Abstract
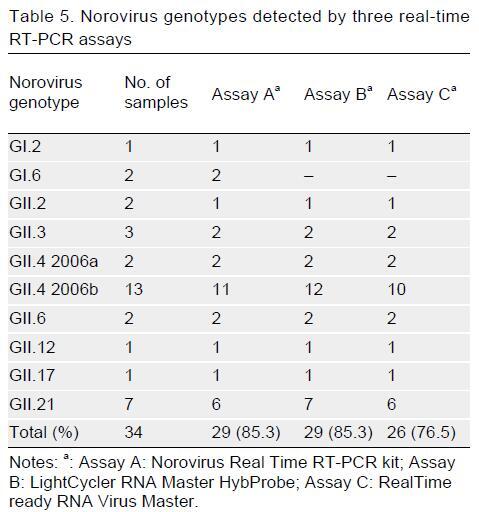
Noroviruses are the leading cause of acute gastroenteritis in humans. Real-time reverse transcription-polymerase chain reaction (real-time RT-PCR) is a promising molecular method for the detection of noroviruses. In this study, the performance of three TaqMan real-time RT-PCR assays was assessed, which were one commercially available real-time RT-PCR kit (assay A: Norovirus Real Time RT-PCR kit) and two in-house real-time RT-PCR assays (assay B: LightCycler RNA Master Hybprobe and assay C: RealTime ready RNA Virus Master). Assays A and B showed higher sensitivity than assay C for norovirus GI, while they all had the same sensitivity (103 DNA copies/mL) for GII DNA standard controls. Assay B had the highest efficiency for both genogroups. No cross-reactivity was observed among GI and GII noroviruses, rotavirus, hepatitis A virus, and poliovirus. The detection rates of these assays in GI and GII norovirus-positive fecal samples were not significantly different. However, the mean quantification cycle (Cq) value of assay B for GII was lower than assays A and C with statistical significance (P-value, 0.000). All three real-time RT-PCR assays could detect a variety of noroviruses including GI.2, GII.2, GII.3, GII.4, GII.6, GII.12, GII.17, and GII.21. This study suggests assay B as a suitable assay for the detection and quantification of noroviruses GI and GII due to good analytical sensitivity and higher performance to amplify norovirus on DNA standard controls and clinical samples.
Keywords: norovirus, genogroup, real-time RT-PCR, quantification
Acknowledgments
This work was supported by research grant from the Thailand Research Fund (TRF) through the Royal Golden Jubilee Ph.D. program (Grant No. PHD/0085/2554), and the Thai Government Budget through Mahidol University, fiscal year 2015‒2017. The authors thank The Language Center, Faculty of Graduate Studies, Mahidol University for editorial assistance.
Footnotes
ORCID: 0000-0003-3519-7862
References
- Aw TG, Gin K E, Oon LL, Chen EX, Woo CH. Prevalence and genotypes of human noroviruses in tropical urban surface waters and clinical samples in Singapore. Appl Environ Microbiol. 2009;75:4984–4992. doi: 10.1128/AEM.00489-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baert L, Uyttendaele M, Debevere J. Evaluation of two viral extraction methods for the detection of human noroviruses in shellfish with conventional and real-time reverse transcriptase PCR. Lett Appl Microbiol. 2007;44:106–111. doi: 10.1111/j.1472-765X.2006.02047.x. [DOI] [PubMed] [Google Scholar]
- Bok K, Abente EJ, Realpe-Quintero M, Mitra T, Sosnovtsev SV, Kapikian AZ, Green KY. Evolutionary dynamics of GII.4 noroviruses over a 34-year period. J Virol. 2009;83:11890–11901. doi: 10.1128/JVI.00864-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bull RA, Tanaka MM, White PA. Norovirus recombination. J Gen Virol. 2007;88:3347–3359. doi: 10.1099/vir.0.83321-0. [DOI] [PubMed] [Google Scholar]
- Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem. 2009;55:611–622. doi: 10.1373/clinchem.2008.112797. [DOI] [PubMed] [Google Scholar]
- Butot S, Le Guyader FS, Krol J, Putallaz T, Amoroso R, Sanchez G. Evaluation of various real-time RT-PCR assays for the detection and quantitation of human norovirus. J Virol Methods. 2010;167:90–94. doi: 10.1016/j.jviromet.2010.03.018. [DOI] [PubMed] [Google Scholar]
- Costafreda MI, Bosch A, Pintó RM. Development, evaluation, and standardization of a real-time TaqMan reverse transcription-PCR assay for quantification of hepatitis A virus in clinical and shellfish samples. Appl Environ Microbiol. 2006;72:3846–3855. doi: 10.1128/AEM.02660-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunbar NL, Bruggink LD, Marshall JA. Evaluation of the RIDAGENE real-time PCR assay for the detection of GI and GII norovirus. Diagn Microbiol Infect Dis. 2014;79:317–321. doi: 10.1016/j.diagmicrobio.2014.03.017. [DOI] [PubMed] [Google Scholar]
- Espy MJ, Uhl JR, Sloan LM, Buckwalter SP, Jones MF, Vetter EA, Yao JD, Wengenack NL, Rosenblatt J E, Cockerill F R 3, Smith T F. Real-time PCR in clinical microbiology: applications for routine laboratory testing. Clin Microbiol Rev. 2006;19:165–256. doi: 10.1128/CMR.19.1.165-256.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farkas T, Singh A, Le Guyader FS, La Rosa G, Saif L, McNeal M. Multiplex real-time RT-PCR for the simultaneous detection and quantification of GI, GII and GIV noroviruses. J Virol Methods. 2015;223:109–114. doi: 10.1016/j.jviromet.2015.07.020. [DOI] [PubMed] [Google Scholar]
- Fuentes C, Guix S, Pérez-Rodriguez FJ, Fuster N, Carol M, Pintó RM, Bosch A. Standardized multiplex one-step qRT-PCR for hepatitis A virus, norovirus GI and GII quantification in bivalve mollusks and water. Food Microbiol. 2014;40:55–63. doi: 10.1016/j.fm.2013.12.003. [DOI] [PubMed] [Google Scholar]
- Green KY. Caliciviridae: the noroviruses. In: Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B, Straus SE, editors. Fields Virology. 2013. pp. 582–608. [Google Scholar]
- Hall AJ, Wikswo ME, Manikonda K, Roberts VA, Yoder JS, Gould LH. Acute gastroenteritis surveillance through the national outbreak reporting system, United States. Emerg Infect Dis. 2013;19:1305–1309. doi: 10.3201/eid1908.130482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyun J, Kim HS, Kim HS, Lee KM. Evaluation of a new real-time reverse transcription polymerase chain reaction assay for detection of norovirus in fecal specimens. Diagn Microbiol Infect Dis. 2014;78:40–44. doi: 10.1016/j.diagmicrobio.2013.09.013. [DOI] [PubMed] [Google Scholar]
- Kageyama T, Kojima S, Shinohara M, Uchida K, Fukushi S, Hoshino FB, Takeda N, Katayama K. Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. J Clin Microbiol. 2003;41:1548–1557. doi: 10.1128/JCM.41.4.1548-1557.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kittigul L, Pombubpa K, Taweekate Y, Diraphat P, Sujirarat D, Khamrin P, Ushijima H. Norovirus GII-4 2006b variant circulating in patients with acute gastroenteritis in Thailand during a 2006–2007 study. J Med Virol. 2010;82:854–860. doi: 10.1002/jmv.21746. [DOI] [PubMed] [Google Scholar]
- Kittigul L, Panjangampatthana A, Pombubpa K, Taweekate Y, Pungchitton S, Diraphat P, Siripanichgon K. Detection and genetic characterization of norovirus in environmental water samples in Thailand. Southeast Asian J Trop Med Public Health. 2012;43:323–332. [PubMed] [Google Scholar]
- Kittigul L, Thamjaroen A, Chiawchan S, Chavalitshewinkoon-Petmitr P, Pombubpa K, Diraphat P. Prevalence and molecular genotyping of noroviruses in market oysters, mussels, and cockles in Bangkok, Thailand. Food Environ Virol. 2016;8:133–140. doi: 10.1007/s12560-016-9228-6. [DOI] [PubMed] [Google Scholar]
- Lees D, CEN WG6 TAG4. International standardisation of a method for detection of human pathogenic viruses in molluscan shellfish. Food Environ Virol. 2010;2:146–155. doi: 10.1007/s12560-010-9042-5. [DOI] [Google Scholar]
- Loisy F, Atmar RL, Guillon P, Le Cann P, Pommepuy M L, Guyader FS. Real-time RT-PCR for norovirus screening in shellfish. J Virol Methods. 2005;123:1–7. doi: 10.1016/j.jviromet.2004.08.023. [DOI] [PubMed] [Google Scholar]
- Lu J, Fang L, Zheng H, Lao J, Yang F, Sun L, Xiao J, Lin J, Song T, Ni T, Raghwani J, Ke C, Faria NR, Bowden TA, Pybus OG, Li H. The evolution and transmission of epidemic GII.17 noroviruses. J Infect Dis. 2016;214:556–64. doi: 10.1093/infdis/jiw208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathijs E, Stals A, Baert L, Botteldoorn N, Denayer S, Mauroy A, Scipioni A, Daube G, Dierick K, Herman L, Van Coillie E, Uyttendaele M, Thiry E. A review of known and hypothetical transmission routes for noroviruses. Food Environ Virol. 2012;4:131–152. doi: 10.1007/s12560-012-9091-z. [DOI] [PubMed] [Google Scholar]
- Mattison K, Grudeski E, Auk B, Brassard J, Charest H, Dust K, Gubbay J, Hatchette TF, Houde A, Jean J, Jones T, Lee BE, Mamiya H, McDonald R, Mykytczuk O, Pang X, Petrich A, Plante D, Ritchie G, Wong J, Booth TF. Analytical performance of norovirus real-time RT-PCR detection protocols in Canadian laboratories. J Clin Virol. 2011;50:109–113. doi: 10.1016/j.jcv.2010.10.008. [DOI] [PubMed] [Google Scholar]
- Myers TW, Gelfand DH. Reverse transcription and DNA amplification by a Thermus thermophilus DNA polymerase. Biochemistry. 1991;30:7661–7666. doi: 10.1021/bi00245a001. [DOI] [PubMed] [Google Scholar]
- Neesanant P, Sirinarumitr T, Chantakru S, Boonyaprakob U, Chuwongkomon K, Bodhidatta L, Sethabutr O, Abente EJ, Supawat K, Mason CJ. Optimization of one-step real-time reverse transcription-polymerase chain reaction assays for norovirus detection and molecular epidemiology of noroviruses in Thailand. J Virol Methods. 2013;194:317–325. doi: 10.1016/j.jviromet.2013.08.033. [DOI] [PubMed] [Google Scholar]
- Patel MM, Widdowson MA, Glass RI, Akazawa K, Vinje J, Parashar UD. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg Infect Dis. 2008;14:1224–1231. doi: 10.3201/eid1408.071114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pang X, Lee BE. Laboratory diagnosis of noroviruses: present and future. Clin Lab Med. 2015;35:345–362. doi: 10.1016/j.cll.2015.02.008. [DOI] [PubMed] [Google Scholar]
- Poddar SK, Sawyer MH, Connor JD. Effect of inhibitors in clinical specimens on Taq and Tth DNA polymerase-based PCR amplification of influenza A virus. J Med Microbiol. 1998;47:1131–1135. doi: 10.1099/00222615-47-12-1131. [DOI] [PubMed] [Google Scholar]
- Schultz AC, Vega E, Dalsgaard A, Christensen LS, Norrung B, Hoorfar J, Vinje J. Development and evaluation of novel one-step TaqMan realtime RT-PCR assays for the detection and direct genotyping of genogroup I and II noroviruses. J Clin Virol. 2011;50:230–234. doi: 10.1016/j.jcv.2010.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siebenga JJ, Vennema H, Zheng DP, Vinjé J, Lee BE, Pang XL, Ho EC, Lim W, Choudekar A, Broor S, Halperin T, Rasool NB, Hewitt J, Greening GE, Jin M, Duan ZJ, Lucero Y, O’Ryan M, Hoehne M, Schreier E, Ratcliff RM, White PA, Iritani N, Reuter G, Koopmans M. Norovirus illness is a global problem: emergence and spread of norovirus GII.4 variants, 2001-2007. J Infect Dis. 2009;200:802–812. doi: 10.1086/605127. [DOI] [PubMed] [Google Scholar]
- Stals A, Uyttendaele M, Van Coillie E. The need for harmonization in detection of human noroviruses in food. J AOAC Int. 2013;96:998–1005. doi: 10.5740/jaoacint.13-020. [DOI] [PubMed] [Google Scholar]
- Taylor S, Wakem M, Dijkman G, Alsarraj M, Nguyen M. A practical approach to RT-qPCR-publishing data that conform to the MIQE guidelines. Methods. 2010;50:S1–S5. doi: 10.1016/j.ymeth.2010.01.005. [DOI] [PubMed] [Google Scholar]
- Van Stelten A, Kreman TM, Hall N, Desjardin LE. Optimization of a real-time RT-PCR assay reveals an increase of genogroup I norovirus in the clinical setting. J Virol Methods. 2011;175:80–84. doi: 10.1016/j.jviromet.2011.04.022. [DOI] [PubMed] [Google Scholar]
- Vinjé J. Advances in laboratory methods for detection and typing of norovirus. J Clin Microbiol. 2015;53:373–381. doi: 10.1128/JCM.01535-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wenzel JJ, Panning M, Kaul KL, Mangold KA, Revell PA, Luna RA, Zepeda H, Perea L, Vazquez-Perez JA, Young S, Rodic-Polic B, Eickmann M, Drosten C, Jilg W, Reischl U. Analytical performance determination and clinical validation of the novel Roche RealTime Ready Influenza A/H1N1 Detection Set. J Clin Microbiol. 2010;48:3088–3094. doi: 10.1128/JCM.00785-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Y, Wang HH, Gao L, Ji JM, Ge ZJ, Zhu XQ, He PY, Chen ZW. A one-step multiplex real-time RT-PCR assay for rapid and simultaneous detection of human norovirus genogroup I, II and IV. J Virol Methods. 2013;189:277–282. doi: 10.1016/j.jviromet.2013.02.004. [DOI] [PubMed] [Google Scholar]


