Abstract
Background
A multifactorial decision support system (mDSS) is a tool designed to improve the clinical decision-making process, while using clinical inputs for an individual patient to generate case-specific advice. The study provides an overview of the literature to analyze current available mDSS focused on prostate cancer (PCa), in order to better understand the availability of decision support tools as well as where the current literature is lacking.
Methods
We performed a MEDLINE literature search in July 2018. We divided the included studies into different sections: diagnostic, which aids in detection or staging of PCa; treatment, supporting the decision between treatment modalities; and patient, which focusses on informing the patient. We manually screened and excluded studies that did not contain an mDSS concerning prostate cancer and study proposals.
Results
Our search resulted in twelve diagnostic mDSS; six treatment mDSS; two patient mDSS; and eight papers that could improve mDSS.
Conclusions
Diagnosis mDSS is well represented in the literature as well as treatment mDSS considering external-beam radiotherapy; however, there is a lack of mDSS for other treatment modalities. The development of patient decision aids is a new field of research, and few successes have been made for PCa patients. These tools can improve personalized medicine but need to overcome a number of difficulties to be successful and require more research.
1. Introduction
Worldwide, prostate cancer (PCa) is the second most occurring type of cancer in men and the most commonly diagnosed cancer for men living in developed countries, making it a very relevant topic for cancer research [1].
A variety of treatment options is available to treat PCa, such as external-beam radiotherapy (EBRT) and radical prostatectomy [2–4], which have similar long-term survival outcomes. Other treatments such as brachytherapy [5–8] are gaining popularity, and active surveillance is an increasingly viable option as well [9, 10], due to the slow progression of some kinds of PCa. Retrospective studies comparing different treatment modalities tend to be conflicting and biased. Consensus on the best treatment choices for men with PCa remains absent because prospective trials for different treatments report different toxicities [4, 11, 12]. Due to this, the treatment choice is largely dependent on both patient and physician subjective preferences, rather than knowledge-based decision-making [13]. Additionally, treatment outcome is dependent on a large number of features, including treatment, patient, tumor, clinical, and genetic features [14]. These factors further complicate the integration of evidence-based decision-making into clinical practice due to the limitations of human cognitive capacity, which can only take a relatively small number of factors into account on which to base a decision [15, 16]. In order to meet the aspiration of personalized medicine, the need for multifactorial decision support systems (mDSS) is growing [17–23]. An mDSS is a tool designed to improve the difficult medical decision-making process. It uses multifactorial inputs (treatment, patient, tumor, clinical, genetic, etc.) for a given patient to generate case-specific advice for patients, clinicians, or other medical professionals. Due to the variety of treatment options for PCa, all equally efficacious for outcome, but having different secondary effects, this disease is an interesting subject for the use of mDSS.
In addition to the need for mDSS for treatment selection, similar systems can be used for the diagnosis of PCa, improving early detection as well as reducing overdiagnosis and unnecessary testing. These mDSS can use imaging, clinical, biological, and other parameters to improve detection and risk classification of PCa in a minimally invasive method to maximize individual treatment.
This study provides an overview of the literature to analyze current available mDSS focused on PCa, in order to better understand the availability of mDSS as well as noting where the current literature is lacking. We aim to provide an update for clinicians about recent advances in mDSS for personalized PCa oncology, which may improve clinical decision-making.
2. Materials and Methods
2.1. Search Strategy
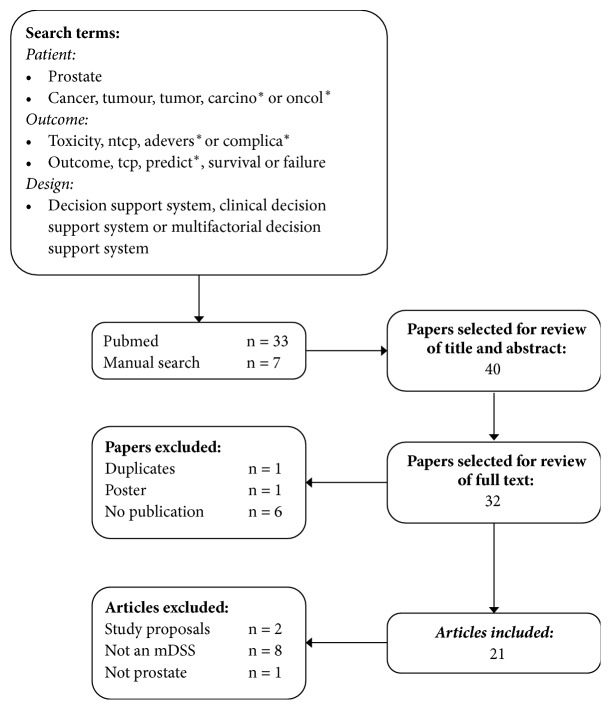
In order to identify all mDSS with relation to treatment for PCa, we performed a MEDLINE/PubMed literature search in July 2018, restricted to English. Details of the strategy we used are shown in Figure 1.
Figure 1.
Flowchart of the results of the literature search in PubMed.
2.2. Selection Criteria
Prior to reviewing full texts, we manually checked the abstracts and titles to select papers for this study. Duplicates, posters, or abstracts that did not include a published work were excluded. Additionally, we excluded studies which clearly did not include an mDSS for PCa. After full text review, we excluded any papers that described study proposals that did not describe an mDSS in PCa. We selected appropriate studies by manually screening and considering the aforementioned criteria.
2.3. Study Characteristics
The studies we included were divided into sections according to the type of mDSS: diagnostic mDSS, which support the staging of PCa or support the decision for more invasive or expensive diagnostic tests; treatment mDSS, which support the decision between treatment modalities or treatment plans; and patient mDSS, which focus on informing the patient.
We described each study using the number of patients, the decision that the system supports, and the system inputs and outcomes. We also commented on the general applicability, the reported performance of the system, and the limitations. In order to assess the reporting quality of the studies, we tested each paper for its compliance to the TRIPOD (Transparent Reporting of studies on prediction models for Individual Prognosis Or Diagnosis) reporting guideline [24]. We reported this in a percentage calculated using the document: “Appendix I: Scoring adherence of prediction model study reports to the TRIPOD reporting guideline”, available on http://www.tripod-statement.org/ (available here).
3. Results
3.1. Included Studies
3.1.1. mDSS for Diagnosis and Diagnostic Interventions
The studies that contained a diagnostic mDSS are listed in Table 1. Two of the studies we found had the goal of supporting the use of a diagnostic tool: Lee et al. (2010) and Mukai et al. (2013). The application tested by Mukai et al. (2013) was meant to support the use of prostate specific antigen (PSA) tests for patients. The mDSS was a web-based application that would aid in the decision to perform PSA tests for general practitioners (GPs) in Denmark. The study leads to the conclusion that it was possible to grant GPs in Denmark easy access to web-based mDSS by replacing certain words in their medical records by hyperlinks. However it also showed that this mDSS did not change PSA-testing behavior. Since this study neither developed nor validated this mDSS, the compliance to the TRIPOD guidelines could not be tested.
Table 1.
Overview of diagnosis support systems.
| Study | N | Decision/Diagnose | Inputs | Outcomes | TRIPOD |
|---|---|---|---|---|---|
| (Roach et al. 1994)[25] | 282 | Low or high risk of LN involvement | PSA,GS, Clinical stage | P LN involvement | 79% |
|
| |||||
| (Diaz et al. 1994)[26] | 217 | Low or high risk of SV involvement | PSA, GS | P SV involvement | 69% |
|
| |||||
| (D'Amico et al. 1998)[27] | 1872 | Patient risk group | PSA, GS, Clinical stage | 5-year PSA outcome | 72% |
|
| |||||
| (Chang et al. 1999)[28] | 43 | Localized vs Advanced PCa | PSA, GS, TRUS, DRE | P advanced PCa and P localized PCa | 65% |
|
| |||||
| (Roach et al. 2000)[29] | 895 | Extracapsular extension | PSA, GS | Extracapsular extension risk | 92%∗∗ |
|
| |||||
| (Lee et al. 2010)[30] | 1077 | Biopsy | Clinical∗, Imaging∗, PSA | P PCa | 86% |
|
| |||||
| (Kim et al. 2011)[31] | 532 | Advanced PCa | TRUS, Clinical, PSA | P advanced PCa | 79% |
|
| |||||
| (Shah et al. 2012)[32] | 31 | Location PCa | MRI image | Cancer probability map | 83% |
|
| |||||
| (Mukai et al. 2013)[33] | 30773 | PSA test | Clinical | Recommendation | -∗∗∗ |
|
| |||||
| (Sadoughi et al. 2014)[34] | 360 | PCa | PSA, Age | P PCa | 33% |
|
| |||||
| (van Leeuwen et al. 2017)[35] | 591 | Significant PCa | Clinical, PSA, PIRADS, DRE | P significant PCa | 97% |
|
| |||||
| (Tosoian, et al. 2017)[36] | 4459 | Pathological Stage | PSA, GS, Clinical stage | % likelihood of given stage | 83% |
Abbreviations. N: number of patients; P: probability; PCa: prostate cancer; PSA: prostate specific antigen; PIRADS: Prostate Imaging Reporting and Data System; TRUS: transrectal ultrasound scan; LN: lymph node; GS: Gleason score; SV: seminal vesicles; TRIPOD: adherence to the TRIPOD statement; DRE: digital rectal examination.
∗Clinical, imaging, and tumor parameters.
∗∗Abstract only.
∗∗∗No development or validation of mDSS: no TRIPOD evaluation possible.
Lee et al. (2010) attempted to support the use of a biopsy by predicting initial biopsy outcomes through three different models: support vector machine (SVM), artificial neural network (ANN), and multiple logistic regression. The study trained each of the models on 600 patients who had undergone transrectal ultrasonography (TRUS)-guided prostate biopsies, tested them on 477, and compared the model performances. The parameters of the models were TRUS findings and clinical parameters, including age and PSA. The area under the receiver operating characteristic (ROC) curve (AUC) for the use of multiple logistic regression analysis, ANN, and the SVM was 0.768, 0.778, and 0.847, respectively, and pairwise comparison of the ROC curves showed that the SVM model had superior performance.
Kim et al. (2011), Sadoughi et al. (2014), Shah et al. (2012), van Leeuwen et al. (2017), and Chang et al. (1999) all aimed to detect, diagnose, or classify PCa using a variety of methods. Kim et al. (2011) performed a study similar to Lee et al. (2010), but with the aim of improving pathological staging, rather than reducing the number of biopsies. Two models were developed, SVM and ANN, for the prediction of advanced PCa and compared based on performance. The models used TRUS-guided biopsy parameters and were tested on 532 patients divided into training and test groups. The SVM model performed significantly better (p=0.02) than the ANN model based on ROC curve, with an AUC of 0.805 while that of the ANN model was 0.719. This study showed that these models could improve objective pathological staging of biopsy-proven PCa patients and could be applied in combination with TRUS-guided biopsies once externally validated.
Another neural network was trained on laboratory results by Sadoughi et al. (2014) who then performed particle swarm optimization. The specific goal of this study was to aid in distinguishing between localized PCa and benign hyperplasia of the prostate. The model was internally validated on 60 patients, and the authors found an accuracy of 98.33%. The description of the methodology was limited, and the reporting conformed only to 33% of the TRIPOD guidelines. The model could potentially improve detection of PCa and possibly reduce the number of biopsies, but external validation is necessary.
Notable were Shah et al. (2012), who also used SVM, but, in contrast to Lee et al. (2011), the model was not trained on biopsy results, but on pathological regions of a magnetic resonance imaging (MRI) scan of postprostatectomy prostates. The aim was not only to diagnose PCa, but to locate it specifically on MRI scans by modeling voxel specific risk analyses. The sensitivity and the specificity of the model with optimized SVM parameters were 90%, and the kappa coefficient was 80%, where the raters were the mDSS and the ground truth histology. The study only included 24 patients, but since the model was trained on specific regions, the training was done on 225 cancer and 264 noncancerous regions. This model could be applied in any hospital with a 3.0 T endorectal multiparametric magnetic resonance imaging (mpMRI) scanner, although it still requires validation.
Van Leeuwen et al. (2017) developed a nomogram, rather than a deep learning algorithm, that included a larger number of parameters to diagnose significant PCa. The nomogram included Prostate Imaging Reporting and Data System (PIRADS), age, PSA, digital rectal examination (DRE), prostate volume, and prior biopsy. The model performed with an AUC of 0.864 on an external validation set, and the paper proposed an optimal strategy to reduce the number of biopsies needed with minimal risk of underdiagnosis. This paper conformed to 97% of the TRIPOD guidelines.
Chang et al. (1999) evaluated the usefulness and the performance of an mDSS, the Prostate Cancer Expert System (PCES), which was validated on 43 patients with confirmed PCa. The PCES system, which utilized PSA, Gleason score, TRUS, and DRE, was used to categorize the patients into localized or advanced PCa, and the same test was applied to four attending physicians and four residents. The results showed that the PCES performed with a higher accuracy than all residents and physicians, though the difference was only higher for two physicians. It also showed that after consultation of the PCES, the staging accuracy of the residents improved to the level of the attending physicians.
A number of prediction tools are currently being applied in the clinic to aid in the further diagnosis of the PCa disease, providing predictions on lymph node (LN) involvement, organ confinement (OC), seminal vesicle (SV) involvement and extracapsular extension, and risk of failure after treatment. The Partin tables [37, 38] are a set of nomograms to predict OC of PCa, initially introduced in 1993 and most recently updated by Tosoian et al. (2017) [36]. Based on these nomograms, Roach derived a set of formulas for the prediction of SV involvement in a paper published by Diaz et al. (1994) [26]. Roach et al. (1994) [25] also derived formulas that predicted LN involvement based on PSA and Gleason score, and Roach et al. (2000) [29] did the same for the risk of failure following radiotherapy (RT) and extracapsular extension in patients with localized PCa.
Tosoian et al. (2017) validated and updated the Partin tables on a cohort of 4459 patients with the goal of predicting the pathological outcome after radical prostatectomy. The performance of the model was tested for binary regression where the AUC was calculated when comparing organ confined (OC) PCa to other pathological outcomes. The model performed best when predicting OC versus LN involvement (AUC = 0.918) and versus seminal vesicle (SV) involvement (AUC = 0.856). The weakest performance was for OC versus extraprostatic extension (AUC = 0.673).
Diaz et al. (1994) split patients into high risk and low risk groups of SV involvement using PSA and Gleason score and tested this on 217 patients. The incidence rate of SV involvement in the low risk group was 7%, while the incidence rate in the high risk group was 37%, resulting in a chi-square of 23.17.
Roach et al. (1994) performed a similar study on 282 patients, but divided the patients into low and high risk groups for LN involvement. This resulted in 6% incidence rate in the low risk group and a 40% incidence rate in the high risk group, resulting in a significant split (p <0.001).
Roach et al. (2000) split 895 patients into low, intermediate, and high risk groups for extracapsular extension. This resulted in an incident rate of 17.8%, 46.7%, and 66.7% in low, intermediate, and high risk groups, respectively, which was a significant split (p <0.01).
D'Amico et al. (1998) suggested a widely accepted risk classification for prostate cancer to help predict biochemical outcome after five years after PCa treatment, stratifying them into low, intermediate, and high risk PCa [27]. This study included 1872 patients who underwent radical prostatectomy, EBRT, or interstitial RT. Cox regression was used to calculate the relative risk between different groups of patients, based on risk level and treatment type. No validation was performed in this paper.
Memorial Sloan Kettering Cancer Center (MSKCC) also has a publically available set of nomograms that is based on data from more than 10,000 patients. They have nomograms available to predict outcome after radical prostatectomy both before and after treatment as well as after PSA elevation (see https://www.mskcc.org/nomograms).
3.1.2. mDSS Supporting Treatment Decisions
The studies that contained treatment mDSS are listed in Table 2. The studies described by Walsh et al. (2018), Smith et al. (2016), and van Wijk et al. (2018) compare different RT treatment plans performed on the same patients and the mDSS selects the best plan with most favorable outcome. This type of personalized mDSS is very suitable for the comparison of RT modalities, as the treatment plans are a predictor for the delivered dose, with consequent treatment outcome. Walsh et al. (2018) used a combination of existing models for tumor control probability (TCP) and normal tissue complication probability (NTCP) to compare different treatment plans with photon and proton RT for 25 patients. The study included extensive corrections for displacements during treatment, which aided in the prediction of delivered dose. As this was a modeling study, no validation was done. However, the concept could be used as a basis for RT plan selection between different modalities and could aid in the optimization of TCP and NTCP. Smith et al. (2016) utilized an advanced Bayesian network to optimize intensity modulated radiotherapy (IMRT) treatment plans based on outcome in terms of progression free survival and toxicity. The models were validated against independent clinical trials for the metastasis free survival and overall survival and resulted in uncertainties of 2.5% and 2%, respectively. This method could potentially be implemented into any IMRT planning system and has the potential to improve the quality of treatment plans, resulting in optimized outcomes.
Table 2.
Overview of treatment support systems.
| Study | N | Decision | Inputs | Outcomes | TRIPOD |
|---|---|---|---|---|---|
| (Hodges et al. 2012)[43] | Model | SBRT, IMRT | Utility, transitions | QALY, Costs, ICER | 80% |
|
| |||||
| (Reed et al. 2014)[44] | Model | ART | Risk group ProsVue slope |
QALY, Cost, ICER | 80% |
|
| |||||
| (Vanneste et al. 2015)[45] | Model | IMRT+IRS, IMRT | Utility, transitions | QALY, Cost, ICER | 81% |
|
| |||||
| (Smith et al. 2016)[46] | Model | RT plan | DVH, Clinical∗ | TCP, NTCP, QALY | 87% |
|
| |||||
| (van Wijk et al. 2018)[47] | 23 | IRS in RT | DVH, Clinical | TCP, NTCP | 84% |
|
| |||||
| (Salem et al. 2018)[48] | 200 | Follow-up | Symptoms, Blood tests | Follow-up suggestion | 71% |
|
| |||||
| (Walsh et al. 2018)[49] | 25 | IMRT, V-mat, PSPT, IMPT | DVH | TCP, NTCP, Robustness, stability | 84% |
Abbreviations. N: number of patients; IMRT: intensity modulated radiotherapy; V-mat: volumetric-modulated arc therapy; PSPT: passively scattered proton therapy; IMPT: image modulated proton therapy; TCP: tumor control probability; NTCP: normal tissue complication probability; DVH: dose-volume histogram; QALY: quality adjusted life year; IRS: implantable rectum spacer; ART: adjuvant radiotherapy; ICER: incremental cost-effectiveness ratio; TRIPOD: adherence to the TRIPOD statement.
∗Clinical parameters.
A device called the implantable rectum spacer (IRS) has been developed to spare the rectum during IMRT by increasing the distance between the anterior rectum wall and the prostate [39, 40]. Van Wijk et al. (2018) made use of image deformation based on a virtual IRS [41, 42]: she published models to predict the sparing effect of an IRS before implanting the IRS. The model was tested in a proof of concept study with 16 patients, comparing the mDSS outcome for the virtual IRS to the real IRS in the patients, and the median discrepancies in outcome were 1.8%. Once validation has taken place, this DSS could be applied to any RT planning system and has the potential to personalize treatment choice.
Two studies that supported the decisions involving follow-up treatment were found. Reed et al. (2014) analyzed the cost-effectiveness (CE) of the use of a nucleic acid detection immunoassay (NADiA) ProsVue™ to support the decision for adjuvant radiotherapy (ART). This model showed that primarily for the intermediate risk patients, NADiA ProsVue had an incremental cost-effectiveness ratio (ICER) lower than $50,000 in 83.6% of the simulations. This analysis in its current state is not applicable outside the USA. Salem et al. (2018) validated a computer-led decision support system aimed at giving additional advice in follow-up treatment strategy. This system uses the clinical profile of the patient in combination with blood test results to propose a follow-up strategy. Validation was done on 200 patients by comparing the computer generated strategy to the advice given by trained urologists. In all cases, the agreement between the experts and the system was better than moderate (kappa >0.6). The paper argued that such a system could significantly reduce costs in follow-up care in the United Kingdom and believed the system can be used by any healthcare worker, regardless of urology background.
Hodges et al. (2012) developed a CE model to analyze the benefit of IMRT with respect to stereotactic body radiotherapy (SBRT). Though this study compared these two treatments, this study carried out analysis on cohort level and not case-specific, as is expected of personalized medicine. The CE analysis was done solely based on probabilistic simulations, thus not taking into account variations in cost, transition rates, or utility values. A sensitivity analysis was performed, revealing that in 66% of the iterations SBRT was cost-effective over IMRT.
Vanneste et al. (2015) constructed a CE model to compare IMRT + IRS with IMRT without IRS. He developed a decision-analytic Markov model to examine the effect of late rectal toxicity and compare the costs and quality-adjusted life years (QALYs). An ICER of €55,880 per QALY was gained. For a ceiling ratio of €80,000, IMRT+S had a 77% probability of being cost-effective.
3.1.3. mDSS Focusing on Providing Patient Information
This section describes the studies that attempted to improve shared decision-making. This type of mDSS fosters patient involvement in therapeutic decisions and emphasizes the provision of information needed to make such decisions (Table 3). Berry et al. (2013) described the testing of a personal patient-profile (P3P) intervention for PCa patients. P3P is a web-based program to help men prepare for shared decision-making about the management of early stage prostate cancer. The study hypothesized that when P3P was used in combination with a consultation with different clinicians to prepare for decision-making, patients were more satisfied with their treatment choice and experienced less regret, but testing did not result in significant improvement. However, this type of system has the capability to take into account patient preferences and priorities, while when only using consultations, these preferences can be misinterpreted by clinicians. The paper suggested similar studies to be performed in the future on larger cohorts.
Table 3.
Summary of patient support systems.
| Study | N | Decision | Inputs | Outcomes | TRIPOD |
|---|---|---|---|---|---|
| (Nguyen et al. 2009)[50] | Literature | Treatment | Various | Various | 86% |
|
| |||||
| (Berry et al. 2013)[51] | 494 | Treatment | P3P | Decision satisfaction | 82% |
Abbreviations. N: number of patients; P3P: personal patient-profile for prostate cancer.
Nguyen et al. (2009) performed an extensive literature research for predictive outcome models for 15 different treatment options for PCa, including active surveillance, combinations of radical prostatectomy, RT, hormonal therapy, and high intensity focused ultrasound. They attempted to create a comprehensive overview of the different outcome combinations, such as survival, metastasis, and various toxicities. This overview was designed to be comprehensive for patients so that they could use their own priorities and preferences to make an informed decision. Though they concluded that there is a great need for additional models, this paper provided a framework and is a step towards evidence-based personalized medicine. Additionally, this framework could be a useful tool for clinical decision-making by medical personnel when adapted for these users.
3.2. Excluded Studies
Two studies, Stacey et al. (2016) [52] and Jayadevappa et al. (2015) [53], were excluded because they contained study proposals. Both these studies will attempt to test patient decision aids. Another study, McRae et al. (2016) [54], was excluded because it was not about prostate cancer.
Eight studies in total were excluded as they did not describe an mDSS, but instead provided tools that could be used in the development of an mDSS. Though these studies can be used to further personalize PCa treatment, these tools cannot be applied in the clinic in their current form. A short overview of these papers is given in Table 4.
Table 4.
Overview of excluded studies that described tools to improve mDSS.
| Study | N | Tool | Inputs | Outcomes |
|---|---|---|---|---|
| (Daemen et al. 2009)[61] | 55 | Genetic integration | DNA, CNV | Cancer outcome |
|
| ||||
| (Kuru et al. 2013)[59] | 50 | Diagnostics | mpMRI | PIRADS |
|
| ||||
| (Zumsteg et al. 2013)[55] | 1024 | Risk stratification | Risk factors, Gleason score, biopsy | Risk group |
|
| ||||
| (Beyan et al. 2014) [62] | Model | Genetic integration | SNPs | Various |
|
| ||||
| (Kent and Vickers 2015)[60] | Model | Diagnostics | Clinical and tumor features | Life expectancy |
|
| ||||
| (Gnanapragasam et al. 2016)[56] | 10139 | Risk stratification | PSA, stage, Gleason score | Risk group |
|
| ||||
| (Epstein et al. 2016)[57] | 26325 | Risk stratification | PSA, stage | Gleason grade |
|
| ||||
| (Gries et al. 2017)[58] | 120 | Utility values | Utility's 18 health states | Utility's 243 heath states |
Abbreviations. N: Number of patients; CNV: copy number variation; SNP: single nucleotide polymorphism; PSA: prostate specific antigen; mpMRI: multiparametric magnetic resonance imaging; PIRADS: Prostate Imaging Reporting and Data system.
Two studies, Daemen et al. (2009) and Beyan et al. (2014), attempted to use genetic information to improve cancer prediction outcomes. Beyan specifically worked to incorporate single nucleotide polymorphisms (SNPs) into the national health information system of Turkey.
Zumsteg et al. (2013) [55], Gnanapragasam et al. (2016) [56], and Epstein et al. (2016) [57] provided new definitions for risk group stratification, increasing upon the current low, intermediate, and high risk groups. They found clear separation in progression free survival in their new risk groups, making these a potential tool for diagnostic mDSS. Gries et al. (2017) [58] provided a method to interpolate the utility values for all combinations of 18 different health states, which could be a valuable tool for mDSS to quantify QALYs. Kuru et al. (2013) [59]showed that a PIRADS score on mpMRI is prognostic for PCa.
Finally Kent et al. (2015) [60] performed a literature search and attempted to create a diagnostic tool which would predict the life expectancy in PCa patients. They concluded that no existing model was suitable for incorporation into an mDSS.
4. Discussion
4.1. Primary Findings
In response to the increasing number of PCa treatment options, the development of mDSS has become a growing topic of interest to provide an aid in difficult medical decisions. Diagnosis and staging mDSS have also been a growing topic of interest, as well as a number of tools to improve cancer detection, predictions of treatment outcome, and outcome stratification. However, there is still a need for new mDSS in treatment decision aids and validation of diagnostic mDSS. Additionally, the field of patient informed decision-making is still in its infancy, but essential for the growth towards individualized medicine.
For the diagnostic tools, there are a number of viable tools available for the diagnosis of local PCa that have been validated on large cohorts [30, 31, 33, 35, 63]. Additionally, a study has been performed for detection of PCa on the voxel level for MRI images [32]. This type of diagnostic mDSS could additionally assist in treatment planning or treatment selection. One study explores diagnosis of PCa with LN metastasis [64], but this field remains largely uncharted, similar to the computer automated detection of biochemical failure or treatment failure, also explored in only one study [48].
For treatment mDSS, the proportion between the number of treatment options and the decision support tools remains somewhat skewed. Nguyen et al. (2009)[50] suggested 15 different combinations of treatment options, and this did not include the use of rectal displacement devices [65, 66] or proton therapy. The treatment mDSS that were found by the current study primarily involved RT, including treatment plan selection [46], proton compared to photon therapy [49], SBRT compared to IMRT [43], and the use of an implantable rectum spacer in EBRT [47]. Nguyen et al. (2009) attempted to create a general overview of prediction tools to create a clear overview for patients, and they found most prediction tools to be focused on RT and radical prostatectomy. Less prediction tools are available for brachytherapy, which is a very viable treatment option for PCa. One study, Alitto et al. (2017) [67], describes an Umbrella Protocol for the standardized development of new mDSS. This protocol could help in improving the application of new mDSS in clinics.
Development of patient decision aids is challenging, as cultural and language barriers are much more present in this field of research. Nguyen et al. (2009) developed a comprehensive treatment overview for patients, but found that the predictive tools available were limited, leaving an incomplete overview. Berry et al. (2013) [51] hypothesized that patients satisfaction was increased after treatment when they were actively involved in the decision-making process and comorbidities like anxiety, depression, and fatigue were reduced, and they proceeded to test this using the P3P intervention. They have found, however, that this method has not increased self-reported preparation for the intervention.
4.2. mDSS Design
The last decade deep learning algorithms have gained popularity in the development of mDSS for the classification of cancers, and the same is true within the field of PCa. Kim et al. (2011) and Lee et al. (2010) tackled a similar problem with both an ANN and an SVM, but the dataset of Kim et al. (2011) was approximately double in size. This increased patient cohort resulted in a higher AUC performance for both models, which confirms that deep learning algorithms rely heavily on large datasets. Also, no external validation was done, so currently these models are not generally applicable. An approach to make these models both more accurate and more applicable for clinical use would be to use distributed learning, where the models are trained on centers all over the world, without the data having to leave the clinics. Shah et al. (2012) used an SVM for a more complex problem than just classification by doing voxel based analyses for the localization of PCa. The drawback of this model is that for training, a large amount of imaging data must be available, as well as 3D pathology, but the advantage is that the usage of subregions in the prostate allows for data augmentation.
Notable is that a number of nomograms initially developed more than two decades ago are currently still in use, though updated using newer datasets. The continued application of simple, easy to interpret models is something to keep in mind in the development of new mDSS. Though artificial intelligence has the potential to improve diagnosis of PCa, transparency plays a large role in clinical application. It also shows that predictive parameters for PCa are very consistent, with persistent usage of Gleason score, PSA, clinical stage, and age. Any new mDSS being developed should be tested against the performance of these parameters to avoid tackling a simple problem using computationally heavy machine learning.
When looking at the available treatment mDSS, we see that most of these are focused on RT. This is likely due to the patient specific treatment planning done in RT, which results in highly detailed dose maps before the start of treatment. This is ideal for mDSS, as different treatment plans can be directly compared, and the different outcomes can be predicted using dose response curves. Other types of treatment, such as prostatectomy or watchful waiting, rely much more heavily on clinical parameters and tumor parameters for predictions of their outcome or on subjective physician decisions. This makes it harder to compare outcomes of different treatments for the same patient. The development of new mDSS comparing completely different treatment types with each other for the same patient would be very beneficial for filling this gap in the current literature.
4.3. Study Limitations
This study focused on mDSS in PCa, and the scope did not include any other cancer types. It is possible that general mDSS, applicable for more than one cancer type, were therefore overlooked. The search was also focused on mDSS and thus did not include any predictive models that could aid in decision-making. The terms describing mDSS may not have been used in interesting studies that could have been included in this overview. ‘Patient decision aids' was also not a search term, which may be the cause of the limited number of mDSS for patients included in this overview. The search performed was only MEDLINE/PubMed linked, so studies not available on these media were overlooked.
Inherent to literature overviews is that negative findings are not always reported, so failed attempts at creating mDSS are often uncommon in overviews and literature reviews, and this may cause a biased view on the subject.
We assessed the reporting quality of each included study using the TRIPOD statement for quantification and comparison. Although the TRIPOD statement has been endorsed by a large number of medical journals and editorial organizations, it is not a universal gold standard. The checklist used for the assessment has limitations, such as the severe point punishment for the lack of specific keywords in title or abstract. This could reduce the score of a well-written paper. Additionally, the adherence to the TRIPOD statement was compressed into a single number, and the specific reporting issues were not named, such as improper analyses, lack of validation, or reporting of study specifics. However, it is a useful tool to show the strength of a report, as when adherence to the TRIPOD statement was high, the paper was clearly written, and all proper steps were taken for the development of quality mDSS tools.
5. Conclusion
A number of mDSS for the primary diagnosis and staging of localized PCa are available. Treatment mDSS were mostly focused on EBRT, for which several tools are available. However, a lack of mDSS for other treatment modalities suggests that the development of new tools is necessary to objectively compare different treatment modalities. The development of patient decision aids is a new field of research, and few successes have been made for PCa patients. Though the idea of informed decision-making by patients is in line with the goal of personalized medicine, the development of these tools needs to overcome a number of barriers to be successful, like comprehensiveness, language barriers, patient cooperation, and physician cooperation. More research needs to be performed to better empower clinical decisions in the diagnosis and treatment process.
Conflicts of Interest
Dr. Philippe Lambin reports grants/sponsored research agreements from Varian Medical, Oncoradiomics, ptTheragnostic, and, outside the submitted work, Health Innovation Ventures and DualTpharma. He received an advisor (SAB)/presenter fee and/or reimbursement of travel costs/external grant writing fee and/or in-kind manpower contribution from Oncoradiomics and, outside the submitted work, BHV and Convert Pharmaceuticals. Dr. Lambin has shares in the company Oncoradiomics SA and, outside the submitted work, Convert Pharmaceuticals SA and is coinventor of two patents on radiomics (PCT/NL2014/050248, PCT/NL2014/050728) licensed to Oncoradiomics; one patent on mtDNA (PCT/EP2014/059089) licensed to ptTheragnostic/DNAmito; three non-patentable inventions (software) licensed to ptTheragnostic/DNAmito, Oncoradiomics, and Health Innovation Ventures. Dr. Seán Walsh is a paid employee at Oncoradiomics. Yvonka van Wijk is an employee at ptTheragnostic. Iva Halilaj is a paid employee at Health Innovations.
Supplementary Materials
The supplementary material we provided contains a checklist of TRIPOD points and the calculation method we used to calculate the adherence to the TRIPOD statement. The version put in the supplementary materials is the one available on the TRIPOD site. It first contains a description, then part A with general description of the study, and then part B where each point is checked.
References
- 1.Bray F., Ferlay J., Soerjomataram I., Siegel R. L., Torre L. A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Hamdy F. C., Donovan J. L., Lane J. A., et al. 10-Year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. The New England Journal of Medicine. 2016;375(15):1415–1424. doi: 10.1056/nejmoa1606220. [DOI] [PubMed] [Google Scholar]
- 3.Lennernäs B., Majumder K., Damber J.-E., et al. Radical prostatectomy versus high-dose irradiation in localized/locally advanced prostate cancer: a swedish multicenter randomized trial with patient-reported outcomes. Acta Oncologica. 2015;54(6):875–881. doi: 10.3109/0284186x.2014.974827. [DOI] [PubMed] [Google Scholar]
- 4.Donovan J. L., Hamdy F. C., Lane J. A., et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. The New England Journal of Medicine. 2016;375(15):1425–1437. doi: 10.1056/NEJMoa1606221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stone N. N., Potters L., Davis B. J., et al. Multicenter analysis of effect of high biologic effective dose on biochemical failure and survival outcomes in patients with gleason score 7-10 prostate cancer treated with permanent prostate brachytherapy. International Journal of Radiation Oncology • Biology • Physics. 2009;73(2):341–346. doi: 10.1016/j.ijrobp.2008.04.038. [DOI] [PubMed] [Google Scholar]
- 6.Sylvester J. E., Grimm P. D., Blasko J. C., et al. 15-Year biochemical relapse free survival in clinical Stage T1-T3 prostate cancer following combined external beam radiotherapy and brachytherapy; Seattle experience. International Journal of Radiation Oncology • Biology • Physics. 2007;67(1):57–64. doi: 10.1016/j.ijrobp.2006.07.1382. [DOI] [PubMed] [Google Scholar]
- 7.Ragde H., Grado G. L., Nadir B. S. Brachytherapy for clinically localized prostate cancer: Thirteen-year disease-free survival of 769 consecutive prostate cancer patients treated with permanent implants alone. Archivos Españoles de Urología. 2001;54(7):739–747. [PubMed] [Google Scholar]
- 8.Grimm P., Billiet I. D., Bostwick., et al. Comparative analysis of prostate-specific antigen free survival outcomes for patients with low, intermediate and high risk prostate cancer treatment by radical therapy. Results from the Prostate Cancer Results Study Group. BJU Int. 2012;109(supplement 1):22–29. doi: 10.1111/j.1464-410X.2011.10827.x. [DOI] [PubMed] [Google Scholar]
- 9.Musunuru H. B., Yamamoto T., Klotz L., et al. Active surveillance for intermediate risk prostate cancer: survival outcomes in the sunnybrook experience. The Journal of Urology. 2016;196(6):1651–1658. doi: 10.1016/j.juro.2016.06.102. [DOI] [PubMed] [Google Scholar]
- 10.Agarwal G., Buethe D., Russell C., Luchey A., Pow-Sang J. M. Long term survival and predictors of disease reclassification in patients on an active surveillance protocol for prostate cancer. Canadian Journal of Urology. 2016;23(2):8215–8219. [PubMed] [Google Scholar]
- 11.Soloway M., Roach M., III Prostate cancer progression after therapy of primary curative intent: A review of data from the prostate-specific antigen era. Cancer. 2005;104(11):2310–2322. doi: 10.1002/cncr.21441. [DOI] [PubMed] [Google Scholar]
- 12.Roach M., III Radical prostatectomy or external beam radiotherapy: One step forward or two steps back? Cancer. 2002;95(2):215–218. doi: 10.1002/cncr.10659. [DOI] [PubMed] [Google Scholar]
- 13.Vanneste B. G. L., van Limbergen E. J., van Lin E. N., et al. Prostate cancer radiation therapy: what do clinicians have to know? BioMed Research International. 2016;2016:14. doi: 10.1155/2016/6829875.6829875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Valentini V., Dinapoli N., Damiani A. The future of predictive models in radiation oncology: from extensive data mining to reliable modeling of the results. Future Oncology. 2013;9(3):311–313. doi: 10.2217/fon.12.197. [DOI] [PubMed] [Google Scholar]
- 15.Dekker A., Vinod S. L., Holloway., et al. Rapid learning in practice: a lung cancer survival decision support system in routine patient care data. Radiotherapy and Oncology. 2014;113(1):47–53. doi: 10.1016/j.radonc.2014.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Abernethy A. P., Etheredge L. M., Ganz P. A., et al. Rapid-learning system for cancer care. Journal of Clinical Oncology. 2010;28(27):4268–4274. doi: 10.1200/JCO.2010.28.5478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lambin P., Van Stiphout R. G. P. M., Starmans M. H. W., et al. Predicting outcomes in radiation oncology-multifactorial decision support systems. Nature Reviews Clinical Oncology. 2013;10(1):27–40. doi: 10.1038/nrclinonc.2012.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lambin P., Zindler J., Vanneste B., et al. Modern clinical research: how rapid learning health care and cohort multiple randomised clinical trials complement traditional evidence based medicine. Acta Oncologica. 2015;54(9):1289–1300. doi: 10.3109/0284186x.2015.1062136. [DOI] [PubMed] [Google Scholar]
- 19.Lambin P., Roelofs E., Reymen B., et al. 'Rapid Learning health care in oncology' - an approach towards decision support systems enabling customised radiotherapy. Radiotherapy & Oncology. 2013;109(1):159–164. doi: 10.1016/j.radonc.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 20.Lambin P., Petit S. F., Aerts H. J. W. L., et al. The ESTRO Breur lecture 2009. from population to voxel-based radiotherapy: exploiting intra-tumour and intra-organ heterogeneity for advanced treatment of non-small cell lung cancer. Radiotherapy & Oncology. 2010;96(2):145–152. doi: 10.1016/j.radonc.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 21.Lambin P., Zindler J. B. G., Vanneste., et al. Decision support systems for personalized and participative radiation oncology. Advanced Drug Delivery Reviews. 2017;109:131–153. doi: 10.1016/j.addr.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 22.Pawloski P. A., Brooks G. A., Nielsen M. E., Olson-Bullis B. A. A systematic review of clinical decision support systems for clinical oncology practice. Journal of the National Comprehensive Cancer Network. 2019;17(4):331–338. doi: 10.6004/jnccn.2018.7104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McNutt T. R., Benedict S. H., Low D. A., et al. Using big data analytics to advance precision radiation oncology. International Journal of Radiation Oncology • Biology • Physics. 2018;101(2):285–291. doi: 10.1016/j.ijrobp.2018.02.028. [DOI] [PubMed] [Google Scholar]
- 24.Moons K. G. M., Altman D. G., Reitsma J. B., Collins G. S. New guideline for the reporting of studies developing, validating, or updating a multivariable clinical prediction model: The TRIPOD statement. Advances in Anatomic Pathology. 2015;22(5):303–305. doi: 10.1097/PAP.0000000000000072. [DOI] [PubMed] [Google Scholar]
- 25.Roach M., III, Marquez C., Yuo H.-S., et al. Predicting the risk of lymph node involvement using the pre-treatment prostate specific antigen and gleason score in men with clinically localized prostate cancer. International Journal of Radiation Oncology • Biology • Physics. 1994;28(1):33–37. doi: 10.1016/0360-3016(94)90138-4. [DOI] [PubMed] [Google Scholar]
- 26.Diaz A., Roach M., III, Marquez C., et al. Indications for and the significance of seminal vesicle irradiation during 3D conformal radiotherapy for localized prostate cancer. International Journal of Radiation Oncology • Biology • Physics. 1994;30(2):323–329. doi: 10.1016/0360-3016(94)90011-6. [DOI] [PubMed] [Google Scholar]
- 27.D'Amico A. V., Whittington R., Bruce Malkowicz S., et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. Journal of the American Medical Association. 1998;280(11):969–974. doi: 10.1001/jama.280.11.969. [DOI] [PubMed] [Google Scholar]
- 28.Chang P. L., Li Y.-C., Wang T. M., Huang S. T., Hsieh M. L., Tsui K. H. Evaluation of a decision-support system for preoperative staging of prostate cancer. Medical Decision Making. 1999;19(4):419–427. doi: 10.1177/0272989X9901900410. [DOI] [PubMed] [Google Scholar]
- 29.Roach 3rd M., Chen A., Song J., et al. Pretreatment prostate-specific antigen and Gleason score predict the risk of extracapsular extension and the risk of failure following radiotherapy in patients with clinically localized prostate cancer. Seminars in Urologic Oncology. 2000;18(2):108–114. [PubMed] [Google Scholar]
- 30.Lee H. J., Hwang S. I., Han S.-M., et al. Image-based clinical decision support for transrectal ultrasound in the diagnosis of prostate cancer: Comparison of multiple logistic regression, artificial neural network, and support vector machine. European Radiology. 2010;20(6):1476–1484. doi: 10.1007/s00330-009-1686-x. [DOI] [PubMed] [Google Scholar]
- 31.Kim S. Y., Moon S. K., Jung D. C., et al. Pre-operative prediction of advanced prostatic cancer using clinical decision support systems: accuracy comparison between support vector machine and artificial neural network. Korean Journal of Radiology. 2011;12(5):588–594. doi: 10.3348/kjr.2011.12.5.588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shah V., Turkbey B., Mani H., et al. Decision support system for localizing prostate cancer based on multiparametric magnetic resonance imaging. Medical Physics. 2012;39(7):4093–4103. doi: 10.1118/1.4722753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mukai T. O., Bro F., Olesen F., Vedsted P. To test or not: A registry-based observational study of an online decision support for prostate-specific antigen tests. International Journal of Medical Informatics. 2013;82(10):973–979. doi: 10.1016/j.ijmedinf.2013.06.010. [DOI] [PubMed] [Google Scholar]
- 34.Sadoughi F., Ghaderzadeh M. A hybrid particle swarm and neural network approach for detection of prostate cancer from benign hyperplasia of prostate. Studies in Health Technology and Informatics. 2014;205:481–485. doi: 10.3233/978-1-61499-432-9-481. [DOI] [PubMed] [Google Scholar]
- 35.van Leeuwen P. J., Hayen A., Thompson J. E., et al. A multiparametric magnetic resonance imaging-based risk model to determine the risk of significant prostate cancer prior to biopsy. BJU International. 2017;120(6):774–781. doi: 10.1111/bju.13814. [DOI] [PubMed] [Google Scholar]
- 36.Tosoian J. J., Chappidi M., Feng Z., et al. Prediction of pathological stage based on clinical stage, serum prostate-specific antigen, and biopsy Gleason score: Partin Tables in the contemporary era. BJU International. 2017;119(5):676–683. doi: 10.1111/bju.13573. [DOI] [PubMed] [Google Scholar]
- 37.Partin A. W., Yoo J., Carter H. B., et al. The use of prostate specific antigen, clinical stage and Gleason score to predict pathological stage in men with localized prostate cancer. The Journal of Urology. 1993;150(1):110–114. doi: 10.1016/S0022-5347(17)35410-1. [DOI] [PubMed] [Google Scholar]
- 38.Partin A. W., Mangold L. A., Lamm D. M., Walsh P. C., Epstein J. I., Pearson J. D. Contemporary update of prostate cancer staging nomograms (Partin Tables) for the new millennium. Urology. 2001;58(6):843–848. doi: 10.1016/S0090-4295(01)01441-8. [DOI] [PubMed] [Google Scholar]
- 39.Vanneste B. G. L., van De Beek K., Lutgens L., Lambin P. Implantation of a biodegradable rectum balloon implant: tips, tricks and pitfalls. International Brazilian Journal of Urology. 2017;43(6):1033–1042. doi: 10.1590/s1677-5538.ibju.2016.0494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vanneste B. G., Buettner F., Pinkawa M., Lambin P., Hoffmann A. L. Ano-rectal wall dose-surface maps localize the dosimetric benefit of hydrogel rectum spacers in prostate cancer radiotherapy. Clinical and Translational Radiation Oncology. 2019;14:17–24. doi: 10.1016/j.ctro.2018.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van Wijk Y., Vanneste B. G. L., Walsh S., et al. Development of a virtual spacer to support the decision for the placement of an implantable rectum spacer for prostate cancer radiotherapy: Comparison of dose, toxicity and cost-effectiveness. Radiotherapy & Oncology. 2017;125(1):107–112. doi: 10.1016/j.radonc.2017.07.026. [DOI] [PubMed] [Google Scholar]
- 42.Vanneste B. G. L., van Wijk Y., Lutgens L. C., et al. Dynamics of rectal balloon implant shrinkage in prostate VMAT: Influence on anorectal dose and late rectal complication risk. Strahlentherapie und Onkologie. 2018;194(1):31–40. doi: 10.1007/s00066-017-1222-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hodges J. C., Lotan Y., Boike T. P., Benton R., Barrier A., Timmerman R. D. Cost-effectiveness analysis of SBRT versus IMRT: an emerging initial radiation treatment option for organ-confined prostate cancer. American Journal of Managed Care. 2012;18(5):e186–e193. [PubMed] [Google Scholar]
- 44.Reed S. D., Stewart S. B., Scales C. D., Jr., Moul J. W. A framework to evaluate the cost-effectiveness of the NADiA prosvue slope to guide adjuvant radiotherapy among men with high-risk characteristics following prostatectomy for prostate cancer. Value in Health. 2014;17(5):545–554. doi: 10.1016/j.jval.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 45.Vanneste B. G. L., Pijls-Johannesma M., Van De Voorde L., et al. Spacers in radiotherapy treatment of prostate cancer: is reduction of toxicity cost-effective? Radiotherapy & Oncology. 2015;114(2):276–281. doi: 10.1016/j.radonc.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 46.Smith W. P., Kim M., Holdsworth C., Liao J., Phillips M. H. Personalized treatment planning with a model of radiation therapy outcomes for use in multiobjective optimization of IMRT plans for prostate cancer. Journal of Radiation Oncology. 2016;11(1, article no. 38) doi: 10.1186/s13014-016-0609-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.van Wijk Y., Vanneste B. G., Jochems A., et al. Development of an isotoxic decision support system integrating genetic markers of toxicity for the implantation of a rectum spacer. Acta Oncologica. 2018;57(11):1499–1505. doi: 10.1080/0284186X.2018.1484156. [DOI] [PubMed] [Google Scholar]
- 48.Salem H. A., Caddeo G., McFarlane J., et al. A multicentre integration of a computer-led follow-up of prostate cancer is valid and safe. BJU International. 2018;122(3):418–426. doi: 10.1111/bju.14157. [DOI] [PubMed] [Google Scholar]
- 49.Walsh S., Roelofs E., Kuess P., et al. Towards a clinical decision support system for external beam radiation oncology prostate cancer patients: Proton vs. photon radiotherapy? a radiobiological study of robustness and stability. Cancers. 2018;10(2) doi: 10.3390/cancers10020055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nguyen C. T., Kattan M. W. Development of a prostate cancer metagram: A solution to the dilemma of which prediction tool to use in patient counseling. Cancer. 2009;115(13):3039–3045. doi: 10.1002/cncr.24355. [DOI] [PubMed] [Google Scholar]
- 51.Berry D. L., Wang Q., Halpenny B., Hong F. Decision preparation, satisfaction and regret in a multi-center sample of men with newly diagnosed localized prostate cancer. Patient Education and Counseling. 2012;88(2):262–267. doi: 10.1016/j.pec.2012.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Stacey D., Taljaard M., Smylie J., et al. Implementation of a patient decision aid for men with localized prostate cancer: evaluation of patient outcomes and practice variation. Implementation Science. 2015;11(1):p. 87. doi: 10.1186/s13012-016-0451-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jayadevappa R., Chhatre S., Gallo J. J., et al. Treatment preference and patient centered prostate cancer care: design and rationale. Contemporary Clinical Trials. 2015;45:296–301. doi: 10.1016/j.cct.2015.09.024. [DOI] [PubMed] [Google Scholar]
- 54.McRae M. P., Simmons G., Wong J., McDevitt J. T. Programmable bio-nanochip platform: a point-of-care biosensor system with the capacity to learn. Accounts of Chemical Research. 2016;49(7):1359–1368. doi: 10.1021/acs.accounts.6b00112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zumsteg Z. S., Spratt D. E., Pei I., et al. A new risk classification system for therapeutic decision making with intermediate-risk prostate cancer patients undergoing dose-escalated external-beam radiation therapy. European Urology. 2013;64(6):895–902. doi: 10.1016/j.eururo.2013.03.033. [DOI] [PubMed] [Google Scholar]
- 56.Gnanapragasam V. J., Lophatananon A., Wright K. A., et al. Improving clinical risk stratification at diagnosis in primary prostate cancer: a prognostic modelling study. PLoS Medicine. 2016;13(8):p. e1002063. doi: 10.1371/journal.pmed.1002063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Epstein J. I., Zelefsky M. J., Sjoberg D. D., et al. A contemporary prostate cancer grading system: a validated alternative to the gleason score. European Urology. 2016;69:428–435. doi: 10.1016/j.eururo.2015.06.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gries K. S., Regier D. A., Ramsey S. D., Patrick D. L. Utility estimates of disease-specific health states in prostate cancer from three different perspectives. Applied Health Economics and Health Policy. 2017;15(3):375–384. doi: 10.1007/s40258-016-0282-x. [DOI] [PubMed] [Google Scholar]
- 59.Kuru T. H., Roethke M. C., Rieker P., et al. Histology core-specific evaluation of the European Society of Urogenital Radiology (ESUR) standardised scoring system of multiparametric magnetic resonance imaging (mpMRI) of the prostate. BJU International. 2013;112(8):1080–1087. doi: 10.1111/bju.12259. [DOI] [PubMed] [Google Scholar]
- 60.Kent M., Vickers A. J. A systematic literature review of life expectancy prediction tools for patients with localized prostate cancer. The Journal of Urology. 2015;193(6):1938–1942. doi: 10.1016/j.juro.2014.11.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Daemen A., Gevaert O., Ojeda F., et al. A kernel-based integration of genome-wide data for clinical decision support. Genome Medicine. 2009;1(4):p. 39. doi: 10.1186/gm39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Beyan T., Aydın Son Y. Incorporation of personal single nucleotide polymorphism (SNP) data into a national level electronic health record for disease risk assessment, part 1: an overview of requirements. JMIR Medical Informatics. 2014;2(2):p. e15. doi: 10.2196/medinform.3169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ghaderzadeh M. Clinical decision support system for early detection of prostate cancer from benign hyperplasia of prostate. Studies in Health Technology and Informatics. 2013;192:p. 928. [PubMed] [Google Scholar]
- 64.Chang P. L., Wang T. M., Huang S. T., Hsieh M. L., Tsui K. H., Lai R. H. Use of a medical decision support system to improve the preoperative diagnosis of prostate cancer with pelvic lymph node metastases. Changgeng Yi Xue Za Zhi. 1999;22(4):556–564. [PubMed] [Google Scholar]
- 65.Wilton L., Richardson M., Keats S., et al. Rectal protection in prostate stereotactic radiotherapy: a retrospective exploratory analysis of two rectal displacement devices. Journal of Medical Radiation Sciences. 2017;64(4):266–273. doi: 10.1002/jmrs.238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mok G., Benz E., Vallee J.-P., Miralbell R., Zilli T. Optimization of radiation therapy techniques for prostate cancer with prostate-rectum spacers: a systematic review. International Journal of Radiation Oncology • Biology • Physics. 2014;90(2):278–288. doi: 10.1016/j.ijrobp.2014.06.044. [DOI] [PubMed] [Google Scholar]
- 67.Alitto A., Gatta R., Vanneste B., et al. PRODIGE: PRediction models in prOstate cancer for personalized meDIcine challenGE. Future Oncology. 2017;13(24):2171–2181. doi: 10.2217/fon-2017-0142. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The supplementary material we provided contains a checklist of TRIPOD points and the calculation method we used to calculate the adherence to the TRIPOD statement. The version put in the supplementary materials is the one available on the TRIPOD site. It first contains a description, then part A with general description of the study, and then part B where each point is checked.