Abstract
The novel understanding that the presence of multiple islet autoantibodies, indicating islet autoimmunity, inevitably leads to type 1 diabetes (T1D) has necessitated the development of a new staging classification system for the condition. Coupled with an improved understanding of the disease course, the realization that T1D appears to be more heterogeneous than previously thought has led to unique opportunities to develop more targeted therapies that may be applied even before the onset of dysglycemia or symptoms. To date, several therapies have been trialed to delay or halt disease progression in both presymptomatic and clinical T1D, each demonstrating varying degrees of effectiveness, toxicity, and utility. Key research supports the eventual implementation of immunotherapy in autoimmune diabetes, potentially calling for a paradigm shift among care providers. It will likely be necessary to develop new approaches to trial design and to address potential barriers to progress before an effective treatment for the disease may be achieved.
1. Introduction
Almost one hundred years after Dr. Frederick Banting and Dr. Charles Best’s landmark discovery of insulin, the diabetes community can now boast multiple types and brands of insulins, many different insulin delivery devices, and high-tech blood glucose monitoring systems. With the recent advent of the artificial pancreas, patients have renewed hope for better diabetes management options and improved glycemic control. However, while such advances have undoubtedly benefited those living with diabetes, they have served no role in actually preventing or altering the pathophysiology of the disease. It is generally accepted that the ultimate goal in the treatment of diabetes entails the prevention or deceleration of the autoimmune process behind beta cell destruction.
In most other autoimmune conditions, immunotherapy is used as a means of preventing disease progression or decreasing disease severity. Such is the case with juvenile idiopathic arthritis (JIA). Prior to the acceptance of the use of immunotherapies in JIA, the goal of treatment was simply to control symptoms of the condition such as joint pain and limited mobility. In the past few decades, immunotherapy has become more widely accepted and is now routinely prescribed to JIA patients in order not only to treat but to prevent joint erosion and disability. The use of such disease-modifying therapies has led to considerable improvements in quality of life for these patients [1]. JIA has a similar prevalence to T1D, a similarly high clinical impact, a comparable long preclinical phase, and a parallel etiopathology in that both autoimmune diseases are considered to be polygenetic with unknown etiologic agents [2]. However, immunotherapy is not typically considered in T1D.
Instead, for almost a century, the primary focus of treatment in T1D has been on correcting hyperglycemia and preventing its associated consequences. Research on glycemic control, diabetes complications, and quality of life reveals that we have much to gain through the development of novel therapies. Studies such as the Diabetes Control and Complications Trial (DCCT) and follow-up Epidemiology of Diabetes Interventions and Complications (EDIC) Trial have shown that poor glycemic control predisposes those with T1D to retinopathy, neuropathy, nephropathy, CVD, and other sequelae [3, 4]. Unfortunately, fewer than 30% of those with T1D are currently meeting A1c goals and mortality rates among patients with T1D remain significantly higher than rates for the general population [5, 6].
A proactive treatment approach, aimed at preventing or slowing beta cell destruction, and consequently thwarting hyperglycemia and its associated complications, has the potential to improve quality of life and decrease T1D’s overwhelming disease burden. While we don’t yet have therapies approved for clinical use, much progress has been made in recent years toward better understanding the T1D disease course and several therapies have already been shown to curb beta cell destruction at least temporarily. Many therapies have demonstrated differences in efficacy among unique subject populations, suggesting that the disease may be more heterogeneous than was previously thought. Such advances in our understanding of T1D will allow for greater opportunity to develop more targeted therapies. Accordingly, it is not unreasonable to imagine the use of disease-modifying therapies for T1D in the not-too-distant future.
2. New Insights into the Disease Course
In 1986, Dr. George Eisenbarth proposed that T1D was likely characterized by an unidentified precipitating event followed by a linear decline in beta cell function, eventually leading to a complete absence of insulin secretion [7]. However, in the last decade, natural history studies have revealed that beta cell decline is not necessarily linear and may not be absolute. Recent data demonstrate that beta cell dysfunction in autoantibody positive individuals is accelerated in the 6–12 months prior to clinical onset. Data from new-onset studies suggest that the decline in insulin secretion is also accelerated in the period shortly following the development of symptomatic disease. This pronounced loss of beta cell function persists for approximately a year following clinical diagnosis, after which insulin secretion continues to diminish but at a decelerated rate [8, 9]. An improved understanding of the clinical course of T1D has implications for the development and administration of therapies, as it can be assumed that efforts to preserve beta cell reserve and function would have the greatest hope of efficacy if applied prior to periods of rapid beta cell functional loss [10].
It has also been recently recognized that not all individuals experience an inevitable absence of insulin secretion. Results from the Joslin Medalist Study, multiple autopsy studies, and organ donor tissue studies show evidence of functional beta cells in those with longstanding T1D [11–13]. In the Medalist Study, 67.4% of participants with a diabetes duration ≥ 50 years had random serum C-peptide levels in the minimal (≥0.03 nmol/l) or sustained (≥0.20 nmol/l) range and postmortem analysis of pancreata from 9 medalists revealed insulin positive beta cells in all participants. Unsurprisingly, individuals with shorter diabetes duration were more likely to exhibit high levels of C-peptide production. Type 1 Diabetes TrialNet, an NIH funded, international clinical trials network aimed at developing disease-modifying therapies, has been following individuals with preclinical, new–onset, and longstanding diabetes for almost 2 decades. Combined data from multiple TrialNet studies show that 88% of those with new-onset T1D have peak C-peptide ≥0.20 nmol/l on mixed meal tolerance test (MMTT) at 12 months post-diagnosis and 66% at 24 months [30]. As discussed below, this is the amount of C-peptide considered essential for clinical benefit. Data from the T1D Exchange Clinic Network demonstrate that almost one out of three individuals with a diabetes duration of 36 months to more than 40 years have measureable residual insulin secretion, although the amount of secretion is almost always very low and rarely approaches clinically important levels [14].
Given the evidence of insulin production in those with longstanding T1D, one might conclude that those with the disease could benefit from therapies aimed at beta cell preservation or restoration even several years or decades after diagnosis. However, it is important to note that while C-peptide may be detectable in an individual, it remains unknown what level or duration of C-peptide production would confer clinical benefits. Post hoc analysis of data from the DCCT showed that a peak C-peptide of ≥0.20 nmol/l was associated with a decreased likelihood of progression to retinopathy or severe hypoglycemia [15]. Among the Medalist cohort, only 2.6% of individuals had this level of C-peptide upon MMTT. Interestingly, further analysis from the DCCT suggests that any detectable C-peptide is better than none and may correlate with a decreased risk of diabetes complications [16]. Similar observations have been made in islet transplant studies, where C-peptide levels insufficient to render participants insulin independent are effective in decreasing hypoglycemic unawareness [17, 18].
3. Starting Point: Genetic Risk
Family and twin studies reveal that genetics certainly play an important role in the development of T1D. In the past couple of decades, researchers have recognized that while the risk of T1D for people in the general population is approximately 1/300, those with a first or second degree relative with T1D have a risk of 1/20. Stated another way, relatives have a 15x increased risk of disease over that of the general population [19]. Among monozygotic twins, the risk is even higher, with disease concordance approaching 40% across age groups. This risk appears to be even higher when the index twin is diagnosed in early childhood [20].
The strongest association between genes and disease, accounting for up to 60% of genetic risk, is seen within the HLA region on chromosome 6 [21]. Specifically, the HLA class II DR3/DR4 genotype is known to confer the greatest risk for disease among Caucasian populations, being found in about 95% of those with T1D and only 60% of those without disease [21–23]. In contrast, HLA class II DQB1*0602 is associated with protection from disease; it is present in greater than 20% of the general Caucasian population, and less than 1% of children who develop T1D [24]. It is important to note that the HLA genotypes most commonly associated with T1D vary among different ethnic populations. For example, the DR3/DR4 genotypes that are so prevalent among Caucasians with T1D are actually very unusual in Japanese populations. Among Japanese, the DR4-DQ (DRB1*0405-DQB1*0401) and DR9 (DRB1*0901-DQB1*0303) HLA haplotypes, which are rare in Europeans, are most commonly associated with disease susceptibility [25, 26]. It is also worth mentioning that the positive predictive value of genotype is low as only about 5% of those with even the highest risk HLA types will ever advance to symptomatic T1D [27–30]. This fact has led to an increased interest in screening family members of those with T1D for alternate disease markers such as diabetes autoantibodies (see section 4) and in preventing the development and progression of disease in this higher risk population [31].
The linkage of T1D with the HLA class II region highlights the importance of adaptive immunity in disease as these genes make proteins responsible for antigen recognition and CD4 T cell activation [25]. T1D predisposition can further be attributed to roughly 50 other non-HLA genes or loci, including the INS, PTPN22, CTLA4, and IL2RA genes, all of which have been shown to have small to moderate effects on disease risk through a variety of mechanisms [32]. Again highlighting the importance of the immune system in T1D, in contrast to the genes that predispose to type 2 diabetes, few of these genes are thought to be related to beta cell function or metabolism.
Genetic studies have also provided clues to understanding the initiation and progression of disease. The Environmental Determinants of Diabetes in the Young (TEDDY) study, which follows genetically at-risk infants and children, initially found that autoantibodies to insulin generally developed prior to autoantibodies to GAD65. However, further analysis revealed that the order of appearance of autoantibodies was related to HLA-DQ genotype. Children with HLA-DQ2/8, DQ8/8, and DQ4/8 primarily developed insulin autoantibodies first, whereas children with DQ2/2 initially developed GAD65 autoantibodies. The TEDDY study also found that early autoantibody seroconversion was more commonly seen in children with the high-risk HLA DR3/4-DQ8 or DR4/4-DQ8/8 genotype. These findings suggest that there may be distinct etiopathologies of T1D related to HLA type [33, 34]. While HLA class II genotype impacts the development of autoantibodies, it does not appear to influence the course of disease after the onset of islet autoimmunity [24]. Conversely, several of the non-HLA T1D susceptibility genes, including IL2, CD25, INSVNTR, IL18RAP, PTPN22, IL10, and others have been shown to influence the rate of disease progression even beyond the development of autoantibodies [35].
4. Diabetes Autoantibodies Instrumental in Identifying Disease
Given the low positive predictive value of genetic factors, the recognition of diabetes autoantibodies such as GAD65 (GADA), zinc transporter 8 (ZnT8A), insulin (IAA), islet cell antibodies (ICA), insulinoma associated proteins (IA-2A and IA-2β), and others as markers of autoimmunity has been critical to the identification and definition of the preclinical stages of T1D. To learn more about the significance of autoantibody positivity, TrialNet designed the Pathway to Prevention Study (TN01). TN01 has served to determine the prevalence and prognostic value of diabetes autoantibodies among first and second degree relatives of those with T1D who, as previously noted, are at increased genetic risk for disease [31]. TrialNet currently offers free autoantibody screening to first degree relatives age 1–45 and second degree relatives age 1–20.
To date, more than 200,000 individuals have been screened for autoantibodies through TN01, with ~15,000 new relatives tested annually. Approximately 5% of those screened through this program are found to be autoantibody positive [19]. Results from this study and similar prospective studies have shed much light on the natural history of the disease [34, 36, 37]. We now understand that 95% of individuals destined to progress to symptomatic T1D before puberty will be autoantibody positive by age 5 [38]. In the TEDDY study, the initial detection of autoantibodies peaked between 9 and 24 months of age [39]. Available data also suggest that that once an individual demonstrates positivity for at least 2 autoantibodies, denoting islet autoimmunity, he will eventually progress to clinical T1D. While several factors, including age and autoantibody profile (see section 6) do affect the rate of progression to symptomatic T1D, multiple autoantibody positivity has been repeatedly shown to be highly predictive of disease across all phenotypes [31, 40]. Among genetically at-risk children followed from birth who progress to multiple autoantibody positivity, the 10-year risk of symptomatic T1D is 70% and lifetime risk approaches 100% [36]. Predictably, once an individual displays not only multiple autoantibody positivity, but also abnormal glucose tolerance, risk of progression to symptomatic T1D is even higher, with 60%−75% of those who fall into this category reaching clinical diagnosis within only 5 years. Again, lifetime risk of disease among this population approaches 100% [31, 41]. The prognostic value of single autoantibody positivity is less clear, although it does appear that the presence of a single autoantibody confers significantly more risk of progression to T1D in children than adults (see section 6)[40].
Some research has shown that the risk conferred by multiple autoantibody positivity is not limited to relatives of those with T1D. For instance, a study recently reported that the risk of progression to symptomatic T1D from the time of seroconversion to multiple autoantibody positivity was nearly 84% within 15 years among three pediatric cohorts from Finland, the U.S., and Germany [36]. Notably, while the German and U.S. participants did include relatives of T1D probands, the Finnish participants were recruited from the general population based on high risk HLA genotype. Despite the fact that two cohorts included T1D family members and one did not, rates of progression to T1D from recognized islet autoimmunity were the same in all three cohorts. This suggests that, once autoantibodies are detectable, disease advances in a similar manner regardless of family history. The TEDDY study, which recruits both neonates from the general population based on high risk HLA type as well as neonates with T1D family members and then follows participants every 3–6 months until age 15 for the development of autoantibodies, confirms these findings, again showing that once multiple autoantibodies appear, the risk of progression to clinical T1D is consistent among all participants [42]. With this new information, TrialNet is now opening its diabetes intervention studies up to all individuals with multiple autoantibody positivity, regardless of family history.
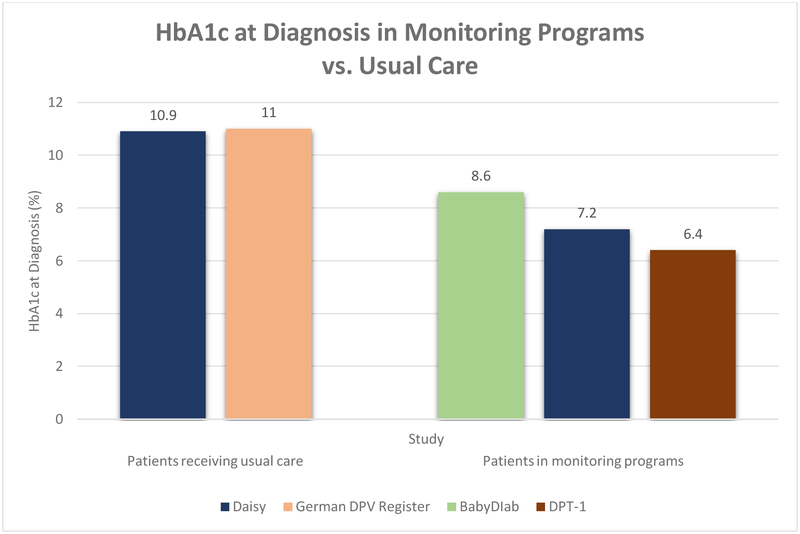
Depending on autoantibody screening results, participants in trials such as TN01 may be offered additional annual or semi-annual monitoring of their autoantibodies and glycemic status or the opportunity to enroll in one of several clinical trials aimed at slowing disease progression (see section 7). A growing body of evidence suggests that individuals at high risk of disease development who are closely monitored for progression are considerably less likely to experience peridiagnostic diabetic ketoacidosis (DKA) and exhibit lower HbA1c levels at diagnosis than those diagnosed in the community via symptom recognition (Fig. 1) [43–45]. In the Diabetes Prevention Trial-Type 1 (DPT-1), where high risk family members underwent semi-annual oral glucose tolerance tests (OGTT) to detect disease progression, only ~4% of individuals with new-onset T1D presented with DKA [7]. In contrast, 20–40% of patients with new-onset T1D present with DKA when diagnosed in the community without the benefit of monitoring programs [46]. Even a slight reduction in incidence of this common and potentially fatal condition is significant. In light of such benefits, the American Diabetes Association (ADA) has recommended since 2009 that family members of individuals with T1D be made aware of the opportunity to screen for autoantibodies in the context of a clinical research trial such as TN01 (see https://trialnet.org/) [47].
Fig. 1a.
DKA prevalence at diagnosis in monitoring programs vs. usual care [43–45, 48–51]
Given the high disease risk identified by multiple autoantibody positivity and the benefits of monitoring high-risk individuals for disease progression, some have posited that screening the general population for autoantibodies is warranted [52]. To date, population-wide screening has only been done in the context of clinical research trials [53, 54]. For instance, the Fr1da study aims to screen 100,000 children aged 2–5 years throughout Bavaria for autoantibodies at routine health examinations performed by their pediatricians. So far, preliminary results show that multiple autoantibodies have been detected in 4 of every 1000 individuals tested. Those who screen positive for autoantibodies are offered additional diabetes education through the study [53]. While screening for autoantibodies is readily feasible, the reduction in HbA1c and DKA at time of clinical diagnosis previously noted was the result of regular monitoring, not just screening. Given the low overall incidence of T1D (<0.5% in the general population), the possibility for false positive results, and the present lack of clinical guidelines concerning what actions should be taken when a positive autoantibody test result is obtained, many financial and logistical barriers must be addressed before population-wide screening is adopted as part of clinical practice [55, 56]. Yet, as soon as a successful disease-modifying therapy is available, routine screening of the general population will be essential to impact disease incidence; while risk remains greatest in relatives, 85% of those who develop T1D have no family history [57]. To prepare for this, multiple approaches to population-wide screening are being tested [58, 59]
5. New Staging Classification System for T1D
In response to the increased understanding of the path to T1D, in October of 2015, JDRF, the Endocrine Society, and the American Diabetes Association released a scientific statement endorsing the adoption of an official staging classification system for the disease (Fig. 2) [33]. According to this new system, T1D can now be categorized into three distinct stages. Prior to stage 1 of disease, it is believed that a yet unidentified inciting event activates the immune system against the beta cells, triggering the production of autoantibodies. In stage 1, individuals officially demonstrate β-cell autoimmunity by exhibiting at least 2 diabetes autoantibodies but remain normoglycemic. In stage 2, individuals maintain multiple autoantibody positivity and remain asymptomatic but display abnormal glucose tolerance as evidenced by impaired fasting glucose levels, an abnormal OGTT, or HbA1c≥ 5.7% [60]. Finally, stage 3 denotes the onset of clinical T1D, which is usually but not always accompanied by symptoms such as polyuria, polydipsia, weight loss, DKA, etc. This staging system introduces the concept that T1D starts at the time of multiple autoantibody development. Thus, clinical trials designed for those in stage 1 and stage 2 T1D can now be considered to treat rather than prevent early disease.
Fig. 2.
the stages of type 1 diabetes. (From: “Type 1 Diabetes: Stage 3” from the TrialNet website. [Image on the Internet]. C2018. Available from https://www.trialnet.org/our-research/long-term-follow-up. Used with permission) [61]
6. Factors Known to Influence Rate of Progression through Stages of T1D
6.1. Age
Multiple factors are known to influence the disease course of T1D, offering the possibility of predicting how rapidly an individual will advance from autoantibody positivity to stage 3 T1D and on to loss of clinically significant insulin secretion. A person’s age is known to have one of the most significant influences over disease progression, with younger individuals moving more rapidly through all three stages of T1D. To illustrate, children with a single autoantibody are more likely to progress to multiple autoantibody positivity than adults [31, 34]. A recent analysis of TrialNet data showed that single Ab + children <8 years had an impressive 35–37% risk of progression to multiple autoantibody positivity within 5 years, whereas adults >18 years had only a 13–16% chance of progression [40]. Further research has shown that young children typically convert from single to multiple antibody positivity within 2 years, with progression less frequent after 4 years of initial seroconversion. Immune responses in adults have been shown to mature over a longer period of time [62, 63]. Similarly, once islet autoimmunity is established, children are more likely to advance to dysglycemia (stage 2 T1D) and from dysglycemia to symptomatic (stage 3) T1D than adults [31]. Interestingly, risk of progression has also been shown to be increased among individuals whose T1D proband was diagnosed at a younger age, once again highlighting the heterogeneous nature of the disease [64].
Age continues to be impactful even after diagnosis, as there is a significantly more drastic decline in C-peptide over the first 2 years post-diagnosis in individuals ≤21 years than in those diagnosed over this age [9]. The effect of age at diagnosis upon residual insulin secretion can also be seen in those with more established T1D. While only 20% of children diagnosed ≤ 18yrs will have any detectable C-peptide 6–9 years after diagnosis, 60% of adults with an equivalent diabetes duration will demonstrate insulin production [14]. The more pronounced beta cell loss seen among children with T1D could signify a more aggressive disease course or may be explained by the fact that young children may have fewer beta cells at diagnosis than their adult counterparts.
6.2. Autoantibody Profile
T1D risk appears to vary with autoantibody number, titer, specificity, and type, as demonstrated by multiple longitudinal studies including TEDDY, DPT-1, and TN01[19, 31, 37, 40, 65]. In the DPT-1, risk of progression to symptomatic T1D intensified with increasing number of autoantibodies. Relatives of probands with T1D across all age groups had a 25% chance of progression to stage 3 T1D over 5 years if positive for 2 Abs, a 40% chance if positive for 3 Abs, and a 50% chance if positive for four Abs [37]. Subsequent data from the TrialNet group confirm these trends. Those with higher autoantibody titers have also been noted to have a higher risk of progression to stage 3 T1D [66, 67]. However, as autoantibody titer is highly correlated with autoantibody number, the utility of this marker is confounded [60]. Recently, an analysis of data from the TrialNet Pathway to Prevention Study showed that the pattern of progression to T1D may also vary by autoantibody type. Single autoantibody positive relatives with IAA were most likely to develop additional autoantibodies in early childhood but risk of progression decreased dramatically after age 8 years. In contrast, individuals with high GAD antibody titers maintained a high risk of progression to multiple autoantibody positivity even after early childhood [40]. This and other data support the idea that T1D may have multiple possible etiologies and pathways.
7. Immunotherapy for TID
Since TID was first described as an autoimmune disease over 30 years ago [7], investigators have used immunotherapy to intervene in the natural history of TID. This approach has been validated by the evidence that stage 1 TID can be identified early with diabetes specific autoantibodies. As with any therapy, immunotherapy must balance risk and burden of treatment with potential benefit. The earliest immunotherapies tested in TID were broad in their targets and often too toxic for widespread clinical use.
One of the earliest immune agents used was cyclosporine (CSA), a non-specific immunosuppressant. Although one year of treatment with CSA produced insulin independence in a majority of children with new-onset (stage 3) TID, remission required chronic therapy, and chronic therapy led to nephrotoxicity [68]. Since the CSA and other first-generation immunotherapy studies, available immunotherapies have become safer and more targeted. Simultaneous with improvements in therapies, disease mechanisms and specific immune targets have been identified in TID. The following section will highlight some general approaches towards immunotherapy in TID. Figure 3 illustrates some of these approaches.
Fig. 3.
Major pathways leading to beta cell destruction and potential mechanisms underlying the use of selected therapies. Figure used with permission from Endotext [69]
7.1. Targeting Inflammation with Specific Anti-Inflammatory Agents
Beta cell inflammation may be a necessary component of beta cell death in TID. As such, both specific and more general anti-inflammatories have been tested, mostly in stage 3 (new onset) disease.
7.1.1. Interleukin-1 (IL-1)
IL-1 is a cytokine important in the normal response to infection and inflammation. IL-1 blockade has been effective for treatment of rheumatoid arthritis and juvenile rheumatoid arthritis and is being tested for treatment of more general inflammatory conditions; in particular atherosclerosis [70]. Two different IL-1 blocking agents (Anakinra and Canakinumab) have been tested in phase 2 studies in stage 3 TID but failed to show any significant C-peptide preservation in treated individuals [71].
7.1.2. Tumor Necrosis Factor alpha (TNF-α)
A 2009 pilot study (n=18) in children with stage 3 disease using the TNF-α inhibitor Etanercept demonstrated an increase in C-peptide level and lower A1c in treated individuals [72]. Golimumab, also a TNF-α blocker, is currently in clinical trial in children with stage 2 [73] and stage 3 TID [74].
7.1.3. Interleukin-12/23 (IL-12/23)
The IL-12/23 cytokine pathway is an attractive therapeutic target due to its involvement in multiple immune pathways, including the release of proinflammatory cytokines and the support of pathogenic T effector cells. The IL-12/23 blocking agent Ustekinumab has been approved for use in psoriasis, psoriatic arthritis and Crohn’s disease. The Pilot Clinical Trial of Ustekinumab in Patients With New-onset TID (USTID) is a small (n=20) JDRF-sponsored phase 1/2 Canadian study testing Ustekinumab in adults with stage 3 TID [75]. USTID has completed enrollment.
7.1.4. Interleukin-6 (IL-6)
IL-6 is a compelling target of autoimmunity due to its effects on both innate and adaptive immunity. In TID, it promotes pathogenic T effector Th17 cells; furthermore, a subset of people with T1D have been shown to have an exaggerated response to IL-6 [76]. Anti-IL-6 therapies currently are used to treat rheumatoid arthritis, juvenile idiopathic arthritis and temporal arteritis. The EXTEND study [77], conducted by the Immune Tolerance Network (ITN) has completed enrollment of both adults and children with stage 3 TID testing whether 6 months of treatment with the IL-6 receptor blocker Tocilizumab can preserve beta cell function.
7.2. Targeting Inflammation with General Anti-Inflammatory Agents
7.2.1. Alpha 1 Antitrypsin (A1AT)
A1AT is a serum protease inhibitor which suppresses pro-inflammatory cytokines including TNF-α, IL-1, and IL-6. A1AT level is normal in TID but there are reports that its activity is impaired [78]. Early small studies in children and adults have demonstrated that A1AT is safe and well tolerated in stage 3 TID [79]. Other studies are testing the effects of A1AT in more established disease [80] [81].
7.2.2. Tyrosine Kinase Inhibition
Imatinib (Gleevac), a tyrosine kinase inhibitor, has broader anti-inflammatory effects and may play a role in TID. Imatinib is known mostly for its efficacy in chronic myelogenous leukemia and other malignancies. In the NOD mouse Imatinib can reverse diabetes [82], a finding which prompted a human trial, Imatinib Treatment in Recent Onset Type 1 Diabetes Mellitus [83]. The preliminary one year results of this study in adults with stage 3 TID were released in June 2017 at the ADA Scientific Session [84]; there is ongoing consideration of further trials in children using imatinib.
7.3. Targeting Antigen Presentation
Antigen presentation is the multi-step process by which proteins are presented to the immune system and then elicit an immune response, either tolerance or activation. Autoimmunity might be a failure of this process. Different strategies have been proposed and tested to redirect the immune response towards tolerance or away from auto-reactivity, some of which will be highlighted below.
7.3.1. Antigen Therapy
Repeated early exposure of an antigen to the naïve immune system may facilitate immune tolerance to that antigen. This approach has been successful in peanut allergies, where in genetically at-risk children, earlier exposure to peanut protein has been shown to decrease rates of future clinical peanut allergy [85]. Antigen therapy has been tested in both islet autoimmunity (stage 1) and in new-onset (stage 3) TID. As described earlier, insulin and GAD65 antibodies mark the presence of an immune response directed at these self-proteins. Studies in stage 1 TID have asked whether repeated exposure to insulin (orally, nasally or parenterally) can prevent or delay stage 3 TID. GAD65 therapy has been tested in both stage 1 and stage 3 TID. While antigen therapy (with insulin or GAD65) appears to be safe and tolerated, none of these approaches has been effective. Notably, trials testing oral insulin in primary prevention, prior to the development of islet autoimmunity, are underway, as described in section 8.3.2.
7.3.2. Target Antigen Presenting Cells
Another strategy is to interfere with specific antigen presenting cells such as the B cell. This has been tested with the B cell-depleting agent Rituximab, which can delay loss of C-peptide in stage 3 disease. A study by Pescovitz et al. published in NEJM in 2009 tested 4 weekly infusions of rituximab, which preserved C-peptide by 8.2 months compared to placebo treated [86]. Rituximab has not been tested in earlier stage disease; but targeting B cells remains attractive and additional trials are in planning stages.
7.3.3. Block MHC Function
Proteins and peptides are presented to the immune system in an MHC-dependent process. Interestingly, antigen presentation can be blocked mechanically with a small molecule, methyldopa. Methyldopa (Aldomet), an FDA approved alpha-adrenergic blocker used for decades to treat hypertension and pregnancy-induced hypertension, has been found to interfere directly with antigen presentation in individuals with DQ8 MHC class II alleles. DQ8 is present in 50–60% of people with TID and of those at risk. Preclinical studies [87] and a phase 1b escalation study [88] suggest that methyldopa can safely and durably block antigen presentation in this fashion. Trial Net aims to launch a study to evaluate the ability of methyldopa to block ongoing immune destruction of beta cells in relatives at risk of TID [89].
7.4. Targeting T cells
Another target of immunotherapy in TID is the T cell. Multiple agents to interfere with T cell activation, reduce the number or function of pathogenic T effector (Teff) cells or increase the number or function of T regulatory (Treg) cells have been tested.
7.4.1. T cell Signaling
Abatacept, a CTLA4 immunoglobulin, is currently used to treat rheumatoid arthritis, juvenile idiopathic arthritis and psoriatic arthritis. It blocks the costimulatory signal necessary for activation of naïve T cells, hence rendering the T cell anergic. Abatacept has demonstrated efficacy in stage 3 disease, where monthly infusions for 2 years delayed C-peptide decline by 9.6 months [90]. In follow up to the promising results in stage 3 disease, TrialNet is enrolling a study to test whether Abatacept given earlier, in stage 1 TID, can delay the onset of stage 2 or stage 3 TID [91].
7.4.2. Anti CD3 Therapy
The anti-CD3 monoclonal antibody binds to the CD3/Tcell receptor (TCR) complex, which leads to disappearance of the CD3/TCR complex and renders the T cell anergic to its target antigen. A number of studies have tested anti CD3 agents, including Teplizumab and Otelixizumab, both of which have demonstrated efficacy in stage 3 TID. A 2002 study published in NEJM by Herold et al., tested a single 14 day course of Teplizumab and demonstrated a delay in loss of C-peptide in Teplizumab treated compared to placebo treated groups [92]. In the AbATE trial, a second 14 day Teplizumab course was given 1 year after the first course, resulting in C-peptide preservation, with an average 15.9 month delay in loss of C-peptide at 2 years [93]. These promising results supported a study looking at earlier-stage disease. In 2010 TrialNet initiated a study asking whether a single 14 day course of Teplizumab in individuals with stage 2 TID can delay or prevent the onset of stage 3 TID [94]. Results of this study are expected in 2019.
7.4.3. Anti CD2 Therapy
It is possible that despite the elimination of a significant portion of Teff cells, destruction of memory Teff cells would also be required to create lasting effects. This has been testing with Alefacept, an anti-CD2 fusion protein which blocks T cell activation and induces apoptosis of memory Teff lymphocytes. An Immune Tolerance Network (ITN) study, TIDAL, results published in 2015, enrolled 49 children and adults (ages 12–35) with stage 3 TID. Participants were treated with two 12-week courses of drug or placebo. Unfortunately, the study was not fully enrolled due to insufficient drug availability. Despite this limitation, at one year, there was a non-significant trend towards preservation of C-peptide, lower insulin usage and fewer hypoglycemic events [95]. These metabolic benefits were sustained at 15 months beyond treatment in treated individuals [96].
7.4.4. Anti-Thymocyte Globulin (ATG)
Anti-thymocyte globulin (ATG) depletes activated T cells; in lower doses, it appears to deplete primarily Teff cells, and in higher doses it depletes both Teff and Treg cells. Low-dose ATG has been used in combination with granulocyte colony stimulating factor (GCSF) which supports leukocyte recovery; in particular, GCSF favors recovery of Tregs. A pilot study (n=24) published in 2015 in people with stage 3 TID (4 months-2 years after diagnosis) suggested the ATG/GCSF combination is safe and there was a trend toward C-peptide preservation [97]. A larger TrialNet study testing the combination of low dose ATG andGCSF vs low dose ATG alone vs placebo in stage 3 disease was completed in 2016. One year after treatment, low-dose ATG alone significantly preserved C- peptide relatives to the placebo group. Thus, this trial demonstrated that GCSF provided no additional benefit. Two-year clinical and mechanistic results are expected in 2019. [98]
7.4.5. Low Dose IL-2
Some individuals with TID have impaired responsiveness to interleukin 2 (IL-2), a cytokine with competing immune effects depending on dosage. In higher doses IL-2 promotes Teff activity, while in lower doses it promotes Treg activity. A pilot study in 2012 tested the combination of IL-2 with rapamycin, the latter of which blocks Teff cells. The combination was predicted to enhance the activity and survival of Tregs while reducing Teff effects. Although this immune benefit was seen, there were clinically-important adverse effects, including a transient decrease in C-peptide [99] whether or not the participants received the rapamycin. The study was terminated early, and mechanistic studies found that IL-2 stimulated both regulatory and NK cells. While subsequent studies are ongoing to test whether a lower dose of IL-2 would boost Treg activity without the adverse effects seen in the pilot study [100], the narrow therapeutic window for beneficial as compared to untoward effects of currently available IL-2 are challenging. Thus, variants of IL-2 which may convey more specificity for T regulatory cells are under development.
7.4.6. Treg Infusion
A more direct method to increase Tregs is by infusion of ex vivo expanded autologous Tregs. A small (n=14) phase 1 dose-finding study reported in 2015 demonstrated safety and tolerability of Treg therapy in stage 3 TID [101]. A similar phase 1 study (n=14) [102] recently completed enrollment. The TILT study, “A Phase I Trial of CD4+CD127lo/-CD25+ Polyclonal Treg Adoptive Immunotherapy with Interleukin-2 for the Treatment of Type 1 Diabetes” asked whether autologous Treg therapy combined with low dose IL-2 treatment immediately after Treg infusion and again one month after infusion can provide additional Treg support [103]. As recently reported (IDS meeting London, 2018) this approach also led to transient impairment in C-peptide.
7.5. Beta Cell Support
Agents to support the beta cell are unlikely to alter the underlying immune defects in TID, but they may be useful in combination with immunotherapy. Multiple agents have been tested; namely, metformin, glucagon-like peptide 1 (GLP1) agonists such as liraglutide, proton pump inhibitors (PPI) such as omeprazole, and hydroxychloroquine (HCQ).
7.5.1. Metformin
While metformin can allow for reduced insulin doses and appears to be safe in established TID, it does not offer any significant clinical benefit as measured by A1c [104]. However, it is unknown whether metformin might protect the beta cell or reduce beta cell stress in earlier stage TID. The Accelerator Trial (adAPT) is a randomized controlled trial [105] to test whether treatment with metformin in children with stage 1 TID can affect markers of beta cell function and reduce the progression towards stage 3 TID.
7.5.2. GLP-1 Agonists
In the presence of hyperglycemia, GLP-1 suppresses glucagon and stimulates insulin secretion [106]. Clinical benefits include improved glycemic control and weight loss without a risk of hypoglycemia [107]. Mouse studies demonstrate that GLP-1 treatment can promote beta cell proliferation and survival, reduce apoptosis, and delay autoimmune diabetes [108]. Furthermore, mouse studies have shown reversal of autoimmune diabetes when GLP-1 agonists are combined with immunotherapies. These effects have not been demonstrated in human disease. In fact, there have been few human trials testing beta cell-specific outcomes with GLP-1 treatment. In patients with longstanding TID, not surprisingly, GLP-1 treatment does not appear to alter C-peptide secretion [109]. However, GLP-1 agents might have a greater effect in earlier stages of disease when beta cell mass is greatest. There are ongoing studies to test this idea in all stages of TID including stage 3 with residual C-peptide [110] [111] [112], stage 2 [113], and stage 1 disease [114].
7.5.3. Proton Pump Inhibition (PPI)
PPIs are used clinically to treat gastroesophageal reflux disease and peptic ulcer disease. Among other effects, PPIs increase gastrin levels, which in animal models, can boost beta cell mass and therefore might be expected to have beneficial effects in human diabetes [115]. However, human studies on the effect of PPIs and glycemia have been mixed [116] [117] [118].
7.5.4. Hydroxychloroquine (HCQ)
HCQ is FDA approved for treatment of malaria, lupus, and rheumatoid arthritis. Interesting, it has been shown to improve insulin sensitivity and beta cell function [119]. In 2018 TrialNet launched a trial to investigate whether HCQ therapy can delay or prevent stage 2 or stage 3 TID in family members with stage 1 TID [120].
8. Other Approaches to Disease-Modifying Therapy
Preclinical studies in autoimmune diabetes suggested that short term treatment with a variety of different immunotherapies could produce a durable disease remission in animals. Based on the success in animal models of TID, this was the strategy employed in human clinical trials; that is, short term therapy to delay the loss of C-peptide. In stage 3 (new-onset) TID, many short term therapies have shown some efficacy and with a reasonable safety profile. However, among these studies, the results are similar: beta cell function is preserved for a period of time, after which C-peptide declines again. This observation is illustrated in figure 4. Although these results consistently illustrate that immunotherapy can alter the natural history of TID, especially in children, no therapy has yet produced a durable remission, which suggests that other strategies may be needed to produce long term C-peptide preservation. However, there may be benefits to even short term C-peptide preservation if it correlates with better glycemic control. The DCCT showed that intensive glycemic control for a mean of 6.5 years leads to better clinical outcomes even years after study completion, after which hemoglobin A1c values in the two groups had converged [121–123]. This sustained benefit to early glycemic control has been termed “metabolic memory” and it may be an additional benefit to short term C-peptide preservation in stage 3 TID.
Fig. 4.
Stylized representation of selected new-onset clinical trial results with positive outcomes. Therapies generally delayed the fall in insulin secretion from ~8 to 12 months. Regardless of the therapy, the most pronounced effects were early after treatment. Figure used with permission from Endotext [69]
8.1. Combination Therapy
With the increasing understanding that TID pathogenesis may involve multiple immune defects and multiple steps, combination therapy may produce a more durable remission. However, combination therapy must be approached with caution, as it may expose participants to additional risk. There may be strategies to mitigate the additional risk; for example, using therapies with non-overlapping but complementary mechanisms. As described earlier, the combination of low dose ATG and GCSF would be expected to deplete T lymphocytes with relative sparing of Tregs; yet the clinical trial found no benefit of the GCSF. Several trials are being considered whereby therapy will be administered sequentially; the aim being to gain the benefits of combined approaches while limiting the potential for increased risks.
8.2. Chronic Therapy
Short term immunotherapy in TID has not produced a durable remission and yet this continues to be the approach to disease modifying therapy. Contrast this to the approach in other autoimmune conditions (rheumatoid arthritis, inflammatory bowel disease, multiple sclerosis), where the standard of care is early and ongoing or intermittent treatment with single or combination disease modifying therapies to alter disease course by preventing tissue destruction and therefore also treat symptoms, including pain and loss of function. It is also understood that any single therapy may eventually lose efficacy, necessitating serial use of multiple therapies over the course of a lifetime. Chronic, sequential, or intermittent therapy has not been used in TID since the cyclosporine studies decades ago; but trials testing these approaches are being planned.
8.3. Earlier Therapy
It makes sense that we would intervene earlier in disease to preserve beta cells before TID becomes clinically apparent. Although we have evidence that beta cells can be preserved in stage 3 disease (after clinical diagnosis), at least for a period of time, less is known about the efficacy of immunotherapy in stage 1 or 2 disease or even earlier, before islet autoimmunity begins.
8.3.1. Selected Therapies to Prevent Stage 1 TID (Islet Autoimmunity)
Observational studies and demographic trends suggest multiple possible environmental triggers of islet autoimmunity. For example, TID and autoimmunity in general have increased with increasing westernization, a phenomenon which contributes to lower vitamin D levels, more hygienic standards of living, changing dietary patterns, etc. There are a limited number of randomized primary prevention trials in TID, in part due to the complexity and expense involved in such human trials and because any intervention directed toward primary prevention must be both safe and tolerable. For decades, observational studies have suggested that breastfeeding is associated with decreased rates of TID, but there were no randomized trials to support this finding. The Trial to Reduce IDDM in the Genetically At Risk (TRIGR) is a multicenter international study that randomized 2160 genetically at-risk infants to two feeding arms: either weaning to conventional cows’ milk formula or weaning to hydrolyzed casein formula, in which proteins are hydrolyzed to be non-immunogenic. In both groups, exclusive breast feeding was recommended until the age of 6 months. There was no benefit to weaning to hydrolyzed formula vs conventional formula in the incidence of either islet autoimmunity [124] or stage 3 TID [125]. Similar to the idea that breastfeeding may be protective, observational studies have suggested that delayed introduction of dietary gluten may be protective [126]. However, this observation has not been supported by a randomized trial. BABYDIET is a German birth cohort study that randomized 150 genetically at-risk infants to two feeding arms: one arm introduced dietary gluten at age 6 months, which is the standard recommendation in Germany, and one arm delayed introduction of gluten until 12 months. Although it was safe to delay gluten exposure, no change was demonstrated in the incidence of islet autoimmunity with delayed introduction of dietary gluten [127].
8.3.2. Selected Previous Studies Testing Therapies to Delay Progression of Stage 1 TID
Nicotinamide
Animal studies had suggested that nicotinamide might delay progression of autoimmune diabetes, perhaps by facilitating beta cell repair. The European Nicotinamide Diabetes Intervention Trial (ENDIT) study asked whether oral nicotinamide might delay the progression from stage 1 to stage 3 TID. Although nicotinamide therapy was shown to be safe, no benefit was demonstrated [128].
Antigen Therapies
Conceptually, antigen therapy might be expected to induce immune tolerance if introduced early and repeatedly to a maturing immune system. However, it is also possible that antigen treatment might “feed the fire” of an autoimmune response. Therefore, in TID, early studies of antigen therapy were performed in stage 3 TID, where clinical disease is already present. Candidate therapies were target antigens or peptides of the autoimmune response including insulin, GAD65 and proinsulin peptide, which is a short immunogenic portion of the larger proinsulin molecule that in animal models of autoimmune diabetes induces a pro-regulatory immune response [129]. Although studies in stage 3 TID demonstrated safety of these therapies, perhaps not surprisingly, they were mostly negative in their ability to preserve C-peptide [130] [131] [132]. In follow up to stage 3 trials, other studies have asked whether antigen therapy can delay or prevent progression in stage 1 TID disease. No effect has been demonstrated with either nasal [133] or parenteral insulin in stage 1 TID [134]. And although a post hoc analysis of data from the DPT-1 trial suggested a benefit to 7.5 mg daily oral insulin therapy in individuals with the highest insulin antibody titers [135] [136] [19], a fully powered TrialNet study showed no benefit in the primary cohort of more than 300 individuals. Interestingly however, an independently randomized cohort of 55 antibody positive individuals treated with 7.5 mg/day oral insulin had a 31 month delay in diagnosis of stage 3 TID as compared to placebo treated. At enrollment, individuals in this cohort had lower first-phase insulin responses but normal glucose tolerance, perhaps indicating a more active disease state. [137]. While the TrialNet study tested a low dose (7.5mg daily) of oral insulin in stage 1 disease, the Global Platform for the Prevention of Autoimmune Diabetes (GPPAD), a European trial consortium, will test a higher dose (67.5 mg daily) in genetically at risk children without islet autoimmunity. In the Primary Oral Insulin Trial (POiNT), genetically at-risk infants will be identified either via cord blood or newborn screening and then randomized to either placebo treatment or daily oral insulin 67.5 mg/day. The higher 67.5 mg dose of oral insulin was supported by the mechanistic Pre-POINT study, in which children ages 2–7 with a family history of TID, high risk HLA genotypes, and no islet autoimmunity were administered either placebo or varying doses of oral insulin. Although Pre-POINT was a small pilot study, results suggested that children who received the highest dose of oral insulin, 67.5 mg/day, had the most salutary immune responses, including regulatory T cell proliferation in response to insulin [138]. The Pre-POINT Early [139] is looking for a similar immune response in a younger cohort, ages 6 months-2 years. Similarly, a TrialNet study called “Immune Effects of Oral Insulin in Relatives at Risk for Type 1 Diabetes Mellitus” tested the effects of both variable oral insulin dosage and dosing schedule on immune responses [140]. In this open label study participants aged 3–45 years were treated either with 67.5 mg per day or with 500 mg every other week for 6 months.
8.3.3. Selected Current Studies Testing Therapies to Delay Progression to Stage 3 TID
There are multiple ongoing studies to test immunotherapies in either stage 1 or stage 2 TID, all with a goal to delay progression to stage 3 TID. Some of these studies are highlighted in Table 1.
Table 1.
Selected immunotherapy studies in Stage 1 or Stage 2 TID
| Study agent, therapeutic mechanism | Study name, references | Study population | Intervention | Results |
|---|---|---|---|---|
| Abatacept, (CTLA4-Ig), T-cell co-stimulation blockade | CTLA4-Ig (Abatacept) for Prevention of Abnormal Glucose Tolerance and Diabetes in Relative At Risk for Type 1 Diabetes [91] | TID relatives; stage 1; identified through TrialNet Pathway to Prevention; age 6–45;n=180 | 14 infusions of abatacept or placebo over 1 year | Enrolling, study outcome expected 2020 |
| Intranasal insulin/antigen therapy, mucosal tolerance | Trial of Intranasal Insulin in Children and Young Adults at Risk of Type 1 Diabetes (INITII) [141] | TID relatives; Stage 1; age 4–30; n=110; | Nasal insulin or placebo; 440 iu daily × 7d then one day/week × 12 months | Estimated completion 2019 |
| Alum-GAD (Diamyd) and Vitamin D3/antigen therapy, tolerance | Prevention Trial: Immune- tolerance With Alum-GAD (Diamyd) and Vitamin D3 to Children With Multiple Islet Autoantibodies (DiAPREV-IT2) [142] | Stage 1; identified through DiPiS, TEDDY or TrialNet; age 4–17; n=80 | 2 sc injections of Alum- GAD (Diamyd) or placebo 30 days apart, combined with vitamin D3 2000 iu/day, starting 30 days prior to first GAD or placebo dose | Enrolling, estimated completion 2022 |
| Teplizumab, anti- CD3/T cell depletion | Teplizumab for Prevention of Type 1 Diabetes in Relatives “At-Risk.” [94] | TID relatives; stage 2, identified through TrialNet Pathway to Prevention; age 8–45;n=71 | Daily iv infusion of teplizumab or placebo for 2 weeks | Enrollment completed, study outcome expected 2019 |
| Golimumab (TNF-α blocker)/anti-inflammatory | A Study to Evaluate SIMPONI (Golimumab) Therapy in Children, Adolescents and Young Adults With Pre-Symptomatic Type 1 Diabetes [73] | Stage 2; age 6–21; n=30 | Weekly weight based sc injection of golimumab or placebo for 26 weeks | Enrolling, estimated completion 2021 |
9. What is the Future of TID Research?
One can envision a future with multiple therapeutic options to prevent islet autoimmunity and to treat TID in all its stages. We will have a greater ability to personalize treatment and to measure response to treatment using disease-specific biomarkers. However, there are multiple steps between our current understanding of the natural history of TID and the clinical use of disease modifying therapies. Despite its challenges, TID research must remain a priority for funding agencies, scientists, clinicians and patients. While a full discussion of the challenges to TID research is outside the scope of this review, some barriers will be highlighted in the next section.
9.1. When does TID begin? Understanding the New Disease Model
As evidenced by the new TID staging model, established islet autoimmunity (stage 1 or 2 TID) inevitably leads to clinical (stage 3) TID; therefore, islet autoimmunity should be considered a disease and not a risk for a disease. This understanding might lead to a greater urgency for population-wide screening for islet autoimmunity, funding for TID research, and participation in studies. Immunotherapy should be incorporated into endocrinology training so that providers who treat people with islet autoimmunity will be familiar and comfortable with this option as future treatments become available. As reviewed earlier, family members of people with TID should be counseled about antibody screening through TrialNet, which currently is the primary means of identifying those with islet autoimmunity in the United States.
9.2. Trials Are Expensive and Slow: What Might Help?
9.2.1. Study Children
The current pathway toward regulatory approval of immunotherapy in TID is to first demonstrate safety and efficacy in adults prior to testing the therapy in children. As with every new therapy, but particularly for children, safety and equipoise are foremost. However, as reviewed earlier, children with TID serve to benefit most from potential beta cell-preserving therapy. Furthermore, the positive results in many TID new-onset trials are driven by the efficacy in children. This was seen in both the Rituximab and Abatacept new-onset trials, shown in figure 5, where the positive result in each study was driven by the response in children under age 18. It is likely that study endpoints could be reached faster and with fewer participants if more children were enrolled. This was one of the conclusions of a panel that met recently to discuss challenges associated with trial design for different age groups [31].
Fig. 5.
The positive results seen in both the rituximab (figure A) and abatacept (figure B) new-onset trials were driven by the response in the age < 18 cohorts. From Wherrett et al. [31].
9.2.2. Use Intermediate Endpoints
With the understanding that established islet autoimmunity is a disease equivalent to TID, the development of islet autoimmunity (stage 1) or impaired glucose tolerance (stage 2) can be used as an intermediate endpoint in study design. For example, the study endpoint for the TrialNet Abatacept in Prevention study [91] currently enrolling individuals with stage 1 disease, is impaired glucose tolerance or stage 2 disease. Mechanistic studies will likely identify earlier and more diverse TID disease biomarkers that can be used as intermediate endpoints and might personalize enrollment criterion. Ultimately, intermediate endpoints might allow investigators to more quickly identify promising therapies before investing in larger, more definitive trials [41].
9.2.3. Single Arm Studies
The natural history of TID is well described after clinical diagnosis (stage 3), with a predictable age-based fall in C-peptide [9]. Given the robust natural history data, it may not be necessary to include a placebo group in all stage 3 intervention studies. There are multiple barriers to clinical trial participation, but a single arm study design may eliminate a barrier for patients who are less likely to enroll if they are not guaranteed treatment with active drug.
9.2.4. Personalize Therapy
In both clinical practice and in clinical trials, it is understood that for any given treatment, only a subset of individuals will have a therapeutic response. In a TID new-onset study, this suggests that a portion of treated participants will maintain C-peptide; whereas the remainder of treated individuals will have a clinical course similar to placebo treated, with the expected decline in C-peptide after diagnosis. The so-called “responders” are identified retrospectively, based on their clinical outcome, without a prior understanding of how they may differ physiologically from non-responders. In fact, there are likely “endotypes” or TID disease subtypes that might be defined by a specific physiologic mechanism. Ongoing and future efforts will describe the physiologic mechanisms underlying endotypes and identify biomarkers to predict and measure response to therapy. With this individualized approach, therapies more likely to be effective for an endotype are selected, and ineffective therapies are discarded sooner. Importantly, whether in a clinical trial or in clinical practice, an individualized approach would produce greater benefits, fewer risks, and lower costs.
10. Conclusions
Nearly 100 years after the first clinical use of insulin for type 1 diabetes, the management of hyperglycemia and its complications remains the primary focus of TID treatment. Although there have been dramatic improvements in diabetes management tools, including more physiologic insulins, better insulin delivery methods, and real-time glucose monitoring systems, a majority of people with TID fail to meet the recommended therapeutic targets, and TID and its complications remain a significant burden to individuals, families, and society. We now understand that TID has a long and measurable preclinical period (stage 1 TID) that progresses inevitably to clinical or stage 3 TID. Additionally, there is ample evidence that the preservation of any remaining endogenous insulin secretion is clinically important; hence beta cell-sparing therapies at any stage of TID are potentially beneficial. However, although several therapies have proven effective in short-term C-peptide preservation, no treatment has durably interrupted the autoimmune destruction of beta cells. As a result, TID is not treated until it becomes clinically apparent, in contrast to the management of other autoimmune conditions. Although continued improvements in hyperglycemia management are welcome and inevitable, in the future, there may be a shift towards treatments that truly modify the disease, whether immunotherapies to disrupt the progression of islet autoimmunity, therapies to support the beta cell, or therapies to replace beta cells. Chronic or combination therapies to durably preserve insulin secretion might be needed, analogous to the treatment of other chronic autoimmune conditions such as rheumatoid arthritis. Importantly, treatment will become more personalized, to reflect the heterogeneity of TID in all of its stages.
Fig. 1b.
HbA1c at diagnosis in monitoring programs vs. usual care [43–45, 50, 51]
Key Points:
Type 1 diabetes may be diagnosed before the development of dysglycemia or symptoms based on the recognition of at least two islet autoantibodies, denoting islet autoimmunity. Early detection grants the opportunity for early intervention.
Several therapies, when used short-term, have demonstrated efficacy in preserving beta cell secretion in individuals newly diagnosed with type 1 diabetes. However, positive effects have not been sustained and are not yet sufficient to warrant routine use of immunotherapy in clinical practice. Therapies that have demonstrated efficacy with transient use have not yet been tested for chronic use in TID.
T1D is a heterogeneous disease, greatly influenced by age at diagnosis and other factors. New and potentially even individualized approaches to therapy, possibly including combination therapies, may be necessary before a cure can be achieved.
Funding:
No funding was used in the preparation of this review article.
Footnotes
Conflict of Interest
Carla Greenbaum reports personal fees from Lilly, grants and personal fees from NovoNordisk, personal fees from Bristol Myers Squibb, grants and personal fees from Janssen, and personal fees from Pfizer.
Sandra Lord and Dana VanBuecken declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including Helsinki declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Contributor Information
Carla Greenbaum, Benaroya Research Institute, 1201 Ninth Avenue, Seattle, WA 98101.
Dana VanBuecken, Benaroya Research Institute.
Sandra Lord, Benaroya Research Institute.
12 References
- 1.Walsh D and McWilliams D, Mechanisms, impact and management of pain in rheumatoid arthritis. Nat Rev Rheumatol, 2014. 10: p. 581–592. [DOI] [PubMed] [Google Scholar]
- 2.Firestein G, Ravinder N, and Romain P, Pathogenesis of rheumatoid arthritis. 2017, UpToDate.
- 3.group D, The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N. Engl. J. Med, 1993. 329(14): p. 977–986. [DOI] [PubMed] [Google Scholar]
- 4.Group, E.o.D.I.a.C.E.R., Epidemiology of Diabetes Interventions and Complications (EDIC). Design, implementation, and preliminary results of a long-term follow- up of the Diabetes Control and Complications Trial cohort. Diabetes Care, 1999. 22(1): p. 99–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Secrest AM, Washington RE, and Orchard TJ, Mortality in Type 1 DIabetes, in Diabetes in America, 3rd Edition 2016, National Institutes of Health; p. 1–16. [Google Scholar]
- 6.Miller KM, et al. , Current State of Type 1 Diabetes Treatment in the U.S.: Updated Data From the T1D Exchange Clinic Registry. Diabetes Care, 2015. 38(6): p. 971–978. [DOI] [PubMed] [Google Scholar]
- 7.Eisenbarth GS, Type I diabetes mellitus. A chronic autoimmune disease. N. Engl. J. Med, 1986. 314(21): p. 1360–1368. [DOI] [PubMed] [Google Scholar]
- 8.Sosenko JM, et al. , Glucose and C-peptide changes in the perionset period of type 1 diabetes in the Diabetes Prevention Trial-Type 1. Diabetes Care, 2008. 31(11): p. 2188–2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Greenbaum CJ, et al. , Fall in C-peptide during first 2 years from diagnosis: evidence of at least two distinct phases from composite Type 1 Diabetes TrialNet data. Diabetes, 2012. 61(8): p. 2066–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Krogvold L, et al. , Function of Isolated Pancreatic Islets From Patients at Onset of Type 1 Diabetes: Insulin Secretion Can Be Restored After Some Days in a Nondiabetogenic Environment In Vitro: Results From the DiViD Study. Diabetes, 2015. 64(7): p. 2506–12. [DOI] [PubMed] [Google Scholar]
- 11.Keenan HA, et al. , Residual insulin production and pancreatic ss-cell turnover after 50 years of diabetes: Joslin Medalist Study. Diabetes, 2010. 59(11): p. 2846–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Atkinson M and Gianani R, The pancreas in human type 1 diabetes: providing new answers to age-old questions. Curr Opin Endocrinol Diabetes Obes, 2009. 16: p. 279–285. [DOI] [PubMed] [Google Scholar]
- 13.Pugliese A, Y. M, Kusmarteva I, The Juvenile Diabetes Research Foundation Network for Pancreatic Organ Donors with Diabetes (nPOD) Program: goals, operational model and emerging findings. Pediatr Diabetes, 2014. February 15 (1): p. 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Davis AK, et al. , Prevalence of detectable C-Peptide according to age at diagnosis and duration of type 1 diabetes. Diabetes Care, 2015. 38(3): p. 476–81. [DOI] [PubMed] [Google Scholar]
- 15.Steffes MW, et al. , Beta-cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care, 2003. 26(3): p. 832–836. [DOI] [PubMed] [Google Scholar]
- 16.Lachin JM, et al. , Impact of C-peptide preservation on metabolic and clinical outcomes in the Diabetes Control and Complications Trial. Diabetes, 2014. 63(2): p. 739–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hirshberg B, et al. , Benefits and risks of solitary islet transplantation for type 1 diabetes using steroid-sparing immunosuppression: the National Institutes of Health experience. Diabetes Care, 2003. 26(12): p. 3288–95. [DOI] [PubMed] [Google Scholar]
- 18.Leitão C, et al. , Restoration of hypoglycemia awareness after islet transplantation. Diabetes Care, 2008. 31(11): p. 2113–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mahon JL, et al. , The TrialNet Natural History Study of the Development of Type 1 Diabetes: objectives, design, and initial results. Pediatr. Diabetes, 2008. [DOI] [PubMed] [Google Scholar]
- 20.Redondo MJ, et al. , Heterogeneity of type I diabetes: Analysis of monozygotic twins in Great Britain and the United States. Diabetologia, 2001. 44: p. 354–362. [DOI] [PubMed] [Google Scholar]
- 21.Noble J and Erlich H, Genetics of type 1 diabetes. Cold Spring Harb Perspect Med, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Noble JA, V. AM, Cook M, Klitz W, Thomson G, and Erlich HA, The role of HLA class II genes in insulin-dependent diabetes mellitus: molecular analysis of 180 Caucasian, multiplex families. American Journal of Human Genetics, 1996. 59(5): p. 1134. [PMC free article] [PubMed] [Google Scholar]
- 23.Pociot F, et al. , Genetics of type 1 diabetes: What’s next? Diabetes, 2010. 59: p. 1561–1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pugliese A, et al. , HLA-DRB1*15:01-DQA1*01:02-DQB1*06:02 Haplotype Protects Autoantibody-Positive Relatives From Type 1 Diabetes Throughout the Stages of Disease Progression. Diabetes, 2016. 65(4): p. 1109–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jerram S and Leslie RD, The genetic architecture of type 1 diabetes. Genes, 2017. 8(8). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ikegami H, et al. , Genetics of type 1 diabetes: Similarities and differences between Asian and Caucasian populations. Ann. N. Y. Acad. Sci, 2006. 1079: p. 51–59. [DOI] [PubMed] [Google Scholar]
- 27.Hagopian WA, et al. , Glutamiate decarboxylase, insulin, and islet cell antibodies and HLA typing to detect diabetes in a general population based study of Swedish children. Journal of Clinical Investigation, 1995. 95(4): p. 1505–1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rewers M, et al. , Newborn screening for HLA markers associated with IDDM: diabetes autoimmunity study in the young (DAISY). Diabetologia, 1996. 39(7): p. 807–12. [DOI] [PubMed] [Google Scholar]
- 29.Winkler C, et al. , A strategy for combining minor genetic susceptibility genes to improve prediction of disease in type 1 diabetes. Genes Immun, 2012. 13: p. 549–555. [DOI] [PubMed] [Google Scholar]
- 30.Achenbach P, et al. , Natural history of type 1 diabetes. Diabetes, 2005. 54 Suppl 2: p. S25–S31. [DOI] [PubMed] [Google Scholar]
- 31.Wherrett DK, et al. , Defining pathways for development of disease-modifying therapies in children with type 1 diabetes: a consensus report. Diabetes Care, 2015. 38(10): p. 1975–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Concannon P, Rich SS, and Nepom GT, Genetics of type 1A diabetes. N. Engl. J. Med, 2009. 360(16): p. 1646–1654. [DOI] [PubMed] [Google Scholar]
- 33.Insel RA, et al. , Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care, 2015. 38(10): p. 1964–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Steck AK, et al. , Predictors of Progression From the Appearance of Islet Autoantibodies to Early Childhood Diabetes: The Environmental Determinants of Diabetes in the Young (TEDDY). Diabetes Care, 2015. 38(5): p. 808–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Achenbach P, et al. , Characteristics of rapid vs slow progression to type 1 diabetes in mulitple islet autoantibody-positive children. Diabetologia. 56: p. 1615–1622. [DOI] [PubMed] [Google Scholar]
- 36.Ziegler AG, et al. , Seroconversion to Multiple Islet Autoantibodies and Risk of Progression to Diabetes in Children. Journal of the American Medical Association, 2013. 309(23): p. 2473–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Orban T, et al. , Pancreatic islet autoantibodies as predictors of type 1 diabetes in the Diabetes Prevention Trial-Type 1. Diabetes Care, 2009. 32(12): p. 2269–2274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Parikka V, N.-S. K, Saarinen M, Simell T, Ilonen J, Hyöty H, Veijola R, Knip M, Simell O, Early seroconversion and rapidly increasing autoantibody concentrations predict prepubertal manifestation of type 1 diabetes in children at genetic risk. Diabetologia, 2012. 55(7): p. 1926–36. [DOI] [PubMed] [Google Scholar]
- 39.Krischer JP, et al. , The 6 year incidence of diabetes-associated autoantibodies in genetically at-risk children: the TEDDY study. Diabetologia, 2015. 58(5): p. 980–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bosi E, et al. , Impact of Age and Antibody Type on Progression From Single to Multiple Autoantibodies in Type 1 Diabetes Relatives. J Clin Endocrinol Metab, 2017. 102(8): p. 2881–2886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Krischer J, The use of intermediate endpoints in the design of type 1 diabetes prevention trials. Diabetologia, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Krischer JP, et al. , The Influence of Type 1 Diabetes Genetic Susceptibility Regions, Age, Sex, and Family History to the Progression from Multiple Autoantibodies to Type 1 Diabetes: A TEDDY Study Report. Diabetes, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Winkler C, et al. , Markedly reduced rate of diabetic ketoacidosis at onset of type 1 diabetes in relatives screened for islet autoantibodies. Pediatric Diabetes, 2012. 13(4): p. 308–313. [DOI] [PubMed] [Google Scholar]
- 44.Elding Larsson H, et al. , Reduced prevalence of diabetic ketoacidosis at diagnosis of type 1 diabetes in young children participating in longitudinal follow-up. Diabetes Care, 2011. 34(11): p. 2347–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Triolo T, et al. , Diabetic subjects diagnosed through the Diabetes Prevention Trial-Type 1 (DPT-1) are often asymptomatic with normal A1C at diabetes onset. Diabetes Care, 2009. 32: p. 769–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Haller MJ, Atkinson MA, and Schatz D, Type 1 diabetes mellitus: etiology, presentation, and management. Pediatr. Clin. North Am, 2005. 52(6): p. 1553–1578. [DOI] [PubMed] [Google Scholar]
- 47.American Diabetes Association, I., American Diabetes Association Standards of Medical Care in Diabetes - 2017. DIabetes Care, 2017. 40(Supplement 1): p. S1–S134.27979885 [Google Scholar]
- 48.Rewers A, et al. , Presence of diabetic ketoacidosis at diagnosis of diabetes mellitus in youth: the Search for Diabetes in Youth Study. Pediatrics, 2008. 121: p. e1258–e1266. [DOI] [PubMed] [Google Scholar]
- 49.Hagopian WA, et al. , The Environmental Determinants of Diabetes in the Young (TEDDY): genetic criteria and international diabetes risk screening of 421 000 infants. Pediatr Diabetes, 2011. 12(8): p. 733–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Neu A, et al. , Ketoacidosis at diabetes onset is still frequent in children and adolescents: a multicenter analysis of 14, 664 patients from 106 institutions. Diabetes Care, 2009. 32: p. 1647–1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Barker J, et al. , Clinical characteristics of children diagnosed with type 1 diabetes through intensive screening and follow-up Author information. Diabetes Care, 2004. 27(6): p. 1399–404. [DOI] [PubMed] [Google Scholar]
- 52.Insel RA, Dunne JL, and Ziegler AG, General population screening for type 1 diabetes: Has its time come? Current Opinion in Endocrinology, Diabetes, and Obesity, 2015. 22(4): p. 270–276. [DOI] [PubMed] [Google Scholar]
- 53.Raab J, et al. , Capillary blood islet autoantibody screening for identifying pre-type 1 diabetes in the general population: design and initial results of the Fr1da study. BMJ Open, 2016. 6(5): p. e011144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.zhao Z, et al. , A miltiplex assay combining insulin, GAD, IA-2, and transglutaminase autoantibodies to facilitate screening for pre-type 1 diabetes and celiac disease. J. Immunol Methods, 2016. 430: p. 28–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Prevention, C.f.D.C.a., National diabetes statistics report, 2017. 2017, Centers for Disease Control and Prevention, U.S. Dept. of Health and Human Services: Atlanta, GA. [Google Scholar]
- 56.Variation and trends in incidence of childhood diabetes in Europe. EURODIAB ACE Study Group. Lancet, 2000. 355(9207): p. 873–876. [PubMed] [Google Scholar]
- 57.Veijola R, et al. , HLA-DQB1-defined genetic susceptibility, beta cell autoimmunity, and metabolic characteristics in familial and nonfamilial insulin-dependent diabetes mellitus. Childhood Diabetes in Finland (DiMe) Study Group. Vol. 98 1996. 2489–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Diabetes, B.D.C.f. ASK Research Program/Autoimmunity Screening for Kids. 2018. 3/16/2018]; Available from: https://www.askhealth.org/.
- 59.GPPAD: Global platform for the prevention of autoimmune diabetes. [cited 2018 March 28]; Available from: https://www.gppad.org/en/.
- 60.Sosenko J, et al. , The prediction of type 1 diabetes by multiple autoantibody levels and their incorporation into an autoantibody risk score in relatives of type 1 diabetic patients. Diabetes Care, 2013. 36(9): p. 2615–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Type 1 Diabetes Trial, N., Type 1 diabetes: Stage 3. 2018.
- 62.Ziegler AG, et al. , Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. JAMA, 2013. 309(23): p. 2473–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chimel R, et al. , Progression from single to multiple islet autoantibodies ofter occurs soon after seroconversion: Implications for early screening. Diabetologia, 2015. 58: p. 411–413. [DOI] [PubMed] [Google Scholar]
- 64.Harjutsalo V, Podar T, and Tuomilehto J, Cumulative incidence of type 1 diabetes in 10,168 siblings of Finnish young-onset type 1 diabetic patients. Diabetes, 2005. 54(2): p. 563–569. [DOI] [PubMed] [Google Scholar]
- 65.Hagopian WA, et al. , TEDDY--The Environmental Determinants of Diabetes in the Young: an observational clinical trial. Ann N Y Acad Sci, 2006. 1079: p. 320–6. [DOI] [PubMed] [Google Scholar]
- 66.Mrena S, et al. , Models for predicting type 1 diabetes in siblings of affected children. Diabetes Care, 2006. 29(3): p. 662–7. [DOI] [PubMed] [Google Scholar]
- 67.Steck A, Johnson K, and Barriga K, Age of islet autoantibody appearance and mean levels of insulin, but not GAD or IA-2 autoantibodies, predict age of diagnosis of type 1 diabetes: Diabetes Autoimmunity Study in the Young. Diabetes Care, 2011. 34: p. 1397–1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bougneres PF, et al. , Factors associated with early remission of type I diabetes in children treated with cyclosporine. N Engl J Med, 1988. 318(11): p. 663–70. [DOI] [PubMed] [Google Scholar]
- 69.Vanbuecken D, Lord S, and Greenbaum C, Changing the course of disease in type 1 diabetes, in Endotext. 2015, South Dartmouth (MA): MDText.com, Inc. [Google Scholar]
- 70.Ridker P, E. B, Thuren T, Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N Engl J Med, 2017. September 21 377 (12): p. 1119–1131. [DOI] [PubMed] [Google Scholar]
- 71.Moran A, et al. , Interleukin-1 antagonism in type 1 diabetes of recent onset: two multicentre, randomised, double-blind, placebo-controlled trials. Lancet, 2013. 381(9881): p. 1905–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mastrandrea L, et al. , Etanercept treatment in children with new-onset type 1 diabetes: pilot randomized, placebo-controlled, double-blind study. Diabetes Care, 2009. 32(7): p. 1244–1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Clinicaltrials.gov, A Study to Evaluate SIMPONI (Golimumab) Therapy in Children, Adolescents and Young Adults With Pre-Symptomatic Type 1 Diabetes. Last updated March 1, 2018. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT03298542.
- 74.Clinicaltrials.gov, Study of SIMPONI® to Arrest Beta-cell Loss in Type 1 Diabetes (T1GER); NCT02846545. 2017.
- 75.ClinicalTrials.gov, Pilot Clinical Trial of Ustekinumab in Patients With New-onset T1D (USTID). Last updated May 25, 2016. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02117765.
- 76.Hundhausen C, et al. , Enhanced T cell responses to IL-6 in type 1 diabetes are associated with early clinical disease and increased IL-6 receptor expression. Sci Transl Med, 2016. 8(356): p. 356ra119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Clinicaltrials.gov, Tocilizumab (TCZ) in New-onset Type 1 Diabetes. 2017.
- 78.Lisowska-Myjak B, P. J, Kaczynska B, Miszkurka G, Kadziela K. Serum protease inhibitor concentrations and total antitrypsin activity in diabetic and non-diabetic children during adolescence. Acta Diabetol, 2006. 43(4): p. 88–92. [DOI] [PubMed] [Google Scholar]
- 79.Gottlieb PA, A. A, Michels AW α1−Antitrypsin therapy downregulates toll-like receptor-induced IL-1β responses in monocytes and myeloid dendritic cells and may improve islet function in recently diagnosed patients with type 1 diabetes. J Clin Endocrinol Metab 2014. 99: p. E1418–E1426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Clinicaltrials.gov, Study of the Safety and Efficacy of Intravenous Alpha-1 Antitrypsin in Type 1 Diabetes Mellitus. Last updated June 9, 2016. Last accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT01304537.
- 81.Clinicaltrials.gov, The Effects of Alpha-1 Antitrypsin (AAT) on the Progression of Type 1 Diabetes. Last updated March 24, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT01319331.
- 82.Louvet C1, S. G, Lang J, Lee MR, Martinier N, Bollag G, Zhu S, Weiss A, Bluestone JA, Tyrosine kinase inhibitors reverse type 1 diabetes in nonobese diabetic mice. Proc Natl Acad Sci U S A, 2008. 105(48): p. 18895–900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Clinicaltrials.gov, Imatinib Treatment in Recent Onset Type 1 Diabetes Mellitus. Last updated June 21, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT01781975.
- 84.Gitelman S, Imatinib (Gleevec) in New-Onset Type 1 Diabetes—Background and Results. Accessed March 23, 2018 https://professional.diabetes.org/webcast/imatinib-gleevec-new-onset-type-1-diabetes%E2%80%94background-and-results Diabetes, 2017. 66 (suppl 1).
- 85.Du Toit G1, R. G, Sayre PH, Bahnson HT, Radulovic S, Santos AF, Brough HA, Phippard D, Basting M, Feeney M, Turcanu V, Sever ML, Gomez Lorenzo M, Plaut M, Lack G; LEAP Study Team., Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015. 372(9): p. 803–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pescovitz MD, et al. , Rituximab, B-Lymphocyte Depletion, and Preservation of Beta-Cell Function. N. Engl. J. Med, 2009. 361(22): p. 2143–2152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ostrov DA, A. A, McDaniel KA, Case S, Baschal EE, Pyle L, Ellis S, Pöllinger B, Seidl KJ, Shah VN, Garg SK, Atkinson MA, Gottlieb PA, Michels AW, Methyldopa blocks MHC class II binding to disease-specific antigens in autoimmune diabetes. J Clin Invest, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Clinicaltrials.gov, Effect of Methyldopa on MHC Class II Antigen Presentation in Type 1 Diabetes. Last updated April 6, 2016. Accessed March 23, 2018 https://clinicaltrials.gov/ct2/show/NCT01883804.
- 89.Clinicaltrials.gov, Methyldopa for Reduction of DQ8 Antigen Presentation in At-Risk Subjects for Type 1 Diabetes. Last updated March 2, 2018. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT03396484.
- 90.Orban T, et al. , Co-stimulation modulation with abatacept in patients with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled trial. Lancet, 2011. 378(9789): p. 412–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Clinicaltrials.gov. CTLA4-Ig (Abatacept)for Prevention of Abnormal Glucose Tolerance and Diabetes in Relatives At -Risk for Type 1; NCT01773707. 2017.
- 92.Herold KC, et al. , Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N. Engl. J. Med, 2002. 346(22): p. 1692–1698. [DOI] [PubMed] [Google Scholar]
- 93.Herold KC, et al. , Teplizumab (anti-CD3 mAb) treatment preserves C-peptide responses in patients with new-onset type 1 diabetes in a randomized controlled trial: metabolic and immunologic features at baseline identify a subgroup of responders. Diabetes, 2013. 62(11): p. 3766–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.(NIDDK), N.I.o.D.a.D.a.K.D., Teplizumab for Prevention of Type 1 Diabetes In Relatives “At-Risk” In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US) 2000- [cited 2017 March 31]. Available from: http://clinicaltrialsgov/show/NCT01030861 NLM Identifier: NCT01030861. [Google Scholar]
- 95.Rigby MR, et al. , Targeting of memory T cells with alefacept in new-onset type 1 diabetes (T1DAL study): 12 month results of a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Diabetes Endocrinol, 2013. 1(4): p. 284–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Rigby MR, et al. , Alefacept provides sustained clinical and immunological effects in new-onset type 1 diabetes patients. J Clin Invest, 2015. 125(8): p. 3285–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Haller MJ, et al. , Anti-thymocyte globulin/G-CSF treatment preserves beta cell function in patients with established type 1 diabetes. J Clin Invest, 2015. 125(1): p. 448–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Haller MJ, et al. , Low-Dose Anti-Thymocyte Globulin (ATG) Preserves beta-Cell Function and Improves HbA1c in New-Onset Type 1 Diabetes. Diabetes Care, 2018. 41(9): p. 1917–1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Long SA, et al. , Rapamycin/IL-2 combination therapy in patients with type 1 diabetes augments Tregs yet transiently impairs beta-cell function. Diabetes, 2012. 61(9): p. 2340–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Clinicaltrials.gov, Low-dose rhIL-2 in Patients With Recently-diagnosed Type 1 Diabetes (DIABIL-2). Last updated Nov 9, 2016. Accessed March 20, 2016 https://clinicaltrials.gov/ct2/show/NCT02411253.
- 101.Bluestone J, B. J, Fitch M, et al. , Type 1 diabetes immunotherapy using polyclonal regulatory T cells Sci Transl Med, 2015. 7(315). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.clinicaltrials.gov, T1DM Immunotherapy Using CD4+CD127lo/-CD25+ Polyclonal Tregs (Treg). Last updated May 20, 2016. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT01210664.
- 103.Clinicaltrials.gov, T1DM Immunotherapy Using Polyclonal Tregs + IL-2 (TILT), in In: ClinicalTrialsgov [Internet] Bethesda (MD): National Library of Medicine (US) 2000- [cited 2017 April 13] Available from: http://clinicaltrials.gov/show/NCT02772679 NLM Identifier: NCT02772679. [Google Scholar]
- 104.Libman IM, M. K, DiMeglio LA, et al. , Effect of Metformin Added to Insulin on Glycemic Control Among Overweight/Obese Adolescents With Type 1 Diabetes: A Randomized Clinical Trial. JAMA, 2015. 314(21): p. 2241–50. [DOI] [PubMed] [Google Scholar]
- 105.Clinicaltrials.gov, Autoimmune Diabetes Accelerator Prevention Trial. Last updated Aug 29, 2016. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02881528.
- 106.Kielgast U1, A. M, Madsbad S, Holst JJ, Effect of glucagon-like peptide-1 on alpha- and beta-cell function in C-peptide-negative type 1 diabetic patients. J Clin Endocrinol Metab. 2010. May 95((5)). [DOI] [PubMed] [Google Scholar]
- 107.Htike Z, Z. F, Papamargaritis D, et al. , Webb DR1,2, Khunti K1,2, Davies MJ1,2. Diabetes Obes Metab. 2017 Apr;19(4):524–536), Efficacy and safety of glucagon-like peptide-1 receptor agonists in type 2 diabetes: A systematic review and mixed-treatment comparison analysis. Diabetes Obes Metab, 2017. April 19(4): p. 524–536. [DOI] [PubMed] [Google Scholar]
- 108.Zhang J1, T. Y, Yamagata K1, Kozawa J1, Sayama K1, Iwahashi H1, Okita K1, Miuchi M2, Konya H2, Hamaguchi T2, Namba M2, Shimomura I1, Miyagawa JI3, Continuous stimulation of human glucagon-like peptide-1 (7–36) amide in a mouse model (NOD) delays onset of autoimmune type 1 diabetes. Diabetologia, 2007. 50(9): p. 1900–1909. [DOI] [PubMed] [Google Scholar]
- 109.Rother KI, et al. , Effects of exenatide alone and in combination with daclizumab on beta-cell function in long-standing type 1 diabetes. Diabetes Care, 2009. 32(12): p. 2251–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Clinicaltrials.gov, Liraglutide Effect on Beta-cell Function in C-peptide Positive Type 1 Diabetes. Last updated Oct 27, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02617654.
- 111.Clinicaltrials.gov, A Clinical Proof-of-principle Trial in Adult Subjects With Newly Diagnosed Type 1 Diabetes Mellitus Investigating the Effect of NNC0114–0006 and Liraglutide on Preservation of Beta-cell Function. Last updated March 9, 2018. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02443155.
- 112.Clinicaltrials.gov, Incretin-based Therapy in Early Diagnosed Type 1 Diabetes. Last updated Oct 26, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02908087.
- 113.Clinicaltrials.gov, Incretin-based Therapy in Late Preclinical Type 1 Diabetes. Last updated Oct 26, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02898506.
- 114.Clinicaltrials.gov, Incretin-based Therapy in Preclinical Type 1 Diabetes in Adults. Last updated Oct 26, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02611232.
- 115.Rooman I, L. J, Bouwens L, Gastrin stimulates β-cell neogenesis and increases islet mass from transdifferentiated but not from normal exocrine pancreas tissue. Diabetes, 2002. 51: p. 686–690. [DOI] [PubMed] [Google Scholar]
- 116.Inci F, A. M, Ozturk M, et al. , Pantoprazole may improve beta cell function and diabetes mellitus. J Endocrinol Invest, 2014. May 37(5): p. 449–54. [DOI] [PubMed] [Google Scholar]
- 117.Han N, O. M, Park SM, et al. , The effect of proton pump inhibitors on glycated hemoglobin levels in patients with type 2 diabetes mellitus. Can J Diabetes, 2015. February 39(1): p. 24–8. [DOI] [PubMed] [Google Scholar]
- 118.Hove KD, B. C, Færch K, et al. , Effects of 12 weeks’ treatment with a proton pump inhibitor on insulin secretion, glucose metabolism and markers of cardiovascular risk in patients with type 2 diabetes: a randomised double-blind prospective placebo-controlled study. Diabetologia, 2013. January 56(1): p. 22–30. [DOI] [PubMed] [Google Scholar]
- 119.Wasko MC, M. C, Kelsey SF, et al. , Antidiabetogenic effects of hydroxychloroquine on insulin sensitivity and beta cell function: a randomised trial. Diabetologia, 2015. October 58(10): p. 2336–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Clinicaltrials.gov, Hydroxychloroquine in Individuals At-risk for Type 1 Diabetes Mellitus. Last updated Feb 12, 2018. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT03428945.
- 121.Nathan DM, B. M, Cleary P, et al. for the DCCT/EDIC Research Group., The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study at 30 Years: Advances and Contributions. Diabetes, 2013. 62: p. 3976–3986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Lachin JM, G. S, Cleary P, Nathan DM. for the EDIC Research Group, Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. N Engl J Med, 2000. 342: p. 381–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Group., E.R., Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA, 2003. 290: p. 2159–2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Knip M, et al. , Hydrolyzed infant formula and early beta-cell autoimmunity: a randomized clinical trial. JAMA, 2014. 311(22): p. 2279–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Knip M, Writing group for teh TRIGR Study Group, Effect of Hydrolyzed Infant Formula vs Conventional Formula on Risk of Type 1 Diabetes. JAMA, 2018. January 319(1): p. 38–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Norris JM, et al. , Timing of initial cereal exposure in infancy and risk of islet autoimmunity. JAMA, 2003. 290(13): p. 1713–1720. [DOI] [PubMed] [Google Scholar]
- 127.Hummel S, P. M, Hummel M, Primary dietary intervention study to reduce the risk of islet autoimmunity in children at increased risk for type 1 diabetes: the BABYDIET study. Diabetes Care, 2011. 34(6): p. 1302–1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Gale EA, et al. , European Nicotinamide Diabetes Intervention Trial (ENDIT): a randomised controlled trial of intervention before the onset of type 1 diabetes. Lancet, 2004. 363(9413): p. 925–931. [DOI] [PubMed] [Google Scholar]
- 129.Gibson V, N. T, Pearce V, et al. , Proinsulin multi-peptide immunotherapy induces antigen-specific regulatory T cells and limits autoimmunity in a humanized model. Clin Exp Immunol. 2015. December 182(3): p. 251–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ludvigsson J, et al. , GAD65 antigen therapy in recently diagnosed type 1 diabetes mellitus. N Engl J Med, 2012. 366(5): p. 433–42. [DOI] [PubMed] [Google Scholar]
- 131.Wherrett DK, et al. , Antigen-based therapy with glutamic acid decarboxylase (GAD) vaccine in patients with recent-onset type 1 diabetes: a randomised double-blind trial. Lancet, 2011. 378(9788): p. 319–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Alhadj Ali M, L. Y, Arif S, et al. , Metabolic and immune effects of immunotherapy with proinsulin peptide in human new-onset type 1 diabetes. Sci Transl Med, 2017. 9(402). [DOI] [PubMed] [Google Scholar]
- 133.Nanto-Salonen K, et al. , Nasal insulin to prevent type 1 diabetes in children with HLA genotypes and autoantibodies conferring increased risk of disease: a double-blind, randomised controlled trial. Lancet, 2008. 372(9651): p. 1746–55. [DOI] [PubMed] [Google Scholar]
- 134.Group, D.P.T.S., Effects of insulin in relatives of patients with type 1 diabetes mellitus. N. Engl. J. Med, 2002. 346(22): p. 1685–1691. [DOI] [PubMed] [Google Scholar]
- 135.Skyler JS, K. J, Wolfsdorf J et al. , Effects of oral insulin in relatives of patients with type 1 diabetes: The Diabetes Prevention Trial--Type 1. Diabetes Care, 2005. 28(5). [DOI] [PubMed] [Google Scholar]
- 136.Skyler J, Update on worldwide efforts to prevent type 1 diabetes. Ann N Y Acad Sci, 2008. 1150: p. 190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Writing Committee for the Type 1 Diabetes TrialNet Oral Insulin Study, G., et al. , Effect of Oral Insulin on Prevention of Diabetes in Relatives of Patients With Type 1 Diabetes: A Randomized Clinical Trial. JAMA, 2017. 318(19): p. 1891–1902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Bonifacio E, et al. , Effects of high-dose oral insulin on immune responses in children at high risk for type 1 diabetes: the Pre-POINT randomized clinical trial. JAMA, 2015. 313(15): p. 1541–9. [DOI] [PubMed] [Google Scholar]
- 139.Clinicaltrials.gov, Pre-POINT-Early Study: NCT02547519.
- 140.Clinicaltrials.gov, Immune Effects of Oral Insulin in Relatives at Risk for Type 1 Diabetes Mellitus, in In: ClinicalTrialsgov [Internet] Bethesda (MD): National Library of Medicine (US) 2000- [cited 2018 Nov 26] Available from: http://clinicaltrials.gov/show/NCT02580877 NLM Identifier: NCT02580877. [Google Scholar]
- 141.Clinicaltrials.gov, Trial of Intranasal Insulin in Children and Young Adults at Risk of Type 1 Diabetes. Last updated Oct 7, 2016. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT00336674.
- 142.Clinicaltrials.gov, Prevention Trial: Immune-tolerance With Alum-GAD (Diamyd) and Vitamin D3 to Children With Multiple Islet Autoantibodies. Last updated August 24, 2017. Accessed March 20, 2018 https://clinicaltrials.gov/ct2/show/NCT02387164.