Abstract
Background
We aimed to investigate the role of PDCD4-mediated Akt signaling pathway in vascular endothelial cell injury caused by ischemia-reperfusion in the lower extremities.
Material/Methods
Ten rats were used as control, while 50 rats were used for creating disease models and were assigned to 5 groups: model group (no injection), NC group (injected with vectors containing PDCD negative control sequence), sh-PDCD4 group (injected with vectors containing sh-PDCD4 sequence), IGF-1 group (injected with IGF-1), and sh-PDCD4+IGF-1 group (injected with IGF-1 and vectors containing sh-PDCD4 sequence).
Results
Compared with the control group, the expression levels of PDCD4 mRNA and protein, as well as levels of circulating endothelial cells, von Willebrand factor, thrombomodulin, and malondialdehyde, increased in the other 5 groups, while the mRNA and protein expression levels of Akt and eNOS, the protein expression levels of p-Akt and p-eNOS, and superoxide dismutase content decreased in these groups (all P<0.05). Compared with the model group, the sh-PDCD4 and sh-PDCD4+1GF-1 groups had lower mRNA and protein expressions of PDCD4 (all P<0.05), whereas the IGF-1 group had similar levels (all P>0.05). These 3 groups had lower levels of circulating endothelial cells, von Willebrand factor, thrombomodulin, and malondialdehyde, and higher mRNA and protein expressions of Akt and eNOS, protein expressions of p-Akt and p-eNOS, and superoxide dismutase content (all P<0.05). The NC group did not differ from the model group (all P>0.05).
Conclusions
PDCD4 gene silencing can activate the Akt signaling pathway and attenuate vascular endothelial cell injury caused by ischemia-reperfusion in the lower extremities in rats.
MeSH Keywords: Apoptosis Regulatory Proteins, Endothelial Cells, Gene Silencing
Background
Ischemia-reperfusion injury (IRI) in the lower extremities refers to a series of physiological and biochemical changes that occur when blood supply returns to tissues after a period of vessel occlusion in the lower limbs, and it is a common pathological change in clinical practice. In some severe cases, lower-extremity IRI can even cause systemic complications such as organ dysfunctions [1,2]. It has been reported that the functionality of vascular endothelial cells (VEC) is associated with the degree of lower-extremity IRI, and the tissue microcirculation can be affected by abnormalities of vascular endothelium and changes in intima permeability, thereby further aggravating the injury [3,4].
Programmed cell death 4 (PDCD4) gene has recently been discovered to be closely related to cell apoptosis. Studies have shown that this gene can regulate programmed death of cells, as well as transcription and translation of proteins, which can influence the growth of cancer cells [5,6]. Moreover, in recent years, some studies have found that PDCD4 plays a crucial role in cardiovascular disease, as it can affect VEC homeostasis [7]. A myocarditis study by Su et al. reported that suppression of PDCD4 expression helps attenuate the progression of myocarditis, indicating that PDCD4 can serve as an essential target in the treatment of this disease [8]. However, there have been few studies on the correlation between PDCD4 expression and lower-extremity IR.
It has been documented that the Akt signaling pathway is involved in cardiocyte apoptosis and myocardial IRI [9]. In their research on myocardial IR, Pei et al. found that activation of the Akt signaling pathway can downregulate the protein expression of Bax in the mouse model of myocardial IR, and this downregulation can noticeably protect the tissue against IRI [10]. However, it is still unclear whether the upstream gene can regulate the role of the Akt signaling pathway in lower-extremity IR. Therefore, in our present study, we created a mouse model of lower-extremity IRI and investigated the relationships among PDCD4 gene, Akt signaling pathway, and VEC injury caused by lower-extremity IR.
Material and Methods
The flow diagram of the study is displayed in Figure 1.
Figure 1.

Flow diagram of the study. PDCD4 – programmed cell death 4; IGF-1 – insulin-like growth factor 1; CEC – circulating endothelial cell; vWF – von Willebrand factor; SOD – superoxide dismutase; MDA – malondialdehyde.
Generating a mouse model of lower-extremity IR
Sixty healthy adult male Sprague-Dawley rats (Shanghai Laboratory Animal Research Center, China) were used for this study. The rats were fasted for 12 h prior to the operation and were given ad libitum access to water. Fifty rats were picked randomly and received intraperitoneal injections of pentobarbital sodium for anesthesia (dosage 50 mg/kg, Haling Biotechnology, China). Then, the rats were fixed on an operating table (Shengruida, China) in supine position with the temperature of the table set at 37°C. An abdominal incision was made and the peritoneum was cut. The aorta (about 10 mm) was exposed and clamped using an atraumatic Bulldog vascular clamp. Next, the abdominal wall was closed with a single-layer suture. No blood flow signal was detected by Laser Doppler Flowmetry (LDF). After 2 h, the laparotomy was performed again, and the clamp was removed to restore the aorta blood flow. The blood flow signal could then be detected by LDF, and the mouse model of lower-extremity IR was thus created [11]. The same procedures, except for the clamping of abdominal aorta, were performed on the remaining 10 rats. The study was approved by the Animal Ethics Committee of the Sir Run Run Shaw Hospital, Zhejiang University School of Medicine (dated 2 April 2014).
Grouping
The 10 rats in which the models were not established as mentioned above were assigned to a control group (untreated), while the other 50 rats were randomized into the following 5 groups of 10 rats each: the model group (no injection of any sequence), the NC group (subjects received injections of vectors containing PDCD negative control sequence through tail vein), the sh-PDCD4 group (subjects received injections of vectors containing sh-PDCD4 sequence through tail vein), the insulin-like growth factor 1 (IGF-1) group (subjects received injections of IGF-1, an activator of Akt signaling pathway, through tail vein), and the sh-PDCD4+IGF-1 group (subjects received injections of IGF-1 and vectors containing sh-PDCD4 sequence through tail vein). All sequences used were synthesized by RiboBio, Guangzhou, China. IGF-1, the activator of the Akt signaling pathway, was purchased from R&D Systems, USA. All rats were fed with a normal diet and had ad libitum access to water. One week after the injections, rats were sacrificed, and blood from the aorta and the vascular endothelium tissues was collected.
Measurement of circulating endothelial cell (CEC) count in each group
CECs were isolated using Hladovec’s method. The serum sample (4.5 mL) containing anticoagulant was centrifuged at 1500 rpm for 20 min. The supernatant was then discarded, CEC precipitation was obtained, and we added 0.1 mL 0.9% NaCl solution. A sample of the solution was then placed into the blood counting chamber, and the CECs were counted under a light microscope.
Determination of thrombomodulin (TM) and von Willebrand factor (vWF) in each group
According to the instruction manual of the vWF kit (ab208980, Abcam, UK) and TM kit (ab209880, Abcam, UK), vWF and TM values were measured as follows: 1) 8 standard wells were used in this assay, and we added 00 μL dilutions to 7 of them, while another well was used as a control for creating a standard curve; 2) samples were loaded into each well (100 μL per well); 3) plates were shaken for mixing and then incubated at 37°C for 2 h; 4) plates were washed in washing buffer 4–6 times and then air dried; 5) enzyme conjugates were added into each well (100 μL per well) for 1-h incubation at 37°C; 6) plates were washed in washing buffer 4–6 times and then air dried; 7) substrate solution was added into each well (100 μL per well) for incubation at 37°C for 10 min in the dark; 8) stop solution was added into each well (50 μL per well), and plates were agitaged to mix the solution; 9) absorbance at 492 nm and 450 nm was measured and the concentrations were calculated based on these absorbance values.
Determination of superoxide dismutase (SOD) value in each group
Serum samples were centrifuged at 3000 rpm for 10 min at room temperature. The supernatant was then collected and kept at −70°C. SOD concentration was measured using xanthine oxidase method according to the manufacturer’s instructions (kit supplier: Nanjing Jiancheng Bioengineering Institute, China) [12]. Colorimetry was conducted at a wavelength of 550 nm. Total SOD activity=((absorbance of control–absorbance of sample)/(absorbance of control))/50%×dilution factor of the reaction system×sample dilution factor before the test.
Determination of malondialdehyde (MDA) level in each group
Blood serum samples were centrifuged at 3000rpm for 10 min at room temperature, and the supernatant was collected and kept at −70°C. The MDA concentration was measured using TBA method (kit supplier: Nanjing Jiancheng Bioengineering Institute, China) [13]. Absorbance at 532 nm was measured in each tube. The formula for calculating MDA content was as follows: MDA value=((absorbance of the sample–absorbance of the blank)/(absorbance of the standard–absorbance of the blank standard))×standard concentration×sample dilution factor before the assay.
qRT-PCR
Total RNAs were extracted from rats’ vascular endothelium using a TRIzol kit (Invitrogen, USA). The extraction was performed in accordance with the manufacturer’s instructions, and ultrapure water treated with DEPC (Sangon, China) was used. Absorbance at 260 nm and 280 nm was measured by use of a UV-Vis spectrophotometer (ND-1000, Nanodrop, Thermo Fisher Scientific, USA). The quality of total RNA was checked and the concentration was adjusted. Next, RNA samples were reversely transcribed using a cDNA reverse transcription kit (K1622, Fermentas, USA). According to the instruction manual, the reaction conditions were set as follows: 70°C for 5 min, ice bath 2 min, 42°C for 50 min, and 70°C for 8 min. The cDNAs obtained from the reverse transcription were kept at −80°C. TaqMan probe method was conducted for qRT-PCR, and the reaction system was set up in accordance with the manufacturer’s instructions (TAG207, Baiaolaibo, China). Primer sequences are presented in Table 1. The running parameters were set as follows: 95°C for 30 s (pre-denaturation), 95°C for 10 s (denaturation), 60°C for 20 s (anneal), and 70°C for 10 s (extension) for 40 cycles. Real-time quantitative PCR (Bio-Rad iQ5, Bio-Rad, USA) was used for the test. U6 was taken as an internal control for miRNA-20b-5p, while β-actin was chosen as an internal control for other target genes. The relative quantitative method was employed and the relative gene expression was calculated by 2−ΔΔCt method. Each reaction was done in triplicate.
Table 1.
Primer sequences for qRT-PCR.
| Gene | Primer |
|---|---|
| PDCD4 | Forward: 5′-GTG ACG CCC TTA GAA GTG GA-3′ |
| Reverse: 5′-CTG CAC CAC CTT TCT TTG GT-3′ | |
| Akt | Forward: 5′-GTGCTGGAGGACAATGACTACGG-3′ |
| Reverse: 5′-AGCAGCCCTGAAAGCAAGGA-3′ | |
| eNOS | Forward: 5′-GCTCTCACTTACTTCCTGGACATCA-3′ |
| Reverse: 5′-GAACCACTTCCATTCTTCGTAGC-3 | |
| GADPH | Forward: 5′-GTTTCTTACTCCTTGGAGGCCAT-3′ |
| Reverse: 5′-TGATGACATCAAGAAGTGGTGAA-3′ |
PDCD4 – programmed cell death 4; eNOS – endothelial NOS.
Western blot analysis
Samples of vascular endothelium were placed in centrifuge tubes and incubated with 100 μL RIPA buffer containing 1 mmol/L PMSF (Solarbio, China) followed by centrifugation at 3000 rpm for 3 min and homogenization for complete lysis. Next, the samples were placed on ice for 30 min and centrifuged again at 1200 rpm at 4°C for 4 min. The supernatant was obtained and kept at −80°C. BCA protein assay kit (AR0146, Boster, China) was used for measuring the protein concentration in tissues. The sample concentration was adjusted to 3 μg/μL before adding loading buffer and heating at 95°C for 10 min. Then, the protein samples were loaded into wells (30 μg per well) and separated by 10% SDS-PAGE. Afterward, the proteins were transferred to a PVDF membrane (Sigma, USA) using a semi-dry transfer method, and were blocked in 5% BSA (Zhongshenglikang, China) for 1 h at room temperature. Samples were then incubated at 4°C overnight with antibodies as follows: anti-PDCD4 primary antibody (rabbit anti-mouse, dilution 1: 1000, ab51495, Abcam, UK), Akt primary antibody (rabbit anti-mouse, dilution 1: 1000, 2966S, Cell Signaling Technology, USA), p-Akt primary antibody (rabbit anti-mouse, dilution 1: 1000, 4070S, Cell Signaling Technology, USA), anti-endothelial NOS (eNOS) primary antibody (rabbit anti-mouse, dilution 1: 1000, ab199956, Abcam, UK), anti-eNOS primary antibody (rabbit anti-mouse, dilution 1: 1000, ab215717, Abcam, UK), and anti-GADPH primary antibody (rabbit anti-mouse, 1 μg/mL, ab37168, Abcam, UK). Afterward, membranes were washed 3 times in TBST (5 min per wash) and we added goat anti-rabbit secondary antibody (dilution 1: 2000, ab6721, Abcam, UK) for 1-h incubation at room temperature before being washed again 3 times (5 min per wash). The Gel Doc EZ Imager (Bio-Rad, USA) was used to yield images, and the gray value of the target band was analyzed by Image J software.
Statistical analysis
SPSS 21.0 statistical software was used for data analysis. Measurement data are expressed as mean ± standard deviation. Comparisons across multiple groups were conducted by one-way ANOVA, and comparisons of 2 groups were performed by t test if the data were normally distributed. A value of P<0.05 was considered statistically significant.
Results
CEC count in each group
Results of the CEC count in rat serum in each group can be seen in Figure 2. In comparison with the control group, the CEC counts in other groups were all elevated considerably (all P<0.05). The sh-PDCD4, IGF-1, and sh-PDCD4+IGF-1 groups had much lower CEC counts than in the model group (all P<0.05), whereas no intergroup differences were found between the model group and the NC group (P>0.05). The sh-PDCD4+IGF-1 group had lower CEC counts than the sh-PDCD4 group (P<0.05), and the CEC count in the IGF-1 group did not differ from that in the sh-PDCD4 group (P>0.05).
Figure 2.
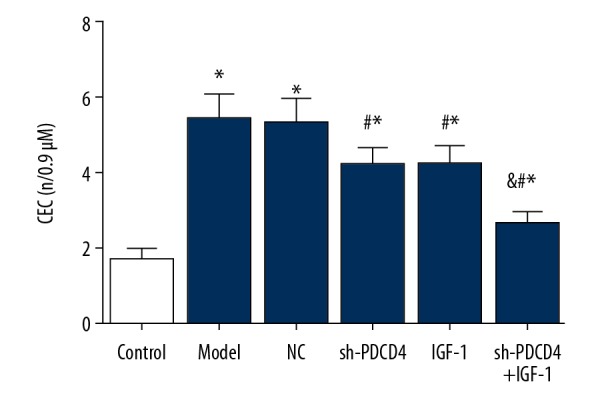
Comparison of CEC count among each group. * P<0.05 vs. the control group, # P<0.05 vs. the model group, & P<0.05 vs. the sh-PDCD4 group. CEC – circulating endothelial cell; PDCD4 – programmed cell death 4; IGF-1 – insulin-like growth factor 1.
TM and vWF values in each group
Results of the TM and vWF levels in rat serum in each group is shown in Figure 3. Compared with the control group, the TM and vWF values in other groups all rose significantly (all P<0.05). Moreover, these 2 values in the sh-PDCD4 group, IGF-1 group, and sh-PDCD4+IGF-1 group were much lower than those in the model group (all P<0.05), whereas no differences were observed between the model group and the NC group (both P>0.05). Compared with the sh-PDCD4 group, the TM and vWF values in the sh-PDCD4+IGF-1 group were lower (all P<0.05), while similar values were found in the IGF-1 group (both P>0.05).
Figure 3.
Comparison of TM and vWF levels among groups. (A) TM level in each group. (B) vWF level in each group. * P<0.05 vs. control group, # P<0.05 vs. model group, & P<0.05 vs. sh-PDCD4 group. TM – thrombomodulin; vWF – von Willebrand factor; PDCD4 – programmed cell death 4; IGF-1 – insulin-like growth factor 1.
SOD level in each group
The SOD levels in rat serum in each group is shown in Figure 4. Compared with the control group, the SOD levels in other groups were much lower (all P<0.05). Meanwhile, the SOD values in the sh-PDCD4 group, IGF-1 group, and sh-PDCD4+IGF-1 group were much higher than that in the model group (P<0.05), and no intergroup difference was found between the NC group and the model group (P>0.05). Moreover, the sh-PDCD4+IGF-1 group had a much higher SOD value (P<0.05), and the IGF-1 group had a similar SOD value, when compared with the sh-PDCD4 group (P>0.05).
Figure 4.

Comparison of SOD level among groups. * P<0.05 vs. control group, # P<0.05 vs. model group, & P<0.05 vs. sh-PDCD4 group. SOD – superoxide dismutase; PDCD4 – programmed cell death 4; IGF-1 – insulin-like growth factor 1.
MDA level in each group
The MDA values in rat serum in each group is shown in Figure 5. Compared with the control group, the MDA values in other groups all increased significantly (all P<0.05), and the MDA levels in the sh-PDCD4, IGF-1, and sh-PDCD4+IGF-1 groups were much lower than that in the model group (all P<0.05). However, no difference was found between the NC group and the model group (P>0.05). Moreover, the MDA level in the sh-PDCD4+IGF-1 group was much lower (P<0.05), and the MDA level in the IGF-1 group was similar (P>0.05), compared with the sh-PDCD4 group.
Figure 5.

Comparison of MDA level among each group. * P<0.05 vs. the control group, # P<0.05 vs. the model group, & P<0.05 vs. the sh-PDCD4 group. PDCD4 – programmed cell death 4; IGF-1 – insulin-like growth factor 1; MDA – malondialde NC.
Levels of PDCD4, Akt, and eNOS mRNAs in endothelium of each group as measured by qRT-PCR
The qRT-PCR results are presented in Figure 6. Compared with the control group, the expression levels of PDCD4 mRNA were significantly upregulated in the other 5 groups, whereas the expression levels of Akt and eNOS mRNAs were markedly downregulated in these groups (all P<0.05). Compared with the model group, the sh-PDCD4 and sh-PDCD4+IGF-1 groups had lower mRNA expression level of PDCD4 and higher mRNA expression levels of Akt and eNOS (all P<0.05), while the IGF-1 group had similar mRNA expression level of PDCD4 (P>0.05) and higher mRNA expression levels of Akt and eNOS (both P<0.05). No differences in these indices were observed between the NC group and the model group (all P>0.05). Moreover, compared with the sh-PDCD4 group, the sh-PDCD4+IGF-1 group had greater reduction in the mRNA expression level of PDCD4 and greater increase in the mRNA expression levels of Akt and eNOS (all P<0.05), whereas the IGF-1 group had higher mRNA expression of PDCD4 (P<0.05) and similar mRNA expression levels of Akt and eNOS (both P>0.05).
Figure 6.

Comparison of mRNA expressions among groups after transfection. * P<0.05 vs. the control group, # P<0.05 vs. the model group, & P<0.05 vs. the sh-PDCD4 group. PDCD4 – programmed cell death 4; eNOS – endothelial NOS; IGF-1 – insulin-like growth factor 1.
Levels of PDCD4, Akt, p-Akt, eNOS, and p-eNOS proteins as measured by Western blot in each group
Results of Western blot analysis can be seen in Figure 7. In comparison with the control group, the protein expressions of PDCD4 were all upregulated and the protein expressions of Akt, p-Akt, eNOS, and p-eNOS were all downregulated in the other groups (all P<0.05). Compared with the model group, the sh-PDCD4 group and the sh-PDCD4+IGF-1 group had lower protein expression levels of PDCD4 and higher protein expression levels of Akt, p-Akt, eNOS, and p-eNOS (all P<0.05), while the IGF-1 group had a similar protein expression level of PDCD4 (P>0.05) and higher protein expression levels of Akt, p-Akt, eNOS, and p-eNOS (all P<0.05). No differences were found between the NC group and the model group (all P>0.05). Compared with the sh-PDCD4 group, the protein expression level of PDCD4 in the sh-PDCD4+IGF-1 group was lower, while the protein expression levels of Akt, p-Akt, eNOS, and p-eNOS in this group were higher (all P<0.05). However, the protein expression level of PDCD4 in the IGF-1 group was higher than in the sh-PDCD4 group (P<0.05), and no differences were found in the protein expression levels of Akt, p-Akt, eNOS, and p-eNOS between the IGF-1 group and the sh-PDCD4 group (all P>0.05).
Figure 7.
Comparison of protein expression in each group after transfection. (A) Western blot result. (B) protein expression level in each group. * P<0.05 vs. control group, # P<0.05 vs. model group, & P<0.05 vs. sh-PDCD4 group. PDCD4 – programmed cell death 4; eNOS – endothelial NOS; IGF-1 – insulin-like growth factor 1.
Discussion
Lower-extremity IRI is a common cardiovascular disease, and oxidative stress is currently believed to be the key mechanism behind the pathogenesis of this disease [14]. When lower-extremity IR occurs in humans, huge amounts of reactive oxygen species (ROS) can exist in reperfused tissue. Meanwhile, due to the oxygen resupply, a series of inflammatory mediators such as lipid mediators and ROS will be generated. Since most of the lipid mediators and ROS come from endothelial cells, endothelial cells are often regarded as an essential component in the occurrence of IRI [15,16].
NOS, which can catalyze NO formation, is one of the essential compounds involved in oxidative stress and inflammatory response [17]. NO can clean up oxygen free radicals through cGMP and suppress the expression of intercellular adhesion molecule 1, thereby reducing neutrophil adhesion and infiltration, and protecting cellular structure and function [18]. However, NO, if overproduced, can combine with superoxide anions to form superoxide nitrite ions, which can lead to cytotoxicity and aggravate damage to cells and tissues [19]. Studies have confirmed that eNOS plays a protective role in vascular injury. It can regulate vasodilation, improve microcirculation, and reduce the occurrence of thrombus [20,21]. After examining the levels of PDCD4, Akt, and eNOS, and activation status of the Akt signaling pathway, we found that PDCD4 gene silencing activated the Akt signaling pathway and promoted the expression of eNOS. These findings demonstrate that PDCD4 gene silencing may play a protective role in vascular injury. Wang et al. have confirmed in their studies that PDCD4 gene participates in vascular remodeling through regulation of the JNK signaling pathway [22].
CEC count is a major indicator used in monitoring vascular endothelial injury [23]. Normally, some CEC exists in human tissues as the result of normal body metabolism. However, under pathological conditions, the CEC count can rise markedly [24]. TM and vWF are also key markers used in monitoring endothelial injury [25–27]. Wu et al. reported that miR-26a can inhibit PFKFB3 in a targeted manner, thereby reducing VEC injury caused by lower-extremity ischemia-reperfusion. After miR-26a overexpression or silencing of PFKFB3 expression in VEC, it was reported that CEC count and expression levels of TM and vWF all were markedly decreased in vascular tissues of mice with IRI [28]. In the present study, we found that the control group had lower CEC, TM, and vWF levels than in the other groups. Moreover, compared with the model group, the CEC count and TM and vWF levels were greatly decreased in the sh-PDCD4, IGF-1, and sh-PDCD4+IGF-1 groups, which further suggests that PDCD4 gene silencing can help activate the Akt signaling pathway, thereby attenuating VEC injury caused by lower-extremity IRI. In addition, Moake et al. found that cerebral cavernous malformation is closely related to PDCD gene mutation [29].
Studies have documented that oxygen free radicals are essential in causing extremity IRI [30]. Excessive production of oxygen free radicals not only harm limbs but also leads to organ injury, as these free radicals are likely to travel to different organs [31]. MDA is a major metabolite in the oxidizing reaction induced by oxygen free radicals in humans, and its level can directly reflect the content of oxygen free radicals [32]. Meanwhile, SOD, as an essential protein in human body that carries a negative charge, can help clean up free radicals in the body. The level of SOD and the ability SOD to scavenge free radicals are closely correlated [33]. From the results of the MDA and SOD levels in each group, it can be seen that the sh-PDCD4 group and sh-PDCD4+IGF-1 group had much lower MDA values and much higher SOD values than in the model group. In addition, the increase in SOD value and the decrease in MDA value in the sh-PDCD4+IGF-1 group were more significant. Tran et al. reported that patients with lower-extremity IRI experience elevation of MDA level and reduction in SOD level, which helps to verify our results [34]. A study by Zeng et al. on the attenuating effect of pretreatment with total flavonoid extract from Dracocephalum moldavica L. on ischemia-reperfusion-induced apoptosis documented that pretreatment with total flavonoid extract from Dracocephalum moldavica L. reduced MDA level and an increase SOD level in IRI mice [35]. Our study has provided some new insights into the treatment of lower-extremity IRI. However, more studies are needed to determine whether the upstream gene is regulated by miRNA.
PDCD4 gene silencing can activate the Akt signaling pathway and attenuate VEC injury caused by lower-extremity IRI in rats. PDCD4 may become an essential target in the treatment of lower-extremity IRI. However, more in vivo and in vitro studies are required for verification of our results.
Conclusions
PDCD4 expression can be markedly elevated in rats with lower-extremity IRI, while silencing PDCD4 can activate the Akt signaling pathway, reduce CEC count and levels of TM and vWF, inhibit MDA level, and increase SOD level in rat serum, which demonstrates the protective effects against VEC injury induced by lower-extremity IRI.
Footnotes
Source of support: This work was supported by a grant from Zhejiang Provincial National Natural Science Foundation of China (Y16H020015)
Conflicts of interest
None.
References
- 1.Comu FM, Kilic Y, Ozer A, et al. Effect of Picroside-2 on erythrocyte deformability and lipid peroxidation in rats subjected to lower extremity ischemia-reperfusion injury. Acta Physiologica. 2015;215:71. [Google Scholar]
- 2.Nagy T, Kovacs V, Hardi P, et al. Inhibition of glutathione S-transferase by ethacrynic acid augments ischemia-reperfusion damage and apoptosis and attenuates the positive effect of ischemic postconditioning in a bilateral acute hindlimb ischemia rat model. J Vasc Res. 2015;52:53–61. doi: 10.1159/000381984. [DOI] [PubMed] [Google Scholar]
- 3.Guo C, Yang M, Jing L, et al. Amorphous silica nanoparticles trigger vascular endothelial cell injury through apoptosis and autophagy via reactive oxygen species-mediated MAPK/Bcl-2 and PI3K/Akt/mTOR signaling. Int J Nanomedicine. 2016;11:5257–76. doi: 10.2147/IJN.S112030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liu L, Fujimoto M, Kawakita F, et al. Anti-vascular endothelial growth factor treatment suppresses early brain injury after subarachnoid hemorrhage in mice. Mol Neurobiol. 2016;53:4529–38. doi: 10.1007/s12035-015-9386-9. [DOI] [PubMed] [Google Scholar]
- 5.Liu JK, Ying M, Zhang JY, et al. Thioridazine upregulates programmed cell death 4 to induce apoptosis in nasopharyngeal carcinoma through the PI3K/Akt signalling pathway. Anti-Cancer Drugs. 2018;29:1. doi: 10.1097/CAD.0000000000000574. [DOI] [PubMed] [Google Scholar]
- 6.Kumar S, Alhasawi N, Marriott CE, et al. Programmed cell death gene 4 (PDCD4), a key regulator of pancreatic beta cell survival. Diabetic Medicine. 2015;32:36. [Google Scholar]
- 7.Kolpa HJ, Peal DS, Lynch SN, et al. miR-21 represses Pdcd4 during cardiac valvulogenesis. Development. 2013;140:2172–80. doi: 10.1242/dev.084475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Su Q, Li L, Liu Y, et al. Ultrasound-targeted microbubble destruction-mediated microRNA-21 transfection regulated PDCD4/NF-kappaB/TNF-alpha pathway to prevent coronary microembolization-induced cardiac dysfunction. Gene Ther. 2015;22:1000–6. doi: 10.1038/gt.2015.59. [DOI] [PubMed] [Google Scholar]
- 9.Yu L, Li F, Zhao G, et al. Protective effect of berberine against myocardial ischemia reperfusion injury: Role of Notch1/Hes1-PTEN/Akt signaling. Apoptosis. 2015;20:796–810. doi: 10.1007/s10495-015-1122-4. [DOI] [PubMed] [Google Scholar]
- 10.Pei YH, Chen J, Xie L, et al. Hydroxytyrosol protects against myocardial ischemia/reperfusion injury through a PI3K/Akt-dependent mechanism. Mediators Inflamm. 2016;2016 doi: 10.1155/2016/1232103. 1232103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Takahashi S, Nakagawa K, Tomiyasu M, et al. Mesenchymal stem cell-based therapy improves lower limb movement after spinal cord ischemia in rats. Ann Thorac Surg. 2018;105:1523–30. doi: 10.1016/j.athoracsur.2017.12.014. [DOI] [PubMed] [Google Scholar]
- 12.Li L, Xiong DF, Liu JW, et al. A cross-sectional study on oxidative stress in workers exposed to extremely low frequency electromagnetic fields. Int J Radiat Biol. 2015;91:420–25. doi: 10.3109/09553002.2015.1012304. [DOI] [PubMed] [Google Scholar]
- 13.Lapo RA, Gogny M, Chatagnon G, et al. Equine digital veins are more sensitive to superoxide anions than digital arteries. Eur J Pharmacol. 2014;740:66–71. doi: 10.1016/j.ejphar.2014.06.016. [DOI] [PubMed] [Google Scholar]
- 14.Kao MC, Yang CH, Chou WC, et al. Cepharanthine mitigates lung injury in lower limb ischemia-reperfusion. J Surg Res. 2015;199:647–56. doi: 10.1016/j.jss.2015.06.041. [DOI] [PubMed] [Google Scholar]
- 15.Gu M, Mordwinkin NM, Kooreman NG, et al. Pravastatin reverses obesity-induced dysfunction of induced pluripotent stem cell-derived endothelial cells via a nitric oxide-dependent mechanism. Eur Heart J. 2015;36:806–16. doi: 10.1093/eurheartj/ehu411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang S, Shaw-Boden J, Banz Y, et al. Effects of C1 inhibitor on endothelial cell activation in a rat hind limb ischemia-reperfusion injury model. J Vasc Surg. 2018;68(6S):209S–21S.e2. doi: 10.1016/j.jvs.2017.10.072. [DOI] [PubMed] [Google Scholar]
- 17.Ghigo D, Todde R, Ginsburg H, et al. Erythrocyte stages of Plasmodium falciparum exhibit a high nitric oxide synthase (NOS) activity and release an NOS-inducing soluble factor. J Exp Med. 1995;182:677–88. doi: 10.1084/jem.182.3.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paul KC, Sinsheimer JS, Rhodes SL, et al. Organophosphate pesticide exposures, nitric oxide synthase gene variants, and gene-pesticide interactions in a case-control study of Parkinson’s disease, California (USA) Environ Health Perspect. 2016;124:570–77. doi: 10.1289/ehp.1408976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Neufeld AH, Shareef S, Pena J. Cellular localization of neuronal nitric oxide synthase (NOS-1) in the human and rat retina. J Comp Neurol. 2000;416:269–75. doi: 10.1002/(sici)1096-9861(20000110)416:2<269::aid-cne11>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 20.Bian M, Xu Q, Xu Y, et al. Investigation on oxidative stress of nitric oxide synthase interacting protein from Clonorchis sinensis. Parasitol Res. 2016;115:77–83. doi: 10.1007/s00436-015-4723-5. [DOI] [PubMed] [Google Scholar]
- 21.Cunha AS, Matheus FC, Moretti M, et al. Agmatine attenuates reserpine-induced oral dyskinesia in mice: Role of oxidative stress, nitric oxide and glutamate NMDA receptors. Behav Brain Res. 2016;312:64–76. doi: 10.1016/j.bbr.2016.06.014. [DOI] [PubMed] [Google Scholar]
- 22.Wang F, Zhao XQ, Liu JN, et al. Antagonist of microRNA-21 improves balloon injury-induced rat iliac artery remodeling by regulating proliferation and apoptosis of adventitial fibroblasts and myofibroblasts. J Cell Biochem. 2012;113:2989–3001. doi: 10.1002/jcb.24176. [DOI] [PubMed] [Google Scholar]
- 23.Wang H, Pan Y, Zhang Z, et al. A novel KRIT1/CCM1 gene insertion mutation associated with cerebral cavernous malformations in a Chinese family. J Mol Neurosci. 2017;61:221–26. doi: 10.1007/s12031-017-0881-5. [DOI] [PubMed] [Google Scholar]
- 24.Dignat-George F, Sampol J. Circulating endothelial cells in vascular disorders: New insights into an old concept. Eur J Haematol. 2000;65:215–20. doi: 10.1034/j.1600-0609.2000.065004215.x. [DOI] [PubMed] [Google Scholar]
- 25.Beije N, Kraan J, Taal W, et al. Prognostic value and kinetics of circulating endothelial cells in patients with recurrent glioblastoma randomised to bevacizumab plus lomustine, bevacizumab single agent or lomustine single agent. A report from the Dutch Neuro-Oncology Group BELOB trial. Br J Cancer. 2015;113:226–31. doi: 10.1038/bjc.2015.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tong H, Chen R, Yin H, et al. Mesenteric lymph duct ligation alleviating lung injury in heatstroke. Shock. 2016;46:696–703. doi: 10.1097/SHK.0000000000000660. [DOI] [PubMed] [Google Scholar]
- 27.Matsubara Y, Matsumoto T, Yoshiya K, et al. Budding uninhibited by Benzimidazole-1 insufficiency prevents acute renal failure in severe sepsis by maintaining anti-coagulant functions of vascular endothelial cells. Shock. 2019;51(3):364–71. doi: 10.1097/SHK.0000000000001147. [DOI] [PubMed] [Google Scholar]
- 28.Wu Y, Zhang MH, Xue Y, et al. Effect of microRNA-26a on vascular endothelial cell injury caused by lower extremity ischemia-reperfusion injury through the AMPK pathway by targeting PFKFB3. J Cell Physiol. 2019;234:2916–28. doi: 10.1002/jcp.27108. [DOI] [PubMed] [Google Scholar]
- 29.Moake JL, Chow TW. Increased von Willebrand factor (vWf) binding to platelets associated with impaired vWf breakdown in thrombotic thrombocytopenic purpura. J Clin Apher. 1998;13:126–32. doi: 10.1002/(sici)1098-1101(1998)13:3<126::aid-jca6>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 30.Conklin DJ, Guo Y, Jagatheesan G, et al. Genetic deficiency of glutathione S-transferase P increases myocardial sensitivity to ischemia-reperfusion injury. Circ Res. 2015;117:437–49. doi: 10.1161/CIRCRESAHA.114.305518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhao S, Qian J, Wang J, et al. Effects of oxygen concentrations on postresuscitation myocardial oxidative stress and myocardial function in a rat model of cardiopulmonary resuscitation. Crit Care Med. 2015;43:e560–66. doi: 10.1097/CCM.0000000000001297. [DOI] [PubMed] [Google Scholar]
- 32.Wu Y, Xia ZY, Zhao B, et al. (−)-Epigallocatechin-3-gallate attenuates myocardial injury induced by ischemia/reperfusion in diabetic rats and in H9c2 cells under hyperglycemic conditions. Int J Mol Med. 2017;40:389–99. doi: 10.3892/ijmm.2017.3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hineno A, Nakamura A, Shimojima Y, et al. Distinctive clinicopathological features of 2 large families with amyotrophic lateral sclerosis having L106V mutation in SOD1 gene. J Neurol Sci. 2012;319:63–74. doi: 10.1016/j.jns.2012.05.014. [DOI] [PubMed] [Google Scholar]
- 34.Tran TP, Tu H, Pipinos II, et al. Tourniquet-induced acute ischemia-reperfusion injury in mouse skeletal muscles: Involvement of superoxide. Eur J Pharmacol. 2011;650:328–34. doi: 10.1016/j.ejphar.2010.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zeng C, Jiang W, Yang X, et al. Pretreatment with total flavonoid extract from Dracocephalum Moldavica L. attenuates ischemia reperfusion-induced apoptosis. Sci Rep. 2018;8:17491. doi: 10.1038/s41598-018-35726-4. [DOI] [PMC free article] [PubMed] [Google Scholar]




