Abstract
Eimeria spp. must be controlled in floor-reared poultry to prevent the onset of coccidiosis. Here we use an oral antibody to chicken IL-10 to prevent growth depression due to Eimeria spp. infection. Egg antibody directed against an antigenic peptide of IL-10 was produced in laying hens and measured using an ELISA. In the first experiment, egg yolk powder containing antibody to chicken IL-10 (vlpramqt conjugate) (anti-IL-10 yolk powder) was fed at 3.4 g/kg feed to determine growth response following mixed Eimeria spp. challenge. Chicks were fed either anti-IL-10 antibodies or control antibodies and challenged (d3) with either sterile saline or a 10× attenuated Eimeria spp. vaccine. Control-fed and Eimeria-challenged chicks grew 8.8% slower than those challenged with saline (P < 0.04), whereas anti-IL-10-fed Eimeria challenged chicks were not different from untreated controls. In the second trial a dose response was performed with doses of either 0 (control antibody), 0.34-, or 3.4-g anti-IL-10 yolk powder/kg feed. Control-fed, Eimeria-challenged chicks grew 10.6% slower than control saline-challenged chicks (P < 0.05); however, anti-IL-10-fed chicks fed either dose of anti-IL-10 were not different from saline-challenged chicks. Finally, the effect of anti-IL-10 on acquired immunity was investigated. Chicks were fed control or anti-IL-10 yolk powder and vaccinated with a 1× dose of Eimeria vaccine at d 3. After 14 d, antibody was removed from the diet. Chicks were either saline or 10× Eimeria challenged at d 17. We found that the anti-IL-10-fed chickens did not show a reduction in growth due to challenge; hence anti-IL-10 does not appear to affect adaptive immunity during the primary immunization. Overall, use of an antibody to IL-10 is a novel method in preventing adverse effects of Eimeria spp. infection in poultry.
Keywords: coccidiosis, egg antibody, anti-IL-10, chicken, Eimeria
INTRODUCTION
The full-length cDNA sequence of chicken interleukin-10 (IL-10) was first characterized using RNA isolated from cecal tonsils of Eimeria tenella-infected chicks (Rothwell et al., 2004). Also found was increased IL-10 mRNA expression in the intestine of E. maxima infected and susceptible chickens, but not Eimeria spp.-resistant chickens. Increased intestinal IL-10 expression 4 d following E. maxima was confirmed by Hong et al. (2006), and Shanmugasundaram et al. (2013) showed that Citristim inhibited Eimeria spp-induced IL-10 mRNA expression in cecal tonsils and reduced oocyst shedding while improving growth and feed efficiency post-infection. These finding suggested that inhibition of Eimeria-induced IL-10 might be protective against Eimeria spp. infection.
Cyktor and Turner (2011) reviewed the mechanisms by which a number of pathogens (including bacteria, protozoa, and fungi) exploit the immunosuppressive effects of IL-10 in facilitating infection. IL-10 removed systemically with either an IL-10 neutralizing antibody or IL-10 gene disruption had increased pathogen clearance and resistance, and improved resolution of the infectious process and disease severity (Cyktor and Turner, 2011). For example, IL-10 knockout mice infected with Cryptosporidium parvum had a 70% reduction in oocyst shedding (Campbell et al., 2002). Paark et al. (2012) identified a mechanism by which pathogens increased IL-10 release. An Anisakis simplex macrophage migratory inhibitory factor homologue (MIFh) increased the release of IL-10 from human peripheral mononuclear blood cells (PMBCs). Pathogen-induced up-regulation of host IL-10 provided a compelling mechanism for evading immune detection, and Jang et al. (2011) demonstrated that use of an Eimeria MIFh (eMIFh) protein as a vaccine in chickens prevented E. tenella-induced growth suppression.
Amino acid homology among the eMIFh is as low as 55%, which suggest that an eMIFh vaccine may not provide broad protection against all species of Eimeria that infect chickens. The recent discovery of an apical IL-10 receptor on intestinal epithelia suggests that IL-10 is secreted into the intestinal lumen (Kominsky et al., 2014), and indeed, fecal IL-10 levels have been detected in humans with intestinal inflammation (Konnikova et al., 2015). Direct targeting of host IL-10 in the lumen of the intestine of the chicken may serve as an alternative to controlling the immunosuppressive effects of host IL-10 up-regulated by Eimeria spp. infection. Previously it had been shown that oral IgY antibodies could be successfully used to target host proteins in the intestinal lumen (Bobeck et al., 2015). Hence, the objective of this study was to determine if an oral antibody to chicken IL-10 could prevent Eimeria-induced decreased growth rates in broiler chicks.
MATERIALS AND METHODS
Commercial recombinant chicken IL-10 was purchased from Kingfisher Biotech Inc. (St. Paul, MN). Chicken anti-IL-10 peptide specific antibodies were affinity purified at GeneTel (Madison, WI). Commercial rabbit anti-IL-10 antibody that cross-reacted with chicken IL-10 was purchased from Bioss (Woburn, MA). Goat anti-rabbit IgG HRP conjugated, and rabbit anti-chicken IgY HRP conjugate were purchased from Bethyl Laboratories (Montgomery, TX). Rabbit isotype control was an antibody to the sodium-phopshate cotransporter 2b, and was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Broiler chicks (Ross 308) for all the studies were purchased from Welp Hatchery (Bancroft, IA).
Chicken Interleukin-10 (IL-10) Peptide Selection
The sequence for chicken interleukin-10 (IL-10) was sourced from pubmed.gov protein: CAF18432 and was based on sequence analysis of Rothwell et al., 2004:
mqtccqalllllaactlpahcleptclhfsellparlrelrvkfeeikdyfqsrddelniqllsselldefkgtfgcqsvsemlrfytdevlpramqtstshqqsmgdlgnmllglkatirrchrfftcekrskaikqiketfekmdengiykamgefdifinyieeyllmrrrk
The bolded portion of the sequence, vlpramqt, was the antigenic peptide (identified by the antigen prediction program BepiPred http://www.cbs.dtu.dk/services/BepiPred/, Technical University Denmark) used for making anti-IL-10 peptide antibody. The chicken IL-10 amino acid sequence was aligned with a human IL-10 amino acid sequence (human IL-10, 2H24_A) and using the crystallized 3-D structure of human IL-10, the likelihood of antibody binding to the chicken peptide was estimated (Bobeck et al., 2015) (Figure 1A). Once the peptide was shown to be located in a region of the IL-10 molecule that could be accessed by an antibody, the 8-amino acid-long peptide sequence was made (GenScript (Piscataway, NJ). Five milligrams of peptide were conjugated to 5 mg of bovine gammaglobulin (BGG, for vaccine) or ovalbumin (OVA, for detecting peptide specific antibody) (Sigma Aldrich, St. Louis, MO) using glutaraldehyde (Fisher Scientific, Pittsburgh, PA) (Cook and Trott, 2010). Peptide-BGG or peptide-OVA conjugates were then dialyzed against phosphate buffered saline overnight using 6000–8000 molecular weight membrane tubing (Spectrapor, Los Angeles, CA). Conjugates were then frozen at –80°C in 1 mg/500 μL aliquots until needed for injection into the laying hens.
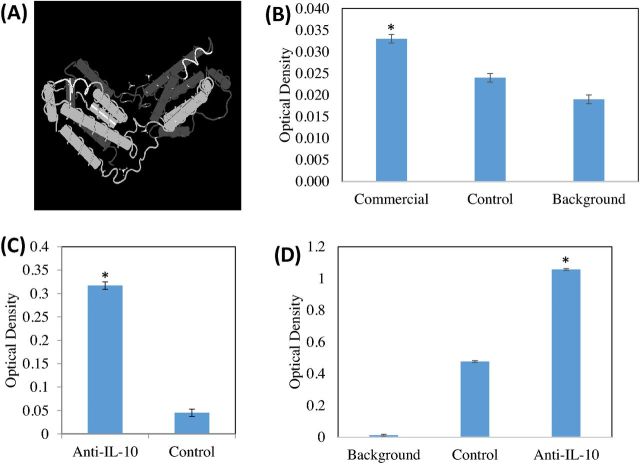
Figure 1.
Anti-IL-10 antibody binds to commercial recombinant IL-10 as well as IL-10 in the lumen of the small intestine. (A) A three-dimensional structure of human IL-10 (2H24_A, pubmed.gov) was used in combination with sequence alignment to determine the placement of the peptide used to bind to chicken IL-10. The region shown in white represented the 8 amino acid peptide used in this study. (B) Commercial rabbit anti-IL-10 antibody was shown to bind commercial recombinant chicken IL-10. This study was conducted to determine the usefulness of commercial rabbit anti-IL-10 as a chicken IL-10 capture antibody. In this experiment, commercial recombinant chicken IL-10 was bound to the plate, and the ability of commercial rabbit anti-IL-10 (commercial), rabbit isotype control (control), or the absence of any primary antibody (background) was compared (secondary antibodies used were goat anti-rabbit IgG-HRP conjugates). *Denotes different binding of commercial recombinant chicken IL-10 by the rabbit anti-IL-10 antibody and the isotype control rabbit antibody and the primary antibody blank (P < 0.05). (C) Commercial recombinant chicken IL-10 was coated on an ELISA plate and control or affinity-purified peptide specific antibody to IL-10 was used to demonstrate the specificity of the antibody in binding recombinant chicken IL-10. We demonstrate that the affinity-purified peptide antibody binds the recombinant IL-10 significantly over control antibody (P < 0.05). (D) IL-10 from intestinal contents of the duodenum of chickens was demonstrated using a capture ELISA (P < 0.05). In this ELISA, commercial rabbit anti-IL-10 was coated on the plate. Duodenal fluids from Eimeria spp.-challenged chicks were incubated on the bound rabbit anti-IL-10. Next incubation was either an affinity-purified chicken anti-IL-10 antibody (anti-IL-10) or control antibody (Control) or the secondary antibody alone (Background). Using an ELISA, IL-10 in the duodenal luminal fluid was qualitatively demonstrated in the duodenal luminal fluid of Eimeria-challenged chicks.
Antibody Production
All procedures involving chickens were approved by the University of Wisconsin College of Agricultural and Life Sciences Animal Care and Use Committee. Procedures that follow were as described in Cook and Trott (2010). Single Comb White Leghorn laying hens raised for life in cages with raised wire floor and never having been vaccinated with Eimeria spp. were injected (100 μg/chicken) with chicken IL-10-BGG-vlpramqt conjugate emulsified with Freund's Complete Adjuvant at 1:1 v/v (Fisher Scientific). Hens injected with adjuvant and BGG only (no peptide conjugate) were used for making control antibodies. Chickens were given a booster injection using the antigens described (BGG or BGG-vlpramqt conjugate) 1 wk following the initial injection; hence the only difference in antibodies from control eggs and anti-IL-10 eggs was antibody to the IL-10 peptide vlpramqt. Eggs were collected beginning 3 wk after the initial injection for a period of 6 wk. Egg yolks were separated from the albumen, lyophilized and stored at 21°C until needed. Enzyme-linked immunosorbent assays (ELISAs) were then used to determine the titer of the antibody. All antibody was fed as dried egg yolk powder.
ELISA
Briefly, 96-well plates were coated with 8.3 μg/mL peptide-OVA conjugate or OVA in coating buffer (1.59 g Na2CO3, 2.93 g NaHCO3, 0.2 g NaN3, pH 9.6, 1,000 mL total volume) overnight to allow for attachment of the peptide-OVA or OVA to the Nunc F MaxiSorp plate (Thermo-Fisher Scientific, Rochester, NY). Plates were then blocked with protein-free blocking buffer (Pierce, Rockford IL) for 1 hour. Plates were then washed 3 times with PBS-0.05% Tween (Fisher Scientific). Primary antibody to the peptide was extracted using acidified phosphate buffered saline (PBS) (pH 4.7) at a concentration of 1:10 (e.g., 0.1 g yolk powder/mL acidified PBS) overnight, and supernates were applied to the plate in 10-fold dilutions from 1:10 to 1:100,000,000. Adjuvant BBG-only injected hen eggs were used as an isotype control and were added to each plate at a 1:10 concentration. Goat anti-chicken secondary antibody (Bethyl Laboratories) was then added in blocking buffer (0.4μL 20antibody/mL blocking buffer) for 30 minutes. Plates were washed 6 times with washing buffer. Substrate was then added (diethanolamine 97 mL, 100 mg MgCl2, 0.2 g NaN3, 800 mL ddH2O, pH 9.8). After a 15-min incubation, the reaction was stopped using 0.5 M H2SO4 and the plate wells were read at 450 nm. Titer (Log2) was the highest dilution of anti-IL-10 egg yolk antibody powder that resulted in an absorbance 2× above a 1:10 dilution of control egg yolk antibody powder.
Validation of Rabbit Anti-IL-10 Binding to Chicken IL-10
Commercial rabbit anti-IL-10 antibody (Bioss) was found to cross react with chicken IL-10. In this experiment, 5 μg of chicken IL-10 was bound to Nunc F MaxiSorp plates overnight in coating buffer. One hundred microliters of a 1:500 dilution of a commercial rabbit anti-IL-10 antibody or rabbit isotype control antibody (Santa Cruz Biotechnology, Santa Cruz, CA) was used to determine if the commercial rabbit anti-IL-10 antibody bound to chicken IL-10 (Figure 1B, see ELISA methods above, except secondary antibody was goat anti-rabbit IgG HRP conjugate, Bethyl Laboratories). An additional control included the exclusion of a primary antibody.
Anti-IL-10 ELISAs
Five micrograms of commercial recombinant chicken IL-10 was bound to Nunc F MaxiSorp plates overnight in coating buffer. One hundred microliters of a 1:500 dilution of chicken affinity-purified anti-IL-10 at a concentration of 0.85 mg/mL or adjuvant control antibody was used to determine if the anti-IL-10 antibody bound the commercial source for IL-10 (see ELISA methods above).
Anti-IL-10 Capture ELISA
A capture ELISA was used to demonstrate the presence of luminal IL-10 in the intestine of Eimeria infected chicks. Duodenal lumen fluid protein (250 μg, determined by BCA assay, Pierce) collected from chicks 4 d post-mixed 10× Eimeria spp. challenge was used. Plates were coated with the commercial rabbit anti-IL-10 antibody (160 ng/mL in coating buffer, Bioss). Centrifuged luminal duodenal fluid supernates (250 μg protein) or saline was then incubated on bound rabbit anti-IL-10 antibody for 24 h at room temperature. Following a 5× wash, affinity-purified chicken anti-IL-10 antibody (anti-IL-10), control antibody (control), or saline (no secondary antibody or blank) was added at 0.85 mg/mL and incubated 24 hours, then washed 5×. Rabbit anti-chicken FC conjugated to HRP (1:10,000 dilution per manufacturer's instructions) was used as the secondary antibody and was allowed to bind for 1 h. Following a 5× wash of unbound secondary antibody, substrate was used and reaction measured as described above (ELISA section).
Dietary anti-IL-10 and Eimeria Challenge Experiments: General Procedures
Three chick experiments were conducted to determine: 1) the effectiveness of dietary anti-IL-10 in preventing reduced body weights due to Eimeria spp. challenge; 2) if a lower dose of dietary anti-IL-10 could be protective against Eimeria-induced body-weight reduction; and 3) if the use of dietary anti-IL-10 interfered with vaccination against Eimeria mixed spp. as measured by body weight post subsequent Eimeria challenge. In all experiments, day-old broiler chicks (Welp's Hatchery, Bancroft, IA) were housed 5 per pen in chick battery brooders with raised wire floors in a temperature-controlled external environment (21°C). Dietary antibody feeding began at placement (d 1) and continued until the completion of the study (d 21) in experiments 1 and 2, or until withdrawal on d 14 in experiment 3 (trials 1 and 2). The antibodies (control or anti-IL-10) were added on top of a complete diet (Sunfresh Purina, Grey Summit, MO), and each antibody was fed to 20 pens of 5 broiler chicks each. To maintain consistent levels of egg yolk powder between diets and to assure the diets were isonitrogenous and isocaloric, anti-IL-10 egg yolk antibody was simply substituted in place of control egg yolk antibody. Hence all diets fed had consistent levels of egg yolk antibody powder (i.e., 3.4 g/kg in experiment 1 and 2, and 0.34 g/kg feed in experiment 3, trials 1 and 2). Pen served as the experimental unit.
Experiment 1. Dietary anti-IL-10 and Eimeria Challenge
At 3 d of age, 10 pens of chicks fed either control or anti-IL-10 were challenged by oral gavage with 10× dose of Eimeria mixed spp. (Advent® Coccidiosis Vaccine, Lincoln, NE, 10 pens) or sterile saline (10 pens/dietary antibody treatment). Chick body weights and feed intake were determined at 21 d of age, and feed conversion was calculated (Table 1). Oocysts were enumerated 7 d post Eimeria challenge (see below).
Table 1.
Experiment 1. Effects of anti-IL-10 on performance of chicks challenged with Eimeria spp.
| Weight (Grams) | Feed Conversion1 | Oocyst/Gram Excreta | ||||
|---|---|---|---|---|---|---|
| Diet | Control | Eimeria | Control | Eimeria | Control | Eimeria |
| Control | 670a | 611b | 1.82 | 1.75 | 4,890 | 119,700 |
| Anti-IL-10 | 633a,b | 659a,b | 1.65 | 1.6 | 6,030 | 31,860 |
| SEM | 27.6 | 0.157 | 12,210 | |||
| P Values | ||||||
| Antibody | 0.78 | 0.19 | 0.06 | |||
| Eimeria | 0.42 | 0.61 | 0.008 | |||
| Antibody × Eimeria | 0.0341 | 0.94 | 0.0745 | |||
1Feed conversion was calculated by divided the feed consumption by the total cage weight n = 10.
a,bMeans with different superscript in the same column were significantly different (P < 0.05).
Experiment 2. Dose of anti-IL-10 and Eimeria spp. Challenge
In this experiment, chicks were fed control antibody (3.4 g/kg feed), anti-IL-10 antibody (3.4 g/kg feed), or 0.34 g/kg feed anti-IL10 antibody (plus 3.06 g/kg feed control antibody). At d 3, chicks were challenged with Eimeria spp. and variables were measured as described in experiment 1 (10 pens of chicks per dietary antibody treatment and Eimeria challenge; Table 2).
Table 2.
Experiment 2. Effect of different doses of anti-IL-10 antibody on the performance of chicks challenged with Eimeria spp.
| Weight (Grams) | Feed Conversion1 | Oocyst/Gram Excreta | ||||
|---|---|---|---|---|---|---|
| Diet | Control | Eimeria | Control | Eimeria | Control | Eimeria |
| Control | 679a | 607b | 1.67b | 1.79a | 2,829 | 109,324 |
| Anti-IL-10 3.4 g/kg | 670a | 650a,b | 1.76a | 1.74a | 5,211 | 27,902 |
| Anti-IL-10 0.34 g/kg | 643a,b | 649a,b | 1.72a,b | 1.72a,b | 3,706 | 47,575 |
| SEM | 30.7 | 0.022 | 15,542 | |||
| P Values | ||||||
| Antibody | 0.54 | 0.434 | 0.117 | |||
| Eimeria | 0.0279 | 0.14 | 0.001 | |||
| Antibody × Eimeria | 0.0472 | 0.01 | 0.0913 | |||
1Feed conversion was calculated by divided the feed consumption by the total cage weight n = 10.
a,bMeans with different superscript in the same column were significantly different (P < 0.05).
Experiment 3, Trials 1 and 2. Anti-IL-10 Effects on Eimeria spp. Vaccination
The third experiment was conducted to determine if the presence of anti-IL-10 in the intestine interfered with the vaccination of young chicks with a 1× dose of Eimeria spp. vaccine. If anti-IL-10 interfered with the vaccination process, a subsequent challenge with a 10× dose of the mixed Eimeria spp. vaccine should result in suppressed growth. Since it was shown in experiments 1 and 2 that anti-IL-10 protected against reduced body weight following challenge, antibody was cleared from the intestine before the 10× challenge. At d 3, 10 pens of 5 chicks on each dietary treatment (0.34 control or 0.34 anti-IL10 antibody/kg feed) were vaccinated by gavage with a 1× dose of Advent® Coccidiosis Vaccine (Lincoln, NE) and 10 pens chicks per dietary treatment received a saline gavage. Chicks were fed either control or anti-IL-10 antibodies diets until 14 d of age (11 d post vaccination), and then antibody was removed from the diet to clear the intestine of antibody for 3 d (Rochell et al., 2012). At 17 d of age, previously vaccinated chicks were challenged with a 10× dose of Advent® Coccidiosis Vaccine. Chick body weights and feed intake were determined at 21 d of age (adequate time previously found to detect reduced body weight due to Eimeria challenge, unpublished) and feed conversion was calculated (Table 3). Oocysts were enumerated 7 d post Eimeria spp. challenge.
Table 3.
Experiment 3, trials 1 and 2. Effect of anti-IL-10 antibody on the performance of Eimeria spp. vaccinated chicks challenged with Eimeria spp.
| Trial 1 | Weight (Grams) | Feed Conversion1 | Oocyst/Gram Excreta | |||
|---|---|---|---|---|---|---|
| Diet | Control | Eimeria | Control | Eimeria | Control | Eimeria |
| Control | 659a | 621b | 1.92 | 1.99 | 0 | 1,290 |
| Anti-IL-10 | 638b | 653a,b | 1.89 | 1.91 | 0 | 780 |
| SEM | 11.4 | 0.056 | 226 | |||
| P Values | ||||||
| Antibody | 0.62 | 0.38 | 0.26 | |||
| Eimeria | 0.32 | 0.45 | 0.001 | |||
| Antibody × Eimeria | 0.016 | 0.661 | 0.285 | |||
| Trial 2 | Weight (Grams) | Feed Conversion1 | Oocyst/Gram Excreta | |||
| Diet | Control | Eimeria | Control | Eimeria | Control | Eimeria |
| Control | 658 | 663 | 1.92 | 1.99 | Nd | Nd |
| Anti-IL-10 | 643 | 660 | 1.89 | 1.91 | Nd | nd |
| SEM | 4.5 | 0.065 | ||||
| P Values | ||||||
| Antibody | 0.19 | 0.45 | Nd | |||
| Eimeria | 0.32 | 0.38 | Nd | |||
| Antibody × Eimeria | 0.49 | 0.66 | Nd | |||
1Feed conversion was calculated by divided the feed consumption by the total cage weight n = 10.
a,bMeans with different superscript in the same column were significantly different (P < 0.05).
Determination of Oocyst Shedding
Each pen's oocysts were enumerated by the Modified McMaster's Method (Hodgson, 1970). The number of oocysts per gram of fecal matter in each sample (one sample per pen) was determined by mixing 0.5 g feces with 14.5 mL supersaturated sodium chloride solution. The homogeneous mixture was then pipetted onto both grids of a McMaster slide and allowed to sit 5 minutes to allow the oocysts to float to the interface between the glass and water. The oocyst count/gram was determined by [(grid 1 + grid 2) × 100] to account for the dilution. Oocyst number was determined on d 7-post coccidia challenge in experiments 1 and 2 and at d 24 in experiment 3. At necropsy, intestines and cecum were examined for Eimeria spp.-induced lesions. Few lesions were found at necropsy; hence lesions scores were not recorded.
Statistical Analysis
Body weight, feed consumption, feed conversion, and oocyst shedding data were analyzed using a two-way ANOVA (SAS Institute Inc., Cary, NC) with antibody and Eimeria as main effects (2 × 2 factorial arrangement of treatments). When interactions were significant (P < 0.05), posthoc analysis was conducted to denote treatment differences using lettered superscripts. Treatment differences for ELISA data were analyzed using a Student's t-test.
RESULTS
Anti-IL-10 Antibody Specificity and Luminal IL-10 Presence
The titer of anti-IL-10 peptide specific egg yolk powder antibody was 10 Log2 relative to control egg yolk powder antibody. In Figure 1C, the specificity of egg yolk antibody (diluted 1: 500) to chicken IL-10 peptide vlpramqt relative to the control antibody (diluted to 1: 500) was demonstrated. Chicken anti-IL-10-vlpramqt showed a 3× increase in absorbance relative to the control antibody when binding a vlpramqt-OVA conjugate. Figure 1D demonstrated the presence of IL-10 in the intestinal luminal contents of the chick duodenum. Chicks were initially treated with a 10× dose of a mixed species Eimeria vaccine, and at d 4, chicks were euthanized and duodenal contents were collected. In this capture ELISA of duodenal contents, chicks vaccinated had significantly more IL-10 protein level when bound with the chicken anti-IL-10 antibody than when bound with control antibody (Figure 1D).
Experiment 1. Dietary Anti-IL-10 and Eimeria spp. Challenge
Chicks fed control antibody and challenged with Eimeria spp. had 3-wk body weights that were reduced 8.8% when compared to unchallenged control, however body weight was not reduced due to Eimeria spp. challenge in chicks fed 3.4 g/kg anti-IL-10 egg yolk antibody powder (antibody × Eimeria interaction, P < 0.05). Antibody feeding and Eimeria spp. challenge had no effect on feed conversion. Eimeria spp. infection increased oocyst shedding (P < 0.01) and the oocyst shedding in between chicks fed anti-IL-10 egg yolk powder or control antibody yolk powder was not significantly different (antibody × Eimeria interaction P = 0.0745).
Experiment 2. Dose of Anti-IL-10 and Eimeria Challenge
A study was conducted to determine if a lower dose of anti-IL-10 egg yolk antibody powder could protect against reduced body weight in Eimeria spp. challenged chicks. As in the first experiment, chicks fed control antibody egg yolk powder and challenged with Eimeria spp. had a reduced 3-wk body weight when compared to unchallenged chicks fed control antibody egg yolk powered (10.6%), while those fed 3.4 or 0.34 g/kg diet anti-IL-10 egg yolk powder showed no reduction in 3-wk body weight when compared to unchallenged chicks (antibody × Eimeria interaction, P < 0.05). Data shows that the use of anti-IL-10 egg yolk antibody powder at 0.34 g/kg diet was as effective as the dose of 3.4 g/kg diet in preventing reduced 3-week body weights due to Eimeria spp. challenge. Oocyst shedding in Eimeria spp. challenged chicks was not significantly influenced by antibody type fed (antibody × Eimeria interaction, P = 0.09). Feed conversion was lowest in chicks fed control antibody and not exposed to Eimeria spp. challenge. All other groups showed increased feed conversion relative to this group (antibody × Eimeria interaction P = 0.01).
Experiment 3. Anti-IL-10 on Vaccinated and Challenged Chicks
Control antibody fed chicks vaccinated with a 1× dose of Eimeria spp. vaccine then challenged with a 10× dose of the vaccine had reduced 3-wk body weight when compared to chicks not exposed to Eimeria spp. (5.8%) in trial 1, but no change in body weight due to Eimeria challenge was observed in anti-IL-10-fed chicks (antibody × Eimeria interaction, P < 0.05). In trial 2, no antibody × Eimeria interaction was observed showing the effects in trial 1 were not repeatable. Eimeria spp. vaccination plus challenge increased oocysts shedding regardless of antibody fed (main effect of Eimeria, P < 0.01, trial 1). No effects of antibody or coccidia on feed conversion were observed in trials 1 or 2.
DISCUSSION
Oral application of antibodies to host proteins represents a new method for control of immunologic events in the lumen of the gastrointestinal system. Experiments have clearly demonstrated that antibodies from egg yolk retain binding activity into the ileum of mice (Bobeck et al., 2015). Using a model of Eimeria spp. infection, we demonstrate that antibody directed at a specific peptide on chicken interleukin-10 (IL-10) can be used to prevent Eimeria spp.-induced decreased body weight.
IL-10 is an anti-inflammatory cytokine through its ability to down-regulate pro-inflammatory cytokines such as TNF-α and IL-6, among others (Trifunovic et al., 2015). Interestingly, IL-10 mRNA is known to be up-regulated early (d 4 post-inoculation) in Eimeria spp. infection, and up to 9 d post-inoculation (Hong et al., 2006). It is this potentially inappropriate activation of IL-10 that allowed us to hypothesize that IL-10 is used by Eimeria spp. to escape immune detection in the intestinal tract. Evidence supporting this hypothesis is the inverse relation of Eimeria spp. infection resistance and intestinal expression of IL-10 in different stains of chickens (Rothwell et al., 2004). Citristim, which reduced oocyte shedding in Eimeria spp.-infected chicks, also reduced IL-10 mRNA in the cecal tonsils (Shanmugasundaram et al., 2013). Also, IL-10 knockout mice that were infected with Cryptosporidium parvum, a close evolutionary relative of Eimeria spp., cleared the infection twice as fast, with less oocyst shedding, than control mice (Campbell et al., 2002). Thus, induction of IL-10 release appears to be critical for Eimeria spp. infection in host animals. In this paper, we showed that feeding an anti-IL-10 peptide egg yolk antibody protected against reduced body weight caused Eimeria spp. challenge. These results suggest that inhibition of IL-10 in the lumen of the gastrointestinal tract might be a viable treatment to control the adverse effect of Eimeria spp. infection.
A question as to how Eimeria spp. can regulate the host release of IL-10 can be deduced by the studies of Jang et al. (2011) and Paark et al. (2012). Jang et al. (2011) showed that Eimeria spp. can produce a macrophage migration inhibitory factor-like protein (unique MIF-like protein per Eimeria species), that when used as a vaccine in chick embryos, resulted in protection against weight loss upon subsequent infection with Eimeria spp. expressing that specific MIF-like protein. Paark et al. (2012) has shown that helminthes also express MIF-like proteins, and that these MIF like protein up-regulate IL-10 production in mononuclear cells. If Eimeria spp. MIF increases IL-10 release from mucosa cells as a means of evading immune detection, then the oral anti-IL-10 antibody may serve as a disruptor of this evasion strategy. Targeting either Eimeria spp. MIF or host IL-10 should demonstrate the same endpoint; resistance to Eimeria spp. infection. Targeting host IL-10 may have an advantage over targeting Eimeria spp. MIF, since each Eimeria species produces a different MIF-like protein (Miska et al., 2007).
Our initial experiments to determine if IL-10 was even present in the lumen of the gastrointestinal tract started with a paper published by Hong et al. (2006), who demonstrated a significant increase in IL-10 mRNA expression in intestinal intraepithelial lymphocytes at d 5 post Eimeria spp. infection (Hong et al., 2006). First, to determine if our antibody was specific to IL-10 from chickens, we bound our anti-IL-10 antibody to chicken IL-10 (Figure 1C). Second, to determine if IL-10 was present in the lumen of the intestine, duodenal luminal contents from chickens challenged with Eimeria spp. were used in a capture ELISA to determine if IL-10 was present. It was demonstrated in Figure 1D that there was IL-10 present in the lumen of the duodenum and the antibody made to IL-10 bound this IL-10. These results indicate that 1) IL-10 is present in the lumen of the gastrointestinal tract, and 2) the antibody we made to IL-10 binds to chicken IL-10.
In the initial feeding study anti-IL-10 was fed at a high rate of 3.4 g/kg of chicken feed. The experiment demonstrated that anti-IL-10 protected against reduced body weight gain in Eimeria spp. infected chicks, however 3.4 g egg yolk antibody/kg diet of anti-IL-10 would not be cost effective for commercial use (experiment 1). The second study, which found that a dose one-tenth the level used in the first experiment was also protective, suggested that the product might have commercial application (experiment 2). Subsequent to this study, we have found that the specific anti-IL-10 antibody content in egg yolk powder is 0.4 mg/g. Based on the molecular weight of IgY and IL-10, and considering the two binding domains on each IgY molecule, 0.4 mg of IgY should be capable of neutralizing up to 80 μg of IL-10. Quantitative analysis of total IL-10 secreted into the intestine should provide a useful estimate of the minimal amount of anti-IL-10 antibody needed in Eimeria spp. challenged chicks.
This last experiment we attempted to determine if anti-IL-10 antibodies interfered with the adaptive immune process. If anti-IL-10 interfered with immunity during vaccination with the 1× dose of vaccine, chicks fed anti-IL-10 should have shown reduced weights on challenge as compared to those fed control antibody. The finding that the body weights of chicks fed the antibody to IL-10 were not adversely affected during challenge even after removal of the anti-IL-10 antibody suggested that anti-IL-10 did not affect adaptive immune function. The finding of decreased body weight following Eimeria spp. challenge in vaccinated chicks fed control antibody (but not in anti-IL-10-fed chicks) in trial 1 is consistent with the literature. Danforth et al. (1997) found in a model similar to the one used in this paper that vaccination with Eimeria mixed spp. (different than the one used in this study) did not protect against weight loss following a high-dose challenge the same vaccine strains. While this study was not designed to test the immune-potentiating effects of feeding anti-IL-10 during an oral vaccination, the improved protection of anti-IL-10 plus vaccine relative to the vaccine alone in trial 1 suggested that anti-IL-10 may have immune-potentiating benefit. For example, anti-IL-10 might be useful in assisting the chick in developing early immunity to select microbes (e.g., Eimeria spp.) that are present within the environment where the chicks are first placed in commercial production facilities. This would eliminate the need for feeding anti-IL-10 throughout the life of the chicken, and may allow the chicken to develop immunity to any and all strains of Eimeria spp. present in the chicken's environment. Better-designed studies could test this hypothesis.
Surprisingly, in these 3 studies there was no significant difference in oocyst shedding between anti-IL-10 and control antibody fed chicks. The P values for the interaction in experiments 1 and 2 would suggest that more power might be needed if oocyst shedding was a desirable endpoint. Oocysts shedding in the non-challenge chicks in experiments 1 and 2 suggest a low level of exposure independent of direct gavage. The level of exposure was not enough to show differential body weight between those fed control or anti-IL-10 antibody and may be in the range that others classify as “too low to count” (Danforth et al., 1997).
In conclusion, using the model described, dietary anti-IL-10 egg yolk antibodies prevented a reduction in chick body weight due to Eimeria spp. challenge. Anti-IL-10 antibodies do not appear to adversely affect adaptive immunity to an Eimeria spp. vaccination and anti-IL-10 antibodies have a minimal effect, if any, on oocyst shedding in Eimeria infected chicks. Findings presented offer a novel host-targeted procedure to improve chick growth when challenged with Eimeria spp.
Acknowledgments
The authors thank Maria Jose, Jason Ren, Jake Olson, Zachary Simons, Christina Stevenson, Dylan Easley, Megan Mezera and Christopher Nguyen for aiding in sample collection and analysis, as well as the University of Wisconsin Animal Care Staff (Dawn Irish and John Kemper) for animal care. Laying hens used for making antibodies used in these studies were kindly donated by S&R Farms, Whitewater, WI.
FUNDING
Research was supported in part by Wisconsin Alumni Research Foundation (WARF) Accelerator Grant, royalties received from WARF and University of Wisconsin School of Veterinary Medicine 12 Month Mentored Research Program, and the College of Agricultural and Life Sciences.
DISCLOSURES
MEC and JMS have an ownership interest in AbE Discovery, LLC, which has licensed technology reported in this publication. All of the remaining authors declare no financial conflict of interest.
REFERENCES
- Bobeck E. A., Hellestad E. M., Sand J. M., Piccione M. L., Bishop J. W., Helvig C., Petkovich M., Cook M. E. Oral peptide specific egg antibody to intestinal sodium-dependent phosphate co-transporter-2b is effective at altering phosphate transport in vitro and in vivo. Poult. Sci. 2015;94:1128–1137. doi: 10.3382/ps/pev085. [DOI] [PubMed] [Google Scholar]
- Campbell L. D., Stewart J. N., Mead J. R. Susceptibility to Cryptosporidium parvum infections in cytokine- and chemokine-receptor knockout mice. J. Parasitol. 2002;88:1014–1016. doi: 10.1645/0022-3395(2002)088[1014:STCPII]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Cook M. E., Trott D. L. IgY - Immune component of eggs as a source of passive immunity for animals and humans. World's Poult Sci. J. 2010;66:215–225. [Google Scholar]
- Cyktor J.C., Turner J. Interleukin-10 and immunity against prokaryotic and eukaryotic interacellular pathogens. Infect. Immun. 2011;79:2964–2973. doi: 10.1128/IAI.00047-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danforth H. D., Lee E. H., Martin A., Dekich M. Evaluation of a gel-immunization technique with two different immunocox vaccine formulations in battery and floor-pen trials with broiler chickens. Parasitol. Res. 1997;83:445–452. doi: 10.1007/s004360050278. [DOI] [PubMed] [Google Scholar]
- Hodgson J. N. Coccidiosis: Oocyst counting technique for coccidiostat evaluation. Exp. Parasit. 1970;28:99–102. doi: 10.1016/0014-4894(70)90073-1. [DOI] [PubMed] [Google Scholar]
- Hong Y. H., Lillehoj H. S., Lee S. H., Dalloul R. A., Lillehoj E. P. Analysis of chicken cytokine and chemokine gene expression following Eimeria acervulina and Eimeria tenella infections. Vet. Immunol. Immunopathol. 2006;114:209–223. doi: 10.1016/j.vetimm.2006.07.007. [DOI] [PubMed] [Google Scholar]
- Jang S. I., Lillehoj H. S., Lee S. H., Kim D. K., Pages M., Hong Y. H., Min W., Lillehoj E. P. Distinct immunoregulatory properties of macrophage migration inhibitory factors encoded by Eimeria parasites and their chicken host. Vaccine. 2011;29:8998–9004. doi: 10.1016/j.vaccine.2011.09.038. [DOI] [PubMed] [Google Scholar]
- Kominsky D. J., Campbell E. L., Ehrentraut S. F., Wilson K. E., Kelly C. J., Glover L. E., Collins C. B., Bayless A. J., Saeedi B., Dobrinskikh E., Bowers B. E., MacManus C. F., Muller W., Colgan S. P., Bruder D. IFN-gamma-mediated induction of an apical IL-10 receptor on polarized intestinal epithelia. J. Immunol. 2014;192:1267–1276. doi: 10.4049/jimmunol.1301757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konnikova Y., Zaman M. M., Makda M., D'Onofrio D. D., Freedman S. D., Martin C. R. Late enteral feedings are associated with intestinal inflammation and adverse neonatal outcomes. PLoS One. 10:e0132924. doi: 10.1371/journal.pone.0132924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miska K. B., Fetterer R. H., Lillehoj H. S., Jenkins M. C., Allen P. C., Harper S. B. Characterisation of macrophage migration inhibitory factor from Eimeria species infectious to chickens. Mol. Biochem. Parasitol. 2007;151:173–183. doi: 10.1016/j.molbiopara.2006.10.020. [DOI] [PubMed] [Google Scholar]
- Paark H. K., Cho M. K., Park H. Y., Kim K. U., Kim Y. S., Lee M. K., Park S. K., Kim D. H., Yu H. S. Macrophage migration inhibitory factor isolated from a parasite inhibited Th2 cytokine production in PBMCs of atopic asthma patients. J. Asthma. 2012;49:10–15. doi: 10.3109/02770903.2011.637593. [DOI] [PubMed] [Google Scholar]
- Rochell S. J., Applegate T. J., Kim E. J., Dozier W. A., III Effects of diet type and ingredient composition on rate of passage and apparent ileal amino acid digestibility in broiler chicks. Poult. Sci. 2012;91:1647–1653. doi: 10.3382/ps.2012-02173. [DOI] [PubMed] [Google Scholar]
- Rothwell L., Young J. R., Zoorob R., Whittaker C. A., Hesketh P., Archer A., Smith A. L., Kaiser P. Cloning and characterization of chicken IL-10 and its role in the immune response to Eimeria maxima. J. Immunol. 2004;173:2675–2682. doi: 10.4049/jimmunol.173.4.2675. [DOI] [PubMed] [Google Scholar]
- Shanmugasundaram R., Sifri M., Selvaraj R. Effect of yeast cell product (Citristim) supplementation on broiler performance and intestinal immune call parameters during an experimental coccidial infection. Poult. Sci. 2013;92:358–363. doi: 10.3382/ps.2012-02776. [DOI] [PubMed] [Google Scholar]
- Trifunovic J., Miller L., Debeljak Z., Horvat V. Pathologic patterns of interleukin 10 expression–a review. Biochem. Med. (Zagreb) 2015;25:36–48. doi: 10.11613/BM.2015.004. [DOI] [PMC free article] [PubMed] [Google Scholar]