In a randomized antiretroviral therapy initiation trial, raltegravir, darunavir/ritonavir, and atazanavir/ritonavir demonstrated similar changes in gut epithelial and microbial translocation products. Lower baseline gut dysfunction was independently associated with increased body mass index and visceral adipose tissue over 96 weeks.
Keywords: gut integrity, microbial translocation, body composition
Abstract
Background
Fat accumulation and insulin resistance remain a threat to the success of antiretroviral therapy (ART). The role of gut dysfunction in metabolic complications associated with ART initiation is unclear.
Methods
Human immunodeficiency virus (HIV)-infected ART-naive participants were randomized to tenofovir disoproxil fumarate/emtricitabine plus atazanavir/ritonavir, darunavir/ritonavir, or raltegravir (RAL). Changes in the gut integrity markers zonulin, lipopolysaccharide-binding protein (LBP), and intestinal fatty acid and ileal bile acid binding proteins (I-FABP and I-BABP) were assessed over 96 weeks. Wilcoxon rank-sum tests were used to compare changes between groups and linear regression models to quantify associations between gut markers, insulin resistance, body mass index (BMI), and visceral, subcutaneous, and total adipose tissue (VAT, SAT, and TAT).
Results
: 90% were male and 48% were White non-Hispanic. The median age was 36 years, HIV-1 ribonucleic acid was 4.56 log10 copies/mL, and CD4 count was 338 cells/µL. An overall 1.7-fold increase in I-FABP was observed throughout 96 weeks, with no difference between arms. Zonulin levels increased with RAL compared to protease inhibitor–based regimens (week 96, P = .02); minimal changes in I-BABP or LBP levels were observed. Higher baseline I-FABP levels were associated with increases in VAT, TAT, and BMI (16%, 9%, and 2.5%, respectively; P < .04) over 96 weeks.
Conclusions
While ART induces changes in the markers of gut barrier dysfunction, the extent to which they improve or worsen the gut barrier function remains unclear. Nevertheless, markers of gut barrier dysfunction in ART-naive individuals predict increases in total and visceral abdominal fat with treatment initiation.
Despite advances in antiretroviral therapy (ART), metabolic complications remain highly prevalent among human immunodeficiency virus (HIV)-infected individuals and have become a substantial threat to treatment success [1]. Initially thought to be linked to the use of protease inhibitors (PI), recent studies have shown that participants initiating therapy with the integrase inhibitor raltegravir (RAL) experienced the same extent of fat gain and insulin resistance as compared to participants initiating PI-based therapies [2, 3]. Although the exact mechanisms leading to these metabolic complications remain unclear [4], HIV-associated inflammation and immune activation likely play a role, with markers of immune activation directly associated with visceral fat accumulation and the incidence of diabetes among treated individuals [5, 6]. The triggering factor linking inflammation to these metabolic complications remains unclear.
Residual inflammation and immune activation while on suppressive ART remain a challenge in the management of HIV-infected individuals [7]. In the past decade, alteration of the gut barrier integrity and subsequent microbial translocation were shown to be drivers of this inflammatory state [8]. In the general population, increased microbial translocation products from low-level inflammatory conditions, such as obesity or diabetes, may play a role in the etiology of these metabolic syndromes [9, 10]. While inflammatory markers have been linked to an increased incidence of diabetes in treated HIV infections [6], the contributions of specific markers of gut epithelial barrier dysfunction and microbial translocation have yet to be explored.
Several markers are used to assess gut wall integrity among patients with HIV, including lipopolysaccharide-binding protein (LBP), an indirect marker of microbial translocation [11]; zonulin, a marker of intestinal permeability [12]; and intestinal fatty acid binding proteins (I-FABP) and ileal bile acid binding proteins (I-BABP), markers of enterocyte damage [13]. I-FABP and I-BABP differ in their localization within the intestines; I-FABP can be found either in the small or large intestine, while I-BABP is exclusively present in the small intestine [14].
The impact of contemporary ART regimens and their differential effects on gut integrity markers and microbial translocation in the chronic phase of untreated HIV is unknown; only a few studies have looked at changes in these gut markers [15, 16].
The objectives of our study were (1) to assess changes in gut integrity markers after the initiation of ART by administering tenofovir disoproxil fumarate/emtricitabine plus either atazanavir/ritonavir (ATV/r), darunavir/ritonavir (DRV/r), or RAL in treatment-naive HIV-infected participants and determine whether there are differences among the 3 regimens; (2) to assess whether baseline gut integrity markers are predictive of increases in visceral and overall adiposity and insulin resistance with newer ART agents; and (3) to assess whether gut integrity markers before or after ART initiation are associated with markers of inflammation and immune activation. Our hypotheses were (1) that gut integrity markers would decrease over time, regardless of regimen used and without differences between regimens; (2) that baseline and changes in gut integrity markers after ART would be predictive of an increase in visceral and overall adiposity and insulin resistance; and (3) that increased gut integrity markers before and after ART initiation would be associated with increased markers of inflammation and immune activation. The aims of this study were assessed utilizing AIDS Clinical Trials Group Study A5260s, the cardiometabolic substudy of A5257.
METHODS
Study Population
In the parent study A5257, HIV-infected treatment-naive participants aged ≥18 years with HIV-1 ribuonucleic acid (RNA) ≥1000 copies/mL were randomized in an open-labeled fashion to receive tenofovir disoproxil fumarate/emtricitabine plus either ATV/r, DRV/r, or RAL. Participants without known cardiovascular disease, diabetes mellitus, uncontrolled thyroid disease, or use of lipid-lowering medications were enrolled in A5260s. Further details have been previously published [2].
The analysis population consisted of a subset of participants who remained on their randomization treatment, achieved virological suppression (HIV-1 RNA <50 copies/mL) by week 24, remained suppressed through week 96, and did not interrupt treatment for more than 7 days. The parent study and substudy (NCT00811954 and NCT00851799) were approved by the institutional review boards at all participating institutions sites, and all participants provided informed written consent.
Study Evaluations
Fasting (≥8 hours) blood samples were collected from all participants at baseline and study weeks 4, 24, and 96, and plasma samples were stored at -70°C until analyzed in batches. Levels of LBP and I-FABP (R&D Systems, Minneapolis, MN), I-BABP (Cloud Clone, Katy, TX), and zonulin (ALPCO, Salem, NH) were measured by enzyme-linked immunosorbent assays in the Dahms Clinical Research Unit at University Hospitals Cleveland Medical Center.
Markers of inflammation were high-sensitivity C-reactive protein, D-dimer, soluble clusters of differentiation 14 and 163 (sCD14 and sCD163), and interleukin-6, which were measured as previously described [17].
Peripheral blood mononuclear cells were collected, cryopreserved, and assessed as previously described [14]. T-cell activation was identified as the percentage of CD4+ or CD8+ cells expressing both human leukocyte antigen-D related and CD38 cells. Monocyte subsets were characterized as inflammatory (CD14+CD16+), patrolling (CD14dimCD16+), and classic (CD14+CD16-).
Trunk and total limb fat, visceral adipose tissue (VAT), subcutaneous abdominal tissue (SAT), and total adipose tissue (TAT) were evaluated at baseline and week 96, as previously detailed [2]. The homeostasis model assessment-insulin resistance (HOMA-IR) index was used to estimate the insulin resistance (HOMA-IR >2.5) defined insulin resistance [18].
Statistical Analysis
Non-normal biomarkers were transformed to the log10 scale for analysis. Absolute levels and changes in gut integrity markers across study weeks were calculated for each treatment group; mean fold-changes in biomarkers were determined by calculating mean differences on the log-scale and then were back-transformed for ease of interpretation. Changes in biomarkers were contrasted between treatment groups with Wilcoxon rank-sum tests. Associations among gut integrity markers and with markers of immune activation and inflammation were examined with Spearman’s rank-based correlations. Associations between measures of body composition and markers of insulin resistance and gut integrity were assessed with linear regression models, with adjustments for demographic factors, HIV-related variables, physical activity, smoking, alcohol use, and drug status. All treatment group comparisons were done pairwise between each of the 3 groups using a 2-sided 2.5% type I error rate; additional statistical tests used 2-sided 5% type I error rates.
RESULTS
Baseline Characteristics
Overall, 231 participants were included in this analysis; 67 participants were randomized to ATV/r, 82 to DRV/r, and 82 to RAL. Overall, 90% were male; 48% were White non-Hispanic, and 19% were Hispanic. The median age was 36 years, median baseline CD4+ cell count was 338 cells/µL, and median HIV-1 RNA was 4.56 log10 copies/mL. Baseline levels of all biomarkers and body composition parameters are presented in Table 1, and did not differ between treatment groups.
Table 1.
Baseline Characteristics by Randomized Treatment Group
| Characteristic | Total (N = 231) |
Atazanavir-Ritonavir (n = 67) |
Raltegravir (n = 82) |
Darunavir-Ritonavir (n = 82) |
|---|---|---|---|---|
| Age, y | 36 (28, 45) | 38 (31, 45) | 36 (27, 45) | 36 (28, 45) |
| Sex, male | 208 (90%) | 63 (94%) | 73 (89%) | 72 (88%) |
| Race/ethnicity | ||||
| White | 110 (48%) | 35 (52%) | 36 (44%) | 39 (48%) |
| Black | 68 (29%) | 21 (31%) | 23 (28%) | 24 (29%) |
| Hispanic | 44 (19%) | 10 (15%) | 16 (20%) | 18 (22%) |
| Smoking history | 127 (55%) | 38 (57%) | 45 (55%) | 44 (54%) |
| CD4 + cell count, cells/mm3 | 338 (190, 450) | 296 (169, 461) | 347 (246, 450) | 337 (171, 425) |
| HIV-1 RNA, log10copies/mL | 4.6 (4.0, 5.0) | 4.8 (4.0, 5.2) | 4.5 (4.0, 4.9) | 4.6 (4.0, 4.9) |
| Body mass index, kg/m2 | 24.9 (22.5, 28.3) | 25.9 (22.7, 28.7) | 24.9 (22.3, 27.8) | 24.5 (22.1, 27.4) |
| Visceral adipose tissue, cm2 | 74.1 (41.6, 110.1) | 76.6 (41.6, 111.3) | 78.0 (41.6, 111.9) | 67.3 (38.8, 105.1) |
| Trunk fat, cm2 | 9.4 (6.1, 12.8) | 9.7 (6.8, 12.9) | 9.9 (6.1, 13.3) | 9.0 (5.9, 11.9) |
| HOMA-IR, log10 | -0.22 (-0.40, 0.14) | -0.21 (-0.40, 0.14) | -0.22 (-0.39, 0.14) | -0.33 (-0.40, 0.12) |
| HOMA-IR | 0.60 (0.40, 1.38) | 0.62 (0.40, 1.38) | 0.60 (0.41, 1.38) | 0.47 (0.40, 1.32) |
| LBP, ng/mL | 1.32 (1.25, 1.43) | 1.33 (1.25, 1.42) | 1.31 (1.24, 1.43) | 1.32 (1.25,1.44) |
| Zonulin, mg/mL | 1.2 (1.11, 1.31) | 1.20 (1.10, 1.28) | 1.21 (1.13, 1.30) | 1.21 (1.11,1.32) |
| I-FABP, pg/mL | 3.27 (3.14, 3.41) | 3.26 (3.16, 3.43) | 3.30 (3.13, 3.41) | 3.25 (3.15,3.39) |
| I-BABP, pg/mL | 2.74 (2.55, 2.96) | 2.68 (2.54, 3.00) | 2.77 (2.56, 3.00) | 2.74 (2.56,2.94) |
Median (first and third quartiles) or number (%).
Abbreviations: HIV, human immunodeficiency virus; HOMA-IR, homeostatic model assessment–insulin resistance; I-BABP, ileal bile acid binding proteins; I-FABP, intestinal fatty acid binding proteins; LBP, lipopolysaccharide-binding protein; RNA, ribonucleic acid.
Changes in Gut Integrity Markers
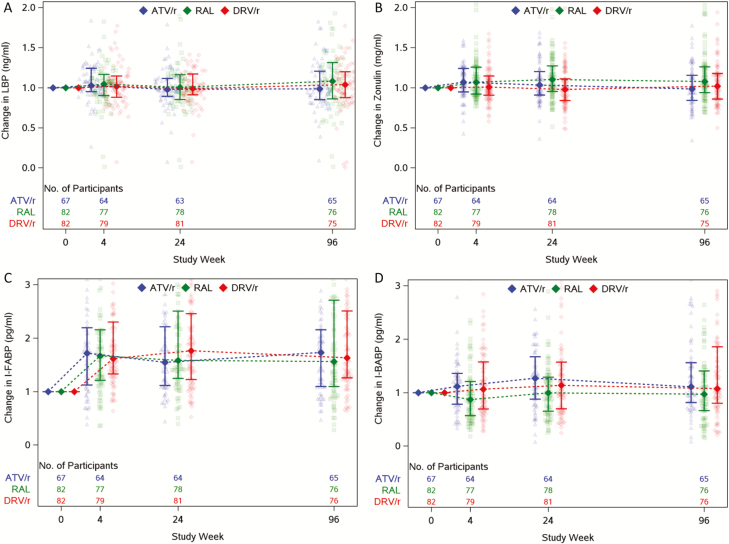
Overall, I-FABP levels increased from baseline to week 4 (mean 1.7-fold change; 95% CI 1.58–1.83), were sustained through week 96 (mean: 1.65 to 1.71), and were consistent for all treatment groups. The increase in I-FABP levels did not differ among the 3 treatment groups at any time point (P > .20).
Overall, zonulin levels modestly increased by week 4 (mean 1.06-fold change; 95% CI 1.02–1.09), with consistent effects observed to 24 and 96 weeks. However, when examining changes within treatment groups, increases were only observed in the RAL treatment group at all time points (mean fold change: 1.09 to 1.12). Zonulin changes were higher in RAL compared to DRV/r at week 24 (relative mean fold change: 1.13; P < .01). Compared to ATV/r, changes in RAL were larger at week 96 (relative mean fold change: 1.14; P = .02) but were not observed at earlier time points.
Although not statistically significant, noteworthy changes in I-BABP levels were observed at weeks 24 and 96 in both protease inhibitors/ritonavir (PI/r) arms (mean fold change: 1.12 to 1.16); slight decreases were observed in the RAL arm at week 4, but they were not sustained. There were statistically significant differences between groups with regard to changes in I-BABP, with larger changes in ATV/r compared to RAL at weeks 4 and 24 (relative mean fold change: 1.22; P < .02) and larger increases in DRV/r compared to RAL at week 4 (relative mean fold change: 1.18; P = .05).
No changes in LBP levels were seen throughout the study period, and no differences were observed among the 3 groups at any time point (all P > .2; Figure 1).
Figure 1.
Changes in gut integrity markers over time by treatment arm. Median fold change in gut integrity markers from baseline by treatment arm. Abbreviations: ATV/r, atazanavir-ritonavir; DRV/r, darunavir/ritonavir; I-BABP, ileal bile-acid binding proteins; I-FABP, intestinal fatty acid binding proteins; LBP, lipopolysaccharide-binding protein; RAL, raltegravir.
Associations Between Baseline Gut Markers With Measures of Body Composition and Insulin Resistance
Linear regression models examined the relationships between baseline gut marker levels and changes in body composition over 96 weeks (for separate models for percentage changes in BMI, VAT, SAT, TAT, and trunk and total fat, see Table 2). In unadjusted models, pre-ART I-FABP levels were significantly associated with percentage changes in body composition over 96 weeks; 2-fold higher I-FABP levels were associated with a 16% increase in VAT, 9% increase in TAT, and 2.5% increase in BMI. The magnitude of these associations remained unchanged after adjusting for clinically relevant factors. Other gut markers were not associated with changes in body composition measures (all P > .06). No associations were found between gut markers and homeostatic model assessment–insulin resistance (all P > .05).
Table 2.
Regression Estimates for Baseline I-FABPa With 96-Week Changes in Body Composition Parameters, Adjusting for Clinically Relevant Factors
| BMI | Visceral Adipose Tissue | Subcutaneous Adipose Tissue | Total Adipose Tissue | Trunk Fat | Total Fat | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Estimateb | P-Value | Estimateb | P-Value | Estimateb | P-Value | Estimateb | P-Value | Estimateb | P-Value | Estimateb | P-Value | |
| Unadjusted I-FABP | 2.5 (0.8–4.2) |
.004 | 15.9 (4.5–27.2) | .006 | 5.6 (-3.2–14.4) |
.216 | 8.6 (0.3–17.0) |
.043 | 2.7 (-4.4– 9.9) |
.456 | 4.4 (-1.7–10.5) |
.155 |
| Adjustment | ||||||||||||
| Age | 2.7 (1.0– 4.5) |
.002 | 17.0 (5.4– 28.6) |
.004 | 6.9 (-2.0–15.8) |
.131 | 9.9 (1.4–18.4) |
.022 | 4.2 (-3.1– 11.5) |
.259 | 5.4 (-0.8– 11.7) |
.087 |
| Sex | 2.6 (0.9– 4.3) |
.003 | 16.0 (4.7– 27.3) |
.006 | 5.8 (-2.8– 14.4) |
.188 | 8.8 (0.6–17.0) |
.035 | 3.1 (-4.0–10.2) |
.396 | 4.7 (-1.4– 10.7) |
.131 |
| Race/ethnicity | 2.1 (0.4– 3.9) |
.015 | 15.5 (3.8– 27.3) |
.009 | 4.2 (-4.8– 13.1) |
.361 | 7.5 (-1.0–16.1) |
.083 | 1.3 (-6.0– 8.6) |
.726 | 3.2 (-3.1– 9.4) |
.321 |
| Smoking history | 2.5 (0.8– 4.2) |
.004 | 16.4 (5.0– 27.8) |
.005 | 5.5 (-3.3– 14.3) |
.224 | 8.6 (0.3–17.0) |
.043 | 2.6 (-4.6– 9.8) |
.480 | 4.4 (-1.7– 10.5) |
.156 |
| Alcohol history | 2.6 (0.9– 4.2) |
.003 | 18.2 (7.3– 29.1) |
.001 | 6.8 (-1.9– 15.6) |
.125 | 9.9 (1.8– 18.1) |
.017 | 3.0 (-4.1– 10.2) |
.406 | 4.5 (-1.6– 10.6) |
.151 |
| Drug history | 2.6 (1.0– 4.3) |
.002 | 17.7 (7.0– 28.4) |
.001 | 6.3 (-2.3– 15.0) |
.150 | 9.6 (1.6–17.6) |
.019 | 3.1 (-3.9– 10.1) |
.385 | 4.7 (-1.3–10.7) |
.123 |
| Physical Activity | 2.4 (0.7– 4.1) |
.006 | 14.6 (3.9– 25.3) |
.008 | 4.5 (-4.4– 13.4) |
.320 | 7.6 (-0.8–16.0) |
.076 | 1.4 (-5.7– 8.6) |
.691 | 3.5 (-2.6– 9.6) |
.260 |
| CD4+ cell count | 1.5 (-0.1– 3.2) | .068 | 12.9 (1.4– 24.5) |
.028 | 3.7 (-5.2– 12.7) |
.414 | 6.5 (-1.9–15.0) |
.130 | -0.2 (-7.3– 6.9) |
.953 | 1.6 (-4.4– 7.6) |
.607 |
| HIV-1 RNA | 1.9 (0.3– 3.5) |
.022 | 12.5 (1.3– 23.6) |
.028 | 3.3 (-5.4– 12.0) |
.462 | 6.0 (-2.2–14.2) |
.150 | -0.0 (-6.8– 6.8) |
.997 | 2.1 (-3.7– 7.9) |
.482 |
| All covariates | 1.6 (0.0– 3.2) |
.046 | 14.7 (4.5– 24.8) |
.005 | 3.2 (-5.5– 11.9) |
.473 | 6.7 (-1.4–14.7) |
.106 | -0.9 (-7.8– 6.0) |
.801 | 0.9 (-5.1– 6.8) |
.773 |
Abbreviations: BMI, body mass index; HIV, human immunodeficiency virus; I-FABP, intestinal fatty acid binding proteins; RNA, ribonucleic acid.
aI-FABP level estimates are presented as per 0.3 log10 units, which is equivalent to a doubling of the marker.
b95% confidence interval.
Associations Between Gut Markers, Human Immunodeficiency Virus Variables, Inflammation, and Immune Activation
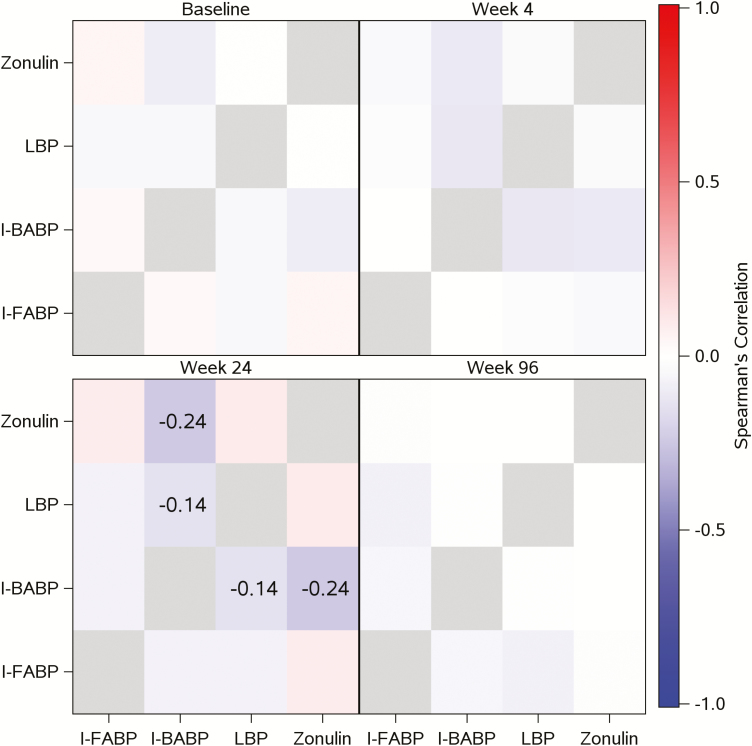
Associations among gut integrity markers were assessed at all study points and are presented in Figure 2.
Figure 2.
Associations among gut integrity markers at different study points. Heat map illustrating the associations between gut integrity markers at different time points. Blue indicates negative correlations, and red indicates positive correlations. Only correlations reaching statistical significance (P ≤ 0.05) are included in the figure. Abbreviations: I-BABP, ileal bile-acid binding proteins; I-FABP, intestinal fatty acid binding proteins; LBP, lipopolysaccharide-binding protein.
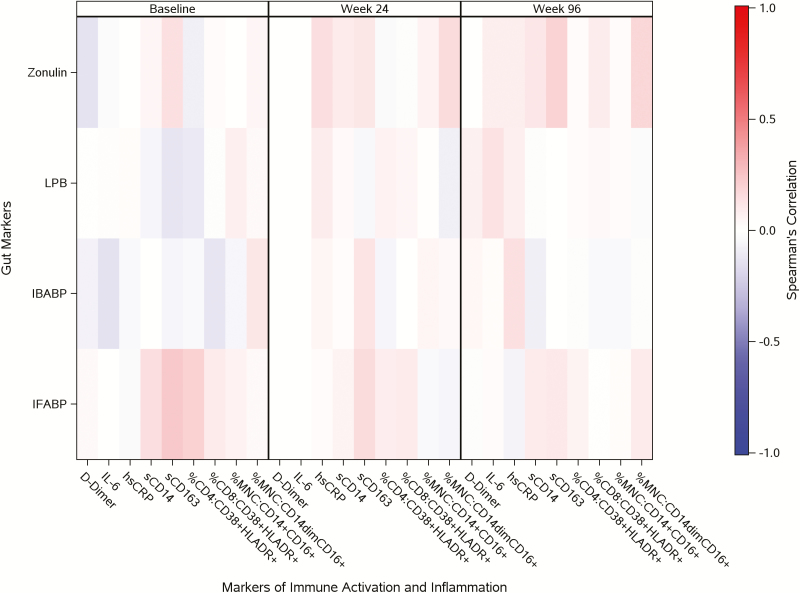
Pre-ART CD4+ T-cell counts were positively correlated with baseline to 96-week changes in I-FABP (rho = 0.18; P < .01).
As shown in Figure 3, pre-ART I-FABP levels were positively associated with sCD163, sCD14, and markers of T-cell activation (CD4+CD38+human leukocyte antigen-D related+T cells; all P < .02); these associations were sustained, but were weaker at on-treatment time points (at weeks 4, 24, and 96).
Figure 3.
Associations between gut integrity markers and markers of inflammation and immune activation. D-dimer and IL-6 were not assessed at week 24. Abbreviations: HLADR, human leukocyte antigen DR; hsCRP, high-sensitivity C-reactive protein; I-BABP, ileal bile-acid binding proteins; I-FABP, intestinal fatty acid binding proteins; IL, interleukin; LBP, lipopolysaccharide-binding protein; MNC, monocyte; sCD, soluble cluster of differentiation.
A modest association was observed between zonulin and sCD163 in treatment-naive participants, which persisted and somewhat strengthened on-treatment time points (all P < .02). After ART initiation, new associations were observed between zonulin with high-sensitivity C-reactive protein and inflammatory monocytes (CD14+CD16+; all P < 0.01). Associations between I-BABP and LBP levels with inflammatory and immune activation markers were weak and inconsistent across all time points.
DISCUSSION
In the context of a large, randomized clinical trial of ART initiation with tenofovir disoproxil fumarate/emtricitabine plus either ATV/r, DRV/r, or RAL, we evaluated the change in gut integrity markers and relationships between these markers and body composition, insulin resistance, inflammation, and immune activation in HIV-infected adults for the first time. Fat accumulation not only causes cosmetic concerns, but is also associated with an increased risk of inflammation, cardiovascular disease, and mortality [19, 20]. Identifying the factors associated with metabolic disturbances encountered in the aging HIV population is therefore important to the long-term management of HIV and could help direct interventions to reduce these ongoing complications. Interestingly, there appear to be large increases in I-FABP and smaller increases in I-BABP and zonulin after ART initiation. Additionally, LBP changes were not observed.
Despite the role of ART in controlling viral replication, gut integrity is not completely restored [21, 22]. In this study, we have shown that I-FABP levels increase after ART initiation without significant changes in LBP. There were 2 previous studies that examined the longitudinal effect of ART initiation on I-FABP and microbial translocation products. Similar to our findings, I-FABP increased in both studies; however, a decrease in microbial translocation products accompanied the rise in I-FABP [15, 16]. Since the impairment of the intestinal mucosal barrier is a prerequisite for microbial translocation [23], the unchanged LBP levels in our study could simply be due to LBP assay variability or to the decrease in sensitivity of this marker compared to more direct measures of microbial products.
In our results, the levels of I-FABP increased in the first 4 weeks of ART initiation and then plateaued throughout the rest of the study period; a similar pattern was also reported by Sereti et al. [15], where I-FABP increased after 2 weeks of ART initiation and then remained elevated for 96 weeks. These findings raise questions as to whether ART has a direct effect on the gut epithelium or whether the increase in I-FABP reflects the natural progression of gut barrier dysfunction among HIV-infected individuals.
Conflicting data exist on the role of I-FABP in predicting mortality in treated HIV-infected individuals [12, 24]. Although plasma I-FABP is frequently considered a marker of intestinal damage [13], we cannot exclude the possibility that the rise in I-FABP might be due to other events. In fact, I-FABP performs different functions within the same type of cell, which is incompletely understood [25, 26]. The increase in I-FABP with ART initiation might be due to the beneficial effect of ART on gut restoration, resulting in the regeneration of epithelial cells and consequent shedding of I-FABP. Indeed, a prior cross-sectional study suggested that gut epithelial barrier integrity, measured directly on gut biopsy tissue, is impaired in untreated HIV infections and associated with blunting of the intestinal villi length, both of which appeared to be largely restored during suppressive ART [27]. Increases in gut epithelial cell numbers may well account for the increases in plasma I-FABP levels, even if gut epithelial cell death is declining on a relative scale. Furthermore, the avid association between CD4+ T cells and I-FABP in our study suggests that participants with lower pre-ART CD4+ T-cell counts may have lower capacities for epithelial cell renewal, and thus less of a treatment-mediated I-FABP increase. Further longitudinal studies sampling intestinal mucosa after ART initiation are needed to confirm this hypothesis and to re-assess the role of the serum marker I-FABP.
Differential Effect of Antiretroviral Therapy Regimens
Conflicting data exist on the effect of different ART regimens on gut barrier structure and microbial translocation [15, 16, 28]. In a randomized trial of ART initiation, an efavirenz (EFV)-based regimen (n = 37) resulted in a higher increase in I-FABP levels compared to lopinavir/ritonavir (LPV/r)-based ART (n = 34) [16]. However, a recent, smaller cross-sectional study in Mexico found higher levels of plasma I-FABP with a PI-based regimen (ATV/r [n = 8] or LPV/r [n = 7]) compared to EFV-based therapy (n = 18) [28]. In both of these small studies, markers of microbial translocation did not differ between treatment groups [16, 28]. While some studies have suggested that older PIs (ritonavir and liponavir) induce endoplasmic reticulum stress and intestinal epithelial apoptosis [29], another study suggested increased oxidative stress–related apoptosis with EFV [30]. It remains unclear whether these previous findings reflect a direct effect of older ART regimens on the gut epithelium or are just a result of gut markers assay variability, heterogeneity, or small patient numbers of these cohorts.
For the first time, we compared the effect of RAL and ritonavir-boosted PIs (ATV and DRV) on gut mucosal integrity and microbial translocation. We found no differences in the plasma levels of I-FABP or LBP in the 3 treatment groups. Interestingly, however, zonulin increased in patients treated with RAL, but not in those treated with PIs. Consistent with these results, we have also previously shown a greater decline in sCD14 with an RAL arm compared to ATV/r or DRV/r [17]. Zonulin, a regulator of epithelial and endothelial functions, is known to increase gut permeability and macromolecule absorption by disassembling tight junctions [31]. Lower levels of zonulin were found to be associated with increased mortality among treated HIV-infected individuals [12], and zonulin, in its innate immune function, was suggested to be protective against HIV infection [32]. Thus, zonulin production may be actively suppressed as a consequence of a disrupted gut epithelial barrier or decreased simply as a consequence of gut epithelial cell death in untreated HIV infection, and then increased during ART as these processes are partially reversed. Consistent with our results, in a smaller study of combination therapy in treatment-naive participants, zonulin increased with RAL plus maraviroc (n = 8) therapy, but did not change with maraviroc (n = 10) or EFV (n = 7) therapy alone [33].
Altogether, these findings might suggest that RAL has a beneficial effect on gut permeability (coinciding with a decrease in microbial translocation) compared to ATV/r and DRV/r regimens. A possible mechanism includes increased RAL penetration into the gut (compared to PIs and non-nucleoside reverse transciptase inhibitor), promoting local control of viral replication, inflammation, and subsequent microbial translocation [34].
Effect of Gut Integrity on Visceral Fat Accumulation
In this study, we have shown for the first time in HIV an association between gut dysfunction and fat gain. Specifically, higher baseline I-FABP levels were found to be a major determinant of the increase in VAT, TAT (unadjusted), and BMI over 96 weeks following ART initiation in HIV-infected ART-naive adults. We have previously shown that pre-ART viral load was also independently associated with fat gain in these patients [2]. Our new findings further highlight the association between the initial severity of HIV infection and consequent gut epithelial dysfunction and fat accumulation in HIV-infected individuals. While the connection between gut dysfunction and fat accumulation remains unclear, disruption of gut mucosal integrity and microbial product translocation lead to the subsequent activation of cytokines and pro-inflammatory pathways in different organs [35, 36]. These inflammatory pathways in the liver may lead to increased fat and triglyceride production, contributing to an increase in visceral fat accumulation [37]. Additionally, both obesity and HIV infection are independently associated with microbial translocation, each contributing to local and systemic inflammation [1, 38] and generating and reinforcing these vicious cycles of inflammation, microbial translocation, and visceral fat expansion.
Our study has several limitations. First, the clinical significance of I-FABP and I-BABP is not fully understood, and the plasma levels of these markers might not correlate with their levels in the gut. Further longitudinal studies assessing gut barrier integrity and permeability from gut biopsies are needed to confirm these results. Additionally, we assessed LBP, which is an indirect marker of microbial translocation that might be less specific than other direct markers such as lipopolysaccharide (LPS) and 16S ribosomal ribonucleic acid (16S rRNA). We did not explore other potential factors, such as diet and gut microbiome, which might have an effect on gut integrity. Finally, most of our participants were men, which limits the generalizability of our results, especially given that these markers may vary with sex in this population [39]. By design, we limited our study to successfully treated participants to avoid the potential effect of viremia and/or lack of adherence to ART on the tested biomarkers.
In summary, in a randomized ART initiation trial of adults with chronic HIV infections, we have shown that the effects of regimins based on RAL, DRV/r, and ATV/r result in similar changes in gut epithelial markers (I-FABP and I-BABP) and microbial translocation products (LBP). However, zonulin, a gut permeability marker, improved with RAL as compared to PI-based regimens. A key finding in our results was that lower baseline gut dysfunction was independently associated with increases in BMI and VAT over 96 weeks, suggesting that the initial severity of HIV infection has the greatest impact on fat accumulation. Based on these results, the clinical significance of gut integrity markers needs to be assessed among HIV-infected individuals initiating ART, and the longitudinal effect of ART on gut epithelial structural integrity requires ongoing investigation.
Notes
Financial support. This work was supported by grants from the National Institute of Health (grant numbers DK118757, HL095132, HL095126, AI069501, AI068636, AI068634, AI69471, and AI56933).
Potential conflicts of interest. C. O. H. served as a consultant to and received grants from Gilead. J. S. C. received research grants from Theratechnologies to her institution. T. T. B. served as a consultant for Gilead, Merck, Theratechnologies, and EMD-Serono. G. A. M. served as a consultant for Gilead, Merck, and ViiV/GSK and received grant support to her institution from BMS, Roche, Astellas, GSK, and Gilead. P. W. H. reports personal fees from Merck, Viiv, and Seres and grants and personal fees from Gilead outside the submitted work. C. M. reports personal fees from Northwestern University Infectious Diseases for statistical consulting work. All other authors report no potential conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Erlandson KM, Lake JE. Fat matters: understanding the role of adipose tissue in health in HIV infection. Curr HIV/AIDS Rep 2016; 13:20–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. McComsey GA, Moser C, Currier J, et al. Body composition changes after initiation of raltegravir or protease inhibitors: ACTG A5260s. Clin Infect Dis 2016; 62:853–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Dirajlal-Fargo S, Moser C, Brown TT, et al. Changes in insulin resistance after initiation of raltegravir or protease inhibitors with tenofovir-emtricitabine: AIDS Clinical Trials Group A5260s. Open Forum Infect Dis 2016; 3:ofw174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lake JE, Stanley TL, Apovian CM, et al. Practical review of recognition and management of obesity and lipohypertrophy in human immunodeficiency virus infection. Clin Infect Dis 2017; 64:1422–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Guaraldi G, Luzi K, Bellistrì GM, et al. CD8 T-cell activation is associated with lipodystrophy and visceral fat accumulation in antiretroviral therapy-treated virologically suppressed HIV-infected patients. J Acquir Immune Defic Syndr 2013; 64:360–6. [DOI] [PubMed] [Google Scholar]
- 6. Brown TT, Tassiopoulos K, Bosch RJ, Shikuma C, McComsey GA. Association between systemic inflammation and incident diabetes in HIV-infected patients after initiation of antiretroviral therapy. Diabetes Care 2010; 33:2244–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hileman CO, Funderburg NT. Inflammation, immune activation, and antiretroviral therapy in HIV. Curr HIV/AIDS Rep 2017; 14:93–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Tincati C, Douek DC, Marchetti G. Gut barrier structure, mucosal immunity an3d intestinal microbiota in the pathogenesis and treatment of HIV infection. AIDS Res Ther 2016; 13:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Nehra V, Allen JM, Mailing LJ, Kashyap PC, Woods JA. Gut microbiota: modulation of host physiology in obesity. Physiology 2016; 31:327–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Bouter KE, van Raalte DH, Groen AK, Nieuwdorp M. Role of the gut microbiome in the pathogenesis of obesity and obesity-related metabolic dysfunction. Gastroenterology 2017; 152:1671–8. [DOI] [PubMed] [Google Scholar]
- 11. Anderson KV. Toll signaling pathways in the innate immune response. Curr Opin Immunol 2000; 12:13–9. [DOI] [PubMed] [Google Scholar]
- 12. Hunt PW, Sinclair E, Rodriguez B, et al. Gut epithelial barrier dysfunction and innate immune activation predict mortality in treated HIV infection. J Infect Dis 2014; 210:1228–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Pelsers MM, Namiot Z, Kisielewski W, et al. Intestinal-type and liver-type fatty acid-binding protein in the intestine. Tissue distribution and clinical utility. Clin Biochem 2003; 36:529–35. [DOI] [PubMed] [Google Scholar]
- 14. Derikx JP, Luyer MD, Heineman E, Buurman WA. Non-invasive markers of gut wall integrity in health and disease. World J Gastroenterol 2010; 16:5272–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sereti I, Krebs SJ, Phanuphak N, et al. ; RV254/SEARCH 010, RV304/SEARCH 013, and SEARCH 011 protocol teams Persistent, albeit reduced, chronic inflammation in persons starting antiretroviral therapy in acute HIV infection. Clin Infect Dis 2017; 64:124–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Vesterbacka J, Nowak P, Barqasho B, et al. Kinetics of microbial translocation markers in patients on efavirenz or lopinavir/r based antiretroviral therapy. PLoS One 2013; 8:e55038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kelesidis T, Tran TT, Stein JH, et al. Changes in inflammation and immune activation with atazanavir-, raltegravir-, darunavir-based initial antiviral therapy: ACTG 5260s. Clin Infect Dis 2015; 61:651–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28:412–9. [DOI] [PubMed] [Google Scholar]
- 19. Scherzer R, Heymsfield SB, Lee D, et al. ; Study of Fat Redistribution and Metabolic Change in HIV Infection (FRAM) Decreased limb muscle and increased central adiposity are associated with 5-year all-cause mortality in HIV infection. AIDS 2011; 25:1405–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Guaraldi G, Murri R, Orlando G, et al. Severity of lipodystrophy is associated with decreased health-related quality of life. AIDS Patient Care STDS 2008; 22:577–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Zevin AS, McKinnon L, Burgener A, Klatt NR. Microbial translocation and microbiome dysbiosis in HIV-associated immune activation. Curr Opin HIV AIDS 2016; 11:182–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Costiniuk CT, Angel JB. Human immunodeficiency virus and the gastrointestinal immune system: does highly active antiretroviral therapy restore gut immunity?Mucosal Immunol 2012; 5:596–604. [DOI] [PubMed] [Google Scholar]
- 23. Nazli A, Chan O, Dobson-Belaire WN, et al. Exposure to HIV-1 directly impairs mucosal epithelial barrier integrity allowing microbial translocation. PLoS Pathog 2010; 6:e1000852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sandler NG, Wand H, Roque A, et al. ; INSIGHT SMART Study Group Plasma levels of soluble CD14 independently predict mortality in HIV infection. J Infect Dis 2011; 203:780–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Makowski L, Hotamisligil GS. Fatty acid binding proteins–the evolutionary crossroads of inflammatory and metabolic responses. J Nutr 2004; 134:2464–8S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Bottasso Arias NM, García M, Bondar C, et al. Expression pattern of fatty acid binding proteins in celiac disease enteropathy. Mediators Inflamm 2015; 2015:738563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Epple HJ, Schneider T, Troeger H, et al. Impairment of the intestinal barrier is evident in untreated but absent in suppressively treated HIV-infected patients. Gut 2009; 58:220–7. [DOI] [PubMed] [Google Scholar]
- 28. Pinto-Cardoso S, Lozupone C, Briceño O, et al. Fecal bacterial communities in treated HIV infected individuals on two antiretroviral regimens. Sci Rep 2017; 7:43741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wu X, Sun L, Zha W, et al. HIV protease inhibitors induce endoplasmic reticulum stress and disrupt barrier integrity in intestinal epithelial cells. Gastroenterology 2010; 138:197–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Pilon AA, Lum JJ, Sanchez-Dardon J, Phenix BN, Douglas R, Badley AD. Induction of apoptosis by a nonnucleoside human immunodeficiency virus type 1 reverse transcriptase inhibitor. Antimicrob Agents Chemother 2002; 46:2687–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev 2011; 91:151–75. [DOI] [PubMed] [Google Scholar]
- 32. Sturgeon C, Fasano A. Zonulin, a regulator of epithelial and endothelial barrier functions, and its involvement in chronic inflammatory diseases. Tissue Barriers 2016; 4:e1251384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Serrano-Villar S, Sainz T, Ma ZM, et al. Correction: effects of combined CCR5/integrase inhibitors-based regimen on mucosal immunity in HIV-infected patients naive to antiretroviral therapy: a pilot randomized trial. PLoS Pathog 2016; 12:e1005540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Patterson KB, Prince HA, Stevens T, et al. Differential penetration of raltegravir throughout gastrointestinal tissue: implications for eradication and cure. AIDS 2013; 27:1413–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cani PD, Amar J, Iglesias MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007; 56:1761–72. [DOI] [PubMed] [Google Scholar]
- 36. Boutagy NE, McMillan RP, Frisard MI, Hulver MW. Metabolic endotoxemia with obesity: is it real and is it relevant?Biochimie 2016; 124:11–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. van Wijk JP, Cabezas MC. Hypertriglyceridemia, metabolic syndrome, and cardiovascular disease in HIV-infected patients: effects of antiretroviral therapy and adipose tissue distribution. Int J Vasc Med 2012; 2012:201027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Jung UJ, Choi MS. Obesity and its metabolic complications: the role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int J Mol Sci 2014; 15:6184–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Krebs SJ, Slike BM, Sithinamsuwan P, et al. ; SEARCH 011 study team Sex differences in soluble markers vary before and after the initiation of antiretroviral therapy in chronically HIV-infected individuals. AIDS 2016; 30:1533–42. [DOI] [PMC free article] [PubMed] [Google Scholar]