Abstract
Barnacles are conspicuous members of rocky intertidal communities and settlement of the final larval stage, the cyprid, is influenced by the presence of biofilms. While modulation of cyprid settlement by biofilms has been studied extensively, the acquisition of a specific microbiome by the settling larva has not. This study investigated settlement in the field of Semibalanus balanoides in two consecutive years when the composition of the benthic bacterial community differed. In both years, settling cyprids adopted a specific sub-set of benthic bacteria that was distinct from the planktonic cyprid and the benthos. This microbiome was consistent, regardless of annual variability in the benthic community structure, and established within hours of settlement. The results imply that a natural process of selection occurs during the critical final transition of S. balanoides to the sessile form. The apparent consistency of this process between years suggests that optimal growth and survival of barnacles could depend upon a complex inter-kingdom relationship, as has been demonstrated in other animal systems.
Keywords: barnacle, larval settlement, microbiome, cyprid, bacteria, 16S sequencing
1. Introduction
Barnacles are key ecosystem engineers [1] as well as commercially and environmentally important marine biofouling species [2]. As adults, they are mostly sessile and gregarious, the latter being critical during larval settlement and for reproduction as mature adults. The cypris larva explores immersed surfaces to determine suitability for permanent attachment. Cyprids are highly discriminatory and surface selection is informed by a range of physical and biological cues [3].
Benthic biofilms are important stimuli for some settling organisms, such as tubeworms [4,5] and algal spores [6,7]. In some cases, specific bacterial taxa are required [8]. The barnacle literature is ambiguous, with conflicting observations from different barnacle species, and biofilms of varying composition [9–13]. Experiments using bacterial isolates from laboratory-cultured barnacles have suggested that the bacterial flora of barnacle shells and the adjacent substratum differ and that shell-associated bacteria have an inductive effect on settlement [14]. The biofilms present in the barnacle's natural environment, however, are more complex and dynamic than those that survive in the laboratory. Suffice it to say that bacterial films are one of the several important cues that enable cyprids to locate a suitable habitat [3].
There is growing evidence that during metamorphosis from the cyprid to the juvenile barnacle [15], and subsequent growth to adulthood [16], bacteria between the metamorphosing cyprid and the surface to which it is attached are either removed or killed. Barnacles nevertheless retain a significant population of bacterial cells post-metamorphosis, and their intervention in the natural development of their microbiome suggests a relationship between barnacles and bacteria that is more complex than simple acquisition of the local microbial consortium. The suggestion that benthic bacteria could, in addition to serving as a marker of a suitable habitat, also serve as an inoculum for the barnacle ‘holobiont’ (e.g. [17]) has never been explored.
The annual settlement of Semibalanus balanoides in the North Sea provides an opportunity to study this process in situ. Adults of the species release nauplius larvae into the water column only once per year, in early spring. The larvae progress through six ecdyses to the cyprid stage, which settles over a short and intense settlement season of around two weeks in late April/early May. This window of settlement activity was exploited to chart the early development of the barnacle microbiome relative to its surroundings. It was presumed that settling larvae would, over the course of their early development, acquire a bacterial consortium similar to that of the surrounding benthos. It is also intuitive that the most dramatic shift in the barnacle's microbial community might be immediately following metamorphosis from the settled cyprid to the juvenile barnacle, at which point feeding commences. Clarification of these points was considered to be an essential basis for further investigation of the natural relationship between barnacles and bacteria.
2. Material and methods
(a). Sample collection
Experiments were conducted in two settlement seasons, May 2017 and May 2018. The site chosen was the north sea-wall at Cullercoats Bay, UK (figure 1a; 55°02′07.1″ N 1°25′51.7″ W). During this period, cyprids of Semibalanus balanoides are the dominant zooplankton and easily distinguishable from other species by their large size (approx. 1 mm in length). In both seasons, cyprids were collected by trawling the water of the bay immediately below the surface, as well as by collecting settled cyprids and juvenile barnacles in three locations along the wall, each separated by 3 m horizontally. Between trawls, the plankton net was soaked overnight in 5% Decon 90® and rinsed thoroughly before reuse. Trawls of less than 30 min were conducted twice in 2016 and three times in 2017 on consecutive high-tides. Thirty larvae were selected at random for sequencing on each occasion from the thousands collected and all samples were processed immediately after collection. Settled individuals were collected (1) after permanent attachment but before metamorphosis to a juvenile, (2) shortly after metamorphosis to a juvenile but before calcification and (3) post-metamorphosis during initial calcification (figure 1b). Thirty individuals of each life stage were collected in each of the three locations 5 h after each high tide. Care was taken during the removal of the individuals to contact only the animal using a sterile mounted needle—one per location and life stage. Laboratory experiments were then designed to replicate the natural settlement process in the absence of the benthic biofilm. Cyprids collected from the final plankton trawl in each year were washed thoroughly using 5× changes of autoclaved seawater. In each year, several hundred larvae were then allowed to settle, metamorphose and calcify in the laboratory in a single 2 l beaker of autoclaved seawater, circulated using a magnetic stirrer and containing a sterile slate block as a settlement substrate, from which settled individuals were sampled. Environmental samples included scrapings from barnacle shells adjacent to settled larvae in the field, rock containing no settled individuals and, in the laboratory, scrapings of the slate substrate. Analysis by PERMANOVA (electronic supplementary material, SUP1) was used to distinguish ‘laboratory versus field’ samples and ‘planktonic versus pooled settled stages versus benthos' samples. For the planktonic cyprid sample n = 7 (2016/17 combined), for pooled settled stages (settled, metamorphosed and calcified) n = 27 (2016/17 combined) and for benthos n = 10 (2016/17 combined). Differences between specific benthic life stages (settled, metamorphosed, calcified) and between years were not analysed formally, and discussion of them is therefore based upon visual interpretation of principal coordinate analysis (PCoA) plots and Bray–Curtis distance (electronic supplementary material, SUP1).
Figure 1.
(a) The sampling location at Cullercoats Bay, North-East England. (b) Barnacles (Semibalanus balanoides) at different life stages. A = adult, S = settled cyprid, M = metamorphosed juvenile, C = calcified juvenile. (c) Shannon diversity index (y-axis), indicating significant differences between planktonic cyprids and pooled settled samples. (d) Bacterial taxa that differentiated significantly between planktonic cyprids and pooled settled samples. (e) PCoA of all developmental stages and replicates for two sampling years in the laboratory and field (statistics refer to PERMANOVA-based comparisons). There is a clear progression of microbiome development in the field (orange ellipse) from planktonic cyprids (black) to settled cyprids (brown), metamorphosed juveniles (orange) and calcified juveniles (light orange), with samples from both years (2016/2017) clustering at each life stage. The same was not true in the laboratory (blue ellipse—settled cyprids dark blue, calcified juveniles lightest blue). Environmental samples from local rocks and barnacle shells were highly variable (green) but appeared to separate by year, in contrast to barnacle samples.
(b). 16S DNA sequencing and analysis
For details of the sequencing procedure, please refer to electronic supplementary material, SUP1. Raw data are available in electronic supplementary material, SUP2.
3. Results and discussion
Although adhered to a surface, the settled cyprid prior to metamorphosis is fundamentally unchanged from the planktonic, lecithotrophic stage. During metamorphosis to a juvenile, the cyprid carapace is lost through ecdysis along with any associated epibionts, and feeding commences. A consequential shift in the bacterial community was expected. It was surprising, therefore, when results indicated that the greatest difference in the associated bacterial community was between the planktonic and settled cyprids. Shannon diversity was significantly higher (p = 0.043) in the combined settled samples (figure 1c), which had a greater relative abundance of e.g. Flavobacteriaceae (unclassified), Lewinella, Granulosicoccus and Maribius (figure 1d). Planktonic cyprids had higher abundance of e.g. Bizionia. While it appeared that settled cyprids and metamorphosed individuals differed in terms of their bacterial communities (‘Metamorphosis’ stage; figure 1e), the magnitude of this difference was less than that between planktonic and settled cyprids (‘Settlement’ stage; figure 1e). The difference in community structure between planktonic cyprids, settled individuals (all stages) and the benthos was significant (PERMANOVA R2 = 0.143, F = 3.25, p ≤ 0.001). PERMDISP and ANOVA were performed to rule out dispersal as a factor and found to be insignificant (p = 0.29).
There was considerable overlap between the taxa associated with the barnacle shell and the adjacent rock, so for presentation in figure 1e, these were combined together as ‘benthos’. PCoA did not cluster benthos data by location, but there was broad separation by year with samples from 2016 and 2017 being separated across principle coordinate 2, suggesting differing community structures (figure 1e). The same was not true for settling barnacles, where 2016 and 2017 data visually clustered in terms of their bacterial communities at all life stages. The microbiome of early barnacle life stages therefore appeared independent of their immediate environment and maintained despite temporal changes in the community structure of the benthos. Since settled cyprids metamorphose to the juvenile within a day, the adoption of the settled-barnacle microbiome must be rapid. After settlement, changes to the microbiome were less dramatic, although there appeared to be a progression through metamorphosis to calcification (figure 1e).
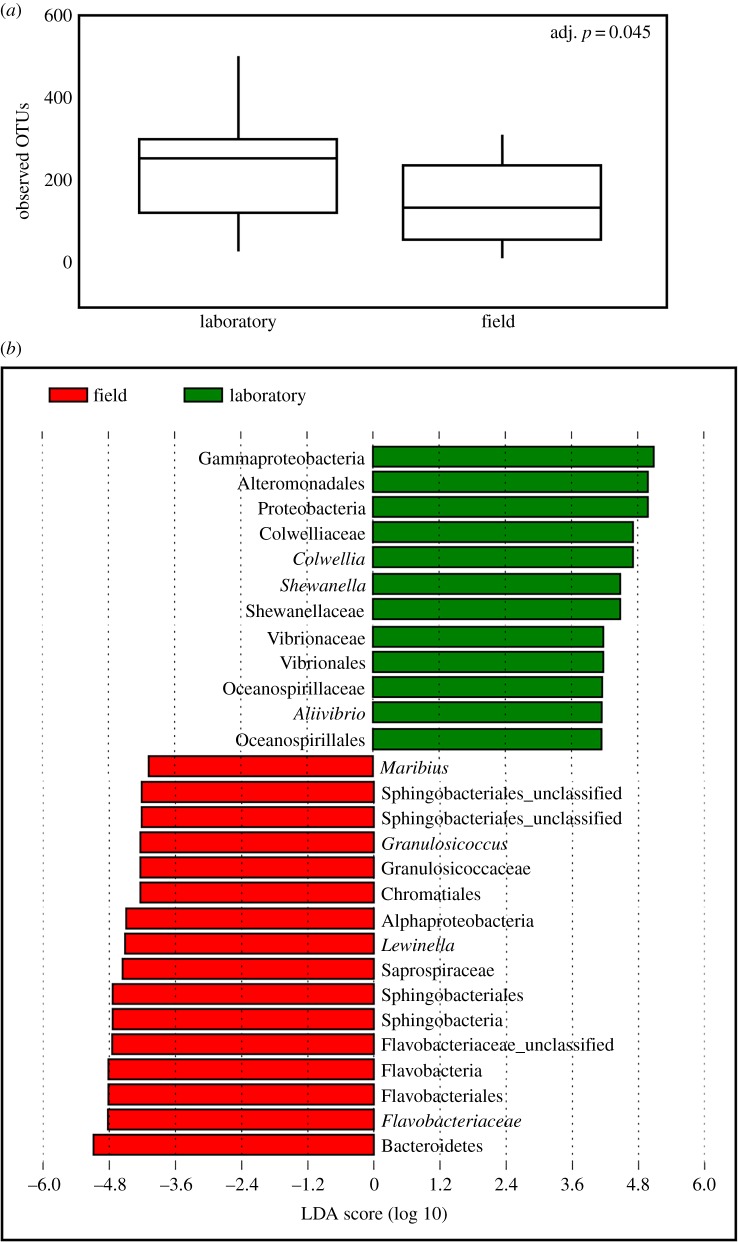
The dramatic shift in the composition of the cyprid microbiome upon settlement in the field did not occur in the laboratory (figure 1e; PERMANOVA R2 = 0.124, F = 5.52, p ≤ 0.001), confirming that the inoculum for development of the juvenile barnacle microbiome was not present on the swimming cyprid and must have been acquired from the benthos. The number of recorded OTUs (figure 2a) was higher in the laboratory, but the abundance of e.g. Shewanella sp. and Colwellia sp. (Figure 2b) on laboratory-reared individuals agrees with their documented tolerance for a broad range of environmental conditions, perhaps enabling them to better survive transfer out of the natural habitat of the host (see electronic supplementary material, SUP3 for alternative presentation of these data).
Figure 2.
(a) The number of bacterial OTUs recorded in field and laboratory experiments. (b) A linear discriminant analysis effect size (LEfSe) plot, presenting bacterial taxa that differentiated between barnacles settled in the laboratory and in the field. Only significant taxa are displayed on the plot (p < 0.05).
It was evident that the microbiome of cyprids changed fundamentally within hours of settlement and that the bacterial consortium acquired from the benthos evolved through metamorphosis and calcification of the juvenile. The results suggest that the same consortium of bacteria was associated with settling larvae in 2 years when the composition of the benthic biofilm differed. However, it is known that bacterial assemblages differ spatially as well as temporally [18]. Pertinent questions for future work would therefore be, how resistant is the barnacle microbiome to environmental change? And how important is microbiome composition to the overall fitness of these important intertidal species [17]?
4. Conclusion
While bacteria were relatively scarce on planktonic cyprids, naturally settled cyprids were colonized rapidly by a precise bacterial consortium that differed relative to planktonic cyprids and the surrounding benthos. Laboratory experiments confirmed that the natural microbiome of juvenile barnacles originated from the benthos. The benthic community differed in two sampling years, but the consortium associated with newly settled barnacles did not. The composition of the benthos was therefore not the major determinant of the barnacle microbiome and this observation provides the first evidence for selective association of specific bacterial taxa with developing barnacles. At this stage, we do not suggest either that the barnacle attracts, passively or actively, specific bacterial strains or that the bacteria most strongly associated with barnacles require the barnacle as a host. Evidently, however, there has evolved a complex inter-kingdom relationship between barnacles and bacteria that may have implications for barnacle growth and survival, and this will be the subject of future work.
Supplementary Material
Supplementary Material
Supplementary Material
Ethics
There are no local or national restrictions on sampling marine invertebrate larvae.
Data accessibility
Raw data are available in electronic supplementary material, SUP2 and Dryad Digital Repository: https://doi.org/10.5061/dryad.85g65v8 [19].
Authors' contributions
N.A. and A.N. designed the study. N.A. collected and prepared samples, while A.N. conducted the 16S sequencing. N.A. and A.N. contributed equally to the preparation of the manuscript. All authors approved the final version of the manuscript and agreed to be held accountable for the content therein.
Competing interests
The authors declare no competing interests.
Funding
The work was funded by a Newcastle University SAgE Faculty Research Fellowship to N.A. as well as Office of Naval Research Award N00014-16-1-3125 to A. S. Clare and N.A.
References
- 1.Martins GM, Neto AI, Cacabelos E. 2016. Ecology of a key ecosystem engineer on hard coastal infrastructure and natural rocky shores. Mar. Environ. Res. 113, 88–94. ( 10.1016/j.marenvres.2015.11.013) [DOI] [PubMed] [Google Scholar]
- 2.Davidson I, Scianni C, Hewitt CL, Holm E, Tanburri M, Ruiz G. 2016. Mini-review: assessing the drivers of ship biofouling management—aligning industry and biosecurity goals. Biofouling 32, 411–428. ( 10.1080/08927014.2016.1149572) [DOI] [PubMed] [Google Scholar]
- 3.Aldred N, Clare AS. 2008. The adhesive strategies of cyprids and development of barnacle-resistant marine coatings. Biofouling 24, 351–363. ( 10.1080/08927010802256117) [DOI] [PubMed] [Google Scholar]
- 4.Shikuma NJ, Pilhofer M, Weiss GL, Hadfield MG, Jensen GJ, Newman DK. 2014. Marine tubeworm metamorphosis induced by arrays of bacterial phage tail-like structures. Science 31, 529–533. ( 10.1126/science.1246794) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shikuma NJ, Antoshechkin I, Medeiros JM, Pilhofer M, Newman DK. 2016. Stepwise metamorphosis of the tubeworm Hydroides elegans is mediated by a bacterial inducer and MAPK signaling. Proc. Natl Acad. Sci. USA 113, 10 097–10 102. ( 10.1073/pnas.1603142113) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Joint I, Tait K, Callow ME, Callow JA, Milton D, Williams P, Camara M. 2002. Cell-to-cell communication across the prokaryote–eukaryote boundary. Science 298, 1207 ( 10.1126/science.1077075) [DOI] [PubMed] [Google Scholar]
- 7.Joint I, Tait K, Wheeler G. 2007. Cross-kingdom signaling: exploitation of bacterial quorum sensing molecules by the green seaweed Ulva. Phil. Trans. R Soc. B. 362, 1223–1233. ( 10.1098/rstb.2007.2047) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Freckelton ML, Nedved BT, Hadfield MG. 2017. Induction of invertebrate larval settlement; different bacteria, different mechanisms? Sci. Rep. 7, 42557 ( 10.1038/srep42557) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maki JS, Rittschof D, Costlow JD, Mitchell R. 1988. Inhibition of attachment of larval barnacles, Balanus amphitrite, by bacterial surface films. Mar. Biol. 97, 199–206. ( 10.1007/BF00391303) [DOI] [Google Scholar]
- 10.Neal AL, Yule AB. 1994. The interaction between Elminius modestus Darwin cyprids and biofilms of Deleya marina NCMB 1877. J. Exp. Mar. Biol. Ecol. 176, 127–139. ( 10.1016/0022-0981(94)90201-1) [DOI] [Google Scholar]
- 11.Wieczorek SK, Clare AS, Todd CD. 1995. Inhibitory and facilitory effects of microbial films on settlement of Balanus amphitrite amphitrite larvae. Mar. Ecol. Prog. Ser. 119, 221–228. ( 10.3354/meps119221) [DOI] [Google Scholar]
- 12.Olivier F, Tremblay R, Bourget E, Rittschof D. 2000. Barnacle settlement: field experiments on the influence of larval supply, tidal level, biofilm quality and age on Balanus amphitrite cyprids. Mar. Ecol. Prog. Ser. 199, 185–204. ( 10.3354/meps199185) [DOI] [Google Scholar]
- 13.Khandeparker L, Anil AC, Raghukumar S. 2006. Relevance of biofilm bacteria in modulating the larval metamorphosis of Balanus amphitrite. FEMS Microbiol. Ecol. 58, 425–438. ( 10.1111/j.1574-6941.2006.00177.x) [DOI] [PubMed] [Google Scholar]
- 14.Bacchetti De Gregoris T, Khandeparker L, Anil AC, Mesbahi E, Burgess JG, Clare AS.. 2012. Characterisation of the bacteria associated with barnacle, Balanus amphitrite, shell and their role in gregarious settlement of cypris larvae. J. Exp. Mar. Biol. Ecol. 413, 7–12. ( 10.1016/j.jembe.2011.11.014) [DOI] [Google Scholar]
- 15.Essock-Burns T, Gohad NV, Orihuela B, Mount AS, Spillmann CM, Wahl KJ, Rittschof D. 2016. Barnacle biology before, during and after settlement and metamorphosis: a study of the interface. J. Exp. Biol. 220, 194–207. ( 10.1242/jeb.145094) [DOI] [PubMed] [Google Scholar]
- 16.Fears KP, Orihuela B, Rittschof D, Wahl KJ. 2018. Acorn barnacles secrete phase-separating fluid to clear surfaces ahead of cement deposition. Adv. Sci. 5, 1700762 ( 10.1002/advs.201700762) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marzinelli EM, Qiu Z, Dafforn KA, Johnston EL, Steinberg PD, Mayer-Pinto M.. 2018. Coastal urbanisation affects microbial communities on a dominant marine holobiont. Biofilms Microb. 4, 1 ( 10.1038/s41522-017-0044-z) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Campbell AH, Marzinelli EM, Gelber J, Steinberg PD. 2015. Spatial variability of microbial assemblages associated with a dominant habitat-forming seaweed. Front. Microbiol. 6, 230 ( 10.3389/fmicb.2015.00230) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Aldred N, Nelson A. 2019. Data from: Microbiome acquisition during larval settlement of the barnacle Semibalanus balanoides. Dryad Digital Repository ( 10.5061/dryad.85g65v8) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Aldred N, Nelson A. 2019. Data from: Microbiome acquisition during larval settlement of the barnacle Semibalanus balanoides. Dryad Digital Repository ( 10.5061/dryad.85g65v8) [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Raw data are available in electronic supplementary material, SUP2 and Dryad Digital Repository: https://doi.org/10.5061/dryad.85g65v8 [19].