Key Points
Microenvironmental stimuli affect EZH2 expression and function in CLL.
Combined B-cell signaling and EZH2 inhibition showed synergistic effects on primary CLL cells.
Introduction
Enhancer of zeste homolog 2 (EZH2) is the catalytic subunit of the polycomb repressive complex 2 (PRC2), which induces gene repression through trimethylation of histone H3 at lysine 27 (H3K27me3).1 Evidence suggests that EZH2 overexpression is associated with clinical aggressiveness in chronic lymphocytic leukemia (CLL), conferring a survival advantage to the malignant cells.2-4 Interestingly, ex vivo inhibition of EZH2 enzymatic activity in primary CLL cells by drugs5,6 induced downregulation of H3K27me3 levels, leading to increased cell apoptosis.2
Microenvironmental interactions of CLL cells with bystander cells mediated by the B-cell receptor (BcR), Toll-like receptors (TLRs), and CD40, among others,7,8 are also critical for CLL development and progression.9 This scenario is reinforced by the remarkable therapeutic efficacy of the Bruton tyrosine kinase inhibitor ibrutinib and the phosphatidylinositol 3-kinase δ inhibitor idelalisib in CLL.10-12 However, despite their overall high efficacy, these drugs only rarely induce complete clinical responses when used as monotherapy, underscoring the existence of additional relevant processes that may underlie resistance and suboptimal outcomes.13-16 Considering the significant role of external triggering in promoting CLL cell survival and proliferation,17 and the emerging role of EZH2 in these processes,18 the current article explored potential links between microenvironmental stimuli and EZH2 expression and whether synergism may exist between EZH2 and signaling inhibitors ex vivo in primary CLL cells.
Methods
Blood samples were collected under informed consent from patients diagnosed with CLL according to the International Workshop on Chronic Lymphocytic Leukemia/National Cancer Institute guidelines.19 The study was approved by the local ethics committee of CERTH (decision on 18 August 2014) and conducted in accordance with the Declaration of Helsinki.
Clinicobiological data for the patient cohort are given in supplemental Table 1. CD19+ B cells were negatively selected from whole blood and cultured in the presence or absence of specific ligands for certain time points, depending on the assay. Quantification of EZH2 messenger RNA (mRNA) levels was achieved by real-time quantitative polymerase chain reaction. Protein expression of p-PLCγ2, EZH2, and H3K27me3 was assessed by using western blotting and/or flow cytometry. Cell viability, Bcl-2, Bcl-xl, Mcl-1, cleaved poly-ADP ribose polymerase (PARP), and cleaved caspase-3 were assessed by using flow cytometry. Purified CLL cells were cultured in the presence of the EZH2 inhibitors GSK126 and GSK343, and/or ibrutinib, idelalisib, and venetoclax, and the combination index, cell viability, and H3K27me3 levels were measured. Coculture system experiments of purified CLL cells with stromal HS-5 cells were performed in the presence of GSK126. Detailed information about the methodology is provided in the supplemental Methods.
Results and discussion
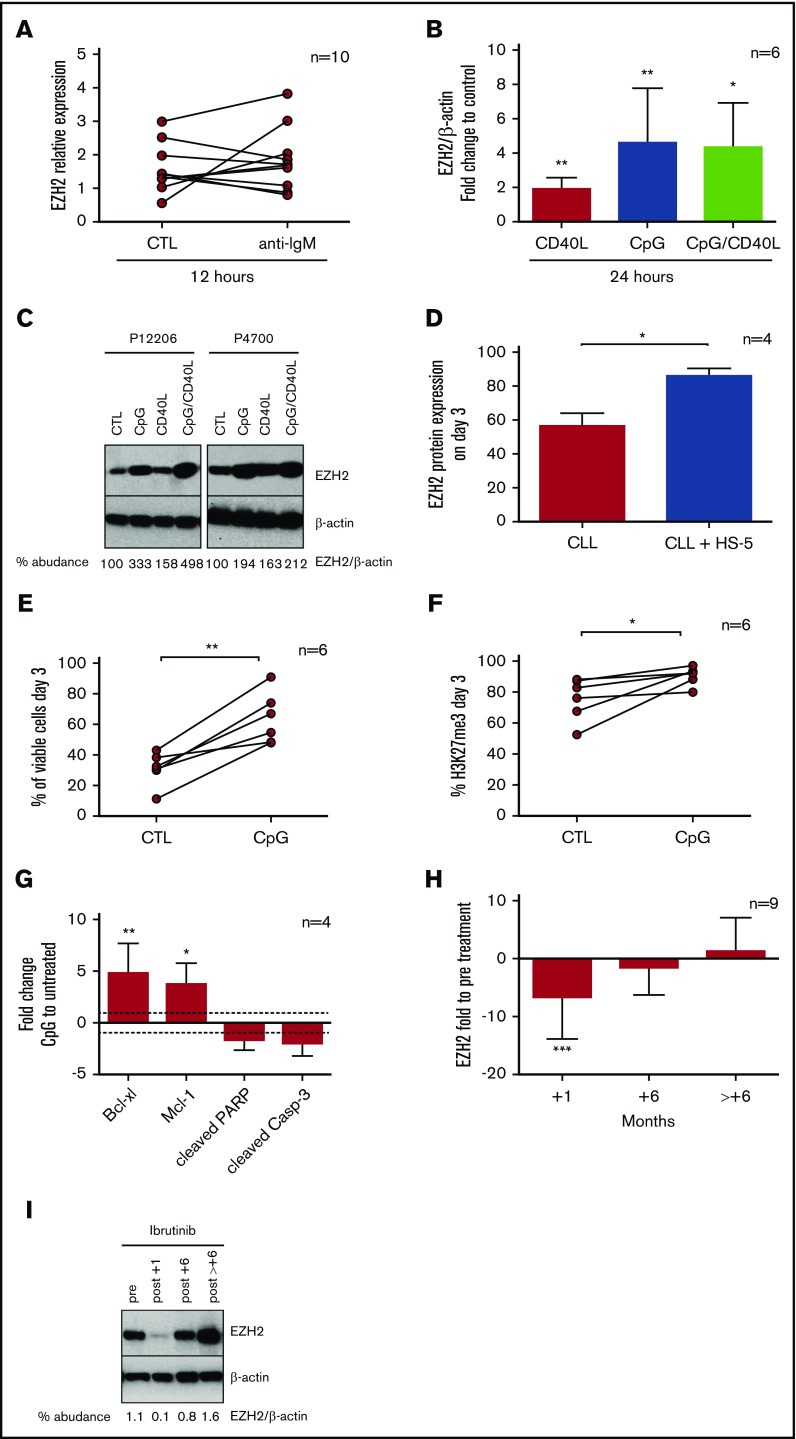
To explore the impact of BcR stimulation on EZH2 expression, we stimulated CD19+ B cells from 10 selected BcR-responsive CLL cases (supplemental Figure 1A-D) with anti–immunoglobulin M for 12 hours (supplemental Figure 1E) and observed that EZH2 mRNA expression was variably affected (Figure 1A). We next investigated the potential impact of other microenvironmental signals and found that in CLL cases responsive to TLR9/CD40 activation (supplemental Figure 1F), EZH2 mRNA expression did not change significantly (fold change [FC] = 1.2) with soluble CD40L (CD40L), whereas TLR9 stimulation with cytosine-phosphorothioate-guanine oligodeoxynucleotides (CpG) or costimulation with CpG/CD40L caused pronounced upregulation of EZH2 mRNA levels (FC = 8.9 [P < .05] and FC = 10.5 [P < .05], respectively) (supplemental Figure 1G). Similar results were obtained at the protein level in cells treated with CD40L (FC = 1.9; P < .01), CpG (FC = 4.6; P < .01), and CpG/CD40L (FC = 4.3; P < .05); the strongest effect was observed after TLR9 stimulation (Figure 1B-C). Moreover, coculture of CLL cells with the HS-5 stromal cell line for 3 days induced EZH2 protein expression (FC = 1.5; P < .05) (Figure 1D). Overall, these findings support the theory that EZH2 expression can be regulated by signals emanating from the tumor milieu. This outcome is in line with observations showing that EZH2 expression is significantly upregulated in the proliferation centers of CLL/SLL lymph nodes18 in which malignant cells receive multiple signals from bystander cells.17
Figure 1.
External stimuli and ibrutinib treatment modulate EZH2 expression. (A) EZH2 mRNA analysis using real-time quantitative polymerase chain reaction of 10 CLL cases after BcR stimulation for 12 hours. In 5 of 10 cases, EZH2 was upregulated (FC = 1.7; P < .05), whereas the remaining were downregulated (FC = 1.4; P < .01). In the graph, 2 connected points represent EZH2 relative expression in 2 different conditions for each patient. (B) Each bar in the graph shows the mean values of the FC of EZH2 protein expression (as analyzed by using western blotting) in cells stimulated with CD40L and/or CpG for 12 hours, normalized to unstimulated control cells. Asterisks indicate significant differences compared with the unstimulated control. (C) Immunoblotting analysis of EZH2 protein expression and β-actin of 2 representative cases. (D) Each bar in the graph shows the mean values with standard deviation of EZH2 expression in CLL cells alone or CLL cells cocultured with HS-5 cells for 3 days (n = 4, FC = 1.5; P < .05). CLL cell viability analysis (E) and H3K27me3 levels (F) at day 3 after TLR9 stimulation, using flow cytometry. In the graph, connected points represent the percentage of viable cells or H3K27me3 levels in 2 conditions: unstimulated control (CTL) and the cells stimulated with CpG (n = 6; FC = 2.1, P < .01; FC = 1.2, P < .05). (G) Each bar in the graph shows the mean values of FC of Bcl-xl, Mcl-1, cleaved PARP, and cleaved caspase-3 of stimulated CLL cells with CpG for 3 days, compared with unstimulated control cells. Asterisks indicate significant differences compared with the unstimulated control (n = 4, FC = 4.8, P < .01; FC = 3.7, P < .05; FC = –1.7 and FC = –2, respectively). (H) CD19+ cells from patients before treatment initiation with ibrutinib and at 1, 6, and >6 months under treatment were lysed for western blotting analysis of EZH2 protein expression. Each bar in the graph represents the median values with range of EZH2/β-actin protein expression normalized to the pretreatment sample (n = 9, FC = 4.3, P < .001). (I) Immunoblotting analysis of EZH2/β-actin levels for 1 representative case. *P < .05, **P < .01, ***P < .001. IgM, immunoglobulin M.
Focusing on TLR9, we observed that TLR9-induced EZH2 upregulation was accompanied by a significant increase in H3K27me3 levels (FC = 1.2; P < .05), cell viability (FC = 2.1; P < .01), and the expression of the antiapoptotic Bcl-xl (FC = 4.8; P < .01) and Mcl-1 (FC = 3.7; P < .05), along with a concomitant decrease in the levels of the pro-apoptotic cleaved PARP (FC = –1.7) and cleaved caspase-3 (FC = –2) (Figure 1E-G). Moreover, coculture of CLL cells with HS-5 cells induced CLL viability (FC= 1.6; P < .05) and H3K27me3 levels (FC = 1.8; P < .05) (supplemental Figure 2A-D). Hence, EZH2 seems to be implicated in the microenvironmentally induced antiapoptotic response in CLL that involves regulation of apoptotic regulators, in line with the literature.20,21
We then studied the effects of ibrutinib on EZH2 protein expression in longitudinal CLL samples from patients under treatment and found significantly reduced EZH2 levels (FC = 3; P < .001) at 1 month; however, at later time points, residual CLL cells expressed EZH2 at levels similar to baseline (Figure 1H-I; supplemental Figure 3A), likely reflecting the fact that they retain the ability to proliferate, as already shown by us and others.2,18 Relative to this, it was recently shown that during ibrutinib therapy, TLR can cooperate with BcR signaling to enhance CLL proliferation and survival, particularly in microenvironmental niches.22 On these grounds, we argue that targeting both immune signaling and EZH2 might prove a potentially beneficial treatment strategy for CLL.
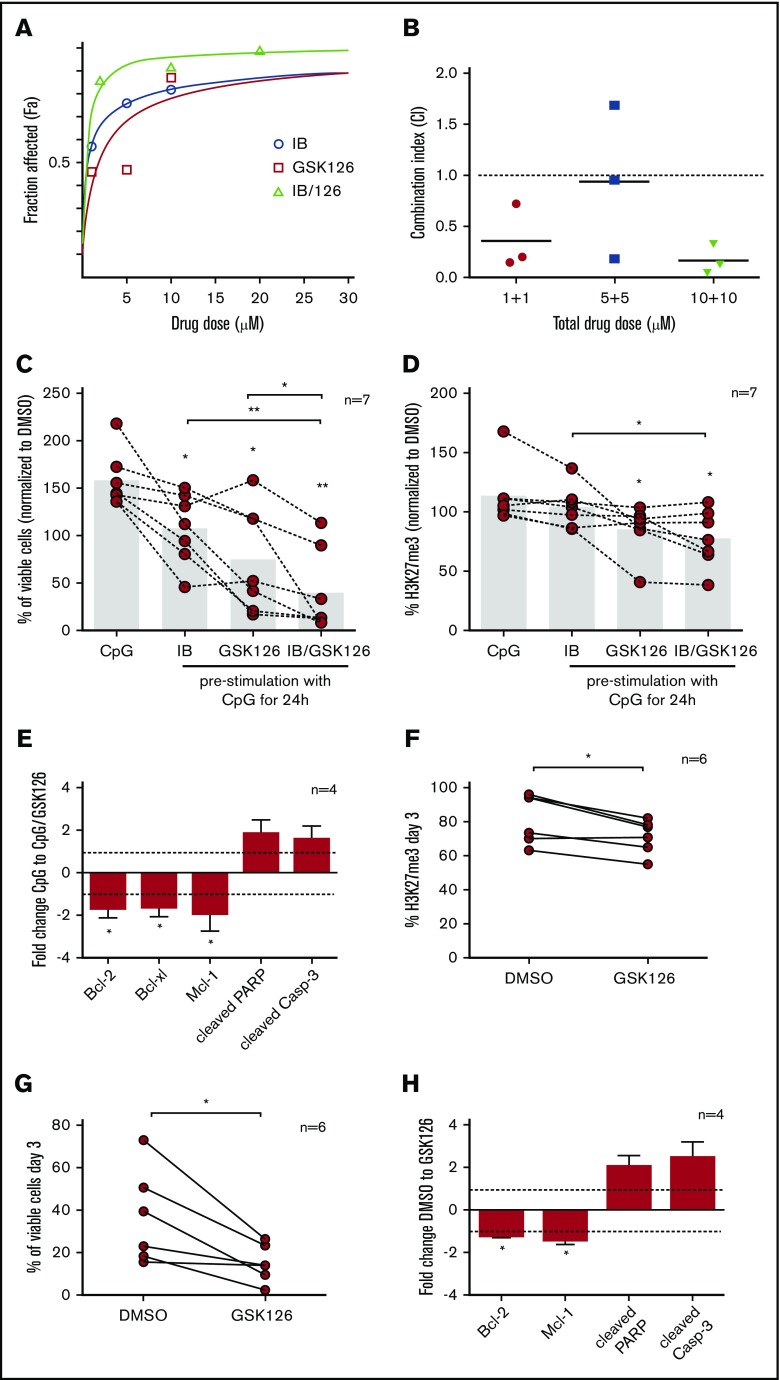
To explore potential synergism of signaling and EZH2 inhibitors, cells were pre-stimulated through TLR9 for 24 hours ex vivo and then exposed to single or combined treatment with ibrutinib + GSK126 (supplemental Figures 3 and 4), revealing synergistic effects (Figure 2A-B). We then explored synergistic effects in an extended group of patients and found that treatment of TLR9-stimulated CLL cells with ibrutinib + GSK126 induced a significant decrease in leukemic cell viability compared with isolated inhibition (FC < 1.2 and P < .05 for all comparisons); concordant results were obtained regarding H3K27me3 levels (Figure 2C-D). This finding was also accompanied by a significant reduction in the antiapoptotic proteins Bcl-2, Bcl-xl, and Mcl-1 (FC = –1.7, FC = –1.6, FC = –1.9; P < .05) and an increase in the levels of the apoptosis mediators, cleaved PARP and cleaved caspase-3 (FC = 1.8, FC = 1.6) (Figure 2E).
Figure 2.
Synergistic effects of ibrutinib (IB) plus GSK126 in primary CLL cells. (A) Negatively selected CD19+ cells from 3 CLL cases were assessed for cell viability after prestimulation with CpG for 24 hours, followed by single IB or GSK126 treatment (1, 5, and 10 μM) or combined treatment with these drugs in a 1:1 ratio at the aforementioned doses for a total of 72 hours. In the graph, the y-axis depicts the fraction affected values and the x-axis depicts either the single drug dose treatment (blue and red labels) or the total drug dose after combinational treatment (green label). All dose–response curves plotted in the graph concern 1 representative case. (B) The interaction between IB and GSK126 at a 1:1 ratio in graded concentrations (1-10 μM) was synergistic (combination index [CI] values <1) in primary CLL cells. Scatter plots in the graph present the mean values of the CI of 3 CLL cases. Effects of incubation with CpG for 24 hours after single or combined treatment of 5 μM IB and/or 10 μM GSK126 for 3 days on CLL cell viability (C) and H3K27me3 levels (D) as measured by using flow cytometry. Connected points in the graphs represent the percentage of viable cells or H3K27me3 levels for each case in all conditions described, normalized to control cells (dimethyl sulfoxide [DMSO]–treated). The bars in the graphs show the mean values. Asterisks above bars indicate significant differences compared with CpG-treated cells (n = 7). (E) Each bar in the graph shows the mean values of FC of Bcl-2, Bcl-xl, Mcl-1, cleaved PARP, and cleaved caspase-3 (Casp-3) of stimulated cells with CpG for 24 hours after treatment with 10 μM GSK126 for 3 days, compared with CpG-stimulated control cells. Asterisks indicate significant differences compared with the CpG-stimulated control cells (n = 4; FC = –1.7, P < .05; FC = –1.6, P < .05; FC = –1.9, P < .05; FC = 1.8; and FC = 1.6, respectively). Effects of incubation with 10 μM of the EZH2 inhibitor GSK126 for 3 days on levels of H3K27me3 (F) and on CLL cell viability (G) using flow cytometry. Connected points in the graphs represent the percentage of H3K27me3 or viable cells treated with GSK126 or DMSO-treated control cells (n = 6). (H) Each bar in the graph shows the mean values of FC of Bcl-2, Mcl-1, cleaved PARP, and cleaved caspase-3 (Casp-3) of cells treated with 10 μM GSK126 for 3 days compared with DMSO-treated cells. Asterisks indicate significant differences compared with control cells (DMSO-treated) (n = 4; FC = –1.2, P < .05; FC = –1.4, P < .05; FC = 2; and FC = 2.5). *P < .05, **P < .01.
Similar experiments were also performed with ibrutinib, idelalisib, and/or another EZH2 inhibitor (GSK343) and with ibrutinib, GSK126, and/or the BCL-2 inhibitor venetoclax. We found that all combinations induced a comparable significant viability reduction compared with the control cells that were not exposed to any drug but also to cells exposed to single inhibitors. Moreover, synergism was confirmed between GSK126 and venetoclax (supplemental Figure 5). Interestingly, cases with low EZH2 levels that were unresponsive to TLR9 stimulation showed no significant differences after single vs combined inhibition (supplemental Figure 6). Additional experiments found that EZH2 inhibition is also effective in the stromal coculture system (supplemental Figure 7). Our results indicate that the identified synergism is likely linked to a particular functional status shaped by immune activation and the resultant EZH2 induction, further emphasizing the value of targeting distinct albeit interconnected mechanisms and pathways contributing prosurvival and drug escape signals to the CLL clone.
Finally, using the EZH2 inhibitors (GSK126 and GSK343), we treated primary cells from samples collected at the time of lack of response in CLL cases resistant to ibrutinib or idelalisib (supplemental Table 1). We found a statistically significant (P < .05) decrease in H3K27me3 levels and CLL cell viability (Figure 2F-G; supplemental Figure 8) accompanied by a significant decrease in the antiapoptotic regulators Bcl-2 and Mcl-1 (FC = –1.2, FC = –1.4; P < .05) and an increase in the proapoptotic proteins cleaved PARP and cleaved caspase-3 (FC = 2, FC = 2.5) (Figure 2H).
In conclusion, EZH2 expression seems to be regulated by signals from the CLL microenvironment, whereas the combination of EZH2 and immune signaling inhibitors has synergistic antitumor effects. Moreover, EZH2 inhibitors are effective in primary CLL cells from cases with suboptimal response to immune signaling inhibitors, further highlighting that EZH2 might represent a rational choice for combination treatment of CLL, capable of improving the efficacy/quality of responses to signaling inhibitors.
Supplementary Material
The full-text version of this article contains a data supplement.
Acknowledgments
This work was supported in part by the TRANSCAN-2 Joint Transnational Call for Proposals 2014 by the European Commission/DG Research and Innovation; cofinanced by Greece and the European Union (European Social Fund-ESF) through the Operational Programme “Human Resources Development, Education and Lifelong Learning” in the context of the project “Reinforcement of Postdoctoral Researchers” (MIS-5001552), implemented by the State Scholarships Foundation (ΙΚΥ); the KRIPIS action, funded by the General Secretariat for Research and Technology of Greece; the Swedish Cancer Society; the Swedish Research Council; the Knut and Alice Wallenberg Foundation; Karolinska Institutet; the Lion’s Cancer Research Foundation; the Marcus Borgström Foundation and Selander’s Foundation; the Italian Association for Cancer Research (AIRC, Special Program Molecular Clinical Oncology, 5 per mille #9965) (P.G.), PRIN 2015ZMRFEA; the Italian Ministry of University and Research–MIUR; and TRANSCAN-143 CGH-CLL, JTC 2015.
Authorship
Contribution: E.C., S.N., N.P., and K.S. were responsible for conception and design; E.C., S.N., K.K., A.R., M.A.A., D.B., and E.F. developed the methods; N.S., A.A., P.G., R.R., and K.S. acquired the data (ie, provided patient samples, provided facilities); E.C., S.N., and N.P. analyzed and interpreted the data (eg, statistical analysis); E.C., S.N., P.G., R.R., T.T., N.P., and K.S. wrote, reviewed, and/or revised the manuscript; and N.P. and K.S. supervised the study.
Conflict-of-interest disclosure: K.S. received honoraria from AbbVie, Gilead Science, and Janssen; and research funding from AbbVie, Gilead, and Janssen. P.G. received honoraria from AbbVie, Acerta, BeiGene, Gilead, and Janssen; and research support from AbbVie, Gilead, Janssen, and Novartis. The remaining authors declare no competing financial interests.
Correspondence: Kostas Stamatopoulos, Institute of Applied Biosciences, Centre for Research and Technology Hellas, 6th km Charilaou-Thermi Rd, GR 57001 Thermi, Thessaloniki, Greece; e-mail: kostas.stamatopoulos@certh.gr.
References
- 1.Cao R, Wang L, Wang H, et al. Role of histone H3 lysine 27 methylation in polycomb-group silencing. Science. 2002;298(5595):1039-1043. [DOI] [PubMed] [Google Scholar]
- 2.Papakonstantinou N, Ntoufa S, Chartomatsidou E, et al. The histone methyltransferase EZH2 as a novel prosurvival factor in clinically aggressive chronic lymphocytic leukemia. Oncotarget. 2016;7(24):35946-35959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Papakonstantinou N, Ntoufa S, Chartomatsidou E, et al. Differential microRNA profiles and their functional implications in different immunogenetic subsets of chronic lymphocytic leukemia. Mol Med. 2013;19:115-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rabello DA, Lucena-Araujo AR, Alves-Silva JC, et al. Overexpression of EZH2 associates with a poor prognosis in chronic lymphocytic leukemia. Blood Cells Mol Dis. 2015;54(1):97-102. [DOI] [PubMed] [Google Scholar]
- 5.Yamagishi M, Uchimaru K. Targeting EZH2 in cancer therapy. Curr Opin Oncol. 2017;29(5):375-381. [DOI] [PubMed] [Google Scholar]
- 6.Morera L, Lübbert M, Jung M. Targeting histone methyltransferases and demethylases in clinical trials for cancer therapy. Clin Epigenetics. 2016;8(1):57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ghia P, Chiorazzi N, Stamatopoulos K. Microenvironmental influences in chronic lymphocytic leukaemia: the role of antigen stimulation. J Intern Med. 2008;264(6):549-562. [DOI] [PubMed] [Google Scholar]
- 8.Ntoufa S, Vardi A, Papakonstantinou N, et al. Distinct innate immunity pathways to activation and tolerance in subgroups of chronic lymphocytic leukemia with distinct immunoglobulin receptors. Mol Med. 2012;18(9):1281-1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sutton LA, Rosenquist R. The complex interplay between cell-intrinsic and cell-extrinsic factors driving the evolution of chronic lymphocytic leukemia. Semin Cancer Biol. 2015;34:22-35. [DOI] [PubMed] [Google Scholar]
- 10.Hallek M. Chronic lymphocytic leukemia: 2017 update on diagnosis, risk stratification, and treatment. Am J Hematol. 2017;92(9):946-965. [DOI] [PubMed] [Google Scholar]
- 11.Byrd JC, Furman RR, Coutre SE, et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med. 2013;369(1):32-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Furman RR, Sharman JP, Coutre SE, et al. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med. 2014;370(11):997-1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nair KS, Cheson B. The role of idelalisib in the treatment of relapsed and refractory chronic lymphocytic leukemia. Ther Adv Hematol. 2016;7(2):69-84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ayed AO, Parikh SA. Management of patients with chronic lymphocytic leukemia at high risk of relapse on ibrutinib therapy. Leuk Lymphoma. 2018;59(10):2287-2296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Burger JA, Tedeschi A, Barr PM, et al. ; RESONATE-2 Investigators . Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373(25):2425-2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Byrd JC, Furman RR, Coutre SE, et al. Three-year follow-up of treatment-naïve and previously treated patients with CLL and SLL receiving single-agent ibrutinib. Blood. 2015;125(16):2497-2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herishanu Y, Pérez-Galán P, Liu D, et al. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood. 2011;117(2):563-574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Szurián K, Csala I, Marosvári D, et al. EZH2 is upregulated in the proliferation centers of CLL/SLL lymph nodes. Exp Mol Pathol. 2018;105(2):161-165. [DOI] [PubMed] [Google Scholar]
- 19.Hallek M, Cheson BD, Catovsky D, et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment and supportive management of chronic lymphocytic leukemia. Blood. 2018;131(25):2745-2760. [DOI] [PubMed] [Google Scholar]
- 20.Iannetti A, Ledoux AC, Tudhope SJ, et al. Regulation of p53 and Rb links the alternative NF-κB pathway to EZH2 expression and cell senescence. PLoS Genet. 2014;10(9):e1004642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Longo PG, Laurenti L, Gobessi S, et al. The Akt signaling pathway determines the different proliferative capacity of chronic lymphocytic leukemia B-cells from patients with progressive and stable disease. Leukemia. 2007;21(1):110-120. [DOI] [PubMed] [Google Scholar]
- 22.Dadashian EL, McAuley EM, Liu D, et al. TLR signaling is activated in lymph node–resident CLL cells and is only partially inhibited by ibrutinib. Cancer Res. 2019;79(2):360-371. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.