Abstract
Type 1 diabetes risk can reliably be predicted by markers of autoimmunity, but approaches to prevent or modify the underlying disease process are needed. We posit this void fundamentally results from a limited understanding of immune-islet cell interactions within the pancreas and relevant immune organs, contributions of β-cells to their own demise, and epigenetic predispositions affecting both immune and islet cells. Because biopsy of the human pancreas and pancreatic lymph nodes carries risk and the pancreas begins to autodigest soon after death, detailed cellular and molecular phenotyping of the human type 1 diabetes pancreas is lacking, limiting our understanding of the mechanisms of β-cell loss. To address these challenges, the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases established the Human Pancreas Analysis Program (HPAP) to procure human type 1 diabetes pancreata for an extensive array of tissue-based, cellular, and epigenetic assays aimed at critical knowledge gaps in our understanding of the local immune attack and loss of β-cells. In this Methodology Review, we describe how HPAP is performing detailed islet and immune cell phenotyping and creating publicly available data sets with the goals of an improved understanding of type 1 diabetes and the development of more effective treatments to prevent or reverse the disease.
Introduction
Type 1 diabetes is widely considered to result from a T-cell–mediated killing of insulin-producing β-cells in the pancreatic islets of Langerhans, resulting in hyperglycemia, lifelong dependence on exogenous insulin, and an increased risk for severe long-term complications, even in those with appropriate disease management. The disorder can be diagnosed at any age, though onset is most commonly considered to occur in childhood, adolescence, or young adulthood (1). Recent work has underscored heterogeneity in the disease pathogenesis and presentation, suggesting that type 1 diabetes is likely a family of phenotypically similar diseases with overlapping but nonidentical pathophysiologies.
Until the late 2000s, most of our knowledge of pancreas pathology in human type 1 diabetes emanated from the study of pancreata collected at autopsy from a limited number of patients, obtained in the 1950–1980s, at the time of or just after symptomatic onset (2–8). For reasons of safety and efficacy, only rarely have patients with type 1 diabetes undergone a pancreatic biopsy. Indeed, the risk of surgical complications and the limited amount of tissue that can be recovered prevent a broader application of this approach to obtain such tissues for research (9–11). With the development of the JDRF-supported Network for Pancreatic Organ donors with Diabetes (nPOD) program in 2007, widespread studies (i.e., those beyond the few cases that were available at individual institutions) characterizing transplant-grade human pancreas, as opposed to autopsy, first became possible (12). From such studies conducted so far, typical pathological features in this disease affirm the long-held notions suggesting loss of pancreatic β-cells and the presence of an inflammatory lesion affecting the pancreatic islets (13). This lesion, termed insulitis, is characterized by the presence of immune cells accumulating around and inside pancreatic islets. More recent studies revealed that cells of the adaptive immune response, reactive to β-cell antigens, are present within the insulitic lesion (14–16) along with a series of local changes in pathology including hyperexpression of HLA class I molecules on β-cells (17), hyaluronan deposition within islets (18), and aberrations reflective of increased endoplasmic reticulum stress and mitochondrial dysfunction in β-cells (19–23). It should also be noted that immune cells are present in the entire pancreas, while more abundant in the endocrine compartment, and that the exocrine pancreas might also be affected in type 1 diabetes (24).
While the recent studies of human type 1 diabetes pancreatic tissue have provided new information, progress to understand the disorder’s pathogenesis and natural history at the molecular and cellular levels has been stymied by several major challenges, including 1) infrequent access to the pancreas from donors with recent-onset type 1 diabetes and lack of procedures for processing these organs for analysis with new and emerging technologies; 2) limited availability of type 1 diabetes pancreatic islets for functional, genomic, and gene expression studies; 3) lack of profiling of immune cells from type 1 diabetes islets and related immune organs such as peripancreatic lymph nodes; 4) lack of multimodal analyses with coregistration of complementary types of data from the same type 1 diabetes samples, coupled with presentation of the data in a publicly available, integrated database; and 5) the emerging consensus that the immunological and endocrine features of human type 1 diabetes have both similarities with and important differences from the nonobese diabetic (NOD) mouse model. To address these limitations and challenges, the National Institutes of Health (NIH), as part of the Human Islet Research Network (HIRN) (https://hirnetwork.org) initiative, created the Human Pancreas Analysis Program (HPAP) (https://hpap.pmacs.upenn.edu; RRID:SCR_016202). This effort is being led by investigators from the University of Pennsylvania (UPenn), Vanderbilt University, and the University of Florida. In this Methodology Review, we summarize the operational model and experimental approaches employed by HPAP investigators, as well as the program’s research objectives and current progress examining the pancreas throughout the natural history of type 1 diabetes.
The Pathogenesis and Natural History of Type 1 Diabetes
The emerging and consensus view on type 1 diabetes progression divides the disease into three stages (25). During stage 1, two or more autoantibodies against β-cell autoantigens (e.g., insulin, GAD, zinc transporter-8 [ZnT8], insulinoma antigen-2 [IA-2]) can be detected in the blood, indicating that the immune system has lost tolerance to β-cells, although the individual remains symptom free (i.e., normoglycemic). Stage 2 is marked by autoantibodies alongside a degree of dysglycemia (i.e., abnormal glucose tolerance tests), yet the individual does not require exogenous insulin and continues to be asymptomatic. Stage 3 is marked by a traditional diabetes diagnosis, which is often accompanied by clinical symptoms such as excessive thirst, frequent urination, weight loss, and fatigue and sometimes diabetic ketoacidosis. This recently developed staging model is important to the type 1 diabetes community, as it considers the disease process as ongoing even in those who are asymptomatic in terms of glycemic control (25).
The causes, triggers, and natural progression of type 1 diabetes remain only partially understood. The disease has a significant genetic component, with up to one-half of the genetic clustering explained by the high-risk DQ2 and DQ8 alleles within the HLA locus (26,27). However, many individuals with the high-risk alleles never develop type 1 diabetes; hence, based on genetic screening alone, we cannot conclusively identify those in the general population, or even in first-degree relatives, who will progress to symptomatic disease. In a broader sense, our inability to predict, with certainty, which at-risk individuals will progress to type 1 diabetes, or when progression will occur, remains a stark reminder that the type 1 diabetes research and clinical communities are hindered by a still rudimentary understanding of the processes that cause the disease. Without question, such efforts would benefit from an understanding of the human pancreas, with a focus on islet cell function, whole pancreas morphology, and the islet-immune interface, for those with or at increased risk for the disorder.
The inability to prevent or reverse disease progression has been, without question, one of the most disappointing outcomes of clinical type 1 diabetes research over multiple decades (28,29). The limited success of immunotherapy-based clinical trials suggests that our understanding of the immune cell attack in type 1 diabetes is still too limited for optimal treatment design and provides a major rationale for the efforts in HPAP. For example, we know very little about the immune cell compartment within the pancreas itself and in the peripancreatic lymph nodes (30), areas that likely play key roles in initiating and perpetuating autoimmunity as the site for T-cell activation and dendritic cell licensing.
HPAP Overview
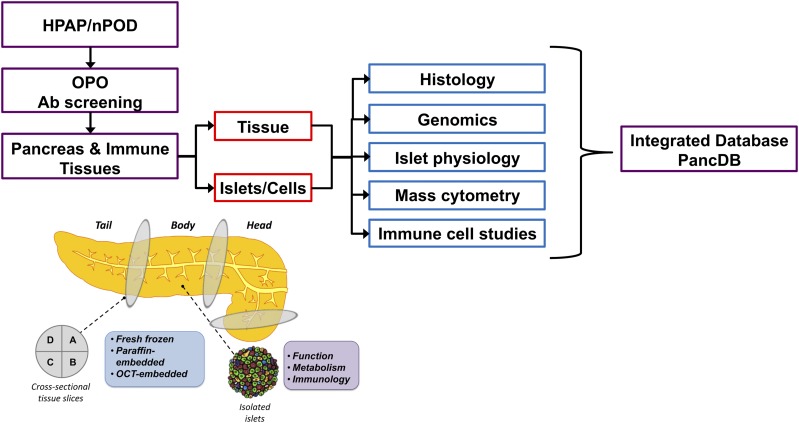
The past decade has seen a dramatic improvement in our ability to phenotype and molecularly profile human tissues with unprecedented resolution at the genomic, epigenomic, protein, and functional levels. Taking advantage of new and improved experimental approaches, the overarching goals of HPAP investigators are 1) to perform deep phenotyping of the endocrine pancreas and its interaction with the immune system to better understand the molecular events that precede and lead to the onset of type 1 diabetes and 2) to accumulate, analyze, and distribute high-value data sets to the diabetes research community through the PANC-DB database (Fig. 1). To this end, HPAP employs state-of-the-art technologies to perform comprehensive analyses of pancreas biology as it pertains to organ donors with type 1 diabetes, autoantibody-positive donors without diabetes, and control donors. Pancreas procurement and analyses take advantage of the expertise and extensive network of Organ Procurement Organizations (OPOs) and autoantibody screening centers established through the JDRF/The Lenona M. and Harry B. Helmsley Charitable Trust (HCT)–funded nPOD program (https://www.jdrfnpod.org). In contrast to nPOD, the major product provided by HPAP is not archived biomaterial subject to broad distribution, but rather, the delivery of extensive and high-quality molecular data sets to the diabetes research community in order to facilitate further discovery. Together, nPOD and HPAP are complementary programs that assist the diabetes community and afford the maximal opportunity for advancing knowledge about the pathogenesis of type 1 diabetes.
Figure 1.
HPAP workflow. HPAP, working with nPOD, identifies organ donors of interest (recent-onset type 1 diabetes, antibody-positive donors, and control subjects). For organ donors <30 years old without diabetes, the OPO, using HPAP/nPOD protocols and reagents, screens for presence of GADAs. If a suitable organ donor is identified, pancreas and immune tissues are shipped to UPenn for processing. The tissue and islets are then analyzed at Vanderbilt University and UPenn. All data are coregistered and integrated in a publicly accessible database (PANC-DB) (https://hpap.pmacs.upenn.edu). Ab, antibody.
Organ Procurement and Tissue Processing by HPAP
HPAP, working collaboratively with nPOD, has established a network to identify organ donors relevant to understanding the pathogenesis of type 1 diabetes. This network includes relationships with the National Disease Research Interchange, the International Institute for the Advancement of Medicine, and all 59 OPOs located throughout the U.S. to identify donors with recent-onset type 1 diabetes, those with type 1 diabetes–associated antibodies, and appropriate controls. HPAP coordinates with nPOD’s established infrastructure to screen for type 1 diabetes–associated autoantibodies against GAD65 (GADA) and IA-2 (IA-2A) from non–type 1 diabetes donor serum in order to identify donors with pre–type 1 diabetes who, had they survived, would have been at high risk of progressing to type 1 diabetes. Such analyses (i.e., autoantibody screening) are also performed on all donors ascribed as having type 1 diabetes to better understand disease heterogeneity and to contribute to diagnosis confirmation. Serology screening laboratories implement rapid single-use combined GADA/IA-2A ELISA kits (Kronus, Inc.) as previously described (31), and the results are sent in real time to both the OPO and the on-call team at the University of Florida, available 24 hours per day, 7 days per week, 365 days per year. The University of Florida team determines whether to accept the donor tissues, with tissues allocated to nPOD or HPAP, based on the inclusion criteria and logistical considerations for each program. Assays, procedures, and data sets generated through each program are shown in Table 1.
Table 1.
Donor groups accepted, standard assays and procedures performed, and data sets generated through the NIH-HIRN–supported HPAP and JDRF/HCT-supported nPOD programs
| Donor groups, procedures, assays, and data sets | HPAP | nPOD* |
|---|---|---|
| Donor groups (pancreas) | ||
| Control (prenatal) | ||
| Control (neonate–10 years of age) | ✓ | ✓ |
| Control (10–30 years of age) | ✓ | ✓ |
| T1D (0–7 years’ duration, <40 years of age) | ✓ | ✓ |
| T1D (7–10 years’ duration, <40 years of age) | ✓ | |
| AAb+ pre-T1D (≤30 years of age) | ✓ | ✓ |
| T2D | ✓ | ✓ |
| Pancreas or SPK transplant recipient | ✓ | |
| Islet transplant recipient | ✓ | |
| Other autoimmunity | ✓ | ✓ |
| Other | ✓ | |
| Additional tissues collected | ||
| Pancreatic draining lymph node | ✓ | ✓ |
| Mesenteric draining lymph node | ✓ | |
| Blood | ✓ | ✓ |
| Spleen | ✓ | ✓ |
| Thymus | ✓ | |
| Intestine | ✓ | ✓ |
| Bone marrow | ✓ | |
| Donor clinical and demographic metadata | ✓ | ✓ |
| Autoantibody screening (rapid ELISA) | ||
| GADA | ✓ | ✓ |
| IA-2A | ✓ | ✓ |
| Autoantibody confirmation (RIA) | ||
| GADA | ✓ | ✓ |
| IA-2A | ✓ | ✓ |
| IAA | ✓ | ✓ |
| ZnT8A | ✓ | ✓ |
| DNA isolation and storage | ✓ | ✓ |
| Genotyping | ||
| High-resolution HLA | ✓ | ✓ |
| UFDIchip | ✓ | |
| Histology | ||
| H-E | ✓ | ✓ |
| IHC: Ki-67/insulin/glucagon | ✓ | ✓ |
| IHC: CD3/insulin/glucagon | ✓ | |
| IHC: CD45/insulin/glucagon | ✓ | |
| IHC: insulin/glucagon/somatostatin/pancreatic polypeptide | ✓ | |
| IHC: digestive enzymes | ✓ | |
| Leukocyte isolation and characterization by FACS and scRNA-Seq | ||
| Intraislet | ✓ | ✓ |
| Spleen | ✓ | |
| Thymus | ✓ | |
| Mesenteric draining lymph nodes | ✓ | |
| Pancreatic draining lymph nodes | ✓ | |
| Treg suppression assays | ✓ | |
| Islet isolation and characterization | ||
| FACS | ✓ | |
| CyTOF | ✓ | |
| Perifusion for insulin secretion | ✓ | |
| Perifusion for glucagon secretion | ✓ | |
| Calcium imaging | ✓ | |
| Patch-clamp analysis | ✓ | |
| Oxygen consumption | ✓ | |
| RNA-Seq | ✓ | |
| ATAC-Seq | ✓ | |
| Bisulfite sequencing for DNA methylation patterns | ✓ | |
| RNA isolation and storage | ||
| Isolated islets | ✓ | |
| Acini | ||
| Unspecified region of tissue | ||
| IMC | ✓ | |
| Slices | ✓ | |
| Perifusion for insulin secretion | ✓ | |
| Perifusion for glucagon secretion | ✓ |
AAb+, autoantibody positive; H-E, hematoxylin-eosin; IAA, insulin autoantibody; RIA, radioimmunoassay; scRNA-Seq, single cell RNA-Seq; SPK, simultaneous pancreas kidney; T1D, type 1 diabetes; T2D, type 2 diabetes; UFDIchip, custom genotyping array with an imputation grid of ∼790,000 single nucleotide polymorphisms providing genome-wide coverage as well as ∼160,000 single nucleotide polymorphisms providing dense coverage in genomic regions implicated in risk for autoimmune disorders; ZnT8A, ZnT8 autoantibody.
*Assays listed here are standardly performed on all tissues acquired by the nPOD Tissue Processing and Pathology Core. For a complete list of all assays and studies being carried out by nPOD investigators worldwide, please see the nPOD website (https://www.jdrfnpod.org/publications/current-npod-projects/).
The HPAP inclusion criteria for deceased organ donors are diagnosis of type 1 diabetes, latent autoimmune diabetes in adults, “other” immune-mediated conditions (e.g., hypothyroidism, etc.), and autoantibody positivity; control donors without diabetes are also included. Both male and female donors are accepted across all ages and stages of diabetes with priority placed on tissues received from young (i.e., childhood or early adult) donors. As of this writing, 36 organs have been processed, including one organ donor with fulminant new-onset (1 day duration) type 1 diabetes and four donors with type 2 diabetes to differentiate immune-mediated disease from metabolic effects on the parameters measured. All organ procurement is coordinated through the transplant surgical team at UPenn where all tissues (i.e., pancreas, spleen, blood, duodenum, and peripancreatic lymph nodes) are processed for histological and molecular analyses and residual tissues are biobanked. Board-certified pathologists review the tissues under blinded conditions and document clinical findings for all cases. All islet isolation takes place at the UPenn Current Good Manufacturing Practice facility to limit product variability.
Donor Metadata
Detailed de-identified demographic and clinical metadata including donor age, sex, race, BMI, ABO blood group, Rh blood type, cause of death, cardiac arrest downtime, cardiopulmonary resuscitation time, admission and cross clamp date and time, cold ischemia time, intraoperative time for pancreas recovery, pancreas weight, serum C-peptide level, HbA1c, infectious disease serology, prior clinical diagnoses (e.g., diabetes, cancer, coronary artery disease, other autoimmune disease), clinical interventions during terminal hospital visit, family medical history, and diabetes duration are made available on the HPAP web portal. Autoantibody status is confirmed by radioimmunoassay performed at the University of Colorado Denver Barbara Davis Center for Childhood Diabetes. Finally, HLA genotype is determined by RT-PCR by the OPO for transplant histocompatibility and later confirmed by high-resolution next-generation sequencing. Excepting rare occasions when the pancreas is not of sufficient quality, islets are isolated, and data including pancreas weight, gross pathology, islet yield and viability, and islet distribution are also reported.
Histopathology
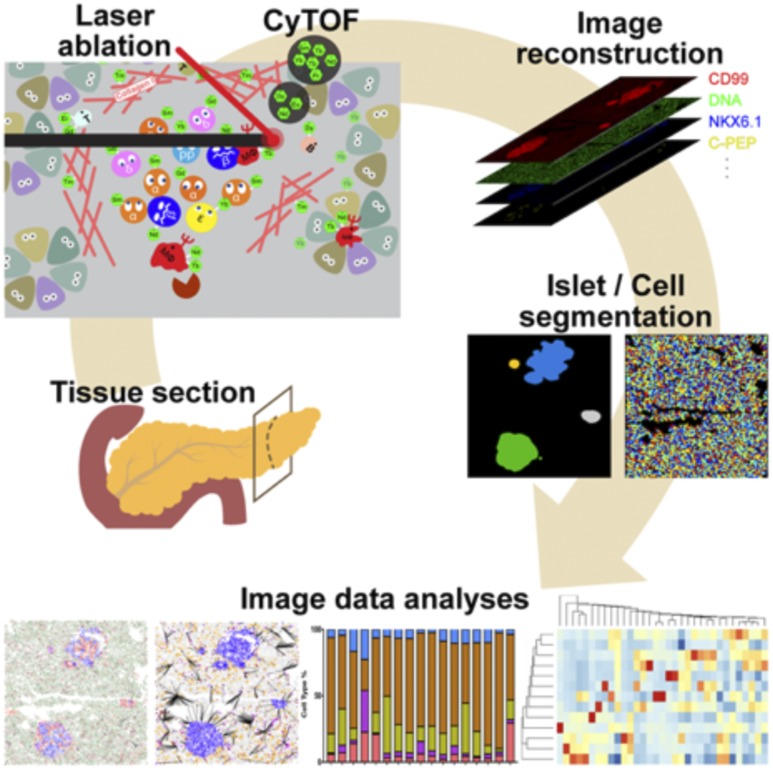
The HPAP group produces a variety of analyses detailing the microscopic structure of the pancreas through histological sectioning and staining. Tissue samples from the head, body, and tail regions of the pancreas are processed through multiple fixation modalities (i.e., formalin fixed paraffin embedded, optimal cutting temperature compound embedded, and fresh frozen [Fig. 1]) to enable the largest number of possible assays. Tissues are routinely stained by hematoxylin-eosin, and immunohistochemistry (IHC) with whole-slide scanned images uploaded to the PANC-DB database. The HPAP group has placed considerable effort on assay development and refinement. For example, HPAP investigators used a validated antibody panel for the simultaneous in situ detection and quantification of 33 disease-relevant protein targets by imaging mass cytometry (IMC) (Fig. 2 [32]). Using this technology, HPAP can visualize alterations in endocrine cell composition, islet architecture, and immune cell representation simultaneously in a single tissue section. Importantly, we predict that with future technological advances, the number of disease-relevant protein targets subject to simultaneous analysis will increase even further.
Figure 2.
IMC of the human pancreas developed by HPAP. Pancreatic tissue sections are staining with 33 antibodies conjugated to heavy metal isotopes. The washed slide is then subjected to laser ablation, and the amount of each isotope per tissue voxel is recorded. The data can be used for image reconstruction or for quantitative analysis of marker protein expression in each cell in the section. Reprinted with permission from Wang et al. (32).
Islet Physiology and Islet Composition
While it was originally thought that type 1 diabetes leads to a complete loss of all pancreatic β-cells, the availability of improved assays for the detection of C-peptide, along with detailed immunostaining studies of pancreatic specimens, demonstrated that even patients with long-standing type 1 diabetes can have residual—or possibly regenerated—β-cells (33,34). In addition, it was recently reported that even in patients with long-standing (>3 years’ duration) type 1 diabetes, proinsulin is detectable in ∼90% of the individuals tested (35). Remarkably, in research supported by HPAP, it was recently shown that when expressed as insulin secretion per insulin content, glucose responsiveness can be exhibited in residual β-cells, albeit with a reduced first-phase amplitude (36). In contrast, pancreatic α-cells, not subject to antigen-specific immune cell attack, were functionally impaired in islets obtained from donors with type 1 diabetes, demonstrating a decreased glucagon secretory response when expressed per glucagon content. These changes in α-cell function were accompanied by an altered expression pattern for key α-cell transcription factors. These findings indicate that endocrine cell function and gene expression may change with type 1 diabetes; however, it remains to be established whether any of these alterations precede the onset of hyperglycemia or whether they are an indirect consequence of the immune cell attack.
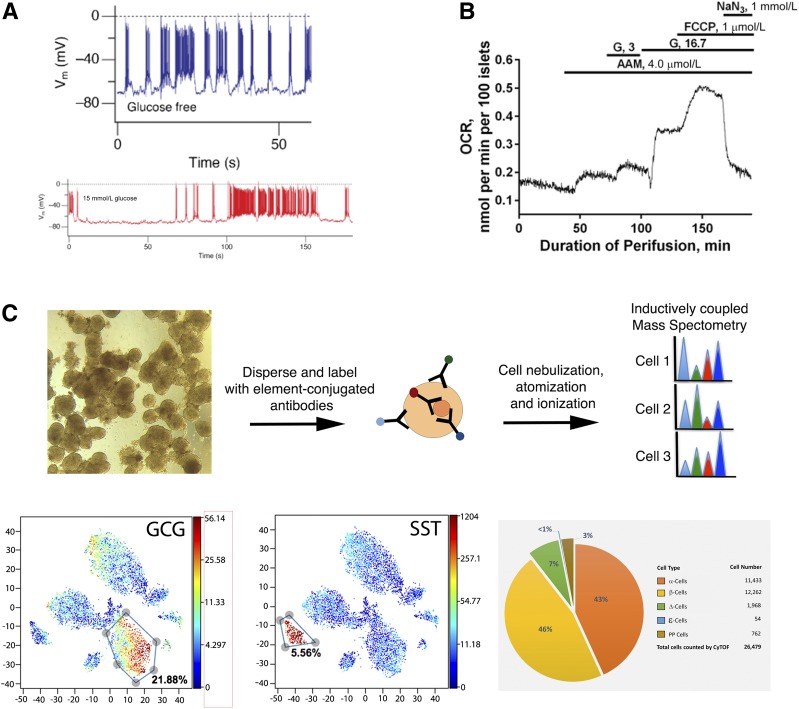
HPAP is determining islet performance in a comprehensive fashion as part of its mission. Routine assays include islet perifusion studies for the determination of glucagon and insulin secretion by the Human Islet Phenotyping Program of the NIH-supported Integrated Islet Distribution Program (IIDP) (https://iidp.coh.org/) so that the data can be easily compared with the large set of islets procured through IIDP. In addition, islet function and health are assessed via calcium imaging using fluorescent calcium chelators, patch-clamp analysis of electrophysiologic properties, and oxygen consumption (Fig. 3A and B).
Figure 3.
Examples of islet assays performed by HPAP. A: Patch clamp electrophysiology recording of a pancreatic β-cell. The top trace shows firing in the absence and the bottom trace (red) in the presence of 15 mmol/L glucose. Note the increased rate of depolarization from 100 to 150 s during the incubation with glucose. B: Oxygen consumption rate (OCR) of human islets. Note the large stimulation by 16.7 mmol/L glucose (G). C: Flow mass cytometry for the precise quantification of islet composition. Top, general principle. Bottom, tSNE plots of islet cell clusters with α-cells (glucagon [GCG]) and δ-cells (somatostatin [SST]) indicated. Pie chart from the HPAP website illustrating endocrine cell proportions.
Key to the interpretation of hormone secretion data is a detailed understanding of islet composition, given that the percentage of the various endocrine cell types varies widely among individuals and across islets from a single pancreas (37). For this purpose, HPAP is employing the recently developed Time of Flight Mass Cytometry (CyTOF) assay for detailed and multiplexed determination of islet composition (Fig. 3C). Gradient-purified islets are digested to yield single cell suspensions, permeabilized and stained for 24 proteins expressed by islet cells (38). Thus, in addition to the precise determination of the endocrine percentages for all five endocrine cell types (α-, β-, δ-, PP, and ε-cells, producing glucagon, insulin, somatostatin, pancreatic polypeptide, and ghrelin, respectively), levels of key transcription factors and rates of cell proliferation are also assessed simultaneously.
Molecular Phenotyping of the Pancreatic Islet
Working from the hypothesis that the interplay between islet cells and the immune system is critical in the early stages of type 1 diabetes pathogenesis, a HPAP goal is to develop a detailed understanding of the molecular changes that occur in individual islet cells over the natural history of type 1 diabetes. Many prior studies employed transcriptome profiling of whole islets (39,40), but because of the aforementioned individual variability in endocrine cell composition, whole islet studies are of poor resolution and limited power. Therefore, HPAP is employing assays based on prior FACS of α- and β-cells using a panel of cell surface antibodies originally developed by the Grompe laboratory at the Oregon Stem Cell Center (41) and recently improved through the addition of an antibody targeting adult β-cells (42). There is a dizzying array of technologies for the determination of steady-state mRNA levels (i.e., transcriptome), native transcripts, open chromatin states, and dozens of histone marks available today. Because of sample limitations, HPAP has opted for the determination of mRNA levels by RNA sequencing (RNA-Seq), the determination of open chromatin status by ATAC-Seq (assay for transposase accessible chromatin with high-throughput sequencing), and the determination of the DNA methylome using bisulfite sequencing because of their information content and the small amount of tissue required. In addition, HPAP is capturing individual variations in gene expression among the islet cell types and islet resident immune cells using single cell RNA-Seq (43–51). RNA and DNA samples are also banked by HPAP for their use in new assays that will be developed in the future.
Analysis of the Systemic and Local Immune System
A key feature of HPAP is the ability to perform immune cell phenotyping that is not restricted to peripheral blood samples but, rather, extends to peripancreatic lymph nodes and the entirety of pancreatic tissue. In addition to blood, spleen, and duodenum, peripancreatic lymph nodes are carefully dissected and analyzed separately from the head, body, and tail regions of the pancreas. T-cell profiles in these efforts include flow cytometric analyses of phenotype, activation state, cytolytic properties, and exhaustion profiles of memory and type 1 diabetes antigen-specific T cells from human pancreatic islet infiltrates, draining lymph nodes, spleen, and peripheral blood. Regulatory T cells (Tregs) are FACS isolated and their function is assessed using in vitro suppression assays to determine the relative suppressive potency of Tregs from individuals with type 1 diabetes as well as the ability of effector T cells from such persons to avoid Treg-mediated suppression.
Data Storage and Availability
Critical to the mission of HPAP is the distribution of data to interested diabetes researchers worldwide. After quality control assessment, all raw and processed data generated through HPAP are directly deposited—prior to publication—in a publicly accessible database (https://hpap.pmacs.upenn.edu). Depending on the assay type, the time from organ procurement to posting of the data on the PANC-DB website can vary from 2 weeks (donor information, islet isolation results, and islet perifusion data) to several months (RNA-Seq results). For optimization of the impact of HPAP and to accelerate discovery, access to these comprehensive data sets requires a minimal login, with only user registration and a commitment to source citing required. Importantly, all data sets are anonymized and compliant with HIPAA regulations. While the ability to download all HPAP data sets is the primary goal of the website, HPAP is continuously developing new data viewers to make the information more readily accessible and useful. For instance, with each deceased organ donor, a detailed “Donor Summary” section presents key patient data as well as information on organ processing. Likewise, an “Islet Isolation” page contains salient features from pancreas weight to islet purity. An image viewer displays preselected antibody channel combinations from IMC as multicolor microscopy images. Thus, users can view the endocrine markers, extracellular matrix proteins, or selected immune cell markers from the head, body, or tail of each pancreas with ease. Of course, after data download, every user can create any number of combinations from the >30 IMC channels collected. The islet composition data, determined with precision using CyTOF with a panel of 24 antibodies (38), are reported as pie charts for each donor (Fig. 3A and B), while islet function can be gleaned from glucagon and insulin secretion studies. Additional data viewers, in particular for the epigenomic and gene expression data, are under development.
Conclusions and Outlook
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)-funded HPAP was established in 2016 to perform in-depth, comprehensive phenotyping of the human pancreas and lymphoid system from individuals with recent-onset type 1 diabetes, autoantibody-positive persons without diabetes considered to have pre–type 1 diabetes, and age-matched control donors. During the first two and a half years in operation, HPAP has collected 36 donor samples of interest for the study of type 1 and prediabetes and has applied newly developed state-of-the-art assays for the determination of pancreas histopathology, islet function, and transcriptome and epigenome studies, as well as detailed immune phenotyping. These valuable data sets are available immediately after curation through the online PANC-DB database for data download and analysis. HPAP investigators are eager to collaborate with the type 1 diabetes research community and welcome suggestions for enhancing HPAP activities, including the functionality of our database and website. Among the accomplishments of the new program thus far is the adoption of IMC for 33 antibodies simultaneously as a routine screening assay and the observation that pancreas-resident CD8+ T cells are highly proliferative, and thus activated, in the type 1 diabetic pancreas regardless of duration of disease (32). We expect that the data produced and insights gained through HPAP’s efforts will help advance the common goals of diabetes researchers worldwide to improve our understanding of the etiology and pathogenesis of type 1 diabetes and ultimately prevent as well as reverse the disease.
Article Information
Funding. Efforts related to the contents of this methodology review were supported by resources and/or funding provided by the NIDDK-supported HIRN (RRID:SCR_014393 [https://hirnetwork.org]) (UC4DK112217 and UC4DK112232).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. K.H.K. conceived of the manuscript and wrote the manuscript. A.C.P., A.N., and M.A.A. contributed to discussion and reviewed and edited the manuscript. K.H.K. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
References
- 1.Katsarou A, Gudbjörnsdottir S, Rawshani A, et al. Type 1 diabetes mellitus. Nat Rev Dis Primers 2017;3:17016. [DOI] [PubMed] [Google Scholar]
- 2.Gepts W. Islet morphology in type I diabetes. Behring Inst Mitt 1984;75:39–41 [PubMed] [Google Scholar]
- 3.Gepts W, Lecompte PM. The pancreatic islets in diabetes. Am J Med 1981;70:105–115 [DOI] [PubMed] [Google Scholar]
- 4.LeCompte PM, Gepts W. The Pathology of Juvenile Diabetes. Volk BWWKF, Ed. New York, Plenum Press, 1977 [Google Scholar]
- 5.Gepts W. Islet changes suggesting a possible immune aetiology of human diabetes mellitus. Acta Endocrinol Suppl (Copenh) 1976;205:95–106 [PubMed] [Google Scholar]
- 6.Gepts W. Pathologic anatomy of the pancreas in juvenile diabetes mellitus. Diabetes 1965;14:619–633 [DOI] [PubMed] [Google Scholar]
- 7.Foulis AK. The pathology of islets in diabetes. Eye (Lond) 1993;7:197–201 [DOI] [PubMed] [Google Scholar]
- 8.Foulis AK, Liddle CN, Farquharson MA, Richmond JA, Weir RS. The histopathology of the pancreas in type 1 (insulin-dependent) diabetes mellitus: a 25-year review of deaths in patients under 20 years of age in the United Kingdom. Diabetologia 1986;29:267–274 [DOI] [PubMed] [Google Scholar]
- 9.Atkinson MA. Pancreatic biopsies in type 1 diabetes: revisiting the myth of Pandora’s box. Diabetologia 2014;57:656–659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Krogvold L, Edwin B, Buanes T, et al. Pancreatic biopsy by minimal tail resection in live adult patients at the onset of type 1 diabetes: experiences from the DiViD study. Diabetologia 2014;57:841–843 [DOI] [PubMed] [Google Scholar]
- 11.Hanafusa T, Miyazaki A, Miyagawa J, et al. Examination of islets in the pancreas biopsy specimens from newly diagnosed type 1 (insulin-dependent) diabetic patients. Diabetologia 1990;33:105–111 [DOI] [PubMed] [Google Scholar]
- 12.Campbell-Thompson M. Organ donor specimens: What can they tell us about type 1 diabetes? Pediatr Diabetes 2015;16:320–330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Campbell-Thompson M, Fu A, Kaddis JS, et al. Insulitis and β-cell mass in the natural history of type 1 diabetes. Diabetes 2016;65:719–731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Babon JA, DeNicola ME, Blodgett DM, et al. Analysis of self-antigen specificity of islet-infiltrating T cells from human donors with type 1 diabetes. Nat Med 2016;22:1482–1487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Michels AW, Landry LG, McDaniel KA, et al. Islet-derived CD4 T cells targeting proinsulin in human autoimmune diabetes. Diabetes 2017;66:722–734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Coppieters KT, Dotta F, Amirian N, et al. Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. J Exp Med 2012;209:51–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Richardson SJ, Rodriguez-Calvo T, Gerling IC, et al. Islet cell hyperexpression of HLA class I antigens: a defining feature in type 1 diabetes. Diabetologia 2016;59:2448–2458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bogdani M, Johnson PY, Potter-Perigo S, et al. Hyaluronan and hyaluronan-binding proteins accumulate in both human type 1 diabetic islets and lymphoid tissues and associate with inflammatory cells in insulitis. Diabetes 2014;63:2727–2743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Phelps EA, Cianciaruso C, Michael IP, et al. Aberrant accumulation of the diabetes autoantigen GAD65 in Golgi membranes in conditions of ER stress and autoimmunity. Diabetes 2016;65:2686–2699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Delong T, Wiles TA, Baker RL, et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 2016;351:711–714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kracht MJ, van Lummel M, Nikolic T, et al. Autoimmunity against a defective ribosomal insulin gene product in type 1 diabetes. Nat Med 2017;23:501–507 [DOI] [PubMed] [Google Scholar]
- 22.Chen J, Chernatynskaya AV, Li JW, et al. T cells display mitochondria hyperpolarization in human type 1 diabetes. Sci Rep 2017;7:10835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Marhfour I, Lopez XM, Lefkaditis D, et al. Expression of endoplasmic reticulum stress markers in the islets of patients with type 1 diabetes. Diabetologia 2012;55:2417–2420 [DOI] [PubMed] [Google Scholar]
- 24.Rodriguez-Calvo T, Ekwall O, Amirian N, Zapardiel-Gonzalo J, von Herrath MG. Increased immune cell infiltration of the exocrine pancreas: a possible contribution to the pathogenesis of type 1 diabetes. Diabetes 2014;63:3880–389063:3880–3890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Insel RA, Dunne JL, Atkinson MA, et al. Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 2015;38:1964–1974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Aly TA, Ide A, Humphrey K, et al. Genetic prediction of autoimmunity: initial oligogenic prediction of anti-islet autoimmunity amongst DR3/DR4-DQ8 relatives of patients with type 1A diabetes. J Autoimmun 2005;25(Suppl.):40–45 [DOI] [PubMed] [Google Scholar]
- 27.Aly TA, Ide A, Jahromi MM, et al. Extreme genetic risk for type 1A diabetes. Proc Natl Acad Sci U S A 2006;103:14074–14079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Atkinson MA, Roep BO, Posgai A, Wheeler DCS, Peakman M. The challenge of modulating β-cell autoimmunity in type 1 diabetes. Lancet Diabetes Endocrinol 2019;7:52–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Roep BO, Wheeler DCS, Peakman M. Antigen-based immune modulation therapy for type 1 diabetes: the era of precision medicine. Lancet Diabetes Endocrinol 2019;7:65–74 [DOI] [PubMed] [Google Scholar]
- 30.Seay HR, Yusko E, Rothweiler SJ, et al. Tissue distribution and clonal diversity of the T and B cell repertoire in type 1 diabetes. JCI Insight 2016;1:e88242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wasserfall CH, Atkinson MA. Autoantibody markers for the diagnosis and prediction of type 1 diabetes. Autoimmun Rev 2006;5:424–428 [DOI] [PubMed] [Google Scholar]
- 32.Wang YJ, Traum D, Schug J, et al.; HPAP Consortium . Multiplexed in situ imaging mass cytometry analysis of the human endocrine pancreas and immune system in type 1 diabetes. Cell Metab 2019;29:769–783.e4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Keenan HA, Sun JK, Levine J, et al. Residual insulin production and pancreatic β-cell turnover after 50 years of diabetes: Joslin Medalist Study. Diabetes 2010;59:2846–2853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oram RA, Jones AG, Besser RE, et al. The majority of patients with long-duration type 1 diabetes are insulin microsecretors and have functioning beta cells. Diabetologia 2014;57:187–191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sims EK, Bahnson HT, Nyalwidhe J, et al.; T1D Exchange Residual C-peptide Study Group . Proinsulin secretion is a persistent feature of type 1 diabetes. Diabetes Care 2019;42:258–264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Brissova M, Haliyur R, Saunders D, et al. α cell function and gene expression are compromised in type 1 diabetes. Cell Reports 2018;22:2667–2676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Brissova M, Fowler MJ, Nicholson WE, et al. Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy. J Histochem Cytochem 2005;53:1087–1097 [DOI] [PubMed] [Google Scholar]
- 38.Wang YJ, Golson ML, Schug J, et al. Single-cell mass cytometry analysis of the human endocrine pancreas. Cell Metab 2016;24:616–626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jackson A, McWilliams C, Kaizer E, et al. Gene expression profiling of human pancreatic islets undergoing a simulated process of instant blood-mediated inflammatory reaction. Transplant Proc 2008;40:430–432 [DOI] [PubMed] [Google Scholar]
- 40.Taneera J, Fadista J, Ahlqvist E, et al. Expression profiling of cell cycle genes in human pancreatic islets with and without type 2 diabetes. Mol Cell Endocrinol 2013;375:35–42 [DOI] [PubMed] [Google Scholar]
- 41.Dorrell C, Abraham SL, Lanxon-Cookson KM, Canaday PS, Streeter PR, Grompe M. Isolation of major pancreatic cell types and long-term culture-initiating cells using novel human surface markers. Stem Cell Res (Amst) 2008;1:183–194 [DOI] [PubMed] [Google Scholar]
- 42.Saunders DC, Brissova M, Phillips N, et al. Ectonucleoside triphosphate diphosphohydrolase-3 antibody targets adult human pancreatic β cells for in vitro and in vivo analysis. Cell Metab 2019;29:745–754.e4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Baron M, Veres A, Wolock SL, et al. A single-cell transcriptomic map of the human and mouse pancreas reveals inter- and intra-cell population structure. Cell Syst 2016;3:346–360.e4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Carrano AC, Mulas F, Zeng C, Sander M. Interrogating islets in health and disease with single-cell technologies. Mol Metab 2017;6:991–1001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li J, Klughammer J, Farlik M, et al. Single-cell transcriptomes reveal characteristic features of human pancreatic islet cell types. EMBO Rep 2016;17:178–187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Muraro MJ, Dharmadhikari G, Grün D, et al. A single-cell transcriptome atlas of the human pancreas. Cell Syst 2016;3:385–394.e3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Segerstolpe Å, Palasantza A, Eliasson P, et al. Single-cell transcriptome profiling of human pancreatic islets in health and type 2 diabetes. Cell Metab 2016;24:593–607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang YJ, Schug J, Won K-J, et al. Single-cell transcriptomics of the human endocrine pancreas. Diabetes 2016;65:3028–3038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xin Y, Kim J, Okamoto H, et al. RNA sequencing of single human islet cells reveals type 2 diabetes genes. Cell Metab 2016;24:608–615 [DOI] [PubMed] [Google Scholar]
- 50.Zeng C, Mulas F, Sui Y, et al. Pseudotemporal ordering of single cells reveals metabolic control of postnatal β cell proliferation. Cell Metab 2017;25:1160–1175.e11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang YJ, Kaestner KH. Single-cell RNA-Seq of the pancreatic islets--a promise not yet fulfilled? Cell Metab 2019;29:539–544 [DOI] [PMC free article] [PubMed] [Google Scholar]