Abstract
Purpose
Vitrectomy is usually only indicated for familial exudative vitreoretinopathy (FEVR) cases with progressive retinal folds or macular dragging. In this report, we present our experience reversing the progression of macular dragging by performing early eye vitrectomies in a 1-month-old male baby with FEVR.
Observations
A 7-day-old, full-term male baby was examined by a pediatric ophthalmologist. His sister had a laser ablation treatment after being diagnosed with FEVR. The ophthalmologist found the baby had avascular retinas, fibrovascular membranes, and vitreous hemorrhages in both eyes, and performed retinal photocoagulations the next day. Although the retinal folds had not yet formed, the arcade vessels began to linearize after the procedure, strongly suggesting disease progression. Therefore, we performed lens-sparing vitrectomies in both eyes on the twenty-ninth day of life. After surgery, the macular dragging reversed, as evidenced by vascular arcade angle measurements. Three years after the surgery, the boy's visual acuity was 0.4 in both eyes.
Conclusions and Importance
In this case, we believe the good postoperative outcomes were due to early vitrectomies before the vitreoretinal traction became severe. In addition, the retinal photocoagulation performed on the eighth day of life may have reduced disease activity, at least partially. This case highlights the importance of prompt diagnosis and appropriate treatment of FEVR.
Keywords: Familial exudative vitreoretinopathy, Vitrectomy, Early treatment, Laser photocoagulation, Retinal fold
1. Introduction
Familial exudative vitreoretinopathy (FEVR) is a hereditary retinal vitreous abnormality caused by failed angiogenesis. Due to retinal blood vessel malformations, this disease can resemble retinopathy of prematurity (ROP), including as retinal vascular abnormalities in the periphery, avascular areas, new blood vessel formation, proliferative membrane formation, and tractional detachment (TRD). Criswick et al. first reported familial retinal vitreous disease exhibiting ROP-like retinopathy in full-term infants in 1969.1 Since then, many studies have investigated the underlying genetic abnormalities and sought to define a staging classification system for FEVR.2, 3 Although sporadic cases have been reported, most cases of FEVR are inherited, due primarily to genetic abnormalities in the Wnt signaling pathway.4, 5, 6, 7, 8, 9, 10, 11 Cases lacking a family history are discovered when a white pupil, strabismus, or visual disturbances are found. Some cases with FEVR progress without effective therapeutic interventions. The point at which retinal folds and TRD are formed is unclear. Depending on the case, the disease image may be completed before birth, with the disease progressing after birth. Herein, we report a case of FEVR with a good outcome after prompt diagnosis and appropriate surgical intervention.
2. Case report
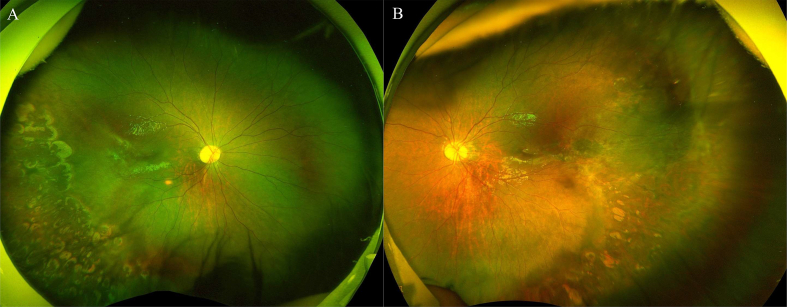
A 7-day-old male baby, born at 36 weeks gestation with a birth weight of 2872 g, was examined by a pediatric ophthalmologist (M.I.) because the patient's sister had a history of FEVR in infancy that had required retinal photocoagulation. The ophthalmologist found our patient had extensive avascular retinas, vitreous hemorrhages, and fibrovascular membranes on the temporal side of the retina in both eyes (Fig. 1A and B), and he performed retinal photocoagulations the next day (Fig. 1C and D). Since a state of high activity persisted after the procedure, the patient was subsequently admitted to our hospital for vitrectomy. Upon our initial examination, we found significant vitreoretinal macular tractions, some linearized arcade vessels, and narrow angles between the arcade vessels and centering on the optic disks. We measured the angles between the temporal inferior and the temporal superior venous arcades and the optic disks using digitized fundi photographs and the Image J system (NIH) software (Fig. 1A and H). Although the retinal folds were not yet formed, the macular anatomy seemed worse, in spite of the photocoagulation.
Fig. 1.
Fundus photographs of the right eye (A) and the left eye (B) at day 7 after birth. An extensive avascular retina, a vitreous hemorrhage, and a fibrovascular membrane on the temporal side of retina are seen. Fundus photograph of the right eye (C) and the left eye (D) at day 8 after birth. The retinal photocoagulation is evident. Fundus photograph of the right eye (E) and the left eye (F) at day 23 after birth. At this point, arcade vessels had begun to linearize, and the angle between the arcade vessels and the centering on the optic disk had narrowed (right eye, 86.7°; left eye, 81.4°). A shallow tractional retinal detachment around the fibrovascular membrane is noted in the right eye. No apparent retinal detachment is seen in the left eye. Fundus photograph of the right eye (G) and the left eye (H) at day 52 after birth. The angle between the arcade blood vessel and the center of the optic disk had widened, and the macular traction had decreased remarkably (right eye, 98.5°; left eye, 98.3°).
We performed bilateral lens-sparing vitrectomies using 3-port, 25-gauge systems 29 days after birth. During the surgery, we dissected the traction between the lenses and the fibrovascular proliferations using vitrectomy probes. After separation of the posterior vitreous, we performed membrane dissections using vitrectomy probes and vertical scissors, followed by laser ablations of the peripheral avascular retinas. During surgery, we noted a shallow TRD around the fibrovascular membrane in his right eye. After the vitrectomies, the angles between the arcade blood vessels and the optic disk centers widened, and the macular traction decreased remarkably (Fig. 1G and H).
Genetic testing revealed a mutation in the FZD4 gene. In particular, a nucleotide substitution at position 313 from adenine to guanine (c.313A > G) resulted in an amino acid change at position 105 from methionine to valine (p.M105V). At 3 years old, the boy's visual acuity was 0.4 in both eyes. Ultra-widefield fundus photographs at that time showed stable conditions with adequate macular reflexes (Fig. 2).
Fig. 2.
Ultra-wide filed fundus images of the right (A) and the left (B) eyes at 3 years post-operation showing stable conditions with macular reflex.
3. Discussion
Since Criswick et al. reported inherited retinal vitreous diseases characterized by ROP-like retinopathy in 1969, many cases of FEVR have been reported.1 In 1971, Gow et al.2 proposed a FEVR staging system, which was modified by Laqua in 1980.3 Since then, several different staging and diagnostic criteria have been proposed, but the most common staging system in use was proposed by Pendergast and Trese.12 These authors also reported vitrectomies in patients as young as 3 months with stage 3B FEVR.
In a series of 31 eyes of 22 patients, Yamane and associates performed surgeries in patients from 1 month to 216 months old, with retinal conditions ranging from retinal folds to total retinal detachments.13 Fei and associates reported the surgical results of lensectomies and vitrectomies in 34 eyes of 25 patients with TRD whose average age was 3.5 ± 5.9 years, and whose final visual acuities ranged from no light perception to 30/200 (n = 17).14
Thus, most reports of surgically treated FEVR cases seem to involve patients treated after the development of retinal folds and/or TRDs after several months of age or older. In contrast, the age of our case at the time of vitrectomy was 1 month. Although freshly formed retinal folds can be unfolded to obtain relatively good visual function preserved by vitrectomy in eyes with Norrie disease (Shima et al.),15 we believe that early vitrectomy i.e., before macular dragging occurs, results in better anatomical and functional results, as seen in our case. Vitrectomy may be more successful at restoring macular anatomy if the traction elements are released before the retinal fold becomes dry.
Careful consideration should be given to the risks and benefits of vitrectomy, and also to the natural course of FEVR. It is believed that FEVR begins to develop during the fetal stages, but can progress after birth and lead to retinal detachment in young people12,16. A key pathological aspect of FEVR is the presence of peripheral retinal avascularity, and fibrosis at the junction between the vascular and avascular retina, which may lead to macular dragging and TRD.17 Our case had wide avascular retinas, fibrosis, and vitreous hemorrhages in both eyes at 7 days of age. The macular dragging seemed to be progressive, and we confirmed this by measuring the vascular arcade angles, even after laser ablation therapy. The cause of the macular dragging was likely the contraction of the fibrotic component after laser ablation, as the vascular activity related to the retinopathy seemed to be reduced, judging from the improvement of posterior vessels dilatation. Retinal folds and TRDs were very likely in our patient, who would probably have developed severe visual function abnormalities if not treated with vitrectomy.
Early diagnosis of FEVR is often difficult. Many patients, on their first visits to eye clinics, show leukocoria and/or strabismus with poor visual acuity. In these cases, the disease may have progressed to the point where patients will have a poor visual prognosis regardless of treatment. Fortunately, early diagnosis was possible in our case because the elder sister had a history of laser ablation therapy for FEVR, and the mother had been instructed by a pediatric ophthalmologist (M.I.) to bring the new baby soon after the delivery. Consequently, early diagnosis 7 days after birth, and early laser ablation at 8 days were achieved. Thus, it is important to screen all family members for early diagnosis and treatment of FEVR.
4. Conclusion
Progressive macular dragging can be reversed by early vitrectomy, and achieved good anatomical and functional results. This case highlights the importance of prompt diagnosis and appropriate treatment of FEVR.
Patient consent
The patient and his parents provided written informed consent for the report.
Funding
SK, KK Grant Support: This study was supported in part by grant-in-aid 17K11440 from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Conflicts of interest
The following authors have no financial disclosures: AI, SK, MI, HK, KK.
Authorship
All authors attest that they meet the current ICMJE criteria for Authorship.
Acknowledgments
None.
References
- 1.Criswick V.G., Schepens C.L. Familial exudative vitreoretinopathy. Am J Ophthalmol. 1969;68(4):578–594. doi: 10.1016/0002-9394(69)91237-9. [DOI] [PubMed] [Google Scholar]
- 2.Gow J., Oliver G.L. Familial exudative vitreoretinopathy. An expanded view. Arch Ophthalmol. 1971;86(2):150–155. doi: 10.1001/archopht.1971.01000010152007. [DOI] [PubMed] [Google Scholar]
- 3.Laqua H. Familial exudative vitreoretinopathy. Arch Von Graefes Arch Klin Exp Ophthalmol. 1980;213(2):121–133. doi: 10.1007/BF00413539. [DOI] [PubMed] [Google Scholar]
- 4.Chen Z.Y., Battinelli E.M., Fielder A. A mutation in the Norrie disease gene (NDP) associated with X-linked familial exudative vitreoretinopathy. Nat Genet. 1993;5(2):180–183. doi: 10.1038/ng1093-180. [DOI] [PubMed] [Google Scholar]
- 5.Robitaille J., MacDonald M.L., Kaykas A. Mutant frizzled-4 disrupts retinal angiogenesis in familial exudative vitreoretinopathy. Nat Genet. 2002;32(2):326–330. doi: 10.1038/ng957. [DOI] [PubMed] [Google Scholar]
- 6.Toomes C., Bottomley H.M., Jackson R.M. Mutations in LRP5 or FZD4 underlie the common familial exudative vitreoretinopathy locus on chromosome 11q. Am J Hum Genet. 2004;74(4):721–730. doi: 10.1086/383202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kondo H., Uchio E., Kusaka S., Higasa K. Risk allele of the FZD4 gene for familial exudative vitreoretinopathy. Ophthalmic Genet. 2018;39(3):405–406. doi: 10.1080/13816810.2017.1401090. [DOI] [PubMed] [Google Scholar]
- 8.Poulter J.A., Ali M., Gilmour D.F. Mutations in TSPAN12 cause autosomal-dominant familial exudative vitreoretinopathy. Am J Hum Genet. 2010;86(2):248–253. doi: 10.1016/j.ajhg.2010.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kondo H., Kusaka S., Yoshinaga A. Mutations in the TSPAN12 gene in Japanese patients with familial exudative vitreoretinopathy. Am J Ophthalmol. 2011;151(6):1095–1100. doi: 10.1016/j.ajo.2010.11.026. e1. [DOI] [PubMed] [Google Scholar]
- 10.Ye X., Wang Y., Cahill H. Norrin, frizzled-4, and Lrp5 signaling in endothelial cells controls a genetic program for retinal vascularization. Cell. 2009;139(2):285–298. doi: 10.1016/j.cell.2009.07.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu J.H., Liu J.H., Ko Y.C. Haploinsufficiency of RCBTB1 is associated with Coats disease and familial exudative vitreoretinopathy. Hum Mol Genet. 2016 15;25(8):1637–1647. doi: 10.1093/hmg/ddw041. [DOI] [PubMed] [Google Scholar]
- 12.Pendergast S.D., Trese M.T. Familial exudative vitreoretinopathy. Results of surgical management. Ophthalmology. 1998;105(6):1015–1023. doi: 10.1016/S0161-6420(98)96002-X. [DOI] [PubMed] [Google Scholar]
- 13.Yamane T., Yokoi T., Nakayama Y., Nishina S., Azuma N. Surgical outcomes of progressive tractional retinal detachment associated with familial exudative vitreoretinopathy. Am J Ophthalmol. 2014;158(5):1049–1055. doi: 10.1016/j.ajo.2014.08.009. [DOI] [PubMed] [Google Scholar]
- 14.Fei P., Yang W., Zhang Q., Jin H., Li J., Zhao P. Surgical management of advanced familial exudative vitreoretinopathy with complications. Retina. 2016;36(8):1480–1485. doi: 10.1097/IAE.0000000000000961. [DOI] [PubMed] [Google Scholar]
- 15.Shima C., Kusaka S., Kondo H., Hasebe H., Fujikado T., Tano Y. Lens-sparing vitrectomy effective for reattachment of newly developed falciform retinal detachment in a patient with Norrie disease. Arch Ophthalmol. 2009;127(4):579–580. doi: 10.1001/archophthalmol.2009.35. [DOI] [PubMed] [Google Scholar]
- 16.Miyakubo H., Hashimoto K., Miyakubo S. Retinal vascular pattern in familial exudative vitreoretinopathy. Ophthalmology. 1984;91(12):1524–1530. doi: 10.1016/s0161-6420(84)34119-7. [DOI] [PubMed] [Google Scholar]
- 17.Tauqeer Z., Yonekawa Y. Familial exudative vitreoretinopathy: pathophysiology, diagnosis, and management. Asia Pac J Ophthalmol (Phila) 2018;7(3):176–182. doi: 10.22608/APO.201855. [DOI] [PubMed] [Google Scholar]