Abstract
Endometrium is the mucosal lining of the uterus which expressed a cyclic process of proliferation, secretion and scaling under the control of hormones secreted by the ovary, and it also plays an indispensable role in the embryo implantation, the constitution of fetal-maternal interface, and the maintaining of pregnancy. In pathophysiological conditions, the abnormality or disorder of endometrium may lead to endometrium-related diseases, such as endometriosis, endometrium hyperplasia and even endometrial carcinoma. In recent years, more and more evidence revealed that autophagy exists in both the endometrium stroma cells and epithelial cells, and the activity of autophagy is changed in the different phases of menstruation, as well as in the endometrium-related diseases. Here, we aim to review the activity level, the regulatory factors and the function of autophagy in physiological and pathophysiological endometria, and to discuss the potential value of autophagy as a target for therapies of endometrium-related diseases.
Keywords: autophagy, endometrium, endometriosis, endometrial carcinoma
Introduction
Autophagy is a highly conserved biological behavior in eukaryotic cells to disassemble unnecessary or dysfunctional components of the cell under exquisite regulation mechanism which can be initiated under the conditions of hypoxia, starvation, lack of nutrition, or extreme pH values 1-3. In macroautophagy (commonly referred to autophagy), substances such as proteins, organelles or cytoplasm are encapsulated in a double-membraned vesicle named as autophagosome, and degraded into small molecules for reuse by the fusion of autophagosome with lysosome 4. Autophagy can also dispose the misfolded proteins, aging organelles, and other damaged cell components to maintain the homeostasis of the cells. Basic level of autophagy is essential for almost all of the eukaryotic cells and many cellular activities, and the absence or aberration of autophagy may lead to a series of pathological states.
Recent studies revealed that autophagy also plays an indispensable role in the physiological and pathophysiological processes related to the endometria, including the cyclic menstruation, the decidualization or reconstitution during normal pregnancy, and the endometrium-related diseases such as endometriosis, endometrial carcinoma and infertility. As a basic biological activity, autophagy is closely related to the cell proliferation and apoptosis, which ubiquitously occur in the dynamic refreshment of endometrium. What's more, the autophagy level in endometrium cells reflects the condition of the disease or physiological process, and can influence the function of cells in some way. Therefore, autophagy in endometrium needs to be further studied and may be beneficial for the future treatment of endometrium-related diseases. And this article is to review the present studies about this topic.
The level of autophagy in endometrium
Endometrium is the mucosal lining of the uterus which can be derived into two functional layers: the transient superficial stratum functionalis and the permanent deeper stratum basalis 5. The superficial stratum functionalis, which is completely shed and regenerated in menstrual cycle, mainly contains glandular epithelium and stromal cells, while the stratum basalis is postulated to be the place in which the stem cells reside 6, 7. In addition, in both layers there exist the blood vessels and immune cells, whose number varies in different phase of a menace cycle in normal endometrium, including NK cells, macrophages, DC cells and so on8, 9. Autophagy is regarded as a ubiquitous physiological process that occurs in all eukaryotic cells. There is no doubt that autophagy also exists in all of the endometrial cells and plays diverse function in different process and diseases, but it was only reported to be detected in endometrial stroma cells and epithelial cells; study is deficient in illustrating the autophagy level in endometrial immune cells and vascular endothelial cells. This may be related to the difficulty in methods to monitoring the autophagy level and the acquiring enough cells for the experiment.
As a marker of autophagy, LC3 was found to be predominantly expressed in epithelial relative to stromal cells in normal endometrium 10 and eutopic or ectopic endometria form endometriosis patients 11. Autophagy in glandular cells was very weakly positive in the early and late proliferative phases; In contrast, intense immunoreactivity for LC3 was detected during the secretory phase and peaked during the late secretory phase of the menstrual cycle in glandular cells 10, 12. A withdraw of progesterone or estrogen can significantly increase the expression level of LC3 in endometrial Ishikawa cells, which may imitate the changes in menstrual phase 10. In a murine ovariectomized rat model, autophagosomes was observed to be significantly increased in the cytoplasm of the uterine epithelial cells 4 weeks postoperation compared with the sham-operated rats; similarly, the level of autophagy was higher in postmenopausal uterine epithelial cells compared to premenopausal uterine epithelial cells 13. Autophagy was also reported to exhibit a dynamic pattern during pregnancy. In the mouse model, LC3 was observed to reach the highest level during days 1 and 2 of pregnancy, and gradually fall from day 4 to day 8. The LC3 level on day 1 of pregnancy can be effectively inhibited by dexamethasone, which is a widely used anti-inflammatory compound, indicating that inflammatory response may be associated with the increased autophagy in the day 1 pregnant uterus 14.
Similar to endometrial epithelium, the level of autophagy in normal endometrial stroma cells (ESCs) significantly increased in secretory phase compared with proliferative phase; while in ectopic ESCs from endometriosis patients, autophagy maintained a constant level throughout the menstrual cycle 12, 15. And during the pregnancy, the autophagy of ESCs was also in a dynamic change, but there was no consecutive observation in human decidua 14, 16. Autophagy in ESCs is relatively low compared to glandular cells; LC3 expression in normal ESCs is quite weak even in the secretory phase 10. What draw our interest is that autophagy level differs in normal ESCs, eutopic ESCs and ectopic ESCs. Actually, there exist contrary opinions on whether autophagy is stronger in normal tissue or endometriotic tissues. Some researchers proved that autophagy level were significantly reduced in eutopic ESCs, ectopic ESCs, serum and peritoneal fluid from women with endometriosis compared with normal women 12, 15, 17-20, while others held the opposite views, revealing that autophagy showed a significant up-regulation in ectopic endometrium compared with normal endometrium 21-24. Similarly, the comparison of autophagy in ectopic ESCs and eutopic ESCs was also not completely clear and consensus 11, 15, 21-24. The differences in researches may due to the different detective method, the individual characteristic of specimen, and the immune microenvironment of patients. Thus, further studied are still needed to find out the truth.
Human endometrium undergoes repetitive cyclic regeneration after the shedding off in menstruation. In the dynamic change, endometrium-derived human mesenchymal stem cells (hMESCs) resided in the basal layer have contributed a lot to enable the rapid regeneration of endometrium after the shedding off in last cycle. The basic level of autophagy seems to be low in hMESCs, while under oxidative stress, the suppression of the p53 transcriptional activity or the calcium chelation caused a significantly increase of autophagy and lead to cell death under oxidative stress 25, 26. Autophagy was discovered to induce G0/G1 arrest and apoptosis of menstrual blood-derived endometrial stem cells via GSK3β/β-catenin pathway 27. On the other hand, in endometrial cancer stem cells (ECSCs), autophagy level seems to be relatively strong. CD133 and CD44 are reported to be important markers of ECSCs. The results from Ran et al. showed that Beclin1, Atg5, Atg7 mRNA levels were significantly enhanced but the P62 was obviously reduced in CD133+/CD44+ cells compared to normal JEC cells (a cell line of endometrial cancer), demonstrated that the CD133+/ CD44+ cells which exhibit CSC properties possess higher autophagy 28 (Table 1).
Table 1.
The autophagy level in endometrium
| Non pregnant | Pregnant | ||||||
|---|---|---|---|---|---|---|---|
| Proliferative phase | Secretory phase | Menstrual phase | First trimester | Second trimester | Term | ||
| Stromal cells | +(-)10, 15 | ++10, 15 | / | +++14 | +14 | ++16 | |
| Epithelial cells | +10 | +++10 | ++13 | +14 | / | / | |
| endometrium-derived human mesenchymal stem cells | Induced by calcium chelation or p53 suppression under oxidative stress25, 26 | ||||||
+++: The autophagy level in normal endometrial epithelial cells in the secretory phase; -: Negative expression of autophagy; /: Not mentioned
Regulatory mechanism of autophagy in endometrium
Autophagy takes part in variety of biological activities, and the level of autophagy can influence the cellular metabolism, the proliferation and program cell death, and the drug resistance or sensibility of cancer cells. Thus, autophagy is precisely regulated in different condition. Factors such as hormones, cytokines, metabolic products and non-coding RNAs can regulate the autophagy level independently or interdependently.
Hormones
Hormones are the main factors that regulate the cyclic proliferation, secretion and shedding of endometrium. Considering that autophagy also changes during the menstrual cycle, it's not difficult to imagine that hormones play important roles in adjusting the level of autophagy in endometrium.
Estrogen is the predominant hormone during the proliferative phase, and many researches have proved that it can inhibit the autophagy activity in endometrium 13, 15, 17, 29, 30. Mei et al. found that estradiol (E2) treatment induced a marked increase in CXCL12 secretion and CXCR4 expression together with decreases in autophagy grade, punctuate LC3B immunofluorescent staining and autophagy-associated protein levels, while the suppression of autophagy induced by E2 and recombinant human CXCL12 protein could be abrogated by an anti-CXCR4 neutralizing antibody, suggesting that estrogen could suppress the autophagy of ESCs by upregulating CXCL12 and CXCR4 expression 29. Estrogen was also reported to induce the autophagy level in normal endometrial cancer cells and endometrial carcinoma stem cells by promoting the expression of estrogen induced gene 121(EIG121), which can induce cytoplasmic vacuolization, induce the formation of autophagosomes and enhance lysosomal degradation of long-lived proteins 31. Knockdown of EIG121 compromises starvation-induced autophagy and sensitizes cells to cell death induced by nutrient deprivation and exposure to cytotoxic agents 28, 31. In a ovariectomized rats menopausal model, autophagy was observed to be increased in uterine endometrium when estrogen was deprived, and the estrogen depletion induced autophagy was possibly via endoplasmic reticulum (ER) stress-dependent arachidonic acid /prostaglandin E2 axis inhibition and Akt-mTOR signaling pathway suppressing in uterine epithelial cells 13.
Relationship between estrogen and progestogen can be both synergistic and antagonistic depending on the tissues or cells receiving the signals. As is the case, their role in regulating the activity of autophagy may be complicated. In the study of Mei et al., the estrogen induced autophagy in ESCs could be reversed by progestogen 15, suggesting that progestogen plays an antagonistic role in controlling the autophagy in ESCs. Consistent with this, Choi et al. 32 found that dienogest, but not progestogen, can enhance autophagy induction in endometriotic cells by impairing activation of AKT, ERK1/2, and mTOR signal pathways. On the contrary, Choi et al. 14 found that progesterone and estrogen both reduced the autophagic response in the uteri in ovariectomized mice model. The differences may occur from the tested samples, actually, the mice uteri contain different kinds of cells, and the total mRNA or protein may not reflect the separate change of autophagy in ESCs, EECs, or other cells precisely.
Other hormones are also involved in regulating the autophagy condition in endometrium. After menopause, the level of estrogen and progestogen goes down while follicle-stimulating hormone (FSH) commonly rises with the decline of ovary function. It was reported that high circulating levels of FSH in postmenopausal women activated the phosphorylation of Smad2/ Smad3 through transforming growth factor beta receptor II (TbRII). The complexes of phosphorylated Smad2/Smad3 subsequently transported into the nucleus and launched the expression and activation of several autophagy-related molecules such as ATG5, ATG12, ATG3, and ATG7. Persistent activation of cell autophagy may directly or indirectly induce cell apoptosis. Thus, FSH can promote the autophagy of endometrial adenocytes 33. Autophagy may also be related with increased androgen availability in PCOS. It was found that autophagy related genes were significantly reduced in anovulatory PCOS compared to healthy endometrium, and was negatively correlated with free androgen index. Treatment of PCOS patients with metformin significantly increased the endometrial mRNA levels of autophagy-associated genes, suggesting that the increased androgen level in PCOS is associated with metformin-sensitive transcriptional downregulation of endometrial autophagy 34. And dexamethasone, a widely used anti-inflammatory compound, was also observed to inhibit the autophagy of mice uteri at the first day of pregnancy, when the autophagy level should have been the highest 14. This effect suggests that inflammatory response may be associated with increased autophagy in the day 1 pregnant uterus. Other hormones, such as luteinizing hormone (LH), anti mullerian hormone (AMH), prolactin (PRL), and Human Chorionic Gonadotropin (HCG), which also take vital part in the menstruation cycle or gestation of fertile women, still need to be investigated on their role of regulating the autophagy of endometrium.
Metabolism and metabolites
Autophagy can be influenced by many metabolites, and it can affect the metabolic condition of cells simultaneously. Rhee et al. found that a high level of palmitic acid, which is commonly observed in obese people as a result of imbalance of energy metabolism, can impaired the autophagy of human endometrial cells during decidualization 16. This may be one of the mechanisms that lead to the poor reproductive outcome and early pregnancy loss in obese women. Folate acid, a widely applied supplement by women during pregnancy, was reported to induce the autophagy of endometrium, thus benefits the decidualization process 35. In another experiment, researchers found that compared to the control group, caloric restriction can significantly increase the autophagy activity and inhibit the p-Akt and mTOR levels in endometriotic lesions of endometriosis mice model 36.
Apart from this, the amino acid metabolism can also regulate the autophagy condition in endometrium. It was detected that the level of phenylalanine, indoleacrylic acid (IAA), phosphocholine and lyso- platelet-activating factor-16 (lyso-PAF) in serum were higher in endometrial carcinoma patients than normal controls, and all of them played a role in regulating the autophagy tumor cells. PAF can induce tumor cell apoptosis and autophagy, while IAA triggered apoptosis and had a biphasic effect on autophagy: inhibiting autophagy with doses <1 mmol/L but inducing at 1 mmol/L. Interestingly, the alterations in apoptosis and autophagy caused by 1 mmol/L IAA coule be reversed by the concomitant treatment of tryptophan (100 mumol/L). Phosphocholine inhibited tumor cell invasion and migration, and promoted cell proliferation and autophagy, all in a dose-dependent manner 37. Other substance, such as retinoic acid, was also reported to enhance the autophagy in ESCs 38. According to this, we can learn that autophagy is widely regulated by many kinds of metabolic products, and closely related to various biological activities.
What's more, the supply of oxygen plays a critical role in the process of cell growth. The lack of oxygen, namely hypoxia, can induce the autophagy of cultured human endometrial stromal cells (HESCs) in a time-dependent manner through hypoxia-inducible factor-1alpha (HIF-1α) 23. LC3-II expression and the number of autophagosomes were gradually increased by hypoxia treatment 22. This autophagy inducing effect of hypoxia was also reported in other cells and diseases, such as preeclampsia, non-alcoholic steatohepatitis, neurodegenerative diseases, and so on 39, 40. Oxidative stress is another good candidate responsible for autophagy induction in endometriotic cells. The persistent oxidative stress, by itself and/or a positive feedback with heme oxygenase-1(HO-1), reactive oxygen species (ROS), and other down-stream response, can stimulate the autophagic process 41-43.
Oxidative stress induces a rapid calcium release from intracellular stores, which play an important role in apoptosis and necrosis, senescence, as well as autophagy. With a sublethal H2O2 treatment, an artificial environment of oxidative stress was formed. It was found that in H2O2-treated human endometrium-derived stem cells, intracellular calcium ([Ca2+]i) chelation by BAPTA-AM can effectively initiate an early onset of autophagy, as observed by both the phosphorylation status of AMPK/mTORC1 pathway and the dynamics of the LC3 lipidization. In other word, the calcium release under oxidative stress could inhibit the activation of autophagy, which may be related to the prevention of the premature senescence and DNA damage 26. Different from the free state of calcium, iron is presented in cells as ferric ammonium citrate (FAC). Treatment with FAC can significantly increase the protein level of LC3-II and the number of autophagosomes in a dose dependent manner in immortalized endometriotic Cells, by the activation of AKT and MAPK pathway 41.
Enzymes, inhibitor and drugs
Serum and glucocorticoid-regulated kinase 1 (SGK1) is a serine/threonine protein kinase that shares structural and functional similarities with the AKT family of kinases 44. It plays an important role in cellular stress response by activating certain potassium, sodium, and chloride channels, suggesting an involvement in the regulation of processes such as cell survival, neuronal excitability, and renal sodium excretion 45-47. In endometrial carcinoma, the expression of SGK1 was upregulated in tumor tissues than normal control, and inhibiting SGK1 with SI113 can induce a reduction of endometrial cancer cells viability as well as a significant increase of autophagy, as revealed by the increase of the markers LC3B-II and beclin I, detected by both immunofluorescence and western blot analysis 48. This effect may be associated to the induction of endoplasmic reticulum stress markers GRP78 and CHOP evaluated by both real-time PCR and western blot analysis.
Inhibitors of the key enzymes can also influence the process of autophagy in endometrium. Liraglutide, which is an agonist of glucagon-like peptide-1 receptor (GLP-1R), can significantly induced autophagy of endometrial carcinoma cells and inhibited Ishikawa cell growth, as well as the elevated AMPK expression 49. Bortezomib is an inhibitor of 26S proteasome, which has been reported to inhibit protein degradation in lysosomesmay by inducing ERK phosphorylation and suppress cathepsin B in endometrial carcinoma cells and enhance chemotherapy efficacy by abolishing chemotherapy-related autophagy 50. A new synthetic histone deacetylase inhibitor, MHY2256, was found to induce apoptosis and autophagy cell death in endometrial cancer cells via p53 acetylation 51. Similarly, as an inhibitor of mTOR, RAD001 significantly decreased phosphorylation of mTOR and inhibited proliferation of progestin- resistant cancer cells and induced a higher sensitivity to paclitaxel-induced apoptosis by promoting autophagy 52, 53. Rapamycin is another inhibitor of mTOR, which is the most commonly used as an inducer of autophagy, but we should notice that it can affect both mTORC1 and mTORC2. What's more, mTOR is a major regulatory protein that is part of several signaling pathways, including for example those that respond to INS/insulin, EGF/epidermal growth factor and amino acids, and it thus controls process other than autophagy 54-57. Therefore, rapamycin will ultimately affect many metabolic pathways and the pleiotropic effects need to be considered 58.
Many drugs, especially those used in the anti-tumor treatment, were discovered with the effect of adjusting the autophagy activation. Metformin, a commonly used medicine for treating type II diabetes, which has recently been reported to possess anti-proliferative properties in a variety of cancers, was reported to regulate the autophagy of tumor cells 59, 60.It was found that metformin could increase the apoptosis, inhibited the growth, and promote the autophagy of endometrial carcinoma cells with different concentrations in a dose- and time-dependent manner 59-62. And the autophagy inducing effect was proved to be related with CEBPD or AMPKα 63, 64 in several tumor cells. Sorafenib is another anti-tumor drug, or more precisely, a multi-target tyrosine and serine/threonine kinase inhibitor approved by FDA for the treatment of hepatocellular carcinoma, advanced renal carcinoma and radioactive iodine-resistant thyroid carcinoma 65, 66. Sorafenib could induce an unfolded protein response (UPR) in EC cells that connects with an autophagic output through a MAPK/JNK-dependent mechanism and this MAPK/JNK-dependent early protective autophagic response in endometrial carcinoma cells may count for the limited effect of sorafenib in a multi-institutional phase II trial in advanced uterine carcinoma patients 67. Programmed cell death 4 (PDCD4), a newly identified tumor suppressor, was uncovered to effectively inhibit the proliferation and suppress the migration and invasion of endometrial cells by inhibiting cell autophagy, probably through NF-κB/MMP2/MMP9 signal pathway 68. Cisplatin was also reported to upregulate cell autophagy in endometrial cancer cells via the PI3K/AKT/mTOR signaling pathway 69. And interestingly, the combination of cisplatin and rapamycin showed a synergetic anti-endometrial cancer effect 58. Also, other drugs 70, including traditional Chinese medicine such as Isoliquiritigenin (ISL), Protopanaxadiol (PPD) showed a potential to regulate the autophagy in endometrium 30, 71, 72. The role of autophagy in endometrial cancer is worthy investigating and is discussed in the next part.
Others
With the constant-depth study, non-coding RNA has been unveiled to participate in more and more biological activities. It was now clear that many micro RNA and long non-coding RNA (lncRNA) have a regulatory effect on the activation of autophagy in endometrium. For example, miR-101-3p was reported to upregulate the expression of autophagy-related proteins LC3-II and beclin-1 in EC cells in a time- and dose-dependent manner, by downregulating the expression of enhancer of zeste homolog 2 (EZH2) 73. EZH2 is a critical component of the polycomb repressive complex 2 with intrinsic histone methyl transferase activity that mediates gene silencing by catalyzing trimethylation on lysine 27 of histone H374. And it can epigenetically repress tuberous sclerosis 2 (TSC2), a negative regulator of the mTOR pathway. Downregulation of TSC2 by EZH2 elicited mTOR activation, which in turn induced the inhibition of autophagy 75. MiR-210, a micro RNA which was significantly induced by hypoxic treatment, was found to promote autophagy partly by inhibiting the Bcl-2 expression 76. MiR-218 can also inhibit the autophagy of endometrial cancer cells by directly bind to the 3'-UTR of HMGB1 gene, which is one of the most important regulators of cellular autophagy 77. lncRNAs, specifically HOTAIR, can regulate the level of autophagy in human endometrial cancer cells by influencing Beclin-1, MDR, and P-gp expression. Interference of HOTAIR enhanced the autophagy activity of cisplatin-resistant Ishikawa cells and further reduced the proliferation of cisplatin- resistant Ishikawa cells 78. lncRNA-MALAT1 also promoted hypoxia-induced autophagy in endometrial stromal cells and reduced their apoptosis, which facilitated the survival of ectopic endometrium79.
DNA mismatch repair (MMR) can induce autophagy in human endometrial tumor cells via signal transferred by p53 after exposure to 6-thioguanine (6-TG), a chemotherapy drug which can be recognized by MMR. Autophagy induced by DNA mismatch repair inhibits apoptosis of endometrial carcinoma cells. An inhibition of autophagy by Atg5 knockdown will significantly promotes the apoptosis of cancer cells after DNA mismatch repair processing of 6-TG, suggesting that autophagy mediated the survival of cancer cells with MMR in the treatment of 6-TG 80. A brief summary of regulators of autophagy in endometrium is shown in Table 2.
Table 2.
Regulatory factors of autophagy in endometrium
| Effect on autophagy | Note | References | ||
|---|---|---|---|---|
| Hormones | Estrogen | downregulate | by upregulating CXCL12 and CXCR4 expression | 13-15, 17, 29, 30 |
| upregulate | by promoting the expression of EIG121 | 28, 31 | ||
| Progestogen | upregulate | impairing activation of AKT, ERK1/2, and mTOR | 28, 31 | |
| downregulate | PR dependent | 14 | ||
| FSH | upregulate | partly through activating the phosphorylation of Smad2/ Smad3 via transforming growth factor beta (TGFβ) | 33 | |
| Androgen | negatively correlated | increased androgen availability in PCOS is associated with downregulation of endometrial autophagy | 34 | |
| Dexamethasone | downregulate | significantly reduced autophagy in the day 1 pregnant mice uterus which may be related to inflammatory response | 14 | |
| Metabolism and Metabolites | Palmitic acid | downregulate | defects in autophagy caused by palmitic acid may contribute to impaired decidualization | 16 |
| Indoleacrylic acid (IAA) | biphasic effect | inhibiting autophagy with doses <1 mmol/L but inducing at 1 mmol/L; autophagy caused by 1 mmol/L IAA coule be reversed by the concomitant treatment of tryptophan (100 mumol/L) | 37 | |
| Phosphocholine | upregulate | in a dose-dependent manner | 37 | |
| lyso-platelet-activating factor-16(lyso-PAF) | upregulate | in a dose-dependent manner | 37 | |
| retinoic acid | upregulate | / | 38 | |
| Folate acid | / | 35 | ||
| calcium | downregulate | calcium chelator induce an early onset of autophagy through AMPK/mTORC1 pathway under oxidative stress | 26 | |
| iron | upregulate | by the activation of AKT and MAPK pathway | 41 | |
| hypoxia | upregulate | through HIF-1α | 22, 23 | |
| Oxidative stress | upregulate | by itself and/or a positive feedback with heme oxygenase-1(HO-1), ROS, and other down-stream responses | 21, 41-43 | |
| Enzymes, inhibitor and drugs | SGK1 | upregulate | associated to the induction of endoplasmic reticulum stress | 48 |
| Liraglutide | upregulate | may be related to the AMPK phosphorylation | 49 | |
| Bortezomib | downregulate | inducing ERK phosphorylation and inhibiting protein degradation in lysosomes | 50 | |
| MHY2256 | upregulate | p53 acetylation | 51 | |
| RAD001 | upregulate | downregulation of AKT/mTOR phosphorylation | 52, 53 | |
| Rapamycin | upregulate | binds to FKBP1A/FKBP12 and inhibits mTORC1 | 30, 58 | |
| Metformin | upregulate | inducing autophagy through CEBPD upregulation or AMPKα | 63, 64 | |
| Sorafenib | upregulate | through a MAPK/JNK-dependent mechanism | 67 | |
| Isoliquiritigenin (ISL) | upregulate | By activating the extracellular signal regulated kinase signaling pathway | 71 | |
| Itraconazole | upregulate | inhibit the expression of mTOR signaling components | 70 | |
| Protopanaxadiol | upregulate | via AMPK/JNK phosphorylation | 30, 72 | |
| PDCD4 | downregulate | may be related to NF-kappaB/MMP2/MMP9 signal pathway | 68 | |
| Cisplatin | upregulate | via the PI3K/AKT/mTOR signalling pathway | 69 | |
| Others | miR-101-3p | upregulate | by downregulating the expression of Enhancer of zeste homolog 2 (EZH2) | 73 |
| miR-210 | upregulate | partly by inhibiting the Bcl-2 expression | 76 | |
| MiR-218 | downregulate | by directly bind to the 3'-UTR of HMGB1 gene | 77 | |
| Lnc-RNA HOTAIR | downregulate | by influencing Beclin-1, MDR, and P-gp expression | 78 | |
| lncRNA-MALAT1 | upregulate | / | 79 | |
| DNA mismatch repair (MMR) | upregulate | via signal transferred by p53 | 80 |
The role of autophagy in physiological conditions
As is mentioned above, the level of autophagy is in a dynamic state during the menstrual cycle. The role of autophagy in menstruation is still not quite clear. Based on the limited research, we can learn that autophagy may be related to the apoptosis induction of endometrial cells by increasing the BAX: BCL2 ratio and activating the subsequent caspases, and is directly involved in the cyclic remodeling of the human endometrium 10.
In the mechanism research of keratinocyte growth factor in treating intrauterine adhesion, autophagy was confirmed to show a close relatively relationship with the proliferation of endometrial epithelial cells and the angiogenesis in endometrial 81. This may also occur in the menstrual period after the shedding of endometrium. Accordingly, autophagy in endometrial cells may be related to the remodeling of the endometrium, as well as the angiogenesis during the menstruation cycle.
After menopause, endometrium undergoes a physiological atrophy with the withdrawal of estrogen and progestogen. Autophagy is detected to play a prominent role in uterine epithelial cell death and is a critical regulator of the uterine epithelium that accounts for endometrial atrophy 13. Similarly, persistent activation of cell autophagy induced by the high level of FSH may directly or indirectly induce cell apoptosis, and finally contribute to the atrophy of endometrial 33. What's more, 3-MA injections significantly increases uterine glycogen content in ovariectomized uteri, suggesting that heightened autophagy under hormone deprivation is associated with glycogen breakdown as an energy provider 14.
Another role of autophagy is the regulatory effect during decidualization. Autophagy pathway plays a vital role in cell proliferation, endometrial function, prostaglandin secretion and cell attachment 82. A high level of autophagy was detected at the first day of pregnancy in mice uteri when the uterus shows an inflammatory response to mating, revealing that uterine autophagy may have undetermined functions as a responsive mechanism to acute inflammation 14. During the decidualization period, the autophagy in endometrium significantly increased, and an impaired autophagy in obese women may be related to a poor outcome of pregnancy, suggesting that autophagy contribute to the endometrial receptivity and embryo implantation 16. Being related to endoplasmic reticulum stress, autophagy was also reported to contribute to early pregnancy success and regulate endometrial function 83. In conclusion, the increasing autophagy is indispensable during the decidualization of endometrium and defects in autophagy may relate to poor reproductive outcomes and early pregnancy loss.
The role of autophagy in endometrium-related diseases
As a fundamental activity of cells, autophagy is essential in biological process of endometrium in both physiology and pathology. In endometrium-related diseases, autophagy plays a multifunctional and complicated role as revealed by more and more evidence, which increased the difficulties for us to understand the effect of it in a certain situation. As a matter of fact, the function of autophagy in different endometrium-related diseased may be totally different. Here, we make a brief discussion of autophagy in endometrium-related diseases separately based on the researches till now, and more thorough studies are needed to help us understand the role of autophagy in endometrium.
Endometrial carcinoma
Endometrial carcinoma is one of the most common malignant diseases in women all over the world. It was reported that autophagy has played important roles in the tumorigenesis, the proliferation, the drug resistance, and the immune regulation of endometrial carcinoma. To investigate the autophagy targeted treatment for endometrial carcinoma may be a potential way instead of the traditional method.
Three autophagy related genes, RB1CC1, ULK4, and WDR45 were found to be significantly mutated in endometrial carcinoma patients compared with normal women, suggesting that aberrant activation or disruption of autophagy plays a role in the tumorigenesis of endometrial carcinoma 84. LC3A reactivity was recognized in three basic patterns: diffuse cytoplasmic, cytoplasmic/juxta-nuclear, and the so-called “stone-like” structures (SLS). The latter has emerged as the hallmark of autophagic activity, being detected exclusively in endometrial carcinomas and mildly in the atypical hyperplasias, while other forms of hyperplasia without cytological atypia and normal endometrial tissues expressed only cytoplasmic staining patterns, suggesting that autophagy is more activated in endometrial carcinoma cells than normal or hyperplastic endometrial 85. The highly activated autophagy may be related the proliferation, apoptosis and other biological activity of tumor cells, which contribute to the oncogenesis of endometrial carcinoma.
Actually, it was reported that autophagy may promote cell survival under nutrient deprivation and other cellular stresses 31. But in other cases, autophagy is more likely to associate with the cell apoptosis and cell death, which is negatively related to proliferation. Several anti-cancer drugs was found to induce the autophagy of cancer cells and inhibit the cell viability, suppress the cell proliferation and downregulate the cell growth 48, 49, 86. Similarly, the lower level of autophagy in progestogen-resistant endometrial cancer cells was related to the increased proliferation 52, and estrogen was also found to promote the cell proliferation and simultaneously inhibit the autophagy in endometrial cancer cells 30. In conclusion, autophagy may play a beneficial role in the initiation of endometrial carcinoma and the survival of tumor cells under stress, but excessive autophagy may lead to the apoptosis and cell death, which is bad for the proliferation of endometrial carcinoma.
Autophagy is also related to the drug resistance of endometrial carcinoma cells for some traditional anti-tumor medicines. For example, cisplatin was reported to upregulate cell autophagy in endometrial cancer cells via the PI3K/AKT/mTOR signalling pathway 69. And by inhibiting the level of autophagy, chloroquine can effectively suppress the proliferation in endometrial cancer cell lines in a dose-dependent manner, the sensitivity of endometrial carcinoma cells to cisplatin was also improved by knocking down ATG5 or ATG7 87. Autophagy has also been shown to be involved in cell resistance to paclitaxel. Paclitaxel-mediated cell death was further potentiated by pretreatment with autophagy inhibitor chloroquine or shRNA against the autophagic gene beclin 1, suggesting that paclitaxel- elicited autophagic response plays a protective role that impedes the eventual death of endometrial carcinoma cell, and autophagy-inhibitor therapy could be an effective and potent strategy to improve paclitaxel treatment outcomes in the treatment of endometrial carcinoma 88. This kind of protective auphagic response was also observed in some new drugs, such as sorafenib. Sorafenib is currently used to treat hepatocellular carcinoma, advanced renal carcinoma and radioactive iodine-resistant thyroid carcinoma, but it has not showed much effect in a multi-institutional phase II trial in advanced uterine carcinoma patients. A recent study shows that targeting autophagy enhances sorafenib cytotoxicity and suppresses tumor growth and pulmonary metastasis progression, which can provide insights into the unopposed resistance of advanced endometrial carcinoma to sorafenib and highlight a new strategy for therapeutic intervention in recurrent endometrial carcinoma 67. For those patients who are insensitive to chemotherapy, inhibiting the autophagy of tumor cells may be one of the new strategies for effective treatment.
On the other hand, autophagy is necessary for the anti-tumor effect in some of the drugs, meaning that they can induce the cancer cell death at least partly by promoting the autophagy. More and more evidence showed that metformin has effects on suppressing the growth of different kinds of tumor cells. It was revealed that metformin can induce the autophagy of endometrial cancer cells, and inhibition of autophagy either by beclin1 knockdown or by 3-methyladenine-mediated inhibition of caspase-3/7 can significantly suppress the anti-proliferative effects of metformin on endometrial cancer cells, indicating that the anti-proliferative effects and apoptosis- inducing effect caused by metformin were partially or completely dependent on autophagy 62. Autophagy was also involved in the cell death inducing effect of suberoylanilide hydroxamic acid (SAHA), a histone deacetylase inhibitor, which was a promising therapeutic agent for endometrial stromal sarcoma 89. These results told us that autophagy may be a double-edged sword in the treatment of endometrial carcinoma. Just like the relationship of autophagy and proliferation, in a certain condition, autophagy may promote the drug resistance or the drug effect according to specific circumstances.
Apart from this, autophagy is related to the immune regulation of the local tumor environment. Exposure with rapamycin results in a significantly increased autophagy in endometrial cancer cells, and it can further enhanced the cytotoxicity of NK cells by upregulating the expression of IL-27 in endometrial cancer cells and IL-27 receptors on NK cells, which can restricted the growth of tumor in Ishikawa- xenografted nude mice 58. In contrast, high expression of Beclin-1 is related to high tumor grade, high myometrial invasion, and a poor 5-year survival in endometrioid adenocarcinoma 90, 91.
Endometriosis
Endometriosis is a chronic, estrogen-dependent disease and characterized by the implantation of endometrial glands and stroma deep and haphazardly into the outside the uterine cavity 92. Autophagy in endometriosis-derived tissues is reduced compared with normal endometriosis tissues, especially in ectopic endometrium and during the secretory phase 93. The impaired autophagy plays critical roles in the process of cell apoptosis and proliferation, invasion and migration, cell immunity, and is associated to the stage and prognosis of endometriosis 93, 94.
As a benign disease characterized by the presence, transfer, invasion, and cultivation of growing endometrial tissue, endometriosis is now found to be more and more closely related to the immune microenvironment at local peritoneal cavity. The impaired autophagy in ectopic ESCs results in the downregulation of hematopoietic cellular kinase (HCK) by inactivating signal transducer and activator of transcription 3 (STAT3), as well as the increased secretion of the downstream molecules CXCL8/IL-8 and IL23-A by ESCs, and this increase induced the upregulation of CD16- NK cells and decline of cytotoxic activity in ectopic lesion microenvironment 17. Similarly, we also found that rapamycin decrease the level of IL-15 receptors in ESCs, which may downregulate the effect of IL-15 on stimulating the growth and invasion of ESCs and helping the immune escape of ESCs by suppressing the cytotoxic activity of NK cells in the ectopic milieu 95. What's more, we proved that autophagy in ectopic ESCs enhances the cytotoxic activity of NK cells and suppresses the growth of ectopic lesions in a mouse EMS model. By increasing the autophagy of ectopic ESCs, protopanaxadiol significantly increased the expression of activating natural cytotoxicity receptors (NKp30 and NKp46) and cytokine IFN-γ, and decreased IL-10 expression in NK cells, and protopanaxadiol can also decreased the number and weight of mouse ectopic lesions 29. Autophagy in ESCs can also influence the function of macrophage around the ectopic lesion. Ruiz et al. noted that autophagic flux inhibitor hydroxychloroquine (HCQ) increase the levels of peritoneal macrophages and the IP-10 (10 kDa interferon-γ-induced protein) chemokine in a mouse model of endometriosis 11, whereas the role of macrophage in the development of endometriosis needs to be deep investigated.
Autophagy is also reported to be related to the proliferation and apoptosis of eutopic or ectopic endometrial tissues from endometriosis patients. By inhibiting the activity of autophagy in endometriotic stromal cells, MK2206 (an AKT inhibitor) and chloroquine markedly reduced cell growth and regrowth after discontinuation of treatment, and reduced the size of endometriotic implants, indicating that autophagy played a cytoprotective role in endometriosis 96. Autophagy was also reported to inhibit the proliferation and colony formation, and the growth of filopodia of the endometriotic cell line CRL-7566 through fascin-120. A newly identified tumor suppressor, PDCD4, can effectively inhibit the proliferation and colony-forming ability and suppress the migration and invasion ability of endometrial cells probably by inhibiting cell autophagy 68. Xu et al. also showed that autophagy in ovarian endometriotic cell line CRL-7566 may contribute to pathological development of endometriosis through enhancing cell survival 76. However, some researcher deemed that autophagy may promote the apoptosis of endometriotic cells. Dienogest treatment of endometriotic cells was found to suppress AKT and ERK1/2 activity, thereby in turn inhibiting mTOR, inducing autophagy and promoting apoptosis 32. Choi et al. also identified that rapamycin treatment induced autophagy and led to apoptosis promotion, which was reversed by the addition of 3-MA, suggesting that mTOR inhibition promotes endometriotic cell apoptosis via autophagy induction 12. And rapamycin was observed to inhibit the proliferation and colony formation and inhibit the growth of filopodia of the endometriotic cell line CRL-7566, which can be reversed by 3-MA 20. Collectively, autophagy may be essential for the proliferation and cell growth of endometriotic cells, but under some certain circumstances, the increased activity of autophagy could lead to their apoptosis.
Migration and invasion are two typical characteristics of endometriotic cells contributing to the development of endometriosis, which are also uncovered to be regulated by the level of autophagy. Human endometrial epithelial cells treated by hypoxia displayed a higher level of autophagy and higher ability of migration and invasion compared to the control cells; however, when co-treated with autophagy inhibitors 3-MA and CQ, the migratory and invasive potential were dramatically decreased. These observations indicated that autophagy facilitates the hypoxia triggered migration and invasion 22. Similar effects were also observed by inhibiting autophagy with specific inhibitors and Beclin1 siRNA in human endometrial stromal cells, the decreased autophagy activity attenuated their hypoxia triggered migration and invasion 23. PDCD4 effectively suppressed the migration and invasion ability of endometrial cells, and the mechanism was also found out to be related to its function of inhibiting the autophagy of endometrial cells 68. Based on the researches, we may conclude that autophagy play an important role in the migration and invasion of endometrial cells.
CA125 is a critical index which can reflect the active inflammatory responses and the development or stage, as well as predict the recurrence of endometriosis 97. As is shown in the study of Ren et al. 98, Beclin 1 protein expression in eutopic endometrial tissues was negatively correlated with serum CA125 (r = -0.307, P = 0.015), and pelvic pain (r = -0.542, P = 0.000). Lu et al. 38 also found that Beclin1 expression showed a negative correlation with the clinical stage of endometriosis, demonstrated that autophagy is lower in the advanced stage of endometriosis. However, our previous work showed that there was no significant difference in autophagy grade between stage I-II and stage III-IV endometriosis-derived ectopic ESCs 15. Zheng et al. 24 clarified that there was a significant positive correlation between serum CA125 level and LC3-II protein level and/or LC3-II/LC3-I ratio in endometrial tissues, whereas the correlation index was separately 0.307 and 0.405, and the CA125 level was negatively related to the mRNA expression of LC3B and Beclin and protein level of Beclin, while the correlation index was quite small and with no significance 24. Since the relationship between autophagy and stage of endometriosis as well as the serum CA125 level remains to be controversial, more samples should be included in the research to figure out the fact, which may help to comprehend the role of autophagy and direct the future treatment of endometriosis with autophagy-related drugs.
Hyperplasia
Autophagy may be related to the endometrial hyperplasia induced by tamoxifen 99. As a first generation of selective estrogen receptor modulators, tamoxifen is usually recommended for the hormone receptor positive breast cancer patients, but one of the side effects of tamoxifen is endometrial hyperplasia even endometrial cancer. Feng et al. found that tamoxifen could significantly induce the mRNA and protein expression of autophagy adaptor P62 in endometrial cells but not breast cancer cells, and inhibiting the expression of P62 retarded tamoxifen- promoted growth of endometrial cancer cells 99, indicating that the selectively promotion of autophagy in endometrial and breast cancer cells may be the reason for tamoxifen-induced hyperplasia. Actually, we may hypothesize that autophagy was associated with hyperplasia of endometrium, not only in the tamoxifen-induced cases. Studies are needed to prove whether this was true in the spontaneous hyperplasia or hormone-related hyperplasia since the evidence was rare.
Endometrium-related infertility
Although many factors contribute to infertility and early pregnancy loss, decidualization defects was regarded as an important factor contributing to compromised endometrial receptivity and poor implantation. As is discussed above, autophagy is beneficial to the process of decidualization, and impaired autophagy in endometrial cells may be related to infertility. In obese women, the increased level of palmitic acid may decrease the autophagy in endometrial cells as well as the process of decidualization, and further cause the implantation failure 16. And autophagy in polycystic ovary syndrome patients was reported to be negative associated to the androgen level, indicating that the inspired autophagy may be related to the increased level of androgen, which inhibits the growth of follicle and is detrimental to the endometrial receptivity 34. Much remains unknown in the field of infertility. Apart from the quality of zygote, the endometrium related process, including the decidualization, the implantation, the remodeling of uterus spiral artery and the endometrial receptivity, are all closely linked to a successful pregnancy. There's no doubt that the condition and function of endometrial or decidual stromal cells play a vital role, and autophagy activity of endometrial or decidual cells deserves more attention and may draw a new light on the way exploring the mechanisms of infertility.
Conclusion and perspectives
Autophagy is a conserved pathway in eukaryotic cells with the function to degrade misfolded proteins, aging organelles, and other damaged cell components, as well as participate in the regulation of biological activities such as proliferation, apoptosis, and immunoreaction. Endometrial cells undergo a dynamic autophagy in the periodic growth and shedding off during the menstrual cycle, and the autophagy level also changed in endometrium-related diseases, as well as the physiological processes including decidualization and atrophy. Although a lot has been done during the past years, much remains unknown related to the autophagy in endometrium. The researches about autophagy limit to the endometrium stromal cells and epithelial, while we believe that autophagy in endometrial immune cells was the same pivotal and even more important. What's more, autophagy influences different biological activities in the same disease or the same kind of cells, these effects may be mutually contradictory. The balance and relationship between these effects, or the synthetic role of autophagy at some certain conditions are still vague to our understanding. On the one side, autophagy could promote the drug resistance of tumor cells and the migration and invasion of ectopic endometrium; on the other side, autophagy may also induce the immune activation and cell apoptosis in the lesions area (Figure 1). There is no constant role of autophagy in different situation; at least the evidences were controversial related the function of autophagy even in the same diseases. For all this, there's no doubt that to determinate the law of autophagy is of great significance in improving the treatment and enhancing the medicine development in endometrium-related diseases.
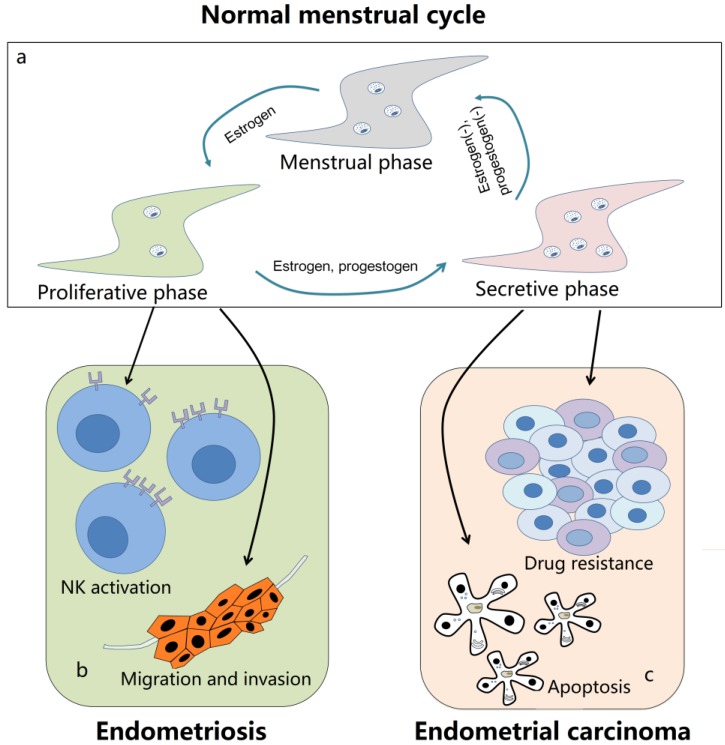
Figure 1.
The role of autophagy in endometrium-related diseases. (a) The level of autophagy in normal menstrual cycle is in a dynamic state. In the proliferative phase, the autophagy activity is relatively low in endometrium cells. When entering into the secretive phase, it significantly increases to the peak in late secretive phase under the control of estrogen and progestogen. And during the menstrual phase, it remains a middle level with the withdrawal of estrogen and progestogen. (b) Autophagy is decreased in endometriosis. On the one hand, the downregulated autophagy can inhibit the activation of NK cells and induce the immune escape of ectopic endometrial stromal cells; on the other hand, the autophagy in endometrial cells may contribute to its migration and invasion. (c) In endometrial carcinoma, the upregulated autophagy is related to its resistance to several drugs, including sorafenib, cisplatin, paclitaxel and so on. Meanwhile, autophagy may also lead to apoptosis of tumor cells and is essential for some other treatment methods, such as metformin and suberoylanilide hydroxamic acid. Autophagy is a double-edged sword in endometrium-related diseases and need to be further explored.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (NSFC) (No. 31671200, 91542108, 81471513 and 81471548), the Shanghai Rising-Star Program (No. 16QA1400800), the Oriented Project of Science and Technology Innovation from Key Lab. of Reproduction Regulation of NPFPC (CX2017-2), and the Program for Zhuoxue of Fudan University.
Author Contributions
Writing and editing of the manuscript and preparing the figures, S.L.Y.; editing the manuscript, H.Y.W. and D.J.L.; writing and editing of the manuscript and figures, M.Q.L.
References
- 1.Gwangwa MV, Joubert AM, Visagie MH. Crosstalk between the Warburg effect, redox regulation and autophagy induction in tumourigenesis. Cellular & Molecular Biology Letters. 2018;23(20):1–19. doi: 10.1186/s11658-018-0088-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhang Z, Lai Q, Li Y. et al. Acidic pH environment induces autophagy in osteoblasts. Scientific Reports. 2017;6(7):46161. doi: 10.1038/srep46161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhong Z, Sanchez-Lopez E, Karin M. Autophagy, Inflammation, and Immunity: A Troika Governing Cancer and Its Treatment. Cell. 2016;166(2):288–298. doi: 10.1016/j.cell.2016.05.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chun Y, Kim J. Autophagy: An Essential Degradation Program for Cellular Homeostasis and Life. Cells. 2018;7(12):1–26. doi: 10.3390/cells7120278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Konrad L, Kortum J, Nabham R. et al. Composition of the Stroma in the Human Endometrium and Endometriosis. Reprod Sci. 2018;25(7):1106–1115. doi: 10.1177/1933719117734319. [DOI] [PubMed] [Google Scholar]
- 6.Amalinei C, Pavaleanu I, Grigoras A. et al. The endometrial regeneration frontiers: from mechanisms to applications in regenerative medicine. Rom J Morphol Embryol. 2018;59(2):407–425. [PubMed] [Google Scholar]
- 7.Valentijn AJ, Palial K, Al-Lamee H. et al. SSEA-1 isolates human endometrial basal glandular epithelial cells: phenotypic and functional characterization and implications in the pathogenesis of endometriosis. Hum Reprod. 2013;28(10):2695–2708. doi: 10.1093/humrep/det285. [DOI] [PubMed] [Google Scholar]
- 8.Vanderstraeten A, Tuyaerts S, Amant F. The immune system in the normal endometrium and implications for endometrial cancer development. J Reprod Immunol. 2015;109:7–16. doi: 10.1016/j.jri.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 9.Zhou JZ, Way SS, Chen K. Immunology of the Uterine and Vaginal Mucosae. Trends Immunol. 2018;39(4):302–314. doi: 10.1016/j.it.2018.01.007. [DOI] [PubMed] [Google Scholar]
- 10.Choi J, Jo M, Lee E. et al. The role of autophagy in human endometrium. Biol Reprod. 2012;86(3):70. doi: 10.1095/biolreprod.111.096206. 1-10. [DOI] [PubMed] [Google Scholar]
- 11.Ruiz A, Rockfield S, Taran N. et al. Effect of hydroxychloroquine and characterization of autophagy in a mouse model of endometriosis. Cell Death Dis. 2016;7:e2059. doi: 10.1038/cddis.2015.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Choi J, Jo M, Lee E. et al. Differential induction of autophagy by mTOR is associated with abnormal apoptosis in ovarian endometriotic cysts. Mol Hum Reprod. 2014;20(4):309–317. doi: 10.1093/molehr/gat091. [DOI] [PubMed] [Google Scholar]
- 13.Zhou S, Zhao L, Yi T. et al. Menopause-induced uterine epithelium atrophy results from arachidonic acid/prostaglandin E2 axis inhibition-mediated autophagic cell death. Sci Rep. 2016;6:31408. doi: 10.1038/srep31408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Choi S, Shin H, Song H. et al. Suppression of autophagic activation in the mouse uterus by estrogen and progesterone. Journal of Endocrinology. 2014;221(1):39–50. doi: 10.1530/JOE-13-0449. [DOI] [PubMed] [Google Scholar]
- 15.Mei J, Zhu XY, Jin LP. et al. Estrogen promotes the survival of human secretory phase endometrial stromal cells via CXCL12/CXCR4 up-regulation-mediated autophagy inhibition. Hum Reprod. 2015;30(7):1677–1689. doi: 10.1093/humrep/dev100. [DOI] [PubMed] [Google Scholar]
- 16.Rhee JS, Saben JL, Mayer AL. et al. Diet-induced obesity impairs endometrial stromal cell decidualization: a potential role for impaired autophagy. Hum Reprod. 2016;31(6):1315–1326. doi: 10.1093/humrep/dew048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mei J, Zhou WJ, Zhu XY. et al. Suppression of autophagy and HCK signaling promotes PTGS2(high) FCGR3(-) NK cell differentiation triggered by ectopic endometrial stromal cells. Autophagy. 2018;14(8):1376–1397. doi: 10.1080/15548627.2018.1476809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang L, Liu Y, Xu Y. et al. The expression of the autophagy gene beclin-1 mRNA and protein in ectopic and eutopic endometrium of patients with endometriosis. Int J Fertil Steril. 2015;8(4):429–436. doi: 10.22074/ijfs.2015.4183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sui X, Li Y, Sun Y. et al. Expression and significance of autophagy genes LC3, Beclin1 and MMP-2 in endometriosis. Exp Ther Med. 2018;16(3):1958–1962. doi: 10.3892/etm.2018.6362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luo X, Cheng W, Wang S. et al. Autophagy Suppresses Invasiveness of Endometrial Cells through Reduction of Fascin-1. Biomed Res Int. 2018;2018:8615435. doi: 10.1155/2018/8615435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Allavena G, Carrarelli P, Del BB. et al. Autophagy is upregulated in ovarian endometriosis: a possible interplay with p53 and heme oxygenase-1. Fertil Steril. 2015;103(5):1244–1251.e1241. doi: 10.1016/j.fertnstert.2015.02.007. [DOI] [PubMed] [Google Scholar]
- 22.Liu H, Du Y, Zhang Z. et al. Autophagy contributes to hypoxia-induced epithelial to mesenchymal transition of endometrial epithelial cells in endometriosis. Biol Reprod. 2018;99(5):968–981. doi: 10.1093/biolre/ioy128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu H, Zhang Z, Xiong W. et al. Hypoxia-inducible factor-1alpha promotes endometrial stromal cells migration and invasion by upregulating autophagy in endometriosis. Reproduction. 2017;153(6):809–820. doi: 10.1530/REP-16-0643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zheng J, Luo X, Bao J, Decreased Expression of HOXA10 May Activate the Autophagic Process in Ovarian Endometriosis. Reprod Sci. 2018; 1933. 719118768704. [DOI] [PubMed] [Google Scholar]
- 25.Borodkina AV, Shatrova AN, Deryabin PI. et al. Tetraploidization or autophagy: The ultimate fate of senescent human endometrial stem cells under ATM or p53 inhibition. Cell Cycle. 2016;15(1):117–127. doi: 10.1080/15384101.2015.1121326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Borodkina AV, Shatrova AN, Deryabin PI. et al. Calcium alterations signal either to senescence or to autophagy induction in stem cells upon oxidative stress. Aging (Albany NY) 2016;8(12):3400–3418. doi: 10.18632/aging.101130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Du J, Zhu X, Guo R. et al. Autophagy induces G0/G1 arrest and apoptosis in menstrual blood-derived endometrial stem cells via GSK3-beta/beta-catenin pathway. Stem Cell Res Ther. 2018;9(1):330. doi: 10.1186/s13287-018-1073-0. 1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ran X, Zhou P, Zhang K. Autophagy plays an important role in stemness mediation and the novel dual function of EIG121 in both autophagy and stemness regulation of endometrial carcinoma JEC cells. Int J Oncol. 2017;51(2):644–656. doi: 10.3892/ijo.2017.4047. [DOI] [PubMed] [Google Scholar]
- 29.Zhang B, Zhou WJ, Gu CJ. et al. The ginsenoside PPD exerts anti-endometriosis effects by suppressing estrogen receptor-mediated inhibition of endometrial stromal cell autophagy and NK cell cytotoxicity. Cell Death Dis. 2018;9(5):574. doi: 10.1038/s41419-018-0581-2. 1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gu CJ, Cheng J, Zhang B. et al. Protopanaxadiol and metformin synergistically inhibit estrogen-mediated proliferation and anti-autophagy effects in endometrial cancer cells. Am J Transl Res. 2017;9(9):4071–4082. [PMC free article] [PubMed] [Google Scholar]
- 31.Deng L, Feng J, Broaddus RR. The novel estrogen-induced gene EIG121 regulates autophagy and promotes cell survival under stress. Cell Death Dis. 2010;1:e32. doi: 10.1038/cddis.2010.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Choi J, Jo M, Lee E. et al. Dienogest enhances autophagy induction in endometriotic cells by impairing activation of AKT, ERK1/2, and mTOR. Fertil Steril. 2015;104(3):655–664.e651. doi: 10.1016/j.fertnstert.2015.05.020. [DOI] [PubMed] [Google Scholar]
- 33.Zhang D, Li J, Xu G. et al. Follicle-stimulating hormone promotes age-related endometrial atrophy through cross-talk with transforming growth factor beta signal transduction pathway. Aging Cell. 2015;14(2):284–287. doi: 10.1111/acel.12278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sumarac-Dumanovic M, Apostolovic M, Janjetovic K. et al. Downregulation of autophagy gene expression in endometria from women with polycystic ovary syndrome. Mol Cell Endocrinol. 2017;440:116–124. doi: 10.1016/j.mce.2016.11.009. [DOI] [PubMed] [Google Scholar]
- 35.Chen Q, Gao R, Geng Y. et al. Decreased autophagy was implicated in the decreased apoptosis during decidualization in early pregnant mice. J Mol Histol. 2018;49(6):589–597. doi: 10.1007/s10735-018-9797-9. [DOI] [PubMed] [Google Scholar]
- 36.Yin B, Liu X, Guo SW. Caloric Restriction Dramatically Stalls Lesion Growth in Mice With Induced Endometriosis. Reprod Sci. 2018;25(7):1024–1036. doi: 10.1177/1933719118756755. [DOI] [PubMed] [Google Scholar]
- 37.Shi K, Wang Q, Su Y. et al. Identification and functional analyses of differentially expressed metabolites in early stage endometrial carcinoma. Cancer Sci. 2018;109(4):1032–1043. doi: 10.1111/cas.13532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lu H, Li S, Wu Q. Retinoic acid regulates endometriotic stromal cell growth through upregulation of Beclin1. Arch Gynecol Obstet. 2018;297(1):93–99. doi: 10.1007/s00404-017-4549-8. [DOI] [PubMed] [Google Scholar]
- 39.Song Y, Du Y, Zou W. et al. Involvement of impaired autophagy and mitophagy in Neuro-2a cell damage under hypoxic and/or high-glucose conditions. Sci Rep. 2018;8(1):3301. doi: 10.1038/s41598-018-20162-1. 1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang X, Ribeiro M, Iracheta-Vellve A. et al. Macrophage-specific HIF-1alpha contributes to impaired autophagic flux in non-alcoholic steatohepatitis. Hepatology. 2019;0(0):1–19. doi: 10.1002/hep.30215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bauckman KA, Haller E, Flores I. et al. Iron modulates cell survival in a Ras- and MAPK-dependent manner in ovarian cells. Cell Death Dis. 2013;4:e592. doi: 10.1038/cddis.2013.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ryter SW, Choi AM. Autophagy: An Integral Component of the Mammalian Stress Response. J Biochem Pharmacol Res. 2013;1(3):176–188. [PMC free article] [PubMed] [Google Scholar]
- 43.Filomeni G, De Zio D, Cecconi F. Oxidative stress and autophagy: the clash between damage and metabolic needs. Cell Death Differ. 2015;22(3):377–388. doi: 10.1038/cdd.2014.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Talarico C, Dattilo V, D'Antona L. et al. SI113, a SGK1 inhibitor, potentiates the effects of radiotherapy, modulates the response to oxidative stress and induces cytotoxic autophagy in human glioblastoma multiforme cells. Oncotarget. 2016;7(13):15868–84. doi: 10.18632/oncotarget.7520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bai J-A, Xu G-F, Yan L-J. et al. SGK1 inhibits cellular apoptosis and promotes proliferation via the MEK/ERK/p53 pathway in colitis. World Journal of Gastroenterology. 2015;21(20):6180–6193. doi: 10.3748/wjg.v21.i20.6180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Conza D, Mirra P, Cali G. et al. The SGK1 inhibitor SI113 induces autophagy, apoptosis, and endoplasmic reticulum stress in endometrial cancer cells. Journal of Cellular Physiology. 2017;232(12):3735–3743. doi: 10.1002/jcp.25850. [DOI] [PubMed] [Google Scholar]
- 47.Lang F, Pelzl L, Hauser S. et al. To die or not to die SGK1-sensitive ORAI/STIM in cell survival. Cell Calcium. 2018;74:29–34. doi: 10.1016/j.ceca.2018.05.001. [DOI] [PubMed] [Google Scholar]
- 48.Conza D, Mirra P, Cali G. et al. The SGK1 inhibitor SI113 induces autophagy, apoptosis, and endoplasmic reticulum stress in endometrial cancer cells. J Cell Physiol. 2017;232(12):3735–3743. doi: 10.1002/jcp.25850. [DOI] [PubMed] [Google Scholar]
- 49.Kanda R, Hiraike H, Wada-Hiraike O. et al. Expression of the glucagon-like peptide-1 receptor and its role in regulating autophagy in endometrial cancer. BMC Cancer. 2018;18(1):657. doi: 10.1186/s12885-018-4570-8. 1-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kao C, Chao A, Tsai CL. et al. Bortezomib enhances cancer cell death by blocking the autophagic flux through stimulating ERK phosphorylation. Cell Death Dis. 2014;5:e1510. doi: 10.1038/cddis.2014.468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.De U, Son JY, Sachan R. et al. A New Synthetic Histone Deacetylase Inhibitor, MHY2256, Induces Apoptosis and Autophagy Cell Death in Endometrial Cancer Cells via p53 Acetylation. Int J Mol Sci. 2018;19(9):2743. doi: 10.3390/ijms19092743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu H, Zhang L, Zhang X. et al. PI3K/AKT/mTOR pathway promotes progestin resistance in endometrial cancer cells by inhibition of autophagy. Onco Targets Ther. 2017;10:2865–2871. doi: 10.2147/OTT.S95267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang H, Li D, Li X. et al. Mammalian target of rapamycin inhibitor RAD001 sensitizes endometrial cancer cells to paclitaxel-induced apoptosis via the induction of autophagy. Oncol Lett. 2016;12(6):5029–5035. doi: 10.3892/ol.2016.5338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liu Y, Okamoto K. The TORC1 signaling pathway regulates respiration-induced mitophagy in yeast. Biochemical and Biophysical Research Communications. 2018;502(1):76–83. doi: 10.1016/j.bbrc.2018.05.123. [DOI] [PubMed] [Google Scholar]
- 55.Manzoni C, Mamais A, Dihanich S. et al. mTOR independent alteration in ULK1 Ser758 phosphorylation following chronic LRRK2 kinase inhibition. Bioscience Reports. 2018;38:1–12. doi: 10.1042/BSR20171669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rahman MA, Terasawa M, Mostofa MG. et al. The TORC1-Nem1/Spo7-Pah1/lipin axis regulates microautophagy induction in budding yeast. Biochemical and Biophysical Research Communications. 2018;504(2):505–512. doi: 10.1016/j.bbrc.2018.09.011. [DOI] [PubMed] [Google Scholar]
- 57.Tyler JK, Johnson JE. The role of autophagy in the regulation of yeast life span. Annals of the New York Academy of Sciences. 2018;1418(1):31–43. doi: 10.1111/nyas.13549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhou WJ, Chang KK, Wu K. et al. Rapamycin Synergizes with Cisplatin in Antiendometrial Cancer Activation by Improving IL-27-Stimulated Cytotoxicity of NK Cells. Neoplasia. 2018;20(1):69–79. doi: 10.1016/j.neo.2017.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yan L, Zhou J, Gao Y. et al. Regulation of tumor cell migration and invasion by the H19/let-7 axis is antagonized by metformin-induced DNA methylation. Oncogene. 2015;34(23):3076–3084. doi: 10.1038/onc.2014.236. [DOI] [PubMed] [Google Scholar]
- 60.Tan BK, Adya R, Chen J. et al. Metformin Treatment Exerts Antiinvasive and Antimetastatic Effects in Human Endometrial Carcinoma Cells. The Journal of Clinical Endocrinology & Metabolism. 2011;96(3):808–816. doi: 10.1210/jc.2010-1803. [DOI] [PubMed] [Google Scholar]
- 61.Zhuo Z, Wang A, Yu H. Metformin targeting autophagy overcomes progesterone resistance in endometrial carcinoma. Arch Gynecol Obstet. 2016;294(5):1055–1061. doi: 10.1007/s00404-016-4148-0. [DOI] [PubMed] [Google Scholar]
- 62.Takahashi A, Kimura F, Yamanaka A. et al. Metformin impairs growth of endometrial cancer cells via cell cycle arrest and concomitant autophagy and apoptosis. Cancer Cell Int. 2014;14:53. doi: 10.1186/1475-2867-14-53. 1-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tsai HH, Lai HY, Chen YC. et al. Metformin promotes apoptosis in hepatocellular carcinoma through the CEBPD-induced autophagy pathway. Oncotarget. 2017;8(8):13832–13845. doi: 10.18632/oncotarget.14640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Li J, Gui Y, Ren J. et al. Metformin Protects Against Cisplatin-Induced Tubular Cell Apoptosis and Acute Kidney Injury via AMPKalpha-regulated Autophagy Induction. Sci Rep. 2016;6:23975. doi: 10.1038/srep23975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.d'Abadie P, Borbath I, Goffette P. et al. Sorafenib Reduced Significantly Heptopulmonary Shunt in a Large Hepatocelullar Carcinoma. Clinical Nuclear Medicine. 2019;44(1):70–71. doi: 10.1097/RLU.0000000000002369. [DOI] [PubMed] [Google Scholar]
- 66.Wang H-T, Xia M. A meta-analysis of efficacy and safety of sorafenib versus other targeted agents for metastatic renal cell carcinoma. Medicine. 2019;98(1):e13779–e13779. doi: 10.1097/MD.0000000000013779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Eritja N, Chen BJ, Rodriguez-Barrueco R. et al. Autophagy orchestrates adaptive responses to targeted therapy in endometrial cancer. Autophagy. 2017;13(3):608–624. doi: 10.1080/15548627.2016.1271512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Li Y, Wang X, Wang X. et al. PDCD4 suppresses proliferation, migration and invasion of endometrial cells by inhibiting autophagy and NF-kappaB/MMP2/MMP9 signal pathway. Biol Reprod. 2018;99(2):360–372. doi: 10.1093/biolre/ioy052. [DOI] [PubMed] [Google Scholar]
- 69.Lin Q, Wang Y, Chen D. et al. Cisplatin regulates cell autophagy in endometrial cancer cells via the PI3K/AKT/mTOR signalling pathway. Oncol Lett. 2017;13(5):3567–3571. doi: 10.3892/ol.2017.5894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tsubamoto H, Inoue K, Sakata K. et al. Itraconazole Inhibits AKT/mTOR Signaling and Proliferation in Endometrial Cancer Cells. Anticancer Res. 2017;37(2):515–519. doi: 10.21873/anticanres.11343. [DOI] [PubMed] [Google Scholar]
- 71.Wu CH, Chen HY, Wang CW. et al. Isoliquiritigenin induces apoptosis and autophagy and inhibits endometrial cancer growth in mice. Oncotarget. 2016;7(45):73432–73447. doi: 10.18632/oncotarget.12369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kang S, Kim JE, Song NR. et al. The ginsenoside 20-O-beta-D-glucopyranosyl-20(S)-protopanaxadiol induces autophagy and apoptosis in human melanoma via AMPK/JNK phosphorylation. PLoS One. 2014;9(8):e104305. doi: 10.1371/journal.pone.0104305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wang C, Liu B. miR-101-3p induces autophagy in endometrial carcinoma cells by targeting EZH2. Arch Gynecol Obstet. 2018;297(6):1539–1548. doi: 10.1007/s00404-018-4768-7. [DOI] [PubMed] [Google Scholar]
- 74.Martin-Mateos R, De Assuncao TM, Pablo Arab J. et al. Enhancer of Zeste Homologue 2 Inhibition Attenuates TGF-beta Dependent Hepatic Stellate Cell Activation and Liver Fibrosis. Cellular and Molecular Gastroenterology and Hepatology. 2019;7(1):197–209. doi: 10.1016/j.jcmgh.2018.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wei FZ, Cao Z, Wang X. et al. Epigenetic regulation of autophagy by the methyltransferase EZH2 through an MTOR-dependent pathway. Autophagy. 2015;11(12):2309–2322. doi: 10.1080/15548627.2015.1117734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Xu TX, Zhao SZ, Dong M. et al. Hypoxia responsive miR-210 promotes cell survival and autophagy of endometriotic cells in hypoxia. Eur Rev Med Pharmacol Sci. 2016;20(3):399–406. [PubMed] [Google Scholar]
- 77.Ran X, Yang J, Liu C. et al. MiR-218 inhibits HMGB1-mediated autophagy in endometrial carcinoma cells during chemotherapy. Int J Clin Exp Pathol. 2015;8(6):6617–6626. [PMC free article] [PubMed] [Google Scholar]
- 78.Sun MY, Zhu JY, Zhang CY. et al. Autophagy regulated by lncRNA HOTAIR contributes to the cisplatin-induced resistance in endometrial cancer cells. Biotechnol Lett. 2017;39(10):1477–1484. doi: 10.1007/s10529-017-2392-4. [DOI] [PubMed] [Google Scholar]
- 79.Liu H, Zhang Z, Xiong W. et al. Long non-coding RNA MALAT1 mediates hypoxia-induced pro-survival autophagy of endometrial stromal cells in endometriosis. J Cell Mol Med. 2019;23(1):439–452. doi: 10.1111/jcmm.13947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zeng X, Yan T, Schupp JE. et al. DNA mismatch repair initiates 6-thioguanine-induced autophagy through p53 activation in human tumor cells. Clin Cancer Res. 2007;13(4):1315–1321. doi: 10.1158/1078-0432.CCR-06-1517. [DOI] [PubMed] [Google Scholar]
- 81.Xu HL, Xu J, Zhang SS. et al. Temperature-sensitive heparin-modified poloxamer hydrogel with affinity to KGF facilitate the morphologic and functional recovery of the injured rat uterus. Drug Deliv. 2017;24(1):867–881. doi: 10.1080/10717544.2017.1333173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yang D, Jiang T, Liu J. et al. CREB3 regulatory factor -mTOR-autophagy regulates goat endometrial function during early pregnancy. Biol Reprod. 2018;98(5):713–721. doi: 10.1093/biolre/ioy044. [DOI] [PubMed] [Google Scholar]
- 83.Yang D, Jiang T, Liu J. et al. Hormone regulates endometrial function via cooperation of endoplasmic reticulum stress and mTOR-autophagy. J Cell Physiol. 2018;233(9):6644–6659. doi: 10.1002/jcp.26315. [DOI] [PubMed] [Google Scholar]
- 84.Lebovitz CB, Robertson AG, Goya R. et al. Cross-cancer profiling of molecular alterations within the human autophagy interaction network. Autophagy. 2015;11(9):1668–1687. doi: 10.1080/15548627.2015.1067362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sivridis E, Giatromanolaki A, Liberis V. et al. Autophagy in endometrial carcinomas and prognostic relevance of 'stone-like' structures (SLS): what is destined for the atypical endometrial hyperplasia? Autophagy. 2011;7(1):74–82. doi: 10.4161/auto.7.1.13947. [DOI] [PubMed] [Google Scholar]
- 86.Hahne JC, Meyer SR, Dietl J. et al. The effect of Cordyceps extract and a mixture of Ganoderma lucidum/Agaricus Blazi Murill extract on human endometrial cancer cell lines in vitro. Int J Oncol. 2014;45(1):373–382. doi: 10.3892/ijo.2014.2414. [DOI] [PubMed] [Google Scholar]
- 87.Fukuda T, Oda K, Wada-Hiraike O. et al. The anti-malarial chloroquine suppresses proliferation and overcomes cisplatin resistance of endometrial cancer cells via autophagy inhibition. Gynecol Oncol. 2015;137(3):538–545. doi: 10.1016/j.ygyno.2015.03.053. [DOI] [PubMed] [Google Scholar]
- 88.Liu S, Li X. Autophagy inhibition enhances sensitivity of endometrial carcinoma cells to paclitaxel. Int J Oncol. 2015;46(6):2399–2408. doi: 10.3892/ijo.2015.2937. [DOI] [PubMed] [Google Scholar]
- 89.Singh AK, Bishayee A, Pandey AK. Targeting Histone Deacetylases with Natural and Synthetic Agents: An Emerging Anticancer Strategy. Nutrients. 2018;10(6):731. doi: 10.3390/nu10060731. 1-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Orfanelli T, Jeong JM, Doulaveris G. et al. Involvement of autophagy in cervical, endometrial and ovarian cancer. Int J Cancer. 2014;135(3):519–528. doi: 10.1002/ijc.28524. [DOI] [PubMed] [Google Scholar]
- 91.Giatromanolaki A, Koukourakis MI, Koutsopoulos A. et al. High Beclin 1 expression defines a poor prognosis in endometrial adenocarcinomas. Gynecol Oncol. 2011;123(1):147–151. doi: 10.1016/j.ygyno.2011.06.023. [DOI] [PubMed] [Google Scholar]
- 92.Zhan L, Li J, Wei B. Autophagy in endometriosis: Friend or foe? Biochem Biophys Res Commun. 2018;495(1):60–63. doi: 10.1016/j.bbrc.2017.10.145. [DOI] [PubMed] [Google Scholar]
- 93.Yang HL, Mei J, Chang KK. et al. Autophagy in endometriosis. Am J Transl Res. 2017;9(11):4707–4725. [PMC free article] [PubMed] [Google Scholar]
- 94.Erekat NS. Autophagy precedes apoptosis among at risk cerebellar Purkinje cells in the shaker mutant rat: an ultrastructural study. Ultrastructural Pathology. 2018;42(2):162–169. doi: 10.1080/01913123.2018.1424744. [DOI] [PubMed] [Google Scholar]
- 95.Yu JJ, Sun HT, Zhang ZF. et al. IL15 promotes growth and invasion of endometrial stromal cells and inhibits killing activity of NK cells in endometriosis. Reproduction. 2016;152(2):151–160. doi: 10.1530/REP-16-0089. [DOI] [PubMed] [Google Scholar]
- 96.Matsuzaki S, Pouly J-L, Canis M. In vitro and in vivo effects of MK2206 and chloroquine combination therapy on endometriosis: autophagy may be required for regrowth of endometriosis. British Journal of Pharmacology. 2018;175(10):1637–1653. doi: 10.1111/bph.14170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mihalyi A, Gevaert O, Kyama CM. et al. Non-invasive diagnosis of endometriosis based on a combined analysis of six plasma biomarkers. Hum Reprod. 2010;25(3):654–664. doi: 10.1093/humrep/dep425. [DOI] [PubMed] [Google Scholar]
- 98.Ren Y, Mu L, Ding X. et al. Decreased expression of Beclin 1 in eutopic endometrium of women with adenomyosis. Arch Gynecol Obstet. 2010;282(4):401–406. doi: 10.1007/s00404-009-1280-0. [DOI] [PubMed] [Google Scholar]
- 99.Feng L, Li J, Yang L. et al. Tamoxifen activates Nrf2-dependent SQSTM1 transcription to promote endometrial hyperplasia. Theranostics. 2017;7(7):1890–1900. doi: 10.7150/thno.19135. [DOI] [PMC free article] [PubMed] [Google Scholar]