Abstract
Purpose:
To compare corneal higher-order aberrations (HOA) following ultrathin Descemet stripping automated endothelial keratoplasty (UT-DSAEK) and Descemet membrane endothelial keratoplasty (DMEK).
Design:
Patient and outcome-masked, randomized controlled clinical trial.
Participants:
Patients with damaged or diseased endothelium from Fuchs endothelial dystrophy or pseudophakic bullous keratopathy who were considered good candidates for DMEK or UT-DSAEK.
Methods:
Corneal anterior and posterior surface HOA were measured with Scheimpflug imaging before surgery and at 3, 6, and 12 months post-operatively. Zernike orders 3-8 were calculated at 4.0- and 6.0-mm-diameter optical zones (OZ). HOA after UT-DSAEK and DMEK were compared and correlation was performed between best spectacle-corrected visual acuity (BSCVA) and HOA at each time point.
Main Outcome Measures:
Higher-order aberrations of the anterior and posterior cornea, expressed as the root mean square deviation from a best fit sphere reference surface.
Results:
At 3, 6, and 12 months after surgery, the posterior corneal surface had significantly less coma (P≤0.003) and total HOA (P≤0.001) in DMEK compared to UT-DSAEK (4.0- and 6.0-mm OZ). Posterior trefoil (P≤0.034), secondary astigmatism (P≤0.042), and tetrafoil (P≤0.045) were lower in DMEK than UT-DSAEK at 3, 6, or 12 months (either 4.0- or 6.0-mm OZ). There were no significant differences in anterior surface HOA between DMEK and UT-DSAEK at any time point. Compared to baseline, total posterior HOA was significantly increased (P≤0.036) in UT-DSAEK at 3, 6, and 12 months, in contrast to DMEK where it was significantly decreased (P≤0.044) at 6 and 12 months (4.0- and/or 6.0-mm OZ). At 6 and 12 months, posterior corneal total HOA correlated moderately with BSCVA (ρ≤0.635, P<0.001) (4.0- and 6.0-mm OZ). There were no moderate or strong correlations between anterior or combined corneal surface HOA at any time point after surgery.
Conclusions:
DMEK results in less posterior corneal HOA compared to UT-DSAEK. DMEK decreases and UT-DSAEK increases posterior corneal HOA compared to pre-surgical values. Total posterior corneal HOA correlates moderately with 6- and 12-month post-operative visual acuity and may partially account for the better visual acuity observed after DMEK.
Précis:
Posterior corneal higher-order aberrations are reduced following DMEK compared to ultrathin DSAEK (UT-DSAEK) and correlate moderately with best corrected visual acuity at 6 and 12 months after surgery.
There have been several proposed mechanisms for the differences in visual outcomes between Descemet stripping automated endothelial keratoplasty (DSAEK) and Descemet membrane endothelial keratoplasty (DMEK) including graft interface haze, higher-order aberrations (HOA), and light scatter.1–8 However, until recently there have been no prospective randomized trials directly comparing the outcomes of these two techniques.
The DETECT trial was a two-surgeon patient and outcome-masked randomized controlled trial comparing outcomes of ultrathin DSAEK (UT-DSAEK) and DMEK which found that DMEK had better visual acuity at 3, 6, and 12 months.9 We hypothesized that central corneal HOA may be a significant contributor to the differences in visual outcomes between these two techniques. Higher levels of HOA have been found to significantly reduce visual acuity after endothelial keratoplasty due to degradation of the retinal image point-spread function and may have a larger impact than induced light scatter.1–5,10–12 However, there is some controversy on which corneal surface aberrations most affect BSCVA as several studies have demonstrated correlations with anterior HOA while others have found that posterior corneal HOA may affect BSCVA to the largest extent.2–5,8,10 This study assesses pre-and post-endothelial keratoplasty HOA of the anterior, posterior, and combined corneal surfaces in the setting of a randomized controlled clinical trial and correlates these aberrations with ETDRS visual acuity.
Methods
The Descemet Endothelial Thickness Comparison Trial (DETECT) was a randomized, patient/evaluator masked, controlled trial comparing clinical outcomes of two types of endothelial keratoplasty. Study participants with isolated endothelial dysfunction at two academic centers were block randomized in a 1:1 fashion to either UT-DSAEK or DMEK. The primary outcome of the trial was BSCVA at 6 months postoperatively. A detailed description of the randomization, blinding, and surgical methods have been previously published.9
Higher-order aberrations (HOA) were measured as a pre-specified secondary outcome of the trial. Three-dimensional scans of the anterior segment were generated with a rotating Scheimpflug camera (Pentacam; Oculus, Wetzlar, Germany) performed under standardized low lighting conditions before surgery and at 3, 6, and 12 months post-operatively. Patients were instructed to blink twice and then look at the fixation device in the Pentacam. Image acquisition was a 1-second scan of 25 rotational Scheimpflug images through the corneal sighting point, the point where the ray of light from the fovea to the fixation device crossed the cornea. Each scan was separated by 7.2 degrees. Acceptable maps had at least 10.0 mm of corneal coverage with no extrapolated data in the central 9.0-mm zone. Scans not meeting acceptable criteria were repeated, but most patients required only 1 image. All Zernike higher orders 3 to 8 of the anterior, posterior, and combined corneal surfaces were calculated at 4.0- and 6.0-mm-diameter optical zones (OZ) using the Pentacam software (version 1.21r3; Oculus) based on measured relative height data between the cornea and a best-fit sphere reference surface. These can be determined independently of pupil size as the measurement is a corneal wavefront measurement and not a total eye wavefront measurement. Root mean square (RMS) deviations from individual aberrations (coma aberrations (Z3±1); trefoil aberrations (Z3±3); spherical aberrations (Z40); secondary astigmatism (Z4±2); tetrafoil aberrations (Z4±4)) were calculated from the Zernike vector values calculated by the software. RMS of higher-order sums for Zernike orders 3 to 8 (total HOA (Z3-8)) were determined in a similar manner as previously described.13
Statistical analysis
Individual and total HOA RMS data of UT-DSAEK and DMEK were found to be non-parametric and were compared with a Mann-Whitney U test at each time point. Spearman correlation was performed between BSCVA and HOA of the entire dataset and within each arm of the study. To account for potential type I errors due to multiple comparisons, a Holm-Bonferroni method was applied to P-value sets. It did not lead to a change in statistical outcome. Missing data were analyzed using the last observation carried forward. All analyses were performed using SPSS Statistics (version 24; IBM).
Ethical Approval
Ethical approval was obtained from the Institutional Review Board at University of California, San Francisco, Oregon Health & Science University, and Stanford University. Written informed consent was obtained from all participants, and the trial adhered to the tenants of the Declaration of Helsinki and HIPAA regulations. The DETECT trial is registered at www.clinicaltrials.gov (Trial # NCT0237137).
Results
216 patients were screened and 50 eyes of 38 patients were randomized to DMEK (25 eyes) or UT-DSAEK (25 eyes). Baseline patient and donor clinical characteristics are outlined in a previous report and are included as Table 1 here (reprinted with permission). There were no previous significant differences in any patient or donor characteristics assessed including refractive error (spherical equivalent).9 Pentacam data were not acquired for 1 patient in the DMEK arm at 6 months.
Table 1.
Baseline Characteristics
| UT-DSAEK (N=25) | DMEK (N=25) | p* | |
|---|---|---|---|
|
Patient characteristics | |||
| Age (years), mean (range) | 68 (51 – 95) | 68 (61 – 81) | 0.96 |
| Female sex, N (%) | 16 (64%) | 13 (52%) | 0.39 |
| Diagnosis, N (%) | |||
| Fuchs | 24 (96%) | 24 (96%) | 1.00 |
| PBK | 1 (4%) | 1 (4%) | |
| ETDRS BSCVA, mean ± SD | |||
| logMAR | 0.27 ± 0.21 | 0.34 ± 0.29 | 0.34 |
| Approximate Snellen | 20/40 | 20/40 | |
| CCT (μm), mean ± SD | 610 ±44** | 608 ± 52 | 0.93 |
| Spherical equivalent (diopters), mean ± SD | −0.4 ± 1.9 | −0.8 ± 2.2 | 0.53 |
| Surgery, N (%) | |||
| Pseudophakic EK | 8 (32%) | 7 (28%) | 0.76 |
| Triple EK | 17 (68%) | 18 (72%) | |
|
Donor characteristics | |||
| Age (years), mean (range) | 64 (54 – 75) | 62 (50 – 76) | 0.38 |
| Female sex, N (%) | 8 (32%) | 5 (20%) | 0.33 |
| Death to preservation (hours), mean ± SD | 9 ± 4 | 9 ± 3 | 0.80 |
| Death to surgery (days), mean ± SD | 6 ± 1 | 6 ± 1 | 0.61 |
| ECC pre-processing (cells/mm2), mean ± SD | 2792±210 | 2797 ± 222 | 0.94 |
| ECC post-processing (cells/mm2), mean ± SD | 2796 ± 238 | 2771 ± 150 | 0.66 |
| Central graft thickness, mean ± SD (range) | 73 ± 12 (37 – 88) | -- | -- |
Abbreviations: UT-DSAEK = ultrathin Descemet stripping automated endothelial keratoplasty, DMEK = Descemet membrane endothelial keratoplasty, PBK = Pseudophakic Bullous Keratopathy, ETDRS = Early Treatment Diabetic Retinopathy Study, BSCVA = best spectacle-corrected visual acuity, SD = standard deviation, logMAR = logarithm of the minimum angle of resolution, CCT = central corneal thickness, EK = endothelial keratoplasty, ECC = endothelial cell count.
t-tests for continuous variables and chi2 tests for categorical variables
one missing value
At baseline, there was a significant difference between groups in secondary astigmatism: anterior 4.0-mm OZ (UT-DSAEK group>DMEK group) and posterior 6.0-mm OZ (DMEK group>UT-DSAEK group) (Tables 2, 3). These differences did not persist after surgery. All other individual and total HOA on anterior, posterior, or combined surfaces had no significant differences at baseline (Tables 2, 3; Fig 1)).
Table 2.
Mean HOA magnitude (RMS in μm) at 4.0-mm optical zone by surgery type at baseline, 3, 6, and 12 months after surgery.
| Baseline |
3 Months |
6 Months |
12 Months |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | |
| RMS coma CF | 0.166 | 0.153 | 0.379 | 0.182 | 0.200 | 0.159 | 0.155 | 0.172 | 0.522 | 0.157 | 0.161 | 0.648 |
| RMS trefoil CF | 0.075 | 0.100 | 0.809 | 0.099 | 0.096 | 0.159 | 0.088 | 0.090 | 0.560 | 0.082 | 0.100 | 0.269 |
| RMS SA CF | 0.078 | 0.059 | 0.455 | 0.068 | 0.085 | 0.204 | 0.072 | 0.067 | 0.362 | 0.076 | 0.077 | 0.946 |
| RMS 2° Astigmatism CF | 0.057 | 0.039 | 0.043 | 0.062 | 0.056 | 0.816 | 0.046 | 0.052 | 0.415 | 0.044 | 0.039 | 0.377 |
| RMS tetrafoil CF | 0.057 | 0.084 | 0.385 | 0.086 | 0.080 | 0.985 | 0.061 | 0.061 | 0.248 | 0.058 | 0.063 | 0.749 |
| RMS total HOA CF | 0.235 | 0.245 | 0.775 | 0.274 | 0.288 | 0.068 | 0.230 | 0.243 | 0.547 | 0.228 | 0.241 | 0.485 |
| RMS coma CB | 0.080 | 0.091 | 0.895 | 0.109 | 0.052 | 0.001 | 0.092 | 0.046 | 0.001 | 0.079 | 0.039 | 0.003 |
| RMS trefoil CB | 0.060 | 0.081 | 0.199 | 0.132 | 0.071 | 0.005 | 0.117 | 0.054 | <0.001 | 0.102 | 0.064 | 0.012 |
| RMS SA CB | 0.040 | 0.050 | 0.173 | 0.047 | 0.044 | 0.491 | 0.045 | 0.039 | 0.420 | 0.041 | 0.036 | 0.304 |
| RMS 2° Astigmatism CB | 0.023 | 0.023 | 0.078 | 0.041 | 0.021 | 0.006 | 0.029 | 0.019 | 0.038 | 0.028 | 0.021 | 0.076 |
| RMS tetrafoil CB | 0.027 | 0.034 | 0.860 | 0.075 | 0.025 | <0.001 | 0.034 | 0.025 | 0.162 | 0.034 | 0.026 | 0.143 |
| RMS total HOA CB | 0.124 | 0.150 | 0.489 | 0.217 | 0.116 | <0.001 | 0.179 | 0.095 | <0.001 | 0.159 | 0.098 | <0.001 |
| RMS coma cornea | 0.184 | 0.157 | 0.404 | 0.244 | 0.233 | 0.954 | 0.198 | 0.190 | 0.938 | 0.209 | 0.175 | 0.516 |
| RMS trefoil cornea | 0.098 | 0.141 | 0.792 | 0.184 | 0.141 | 0.839 | 0.155 | 0.116 | 0.252 | 0.137 | 0.138 | 0.954 |
| RMS SA cornea | 0.065 | 0.053 | 0.717 | 0.050 | 0.076 | 0.049 | 0.066 | 0.056 | 0.362 | 0.067 | 0.067 | 0.854 |
| RMS 2° Astigmatism cornea | 0.066 | 0.041 | 0.097 | 0.092 | 0.072 | 0.491 | 0.062 | 0.064 | 0.946 | 0.058 | 0.052 | 0.560 |
| RMS tetrafoil cornea | 0.072 | 0.095 | 0.116 | 0.112 | 0.097 | 0.467 | 0.074 | 0.071 | 0.522 | 0.069 | 0.077 | 0.426 |
| RMS total HOA cornea | 0.267 | 0.279 | 0.709 | 0.383 | 0.343 | 0.720 | 0.317 | 0.276 | 0.438 | 0.303 | 0.279 | 0.662 |
Abbreviations: RMS = root mean square; HOA = higher-order aberration; CF = anterior cornea; CB = posterior cornea; Cornea = combined corneal surfaces; Coma = coma aberrations (Z3±1); Trefoil = trefoil aberrations (Z3±3); SA = spherical aberration (Z40); 2° Astigmatism = secondary astigmatism (Z4±2); Tetrafoil = tetrafoil aberrations (Z4±4); Total HOA = total higher order aberration (Z3-8). P = difference between surgical groups by Mann-Whitney U test. Significant differences in bold.
Table 3.
Mean HOA magnitude (RMS in μm) at 6.0-mm optical zone by surgery type at baseline, 3, 6, and 12 months after surgery.
| Baseline |
3 Months |
6 Months |
12 Months |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | UT-DSAEK | DMEK | P | |
| RMS coma CF | 0.390 | 0.419 | 0.503 | 0.461 | 0.453 | 0.742 | 0.383 | 0.414 | 0.977 | 0.402 | 0.392 | 0.684 |
| RMS trefoil CF | 0.276 | 0.300 | 0.575 | 0.430 | 0.279 | 0.900 | 0.325 | 0.257 | 0.655 | 0.297 | 0.267 | 0.861 |
| RMS SA CF | 0.353 | 0.306 | 0.423 | 0.340 | 0.376 | 0.449 | 0.309 | 0.336 | 0.479 | 0.327 | 0.348 | 0.522 |
| RMS 2° Astigmatism CF | 0.135 | 0.176 | 0.717 | 0.173 | 0.168 | 0.816 | 0.153 | 0.142 | 0.383 | 0.137 | 0.131 | 0.443 |
| RMS tetrafoil CF | 0.165 | 0.236 | 0.869 | 0.243 | 0.241 | 0.823 | 0.229 | 0.173 | 0.105 | 0.182 | 0.175 | 0.503 |
| RMS total HOA CF | 0.716 | 0.813 | 0.860 | 0.897 | 0.829 | 0.347 | 0.739 | 0.724 | 0.900 | 0.725 | 0.723 | 0.839 |
| RMS coma CB | 0.167 | 0.178 | 0.982 | 0.237 | 0.136 | 0.002 | 0.225 | 0.110 | 0.002 | 0.209 | 0.098 | <0.001 |
| RMS trefoil CB | 0.140 | 0.193 | 0.089 | 0.317 | 0.193 | 0.034 | 0.254 | 0.151 | 0.017 | 0.234 | 0.158 | 0.125 |
| RMS SA CB | 0.156 | 0.192 | 0.160 | 0.193 | 0.166 | 0.426 | 0.184 | 0.153 | 0.342 | 0.166 | 0.143 | 0.560 |
| RMS 2° Astigmatism CB | 0.066 | 0.066 | 0.042 | 0.158 | 0.052 | <0.001 | 0.121 | 0.046 | <0.001 | 0.114 | 0.048 | <0.001 |
| RMS tetrafoil CB | 0.073 | 0.104 | 0.621 | 0.216 | 0.082 | <0.001 | 0.108 | 0.075 | 0.093 | 0.103 | 0.074 | 0.045 |
| RMS total HOA CB | 0.329 | 0.401 | 0.339 | 0.621 | 0.346 | <0.001 | 0.505 | 0.288 | <0.001 | 0.459 | 0.284 | <0.001 |
| RMS coma cornea | 0.451 | 0.435 | 0.965 | 0.578 | 0.541 | 0.992 | 0.492 | 0.471 | 0.479 | 0.500 | 0.440 | 0.256 |
| RMS trefoil cornea | 0.283 | 0.367 | 0.668 | 0.623 | 0.381 | 0.313 | 0.489 | 0.344 | 0.165 | 0.419 | 0.355 | 0.528 |
| RMS SA cornea | 0.303 | 0.251 | 0.475 | 0.297 | 0.341 | 0.438 | 0.302 | 0.304 | 0.961 | 0.318 | 0.323 | 0.892 |
| RMS 2° Astigmatism cornea | 0.157 | 0.175 | 0.733 | 0.268 | 0.200 | 0.432 | 0.198 | 0.166 | 0.105 | 0.185 | 0.158 | 0.362 |
| RMS tetrafoil cornea | 0.197 | 0.286 | 0.660 | 0.327 | 0.292 | 0.503 | 0.283 | 0.211 | 0.295 | 0.234 | 0.211 | 0.869 |
| RMS total HOA cornea | 0.757 | 0.859 | 0.878 | 1.154 | 0.966 | 0.655 | 0.954 | 0.812 | 0.256 | 0.897 | 0.805 | 0.313 |
Abbreviations: RMS = root mean square; HOA = higher-order aberration; CF = anterior cornea; CB = posterior cornea; Cornea = combined corneal surfaces; Coma = coma aberrations (Z3±1); Trefoil = trefoil aberrations (Z3±3); SA = spherical aberration (Z40); 2° Astigmatism = secondary astigmatism (Z4±2); Tetrafoil = tetrafoil aberrations (Z4±4); Total HOA = total higher order aberration (Z3-8). P = difference between surgical groups by Mann-Whitney U test. Significant differences in bold.
Figure 1.
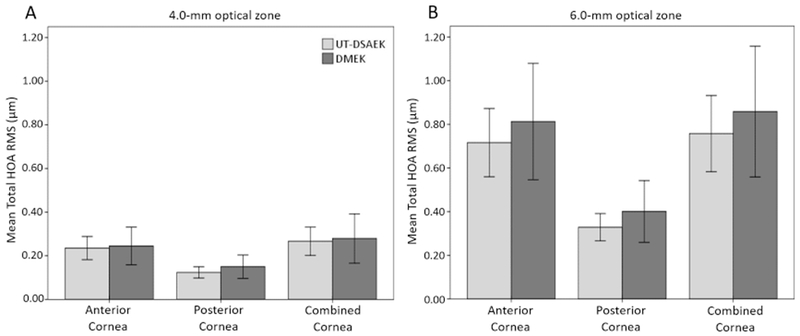
Baseline mean total higher-order aberration (HOA) (root-mean-square microns) of DMEK and UT-DSAEK groups by corneal surface at the 4.0-mm optical zone (A) and 6.0-mm optical zone (B). Total HOA = total higher-order aberration (Z3-8); Error bars = 95% confidence interval.
Post-operative HOA analysis comparing UT-DSAEK and DMEK (Table 2,3)
At 3, 6, and 12 months after surgery, there were no significant differences in anterior surface or combined surface HOA between UT-DSAEK and DMEK with one exception: combined surface spherical aberration at 3 months (4.0-mm OZ, DMEK>UT-DSAEK, Table 2).
For posterior surface aberrations of the cornea there were several significant differences between DMEK and UT-DSAEK. In all instances DMEK had lower magnitude HOA than UT-DSAEK. Coma and total HOA were significantly lower in DMEK at 3, 6, and 12 months (4.0- and 6.0-mm OZ). At 3 and 6 months, trefoil and secondary astigmatism were significantly lower in DMEK (4.0- and 6.0-mm OZ), and trefoil continued to be lower at 12 months in the 4.0-mm OZ. Tetrafoil was significantly lower in DMEK at 3 months in the 4.0-mm OZ (Table 2) and3 and 12 months in the 6.0-mm OZ (Table 3).
Post-operative HOA analysis of UT-DSAEK compared to baseline (Table 4; Fig 2, 3)
Table 4.
Mean HOA magnitude (RMS in μm) at 3, 6, and 12 months after UT-DSAEK compared to baseline at 4.0- and 6.0-mm optical zones.
| 3 Months |
6 Months |
12 Months |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4.0-mm optical zone |
6.0-mm optical zone |
4.0-mm optical zone |
6.0-mm optical zone |
4.0-mm optical zone |
6.0-mm optical zone |
|||||||
| Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | |
| Coma CF | 0.182 | 0.984 | 0.461 | 0.570 | 0.155 | 0.733 | 0.383 | 0.885 | 0.157 | 0.628 | 0.402 | 0.749 |
| Trefoil CF | 0.099 | 0.710 | 0.430 | 0.942 | 0.088 | 0.278 | 0.325 | 0.657 | 0.082 | 0.536 | 0.297 | 0.853 |
| SA CF | 0.068 | 0.934 | 0.340 | 0.804 | 0.072 | 0.901 | 0.309 | 0.337 | 0.076 | 0.726 | 0.327 | 0.502 |
| 2° Astigmatism CF | 0.062 | 0.918 | 0.173 | 0.364 | 0.046 | 0.718 | 0.153 | 0.146 | 0.044 | 0.741 | 0.137 | 0.489 |
| Tetrafoil CF | 0.086 | 0.180 | 0.243 | 0.079 | 0.061 | 0.992 | 0.229 | 0.079 | 0.058 | 0.591 | 0.182 | 0.710 |
| Total HOA CF | 0.274 | 0.942 | 0.897 | 0.820 | 0.230 | 0.967 | 0.739 | 0.934 | 0.228 | 0.885 | 0.725 | 0.733 |
| Coma CB | 0.109 | 0.119 | 0.237 | 0.081 | 0.092 | 0.421 | 0.225 | 0.132 | 0.079 | 0.992 | 0.209 | 0.164 |
| Trefoil CB | 0.132 | <0.001 | 0.317 | 0.001 | 0.117 | <0.001 | 0.254 | 0.004 | 0.102 | 0.003 | 0.234 | 0.023 |
| SA CB | 0.047 | 0.142 | 0.193 | 0.187 | 0.045 | 0.391 | 0.184 | 0.317 | 0.041 | 0.765 | 0.166 | 0.942 |
| 2° Astigmatism CB | 0.041 | 0.037 | 0.158 | <0.001 | 0.029 | 0.231 | 0.121 | 0.003 | 0.028 | 0.457 | 0.114 | 0.001 |
| Tetrafoil CB | 0.075 | <0.001 | 0.216 | <0.001 | 0.034 | 0.332 | 0.108 | 0.239 | 0.034 | 0.302 | 0.103 | 0.056 |
| Total HOA CB | 0.217 | 0.001 | 0.621 | <0.001 | 0.179 | 0.002 | 0.505 | 0.001 | 0.159 | 0.036 | 0.459 | 0.009 |
| Coma cornea | 0.244 | 0.112 | 0.578 | 0.265 | 0.198 | 0.613 | 0.492 | 0.628 | 0.209 | 0.212 | 0.500 | 0.270 |
| Trefoil cornea | 0.184 | 0.023 | 0.623 | 0.036 | 0.155 | 0.045 | 0.489 | 0.013 | 0.137 | 0.065 | 0.419 | 0.076 |
| SA cornea | 0.050 | 0.549 | 0.297 | 0.733 | 0.066 | 0.718 | 0.302 | 0.934 | 0.067 | 0.563 | 0.318 | 0.657 |
| 2° Astigmatism cornea | 0.092 | 0.163 | 0.268 | 0.015 | 0.062 | 0.529 | 0.198 | 0.081 | 0.058 | 0.657 | 0.185 | 0.380 |
| Tetrafoil cornea | 0.112 | 0.103 | 0.327 | 0.017 | 0.074 | 0.733 | 0.283 | 0.114 | 0.069 | 0.885 | 0.234 | 0.297 |
| Total HOA cornea | 0.383 | 0.034 | 1.154 | 0.021 | 0.317 | 0.122 | 0.954 | 0.049 | 0.303 | 0.170 | 0.897 | 0.114 |
Abbreviations: RMS = root mean square; HOA = higher-order aberration; CF = anterior cornea; CB = posterior cornea; Cornea = combined corneal surfaces; Coma = coma aberrations (Z3±1); Trefoil = trefoil aberrations (Z3±3); SA = spherical aberration (Z40); 2° Astigmatism = secondary astigmatism (Z4±2); Tetrafoil = tetrafoil aberrations (Z4±4); Total HOA = total higher order aberration (Z3-8). P = difference between time point and baseline by Mann-Whitney U test. Significant differences in bold.
Figure 2.
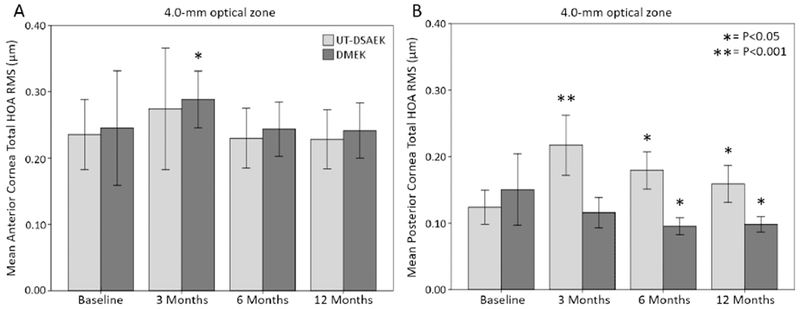
Mean total higher-order aberration (HOA) (root-mean-square microns) at the 4.0-mm optical zone of the anterior corneal surface (A) and the posterior corneal surface (B) at baseline, 3 months, 6 months, and 12 months after DMEK or UT-DSAEK. Total HOA = total higher-order aberration (Z3-8). Error bars = 95% confidence interval. P indicates difference of group from baseline HOA.
Figure 3.
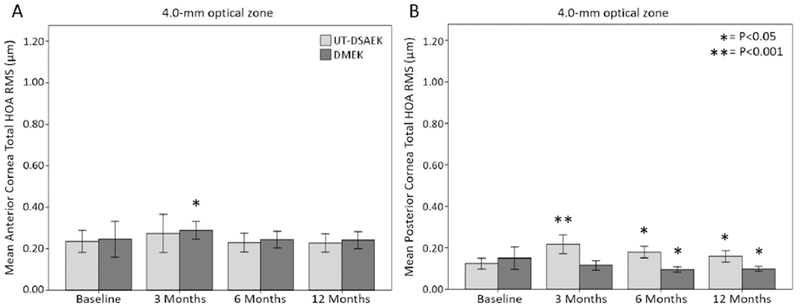
Mean total higher-order aberration (HOA) (root-mean-square microns) at the 6.0-mm optical zone of the anterior corneal surface (A) and the posterior corneal surface (B) at baseline, 3 months, 6 months, and 12 months after DMEK or UT-DSAEK. Total HOA = total higher-order aberration (Z3-8). Error bars = 95% confidence interval. P indicates difference of group from baseline HOA.
At 3, 6, and 12 months after surgery there were no significant differences in anterior surface HOA compared to baseline for UT-DSAEK at 4.0- or 6.0-mm OZ (Fig 2A, 3A).
For the combined surfaces, trefoil was higher at 3 and 6 months (4.0- and 6.0-mm OZ) compared to baseline. Secondary astigmatism and tetrafoil were higher at 3 months (6.0-mm OZ). Total HOA were higher in the 4.0- and 6.0-mm OZ at 3 months, but only the 6.0-mm OZ at 6 months.
For the posterior surface there were multiple significant differences in HOA compared to baseline, some of which persisted to the 12-month follow-up. At 3, 6, and 12 months, UT-DSAEK had higher trefoil and total HOA in both 4.0- and 6.0-mm OZ compared to baseline (Fig 2B, 3B). Secondary astigmatism was higher in both 4.0- and 6.0-mm OZ at 3 months and in the 6.0-mm OZ alone at 6 and 12 months. At 3 months, tetrafoil was higher in the 4.0- and 6.0-mm OZ.
Post-operative HOA analysis of DMEK compared to baseline (Table 5; Fig 2, 3)
Table 5.
Mean HOA magnitude (RMS in μm) at 3, 6, and 12 months after DMEK compared to baseline at 4.0- and 6.0-mm optical zones.
| 3 Months |
6 Months |
12 Months |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4.0-mm optical zone |
6.0-mm optical zone |
4.0-mm optical zone |
6.0-mm optical zone |
4.0-mm optical zone |
6.0-mm optical zone |
|||||||
| Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | Mean RMS | P | |
| Coma CF | 0.200 | 0.035 | 0.453 | 0.703 | 0.172 | 0.197 | 0.414 | 0.718 | 0.161 | 0.386 | 0.392 | 0.718 |
| Trefoil CF | 0.096 | 0.134 | 0.279 | 0.726 | 0.090 | 0.180 | 0.257 | 0.563 | 0.100 | 0.163 | 0.267 | 0.992 |
| SA CF | 0.085 | 0.076 | 0.376 | 0.170 | 0.067 | 0.773 | 0.336 | 0.687 | 0.077 | 0.140 | 0.348 | 0.348 |
| 2° Astigmatism CF | 0.056 | 0.066 | 0.168 | 0.122 | 0.052 | 0.016 | 0.142 | 0.563 | 0.039 | 0.733 | 0.131 | 0.869 |
| Tetrafoil CF | 0.080 | 0.918 | 0.241 | 0.151 | 0.061 | 0.687 | 0.173 | 0.741 | 0.063 | 0.695 | 0.175 | 0.687 |
| Total HOA CF | 0.288 | 0.015 | 0.829 | 0.227 | 0.243 | 0.288 | 0.724 | 0.929 | 0.241 | 0.332 | 0.723 | 0.877 |
| Coma CB | 0.052 | 0.051 | 0.136 | 0.327 | 0.046 | 0.046 | 0.110 | 0.167 | 0.039 | 0.007 | 0.098 | 0.081 |
| Trefoil CB | 0.071 | 0.885 | 0.193 | 0.650 | 0.054 | 0.110 | 0.151 | 0.451 | 0.064 | 0.620 | 0.158 | 0.695 |
| SA CB | 0.044 | 0.680 | 0.166 | 0.364 | 0.039 | 0.204 | 0.153 | 0.204 | 0.036 | 0.044 | 0.143 | 0.063 |
| 2° Astigmatism CB | 0.021 | 0.342 | 0.052 | 0.861 | 0.019 | 0.450 | 0.046 | 0.464 | 0.021 | 0.157 | 0.048 | 0.967 |
| Tetrafoil CB | 0.025 | 0.445 | 0.082 | 0.657 | 0.025 | 0.577 | 0.075 | 0.451 | 0.026 | 0.702 | 0.074 | 0.433 |
| Total HOA CB | 0.116 | 0.132 | 0.346 | 0.427 | 0.095 | 0.017 | 0.288 | 0.078 | 0.098 | 0.025 | 0.284 | 0.044 |
| Coma cornea | 0.233 | 0.005 | 0.541 | 0.201 | 0.190 | 0.135 | 0.471 | 0.657 | 0.175 | 0.302 | 0.440 | 0.901 |
| Trefoil cornea | 0.141 | 0.035 | 0.381 | 0.749 | 0.116 | 0.157 | 0.344 | 0.170 | 0.138 | 0.085 | 0.355 | 0.297 |
| SA cornea | 0.076 | 0.151 | 0.341 | 0.095 | 0.056 | 0.869 | 0.304 | 0.427 | 0.067 | 0.348 | 0.323 | 0.127 |
| 2° Astigmatism cornea | 0.072 | 0.011 | 0.200 | 0.034 | 0.064 | 0.014 | 0.166 | 0.489 | 0.052 | 0.160 | 0.158 | 0.496 |
| Tetrafoil cornea | 0.097 | 0.749 | 0.292 | 0.183 | 0.071 | 0.219 | 0.211 | 0.765 | 0.077 | 0.483 | 0.211 | 0.687 |
| Total HOA cornea | 0.343 | 0.007 | 0.966 | 0.037 | 0.276 | 0.219 | 0.812 | 0.380 | 0.279 | 0.183 | 0.805 | 0.409 |
Abbreviations: RMS = root mean square; HOA = higher-order aberration; CF = anterior cornea; CB = posterior cornea; Cornea = combined corneal surfaces; Coma = coma aberrations (Z3±1); Trefoil = trefoil aberrations (Z3±3); SA = spherical aberration (Z40); 2° Astigmatism = secondary astigmatism (Z4±2); Tetrafoil = tetrafoil aberrations (Z4±4); Total HOA = total higher order aberration (Z3-8). P = difference between time point and baseline by Mann-Whitney U test. Significant differences in bold.
For the anterior surface, coma and total HOA (Fig 2A) were higher in the 4.0-mm OZ in DMEK at 3 months compared to preoperative baseline. Secondary astigmatism was higher at 6 months (4.0-mm OZ). No differences were detected at 12 months.
For the combined surface, coma and trefoil were higher in DMEK at 3 months (4.0-mm OZ). Secondary astigmatism was higher in DMEK at 3 months (4.0- and 6.0-mm OZ) and 6 months (4.0-mm OZ). Total HOA was higher at 3 months in the 4.0- and 6.0-mm OZ. No differences were detected at 12 months.
For posterior surface, there were no differences detected at 3 months in DMEK compared to baseline. Coma and total HOA were significantly lower at 6 and 12 months (4.0-mm OZ) and total HOA was also lower in the 6.0-mm OZ at 12 months (Fig 2B, 3B). Spherical aberration was lower at 12 months (4.0-mm OZ).
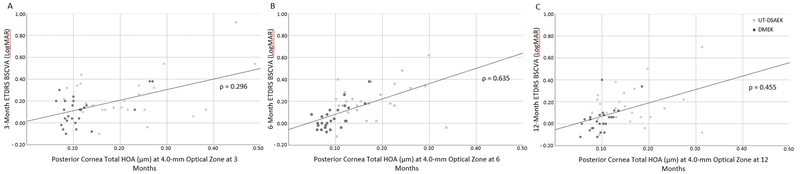
Correlation of ETDRS Visual acuity with HOA (Table 6; Fig 4A, B, C)
Table 6.
Spearman correlation between best spectacle-corrected visual acuity and higher order aberrations of the central 4.0- and 6.0-mm optical zones at 3, 6, and 12 months after surgery.
| 3 Months |
6 Months |
12 Months |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4.0-mm optical zone | 6.0-mm optical zone | 4.0-mm optical zone | 6.0-mm optical zone | 4.0-mm optical zone | 6.0-mm optical zone | |||||||
| ρ | P | ρ | P | ρ | P | ρ | P | ρ | P | ρ | P | |
| RMS coma CF | 0.131 | 0.364 | 0.159 | 0.271 | 0.171 | 0.236 | 0.234 | 0.101 | 0.041 | 0.778 | 0.159 | 0.271 |
| RMS trefoil CF | 0.129 | 0.374 | 0.103 | 0.477 | 0.168 | 0.243 | 0.125 | 0.386 | 0.040 | 0.782 | 0.050 | 0.731 |
| RMS SA CF | 0.051 | 0.727 | 0.075 | 0.603 | 0.165 | 0.251 | −0.086 | 0.551 | −0.120 | 0.407 | −0.172 | 0.233 |
| RMS 2° Astigmatism CF | 0.042 | 0.775 | 0.018 | 0.904 | 0.041 | 0.778 | 0.128 | 0.374 | 0.095 | 0.512 | 0.157 | 0.278 |
| RMS tetrafoil CF | 0.251 | 0.078 | 0.262 | 0.066 | 0.070 | 0.627 | 0.233 | 0.103 | 0.329 | 0.020 | 0.250 | 0.080 |
| RMS total HOA CF | 0.177 | 0.220 | 0.200 | 0.165 | 0.209 | 0.145 | 0.204 | 0.156 | 0.095 | 0.513 | 0.173 | 0.230 |
| RMS coma CB | 0.299 | 0.035 | 0.188 | 0.190 | 0.443 | 0.001 | 0.409 | 0.003 | 0.226 | 0.114 | 0.292 | 0.039 |
| RMS trefoil CB | 0.133 | 0.356 | 0.043 | 0.769 | 0.518 | <0.001 | 0.388 | 0.005 | 0.399 | 0.004 | 0.351 | 0.012 |
| RMS SA CB | 0.073 | 0.615 | 0.124 | 0.391 | 0.178 | 0.216 | 0.338 | 0.016 | 0.103 | 0.477 | 0.227 | 0.112 |
| RMS 2° Astigmatism CB | 0.223 | 0.120 | 0.354 | 0.012 | 0.331 | 0.019 | 0.528 | <0.001 | 0.001 | 0.996 | 0.118 | 0.413 |
| RMS tetrafoil CB | 0.292 | 0.040 | 0.246 | 0.085 | 0.245 | 0.087 | 0.468 | 0.001 | 0.165 | 0.252 | 0.239 | 0.094 |
| RMS total HOA CB | 0.296 | 0.037 | 0.269 | 0.058 | 0.635 | <0.001 | 0.580 | <0.001 | 0.455 | 0.001 | 0.456 | 0.001 |
| RMS coma cornea | 0.309 | 0.029 | 0.204 | 0.156 | 0.308 | 0.030 | 0.260 | 0.068 | 0.091 | 0.531 | 0.153 | 0.289 |
| RMS trefoil cornea | 0.003 | 0.984 | 0.076 | 0.602 | 0.179 | 0.213 | 0.205 | 0.152 | 0.156 | 0.279 | 0.169 | 0.241 |
| RMS SA cornea | −0.029 | 0.841 | 0.053 | 0.716 | 0.180 | 0.210 | −0.056 | 0.698 | −0.105 | 0.470 | −0.142 | 0.326 |
| RMS 2° Astigmatism cornea | 0.179 | 0.214 | 0.261 | 0.067 | 0.109 | 0.450 | 0.112 | 0.439 | 0.043 | 0.767 | 0.099 | 0.493 |
| RMS tetrafoil cornea | 0.148 | 0.306 | 0.139 | 0.336 | 0.089 | 0.540 | 0.211 | 0.142 | 0.314 | 0.026 | 0.284 | 0.046 |
| RMS total HOA cornea | 0.276 | 0.052 | 0.242 | 0.091 | 0.327 | 0.020 | 0.279 | 0.049 | 0.186 | 0.196 | 0.194 | 0.178 |
Abbreviations: RMS = root mean square; HOA = higher-order aberration; CF = anterior cornea; CB = posterior cornea; Cornea = combined corneal surfaces; Coma = coma aberrations (Z3±1); Trefoil = trefoil aberrations (Z3±1); SA = spherical aberration (Z40); 2° Astigmatism = secondary astigmatism (Z4±2); Tetrafoil = tetrafoil aberrations (Z4±4); Total HOA = total higher order aberration (Z3-8). ρ = Spearman’s correlation coefficient. P = significance of Spearman correlation. Significant correlations in bold.
Figure 4.
Correlation of ETDRS LogMAR best spectacle-corrected visual acuity (BSCVA) with posterior corneal total higher-order aberration (HOA) at the 4.0-mm optical zone 3 months (A), 6 months (B), and 12 months (C) after DMEK or UT-DSAEK. Total HOA = total higher-order aberration (Z3-8). ρ = Spearman’s rho.
Table 6 gives Spearman correlation values (ρ) with relevant P-values for all time points and HOA measured. A glance at the table demonstrates that there are no strong positive or negative correlations between any HOA category and logMAR BSCVA (assuming a strong correlation to be |ρ| ≤ 0.7). All significant correlations are positive. Moderate positive correlations (0.4 ≤ ρ < 0.7) exist in only the posterior surface HOA sets. These include: coma (4.0- and 6.0-mm OZ), trefoil (4.0-mm OZ), secondary astigmatism (6.0-mm OZ), tetrafoil (6.0-mm OZ) at 6 months. Contributions of total HOA correlate moderately with BSCVA in both optical zones at 6 and 12 months (ρ≤0.635, Fig 4B, C). There are many weak correlations (0.2 ≤ ρ < 0.4) observed in the combined surface analysis, but all correspond to weak correlations in the posterior group except for tetrafoil at 12 months (corresponding to a weak correlation of the anterior surface).
Discussion
Visual acuity at 6 months was the primary outcome in the DETECT randomized controlled trial and was significantly better in the DMEK patients.9 In this study, we found that total posterior corneal HOA correlated moderately well with ETDRS visual acuity at 6 and 12 months. Complementing these findings, we also showed significantly less posterior corneal individual and total HOA in the DMEK arm of the study compared to UT-DSAEK at 3, 6, and 12 months in 4.0- and 6.0-mm optical zones. Additionally, posterior HOA decreased after DMEK, while posterior HOA increased after UT-DSAEK.
Ocular aberrations can be measured by a variety of techniques, some of which include measurements of whole eye wavefronts. Scheimpflug imaging specifically allows us to focus on changes in anterior and posterior corneal surfaces which are most relevant to studying the optical effects of endothelial keratoplasty. In contrast to previous studies that noted significance of anterior HOA,2–4 we found that certain individual and total HOA of the posterior corneal surface correlated strongest with the BSCVA at 6 and 12 months following DMEK or UT-DSAEK. In our cohort, HOA of the anterior corneal surface did not differ significantly between DMEK and UT-DSAEK at any postoperative timepoint (Tables 2, 3) and only anterior trefoil correlated weakly with BSCVA at the 12-month time point (Table 6). This contrasts with previous studies looking at DSEK only.3,8 The difference between these results and ours may reflect graft thickness differences, which were tightly controlled and thinner in our UT-DSAEK arm, and possibly the methods used to measure and calculate HOA. A previous study comparing anterior and posterior corneal aberrations using Scheimpflug imaging found no significant differences between DMEK and DSEK in anterior HOA and found certain posterior individual and total HOA to be significantly different between the 2 types of grafts, similar to our results. However, they were unable to find a positive correlation between these HOA and visual acuity. This retrospective study also differed from ours in that postop measurement times were not matched between patients and DSAEK graft thickness was not tightly controlled.5 In our study, we did observe wider ranges in both individual and total HOA in the UT-DSAEK group as compared to DMEK (Fig 4), which could reflect variation in pre- and postoperative graft thicknesses and uniformity. The subjects with lowest BSCVA (Fig 4) did not have any slit lamp findings or preoperative conditions (Table 1) to explain their poorer outcomes. Our data suggests that posterior corneal HOA play an important role in visual acuity following endothelial) keratoplasty.
Interestingly, we found that posterior corneal HOA actually increased significantly in the UT-DSAEK group from baseline to 3 months after surgery and remained significantly larger at 6 and 12 months. Similar increases in HOA after DSAEK have been previously demonstrated and this is perhaps not surprising given the meniscus-shaped graft and changes in posterior corneal surface after DSAEK.14,15 In contrast, the DMEK group demonstrated a significant decrease in total HOA from baseline to 6 and 12 months after surgery, supporting the faster visual recovery demonstrated by the DETECT trial and other studies.5,9,16,17 Dickman and colleagues demonstrated improved posterior corneal surface regularity, graft symmetry, and decreased HOA with the use of thinner DSAEK grafts.18 It appears that the near anatomic replacement of tissue in DMEK reduces the amount of induced posterior corneal surface distortion thereby contributing to improved visual acuity compared to UT-DSAEK.
Although the refractive indices between aqueous and posterior cornea are small, the refractive effect of the posterior surface is not insignificant. Measurements of posterior corneal curvature are very important for toric IOL planning.19 Reported contributions of the posterior cornea have included spherical aberration,20 a fraction of the total corneal astigmatism in normal and post keratoplasty eyes,21,22 alterations in posterior corneal radius of curvature,23 and coma contributions in keratoconus patients.24 In addition, the internal optics of the eye, which include both crystalline lens and posterior cornea, are known to offset aberrations created by the anterior corneal surface.21,22,25 HOA of the posterior corneal surface after UT-DSAEK appear to improve over time (Fig 2, 3). Previous studies have found that BSCVA and posterior corneal HOA after DSAEK may slowly improve up to several years after surgery.14,26 The DETECT trial has pre-specified, masked follow-up time points up to 24 months after surgery, which may demonstrate that both HOA and visual acuity differences between DMEK and UT-DSAEK decrease with more time.
We observed that patients with UT-DSAEK gain better vision after surgery despite worsening of posterior HOA. Post-operatively in the UT-DSAEK group, there must be other factors that also correlate positively with visual outcome and balance the deleterious effect of posterior HOA. The most obvious would be elimination of central guttae and the improvement in corneal thickening/edema following surgery. Changes in the ultrastructure of the corneal collagen with improved endothelial pump function may improve overall BSCVA despite increased posterior HOA.
Limitations of this study include the fact that HOA was a pre-specified secondary outcome of DETECT and thus the study may not have been powered to determine all differences. A larger sample size may have found more differences on the anterior and combined surfaces. The differences in visual outcomes between groups may reflect other variables as well including forward light scatter, corneal backscatter, and recipient age;1 however, there was no difference in preoperative age between cohorts in this study. We did not find a strong or moderate correlation between anterior corneal HOA and visual acuity which may be due to the fact that most of our patients had relatively clear corneas at baseline with central guttae rather than edema being the visually symptomatic finding. Severe or longstanding pre-operative corneal edema can lead to subepithelial fibrosis and other anterior stromal irregularities which may have an impact on post-op acuity.2 In these severe cases, either type of endothelial keratoplasty may be less likely to resolve these corneal changes. Worse pre-operative disease severity may therefore diminish the gains seen with DMEK in our study and could explain some of the variation seen in the literature. Additionally, correlation does not prove causation and categorization of correlation values as weak, moderate, or strong varies considerably in the literature.
A “nanothin” DSAEK technique has recently been reported which may further reduce HOA and improve visual acuity while maintaining the advantages of simpler tissue handling and reduced graft detachments.27 Ultimately, the differences in BSCVA between DMEK and UT-DSAEK were small and reflect the excellent outcomes that are often obtained with either technique. However, even small differences in higher-order aberrations may produce clinically significant variations in visual recovery time, contrast sensitivity, and patient satisfaction.
In conclusion, DMEK results in reduction of posterior corneal HOA while UT-DSAEK actually increases posterior corneal HOA at least up to 12 months post-operatively. Further, posterior corneal HOA are shown to correlate more strongly with post-operative BSCVA at 6 and 12 months after DMEK and UT-DSAEK than anterior corneal aberrations. These observations support the concept that differences in posterior corneal contour between endothelial keratoplasty techniques have a clinically significant impact on visual outcomes following DMEK and UT-DSAEK.
Acknowledgments
Financial Support:
Supported by the National Institutes of Health (Bethesda, MD) grant K23 EY025025 (J.N.) and P30 EY010572 (CEI departmental core grant); and by unrestricted departmental funding (UCSF and CEI) from Research to Prevent Blindness (New York, NY).
Lions VisionGift, Portland, OR - Contributed to the design and implementation of the study, providing all of the tissue for the study, and randomizing all eyes to treatment arm.
The remaining sponsors or funding organizations had no role in the design or conduct of this research
Abbreviations and Acronyms:
- BSCVA
best spectacle-corrected visual acuity
- DMEK
Descemet membrane endothelial keratoplasty
- DSAEK
Descemet stripping automated endothelial keratoplasty
- EK
endothelial keratoplasty
- HOA
higher-order aberration
- logMAR
logarithm of the minimum angle of resolution
- RMS
root mean square
- UT-DSAEK
ultrathin Descemet stripping automated endothelial keratoplasty
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Meeting Presentation: Some data previously presented at the Association for Research in Vision and Ophthalmology Annual Meeting, 2018
Conflict of Interest: No conflicting relationship exists for any author
References:
- 1.Patel S, Baratz K, Hodge D, et al. The effect of corneal light scatter on vision after Descemet stripping with endothelial keratoplasty. Arch Ophthalmol 2009;12:153–160. [DOI] [PubMed] [Google Scholar]
- 2.Van Dijk K, Droutsas K, Hou J, et al. Optical quality of the cornea after Descemet membrane endothelial keratoplasty. Am J Ophthalmol 2014;158:71–79. e1. [DOI] [PubMed] [Google Scholar]
- 3.Nielsen E, Ivarsen A, Kristensen S, Hjortdal J. Fuchs’ endothelial corneal dystrophy: a controlled prospective study on visual recovery after endothelial keratoplasty. Acta Ophthalmol 2016;94:780–787. [DOI] [PubMed] [Google Scholar]
- 4.Hayashi T, Yamaguchi T, Yuda K, et al. Topographic characteristics after Descemet’s membrane endothelial keratoplasty and Descemet’s stripping automated endothelial keratoplasty. PLoS One 2017;12:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rudolph M, Laaser K, Bachmann BO, et al. Corneal Higher-Order Aberrations after Descemet’s Membrane Endothelial Keratoplasty. Ophthalmology 2012;119:528–535. [DOI] [PubMed] [Google Scholar]
- 6.Singh A, Zarei-Ghanavati M, Avadhanam V, Liu C. Systematic Review and Meta-Analysis of Clinical Outcomes of Descemet Membrane Endothelial Keratoplasty Versus Descemet Stripping Endothelial Keratoplasty/Descemet Stripping Automated Endothelial Keratoplasty. Cornea 2017;36:1437–1443. [DOI] [PubMed] [Google Scholar]
- 7.Pavlovic I, Shajari M, Herrmann E, et al. Meta-Analysis of Postoperative Outcome Parameters Comparing Descemet Membrane Endothelial Keratoplasty Versus Descemet Stripping Automated Endothelial Keratoplasty. Cornea 2017;36:1445–1451. [DOI] [PubMed] [Google Scholar]
- 8.Patel SV, Baratz KH, Maguire LJ, et al. Anterior Corneal Aberrations after Descemet’s Stripping Endothelial Keratoplasty for Fuchs’ Endothelial Dystrophy. Ophthalmology 2012;119:1522–1529. [DOI] [PubMed] [Google Scholar]
- 9.Chamberlain W, Lin CC, Austin A, et al. Descemet Endothelial Thickness Comparison Trial: A Randomized Trial Comparing Ultrathin Descemet Stripping Automated Endothelial Keratoplasty with Descemet Membrane Endothelial Keratoplasty. Ophthalmology 2018:1–8. [DOI] [PubMed] [Google Scholar]
- 10.Yamaguchi T, Negishi K, Yamaguchi K, et al. Effect of Anterior and Posterior Corneal Surface Irregularity on Vision after Descemet-stripping Endothelial Keratoplasty. J Cataract Refract Surg 2009;35:688–694. [DOI] [PubMed] [Google Scholar]
- 11.Pantanelli SM, Sabesan R, Ching SST, et al. Visual performance with wave aberration correction after penetrating, deep anterior lamellar, or endothelial keratoplasty. Investig Ophthalmol Vis Sci 2012;53:4797–4804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McLaren JW, Patel SV. Modeling the effect of forward scatter and aberrations on visual acuity after endothelial keratoplasty. Investig Ophthalmol Vis Sci 2012;53:5545–5551. [DOI] [PubMed] [Google Scholar]
- 13.Salmon TO, van de Pol C. Normal-eye Zernike coefficients and root-mean-square wavefront errors. J Cataract Refract Surg 2006;32:2064–2074. [DOI] [PubMed] [Google Scholar]
- 14.Hindman HB, Huxlin KR, Pantanelli SM, et al. Post-DSAEK optical changes: A comprehensive prospective analysis on the role of ocular wavefront aberrations, haze, and corneal thickness. Cornea 2013;32:1567–1577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Muftuoglu O, Prasher P, Bowman RW, et al. Corneal Higher-Order Aberrations after Descemet’s Stripping Automated Endothelial Keratoplasty. Ophthalmology 2010;117:878–884. e6. [DOI] [PubMed] [Google Scholar]
- 16.Heinzelmann S, Böhringer D, Eberwein P, et al. Outcomes of Descemet membrane endothelial keratoplasty, Descemet stripping automated endothelial keratoplasty and penetrating keratoplasty from a single centre study. Graefe’s Arch Clin Exp Ophthalmol 2016;254:515–522. [DOI] [PubMed] [Google Scholar]
- 17.Droutsas K, Lazaridis A, Papaconstantinou D, et al. Visual Outcomes after Descemet Membrane Endothelial Keratoplasty Versus Descemet Stripping Automated Endothelial Keratoplasty-Comparison of Specific Matched Pairs. Cornea 2016;35:765–771. [DOI] [PubMed] [Google Scholar]
- 18.Dickman MM, Cheng YYY, Berendschot TTJM, et al. Effects of Graft Thickness and Asymmetry on Visual Gain and Aberrations after Descemet Stripping Automated Endothelial Keratoplasty. JAMA Ophthalmol 2013;131:737–744. [DOI] [PubMed] [Google Scholar]
- 19.Koch DD, Jenkins RB, Weikert MP, et al. Correcting Astigmatism with Toric Intraocular Lenses: Effect of Posterior Corneal Astigmatism. J Cataract Refract Surg 2013;39:1803–1809. [DOI] [PubMed] [Google Scholar]
- 20.Sicam V, Dubbelman M, van der Heijde RGL. Spherical aberration of the anterior and posterior surface of the human cornea. J Opt Soc Am A Opt Image Sci Vis 2006;23:544–549. [DOI] [PubMed] [Google Scholar]
- 21.Dubbelman M, Sicam VADP, Van Der Heijde GL. The shape of the anterior and posterior surface of the aging human cornea. Vision Res 2006;46:993–1001. [DOI] [PubMed] [Google Scholar]
- 22.Yamaguchi T, Ohnuma K, Tomida D, et al. The contribution of the posterior surface to the corneal aberrations in eyes after keratoplasty. Investig Ophthalmol Vis Sci 2011;52:6222–6229. [DOI] [PubMed] [Google Scholar]
- 23.Chen M, Sabesan R, Ahmad K, Yoon G. Correcting anterior corneal aberration and variability of lens movements in keratoconic eyes with back-surface customized soft contact lenses. Opt Lett 2007;32:3203. [DOI] [PubMed] [Google Scholar]
- 24.Chen M, Yoon G. Posterior corneal aberrations and their compensation effects on anterior corneal aberrations in keratoconic eyes. Investig Ophthalmol Vis Sci 2008;49:5645–5652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Artal P, Guirao A, Berrio E, Williams DR. Compensation of Corneal Aberrations by the Internal Optics of the Eyes. J Vis 2001;1:1–8. [DOI] [PubMed] [Google Scholar]
- 26.Li JY, Terry MA, Goshe J, et al. Three-year visual acuity outcomes after Descemet’s stripping automated endothelial keratoplasty. Ophthalmology 2012;119:1126–1129. [DOI] [PubMed] [Google Scholar]
- 27.Kurji KH, Cheung AY, Eslani M, et al. Comparison of Visual Acuity Outcomes Between Nanothin Descemet Stripping Automated Endothelial Keratoplasty and Descemet Membrane Endothelial Keratoplasty. Cornea 2018;37:1226–1231. [DOI] [PubMed] [Google Scholar]