6 antioxidant enzymes and was involved in the drought-stress response through OST1, DREB6, LOS5 regulation, the cold-stress response through CBF1, CBF2, and ZAT10 upregulation, and the high-salinity-stress response through SOS1, SOS2 and SOS3 regulation. Comparatively, ABI5-5b overexpression caused upregulation of NCED2 and DREB6, which were involved in drought regulation, CBF1, CBF2, ZAT10, which were involved in cold regulation, and only SOS3, which was involved in high-salinity regulation. The results from transgenic apple calli in response to drought, cold and high-salinity stresses also suggested these five common DEGs were involved in multiple pathways and improved comprehensive resistance to stress.
In addition, we also observed examples of the suppression of abiotic stress-inducible genes, such as CAT1, DREB2B, CBF3, ICE1, and NHX1, by these five common DEGs. The overexpression of protein phosphatases or transcription factors by common DEGs could directly or indirectly decrease the expression of relative abiotic stress genes, by which compensation regulation, especially in gene families, could occur. The consistency of the expression patterns of CBF3 and its transcription factors ICE1 and ZAT10 and its putative target gene CSD1 indicated that the multi-functionality of the five common DEGs might be upstream of the stress genes detected.
As previous studies have shown, a systematic and organismal response occurs when plants suffer one or more bio/abiotic stress, mediated by Ca2+, ROS, phytohormones, and cellular, genetic or metabolic processing44,45. We only investigated the correlation of five co-upregulated genes with other well-identified resistance genes; however, the complexity of the regulatory networks was greater than expected (Fig. 6). The overexpression of these five DEGs promoted the expression of many genes in the antioxidant pathway, caused a decrease in CAT1 expression and consistent increase in the expression of DREB6, CBF1 and ZAT10, and had different regulatory effects on other genes. Positive functions on abiotic stress resistance genes, such as ICE1 and CBF3 for cold stress46,47 or NHX1 for high-salinity stress48, were not well reflected under the overexpression of the five common DEGs, showing that specificity and coordination coexisted in multiple regulatory pathways.
Figure 6.
Crosstalk between drought, cold and high-salinity stress pathways linked to five common differentially expressed genes (DEGs) in apple. ABA, abscisic acid; CTK, cytokinins; AUX, auxin; BR, brassinolide.
Taken together, the comparative transcriptome analysis revealed 377 commonly upregulated and 211 commonly downregulated DEGs involved in the crosstalk between drought, cold and high-salinity stress in apple. GO classification and KEGG pathway analyses indicated that these common DEGs participate in various pathways related to signal transduction, antioxidants, metabolism, growth and developmental processes. Seventeen DEGs from the ‘environmental information processing’ pathway were selected for qRT-PCR analysis, five of which were overexpressed in apple calli and shown to regulate well-known genes and improve the abiotic stress resistance of transgenic calli. Overall, our study provided new insights into the crosstalk mechanism involved in drought, cold and high-salinity stress responses in apple and laid a reference for the investigation of other common DEGs for resistance breeding and functional studies.
Materials and Methods
Plant material and stress treatments
In vitro plantlets of the dwarf rootstock, Malus × domestica cv. ‘SH6’, were cultured on Murashige and Skoog (MS) medium supplemented with 1.0 mg/L 6-benzylaminopurine (6-BA), 0.4 mg/L 3-indolebutyric acid (IBA), 30 g/L sucrose and 7.0 g/L agar at 25 °C under a 16-h light/8-h dark photoperiod. After 15 days of rooting cultivation on MS medium (1/2 MS, 0.4 mg/L IBA), uniformly developing plants were transferred to stress treatment conditions. Drought and cold stress treatments were carried out with 40% PEG8000 and at 4 °C, respectively, as previously reported49. Plantlets cultured on the same MS medium, with or without 150 mM NaCl, were used for the high-salinity treatment and the control, respectively. All treatments were carried out for 4 hours, and leaves were harvested in three biological replicates and immediately frozen in liquid nitrogen.
Apple fruit calli (cv. Orin) were grown on MS medium supplemented with 0.5 mg/L indole-3-acetic acid (IAA) and 1.5 mg/L 6-BA at 25 °C in the dark and used for Agrobacterium tumefaciens infection, as described by Li et al.50. Transgenic calli were grown on the same medium at 16 °C supplemented with 40% PEG8000 and 100 mM NaCl for stress treatment.
RNA extraction, library construction, and RNA-seq analysis
Total RNA from leaf samples of treated plantlets was extracted using the EASYspin plant RNA extraction kit (Biomed, China), and the integrity, concentration and purity of each sample were evaluated using 1% agarose gel electrophoresis and a NanoPhotometer spectrophotometer (IMPLEN, CA, USA). RNA-seq libraries were constructed according to the manuals provided by Illumina Inc. (San Diego, USA) and were sequenced on an Illumina HiSeq 2500 platform (Biomarker Technology Co.), which generated raw data of 150-bp paired-end reads. Mixed cDNA libraries from ten uniformly sized in vitro apple plants from each of the drought, cold, and high-salinity treatments and a control were used for RNA-seq.
Mapping and annotation of sequencing reads
After quality assessment and data filtering, clean reads were obtained by removing the reads containing the adapter and low-quality reads, and these were then mapped to the Malus × domestica genome (GDDH13 Version 1.1) using HISAT2 software51 and de novo assembled using the paired-end method and StringTie software52. Subsequently, unigenes were functionally annotated using diverse protein databases, including the National Center for Biotechnology Information (NCBI) non-redundant protein (NR) database, the Swiss-Prot database, the Gene Ontology (GO) database, the Clusters of Orthologous Groups of proteins (COG) database, the EuKaryotic Orthologous Groups (KOG) database, the Protein Family (Pfam) database, and the Kyoto Encyclopedia of Genes and Genomes (KEGG) database. The gene coding sequences (CDS) were predicted with TransDecoder software (http://transdecoder.github.io).
DEG analysis
Gene expression levels were analysed using fragments per kilobase of the transcript per million mapped reads (FPKM) method53. DESeq software was used to identify DEGs in pair-wise comparisons54, and the results of all statistical tests were revised to account for multiple testing with the Benjamini–Hochberg false discovery rate (FDR < 0.01). Sequences were determined to be significantly differentially expressed at a P value of <0.01 and a fold change (FC) of >2.0. DEGs shared between the three treatments were then analysed using KEGG pathway enrichment analysis, and the sequences of DEGs of interest were retrieved from the apple genome sequence (https://iris.angers.inra.fr/gddh13/).
Genetic transformation of apple calli
To investigate the crosstalk between genes involved in multiple abiotic stress resistance, the complete CDS of one of the five selected common DEGs was inserted into the pCAMBIA1304 vector for overexpression and transformed into A. tumefaciens strain EHA105. The preparation of the infection suspension and overexpression of DEGs in apple calli were performed as previously described55.
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted using the EASYspin plant RNA extraction kit (Biomed, China), and first-strand cDNAs were synthesized using M-MLV reverse transcriptase (Promega, USA) according to the manufacturers’ instructions. The 2 × SYBR-Green I RT-PCR Master Mix (Takara, Japan) was used as the labelling agent, and qRT-PCR was performed on a Bio-RAD C1000TM Thermal Cycler with a CFX384TM Real Time System (Bio-Rad, USA). The reaction mixture (10 µL) contained 5 µL 2 × Master Mix, 10 µmol·L−1 forward and reverse primers (0.5 µL each), 0.8 µL template cDNA, and 3.2 µL ddH2O. The PCR program was 95 °C for 1 min, followed by 40 cycles at 94 °C for 20 s, 60 °C for 20 s and 72 °C for 20 s. Three independent biological replicates were analysed for each sample, and β-Actin (XM008356922) was amplified along with the target genes as the internal control. A heat map was generated using custom scripts in R Project for Statistical Computing v3.3.1.
Histochemical staining
Transgenic calli were stained with NBT for superoxide activity analysis and with DAB for H2O2 concentration detection in accordance with the methods of Hu et al.56. For NBT staining, 2 mM NBT (Sigma, Germany) solution was prepared in 20 mM phosphate buffer (pH 7.0). In addition, 1.0 mg/ml DAB (Sigma, Germany) was prepared in ddH2O and adjusted to pH 6.0 with Tris–HCl buffer (pH 7.5). Calli were incubated in NBT solution for 3 h and in DAB solution for 8 h separately in darkness and then kept in a solution of 3:1:1 ethanol:lactic acid:glycerol and photographed.
Statistical analysis
All experiments were performed with at least three biological replicates and three technical replicates. Data are presented as the mean ± SD. Statistical differences between groups were evaluated using Duncan’s multiple-range test. P values < 0.05 were considered statistically significant.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Supplementary information
Acknowledgements
This work was financially supported by the Science and Technology Innovation Ability Construction Projects of Beijing Academy of Agriculture and Forestry Science (KJCX20180701) and the Ministry of Agriculture of the People’s Republic of China (CARS-27).
Author Contributions
The project was designed and conceived by Xingliang Li and Junke Zhang. The experimental work was carried out by Xingliang Li, Minji Li, Beibei Zhou and Yuzhang Yang; Xingliang Li and Qinping Wei analysed the data. Xingliang Li wrote the manuscript. All authors have read and approved the final version of this manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-45266-0.
References
- 1.Wu YQ, et al. Comparative transcriptome profiling of a desert evergreen shrub, Ammopiptanthus mongolicus, in response to drought and cold stresses. BMC Genomics. 2014;15:671. doi: 10.1186/1471-2164-15-671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang GT, et al. Signal transduction during cold, salt, and drought stresses in plants. Mol Biol Rep. 2012;39:969–987. doi: 10.1007/s11033-011-0823-1. [DOI] [PubMed] [Google Scholar]
- 3.Turkan I. Emerging roles for ROS and RNS–versatile molecules in plants. J Exp Bot. 2017;68:4413–4416. doi: 10.1093/jxb/erx236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haak DC, et al. Multilevel regulation of abiotic stress responses in plants. Front Plant Sci. 2017;8:1–24. doi: 10.3389/fpls.2017.01564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shinozaki K, Yamaguchi-Shinozaki K. Gene networks involved in drought stress response and tolerance. J Exp Bot. 2006;58:221–227. doi: 10.1093/jxb/erl164. [DOI] [PubMed] [Google Scholar]
- 6.Chinnusamy V, Zhu JH, Zhu JK. Cold stress regulation of gene expression in plants. Trends Plant Sci. 2007;12:444–451. doi: 10.1016/j.tplants.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 7.Ji HT, et al. The salt overly sensitive (SOS) pathway: established and emerging roles. Mol Plant. 2013;6:275–286. doi: 10.1093/mp/sst017. [DOI] [PubMed] [Google Scholar]
- 8.Hossain MA, Mostofa MG, Fujita M. Cross protection by cold-shock to salinity and drought stress-induced oxidative stress in mustard (Brassica campestris L.) seedlings. Mol. Plant Breeding. 2013;4:50–70. [Google Scholar]
- 9.Oliveira TMD, et al. Analysis of the NAC transcription factor gene family in citrus reveals a novel member involved in multiple abiotic stress responses. Tree Genet Genomes. 2011;7:1123–1134. doi: 10.1007/s11295-011-0400-8. [DOI] [Google Scholar]
- 10.Zandkarimi H, Ebadi A, Salami SA, Alizade H, Baisakh N. Analyzing the expression profile of areb/abf and dreb/cbf genes under drought and salinity stresses in grape (vitis vinifera l.) Plos One. 2015;10:e0134288. doi: 10.1371/journal.pone.0134288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yoon SK, et al. Response to drought and salt stress in leaves of poplar (populus alba × populus glandulosa): expression profiling by oligonucleotide microarray analysis. Plant Physiol Bioch. 2014;84:158–168. doi: 10.1016/j.plaphy.2014.09.008. [DOI] [PubMed] [Google Scholar]
- 12.Wang ML, et al. Overexpression of a Camellia sinensis DREB transcription factor gene (CsDREB) increases salt and drought tolerance in transgenic Arabidopsis thaliana. J Plant Biol. 2017;60:452–461. doi: 10.1007/s12374-016-0547-9. [DOI] [Google Scholar]
- 13.Seki M, Narusaka M, Ishida J, Nanjo T, Fujita M. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. The Plant J. 2002;31:279–292. doi: 10.1046/j.1365-313X.2002.01359.x. [DOI] [PubMed] [Google Scholar]
- 14.Li PC, et al. Transcriptomic profiling of the maize (zea mays l.) leaf response to abiotic stresses at the seedling stage. Front Plant Sci. 2017;8:290. doi: 10.3389/fpls.2017.00290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou ML, et al. Improvement of drought and salt tolerance in Arabidopsis and Lotus corniculatus by overexpression of a novel DREB transcription factor from Populus euphratica. Gene. 2012;506:10–17. doi: 10.1016/j.gene.2012.06.089. [DOI] [PubMed] [Google Scholar]
- 16.Miao YC, et al. An arabidopsis glutathione peroxidase functions as both a redox transducer and a scavenger in abscisic acid and drought stress responses. Plant Cell. 2006;18:2749–2766. doi: 10.1105/tpc.106.044230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhou XN, et al. Molecular characterization of an ABA insensitive 5 orthologue in Brassica oleracea. Biochem Biophys Res Commun. 2013;430:1140–1146. doi: 10.1016/j.bbrc.2012.12.023. [DOI] [PubMed] [Google Scholar]
- 18.Wang QH, et al. Transcriptome analysis around the onset of strawberry fruit ripening uncovers an important role of oxidative phosphorylation in ripening. Sci Rep. 2017;7:41477. doi: 10.1038/srep41477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rehman HM, et al. Comparative genomic and transcriptomic analyses of Family-1 UDP glycosyltransferase in three Brassica species and Arabidopsis indicates stress-responsive regulation. Sci Rep. 2018;8:1875. doi: 10.1038/s41598-018-19535-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kumar D, Hazra S, Datta R, Chattopadhyay S. Transcriptome analysis of Arabidopsis mutants suggests a crosstalk between aba, ethylene and GSH against combined cold and osmotic stress. Sci Rep. 2016;6:36867. doi: 10.1038/srep36867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Matsui A, et al. Arabidopsis transcriptome analysis under drought, cold, high-salinity and ABA treatment conditions using a tiling array. Plant Cell Physiol. 2008;49:1135–1149. doi: 10.1093/pcp/pcn101. [DOI] [PubMed] [Google Scholar]
- 22.Sham A, et al. Transcriptome analysis reveals genes commonly induced by botrytis cinerea infection, cold, drought and oxidative stresses in arabidopsis. PLoS One. 2014;9:e113718. doi: 10.1371/journal.pone.0113718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shan XH, et al. Transcriptome profile analysis of maize seedlings in response to high-salinity, drought and cold stresses by deep sequencing. Plant Mol Biol Rep. 2013;31:1485–1491. doi: 10.1007/s11105-013-0622-z. [DOI] [Google Scholar]
- 24.Rabbani MA. Monitoring expression profiles of rice genes under cold, drought, and high-salinity stresses and abscisic acid application using cDNA microarray and RNA gel-blot analyses. Plant Physiol. 2003;133:1755–1767. doi: 10.1104/pp.103.025742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marini RP, Fazio G. Apple rootstocks: history, physiology, management, and breeding. Hortic Rev. 2018;45:197–312. [Google Scholar]
- 26.Li XL, et al. Influence of six dwarfing interstocks on the ‘Fuji’ apple under drought stress. Indian J Hortic. 2017;74:346–350. doi: 10.5958/0974-0112.2017.00069.X. [DOI] [Google Scholar]
- 27.Wang YX, et al. Physiological mechanisms of resistance to cold stress associated with 10 elite apple rootstocks. J IntegrAgr. 2018;17:857–866. [Google Scholar]
- 28.Zhou BB, et al. Dwarfing apple rootstock responses to elevated temperatures: A study on plant physiological features and transcription level of related genes. J IntegrAgr. 2016;15:1025–1033. [Google Scholar]
- 29.Xing LB, et al. Transcription profiles reveal sugar and hormone signaling pathways mediating flower induction in apple (Malus domestica Borkh) Plant Cell Physiol. 2015;56:2052–2068. doi: 10.1093/pcp/pcv124. [DOI] [PubMed] [Google Scholar]
- 30.Liu KD, et al. Transcriptome analysis and identification of genes associated with floral transition and flower development in sugar apple (Annona squamosa L.) Front Plant Sci. 2016;7:1695. doi: 10.3389/fpls.2016.01695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Costa F, et al. Use of homologous and heterologous gene expression profiling tools to characterize transcription dynamics during apple fruit maturation and ripening. BMC Plant Biol. 2010;10:229. doi: 10.1186/1471-2229-10-229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.El-Sharkawy I, Liang D, Xu K. Transcriptome analysis of an apple (Malus × domestica) yellow fruit somatic mutation identifies a gene network module highly associated with anthocyanin and epigenetic regulation. J Exp Bot. 2015;66:7359–7376. doi: 10.1093/jxb/erv433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mellidou I, et al. Transcriptomic events associated with internal browning of apple during postharvest storage. BMC Plant Biol. 2014;14:328. doi: 10.1186/s12870-014-0328-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Porto DD, et al. Transcription profiling of the chilling requirement for bud break in apples: a putative role for FLC-like genes. J Exp Bot. 2015;66:2659–2672. doi: 10.1093/jxb/erv061. [DOI] [PubMed] [Google Scholar]
- 35.Du F, Xu JN, Li D, Wang XY. The identification of novel and differentially expressed apple-tree genes under low-temperature stress using high-throughput Illumina sequencing. Mol Biol Rep. 2015;42:569–580. doi: 10.1007/s11033-014-3802-5. [DOI] [PubMed] [Google Scholar]
- 36.Zhu LM, et al. Transcriptomics analysis of apple leaves in response to alternaria alternata apple pathotype infection. Front Plant Sci. 2017;8:22. doi: 10.3389/fpls.2017.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Balan B, Marra FP, Caruso T, Martinelli F. Transcriptomic responses to biotic stresses in Malus x domestica: a meta-analysis study. Sci Rep. 2018;8:1970. doi: 10.1038/s41598-018-19348-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang JY, et al. Identification of wheat stress-responding genes and TaPR-1-1function by screening a cDNA yeast library prepared following abiotic stress. Sci Rep. 2019;9:141. doi: 10.1038/s41598-018-37859-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li SX, et al. Global gene expression analysis reveals crosstalk between response mechanisms to cold and drought stresses in cassava seedlings. Front Plant Sci. 2017;8:1259. doi: 10.3389/fpls.2017.01259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhou AM, et al. Transcriptome sequencing of dianthus spiculifolius and analysis of the genes involved in responses to combined cold and drought stress. Inter J Mol Sci. 2017;18:849. doi: 10.3390/ijms18040849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fahad S, et al. Phytohormones and plant responses to salinity stress: a review. Plant Growth Regul. 2015;75:391–404. doi: 10.1007/s10725-014-0013-y. [DOI] [Google Scholar]
- 42.Tiwari S, Lata C, Chauhan PS, Prasad V, Prasad M. A functional genomic perspective on drought signalling and its crosstalk with phytohormone-mediated signalling pathways in plants. Curr Genomics. 2017;18:469–482. doi: 10.2174/1389202918666170605083319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xue TT, et al. Genome-wide and expression analysis of protein phosphatase 2C in rice and Arabidopsis. BMC Genomics. 2008;9:550. doi: 10.1186/1471-2164-9-550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhu YM, Shao J, Zhou Z, Davis RE. Comparative transcriptome analysis reveals a preformed defense system in apple root of a resistant genotype of G.935 in the absence of pathogen. Int J Plant Genom. 2017;2017:1–14. doi: 10.1155/2017/8950746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Das P, Majumder AL. Transcriptome analysis of grapevine under salinity and identification of key genes responsible for salt tolerance. Funct Integr Genomics. 2019;19:61–73. doi: 10.1007/s10142-018-0628-6. [DOI] [PubMed] [Google Scholar]
- 46.Mahajan S, Tuteja N. Cold, salinity and drought stresses: An overview. Arch Biochem Biophys. 2005;444:139–158. doi: 10.1016/j.abb.2005.10.018. [DOI] [PubMed] [Google Scholar]
- 47.Yin Y, et al. Functional Analysis of CsCBF3 transcription factor in tea plant (Camellia sinensis) under cold stress. Plant Growth Regul. 2016;80:335–343. doi: 10.1007/s10725-016-0172-0. [DOI] [Google Scholar]
- 48.Li YH, et al. Overexpression of a Malus vacuolar Na+/H+ antiporter gene (MdNHX1) in apple rootstock M.26 and its influence on salt tolerance. Plant Cell Tiss Org. 2010;102:337–345. doi: 10.1007/s11240-010-9738-0. [DOI] [Google Scholar]
- 49.Li XL, et al. Genome-wide analysis of antioxidant enzyme gene families involved in drought and low-temperature responses in Apple (Malus domestica) J Hortic Sci Biotechnol. 2018;93:337–346. doi: 10.1080/14620316.2017.1382314. [DOI] [Google Scholar]
- 50.Li T, et al. Apple (Malus domestica) MdERF2 negatively affects ethylene biosynthesis during fruit ripening by suppressing mdacs1 transcription. Plant J. 2016;88:735–748. doi: 10.1111/tpj.13289. [DOI] [PubMed] [Google Scholar]
- 51.Kim D, Langmead B, Salzberg SL. HISAT: a fast spliced aligner with low memory requirements. Nat Methods. 2015;12:357–360. doi: 10.1038/nmeth.3317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pertea M, et al. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol. 2015;33:290–295. doi: 10.1038/nbt.3122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Florea L, Song L, Salzberg SL. Thousands of exon skipping events differentiate among splicing patterns in sixteen human tissues. F1000Research. 2013;2:188. doi: 10.12688/f1000research.2-188.v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang LK, Feng ZX, Wang X, Wang XW, Zhang XG. DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics. 2010;26:136–138. doi: 10.1093/bioinformatics/btp612. [DOI] [PubMed] [Google Scholar]
- 55.Xie XB, et al. The bHLH transcription factor MdbHLH3 promotes anthocyanin accumulation and fruit colouration in response to low temperature in apples. Plant Cell Environ. 2012;35:1884–1897. doi: 10.1111/j.1365-3040.2012.02523.x. [DOI] [PubMed] [Google Scholar]
- 56.Hu YM, et al. ACCERBATIN, a small molecule at the intersection of auxin and reactive oxygen species homeostasis with herbicidal properties. J Exp Bot. 2017;68:4185–4203. doi: 10.1093/jxb/erx242. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.