Abstract
Mesenchymal origin of primary thyroid angiosarcomas (TAS) is extremely rare and comprises less than 1% of primary thyroid cancer worldwide. While TAS are most commonly occurring in the Alpine region, there are multiple reported cases of TAS in non-Alpine regions. Diagnosis of TAS is commonly made after thyroidectomy as cytologic diagnosis can be challenging due to paucity of cells, presence of necrosis and unawareness of the disease due to rarity. We report a case of primary TAS diagnosed by cytology in a 56-year-old man who presented with a sudden onset of left neck pain, swelling and haemoptysis. He was later noted to have suspicious nodules on both lobes of thyroid on ultrasound. Fine needle aspiration of thyroid nodules showed malignant epithelioid cells. The diagnosis of TAS was made based on positive endothelial markers such as thrombomodulin and CD31, with many pertinent negatives, including negative cytokeratins,thyroid transcription factor (TTF1), thyroglobulin, calcitonin and carcinoembryonic antigen (CEA).
Keywords: thyroid disease, cancer intervention, endocrine cancer
Background
Angiosarcomas are malignant vascular tumours of endothelial origin and seen commonly in the skin and superficial soft tissue. Primary angiosarcomas at other sites including thyroid are less common. Cytologic diagnosis of thyroid angiosarcomas (TAS) is challenging due to paucity of cells, presence of necrosis and rarity of the disease. Diagnosis of TAS is typically made after thyroidectomy. However, the cytologic diagnosis of TAS is possible with a high index of clinical suspicion and the help of immunohistochemistry stains.
Case presentation
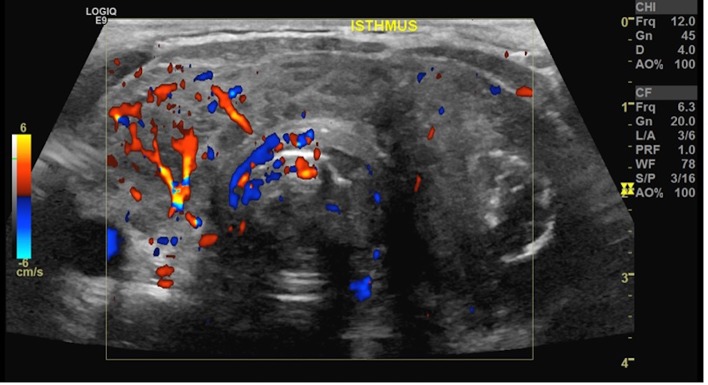
A 56-year-old Caucasian man with hypothyroidism presented with sudden onset of left neck pain and swelling in April of 2017. Neck CT showed bilateral thyroid masses with displacement of the trachea to the right. Thyroid ultrasound showed 3–4 cm irregular margin thyroid masses on both lobes of thyroid with a diffuse microcalcification and increased vascularity on the right thyroid mass and a ring-shaped macrocalcification inside the left thyroid mass (figure 1). While waiting for fine needle aspiration (FNA), he presented to the hospital 2 months later with haemoptysis, changes in voice, increased fullness of the neck and odynophagia.
Figure 1.
Thyroid ultrasound showed an ill-defined right thyroid nodule with microcalcification and a left thyroid nodule with a ring-shaped macrocalcification.
Investigations
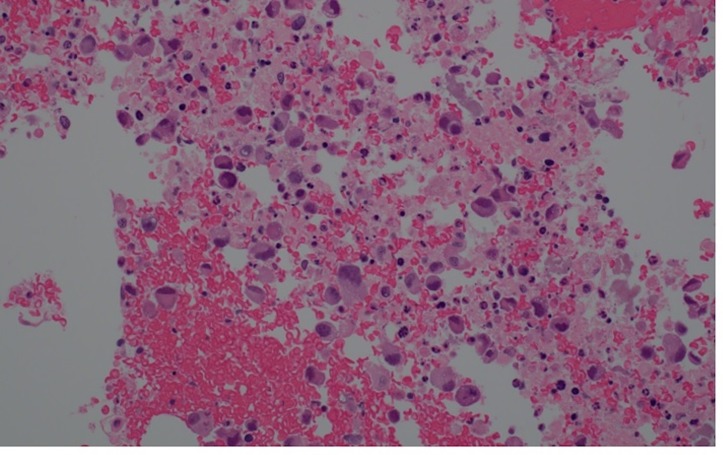
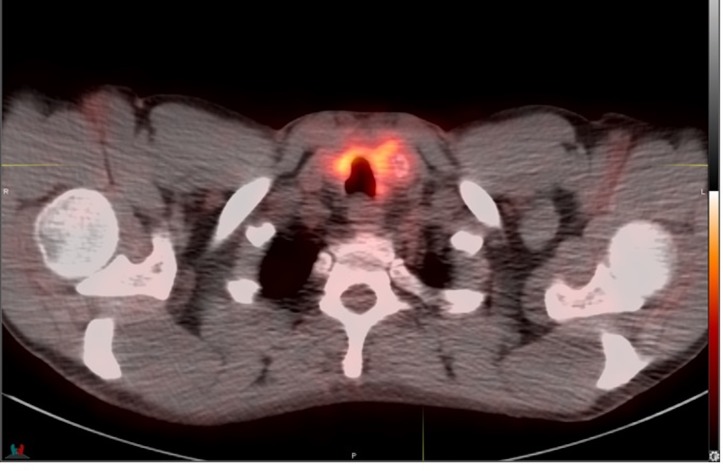
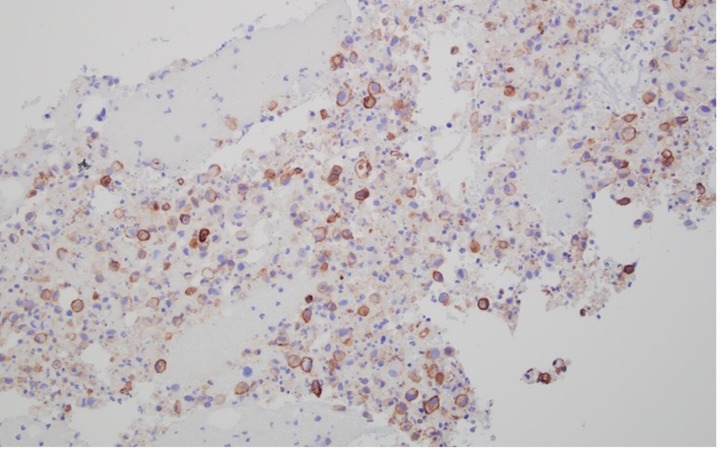
Flexible laryngoscopy showed areas of granulation in the subglottic region with no significant stenosis and normal vocal cords. FNA of bilateral thyroid masses showed abundant epithelioid cells with a plasmacytoid appearance. The nuclei were irregular with large nucleoli, small vacuoles within the cytoplasm and occasional degenerating/necrotic cells (figure 2). The initial cytological impression was malignant epithelioid cell with differential diagnosis of medullary thyroid cancer versus metastasis. However, medullary thyroid cancer was unlikely as the stains for calcitonin, thyroid transcription factor (TTF1), thyroglobulin, carcinoembryonic antigen (CEA) and synaptophysin along with serum tumour markers for neuroendocrine tumour such as CEA, chromogranin A and calcitonin were negative. Positron emission tomography (PET)–CT scan showed bilateral fluorodeoxyglucose (FDG) avid lesions on both lobes of the thyroid (figure 3), FDG avid cervical and superior mediastinal lymphadenopathy, and FDG accumulation in the manubrium. As the cytology sample of initial thyroid nodule was inadequate to perform further immunohistochemical stains, repeat biopsy of right thyroid nodule and manubrium was performed which showed similar malignant epithelioid cells with positive staining for thrombomodulin (figure 4), CD99, CD31 (focal weak) (figure 5), CD30 (partial) and smooth muscle actin (SMA) (focal weak). The tumour was negative for CD34, D2-40, PAX8, p53 (wild-type pattern), caldesmon, desmin, smooth muscle myosin, Myo-D1, CD117, CD15, ALK1, SALL4, calretinin, cytokeratins (CKAE1/AE3), EMA, CD45, Mart-1/MelanA, HMB45 and S100. The diagnosis of poorly differentiated sarcoma, most consistent with angiosarcoma, was made based on positive endothelial markers such as thrombomodulin and CD31, with many pertinent negatives, including negative cytokeratins, TTF1, thyroglobulin, PAX8 and p53. Though CD31 stain was weak in our case, we believed the tumour expressed CD31 as there was no background or non-specific staining noticed with any of the other negative stains, allowing a high degree of confidence when interpreting the somewhat weak CD31 stain. Additional stains for vascular differentiation such as erythroblastosis virus E26 oncogene homolog (ERG) and Freund’s leukaemia integration site 1 (FLI1) were not available at our facility, and were deemed unnecessary due to the morphology, immune profile with two positive vascular markers (CD31 and thrombomodulin) and many pertinent negatives.
Figure 2.
Malignant epithelioid cells with prominent nucleoli on cell block (×400).
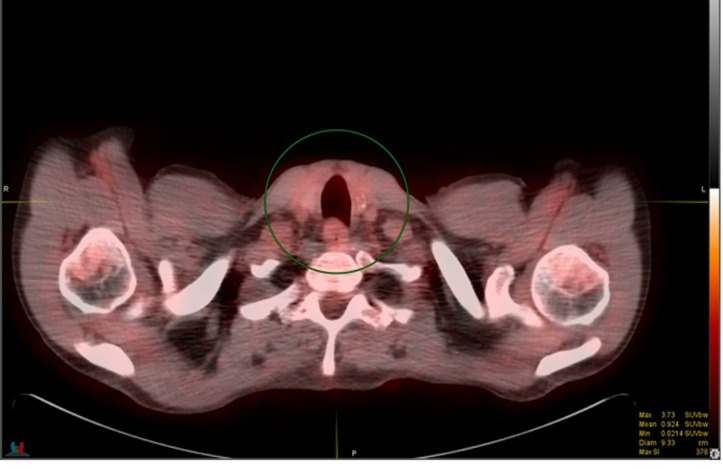
Figure 3.
PET–CT scan with bilateral FDG avid lesions on both lobes of the thyroid. The initial standardized uptake values (SUV) of the tumour was 7.2 and it was increased to 10.8 on the delayed images.
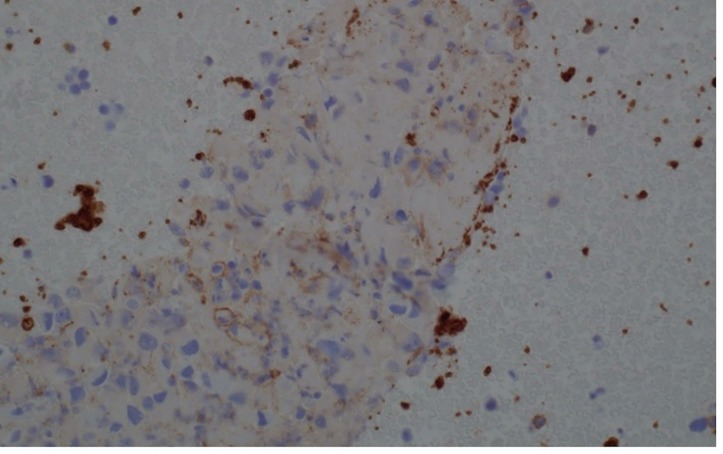
Figure 4.
Positive thrombomodulin (×200).
Figure 5.
Positive CD31 (focal and weak) (×400).
Treatment
After the cytologic diagnosis of TAS, the treatment options were discussed at the tumour board. Based on presentation with haemoptysis and radiological findings of FDG avid lesions at manubrium, bilateral cervical and mediastinal lymph nodes, the surgical approach might not be limited to total thyroidectomy and bilateral neck dissection. Surgery might involve total laryngectomy, tracheal resection, esophagectomy with gastric pull-up, resection of sternum, gastric tube and a mediastinal airway. Our patient declined local tumour control with radical surgery as he preferred not to have a tracheostomy or gastric tube even as a temporary measure. He completed six cycles of chemotherapy with combination of doxorubicin, ifosfamide and mesna every 21 days. There was a resolution of FDG avid lesions in the thyroid, neck and manubrium on PET/CT acquired at a month (figure 6) and 7 months after the completion of chemotherapy. As he had a complete response to initial chemotherapy, consolidative XRT (radiation therapy) to the thyroid bed was started 6 months after the completion of chemotherapy. He received a total nominal dose of 6000 cGy to the thyroid bed in 30 fractions over 51 elapsed days and tolerated well.
Figure 6.
Resolution of FDG avid lesions in thyroid on PET/CT obtained a month after the completion of chemotherapy.
Outcome and follow-up
He is clinically doing well 23 months after initial presentation with no radiologic evidence of disease recurrence at the time of our case submission.
Discussion
Primary thyroid sarcomas (PTS) are rare sarcomas and represent less than 1% of all sarcoma diagnoses.1 PTS consist of angiosarcomas, malignant haemangioendotheliomas, fibrous histiocytomas, leiomyosarcomas, fibrosarcomas, osteosarcomas and liposarcomas. Surov et al’s review of PTS yielded 113 reported cases of PTS between 1990 and 2014. TAS were the most common with 29 cases.2 Typically, TAS are seen in elderly patients with a history of goitre in the Alpine area.3 4 It represents 2%–10% of all thyroid cancers in the central European Alpine regions of Northern Italy, Switzerland and Austria.5 6 However, a recent report quoted TAS as 15%–20% of all thyroid malignancies in people living in European Alpine regions.7 This is thought to be due to an iodine deficiency leading to goitres in those areas.4 8 While most commonly occurring in the Alpine region, there are multiple reported cases of angiosarcomas in non-Alpine regions.5 9 Rotellini et al accounted for 24 cases of TAS in a non-Alpine region from 1990 to 2015.10 Most recently, De Felice et al identified 61 patients with TAS reported in English literature. Only five patients in this case series report were from the USA.7
The exact aetiology of TAS is unknown; however, prevalence is higher in mountainous Alpine regions of Europe where iodine deficiency and endemic goitre are common.3 4 7 History of exposure with ionising radiation, polyvinyl chloride and thorium dioxide are considered potential risk factors for TAS.7 Typical age of presentation is between 50 and 80 years of age. It is female predominate with female to male ratio of 9:3.3–5 9 Majority of these tumours are painless and large; however, size can vary from 2.5 cm to 13 cm with an average of 6 cm tumour size.5 6 11 They are locally aggressive and can present with sudden onset of pain from intra-nodular haemorrhage or compressive symptoms from sudden growth.12 In addition to loco-regional involvement, metastases to skin, bone, bone marrow, brain and lung have been reported.11 13 Some may present with complications from metastasis such as cough, haemoptysis, anaemia and hypercalcaemia.13 14
The diagnosis of TAS is commonly made after thyroidectomy. The presence of angioinvasion, extrathyroidal extension (ETE) and distant metastasis at the time of presentation generally carries poor prognosis.5 7 15 Early diagnosis may have an impact on clinical outcome and prognosis by enabling an individualised treatment plan. However, cytologic diagnosis can be challenging due to paucity of cells, presence of necrosis and unawareness of the disease due to rarity. Angiosarcomas can have a spindled or epithelioid appearance, with deep soft tissue sites and thyroid more commonly having an epithelioid morphology. Typically, large rounded epithelioid cells with an abundant amphophilic or eosinophilic cytoplasm and large vesicular nuclei are identified on cytology. Histologically, those tumours typically show extensive necrosis with intratumour haemorrhage with or without extensive surrounding structure infiltration. Tumour cells can express both endothelial markers and cytokeratins. The presence of intracytoplasmatic rod-shaped membrane-bound Weibel-Palade bodies also suggests endothelial differentiation of tumour cells.6 Without the aids of immunohistochemical stain, TAS can be misdiagnosed as an angiomatoid variant of anaplastic carcinoma of thyroid or metastasis. The use of immunohistochemical stains for endothelial origin such as CD31, CD34, thrombomodulin, FLI1, ERG and factor VIII-related antigen help to differentiate primary angiosarcoma of thyroid from an endothelial variant of undifferentiated thyroid carcinoma. CD31 is considered the most sensitive and specific marker for endothelial differentiation, being expressed in 90% of angiosarcomas. While in this case the immunohistochemistry profile was most consistent with angiosarcoma, the distinction between primary angiosarcoma of the thyroid and angiomatoid variant of undifferentiated (anaplastic) thyroid carcinoma has been controversial and difficult, with some authors reporting the two as existing on a spectrum.16 There were some additional studies that examined the immunophenotypic and genetic profile differences of TAS and anaplastic carcinoma of thyroid. A recent case series study on the clinico-morphologic and genetic characteristics of 10 angiosarcomas and 22 anaplastic carcinomas by Kuhn et al found similar findings with CD31 and ERG as the most reliable endothelial markers for angiosarcomas as all cases of angiosarcoma in their series had positive CD31 and ERG. On the other hand, positivity of mutated pattern of p53 and PAX8 favoured a diagnosis of anaplastic carcinoma, as all angiosarcomas cases had negative mutated pattern of p53 and PAX8 whereas 71% and 50% of anaplastic carcinoma patients in their series had positive mutated pattern p53 and PAX8, respectively. In addition, authors explored the genetic profile of each group by using next-generation sequencing with findings of single-event mutation in angiosarcomas with no TP53 or TERT promoter mutations which were the most frequent genetic alterations in anaplastic carcinomas.17 A different study found no mRNA expression for thyroglobulin by cases of angiosarcoma with weak expression by cases of anaplastic carcinoma, suggesting that they have unrelated cells of origin.18 While we believe they are distinct entities, the significance of distinction is debatable, as the treatment and prognosis for both malignancies are currently similar.10 19
In patients with TAS, total thyroidectomy is widely accepted4 7; however, high vascularity and presence of ETE raise the concern for development of life-threatening haemorrhage during and after radical thyroidectomy.4 Adjuvant radiotherapy with total dose ranging from 53 Gy to 65 Gy has been well tolerated and may impact survival.7 11 14 20 Hence, neo-adjuvant or adjuvant chemotherapy and/or radiotherapy for both local and systemic tumour control may be considered based on presentation, patients’ preferences and presence or absence of ETE or systemic metastasis.
Despite a combined treatment modality, TAS carry a very poor prognosis. Metastatic disease is associated with a poor prognosis, with mean survival time of only a few months after diagnosis and surgical treatment. Most patients die in less than 6 months regardless of treatment with a few surviving up to 5 years. Goh et al showed a 5-year survival rate of 33.3%.5 Collini et al presented six cases with median follow-up of 59 months. Two out of six patients from their case series showed local relapse within 5 months of diagnosis and died from disease earlier than those who had no local relapse.11 Absence of ETE generally allows longer survival.21 Hence, early diagnosis is essential and can guide an individualised treatment plan. Unfortunately, the majority of cases were diagnosed after thyroidectomy. However, early diagnosis is possible with clinical awareness of this rare disease and appropriate application of immunohistochemical stains. In our case, as we could diagnose TAS with FNA and cell block, our patient could make an informed personal medical decision and avoid surgery.
Learning points.
Thyroid angiosarcomas (TAS) are locally aggressive and can present with sudden onset of pain from intra-nodular haemorrhage or compressive symptoms from sudden growth.
While diagnosis is typically made after thyroidectomy, awareness of this disease entity in the setting of a poorly differentiated tumour, discussion with the pathologist and use of appropriate immunohistochemistry staining make early diagnosis with fine needle aspiration possible.
CD31, Freund’s leukaemia integration site 1 and erythroblastosis virus E26 oncogene homolog are some of the most useful markers to show endothelial differentiation in tumours, including primary angiosarcoma of the thyroid. CD31 can be weak or focal as in this case.
There is no established consensus or optimal treatment approach for TAS due to rarity of disease and heterogeneity in previous therapeutic approach.
Prognosis is poor with the presence of extrathyroidal extension and distant metastasis at the time of presentation.
Footnotes
Contributors: All authors contributed equally to this manuscript: discuss planning, conduct, reporting, conception and design, acquisition of data as well as analysis and interpretation of data. AK: data gathering, reviewing and writing. LR: interpretation, data gathering, reviewing and writing. LD: interpretation, data gathering and reviewing. YO: data gathering, interpretation, writing and reviewing.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Obtained.
References
- 1. Weiss SW GJ. Soft tissue tumors. St Louis: Mosby, 2001. [Google Scholar]
- 2. Surov A, Gottschling S, Wienke A, et al. Primary Thyroid Sarcoma: a systematic review. Anticancer Res 2015;35:5185–91. [PubMed] [Google Scholar]
- 3. Petronella P, Scorzelli M, Luise R, et al. Primary thyroid angiosarcoma: an unusual localization. World J Surg Oncol 2012;10:73 10.1186/1477-7819-10-73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kalitova P, Plzak J, Kodet R, et al. Angiosarcoma of the thyroid. Eur Arch Otorhinolaryngol 2009;266:903–5. 10.1007/s00405-008-0820-8 [DOI] [PubMed] [Google Scholar]
- 5. Goh SG, Chuah KL, Goh HK, et al. Two cases of epithelioid angiosarcoma involving the thyroid and a brief review of non-Alpine epithelioid angiosarcoma of the thyroid. Arch Pathol Lab Med 2003;127:E70–3. [DOI] [PubMed] [Google Scholar]
- 6. Ryska A, Ludvíková M, Szépe P, et al. Epithelioid haemangiosarcoma of the thyroid gland. Report of six cases from a non-Alpine region. Histopathology 2004;44:40–6. 10.1111/j.1365-2559.2004.01772.x [DOI] [PubMed] [Google Scholar]
- 7. De Felice F, Moscatelli E, Orelli S, et al. Primary thyroid angiosarcoma: a systematic review. Oral Oncol 2018;82:48–52. 10.1016/j.oraloncology.2018.05.004 [DOI] [PubMed] [Google Scholar]
- 8. Kaur A, Didolkar MS, Thomas A. Angiosarcoma of the thyroid: a case report with review of the literature. Endocr Pathol 2013;24:156–61. 10.1007/s12022-013-9253-z [DOI] [PubMed] [Google Scholar]
- 9. Maiorana A, Collina G, Cesinaro AM, et al. Epithelioid angiosarcoma of the thyroid. Clinicopathological analysis of seven cases from non-Alpine areas. Virchows Arch 1996;429(2-3):131–7. [DOI] [PubMed] [Google Scholar]
- 10. Rotellini M, Vezzosi V, Bianchi S. Epithelioid angiosarcoma of the thyroid: report of a case from an italian non-alpine area and review of the literature. Endocr Pathol 2015;26:152–6. 10.1007/s12022-015-9372-9 [DOI] [PubMed] [Google Scholar]
- 11. Collini P, Barisella M, Renne SL, et al. Epithelioid angiosarcoma of the thyroid gland without distant metastases at diagnosis: report of six cases with a long follow-up. Virchows Arch 2016;469:223–32. 10.1007/s00428-016-1964-3 [DOI] [PubMed] [Google Scholar]
- 12. Binesh F, Akhavan A, Navabii H, et al. Primary angiosarcoma of the thyroid gland in an young Iranian woman. Case Rep Child Meml Hosp Chic 2011;2011:bcr0320114042 10.1136/bcr.03.2011.4042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Yilmazlar T, Kirdak T, Adim S, et al. A case of hemangiosarcoma in thyroid with severe anemia due to bone marrow metastasis. Endocr J 2005;52:57–9. 10.1507/endocrj.52.57 [DOI] [PubMed] [Google Scholar]
- 14. Yoon Moon S, Su Park H, Young Woo J, et al. Primary Thyroid Angiosarcoma with Tracheal Invasion. Intern Med 2016;55:1165–9. 10.2169/internalmedicine.55.5447 [DOI] [PubMed] [Google Scholar]
- 15. Nechifor-Boilă A, Decaussin-Petrucci M, Varga-Ilyés A, et al. Angioinvasion as a factor for predicting aggressive outcome in primary thyroid angiosarcoma: three case reports and literature review. Pol J Pathol 2018;69:53–61. 10.5114/pjp.2018.75337 [DOI] [PubMed] [Google Scholar]
- 16. Mills SE, Gaffey MJ, Watts JC, et al. Angiomatoid carcinoma and ’angiosarcoma' of the thyroid gland. A spectrum of endothelial differentiation. Am J Clin Pathol 1994;102:322–30. 10.1093/ajcp/102.3.322 [DOI] [PubMed] [Google Scholar]
- 17. Kuhn E, Ragazzi M, Ciarrocchi A, et al. Angiosarcoma and anaplastic carcinoma of the thyroid are two distinct entities: a morphologic, immunohistochemical, and genetic study. Mod Pathol 2019. 10.1038/s41379-018-0199-z [DOI] [PubMed] [Google Scholar]
- 18. Papotti M, Volante M, Negro F, et al. Thyroglobulin mRNA expression helps to distinguish anaplastic carcinoma from angiosarcoma of the thyroid. Virchows Arch 2000;437:635–42. 10.1007/s004280000308 [DOI] [PubMed] [Google Scholar]
- 19. Talbott I, Wakely PE. Undifferentiated (anaplastic) thyroid carcinoma: Practical immunohistochemistry and cytologic look-alikes . Semin Diagn Pathol 2015;32:305–10. 10.1053/j.semdp.2014.12.012 [DOI] [PubMed] [Google Scholar]
- 20. Rhomberg W, Boehler F, Eiter H, et al. Treatment options for malignant hemangioendotheliomas of the thyroid. Int J Radiat Oncol Biol Phys 2004;60:401–5. 10.1016/j.ijrobp.2004.03.023 [DOI] [PubMed] [Google Scholar]
- 21. Lamovec J, Zidar A, Zidanik B. Epithelioid angiosarcoma of the thyroid gland. Report of two cases. Arch Pathol Lab Med 1994;118:642–6. [PubMed] [Google Scholar]