Abstract
Speech Motor Delay (SMD) is a recently proposed childhood motor speech disorder characterized by imprecise and unstable speech, prosody, and voice that does not meet criteria for either Childhood Dysarthria or Childhood Apraxia of Speech. The goals of the present research were to obtain information on the phenotype of SMD and initial information on the persistence of SMD in children receiving treatment for idiopathic Speech Delay (SD). Five questions about the phenotype and persistence of SMD were posed using a database of audio-recordings and participant records and longitudinal data from audio-recordings of children with early SMD treated for SD. Three phenotype questions examined associations between participant risk factors and prevalence of SMD, and described the most frequent speech, prosody, and voice signs of early SMD. To provide initial estimates of the persistence of SMD, two questions examined associations between the persistence of SMD and participant risk factors using the audio-recordings of 14 participants with SMD treated for idiopathic SD. Phenotype findings indicated that SMD is characterized by across-the-board delays in the spatiotemporal precision and stability of speech, prosody, and voice production. Persistence findings indicated that although most participants normalized early SMD by 6 years of age, SMD persisted until at least late adolescence in 21.4% of participants. Findings are interpreted to support the construct validity of SMD and the potential for research using additional assessment modalities to explicate its genomic and neuromotor causal pathways.
Keywords: Apraxia, dysarthria, speech delay, speech motor delay, speech sound disorders
Motor speech disorders in children
Recent research in motor speech disorders in children suggests that there is need for three motor speech classifications (Shriberg, Kwiatkowski, & Mabie, 2019).
Childhood dysarthria, and childhood apraxia of speech
The most common context for childhood motor speech disorders is Childhood Dysarthria (CD) as it occurs in children with congenital neurodevelopmental disorders such as cerebral palsy, or in children who acquire a disorder such as traumatic brain injury during the developmental period (Duffy, 2013; Morgan & Liégeois, 2010). Childhood Apraxia of Speech (CAS), in contrast, has most often been studied as an idiopathic disorder, with more recent interdisciplinary interest in the genomic antecedents of CAS in both idiopathic contexts and in the context of complex neurodevelopmental disorders (American Speech-Language-Hearing Association, 2007; Royal College of Speech and Language Therapists, 2011).
Speech motor delay
In addition to the motor speech literatures in CD and CAS, the literature in idiopathic Speech Delay (SD) includes proposals, findings, and discussions suggesting a ‘motor component’ in some children with idiopathic SD (Shriberg, Kwiatkowski, & Mabie, 2019). The general hypothesis is that just as there are clinical entities for idiopathic delays in other developmental domains (e.g., cognitive delay, language delay, speech delay) there is need in Speech Sound Disorders (SSD) for a classification entity for children with speech motor delay.
A research program in SSD included the goals of developing valid and reliable measures to identify CAS, CD, and to identify a motor speech disorder that does not meet criteria for either CAS or CD (Shriberg, 2010b). The research initially used the provisional term Motor Speech Disorder-Not Otherwise Specified as a placeholder classification for this latter putative disorder (Shriberg, 2010a, 2010b; Shriberg et al., 2010a; Shriberg, Potter, & Strand, 2011). Findings from subsequent research with children with idiopathic SD (Shriberg, 2017) and children with SD in the context of complex neurodevelopmental disorders (Baylis & Shriberg, 2018) supported replacing the provisional term with the present term, Speech Motor Delay (SMD). Specifically, perceptual and acoustic analyses reported in Shriberg (2017) indicated that the early speech-prosody-voice phenotypes of children meeting criteria for the placeholder classification Motor Speech Disorder-Not Otherwise Specified is consistent with a delay in neuromotor precision-stability (i.e., the lower tail of speech motor development).
A Supplement for this research series provides detailed information on classification methods [Supplementary Data]. The Supplement includes a copy of the Precision-Stability Index (PSI), the 32-sign measure developed to identify SMD. Psychometric and reference data for the PSI are provided in two technical reports (Mabie & Shriberg, 2017; Shriberg & Mabie, 2017).
Statement of purpose
The goals of the present paper are to describe initial findings for the behavioural phenotype of SMD and its persistence. Using information in a database described in Shriberg, Kwiatkowski, & Mabie (2019), three questions were posed about the phenotype of SMD in children with idiopathic SD. Two additional questions focusing on the persistence of SMD were posed using information from retrospective longitudinal audio-recordings of 14 children with early SMD in treatment for SD. The rationales for each question address initial phenotype and persistence questions that can be posed using the present retrospective data. Following are the five questions, each with implications for theory and practice in children with SSD:
Were estimates of the prevalence of SMD in children with idiopathic SD associated with any participant variables?
Which were the most frequent early signs of SMD in participants with idiopathic SD and SMD?
Did participants with SMD differ significantly from participants with No Motor Speech Disorder (No MSD) on any participant variables?
Did early SMD persist past 6 or 9 years of age in any participant treated for idiopathic SD?
Were any demographic or early speech variables significantly associated with the persistence of SMD in participants treated for SD?
Methods
Participants for the phenotype questions
Table 1 is a summary of demographic, cognitive-language, and speech competence findings for four classifications of Motor Speech Disorder (MSD) described in Shriberg, Kwiatkowski, & Mabie (2019). The Appendix (adapted from Table 2 in Shriberg, Kwiatkowski, & Mabie 2019) and the Supplement for the present series of articles [Supplementary Data] provide detailed information on the measures and procedures used for the four MSD classifications. An overview of the measure used to identify SMD is provided in a subsequent section.
Table 1.
Motor speech classifications and demographic, cognitive-language, and speech information for 415 participants with Speech Delay.
| Motor Speech Classification | Demographic |
Cognitive-Language |
Speech |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (yrs) |
Sex (%) |
Cognitiona |
Languageb |
PCCc |
IId |
|||||||||
| n | M | SD | Range | Female | Male | M | SD | M | SD | M | SD | M | SD | |
| No MSD | 341 | 4.8 | 1.7 | 3–16 | 30.0 | 70.0 | 100.6 | 14.2 | 96.1 | 15.6 | 74.5 | 12.2 | 92.1 | 7.8 |
| SMD | 50 | 5.1 | 1.7 | 3–10 | 20.0 | 80.0 | 100.6 | 12.1 | 97.2 | 13.6 | 71.8 | 14.3 | 88.8 | 10.2 |
| CD | 14 | 4.7 | 1.6 | 3–8 | 21.4 | 78.6 | 109.8 | 11.1 | 105.0 | 17.0 | 70.0 | 14.8 | 83.4 | 14.6 |
| CAS | 10 | 5.0 | 1.7 | 3–9 | 20.0 | 80.0 | 63 | * e | 77 | * e | 76.3 | 9.6 | 84.4 | 15.1 |
| Total/Average | 415 | 4.9 | 1.7 | 3–16 | 28.4 | 71.6 | 100.8 | 14.1 | 96.6 | 15.4 | 74.1 | 12.6 | 91.2 | 8.8 |
| Group-Averaged | 4.9 | 1.7 | 22.9 | 77.2 | 93.5 | 12.5 | 93.8 | 15.4 | 73.2 | 12.7 | 87.2 | 11.9 | ||
Notes: PCC = Percentage of Consonants Correct; II = Intelligibility Index; No MSD = No Motor Speech Disorder; SMD = Speech Motor Delay; CD = Childhood Dysarthria; CAS = Childhood Apraxia of Speech.
Scores for Cognition: IQ Composite (n = 25) from Kaufman Brief Intelligence Test – Second Edition; Kaufman and Kaufman (2004).
Total Score Standard Score (General Ability; n = 64) from Detroit Tests of Learning Aptitude-Primary: Third Edition; Hammill and Bryant (2005).
Full IQ (n = 16) from Weschler Abbreviated Scale of Intelligence; Wechsler (1999).
Scores for Language: Oral Composite Standard Score (n = 27) from Oral and Written Language Scales (OWLS); Carrow-Woolfolk (1995).
Oral Composite Standard Score (n = 66) from Oral and Written Language Scales, Second Edition (OWLS-II); Carrow-Woolfolk (2011).
Core Language Standard Scores (n = 23) from Clinical Evaluation of Language Fundamentals-4 (CELF–4); Semel, Wiig, and Secord (2003).
Standardized cognition and language measures available for only one participant.
Table 2.
Demographic, language, and speech information for 14 participants in treatment for idiopathic Speech Delay.a.
| Sex |
Speech |
|||||
|---|---|---|---|---|---|---|
| Participant | Age (yrs; mos) | Female | Male | Languageb | PCCc | IId |
| 1 | 3;1 | X | PPVT-R SS: 99 | 53.4 | 82.9 | |
| PLS: Auditory Comprehension Quotient: 100; Verbal Ability | ||||||
| Quotient: 87 | ||||||
| 2 | 3;6 | X | OWLS: Oral Composite SS: 99 | 61.7 | 81.8 | |
| 3 | 3;6 | X | PPVT-R SS: 66 | 66.5 | 76.6 | |
| TOLD-P: Subtest I SS: 63; Subtest II: 16; Subtest III: 9; Subtest IV: 9; | ||||||
| Subtest V: 9 | ||||||
| 4 | 3;10 | X | * | 68.2 | 70.4 | |
| 5 | 4;1 | X | PPVT-R SS: 95 | 63.1 | 80.6 | |
| 6 | 4;2 | X | * | 80.1 | 95.2 | |
| 7 | 4;4 | X | OWLS: Oral Composite SS: 101 | 70.9 | 96.2 | |
| 8 | 4;5 | X | PLS-3: Receptive: 76; Expressive: 67 | 42.1 | 75.2 | |
| 9 | 4;7 | X | * | 80.1 | 95.4 | |
| 10 | 5;0 | X | * | 71.3 | 96.2 | |
| 11 | 5;1 | X | * | 73.8 | 73.2 | |
| 12 | 5;4 | X | PPVT-R Percentile: 99th | 66.7 | 97.2 | |
| 13 | 5;7 | X | PPVT-R: Age equivalent 4;9 | 67.4 | 100.0 | |
| The Miller-Yoder Test of Grammatical Comprehension: ‘passed all 5 | ||||||
| and 6 year level items’ | ||||||
| The Token Test for Children: scored within age level on all 5 | ||||||
| subtests | ||||||
| 14 | 5;9 | X | * | 82.9 | 91.8 | |
| Mean | 4;5 | 67.7 | 86.6 | |||
| SD | 10 mos | 10.8 | 10.4 | |||
| % | 14.3 | 85.7 | ||||
Notes: PPVT-R = Peabody Picture Vocabulary Test-Revised; SS = Standard Score; PLS = Preschool Language Scale; OWLS = Oral and Written Language Scales; TOLD-P = Test of Language Development-Primary; PLS-3 = Preschool Language Scale-3; II = Intelligibility Index and PCC = Percentage of Consonants Correct.
Participants are ordered by age at first assessment: language and speech data were obtained at the same or approximate age. All speech data were obtained from retrospective longitudinal audio-recordings.
Peabody Picture Vocabulary Test-Revised (Dunn & Dunn, 1981); Preschool Language Scale (Zimmerman, 1969); Oral and Written Language Scales (Carrow-Woolfolk, 1995); TOLD-P = Test of Language Development-Primary (Newcomer & Hammill, 1988); Preschool Language Scale-3 (Zimmerman, Steiner, & Pond, 1992); The Miller-Yoder Test of Grammatical Comprehension (Miller & Yoder, 1975); The Token Test for Children (DiSimoni, 1978).
= no data.
The 415 participants in Table 1 had been recruited for studies of idiopathic SD in local and collaborative studies conducted over several decades in six cities in the U.S. All participants had been assented and/or consented to participate in the research using procedures and forms approved by internal review boards at the University of Wisconsin-Madison and five other collaborative research sites. As shown in Table 1, 341 (82.2%) participants in the six data sets met criteria for No MSD, 50 (12%) met criteria for SMD, 14 (3.4%) met criteria for CD, and 10 (2.4%) of the participants met criteria for CAS (Shriberg, Kwiatkowski, & Mabie, 2019). There were no participants in the present database of children with idiopathic SD who met criteria for concurrent CD & CAS. The focus of the present paper is on the 50 participants who met criteria for SMD. Findings from the 341 participants with No MSD provided control data for the descriptive and inferential statistical analyses.
Participants for the persistence questions
Table 2 is a summary of demographic, language, and speech characteristics of 14 children in treatment for idiopathic SD who were retrospectively identified as having early SMD. Participants were selected from collaborative research at two university research sites at which databases of audio-recorded conversational samples were available. All participants had been assented and/or consented to participate in one or more studies of idiopathic SD using procedures and forms approved by internal review boards at the University of Wisconsin-Madison and Case Western Reserve University, Cleveland, Ohio. Conversational samples were obtained weekly to multi-yearly to monitor children’s normalization of speech sound deletions and substitutions. Examiners at each site had used conversational speech sampling procedures designed to be naturalistic and maximally productive for children with low verbal output (e.g., McSweeny, 1998; Shriberg, Kwiatkowski, & Mabie (2019).
The 14 participants in Table 2 were selected from a total of 104 participants for whom retrospective longitudinal data were available. There were two criteria for selecting participants and speech samples for the present study: 1) the participant met the Speech Disorders Classification System criterion for SMD on the PSI (i.e., < 70%) on at least one occasion when a conversational speech sample was available before the participant was 6 years of age, and 2) the participant either met criteria for No MSD on at least one later recording date or continued to meet the criterion for SMD on at least one later recording date. Perceptual and acoustic data reduction of all recordings from the 14 participants were completed for the present analyses by the research personnel who completed data reduction in Shriberg, Kwiatkowski, & Mabie (2019).
As shown in Table 2, the 14 participants were from 3 years, 1 month of age to 5 years, 9 months of age at the earliest available conversational speech sample. The summary demographic statistics for the 14 children are generally similar to those for the 50 participants with SMD in Table 1, with mean values on the continuous variables within a standard deviation of one another. Table 2 includes the several full-scale and subscale language scores available for each participant and the two speech competence scores available from all children at their earliest recorded sample. These scores were also consistent with the range of language and speech competence scores of participants in the phenotype database (Table 1).
Motor speech classification
The following is a brief description of the PSI. Readers may wish to review classification information for this measure in the Appendix and in the Supplement. The Supplement includes a copy of the PSI that provides computational information on each of the 32 PSI signs of SMD.
The 13 perceptual and 19 acoustic items that comprise the PSI assess the spatiotemporal precision and stability of speech (18 signs), prosody (5 signs), and voice (9 signs). The signs of imprecision and instability were selected from research and clinical discussions of typical and atypical motor speech development, together with anecdotal observations of the speech, prosody, and voice profiles of children suspected to have a ‘motor’ component to their idiopathic SD. An age-sex standardized value (z-score) for each sign is obtained from the conversational speech samples from each participant. Sources for procedural information on narrow phonetic transcription, prosody-voice coding, and acoustic data reduction for the PSI are included in Shriberg et al. (2010a) and Mabie and Shriberg (2017). A database of 200 typical speakers, 3 to 80 years of age, was used to standardize PSI scores (Potter et al., 2012; Scheer-Cohen et al., 2013). A z-score less than −1.25 standard deviation units on each sign indicates performance below the typical age-sex adjusted mean for the sign. The PSI percentage score is calculated by dividing the number of standardized signs lower than −1.25 by 32 (or less if missing data) and subtracting the quotient from 100, so that lower percentage scores reflect reduced precision and stability. Speakers with PSI scores lower than 70% are classified as PSI+ (i.e., SMD). Speakers who meet the Speech Disorders Classification System criteria for both SMD and any of the other three MSD classifications – CD, CAS, or CD & CAS – are classified as positive on the relevant latter, putatively more severe MSD classifications.
Data reduction and reliability estimates
Four transcriptionists and/or acoustic analysts completed narrow phonetic transcription, prosody-voice coding, and acoustic analyses of the continuous speech samples. A research assistant error-checked the transcripts and entered them into software titled Programs to Examine Phonetic and Phonologic Evaluation Records (PEPPER, 2019). Table 3 in Shriberg, Kwiatkowski, & Mabie (2019) provides a summary of the point-to-point interjudge and intrajudge percentage of agreement findings for 60 conversational samples from the 415 participants randomly selected to be proportionate to the prevalence obtained for each MSD. As reported, the mean agreement across 26 estimates was 87.9% (SD = 5.9%), which is consistent with reliability estimates for comparable perceptual and acoustic variables in SSD research (Shriberg, Kwiatkowski, & Mabie, 2019).
Table 3.
Description of participant samples with high (Sample 1) and low (Sample 2) prevalence of Speech Motor Delay (SMD).a.
| Sample 1: High Prevalence of SMD (22.4%) |
Sample 2: Low Prevalence of SMD: (2.8%) |
Between-group Comparisons |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | n | M | SD | % | n | M | SD | % | Confidence Interval | Effect Sizeb | p-valuec |
| Demographic | |||||||||||
| Age (yrs) | 67 | 5.1 | 1.5 | 72 | 4.0 | 0.7 | 0.59, 1.30 | 0.95 L | |||
| Gender (male) | 77.6 | 73.6 | 0.694 | ||||||||
| Parent Report-Risk Factors | |||||||||||
| Speech, Language, & Learning | |||||||||||
| Articulation/Phonology | 60 | 92.3 | 44 | 68.8 | 0.001 | ||||||
| Apraxia | 6 | 9.2 | 3 | 4.8 | 0.492 | ||||||
| Dysarthria | 3 | 4.6 | 0 | 0.0 | 0.244 | ||||||
| Receptive language disorder | 7 | 10.8 | 2 | 2.9 | 0.090 | ||||||
| Expressive language disorder | 17 | 26.2 | 3 | 4.8 | 0.001 | ||||||
| Structural disorder | 5 | 7.7 | 2 | 3.1 | 0.440 | ||||||
| Fluency disorder | 6 | 9.2 | 1 | 1.4 | 0.057 | ||||||
| Voice Disorder | 3 | 4.6 | 1 | 1.5 | 0.358 | ||||||
| Other Disorder | 4 | 6.2 | 0 | 0.0 | 0.058 | ||||||
| Learning disability | 3 | 4.5 | 3 | 4.2 | 1.000 | ||||||
| Hearing | |||||||||||
| History of hearing problems | 5 | 7.7 | 4 | 5.7 | 0.738 | ||||||
| OME 16+ in first 18 months | 2 | 3.4 | 4 | 6.8 | 0.679 | ||||||
| PE tubes placed | 11 | 16.4 | 9 | 12.5 | 0.630 | ||||||
| Genetics | |||||||||||
| Nuclear family member with | 32 | 49.2 | 31 | 46.3 | 0.862 | ||||||
| verbal trait disorder | |||||||||||
| Cognitive-Linguistic | |||||||||||
| Syllable Repetition Taskd | |||||||||||
| Performance | 66 | −1.16 | 1.42 | 71 | −0.58 | 1.02 | −0.81, −0.13 | −0.47 S | |||
| Encoding | 66 | −0.44 | 1.09 | 69 | −0.14 | 1.03 | −0.62, 0.06 | −0.28 | |||
| Memory | 67 | −1.01 | 1.78 | 72 | 0.13 | 0.81 | −1.18, −0.48 | −0.83 L | |||
| Language | |||||||||||
| Average Words/Utterance-Z | 67 | −1.38 | 1.14 | 72 | −1.94 | 1.03 | 0.18, 0.85 | 0.51 M | |||
| Speech | |||||||||||
| PCC | 67 | 80.6 | 11.2 | 72 | 70.0 | 9.6 | 0.66, 1.37 | 1.01 V | |||
| PCC-Z | 67 | −1.86 | 1.58 | 72 | −2.44 | 1.19 | 0.08, 0.75 | 0.41 S | |||
| PVC | 67 | 93.3 | 5.3 | 72 | 91.7 | 5.3 | −0.03, 0.63 | 0.30 | |||
| PVC-Z | 67 | −2.61 | 2.01 | 72 | −3.21 | 1.71 | −0.01, 0.66 | 0.32 | |||
| II | 67 | 94.4 | 7.2 | 72 | 88.5 | 8.9 | 0.38, 1.07 | 0.72 M | |||
| II-Z | 67 | −1.79 | 2.10 | 72 | −3.54 | 1.94 | 0.51, 1.21 | 0.86 L | |||
| OIIe: % Lowered Intelligibility | 10.4 | 23.6 | 0.046 | ||||||||
Notes: OME = Otitis Media with Effusion; PE = Pressure Equalization; PCC-Z = Percentage of Consonants Correct-Z-score; PVC = Percentage of Vowels Correct; PVC-Z = Percentage of Vowels Correct-Z-score; II-Z = Intelligibility Index-Z-score; OII = Ordinal Intelligibility Index.
See text for description of treatment histories for each group. Statistically significant findings are bolded.
Effect Size: Small (S) >0.2; Medium (M) >0.5; Large (L) >0.8; Very Large (V) >1.0.
Fisher’s exact tests.
Mabie and Shriberg (2017). Lowered intelligibility: <85%.
Results and discussion
It is efficient to organize this section by the five questions posed in the Statement of Purpose, with each of the following sections including rationale for the question, findings, and discussion. Analyses and discussion are limited by several design considerations, notably those associated with retrospective data and cell sizes.
Question 1: were estimates of the prevalence of SMD in children with idiopathic SD associated with any participant variables?
Rationale
The prevalence of SMD in the six samples of children with idiopathic SD (Shriberg, Kwiatkowski, & Mabie, 2019) was estimated at 12%, the mean of the samples at six study sites. The range of the prevalence estimates was notably wide, from 2.8% at one site to 22.4% at another. Because the speech sampling procedures and recording instrumentation were similar at each site, and the measures, data reduction procedures, data reduction personnel, and classification software were exactly the same for all six estimates, the differences in the prevalence estimates obtained at the study sites were assumed to be associated with one or more participant variables at the different sites. The first question examines the support for that explanation, which may inform the phenotypic characteristics of SMD.
Findings
Table 3 includes descriptive findings for the study sample with the highest (22.4%) and lowest (2.8%) prevalence of SMD. The two study samples were comparable in size (ns of 67 and 72 participants). Between-group statistical comparisons for each variable in Table 3 were completed using the inferential tests for categorical and continuous variables described in the table. Analyses were treated family-wise to minimize Type II errors due to the many comparisons with relatively few participant data (Feise, 2002; Nakagawa, 2004; Perneger, 1998). Four statistically significant findings in Table 3 are described below, followed by discussion.
The first significant finding in Table 3 is that participants in the study sample with a high prevalence of SMD (22.4%) were approximately one year older than participants in the study sample with a low prevalence of SMD (2.8%). Participants in the high prevalence of SMD sample averaged 5.1 years of age (SD: 1.5 yrs), compared to an average of 4.0 years of age (SD: 0.07 yrs) for participants in the low prevalence sample. Second, parent report questionnaires indicated that significantly more participants in the high than the low SMD prevalence sample were reported to have concurrent speech deficits, and notably, expressive language disorders. Third, significantly more participants in the high than the low prevalence of SMD sample had lower standardized scores on the performance and memory subscales of the Syllable Repetition Task (Shriberg et al., 2009). Last, four findings for the speech and intelligibility measures in Table 3 indicated that significantly more participants in the high than low prevalence of SMD sample had higher average speech and intelligibility scores compared to the average speech and intelligibility scores of participants in the low SMD prevalence sample.
Discussion
The finding that the study sample with the high prevalence of SMD was significantly older than the group with the low prevalence is not consistent with a developmental explanation of SMD. That is, if the prevalence of SMD is associated with age, older participants would be more likely to have a lower prevalence of SMD. A significant difference in the treatment histories of participants in the two study groups prior to their enrollment in each study group (as discussed in the next paragraph) provides a possible explanation of the four significant findings in Table 3.
The inclusionary/exclusionary participation criteria for the six studies was moderate to severe SD, but the treatment histories of participants were not similar. Inspection of participant records indicated that almost all (95%) of the participants in the high prevalence SMD group in Table 3 (SMD prevalence = 22.4%) had received treatment prior to enrollment in the study, whereas none (0%) of the participants in the low prevalence SMD group (SMD prevalence = 2.8%) had received treatment prior to enrollment in the present study. Findings are interpreted as proof of concept for the construct of SMD. As previously reviewed, a motivation for the development of a third type of motor speech disorder in addition to dysarthria and apraxia of speech was the frequent observation in research and clinical SSD literatures that some children are suspected to have a motor component to their idiopathic SD. This observation may sometimes be suspected for children who are not making expected progress in treatment for idiopathic SD. Although the older participants in the present study sample with a high prevalence of SMD had significantly higher speech competence and higher intelligibility (Table 3), their referral by parents and local clinicians to a study on moderate-severe SSD was plausibly consistent with their lowered memory and lowered expressive language scores, and the percept by parents and clinicians that they appeared to continue to have speech production needs. That is, although 95% of the significantly older participants in this study sample had been in treatment for SD, and their average speech and intelligibility scores (adjusted for age and sex) were significantly higher than the average scores of the younger participants in the comparison study sample that had not had treatment, a significantly higher percentage of these participants met criteria for SMD (Fisher’s exact test = 0.000).
Question 2: which were the most frequent early signs of SMD in participants with idiopathic SD and SMD?
Rationale
In a research series on CAS, one of four criteria proposed for a core phenotypic sign of a motor speech classification was that the sign is present at both the earliest and latest stages of expression of the disorder (Shriberg et al., 2017). To address the second phenotype question, the present analyses examined findings for the 32 PSI signs for each of the 50 participants with SMD to determine which PSI signs were most often present at the earliest available assessment. The present database did not include follow-up data to assess whether the same signs also persisted until SMD normalized, and there were too few participants with persistent SMD in the second database (Table 2) to address the question.
Findings
Table 4 includes information for PSI signs on which more than 50% of theparticipants with SMD (i.e., at least 26 of the 50 participants) met the standardized criteria for imprecision or instability. This arbitrary proportional criterion yielded 10 (31.3%) of the 32 PSI signs operationally defined as possible core phenotypic features of SMD. As shown in Table 4, the percentage of the 50 participants with SMD who had z-scores lower than 1.25 on each of the 10 signs ranged from 52% to 66.7% (M: 56.3; SD: 5.6). In comparison, the percentage of the 341 participants with No MSD with z-scores lower than 1.25 on each of the 10 signs ranged from 11.1% to 47.7% (M: 25.8; SD: 11.8). It should be noted that Sign No. 12, the sign on which the highest percentage of participants with No MSD were positive (47.7%), was the acoustic marker for dentalized /s/ initial singletons, one of the most common phoneme distortions in children with any type of SSD.
Table 4.
Ten most frequent earliest available PSI signs for participants with Speech Motor Delay (SMD) and with No Motor Speech Disorder (No MSD).
| System |
Domain |
PSI Sign No. | PSI Sign Description | Data Reduction |
% of Participants Positive on Sign |
Attributes |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Perceptual | Acoustic | SMD | No MSD | Precision | Stability | Spatial | Temporal | ||||
| Speech | Vowels | ||||||||||
| 5 | Increased Duration for Mid-Vowels and Diphthongs | X | 56.0 | 25.0 | X | X | |||||
| 6 | Reduced % Vowel Phoneme Target Consistency | X | 65.0 | 32.4 | X | X | |||||
| 7 | Reduced % Vowel Target Consistency | X | 66.7 | 28.7 | X | X | |||||
| Consonants | |||||||||||
| 8 | Reduced % Correct Glides | X | 52.0 | 31.7 | X | X | |||||
| 12 | Decreased 1st Moment on/s/Initial Singletons | X | 53.3 | 47.7 | X | X | |||||
| Vowels & Consonants | |||||||||||
| 17 | Increased Diacritic Modification Index Class: Duration % | X | 54.0 | 24.3 | X | X | |||||
| 18 | Increased % of Epenthesis Errors | X | 60.0 | 33.1 | X | X | |||||
| Prosody | Rate | ||||||||||
| 21 | Increased Average Syllable Duration (ms) (without pauses) | X | 52.0 | 7.7 | X | X | |||||
| Stress | |||||||||||
| 23 | Increased % of Prosody-Voice Codes15/16 Excessive Equal codes of all Prosody-Voice Codes 15/16 (uncircled & circled) | X | 52.3 | 11.1 | X | X | |||||
| Voice | Laryngeal Quality | ||||||||||
| 29 | Decreased Harmonic-to-Noise Ratio (dB) for Vowels | X | 52.0 | 16.5 | X | __a | __a | ||||
| Totals | 6 | 4 | 7 | 3 | 5 | 4 | |||||
| Mean (SD) | 56.3 (5.6) | 25.8 (11.8) | |||||||||
Notes: PSI = Precision-Stability Index; ms = millisecond; dB = decibel.
Indeterminate.
The primary finding toward the goal of characterizing the phenotype of SMD is that, as shown in the left-most column in Table 4, the 10 most frequent signs of SMD involve motor speech processes within each of the three domains of Speech, Prosody, and Voice. Specifically, the 10 most frequent signs of early SMD included age-inappropriate motor behaviours in subdomains of Speech (Vowels, Consonants, and both Vowels and Consonants), Prosody (Rate, Stress), and Voice (Laryngeal Quality). A second finding in Table 4 is that 7 of the 10 most frequent signs of SMD involved speech production. A third finding is that the items were scored using both perceptual (6 signs) and acoustic (4 signs) methods. Last, as shown in the totals in the bottom rows of the right-most four columns in Table 4, the most frequent signs of SMD included both precision (7 signs) and stability (3 signs) attributes and spatial (5 signs) and temporal (4 signs) features.
Discussion
Findings in Table 4 are interpreted to indicate that rather than being circumscribed to only certain motor domains, the SMD phenotype is characterized by an ‘across-the-board’ delay in the acquisition of precise and stable speech, prosody, and voice, predominantly in movements subserving speech production. Moreover, the potentially core signs of SMD in Table 4 include delays in development of both spatial and temporal precision and stability. A technical report provides reference data for children with SD with and without concurrent SMD (Shriberg, Campbell, Mabie, & McGlothlin, 2019).
What does not emerge from these initial findings is a speech, prosody, and voice profile of SMD that provides quantitative support to SMD as a unique clinical entity relative to CD and CAS. Alternative multivariate statistical techniques to the present univariate analyses, which would require many more participants in each of the four motor speech classifications, could be informative. At present, SMD is proposed as a clinical classification that provides psychometric support for the concept of a delay in speech motor development, with implications for prediction, treatment, and eventually, prevention.
Question 3: did participants with SMD differ significantly from participants with No MSD on any participant variables?
Rationale
The phenotype analysis examined whether a participant’s status on any demographic, parent report, cognitive-linguistic, or speech variable was significantly associated with risk for SMD. Table 5 includes findings for the 50 participants in Shriberg et al. (2019) with SMD, with findings from the 341 participants with No MSD providing the control data.
Table 5.
Information for participants with Speech Motor Delay (SMD) and participants with No Motor Speech Disorder (No MSD).a.
| SMD |
No MSD |
Inferential Statistics |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | n | M | SD | % | n | M | SD | % | Confidence Interval | Effect Sizeb | p-valuec |
| Demographic | |||||||||||
| Age (yrs) | 50 | 5.1 | 1.7 | 341 | 4.8 | 1.7 | −0.12, 0.47 | 0.18 | |||
| Gender (male) | 80.0 | 70.0 | 0.181 | ||||||||
| Parent Report-Risk Factors | |||||||||||
| Speech, Language, & Learning | |||||||||||
| Articulation/Phonology | 19 | 94.7 | 129 | 79.8 | 0.200 | ||||||
| Apraxia | 19 | 15.8 | 128 | 7.0 | 0.189 | ||||||
| Dysarthria | 19 | 0.0 | 130 | 2.3 | 1.000 | ||||||
| Receptive language disorder | 20 | 15.0 | 133 | 7.5 | 0.380 | ||||||
| Expressive language | 19 | 36.8 | 127 | 14.2 | 0.023 | ||||||
| disorder | |||||||||||
| Structural disorder | 19 | 5.3 | 130 | 4.6 | 1.000 | ||||||
| Fluency disorder | 20 | 10.0 | 133 | 4.5 | 0.281 | ||||||
| Voice Disorder | 20 | 5.0 | 132 | 2.3 | 0.435 | ||||||
| Other Disorder | 19 | 0.0 | 131 | 3.1 | 1.000 | ||||||
| Learning disability | 22 | 4.5 | 137 | 3.6 | 1.000 | ||||||
| Hearing | |||||||||||
| History of hearing problems | 20 | 5.0 | 134 | 9.0 | 1.000 | ||||||
| OME 16+ in first 18 months | 18 | 0.0 | 119 | 5.0 | 1.000 | ||||||
| PE tubes placed | 22 | 18.2 | 137 | 15.3 | 0.754 | ||||||
| Genetics | |||||||||||
| Nuclear family member with | 20 | 30.0 | 132 | 48.5 | 0.152 | ||||||
| verbal trait disorder | |||||||||||
| Cognitive-Linguistic | |||||||||||
| Syllable Repetition Taskd | |||||||||||
| Performance | 24 | −1.18 | 1.39 | 174 | −0.83 | 1.36 | −0.68, 0.17 | −0.26 | |||
| Encoding | 25 | −0.71 | 0.79 | 171 | −0.25 | 1.10 | −0.85, −0.01 | −0.43 S | |||
| Memory | 25 | −1.02 | 1.91 | 177 | −0.49 | 1.48 | −0.76, 0.08 | −0.34 | |||
| Language | |||||||||||
| OWLS: Oral Compositee | 20 | 97.2 | 13.6 | 58 | 94.7 | 14.4 | −0.33, 0.68 | 0.17 | |||
| Average Words/Utterance | 50 | −1.43 | 0.97 | 341 | −0.93 | 1.44 | −0.66, −0.06 | 0.36 S | |||
| Speech | |||||||||||
| PCC | 50 | 71.8 | 14.3 | 341 | 74.5 | 12.2 | −0.51, 0.08 | −0.22 | |||
| PCC-Z | 50 | −3.06 | 1.42 | 341 | −2.51 | 1.51 | −0.66, −0.07 | −0.37 S | |||
| PVC | 50 | 91.9 | 6.1 | 341 | 93.6 | 4.4 | −0.66, −0.07 | −0.36 S | |||
| PVC-Z | 50 | −3.24 | 1.97 | 341 | −2.68 | 1.84 | −0.60, 0.00 | −0.30 | |||
| II | 50 | 88.8 | 10.2 | 341 | 92.1 | 7.8 | −0.70, −0.11 | −0.40 S | |||
| II-Z | 50 | −3.35 | 2.02 | 341 | −2.65 | 2.10 | −0.63, −0.04 | −0.33 S | |||
| OIIf: % Lowered Intelligibility | 34.0 | 14.1 | 0.002 | ||||||||
Notes: OME = Otitis Media with Effusion; PE = Pressure Equalization; PCC-Z = Percentage of Consonants Correct-Z-score; PVC = Percentage of Vowels Correct; PVC-Z = Percentage of Vowels Correct-Z-score; II-Z = Intelligibility Index-Z-score; OII = Ordinal Intelligibility Index.
The right-most three columns include findings from between-group inferential statistical analyses of each variable. Statistically significant findings are bolded.
Effect Size: Small (S) >0.2; Medium (M) >0.5; Large (L) >0.8.
Fisher’s exact tests.
Oral Composite Standard Score from Oral and Written Language Scales (OWLS; Carrow-Woolfolk, 1995).
Mabie and Shriberg (2017). Lowered intelligibility: <85%.
Findings
Two sets of findings in Table 5 provide additional information on the SMD phenotoype. First, other than speech status, the three variables in Table 4 that significantly differentiated participants with SMD from those with No MSD were each associated with language processes. Significantly more participants with SMD had a parent-reported expressive language disorder, lower encoding scores on a nonword repetition task, and a lower average number of words per utterance in conversational speech. Second, relative to the speech variables, participants with SMD had significantly lower scores than participants with No MSD on Percentage of Consonants Correct-Z-Score, Percentage of Vowels Correct, Intelligibility Index, and Intelligibility Index-Z-score, and they had a significantly higher percentage of participants with Lowered intelligibility on the Ordinal Intelligibility Index. Relative to the speech variables, the summary consonant, vowel, and intelligibility findings in Table 5, adjusted for age and sex, document lowered speech competence as a significant feature of the SMD phenotype.
Discussion
A central research and clinical question is whether the type and/or severity of language impairment in SD in children with concurrent SMD differs significantly from the language profiles of children with SD but not SMD, a question that could not be addressed due to the diverse language measures used in the present database. Clearly, associations between neurocognitive and neuromotor developmental processes are of central interest in speech-language disorders, with SMD a possibly useful research entity to bridge research in the two domains.
Question 4: did early SMD persist past 6 or 9 years of age in any participant treated for idiopathic SD?
Rationale
Children with developmental delays in the acquisition of behaviours may self-correct without treatment (i.e., a primary rationale for the clinical recommendation to ‘watch and wait’). Consistent with contemporary clinical practice, each of the 14 children with SD in the database described in Table 2 were provided treatment beginning at preschool age. The last two questions addressed in the present study examined the persistence of SMD as rationale for replacing the provisional term Motor Speech Disorder-Not Otherwise Specified with Speech Motor Delay (SMD). We use the conventional ages of 6 years as the terminus point for typical acquisition of most English consonants (e.g., Campbell et al., 2003; Shriberg, Kwiatkowski, & Gruber, 1994), and Persistent SD for consonant deletions and substitutions that persist past 9 years (Shriberg, Gruber, & Kwiatkowski, 1994). Lacking empirical support at present, it would seem that a conservative arbitrary designation would be to term SMD after 9 years of age as Persistent SMD.
Findings
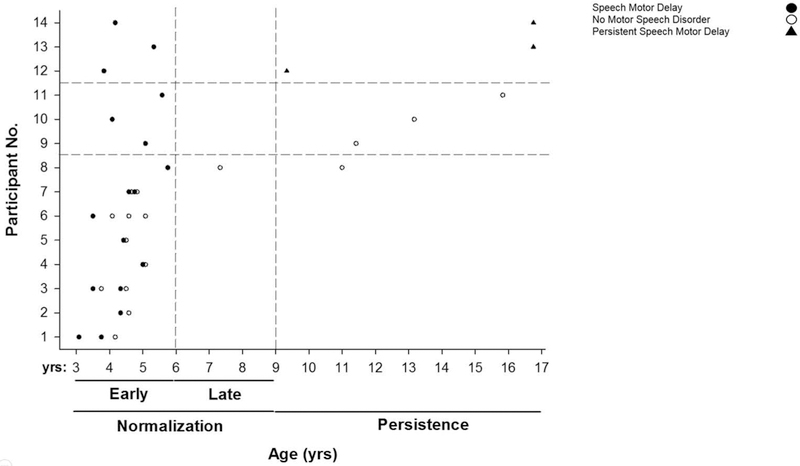
Table 2 includes descriptive information for 14 children treated for idiopathic SD, some over a lengthy period in which multiple conversational speech samples were obtained for clinical probes and/or research needs. Figure 1 includes speech motor classification findings for each of the 14 participants in Table 2 organized to address the present question of whether early SMD persisted past 6 or 9 years in any of the participants. The symbols in Figure 1 indicate participants with early SMD (filled circles) who normalized (No MSD at assessment; open circles) by 6 years or 9 years of age or had Persistent SMD at assessment (filled triangles) after 9 years of age.
Figure 1.
Persistence of Speech Motor Delay (SMD) in 14 participants treated for idiopathic Speech Delay (SD).
As shown in Figure 1, SMD persisted in 3 of the 14 (21.4%) of the participants after 9 years of age. The remaining 11 (78.6%) participants normalized SMD before 9 years, with no available data points between 6 and 9 years for participants 9, 10, and 11 to estimate if they may have normalized during that period. Correlation analyses of the early PSI percentage scores did not indicate any association between the magnitudes of the percentage scores and persistent SMD.
Discussion
The findings in Figure 1 indicating that nearly 80% of the present participants normalized SMD (i.e., met criteria for No MSD by 9 years of age) are viewed as support for the construct validity of a motor speech disorder characterized by a speech motor delay. The present data are not informative on whether normalization by 9 years of age would have occurred if these latter participants had not been in treatment for SD or what type(s) of treatment might have been focused on motor speech targets. As well, the present findings raise the question of whether the three participants with persistent SMD may have normalized if their SMD had been identified and at least partially treated using principles of motor learning (e.g., Maas et al., 2008).
Question 5: were any demographic or early speech variables significantly associated with the persistence of SMD in participants treated for SD?
Rationale
The primary goal of the final analyses was to examine whether age, sex, or any of the speech measures for the 14 participants with SMD prior to 6 years of age were significantly associated with the persistence of SMD past 9 years of age.
Findings
Table 6 includes findings for the two demographic variables and the nine raw and standardized measures of consonants, vowels, and intelligibility. None of the 11 comparisons were statistically significant. Notably, differences in the severity of early SD were not associated with SMD persistence findings. Specifically, participants whose SMD normalized by 9 years of age averaged poorer early speech production and intelligibility scores (see Table 2 for ages of participants at preschool assessment) than the participants whose SMD persisted after 9 years of age. For example, the averages of the Percentage of Consonants Correct raw and z-scores of participants with Normalized SMD were lower than the average Percentage of Consonants Correct scores of participants with Persistent SMD.
Table 6.
Speech findings for 14 participants with concurrent Speech Delay and early Speech Motor Delay (SMD) treated for Speech Delay.
| Normalized SMD by 9 years of age (n = 11) |
Persistent SMD (n = 3) |
Between-group Comparisons |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | n | M | SD | % | n | M | SD | % | Confidence Interval | Effect Size | p-valuea |
| Demographic | |||||||||||
| Gender (male) | 11 | 81.8 | 3 | 100.0 | 1.000 | ||||||
| Age (yrs) | 11 | 4.5 | 0.9 | 3 | 4.3 | 0.6 | −1.06, 1.50 | 0.22 | |||
| Speechb | |||||||||||
| PCC | 11 | 66.6 | 11.6 | 3 | 71.6 | 7.3 | −1.71, 0.86 | −0.43 | |||
| PCC-Z | 11 | −3.30 | 1.15 | 3 | −2.37 | 1.50 | −2.02, 0.59 | −0.72 | |||
| PCCR | 11 | 72.0 | 12.2 | 3 | 80.5 | 8.0 | −1.99, 0.62 | −0.69 | |||
| PCCR-Z | 11 | −4.19 | 1.04 | 3 | −2.97 | 1.89 | −2.26, 0.39 | −0.93 | |||
| PVC | 11 | 90.6 | 5.5 | 3 | 91.2 | 0.9 | −1.39, 1.17 | −0.11 | |||
| PVC-Z | 11 | −3.58 | 1.67 | 3 | −4.74 | 0.45 | −0.60, 2.01 | 0.71 | |||
| II | 11 | 86.4 | 9.7 | 3 | 87.6 | 14.9 | −1.38, 1.17 | −0.10 | |||
| II-Z | 11 | −3.55 | 2.12 | 3 | −2.65 | 2.09 | −1.68, 0.89 | −0.40 | |||
| OIIc: % Lowered Intelligibility | 54.5 | 33.3 | 1.000 | ||||||||
Notes: PCC = Percentage of Consonants Correct; PCC-Z = Percentage of Consonants Correct-Z-score; PCCR = Percentage of Consonants Correct-Revised; PCCR-Z = Percentage of Consonants Correct-Revised-Z-score; PVC = Percentage of Vowels Correct; PVC-Z = Percentage of Vowels Correct-Z-score; II = Intelligibility Index; II-Z – Intelligibility Index-Z-score; OII = Ordinal Intelligibility Index.
Fisher’s exact tests.
At first assessment.
Mabie and Shriberg (2017). Lowered intelligibility: <85%.
Discussion
There were too few available data on demographic and language variables to adequately explore their potential association with the phenotype and persistence of SMD. Specifically, the available retrospective data are not sufficient to support speculation on the interaction of language variables and the severity and persistence of SD with and without early concurrent SMD. If the trends in the present study are obtained in prospective cross-validation studies, they would support the construct validity of SMD as a clinical entity, perhaps particularly for children with PSD.
Conclusion
Two methodological limitations of the present research warrant comment. First, the behavioural measures of SMD, CD, and CAS used in this research have only recently become available, warranting replication to estimate their internal and external validity. Second, SMD findings in the present paper are based on retrospective data. Prospective phenotype and persistence studies are needed to assess the generalizability of findings with large, representative samples from diverse clinical communities. Findings from recent treatment studies by Namasivayam and colleagues (Namasivayam, Granata, Huynh, & Van Lieshout, 2018; Namasivayam et al., in press) support the construct validity of SMD, using rationale and methods similar to those described in Shriberg (2017).
The present phenotype and persistence findings for SMD are interpreted to support SMD as a valid childhood motor speech disorder. As with CD and CAS, basic and applied research in SMD in idiopathic and symptomatic contexts is needed to explicate its biobehavioural origins and their implications for prevention and treatment. A possibility is that although the speech processing phenotypes of SMD, CD, and CAS presumably differ, the prevalence, phenotype, and persistence findings reported support the likelihood of common biologic pathways across the three disorders. Specifically, the reported comorbidity of speech, motor speech, and language disorder is consistent with the possibility of identifying causal genes that confer risk for pleiotropic effects within and among neurodevelopmental domains (Liégeois et al., 2019; Miscimarra et al., 2007; Peter, Matsushita, & Raskind, 2012; Smith, Pennington, Boada, & Shriberg, 2005; Stein et al., 2006, 2004).
Supplementary Material
Acknowledgments
We thank each of the following colleagues for her or his contribution to this research: Richard Boada, Roger Brown, Joseph Duffy, Marios Fourakis, Lisa Freebairn, Jordan Green, Sheryl Hall, Katherina Hauner, Kathy Jakielski, Heather Karlsson, Barbara Lewis, Jane McSweeny, Christopher Moore, Katharine Odell, Bruce Pennington, Nancy Potter, Erin Redle, Heather Rusiewicz, Carmen Rasmussen, Alison Scheer-Cohen, Kristie Spencer, Edythe Strand, Christie Tilkens, Jennifer Vannest, Jennell Vick, and David Wilson.
Funding
This work was supported by a grant from the National Institute on Deafness and Other Communication Disorders [DC000496] and a core grant to the Waisman Center from the National Institute of Child Health and Human Development [U54 HD090256].
Abbreviations:
- CAS
Childhood Apraxia of Speech
- CD
Childhood Dysarthria
- MSD
Motor Speech Disorder
- No MSD
No Motor Speech Disorder
- PSI
Precision-Stability Index
- SD
Speech Delay
- SMD
Speech Motor Delay
- SSD
Speech Sound Disorders
Appendix. Speech, motor speech, and dysarthria subtype classifications in the Speech Disorders Classification System (SDCS).
| The five Speech classifications and five Motor Speech classifications in the SDCS are each mutually exclusive. The five dysarthria subtype classifications are not mutually exclusive. That is, a speaker can meet percentile criteria for more than one of the five listed dysarthria subtype classifications (i.e., mixed dysarthria). See Supplement for the procedures and measures used to classify each motor speech disorder. | ||||
|---|---|---|---|---|
| SDCS Classifications and Dysarthria Subtypes | Abbreviation | Age (yrs;mos) at Assessment | Description | Referencesa |
| Five Speech Classifications | ||||
| Normal(ized) Speech Acquisition | NSA | 3–80 | Does not meet criteria for any of the four Speech Disorder classifications | 2, 3, 4 |
| Speech Errors | SE | 6–8;11 | Age-inappropriate speech sound distortions | 3, 4 |
| Persistent Speech Errors | PSE | 9–80 | Age-inappropriate speech sound distortions that persist past 9 years of age | 4, 5 |
| Speech Delay | SD | 3–8;11 | Age-inappropriate speech sound deletions and/or substitutions | 3, 4 |
| Persistent Speech Delay | PSD | 9–80 | Age-inappropriate speech sound deletions and/ or substitutions that persist past 9 years of age | 3, 4, 5 |
| Five Motor Speech Classifications | ||||
| No Motor Speech Disorder | No MSD | 3–80 | Does not meet criteria for any of the four Motor Speech Disorders classifications | 2, 6, 8 |
| Speech Motor Delay | SMD | 3–80 | Meets PSI criterion for SMD | 2, 6, 8 |
| Childhood Dysarthria | CD | 3–80 | Meets DI and DSI criteria for CD | 2, 6, 8 |
| Childhood Apraxia of Speech | CAS | 3–80 | Meets PM criterion for CAS | 6, 7, 8 |
| Childhood Dysarthria & Childhood Apraxia of Speech | CD & CAS | 3–80 | Meets SDCS criteria for CD & CAS | 2, 6, 8 |
| Five Dysarthria Subtypes | ||||
| Ataxic | 3–80 | Cerebellar disorder | 1, 2 | |
| Spastic | 3–80 | Upper motor neuron disorder | 1, 2 | |
| Hyperkinetic | 3–80 | Basal ganglia disorder; increased movement | 1, 2 | |
| Hypokinetic | 3–80 | Basal ganglia disorder; decreased movement | 1, 2 | |
| Flaccid | 3–80 | Lower motor neuron disorder | 1, 2 | |
Notes: PSI = Precision-Stability Index; DI = Dysarthria Index; DSI = Dysarthria Subtype Index; PM = Pause Marker.
Footnotes
Disclosure Statement
The authors report no declarations of interest.
References
- American Speech-Language-Hearing Association. (2007). Childhood apraxia of speech (Technical report) Retrieved from http://www.asha.org/public/speech/disorders/ChildhoodApraxia/doi: 10.1094/PDIS-91-4-0467B [DOI] [Google Scholar]
- Baylis AL, & Shriberg LD (2018). Estimates of the prevalence of speech and motor speech disorders in youth with 22q11.2 Deletion syndrome. American Journal of Speech-Language Pathology Advance online publication. doi: 10.1044/2018_AJSLP-18-0037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell TF, Dollaghan CA, Rockette HE, Paradise JL, Feldman HM, Shriberg LD, … Kurs-Lasky M (2003). Risk factors for speech delay of unknown origin in 3-year-old children. Child Development, 74, 346–357. [DOI] [PubMed] [Google Scholar]
- Carrow-Woolfolk E (1995). Oral and Written Language Scales (OWLS) Bloomington, MN: Pearson Assessment. [Google Scholar]
- Carrow-Woolfolk E (2011). Oral and Written Language Scales (2nd ed.). San Antonio, TX: Pearson Assessments. [Google Scholar]
- DiSimoni F (1978). The token test for children Boston, MA: Teaching Resources Corporation. [Google Scholar]
- Duffy JR (2013). Motor speech disorders: Substrates, differential diagnosis, and management (3rd ed.). St. Louis, MO: Mosby. [Google Scholar]
- Dunn M, & Dunn LM (1981). Peabody picture vocabulary test—revised Circle Pines, MN: AGS. [Google Scholar]
- Feise RJ (2002). Do multiple outcome measures require p-value adjustment? BMC Medical Research Methodology, 2, 8–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammill D, & Bryant B (2005). Detroit Tests of Learning Aptitude-Primary – Third Edition (DTLA-P-3) Austin, TX: Pro-Ed. [Google Scholar]
- Kaufman AS, & Kaufman NL (2004). Kaufman Brief Intelligence Test – Second Edition (KBIT-2) San Antonio, TX: Pearson Assessments. [Google Scholar]
- Liégeois FJ, Turner SJ, Mayes A, Bonthrone AF, Boys A, Smith L, … Morgan AT (2019). Dorsal language stream anomalies in an inherited speech disorder. Brain doi: 10.1093/brain/awz018 [DOI] [PubMed] [Google Scholar]
- Maas E, Robin DA, Austermann Hula SN, Freedman SE, Wulf G, Ballard KJ, & Schmidt RA (2008). Principles of motor learning in treatment of motor speech disorders. American Journal of Speech-Language Pathology, 17, 277–298. doi: 10.1044/1058-0360(2008/025) [DOI] [PubMed] [Google Scholar]
- Mabie HL, & Shriberg LD (2017). Speech and motor speech measures and reference data for the Speech Disorders Classification System (SDCS). (Technical Report No. 23). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- McSweeny J (1998). Procedures to obtain extended conversational speech samples for prosody-voice analysis (Technical Report No. 7). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Miller JF, & Yoder DE (1975). The miller-yoder test of grammatical comprehension Madison: The University of Wisconsin-Madison. [Google Scholar]
- Miscimarra L, Stein C, Millard C, Kluge A, Cartier K, Freebairn L, … Iyengar SK (2007). Further evidence of pleiotropy influencing speech and language: Analysis of the DYX8 region. Human Heredity, 63, 47–58. doi: 10.1159/000098727 [DOI] [PubMed] [Google Scholar]
- Morgan AT, & Liégeois F (2010). Re-thinking diagnostic classification of the dysarthrias: A developmental perspective. Folia Phoniatrica Et Logopaedica, 62, 120–126. doi: 10.1159/000287210 [DOI] [PubMed] [Google Scholar]
- Nakagawa S (2004). A farewell to Bonferroni: The problems of low statistical power and publication bias. Behavioral Ecology, 15, 1044–1045. doi: 10.1093/beheco/arh107 [DOI] [Google Scholar]
- Namasivayam AK, Granata F, Huynh A, & Van Lieshout PHHM (2018, November). Randomized control trial of PROMPT intervention for children with severe speech sound disorders. Paper presented at the Annual Convention of the American Speech-Language-Hearing Association, Boston, MA. [Google Scholar]
- Namasivayam AK, Pukonen M, Goshulak D, Francesca G, Le DL, Kroll R, & Van Lieshout PHHM (in press). Investigating intervention dose frequency for children with speech sound disorders and motor speech involvement. International Journal of Language & Communication Disorders [DOI] [PubMed]
- Newcomer P, & Hammill D (1988). Test of Language Development-Primary (TOLD-P) Austin, TX: Pro-Ed. [Google Scholar]
- Perneger TV (1998). What’s wrong with Bonferroni adjustments? British Medical Journal, 316, 1236–1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peter B, Matsushita M, & Raskind WH (2012). Motor sequencing deficit as an endophenotype of speech sound disorder: A genome-wide linkage analysis in a multigenerational family. Psychiatric Genetics, 22, 226–234. doi: 10.1097/YPG.0b013e328353ae92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potter NL, Hall S, Karlsson HB, Fourakis M, Lohmeier HL, McSweeny JL, … Shriberg LD (2012). Reference data for the madison speech assessment protocol (MSAP): A database of 150 participants 3-to-18 years of age with typical speech. (Technical Report No. 18). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Programs to Examine Phonetic and Phonologic Evaluation Records [PEPPER: Computer software] (2019). Madison: Waisman Center, University of Wisconsin–Madison. [Google Scholar]
- Royal College of Speech and Language Therapists. (2011). Developmental Verbal Dyspraxia London, England: Royal College of Speech and Language Therapists. [Google Scholar]
- Scheer-Cohen AR, Holt AS, Karlsson HB, Mabie HL, McSweeny JL, Tilkens CM, & Shriberg LD (2013). Reference data for the madison speech assessment protocol (MSAP): A database of fifty 20-to-80 year old participants with typical speech. (Technical Report No. 20). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Semel E, Wiig E, & Secord W (2003). Clinical Evaluation of Language Fundamentals-4 (CELF– 4) San Antonio, TX: Harcourt Assessment. [Google Scholar]
- Shriberg LD (1993). Four new speech and prosody-voice measures for genetics research and other studies in developmental phonological disorders. Journal of Speech and Hearing Research, 36, 105–140. doi: 10.1044/jshr.3601.105 [DOI] [PubMed] [Google Scholar]
- Shriberg LD (2010a). Childhood speech sound disorders: From post-behaviorism to the postgenomic era. In Paul R & Flipsen P (Eds.), Speech sound disorders in children (pp. 1–34). San Diego, CA: Plural Publishing. [Google Scholar]
- Shriberg LD (2010b). A neurodevelopmental framework for research in childhood apraxia of speech. In Maassen B & van Lieshout P (Eds.), Speech motor control: New developments in basic and applied research (pp. 259–270). New York, NY: Oxford University Press. [Google Scholar]
- Shriberg LD (2017, July). Motor speech disorder-not otherwise specified: Prevalence and phenotype. Paper presented at the 7th International Conference on Speech Motor Control, Groningen, the Netherlands. [Google Scholar]
- Shriberg LD, Austin D, Lewis BA, McSweeny JL, & Wilson DL (1997). The Percentage of Consonants Correct matrix (PCC): Extensions and reliability data. Journal of Speech, Language, and Hearing Research, 40, 708–722. doi: 10.1044/jslhr.4004.708 [DOI] [PubMed] [Google Scholar]
- Shriberg LD, Campbell TF, Mabie HL, & McGlothlin JH (2019). Reference data for children with idiopathic speech delay with and without concurrent speech motor delay. (Technical Report No. 26). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Shriberg LD, Fourakis M, Hall S, Karlsson HB, Lohmeier HL, McSweeny JL, … Wilson DL (2010a). Extensions to the Speech Disorders Classification System (SDCS). Clinical Linguistics & Phonetics, 24, 795–824. doi: 10.3109/02699206.2010.503006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shriberg LD, Fourakis M, Hall S, Karlsson HB, Lohmeier HL, McSweeny JL, … Wilson DL (2010b). Perceptual and acoustic reliability estimates for the Speech Disorders Classification System (SDCS). Clinical Linguistics & Phonetics, 24, 825–846. doi: 10.3109/02699206.2010.503007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shriberg LD, Gruber FA, & Kwiatkowski J (1994). Developmental phonological disorders III: Long-term speech-sound normalization. Journal of Speech and Hearing Research, 37, 1151–1177. doi: 10.1044/jshr.3705.1151 [DOI] [PubMed] [Google Scholar]
- Shriberg LD, & Kwiatkowski J (1985). Continuous speech sampling for phonologic analyses of speech-delayed children. Journal of Speech and Hearing Disorders, 50, 323–334. [DOI] [PubMed] [Google Scholar]
- Shriberg LD, Kwiatkowski J, & Gruber FA (1994). Developmental phonological disorders II: Short-term speech-sound normalization. Journal of Speech and Hearing Research, 37, 1127–1150. doi: 10.1044/jshr.3705.1127 [DOI] [PubMed] [Google Scholar]
- Shriberg LD, Kwiatkowski J, & Mabie HL (2019). Estimates of the prevalence of motor speech disorders in children with idiopathic speech delay. Clinical Linguistics & Phonetics doi: 10.1080/02699206.2019.1595731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shriberg LD, & Lohmeier HL (2008). The syllable repetition task (Technical Report No. 14). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Shriberg LD, Lohmeier HL, Campbell TF, Dollaghan CA, Green JR, & Moore CA (2009). A nonword repetition task for speakers with misarticulations: The Syllable Repetition Task (SRT). Journal of Speech, Language, and Hearing Research, 52, 1189–1212. doi: 10.1044/1092-4388(2009/08-0047) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shriberg LD, & Mabie HL (2017). Speech and motor speech assessment findings in eight complex neurodevelopmental disorders. (Technical Report No. 24). Phonology Project, Waisman Center, University of Wisconsin-Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Shriberg LD, Potter NL, & Strand EA (2011). Prevalence and phenotype of childhood apraxia of speech in youth with galactosemia. Journal of Speech, Language, and Hearing Research, 54, 487–519. doi: 10.1044/1092-4388(2010/10-0068) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shriberg LD, Strand EA, Fourakis M, Jakielski KJ, Hall SD, Karlsson HB, … Wilson DL (2017). A diagnostic marker to discriminate childhood apraxia of speech from speech delay: III. Theoretical coherence of the pause marker with speech processing deficits in childhood apraxia of speech. Journal of Speech, Language, and Hearing Research, 60, S1135–S1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith SD, Pennington BF, Boada R, & Shriberg LD (2005). Linkage of speech sound disorder to reading disability loci. Journal of Child Psychology and Psychiatry, 46, 1057–1066. doi: 10.1111/j.1469-7610.2005.01534.x [DOI] [PubMed] [Google Scholar]
- Stein CM, Millard C, Kluge A, Miscimarra LE, Cartier KC, Freebairn LA, … Iyengar SK (2006). Speech sound disorder influenced by a locus in 15q14 region. Behavior Genetics, 36, 858–868. doi: 10.1007/s10519-006-9090-7 [DOI] [PubMed] [Google Scholar]
- Stein CM, Schick JH, Gerry Taylor H, Shriberg LD, Millard C, Kundtz-Kluge A, … Iyengar SK (2004). Pleiotropic effects of a chromosome 3 locus on speech-sound disorder and reading. American Journal of Human Genetics, 74, 283–297. doi: 10.1086/381562 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tilkens CM, Karlsson HB, Fourakis M, Hall SD, Mabie HL, McSweeny JL, …, Shriberg LD (2017). A diagnostic marker to discriminate Childhood Apraxia of Speech (CAS) from Speech Delay (SD). (Technical Report No. 22). Phonology Project, Madison, WI: Waisman Center, University of Wisconsin–Madison; Retrieved from Phonology Project website: http://www.waisman.wisc.edu/phonology/ [Google Scholar]
- Wechsler D (1999). Wechsler abbreviated scale of intelligence San Antonio, TX: Pearson Corporation. [Google Scholar]
- Zimmerman IL (1969). Preschool language scale Columbus, OH: Charles E. Merrill Publishing Company. [Google Scholar]
- Zimmerman IL, Steiner VG, & Pond RE (1992). Preschool Language Scale: Third Edition (PLS-3) San Antonio, TX: Psychological Corporation. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.