Abstract
Deep vein thrombosis (DVT), caused by alterations in venous homeostasis, is the third most common cause of cardiovascular mortality, however, key molecular determinants in venous thrombosis have not been fully elucidated. Several lines of evidence indicate that DVT occurs at the intersection of dysregulated inflammation and coagulation. The enzyme ectonucleoside tri(di)phosphohydrolase (ENTPD1, also known as CD39) is a vascular ecto-apyrase on the surface of leukocytes and the endothelium that inhibits intravascular inflammation and thrombosis by hydrolysis of phosphodiester bonds from nucleotides released by activated cells. Here, we evaluated the contribution of CD39 to venous thrombosis in a restricted-flow model of murine inferior vena cava stenosis. CD39 deficiency conferred a greater than 2-fold increase in venous thrombogenesis, characterized by increased leukocyte engagement, neutrophil extracellular trap formation, fibrin, and local activation of tissue factor in the thrombotic milieu. This venous thrombogenesis was orchestrated by increased phosphorylation of the p65 subunit of NF-κB, activation of the NLR family pyrin domain–containing 3 (NLRP3) inflammasome, and IL-1β release in CD39-deficient mice. Substantiating these findings, an IL-1β–neutralizing antibody or the IL-1 receptor inhibitor anakinra attenuated the thrombosis risk in CD39-deficient mice. These data demonstrate that IL-1β is a key accelerant of venous thrombo-inflammation, which can be suppressed by CD39. CD39 inhibits in vivo crosstalk between inflammation and coagulation pathways and is a critical vascular checkpoint in venous thrombosis.
Keywords: Inflammation, Vascular Biology
Keywords: Cardiovascular disease, Innate immunity, Thrombosis
Introduction
The cell-surface enzyme ectonucleoside tri(di)phosphohydrolase-1 (ENTPD1), also known as CD39), is a key molecular factor protecting blood fluidity at the vessel-wall interface by catalyzing the hydrolytic cleavage of phosphodiester bonds, thereby dissipating the extracellular purinergic nucleotides ATP and ADP. These extracellular purines function as intravascular autocrine and paracrine “danger” signals to a variety of cells including innate immune cells, platelets, and endothelium, activating inflammatory and thrombotic pathways (1, 2). Our group has recently shown that CD39 haploinsufficiency is a critical mediator of arterial leukocyte trafficking, atherosclerosis, and platelet hyperactivity (3–5), but the role for CD39 in the venous milieu has not been fully elucidated. In contrast to arterial beds, veins are characterized by low shear, an absence of atherosclerotic plaque, and an emphasis on tissue factor–driven thrombosis. Despite the substantial morbidity and mortality burden of deep vein thrombosis (DVT) (6), proximate mechanisms that trigger and propagate venous thrombosis are incompletely understood. As a window to understanding venous thrombosis pathogenesis, many acquired risk factors are associated with DVT, including trauma, infection, and chronic inflammatory diseases, suggesting an integral association between inflammation and coagulation (7). Although multiple, sometimes coalescing, pathologic drivers can result in venous thrombosis, current studies have largely focused on coagulation, while the precise mechanisms underlying inflammatory triggers and their endogenous checkpoints remain unknown. We hypothesized that CD39-haploinsufficient mice would have impaired protection against DVT, enhanced leukocyte recruitment and activation, and unrestrained inflammatory signaling.
Here, we show that CD39 plays a critical role in protection from venous thrombogenesis in restricted venous flow conditions by checking leukocyte engagement and inflammasome activation. Our data show that loss of a single Cd39 allele increases venous thrombosis and propagation, with enhanced tissue factor and fibrin deposition, neutrophil extracellular trap (NET) formation, and canonical inflammasome activation, under control of NF-κB, the NOD-like receptor pyrin domain–containing 3 protein (NLRP3) inflammasome, and IL-1β expression. Taken together, these data provide evidence that CD39 serves as an indispensable checkpoint of venous inflammation and coagulation at the blood-vessel interface by restraining innate immune activation.
Results and Discussion
CD39 protects against venous thrombosis following inferior vena cava flow restriction.
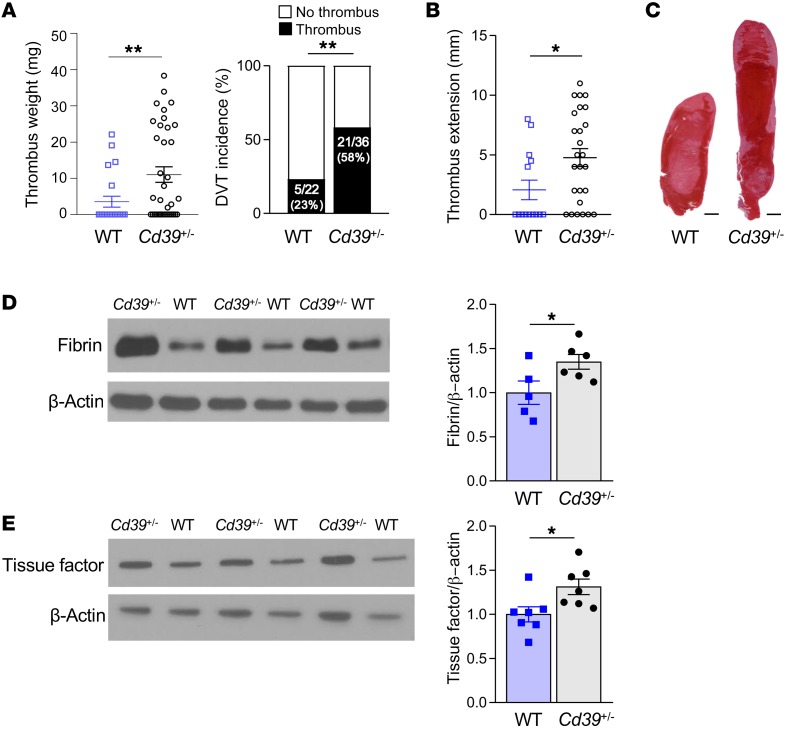
Our group and others have previously shown that CD39 has a potent antithrombotic and antiinflammatory effect in the context of arterial flow conditions (3, 4, 8). We first validated our model of flow-restricted venous thrombosis in mice with a normal complement of CD39 (WT), with thrombus occurring in 23% of mice (Figure 1A), consistent with prior reports in this model (9–11). These thrombi contained regions that were RBC rich and RBC poor, similar to the morphology seen in human venous thrombi (12). Given that expression of CD39 can be dynamically downregulated by systemic cytotoxic stress or local turbulent blood flow patterns and that it is being studied as an inhibition target in cancer, we investigated whether partial deficiency of CD39 could contribute to venous thrombosis under flow-restricted conditions. We used CD39-haploinsufficient mice to test the effect of CD39 deficiency on venous thrombosis. Cd39+/– mice have similar circulating leukocyte, RBC, and platelet counts when compared with those of genotype controls (WT) containing the nonexcised LoxP sites used to generate the Cd39+/– mice (13). When subjected to inferior vena cava (IVC) flow restriction, Cd39+/– mice developed significantly more venous thrombosis compared with WT controls (Figure 1A), coupled with more thrombus propagation, sometimes extending into the iliac veins (Figure 1, B and C). As activation of the coagulation cascade results in thrombin-induced cleavage of fibrinogen to fibrin, we examined the effect of CD39 haploinsufficiency on fibrin formation in the venous thrombus milieu. Using a fibrin-specific antibody, we determined that Cd39+/– mice had greater fibrin accumulation than did WT mice, suggesting an increase in the insoluble fibrin networks in the vein lumen that contributed to heightened thrombus stabilization in Cd39+/– mice (Figure 1D). Whether the increased fibrin content in venous thrombi from Cd39+/– mice reflects greater fibrin deposition or deficient fibrinolysis is unknown and remains to be elucidated. Tissue factor, a significant determinant of thrombus initiation and propagation, is released by hematopoietic cells under venous flow–restricted conditions and complexes with factor VII/VIIa in blood to activate the tissue factor (extrinsic) coagulation pathway (14). Consistent with our findings of increased venous thrombogenesis, thrombus lysate from Cd39+/– mice contained more tissue factor when compared with WT controls (Figure 1E).
Figure 1. Increased venous thrombogenesis, clot extension, and fibrin content in Cd39+/– mice compared with genotype controls.
(A) Thrombus weights and thrombus frequency 2 days after IVC flow restriction (stenosis) (WT n = 22, Cd39+/– n = 36). (B) Thrombus extension (WT n = 14, Cd39+/– n = 26). (C) Representative H&E-stained sections of thrombi (n = 5, each). Scale bars: 1 mm. (D) Immunoblots of fibrin content of thrombus lysates (WT n = 5, Cd39+/– n = 6) and (E) tissue factor (n = 7 for each). Data represent the mean ± SEM. *P < 0.05 and **P < 0.01, by 2-tailed Student’s t test (D and E) with Welch’s correction (A and B). Data shown in the right panel in A were analyzed using the χ2 method.
CD39 haploinsufficiency increases NET formation in venous thrombosis.
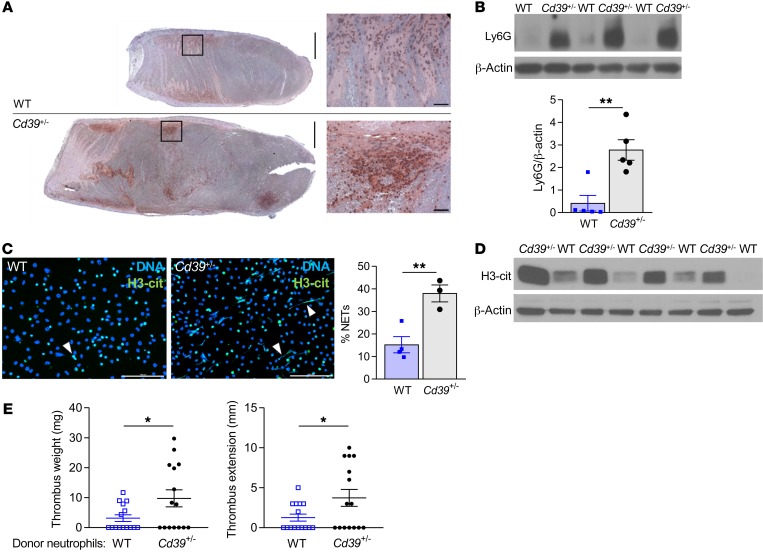
We next examined whether the increased thrombogenesis in Cd39+/– mice could in part be explained by leukocyte engagement. Venous thrombi in Cd39+/– mice revealed enhanced leukocyte recruitment, driven primarily by neutrophils (Figure 2, A and B). Neutrophil activation with chromatin decondensation and release of web-like traps facilitate heterotypic cell interactions in venous thrombosis under flow restriction, serving as a scaffold for platelet aggregation, neutrophil and monocyte recruitment, and fibrin deposition (14, 15). During thrombosis, NETs also construct a procoagulant milieu, sequestering vWF, activating factor XII in the intrinsic coagulation cascade, and inactivating the tissue factor pathway inhibitor (14, 16, 17). The prominent leukocyte recruitment and fibrin deposition observed in the developing thrombus in Cd39+/– mice led us to next examine whether CD39 deficiency would be sufficient to trigger an increase in neutrophil activation and NET formation. Neutrophils from Cd39+/– mice had reduced expression of CD39 compared with WT neutrophils (Supplemental Figure 1A; supplemental material available online with this article; https://doi.org/10.1172/JCI124804DS1). Following in vitro stimulation with PMA, a potent inducer of NADPH oxidase–dependent NET formation, mature neutrophils from naive Cd39+/– mice, as compared with genotype controls, had a marked, greater than 2.5-fold increase in expression of a citrullinated form of histone H3 (H3-cit), which is a NET-associated histone modification that results in loosened chromatin structures that can be extruded as NETs (Figure 2C). Consistent with these in vitro findings, immunoblots of venous thrombus lysate from Cd39+/– mice also showed significantly higher levels of H3-cit expression than the levels seen in WT mice (Figure 2D and Supplemental Figure 1B). Confirming the role of neutrophil CD39 haploinsufficiency in promoting thrombosis, Cd39+/– neutrophils administered to WT mice increased thrombus size compared with administration of WT neutrophils (Figure 2E). Taken together, these data suggest that during venous thrombogenesis, CD39 is an obligatory restraint on H3-cit–positive NET formation in vitro and in vivo.
Figure 2. CD39 tempers thrombus cellularity and leukocyte recruitment to growing thrombus.
(A) Neutrophil-stained (Ly6G) venous thrombi (brown, Ly6G; n = 5 for each). Scale bars: 1 mm and 50 μm (insets). (B) Immunoblot for Ly6G in venous thrombi from WT and Cd39+/– mice (n = 5 for each). (C) H3-cit expression indicates NET formation by immunofluorescence microscopy following ex vivo stimulation with 100 nM PMA (green, H3-cit; blue, DNA; white arrowheads, NETs). WT n = 4, and Cd39+/– n = 3. Scale bars: 100 μm. Representative images are shown. (D) H3-cit content in venous thrombus lysates (n = 4 for each). (E) Venous thrombus weight and extension in WT mice following adoptive transfer of WT or Cd39+/– neutrophils (n = 15 for each). Data represent the mean ± SEM. *P < 0.05 and **P < 0.01, by 2-tailed Student’s t test.
Exaggerated inflammasome activation in venous thrombosis in CD39-deficient mice.
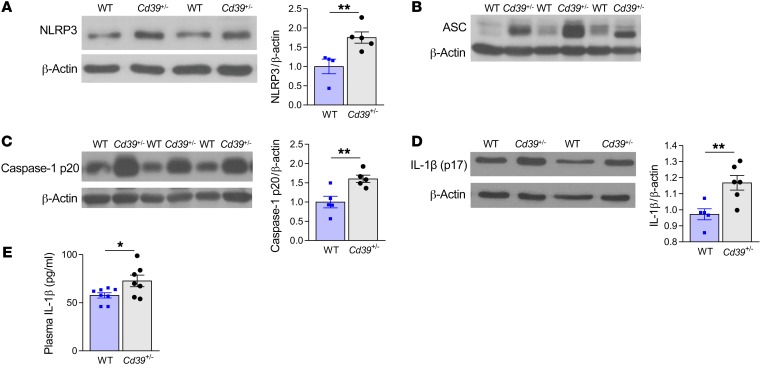
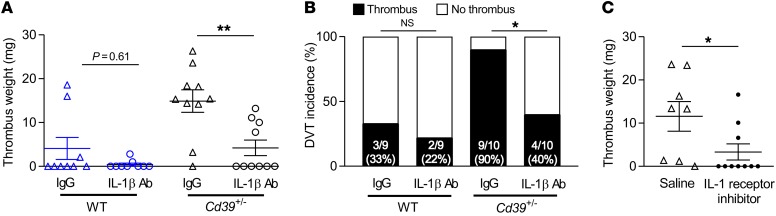
Extracellular release of ATP and ADP through cell death, injury, or activation is a potent stress response, altering the local microenvironment to activate paracrine and autocrine signaling pathways (18, 19). Binding of extracellular ATP to the plasma membrane receptor ionophore P2X7 activates a potent stress-response–signaling pathway characterized by potassium efflux, which triggers assembly and activity of the inflammasome, a multiprotein oligomer that activates highly proinflammatory cytokines including IL-1β (20). Gupta et al. recently reported increased NLRP3 inflammasome assembly in patients at high altitude at risk for DVT (21). Canonical inflammasome activation requires a “priming” step marked by NF-κB activation and inflammasome component transcription (20). A second signal initiates NLRP3-mediated assembly and oligomerization of inflammasome component fibers, proteolytic cleavage of pro–caspase-1 to caspase-1 to generate mature IL-1β, and release of prothrombotic tissue factor–rich microparticles (22, 23). As ATP, the substrate for the phosphohydrolytic activity of CD39, is the primary activating ligand for the vascular P2X7 receptor (24), we hypothesized that CD39 could suppress inflammasome activation in venous thrombosis and that haploinsufficiency would thus drive a proinflammatory state. We examined the expression of NLRP3 inflammasome components in venous thrombi from Cd39+/– and WT mice and observed markedly enhanced phosphorylation of the transactivator serine534 site (murine homolog of human serine536) of the NF-κB p65 subunit in Cd39+/– thrombi (Supplemental Figure 2A). This was accompanied by increased expression of NLRP3, apoptosis-associated speck-like protein containing a CARD (ASC), and active caspase-1 in Cd39+/– mice (Figure 3, A–C, and Supplemental Figure 2B), as well as exaggerated mature (processed) IL-1β content (Figure 3D) compared with WT thrombi. Concordant with these data, we detected elevated plasma levels of IL-1β in Cd39+/– mice compared with levels in WT mice following venous thrombosis (Figure 3E). A recent large clinical trial demonstrated reduced arterial cardiovascular events in high-risk patients following treatment with a monoclonal antibody targeting IL-1β (25). To determine whether the exaggerated venous thrombosis conferred by CD39 haploinsufficiency could be rescued by reduced IL-1β bioavailability, we next performed IVC flow restriction in WT and Cd39+/– mice treated with an IL-1β–neutralizing antibody (Figure 4, A and B). IL-1β blockade resulted in a striking decrease in venous thrombogenesis in CD39-deficient mice; specifically, Cd39+/– mice treated with a neutralizing IL-1β antibody developed smaller and fewer venous thrombi than did isotype IgG–injected Cd39+/– mice under flow-restricted conditions, whereas WT mice treated with neutralizing IL-1β antibody had rates of thrombogenesis similar to those of IgG controls and a nonsignificant decrease in thrombus size. IL-1β blockade did not prolong the tail bleeding time (data not shown). To exclude an IgG-dependent effect of IL-1β blockade on Cd39+/– venous thrombosis, Cd39+/– mice were next treated with anakinra, a synthetic inhibitor of the IL-1 receptor, or saline vehicle control. Concordant with IL-1β neutralization experiments, IL-1 receptor inhibition resulted in lower thrombotic burden than was observed in saline-treated Cd39+/– mice in contemporaneous experiments (Figure 4C). Taken together, these experiments show a pivotal role for CD39, the dominant vascular ectonucleotidase, in inhibiting NLRP3 inflammasome activation and IL-1β signaling during venous thrombogenesis.
Figure 3. CD39 regulates inflammasome activation in mice with flow-restricted venous thrombosis.
Expression of (A) NLRP3 (WT n = 4, Cd39+/– n = 5), (B) ASC, (C) cleaved caspase-1 (p20 fragment) (n = 5 for each), and (D) mature IL-1β in thrombus lysates (WT n = 5, Cd39+/– n = 6). (E) plasma IL-1β expression by ELISA (WT n = 8, Cd39+/– n = 7). Data represent the mean ± SEM. *P < 0.05 and **P < 0.01, by 2-tailed Student’s t test.
Figure 4. IL-1β inhibition reduces the incidence of venous thrombosis in CD39-deficient mice.
(A) Thrombus size and (B) frequency of DVT in WT and Cd39+/– mice administered IL-1β–neutralizing or IgG isotype control antibody (200 μg i.p. 24 hours prior to surgery, n = 9–10 for each). (C) Thrombus size in Cd39+/– mice treated with vehicle control or the IL-1 receptor inhibitor anakinra (100 mg/kg/day i.p., n = 8–10 for each). Data represent the mean ± SEM. *P < 0.05 and **P < 0.01, by 2-tailed Student’s t test with Welch’s correction (A and C) and χ2 method (B).
In summary, our findings demonstrate for the first time to our knowledge that inflammasome-mediated IL-1β is essential for venous thrombogenesis under vascular conditions which include limited flow. These data also reveal an indispensable role for vascular CD39 in protecting against venous thrombogenesis, as it alters multiple aspects of venous clot formation. A functional paradigm exists, wherein venous thrombosis develops as a convergence of multiple inputs from several cell lineages, including neutrophils, monocytes, platelets, and endothelial cells. In addition to prothrombotic and proinflammatory mediators, these vascular and innate immune effector cells possess the machinery for inflammasome activation and IL-1β processing (14, 20, 26). Our data show that CD39 is vital for reducing intravascular fibrin deposition, which otherwise provides mechanical and structural support to nascent thrombi; for restraining NET formation; for NLRP3 inflammasome activation; and, critically, for IL-1β accessibility during venous thrombosis. IL-1β may facilitate venous thrombosis through several effector mechanisms in the thrombotic milieu including leukocyte recruitment (27, 28), remote signaling via thrombogenic microparticles (23), and platelet integrin activation (29). Indeed, platelets in Cd39+/– mice are also hyperactive (4), suggesting that CD39-based targeted therapeutics may be effective in interrupting a positive feedback loop of thrombo-inflammation (5, 30). Taken together, these data present a compelling role for CD39 as a vascular checkpoint in impeding venous thrombus accretion. Treatments for venous thrombosis have largely targeted the coagulation pathway, leading to incomplete protection from DVT. Strategies aimed at the nexus of inflammation and coagulation will be critical for progress in the development of effective therapeutics in venous thrombosis.
Methods
Detailed Methods are provided in the Supplemental Methods.
Statistics.
Data analysis was performed using GraphPad Prism 8 software. Normally distributed data were analyzed using an unpaired, 2-tailed Student’s t test, and skewed data were analyzed using Welch’s correction. Thrombus frequency was analyzed by χ2 analysis. Statistical significance was defined as a P value of less than 0.05.
Study approval.
All experiments were conducted in accordance with NIH guidelines for the use of live animals and were approved by the IACUC of the University of Michigan.
Author contributions
YK and DJP were responsible for the study concept and design. YK, VY, LC, AB, RZ, HL, BNJ, SY, and BET acquired data. YK, VY, LC, AB, RZ, HL, and BET performed data analysis. YK, BET, DJP, JSK, JMK, SHV, MAH, and ACA interpreted the data. SK and HL were responsible for mouse strain generation and colony management. YK and DJP supervised the study, obtained funding, and drafted the manuscript.
Supplementary Material
Acknowledgments
The authors would like to thank Cathy Luke, Peter Henke, and Gabriel Nunez (University of Michigan) for helpful discussions. The authors also acknowledge members of the University of Michigan Dental School Histology Core. This work was supported in part by funding from the following: the NIH (K08HL131993, to YK; T32HL007853, to BNJ and ACA; HL127151 and NS087147, to DJP; GM105671 and HL114405, to MH; K99HL136784, to BET; HL134846, to JSK; AR072107, to JMK; and HL119623, to SHV); a J. Griswold Ruth MD and Margery Hopkins Ruth Professorship; the Haller Family Foundation; the A. Alfred Taubman Medical Research Institute (to DJP); a McKay Grant; the Bo Schembechler Heart of a Champion Foundation; the Jobst-American Venous Forum Foundation (to YK); and the University of Michigan Frankel Cardiovascular Center.
Version 1. 04/16/2019
In-Press Preview
Version 2. 05/28/2019
Electronic publication
Version 3. 07/01/2019
Print issue publication
Footnotes
Conflict of interest: YK and DJP are co-inventors on an unrelated patent (application no. 20180369278). DJP is an inventor on unrelated patents (patent application no. 2017192553A1; and US patent nos. 9789133, held by Proterris Inc., 8128963, held by Columbia University, and 6972175). MH is an inventor on unrelated patents (US patent nos. 10266488, 9750757, and 9789087B2; and patent application nos. PCT/US17/38994, PCT/US19/27785, and PCT/US19/27881).
Copyright: © 2019, American Society for Clinical Investigation.
Reference information: J Clin Invest. 2019;129(7):2872–2877.https://doi.org/10.1172/JCI124804.
Contributor Information
Vinita Yadav, Email: vinitay@med.umich.edu.
Liguo Chi, Email: cliguo@med.umich.edu.
Raymond Zhao, Email: zhaoray@umich.edu.
Srilakshmi Yalavarthi, Email: syalavar@med.umich.edu.
Benjamin N. Jacobs, Email: benjac@umich.edu.
Alison Banka, Email: albanka@umich.edu.
Hui Liao, Email: huiliao@umich.edu.
Sharon Koonse, Email: koonse@med.umich.edu.
Anuli C. Anyanwu, Email: canyanwu@med.umich.edu.
J. Michelle Kahlenberg, Email: mkahlenb@med.umich.edu.
Jason S. Knight, Email: jsknight@umich.edu.
David J. Pinsky, Email: dpinsky@umich.edu.
Yogendra Kanthi, Email: ykanthi@med.umich.edu.
References
- 1.Marcus AJ, Broekman MJ, Pinsky DJ. COX inhibitors and thromboregulation. N Engl J Med. 2002;347(13):1025–1026. doi: 10.1056/NEJMcibr021805. [DOI] [PubMed] [Google Scholar]
- 2.Kanthi YM, Sutton NR, Pinsky DJ. CD39: Interface between vascular thrombosis and inflammation. Curr Atheroscler Rep. 2014;16(7):425. doi: 10.1007/s11883-014-0425-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hyman MC, et al. Self-regulation of inflammatory cell trafficking in mice by the leukocyte surface apyrase CD39. J Clin Invest. 2009;119(5):1136–1149. doi: 10.1172/JCI36433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kanthi Y, et al. Flow-dependent expression of ectonucleotide tri(di)phosphohydrolase-1 and suppression of atherosclerosis. J Clin Invest. 2015;125(8):3027–3036. doi: 10.1172/JCI79514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pinsky DJ, et al. Elucidation of the thromboregulatory role of CD39/ectoapyrase in the ischemic brain. J Clin Invest. 2002;109(8):1031–1040. doi: 10.1172/JCI10649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Yusuf HR, Tsai J, Atrash HK, Boulet S, Gross SD. Venous thromboembolism in adult hospitalizations-United States, 2007-2009. Centers for Disease Control and Prevention. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6122a1.htm?s_cid=mm6122a1_w Updated June 8, 2012. Accessed April 23, 2019.
- 7.Wolberg AS, et al. Venous thrombosis. Nat Rev Dis Primers. 2015;1:15006. doi: 10.1038/nrdp.2015.6. [DOI] [PubMed] [Google Scholar]
- 8.Covarrubias R, et al. Role of the CD39/CD73 purinergic pathway in modulating arterial thrombosis in mice. Arterioscler Thromb Vasc Biol. 2016;36(9):1809–1820. doi: 10.1161/ATVBAHA.116.307374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Geddings JE, et al. Tissue factor-positive tumor microvesicles activate platelets and enhance thrombosis in mice. J Thromb Haemost. 2016;14(1):153–166. doi: 10.1111/jth.13181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meng H, et al. In Vivo Role of Neutrophil Extracellular Traps in Antiphospholipid Antibody-Mediated Venous Thrombosis. Arthritis Rheumatol. 2017;69(3):655–667. doi: 10.1002/art.39938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brill A, et al. von Willebrand factor-mediated platelet adhesion is critical for deep vein thrombosis in mouse models. Blood. 2011;117(4):1400–1407. doi: 10.1182/blood-2010-05-287623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Savchenko AS, et al. Neutrophil extracellular traps form predominantly during the organizing stage of human venous thromboembolism development. J Thromb Haemost. 2014;12(6):860–870. doi: 10.1111/jth.12571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Visovatti SH, et al. Purinergic dysregulation in pulmonary hypertension. Am J Physiol Heart Circ Physiol. 2016;311(1):H286–H298. doi: 10.1152/ajpheart.00572.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.von Brühl ML, et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J Exp Med. 2012;209(4):819–835. doi: 10.1084/jem.20112322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martinod K, et al. Neutrophil histone modification by peptidylarginine deiminase 4 is critical for deep vein thrombosis in mice. Proc Natl Acad Sci U S A. 2013;110(21):8674–8679. doi: 10.1073/pnas.1301059110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Massberg S, et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat Med. 2010;16(8):887–896. doi: 10.1038/nm.2184. [DOI] [PubMed] [Google Scholar]
- 17.Carestia A, Rivadeneyra L, Romaniuk MA, Fondevila C, Negrotto S, Schattner M. Functional responses and molecular mechanisms involved in histone-mediated platelet activation. Thromb Haemost. 2013;110(5):1035–1045. doi: 10.1160/TH13-02-0174. [DOI] [PubMed] [Google Scholar]
- 18.Carta S, et al. Cell stress increases ATP release in NLRP3 inflammasome-mediated autoinflammatory diseases, resulting in cytokine imbalance. Proc Natl Acad Sci U S A. 2015;112(9):2835–2840. doi: 10.1073/pnas.1424741112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen Y, et al. ATP release guides neutrophil chemotaxis via P2Y2 and A3 receptors. Science. 2006;314(5806):1792–1795. doi: 10.1126/science.1132559. [DOI] [PubMed] [Google Scholar]
- 20.Franchi L, Kanneganti TD, Dubyak GR, Núñez G. Differential requirement of P2X7 receptor and intracellular K+ for caspase-1 activation induced by intracellular and extracellular bacteria. J Biol Chem. 2007;282(26):18810–18818. doi: 10.1074/jbc.M610762200. [DOI] [PubMed] [Google Scholar]
- 21.Gupta N, et al. Activation of NLRP3 inflammasome complex potentiates venous thrombosis in response to hypoxia. Proc Natl Acad Sci U S A. 2017;114(18):4763–4768. doi: 10.1073/pnas.1620458114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Enjyoji K, et al. Targeted disruption of cd39/ATP diphosphohydrolase results in disordered hemostasis and thromboregulation. Nat Med. 1999;5(9):1010–1017. doi: 10.1038/12447. [DOI] [PubMed] [Google Scholar]
- 23.Rothmeier AS, et al. Caspase-1-mediated pathway promotes generation of thromboinflammatory microparticles. J Clin Invest. 2015;125(4):1471–1484. doi: 10.1172/JCI79329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lévesque SA, Kukulski F, Enjyoji K, Robson SC, Sévigny J. NTPDase1 governs P2X7-dependent functions in murine macrophages. Eur J Immunol. 2010;40(5):1473–1485. doi: 10.1002/eji.200939741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ridker PM, et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N Engl J Med. 2017;377(12):1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 26.Kahlenberg JM, Thacker SG, Berthier CC, Cohen CD, Kretzler M, Kaplan MJ. Inflammasome activation of IL-18 results in endothelial progenitor cell dysfunction in systemic lupus erythematosus. J Immunol. 2011;187(11):6143–6156. doi: 10.4049/jimmunol.1101284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Karmakar M, et al. Neutrophil IL-1β processing induced by pneumolysin is mediated by the NLRP3/ASC inflammasome and caspase-1 activation and is dependent on K+ efflux. J Immunol. 2015;194(4):1763–1775. doi: 10.4049/jimmunol.1401624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Saxena A, et al. IL-1 induces proinflammatory leukocyte infiltration and regulates fibroblast phenotype in the infarcted myocardium. J Immunol. 2013;191(9):4838–4848. doi: 10.4049/jimmunol.1300725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Qiao J, et al. NLRP3 regulates platelet integrin αIIbβ3 outside-in signaling, hemostasis and arterial thrombosis. Haematologica. 2018;103(9):1568–1576. doi: 10.3324/haematol.2018.191700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hohmann JD, et al. Delayed targeting of CD39 to activated platelet GPIIb/IIIa via a single-chain antibody: breaking the link between antithrombotic potency and bleeding? Blood. 2013;121(16):3067–3075. doi: 10.1182/blood-2012-08-449694. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.