Abstract
Objective
Chronic constriction injury (CCI) of the sciatic nerve is a peripheral nerve injury widely used to induce mononeuropathy. This study used machine learning methods to identify the best gait analysis parameters for evaluating peripheral nerve injuries.
Methods
Twenty-eight male Wistar rats (weighing 270±10 g), were used in the present study and divided into the following 4 groups: CCI with 4 ligatures around the sciatic nerve (CCI-4L; n=7), a modified CCI model with 1 ligature (CCI-1L; n=7), a sham group (n=7), and a healthy control group (n=7). All rats underwent gait analysis 7 and 28 days postinjury. The data were evaluated using Kinovea and WeKa software (machine learning and neural networks).
Results
In the machine learning analysis of the experimental groups, the pre-swing (PS) angle showed the highest ranking in all 3 analyses (sensitivity, specificity, and area under the receiver operating characteristics curve using the Naive Bayes, k-nearest neighbors, radial basis function classifiers). Initial contact (IC), step length, and stride length also performed well. Between 7 and 28 days after injury, there was an increase in the total course time, step length, stride length, stride speed, and IC, and a reduction in PS and IC-PS. Statistically significant differences were found between the control group and experimental groups for all parameters except speed. Interactions between time after injury and nerve injury type were only observed for IC, PS, and IC-PS.
Conclusion
PS angle of the ankle was the best gait parameter for differentiating nonlesions from nerve injuries and different levels of injury.
Keywords: Peripheral nerve injury, Mononeuropathy, Chronic constriction injury, Sciatic nerve, Motor deficits functions, Gait analysis
INTRODUCTION
Animal research is an integral and indispensable part of the development of modern medicine, as research material in neurosciences, studies related to neurological disorders in humans reproduced in experimental animal model [1-5]. In this way, the experimental model favors the representation of specific standardized events and has aroused great interest in the knowledge of histopathological, biochemical, behavioral and functional changes, mainly in the search for prevention and treatment methods [5].
Injuries to the central (variety of causes, including trauma, stroke, tumor, substance abuse, or degenerative neurological diseases) and peripheral nervous system (PNS) can substantially affect a person's long-term quality of life, resulting in significant cause of disability (e.g., loss of ambulation, balance, coordination, fine motor skills, strength, and endurance) and large social and economic burden [6,7].
According to Castillo-Galvan et al. [8], the trauma-associated PNS has a prevalence of 1.3%–2.8%, for example, regarding injuries in the central nervous system (CNS; traumatic brain), McAllister [9] describes a rate of 1 to 2 million Americans are injured each year. In the PNS, axonal regeneration is better when compared to CNS. Although the improvement of motor function occurs in patients suffering from spinal cord injuries, stroke patients, and head trauma, axons of CNS do not spontaneously regenerate after injury [10].
However, although easily assessed in humans, analyzing the progress of functional recovery in animal models for a range of conditions related to central and peripheral injury requires tests that are sensitive enough to characterize distinct aspects of motor function reorganization [11]. This situation is due to the fact that the lesions in the nervous system in rats result in varied deficits in motor control, such as skilled reaching, including the ability to reach, grasp, and eat a piece of food reach eat and skilled walking, this condition is associated with a remarkable recovery of locomotion, making it difficult to analyze and distinguish between true recovery and compensatory changes [12-14].
With regard to functional assessment, to increase the effectiveness of tests, they should allow for descriptive measures of motor function and be sensitive to changes with the ability to distinguish details related to the different stages of functional recovery and compensatory alterations after CNS injury or PNS [11,14].
This method of reproducible experimental functional gait analysis in rats can be performed by temporal and spatial parameters evaluation, which offer reliability when applied in several stages of the experiment and can be used to help the researcher in the evaluation process on dysfunctions after nervous system injury [15].
In this context, machine learning was used through artificial neural networks as a mathematical model inspired by the neural structure of intelligent organisms to acquire knowledge through experience with the best parameters and attributes to differentiate gait [16].
This mechanical process requires a coordinated function, which involves sensory input, motor response, and cortical integration. In this aspect to study it through computerized analysis, it is possible to generate objective and reliable data on the performance of the members [17]. The obtaining of such data derives from numerical/quantitative characteristics of the computerized images. Medical diagnostic logic via image analysis can be objectively coded and consistently applied in the routine of repetitive tasks [18].
The use of machine learning and neural networks allows a more refined study to identify ideal parameters on clinical conditions differentiation. Therefore, our objective is, through the use of machine learning methods, to identify the best parameters in the gait analysis for evaluation of peripheral nerve lesions.
MATERIALS AND METHODS
1. Study Design
Prospective, observational and longitudinal study.
2. Animals
Twenty-eight male Wistar rats (weighing 270±10 g), were used in the present study. All protocols were in compliance with the recommendations of the Committee for Ethics in Animal Experimentation (CETEA) of the Ribeirão Preto Medical School of the University of São Paulo (FMRP-USP) (Process 015/2005 and 036/2017), agreement with the Animal Research Ethics adopted by the National Council for Animal Experimentation Control (CONCEA), affiliated to the International Council for Laboratory Animal Science (ICLAS). All efforts were made to minimize suffering, as well as the number of animals used in this study.
3. Experimental Groups
A unilateral mononeuropathy was produced in rats using the chronic constriction injury (CCI) of the sciatic nerve by Bennet and Xie’s model [19] that consists in a surgical procedure by placing four loose ligatures around the sciatic nerve (CCI-4L group; n=7). In addiction, was done in the independent group a modified CCI model using a single loose ligature around the sciatic nerve (CCI-1L group; n=7). In addition, sham procedure without nerve injury by constriction was made as a sham group (n=7). A control group was formed by healthy rats without any injury or lesion (n=7).
4. CCI of the Sciatic Nerve – a Model of Peripheral Mononeuropathy
The peripheral nerve injuries in rodents result in various degrees of neuropathies. A unilateral mononeuropathy was produced in rats using the CCI of the sciatic nerve by Bennet and Xie’s model [20] that consists in a surgical procedure by placing 4 loose ligatures around the sciatic nerve (CCI-4L group). In addition, was done in the independent group a modified CCI model using a single loose ligature around the sciatic nerve (CCI-1L group).
Concerning the procedure, all groups of rats were anaesthetized by a mixture of ketamine/xylazine (92 mg/kg and 9.2 mg/kg, respectively, intraperitoneal). The right hind paw was shaved, moistened with a disinfectant and by a blunt dissection through the biceps femoris the sciatic nerve was exposed. At the level of the middle of the thigh and proximal to trifurcation, about 7 mm of the sciatic was freed from adhering tissues and four or one loose ligature (4.0 chromic gut). When used 4 ligatures were also were tied with about 1-mm spacing. The wound was then closed by suturing the muscle using chromic catgut with a continuous suture pattern. Finally, the skin was closed with 3.0 black braided silk sutures using a horizontal mattress suture pattern. As control group was performed the sham procedure. Sham surgery was done by exposing the sciatic nerve as described above, without ligatures neither nerve-damaging. All animals were then transferred to their home cage and left to recover. In our study, the rats were examined daily after surgery and about weekly until 28 days. During this inspection each animal was placed upon a table and carefully observed the condition of affected hind paw.
5. Gait Analysis
The rats walked on an acrylic platform (length×wide: 100 cm×5 cm), similar to CatWalk model, attached to a 20×20×20-cm chamber that serves as shelter after the walking end. The chamber is a strong stimulus attractive to animals.
Regarding the execution of the task of walking, according to previous protocols [21]. we first performed the adaptation and training of the animals during three consecutive days on the platform. Initially (day 1), the animals were placed in the housing chamber for 60 seconds, this period was enough to familiarize the animals with the environment. The chamber is a strong stimulus attractive to animals.
After this initial period of adaptation, during 2 days these animals made 3 attempts that consisted of reaching the housing chamber coming from a point increasingly distant from the chamber [21]. They learned to walk without exploratory pauses and at a steady speed in 2 or 3 trials. On the day of the evaluation, it was considered valid the attempt that the animal crossed the route without intercurrences or distractions. All analyzes were performed after 7 and 28 days of lesion induction (Fig. 1).
Fig. 1.
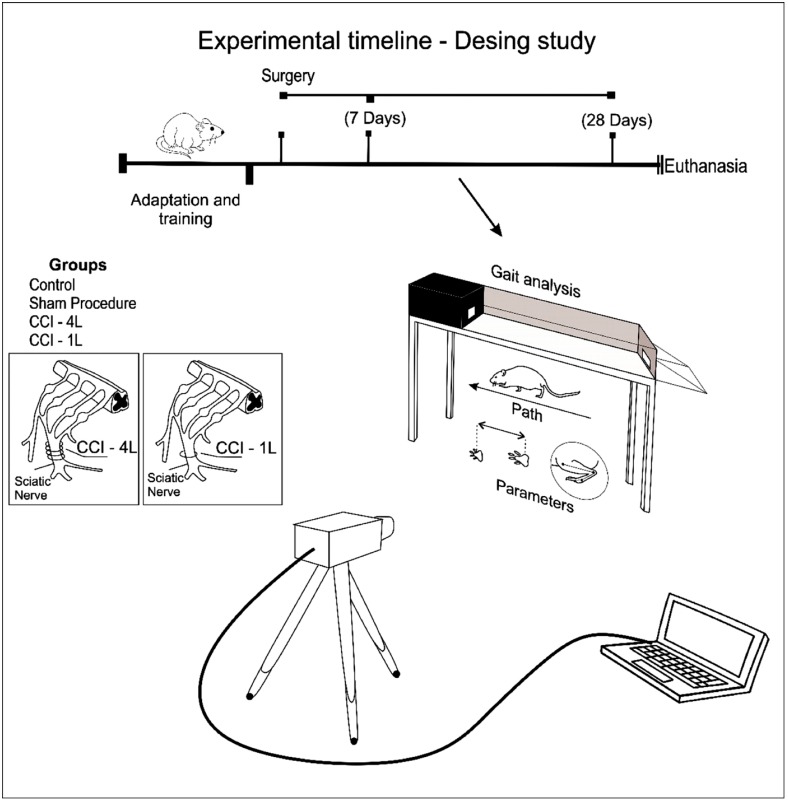
Schematic timeline of procedures and data collection during the study. Twenty-one male Wistar rats, distributed 3 experimental groups. Two groups underwent a unilateral mononeuropathy was produced in rats using the CCI of the sciatic (4 and 1 ligature); the third group was performed sham procedure. After 28 days, gait analysis was performed in the 3 groups, to evaluate the gait and selection of the parameters that best represent the functional recovery after peripheral nerve injury. CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve.
The gait was recorded by a video camera (Sony-DCR-SX85-Handycam-Camcorder-Silver) on the sagittal plane. The functional gait analysis was performed through video images using the Software Kinovea version 0.8.21. The videos were analyzed to select the frames of the gait cycle: initial contact (IC), medium support, final support, and balance for both posterior limbs. The mean angular values of the dorsiflexion movement on a posterior limb were calculated during IC and pre-swing (PS) gait stages (Fig. 2). The temporospatial parameters were calculated: mean step length, mean stride length, mean stride speed, total course time, and course speed. These gait parameters were measured at three different times during the total course and their mean values were calculated.
Fig. 2.
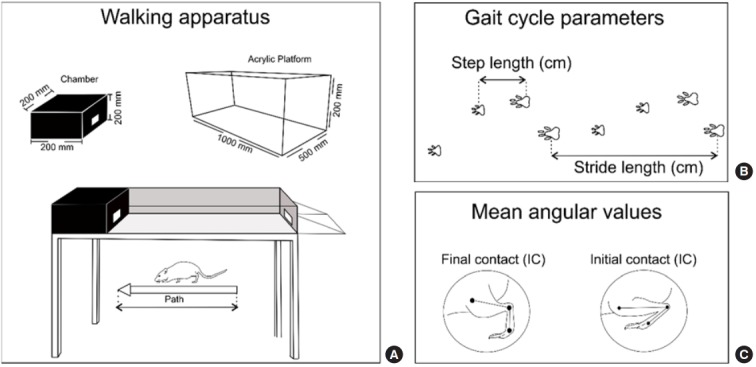
(A) Representation of the gait analysis platform with description of measurements (1,000 mm long, 50 mm wide), at the end of the platform there is a 200×200×200-mm chamber that serves as a shelter after the end of the platform walking. (B) Linear parameters of the gait. (C) Parameters angular gait.
6. Machine Learning
Selection of relevant features was performed by the ReliefF method [22], with a ranking search function. Relief basically estimates features according to the potential to distinguish nearest instances from each other in multiclass problems. ReliefF and the ranking function used in this work are implemented in the data mining and machine learning software [23] Weka v3.6.12, with a parameter often nearest instance. With the ranking search function, features are listed in descending order according to the relevance attributed by ReliefF.
Features were normalized in the range 0–1 for the classification process. We used 3 traditional machine learning algorithms to classify the instances: the Naive Bayes (NB) classifier, the k-nearest neighbors (KNN) method, and the radial basis function-based (RBF) artificial neural network. All algorithms used in this work were also implemented in Weka [23] v3.6.12.
The NB method is a probabilistic classifier based on Bayes’ theorem with features independence assumption [24].
The KNN, also known as an instance-based learning algorithm, is a nonparametric classifier that chooses the majority class among k neighbors to classify the unknown test instance [24]. In this work, the value of k varied from 1 to the number of instances.
The RBF artificial neural network generally has 3 layers of neurons: 1 input (whose neurons correspond to features), 1 hidden (whose neurons correspond to radial units), and 1 output (whose neurons correspond to classes). In this work, the RBF training was performed by a k-means clustering algorithm to provide the radial units [20]. The number of hidden layer neurons varied from 1 to the number of training instances.
The number of features selected for the classification varied from 1 to the all 9 features ranked by the ReliefF. The classification was assessed by the area under the receiver operating characteristic (ROC) curve (AUC), and the parameters of sensitivity and specificity, using the leave-one-out validation method.
7. Statistical Analysis
Kolmogorov-Smirnov test was used to verify the sample distribution. One-way analysis of variance with Bonferroni post hoc test has compared the gait parameters between control, bennett, and CCI groups. We used GraphPad Prism v.5 (GraphPad Software, San Diego, CA, USA) for statistical analysis.
RESULTS
1. Influence of Longitudinal Analysis: 7 and 28 Days After Injury
Table 1 presents the values of the temporospatial and angular gait cycle parameters for the control, sham, CCI-1L and CCI-4L groups expressed as a mean and standard deviation. Fig. 3 shows the graphs comparing gait parameter changes between 7 and 28 days. Except for the variable Speed (p=0.18), there was interference of the time variable between period 7- and 28-day follow-up for all other parameters. Within this period, there was an increase in the total course time (p=0.03), step length (p=0.0002), stride length (p=0.0003), stride speed, and a reduction in PS (p=0.002) and IC-PS (p=0.0001).
Table 1.
Gait cycle parameters of rats with and without peripheral nerve injury on 7 and 28 days after injury
| Parameter | Days | Control | Sham | CCI-1L | CCI-4L |
|---|---|---|---|---|---|
| Total course time (sec) | 7 | 5.56 ± 1.89 | 4.88 ± 1.79 | 4.75 ± 1.32 | 5.23 ± 1.61 |
| 28 | 7.05 ± 3.04 | 7.16 ± 4.54 | 5.30 ± 2.42 | 8.60 ± 5.24 | |
| Course speed (cm/sec) | 7 | 19.49 ± 5.82 | 23.25 ± 9.28 | 22.87 ± 8.13 | 21.28 ± 8.90 |
| 28 | 16.88 ± 7.69 | 18.64 ± 10.59 | 22.12 ± 9.64 | 16.12 ± 9.14 | |
| Step length (cm) | 7 | 4.62 ± 0.73 | 8.31 ± 1.00 | 6.71 ± 9.98 | 6.89 ± 0.28 |
| 28 | 6.72 ± 1.26 | 8.56 ± 1.03 | 7.79 ± 1.07 | 8.38 ± 1.53 | |
| Stride length (cm) | 7 | 9.89 ± 1.49 | 16.24 ± 1.93 | 12.39 ± 3.19 | 13.96 ± 0.46 |
| 28 | 13.60 ± 4.28 | 17.05 ± 2.08 | 15.57 ± 2.25 | 16.81 ± 3.30 | |
| Stride speed (cm/sec) | 7 | 15.44 ± 5.77 | 56.56 ± 6.16 | 28.30 ± 22.27 | 39.93 ± 9.06 |
| 28 | 23.29 ± 10.08 | 26.14 ± 17.31 | 28.32 ± 8.49 | 22.10 ± 9.75 | |
| Initial contact angle/IC (º) | 7 | 64.28 ± 3.03 | 53.09 ± 22.50 | 29.16 ± 5.64 | 31.85 ± 13.51 |
| 28 | 65.66 ± 11.47 | 61.71 ± 18.75 | 70.28 ± 15.12 | 62.09 ± 21.44 | |
| Pre-swing angle/PS (º) | 7 | 95.57 ± 7.98 | 86.71 ± 14.60 | 49.94 ± 17.11 | 41.71 ± 24.12 |
| 28 | 90.38 ± 15.55 | 64.85 ± 19.28 | 38.89 ± 8.03 | 24.47 ± 8.23 | |
| IC-PS | 7 | 31.28 ± 5.99 | 33.61 ± 11.53 | 20.77 ± 12.98 | 9.85 ± 16.81 |
| 28 | 24.71 ± 7.35 | 3.14 ± 31.09 | -31.39 ± 14.0 | -37.61 ± 24.80 |
Values are presented as mean±standard deviation.
CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve; IC, initial contact; PS, pre-swing.
Fig. 3.
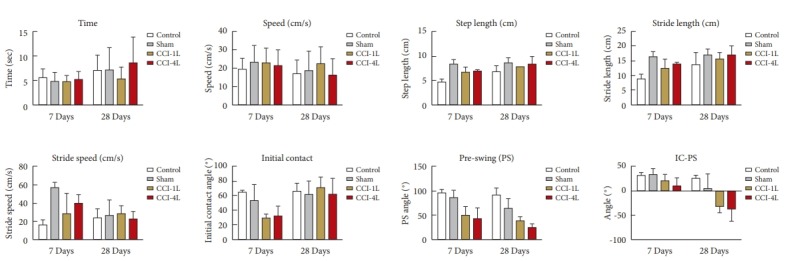
Comparison of gait parameters (temporal-spatial and angular) between sham, CCI-4L and CCI-1L groups. CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve; IC, initial contact; PS, pre-swing. *p<0.05, statistically significant difference.
2. Influence of Nerve Injury Type
There was a difference between the type of injury for the stride speed (p=0.0001), IC (p=0.01), PS (p=0.0001), and IP-PS (p=0.0001). The control group was statistically different from the sham, CCI-1L, and CCI-4L groups for all above mentioned gait parameters.
In the Bonferroni posttest, the variables step length, stride length, stride speed, and IC were different between the sham group and the injured groups CCI-1L and CCI-4L only 7 days after the injury (p<0.05). With 28 days of injury no differences were found. Regarding the PS and IC-PS parameters there were differences between Sham and the injured groups at 7 and 28 days after injury. In all parameters, the sham group obtained higher values in relation to the injured groups (Fig. 3).
There were no differences in the values of CCI-1L and CCI-4L in any of the gait parameters used. Regarding the interaction between the injury time factors (7 or 28 days) and the type of injury, it was possible to observe time influence on the type of injury only in the IC, PS and IC-PS variables.
3. Machine Learning Analysis
Tables 2 and 3 present the ranking values of the best parameters obtained through machine learning for comparison between experimental groups.
Table 2.
Ranking of gait parameters selected by the RelieF method to implement machine learning
| Ranking | Control vs. sham |
Control vs. CCI-1L |
Control vs. CCI-4L |
Sham vs. CCI-4L |
Sham vs. CCI-1L |
CCI-1L vs. CCI-4L |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | |
| 1 | 0.5834 | Stride speed | 0.6417 | Initial contact | 0.7336 | Pre-swing | 0.37005 | Pre-swing | 0.27464 | Pre-swing | 0.13176 | Pre-swing |
| 2 | 0.4864 | Step length | 0.467 | Pre-swing | 0.5614 | Initial contact | 0.01873 | Step length | 0.03333 | IC-PS | 0.02508 | Time |
| 3 | 0.4623 | Stride length | 0.2934 | Step length | 0.5383 | Stride length | 0.01341 | IC-PS | 0.02410 | Stride length | 0.01128 | Step length |
| 4 | 0.1493 | Initial contact | 0.1342 | Stride length | 0.5227 | Step length | -0.00217 | Stride speed | -0.00117 | Stride speed | 0.00269 | Speed |
| 5 | 0.0607 | Pre-swing | 0.1291 | IC-PS | 0.4073 | Stride speed | -0.00115 | Stride length | -0.00654 | Initial contact | 0.00113 | Stride speed |
| 6 | 0.0192 | IC-PS | 0.0402 | Stride speed | 0.3884 | IC-PS | -0.00027 | Speed | -0.02065 | Time | -0.01332 | Initial contact |
| 7 | -0.0166 | Speed | -0.0361 | Speed | -0.0321 | Speed | -0.00029 | Initial contact | -0.03449 | Speed | -0.01776 | Stride length |
| 8 | -0.0166 | Time | -0.047 | Time | -0.0331 | Time | -0.0332 | Time | -0.08334 | Step length | -0.01776 | IC-PS |
This analysis was performed on 7 days after nerve injury.
CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve; IC, initial contact; PS, pre-swing.
Table 3.
Ranking of gait parameters selected by the RelieF method to implement machine learning
| Ranking | Control vs. sham |
Control vs. CCI-1L |
Control vs. CCI-1L |
Sham vs. CCI-4L |
Sham vs. CCI-1L |
CCI-1L vs. CCI-4L |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | Value | Parameter | |
| 1 | 0.117579 | Step length | 0.45817 | Pre-swing | 0.52193 | Pre-swing | 0.2996 | Pre-swing | 0.188 | Pre-swing | 0.10775 | Pre-swing |
| 2 | 0.085039 | Pre-swing | 0.01465 | IC-PS | 0.02950 | Step length | 0.0814 | IC-PS | 0.1015 | IC-PS | 0.02779 | Step length |
| 3 | 0.01670 | Stride length | -0.00123 | Initial contact | 0.01229 | IC-PS | -0.0145 | Stride speed | 0.0241 | Stride length | 0.0274 | Stride length |
| 4 | -0.00009 | Initial contact | -0.00138 | Step length | 0.01046 | Stride length | -0.0275 | Speed | 0.0219 | Step length | 0.00471 | Time |
| 5 | -0.00220 | IC-PS | -0.0247 | Stride length | -0.00029 | Time | -0.031 | Time | -0.0117 | Stride speed | 0.00113 | Speed |
| 6 | -0.01014 | Stride speed | -0.01402 | Speed | -0.00096 | Initial contact | -0.0332 | Stride length | -0.018 | Initial contact | -0.01683 | IC-PS |
| 7 | -0.03043 | Time | -0.02957 | Time | -0.03006 | Speed | -0.0345 | Initial contact | -0.0242 | Time | -0.02671 | Stride speed |
| 8 | -0.03574 | Speed | -0.04865 | Stride speed | -0.05391 | Stride speed | -0.0428 | Step length | -0.0361 | Speed | -0.02766 | Initial contact |
This analysis was performed on 28-day follow-up.
CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve; IC, initial contact; PS, pre-swing.
At 7 days after the injury (Table 2), we can see that PS, step length and stride length were the parameters that presented the highest rankings in the analysis between groups. The PS angle parameter was the one that presented the highest values of overall ranking, showing the best parameter to differentiate the different injury types (sham, CCI-1L, and CCI-4L). However, to differentiate the control group from the injured groups, stride length was better to differentiate control and sham, whereas IC was better to differentiate between the control and the CCI-1L groups.
At 28 days after the injury (Table 3), we can see that PS, step length, and IC-PS were the parameters that presented the highest rankings most frequently in the analysis between groups. PS angle was the best parameter to differentiate the control, sham and the 2 injury type groups. Just to differentiate the sham versus control group, stride length proved to be the best parameter.
Tables 4 and 5 show the values of sensitivity, specificity, and area under the ROC curve calculated using the classifiers NB, KNN, and RBF. These algorithms selected up until 7 parameters (features) with the best ranking in descending order (Tables 2, 3) for analyzes between groups.
Table 4.
Sensitivity, specificity, and area under the ROC curve values for detection of peripheral nerve injury based on gait parameters evaluated 7 days after nerve injury
| Group | AUC | Sensitivity | Specificity | Features |
|---|---|---|---|---|
| Control vs. sham | ||||
| NB | 1.00 | 1.00 | 1.00 | 1 |
| KNN | 1.00 | 1.00 | 1.00 | 1 |
| RBF | 1.00 | 1.00 | 1.00 | 1 |
| Control vs. CCI-1L | ||||
| NB | 1.00 | 1.00 | 1.00 | 1 |
| KNN | 1.00 | 1.00 | 1.00 | 1 |
| RBF | 1.00 | 1.00 | 1.00 | 1 |
| Control vs. CCI-4L | ||||
| NB | 1.00 | 1.00 | 1.00 | 1 |
| KNN | 1.00 | 1.00 | 1.00 | 1 |
| RBF | 1.00 | 1.00 | 1.00 | 1 |
| Sham vs. CCI-1L | ||||
| NB | 0.92 | 1.00 | 0.83 | 3 |
| KNN | 0.92 | 0.83 | 0.83 | 4 |
| RBF | 0.83 | 0.83 | 0.83 | 2 |
| Sham vs. CCI-4L | ||||
| NB | 0.88 | 0.83 | 0.50 | 6 |
| KNN | 1.00 | 0.83 | 1.00 | 7 |
| RBF | 0.83 | 0.83 | 0.83 | 1 |
| CCI-4L vs. CCI-1L | ||||
| NB | 0.83 | 0.83 | 0.83 | 1 |
| KNN | 0.84 | 0.83 | 0.67 | 1 |
| RBF | 0.92 | 0.83 | 0.67 | 1 |
ROC, receiver operating characteristic; AUC, area under the curve; NB, Naive Bayes; KNN, k-nearest neighbors; RBF, radial basis function; CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve.
Table 5.
Sensitivity, specificity and area under the ROC curve values for detection of peripheral nerve injury based on gait parameters evaluated 28 days after nerve injury
| Group | AUC | Sensitivity | Specificity | Features |
|---|---|---|---|---|
| Control vs. sham | ||||
| NB | 0.92 | 0.83 | 1.00 | 2 |
| KNN | 0.89 | 0.83 | 0.83 | 3 |
| RBF | 0.72 | 0.67 | 0.67 | 1 |
| Control vs. CCI-1L | ||||
| NB | 1.00 | 0.83 | 1.00 | 1 |
| KNN | 1.00 | 0.83 | 1.00 | 1 |
| RBF | 1.00 | 1.00 | 1.00 | 1 |
| Control vs. CCI-4L | ||||
| NB | 1.00 | 1.00 | 1.00 | 1 |
| KNN | 1.00 | 1.00 | 1.00 | 1 |
| RBF | 1.00 | 0.83 | 1.00 | 1 |
| Sham vs. CCI-1L | ||||
| NB | 0.83 | 0.83 | 0.83 | 1 |
| KNN | 0.92 | 1.00 | 0.83 | 3 |
| RBF | 0.89 | 0.83 | 0.83 | 1 |
| Sham vs. CCI-4L | ||||
| NB | 0.81 | 0.83 | 0.83 | 1 |
| KNN | 0.85 | 0.83 | 0.50 | 3 |
| RBF | 0.83 | 0.68 | 0.83 | 3 |
| CCI-4L vs. CCI-1L | ||||
| NB | 0.89 | 0.67 | 0.83 | 1 |
| KNN | 0.83 | 1.00 | 0.83 | 1 |
| RBF | 0.92 | 0.83 | 0.83 | 3 |
ROC, receiver operating characteristic; AUC, area under the curve; NB, Naive Bayes; KNN, k-nearest neighbors; RBF, radial basis function; CCI, chronic constriction injury; CCI-1L, modified CCI model with 1 ligature; CCI-4L, CCI with 4 ligatures around the sciatic nerve.
In Table 4 we observed that using only the best parameter (with better ranking) it was possible to completely differentiate the control group from the others (maximum sensitivity and specificity) after 7 days of surgery. In the differentiation of the experimental groups, the values remained good but fell somewhat in relation to the control group. The use of the PS parameter guaranteed satisfactory AUC values and high sensitivity for the three classifiers, especially on nerve injury differentiation (CCI-1L vs. CCI-4L). Then, step length and IC were the next parameters that highlighted in the 7 days analyzes.
In the 28-day analysis (Table 5), we can see that specificity and sensitivity values fell in relation to the tests performed 7 days after the injury. Again, PS and step length were the best parameters for group differentiation. However, whereas in the 7-day analysis several parameters were necessary in order to differentiate the sham group from the nerve injury groups, after 28 days of injury, one to three parameters were required. The differentiation between CCI-1L and CCI-4L was more specific 28 days after injury. In general, regarding machine learning, there were no significant changes in the use of parameters between analyzes of 7 or 28 days after injury, except to compare the 2 types of nerve injury.
DISCUSSION
Through a prediction method based on machine learning and artificial neural networks, our study identified that in gait analysis of animals submitted to peripheral nerve injury, the angular parameters of the joints during gait performed better to specify the different groups.
Over the years, different methods have been developed to analyze locomotor function in experimental animal models, thus, many tests have been developed for the research of central and peripheral nerve injuries with the use of small animal models to determining meaningful outcome variables [25-27].
Considering the methods of functional evaluation, we can select some tests that determine the degree of recovery after injury to the nervous system, e.g., sciatic functional index, toe-out-angle (TOA) [28,29] to evaluate changes in the locomotion pattern of the rats over time [11]. However, these analyzes may be influenced by changes in neural response and axonal regeneration, translating into biomechanical changes and compensations due to interactions and complex changes in the weeks following the injury [17].
Following any analysis strategy, functional outcome is the important evidence of function recovery [11,28,30], so quantifying locomotion with computerized gait analysis devices is a great way to evaluate dynamic and static walking patterns in small animals for quantifying and qualify the changes related to the recovery of function.
In this context, based on the statistical analysis the data presented in this study demonstrate that the animals in the control group had significantly better results than the experimental groups. It is interesting to note that even the sham group presented this difference, showing that even a skin lesion would already be enough to alter gait. The types of nervous lesions CCI-1L and CCI-4L did not vary among themselves, but gait parameters were worse than the control and sham groups.
In the longitudinal evaluation, we did not observe changes in the speed variable, but we registered an increase in the values of the following parameters: total course time, step length, stride length, stride speed, IC and reduction in PS in experimental groups. Therefore, the maintenance of speed during gait may have occurred due to changes in neural recruitment and hind limb mechanics [31]. The changes in mentioned above parameters are related to the reorganization in the muscular forces generation to support the body weight and forces in different rhythms to locomotion [32].
Generally, the rat cannot discharge all the weight on the injured limb, causing an eversion of the affected foot, shorter support time and consequent greater weight discharge on the healthy limb [33]. Eftaxiopoulou et al. [34], verified this asymmetry in weight loss and did not observe a correlation between asymmetry and gait speed. This corroborates the idea that the animals perform compensations of the movement to supply the deficit caused by the neuronal injury to try to maintain the gait pattern near the initial (speed and time) [35,36].
The second aspect regarding compensations was observed through the PS variable in the comparison between groups sham, CCI-1L, and CCI-4L. These changes correlate with the long-term effects of sciatic nerve constriction, favoring the chronic contractions development that generate the joint function restriction during locomotion, and significantly altering the kinematics of the injured posterior limb joint [37,38].
Angular changes identified through the PS variable may be linked with changes in the IC and step length parameters and corroborate our results, so we can understand that due to the sensory perception deficit and limb support in the contact moment between paw and soil occurs the loss of the reference signal generated by the necessary force to support the animal body weight [32].
Considering compensation mechanisms when comparing the groups submitted to injury to the control group observed changes in stride length variable, this change may cause increased affected hind limb circumduction and changing the stride length for the gait maintenance [38]. The literature points to several compensatory factors that may prevent gait impairment. After the nerve injury, there is a decrease in the range of motion of the knee but is compensated by an increase in the range of motion of the hip; as well as a contralateral compensation on the healthy limb: increase in hip amplitude and ankle angle in the PS phase [34].
Varejao et al. [39] observed that in the healthy rat, most of the angular movements occur in the sagittal plane and after the nerve injury is accompanied by an external rotation, also using the transverse plane. After the fourth week of injury, he observed improvement in movements in the sagittal plane with an increase in TOA. This compensated external rotation causes greater control of the ankle joint since the plantar flexors are affected [11,39].
Observing the results of the statistical analysis associated to the machine learning analysis, it is interesting to note that the parameters linear presented relatively low results compared to angles.
Generally, the use of the PS angle parameter, IC angle and the difference between IC-PS presented high values of specificity and sensitivity for the detection of gait change. Among the linear parameters step length and stride length presented the highest rankings in the comparison between groups.
The changes in the values of PS and IC-PS may be related to compensations in the sagittal axis of the animals’ body, as Varejao et al. [39] and Eftaxiopoulou et al. [34], have demonstrated in their studies. In addition, we must also consider, according to Lakes and Allen [40], that step length and stride are highly correlated with maintaining walking speed, corroborating the findings of our study.
In this study, the neural networks correlated the longitudinal follow-up after nerve damage (7 and 28 days) and the injury type influence, our results infer that the presented benefits were: reduction of the data dimensionality with minimum loss of information content. The classification and grouping of data according to their similarities resulted in the new indicators selection to quick and objective interpretation of the great amount of information that can be obtained in the gait analysis.
Considering the notes of Varejao et al. [39], Eftaxiopoulou et al. [34], the results of this research new studies may be proposed to associate the data from this research with movements related to the hip, these data would be useful to strengthen and describe the gait profile, as well as compare and differentiate this model of nerve injury from other movement disorders.
Thus, from the results of this research, new, deeper studies, based on the specific parameters (linear and angular) established in this research, make a comparison between the different moments of the functional recovery. Based on the premise of functional rehabilitation, establishing the reassessment periods are fundamental to evaluate a greater or lesser influence of the injury on gait parameters. Essentially, we can see that PS, IC, step length and stride length were among the best parameters in the 7- and 28-day evaluation (Tables 2, 3). However, in a comparison on 7-day evaluation (sham vs. CCI-4L) more parameters were necessary for a better differentiation. It was also possible to observe that it was easier to differentiate the groups 28 days after injury (higher sensitivity and specificity) than 7 days after injury (Tables 4, 5). Perhaps the easiness to classify old lesions has occurred due to the more pronounced movements pattern.
This article has limitations that need to be mentioned. First, pain may be a critical factor to influence the gait. We do not control or correlated pain related factors with gait parameters because was not the scope study. Also, limping is observed in almost all types of nerve injury and and we could have analyzed this parameter. It is interesting that future studies analyzes this relationships.
This paper can favor studies related to the understanding of the primary effects of nervous system lesions, to the effects of therapeutic resources to promote the regeneration of the nerves and help directed studies about gait specifically, but not limited to, a sciatic lesion rat model.
CONCLUSION
We concluded that the angle of the lower limb during the PS angle was considered the best gait parameter to identify alterations due to a tibial nerve injury. IC, step length, and stride length are good parameters to complement the gait analysis. Parameters such as speed and total course time were not efficient to differentiate the presence of nerve injury or to differentiate different lesion degrees.
Acknowledgments
This research was supported by São Paulo Research Foundation (FAPESP) (Research Grant 2013/12916-0 and Multi-user Equipment Grant 2014/11869-0). Renato Leonardo de Freitas was supported by FAPESP (Scientific Initiation Scholarship 2001/03752-6, M.Sc. fellowship 2003/05256-1, post-doctoral fellowship 2009/17258-5, and researcher fellowship 2014/07902-2). FAPESP supported Priscila de Medeiros (Sc.D. fellowship process 2012/25167-2). Matias Jr, I.; was supported by the São Paulo Research Foundation - FAPESP (Master in Science scholarship, process 2013/10543-1) and Matias Jr, I. (process 1535785); Menezes-Reis, R. and Vicente-César, H., were supported by Coordination for the Improvement of Higher Education Personnel – CAPES (Scientiae Doctor Fellowship).
Footnotes
The authors have nothing to disclose.
REFERENCES
- 1.Canazza A, Minati L, Boffano C, et al. Experimental models of brain ischemia: a review of techniques, magnetic resonance imaging, and investigational cell-based therapies. Front Neurol. 2014;5:19. doi: 10.3389/fneur.2014.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jaggi AS, Jain V, Singh N. Animal models of neuropathic pain. Fundam Clin Pharmacol. 2011;25:1–28. doi: 10.1111/j.1472-8206.2009.00801.x. [DOI] [PubMed] [Google Scholar]
- 3.Kjell J, Olson L. Rat models of spinal cord injury: from pathology to potential therapies. Dis Model Mech. 2016;9:1125–37. doi: 10.1242/dmm.025833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Matias I, Elias-Filho DH, Garcia CAB, et al. A new model of experimental hemispherotomy in young adult Rattus norvegicus: a neural tract tracing and SPECT in vivo study. J Neurosurg. 2018 Jun 1;:1–14. doi: 10.3171/2017.12.JNS171150. [Epub]. [DOI] [PubMed] [Google Scholar]
- 5.Cenci MA, Whishaw IQ, Schallert T. Animal models of neurological deficits: how relevant is the rat? Nat Rev Neurosci. 2002;3:574–9. doi: 10.1038/nrn877. [DOI] [PubMed] [Google Scholar]
- 6.Caeyenberghs K, Clemente A, Imms P, et al. Evidence for training-dependent structural neuroplasticity in brain-injured patients: a critical review. Neurorehabil Neural Repair. 2018;32:99–114. doi: 10.1177/1545968317753076. [DOI] [PubMed] [Google Scholar]
- 7.Pandit V, Khan M, Zakaria ER, et al. Continuous remote ischemic conditioning attenuates cognitive and motor deficits from moderate traumatic brain injury. J Trauma Acute Care Surg. 2018;85:48–53. doi: 10.1097/TA.0000000000001835. [DOI] [PubMed] [Google Scholar]
- 8.Castillo-Galván ML, Martínez-Ruiz FM, de la Garza-Castro O, et al. Study of peripheral nerve injury in trauma patients. Gac Med Mex. 2014;150:527–32. [PubMed] [Google Scholar]
- 9.McAllister TW. Neurobiological consequences of traumatic brain injury. Dialogues Clin Neurosci. 2011;13:287–300. doi: 10.31887/DCNS.2011.13.2/tmcallister. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Huebner EA, Strittmatter SM. Axon regeneration in the peripheral and central nervous systems. Results Probl Cell Differ. 2009;48:339–51. doi: 10.1007/400_2009_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Varejão AS, Melo-Pinto P, Meek MF, et al. Methods for the experimental functional assessment of rat sciatic nerve regeneration. Neurol Res. 2004;26:186–94. doi: 10.1179/016164104225013833. [DOI] [PubMed] [Google Scholar]
- 12.Jones TA. Motor compensation and its effects on neural reorganization after stroke. Nat Rev Neurosci. 2017;18:267–80. doi: 10.1038/nrn.2017.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marino R, Jr, Machado AG, Timo-Iaria C. Functional recovery after combined cerebral and cerebellar hemispherectomy in the rat. Stereotact Funct Neurosurg. 2001;76:83–93. doi: 10.1159/000056497. [DOI] [PubMed] [Google Scholar]
- 14.Pagnussat AS, Michaelsen SM, Achaval M, et al. Effect of skilled and unskilled training on nerve regeneration and functional recovery. Braz J Med Biol Res. 2012;45:753–62. doi: 10.1590/S0100-879X2012007500084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Couto PA, Filipe VM, Magalhães LG, et al. A comparison of two-dimensional and three-dimensional techniques for the determination of hindlimb kinematics during treadmill locomotion in rats following spinal cord injury. J Neurosci Methods. 2008;173:193–200. doi: 10.1016/j.jneumeth.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 16.Clayton HM. Instrumentation and techniques in locomotion and lameness. Vet Clin North Am Equine Pract. 1996;12:337–50. doi: 10.1016/s0749-0739(17)30285-7. [DOI] [PubMed] [Google Scholar]
- 17.Takakusaki K. Functional neuroanatomy for posture and gait control. J Mov Disord. 2017;10:1–17. doi: 10.14802/jmd.16062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rangayyan RM. Biomedical image analysis. In: LLC CP, editor. The nature of biomedical image. Boca Raton: CRC Press; 2005. p. 1311. [Google Scholar]
- 19.Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 1988;33:87–107. doi: 10.1016/0304-3959(88)90209-6. [DOI] [PubMed] [Google Scholar]
- 20.Frank E. Fully supervised training of Gaussian radial basis function networks in WEKA. Hamilton (NZ): Department of Computer Science, The University of Waikato; 2014. [Google Scholar]
- 21.Hornberger TA, Jr, Farrar RP. Physiological hypertrophy of the FHL muscle following 8 weeks of progressive resistance exercise in the rat. Can J Appl Physiol. 2004;29:16–31. doi: 10.1139/h04-002. [DOI] [PubMed] [Google Scholar]
- 22.Kononenko I. Estimating attributes: analysis and extensions of RELIEF. In: Bergadano F, De Raedt L, editors. Machine learning: ECML-94. ECML 1994. Lecture Notes in Artificial Intelligence; European Conference on Machine Learning; 1994 Apr 6-8; Catania, Italy. Berlin: Springer; 1994. [Google Scholar]
- 23.Witten IH, Frank E, Hall MA. Data mining: practical machine learning tools and techniques. 3rd ed. Amsterdam: Elsevier; 2011. [Google Scholar]
- 24.John GH, Langley P. Estimating continuous distributions in Bayesian classifiers. Proceedings of the Eleventh conference on Uncertainty in artificial intelligence; 1995 Aug 18-20; Montréal, Québec, Canada. San Francisco (CA): Morgan Kaufmann Publishers Inc.; 1995. pp. 338–45. [Google Scholar]
- 25.Nakamura A, Funaya H, Uezono N, et al. Low-cost three-dimensional gait analysis system for mice with an infrared depth sensor. Neurosci Res. 2015;100:55–62. doi: 10.1016/j.neures.2015.06.006. [DOI] [PubMed] [Google Scholar]
- 26.Rui J, Runge MB, Spinner RJ, et al. Gait cycle analysis: parameters sensitive for functional evaluation of peripheral nerve recovery in rat hind limbs. Ann Plast Surg. 2014;73:405–11. doi: 10.1097/SAP.0000000000000008. [DOI] [PubMed] [Google Scholar]
- 27.Vandeputte C, Taymans JM, Casteels C, et al. Automated quantitative gait analysis in animal models of movement disorders. BMC Neurosci. 2010;11:92. doi: 10.1186/1471-2202-11-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kappos EA, Sieber PK, Engels PE, et al. Validity and reliability of the CatWalk system as a static and dynamic gait analysis tool for the assessment of functional nerve recovery in small animal models. Brain Behav. 2017;7:e00723. doi: 10.1002/brb3.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sabatier MJ, To BN, Nicolini J, et al. Effect of slope and sciatic nerve injury on ankle muscle recruitment and hindlimb kinematics during walking in the rat. J Exp Biol. 2011;214(Pt 6):1007–16. doi: 10.1242/jeb.051508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hsieh TH, Chen JJ, Chen LH, et al. Time-course gait analysis of hemiparkinsonian rats following 6-hydroxydopamine lesion. Behav Brain Res. 2011;222:1–9. doi: 10.1016/j.bbr.2011.03.031. [DOI] [PubMed] [Google Scholar]
- 31.Heitz RP. The speed-accuracy tradeoff: history, physiology, methodology, and behavior. Front Neurosci. 2014;8:150. doi: 10.3389/fnins.2014.00150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Giszter SF, Davies MR, Graziani V. Coordination strategies for limb forces during weight-bearing locomotion in normal rats, and in rats spinalized as neonates. Exp Brain Res. 2008;190:53–69. doi: 10.1007/s00221-008-1451-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bozkurt A, Deumens R, Scheffel J, et al. CatWalk gait analysis in assessment of functional recovery after sciatic nerve injury. J Neurosci Methods. 2008;173:91–8. doi: 10.1016/j.jneumeth.2008.05.020. [DOI] [PubMed] [Google Scholar]
- 34.Eftaxiopoulou T, Macdonald W, Britzman D, et al. Gait compensations in rats after a temporary nerve palsy quantified using temporo-spatial and kinematic parameters. J Neurosci Methods. 2014;232:16–23. doi: 10.1016/j.jneumeth.2014.04.011. [DOI] [PubMed] [Google Scholar]
- 35.Cendelín J, Voller J, Vozeh F. Ataxic gait analysis in a mouse model of the olivocerebellar degeneration. Behav Brain Res. 2010;210:8–15. doi: 10.1016/j.bbr.2010.01.035. [DOI] [PubMed] [Google Scholar]
- 36.Vrinten DH, Hamers FF. ‘CatWalk’ automated quantitative gait analysis as a novel method to assess mechanical allodynia in the rat; a comparison with von Frey testing. Pain. 2003;102:203–9. doi: 10.1016/s0304-3959(02)00382-2. [DOI] [PubMed] [Google Scholar]
- 37.Dellon AL, Mackinnon SE. Sciatic nerve regeneration in the rat. Validity of walking track assessment in the presence of chronic contractures. Microsurgery. 1989;10:220–5. doi: 10.1002/micr.1920100316. [DOI] [PubMed] [Google Scholar]
- 38.Santos PM, Williams SL, Thomas SS. Neuromuscular evaluation using rat gait analysis. J Neurosci Methods. 1995;61:79–84. doi: 10.1016/0165-0270(95)00026-q. [DOI] [PubMed] [Google Scholar]
- 39.Varejão AS, Cabrita AM, Meek MF, et al. Ankle kinematics to evaluate functional recovery in crushed rat sciatic nerve. Muscle Nerve. 2003;27:706–14. doi: 10.1002/mus.10374. [DOI] [PubMed] [Google Scholar]
- 40.Lakes EH, Allen KD. Gait analysis methods for rodent models of arthritic disorders: reviews and recommendations. Osteoarthritis Cartilage. 2016;24:1837–49. doi: 10.1016/j.joca.2016.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]


