Abstract
Background
The identification of patients with advanced liver fibrosis secondary to non-alcoholic fatty liver disease (NAFLD) remains challenging. Using non-invasive liver fibrosis tests (NILT) in primary care may permit earlier detection of patients with clinically significant disease for specialist review, and reduce unnecessary referral of patients with mild disease. We constructed an analytical model to assess the clinical and cost differentials of such strategies.
Methods
A probabilistic decisional model simulated a cohort of 1000 NAFLD patients over 1 year from a healthcare payer perspective. Simulations compared standard care (SC) (scenario 1) to: Scenario 2: FIB-4 for all patients followed by Enhanced Liver Fibrosis (ELF) test for patients with indeterminate FIB-4 results; Scenario 3: FIB-4 followed by fibroscan for indeterminate FIB-4; Scenario 4: ELF alone; and Scenario 5: fibroscan alone. Model estimates were derived from the published literature. The primary outcome was cost per case of advanced fibrosis detected.
Results
Introduction of NILT increased detection of advanced fibrosis over 1 year by 114, 118, 129 and 137% compared to SC in scenarios 2, 3, 4 and 5 respectively with reduction in unnecessary referrals by 85, 78, 71 and 42% respectively.
The cost per case of advanced fibrosis (METAVIR ≥F3) detected was £25,543, £8932, £9083, £9487 and £10,351 in scenarios 1, 2, 3, 4 and 5 respectively. Total budget spend was reduced by 25.2, 22.7, 15.1 and 4.0% in Scenarios 2, 3, 4 and 5 compared to £670 K at baseline.
Conclusion
Our analyses suggest that the use of NILT in primary care can increases early detection of advanced liver fibrosis and reduce unnecessary referral of patients with mild disease and is cost efficient. Adopting a two-tier approach improves resource utilization.
Keywords: Enhanced Liver fibrosis (ELF), Fibroscan, |NAFLD, Cirrhosis detection, Cost savings
Background
The health, societal and economic burden of chronic liver disease (CLD) is substantial and represents a public health priority [1, 2]. CLD is the 5th commonest cause of death in the United Kingdom, and the only one in the top five that is increasing [3]. With rising prevalence of risk factors for liver disease including obesity and alcohol, pressure on healthcare resources is likely to intensify. Better and earlier detection of CLD in primary care is key to improving health outcomes and associated costs [4].
Non alcoholic fatty liver disease (NAFLD) is the commonest cause of deranged liver function tests (LFTs) in primary care [5]. Only a minority (5%) of these cases progresses to clinically significant liver disease [6] whilst evidence highlights fibrosis severity as the key determinant of liver related morbidity and mortality [7, 8]. The identification of patients with significant liver disease is a primary care challenge, where accurate fibrosis assessment is limited by a reliance on LFTs, which are poor discriminators of liver fibrosis [9].
In the current ‘standard care’ (SC), primary care physicians (PCP) assess the severity of a patient’s liver disease and subsequent need for specialist referral based on history, examination, blood tests including LFTs and ultrasound.
Patients referred to secondary care deemed to have significant liver disease due to NAFLD may benefit from active management (including consideration for clinical trials of emerging therapies for NAFLD and fibrosis) [2, 10]. Patients with cirrhosis will be enrolled in pathways of care that improve outcomes through targeted screening and treatment for complications of cirrhosis including portal hypertension [11] and hepatocellular carcinoma (HCC) [12]. However, current primary care approaches result in the referral of many patients that do not have significant liver fibrosis, placing a burden on secondary care services, incurring unwarranted costs and generating unnecessary inconvenience and anxiety for patients [13]. Furthermore a significant number of patients with advanced liver fibrosis are missed and not referred to secondary care. These patients have been falsely reassured and will silently progress before presenting with end-stage liver disease or liver cancer.
The use of non-invasive liver fibrosis tests (NILT) [14] may improve PCP staging of disease [4, 15] and referral practice but there is a lack of health-economic evidence about the use of NILT in fatty liver disease to inform clinicians, commissioners and policy makers about the value of such strategies. In this study, we developed a probabilistic decision analytical model to investigate the clinical and cost impact of primary care risk stratification of patients with NAFLD.
Methods
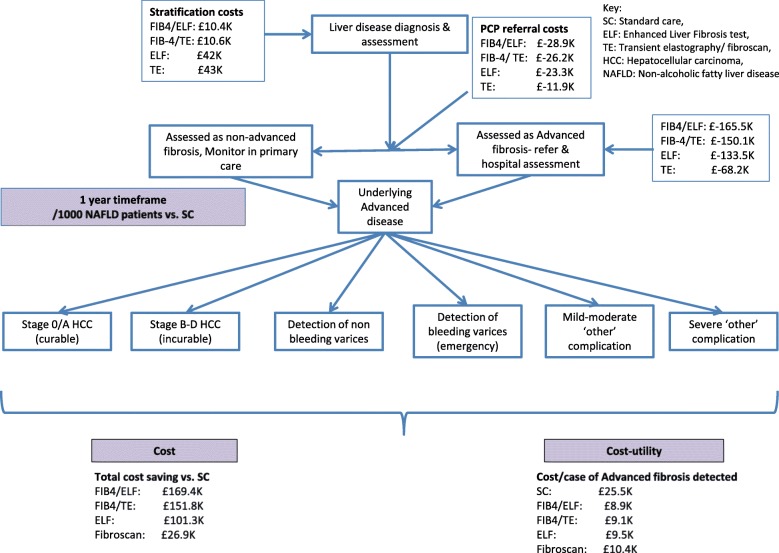
A liver working group, comprising primary care physicians and secondary care liver specialists, commissioners, public health practitioners and patient representatives was formed in the London Boroughs of Camden and Islington to develop new pathways of care for patients with NAFLD. Part of the strategy was to establish current practice (standard care (SC)) (Fig. 1) and to establish pragmatic guidance on how to improve the identification of NAFLD cases with advanced liver disease for referral to secondary care. In the first instance, PCP favoured the selection of patients with deranged liver function tests (LFT) even though it was agreed that this would miss a minority of cases with liver fibrosis who have normal LFTs.
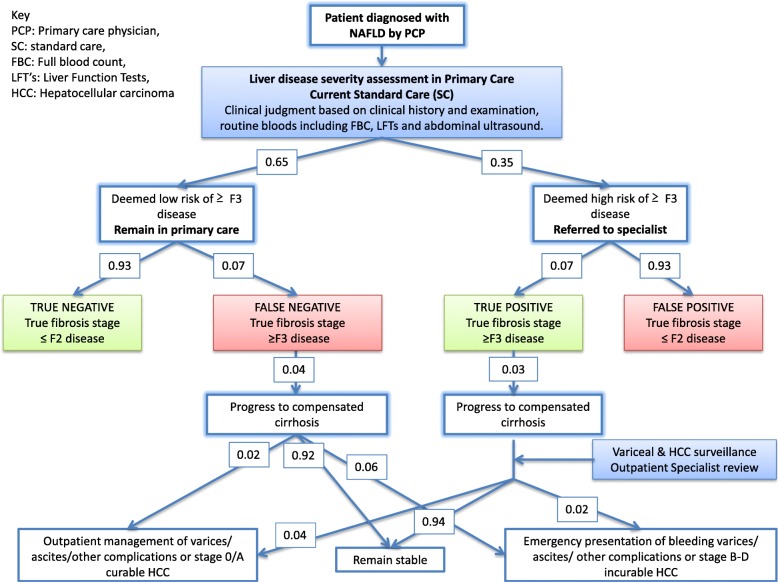
Fig. 1.
Schematic simulating current ‘standard of care’ patient journey. Simplified simulated journey of a patient with NAFLD through the healthcare system after primary care assessment using standard of care over a 1-year timeframe (see Table 1 and Table 2 for references). The diagnostic performance of the primary care assessment has four outcomes - 1) ‘True positive’; Patients deemed to be at high risk for advanced fibrosis subsequently confirmed as having ≥ F3 fibrosis after specialist assessment. Patients will be actively managed in secondary care (including consideration for clinical trials). Patients with cirrhosis will be enrolled in pathways of care that improve outcomes through targeted screening and treatment for portal hypertension and hepatocellular carcinoma (HCC). 2) ‘True negative’; Patients deemed to be at low risk for advanced fibrosis found to have ≤ F2 disease. These patients are unlikely to suffer morbidity from their liver disease. Management in primary care should be focussed on managing reversible metabolic disorders. 3) ‘False positive’; Patients deemed to be at high risk for advanced fibrosis in primary care but found to have ≤ F2 fibrosis. The pathway can be considered to have failed this group of patients, whom can be managed effectively in primary care with weight loss and exercise. 4) ‘False negative’; Patients deemed to be at low risk for advanced fibrosis who have ≥ F3 fibrosis. This cohort of patients have been falsely reassured and represent a failure of the pathway as they remain in primary care unless they present with complications of CLD if their disease progresses, at which point interventions are increasingly limited
Probabilistic decision model
A probabilistic decision analytical simulation model was created using Microsoft Excel Software (version16.23, 2019). The model piloted competing primary care risk stratification diagnostic strategies for 1000 patients with a confirmed diagnosis of NAFLD (Fig. 2). The average patient was 50 years old with elevated transaminases. The cycle length was 1 year.
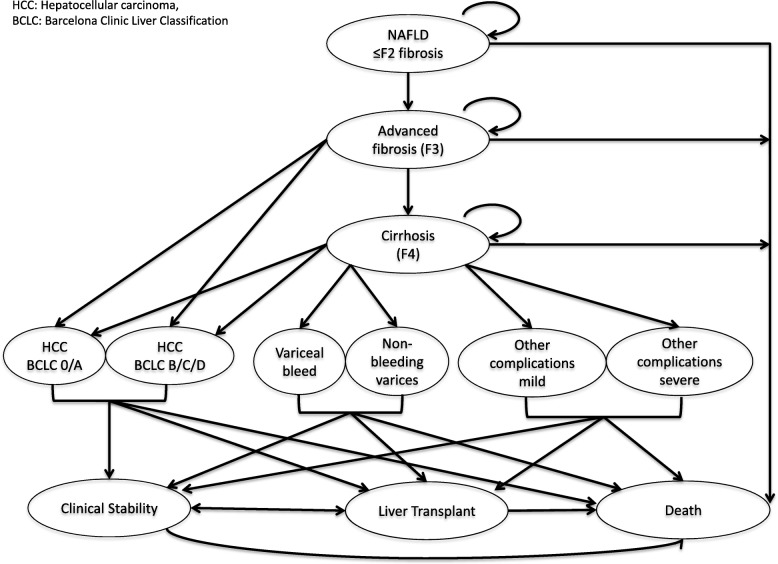
Fig. 2.
Decision tree presenting overview of transition of a patient with NAFLD through the model. In the model, a patient with NAFLD and ≤ F2 fibrosis could remain well, progress to F3 fibrosis or die. A patient with F3 fibrosis could remain well, progress to compensated cirrhosis, develop HCC or die. Patients with compensated cirrhosis could remain stable, develop a complication of cirrhosis, undergo liver transplantation or die. The model differentiated early stage complications (non-bleeding varices detected by surveillance endoscopy, Barcelona Clinic Liver Cancer (BCLC) stage 0/A HCC and mild/moderate ‘other’ complication including ascites, jaundice and hepatic encephalopathy managed as outpatient), from late stage complications (bleeding varices, BCLC stage B-D HCC and severe ‘other’ CLD complications necessitating inpatient admission)
Competing Strategies in the Model and Analyses
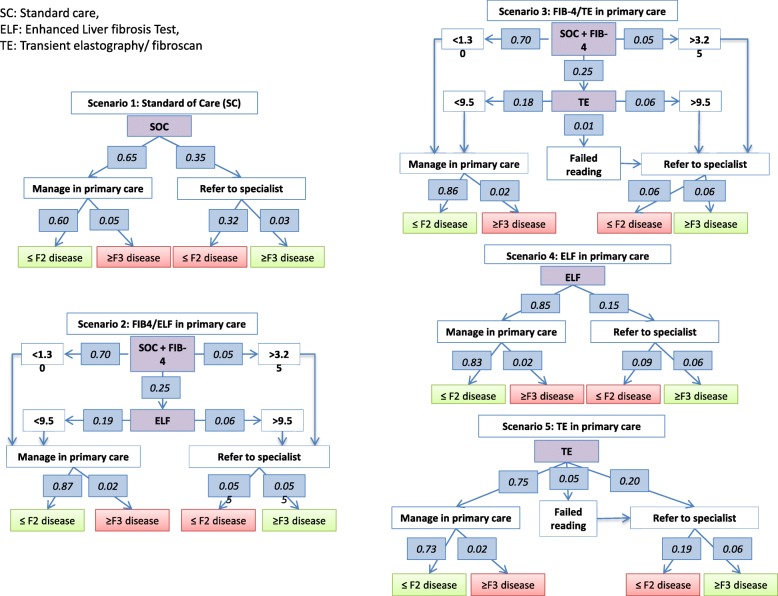
We modelled the standard care in the UK National Health Service (NHS) (scenario 1). The use of FIB-4 and ELF in a two-tier stratification approach (scenario 2) was modelled to replicate a local pilot pathway - the Camden and Islington NAFLD pathway [13]. Following an independent evaluation of NILT public health consultants favoured the use of FIB-4 over the NAFLD Fibrosis Score, in part due to a lack of standardization in the diagnosis of diabetes. Fibroscan is increasingly established in secondary care practice, and was incorporated to assess its performance in place of ELF in a two-tier strategy (Scenario 3). One-tier approaches were also considered in which SC was supported by ELF (scenario 4), or fibroscan alone (Scenario 5).
In all scenarios SC delivered by PCPs included history, physical assessment followed by investigation of liver function, tests for viral, immune and metabolic causes of liver disease and an ultrasound scan in order to make an assessment of the risk of advanced liver fibrosis. This was classified as a binary outcome with the case deemed to be at ‘high risk’ of advanced liver disease necessitating referral to a specialist, or at ‘low risk’ and thus appropriate for management in primary care. This decision process required 3 PCP consultations, 3 routine bloods tests and 1 ultrasound scan. In scenarios 2 and 3, SC was supported by the calculation of a FIB-4 score in all patients to improve the identification of patients at risk of advanced fibrosis (METAVIR ≥ F3). Low risk patients (FIB-4 < 1.30) were managed in primary care whilst high-risk patients (FIB-4 > 3.25) were referred to a specialist in secondary care. Patients with indeterminate scores (FIB-4 1.30–3.25) required an ELF test (Scenario 2) or a community fibroscan (Scenario 3). Published cut-offs were used to identify cases at increased risk of advanced fibrosis (≥10.3 for ELF and ≥ 7.9 kPa for Fibroscan). A fibroscan failure rate of 5% was assumed [47]. For patients managed in Scenario 4, SC was followed by ELF test for all patients and in Scenario 5 SC was followed by fibroscan for all patients.
Patients identified as being at high-risk of advanced fibrosis were referred to a secondary care specialist. Evaluation included further blood tests, fibroscan, imaging including US scan (50% of cases, informed by local audit), CT scan (5% of cases, informed by local audit), MRI Liver (5%, informed by local audit) and liver biopsy (15% of cases, informed by local audit). Patients deemed to not have advanced fibrosis (false positive) would be discharged to primary care, whilst those confirmed with advanced fibrosis would enter recognised surveillance pathways.
The time horizon for the base-case was 1 year to assess short-term benefits, likely to relate to resource utilisation. A 5- year timeframe was applied to assess the longer-term implications.
Clinical data inputs
A comprehensive literature search informed model parameters. The data were critically assessed to ensure suitability for this study and were supplemented by expert opinion when required.
The model assumed an intention-to-diagnose strategy. All patients would be managed according to the pathways. SC performance is poorly documented, but estimates were extrapolated from available studies [13, 16, 17]. Advanced fibrosis (≥F3 fibrosis) prevalence was set at 7.5% [5] and published sensitivities and specificities of FIB-4 [18, 19], ELF [19, 20] and fibroscan [19, 21] were used to predict stratification rates for low- and high- risk of advanced fibrosis (Table 1 and Fig. 3). Estimates of NAFLD disease progression were used to inform pathway performance (Table 2).
Table 1.
Test performance and Disease prevalence estimates
| Test characteristics | Sensitivity | Specificity | Reference |
|---|---|---|---|
| Standard of care | 0.35 | 0.65 | Expert Opinion [16, 17] |
| FIB-4 (cut off 1.30) | 0.84 | 0.74 | [18, 19] |
| FIB-4 (cut off 3.25) | 0.38 | 0.97 | [18, 19] |
| ELF | 0.80 | 0.90 | [19, 20] |
| Fibroscan | 0.82 | 0.84 | [19, 21] |
| Population and disease characteristics | Transition probability | ||
| Prevalence of advanced fibrosis in the general population | 0.075 | [5] | |
Published test characteristics of non-invasive liver fibrosis tests to detect advanced fibrosis (METAVIR ≥F3) in patients with non-alcoholic fatty liver disease
Fig. 3.
Flow diagram depicting patient flow in each simulated scenario. Published test performances allowed prediction of true and false positive and negative rates for detection of advanced fibrosis (≥F3) in each scenario
Table 2.
Transitional probability estimates used to populate the probabilistic analytical model for the base case (annual progression rates)
| Parameter/ Health state | Transition probability | References | |
|---|---|---|---|
| Population and disease characteristics | |||
| Prevalence of advanced fibrosis in the general population | 0.075 | [5] | |
| Reduction in fibrosis progression after GP management | 0.01 | [22], Author assumption | |
| Reduction in fibrosis progression after specialist review | 0.025 | [22] | |
| Mild/ No fibrosis (F0, F1, F2) | |||
| Remain healthy | 0.99 | [23–26] | |
| Develop F3 disease | 0.001 | [23, 24] | |
| Mortality (all cause) | 0.005 | [25, 27, 28] | |
| Discharge from specialist services | 0.7 | Unpublished audit | |
| Advanced fibrosis (F3) | |||
| Remain healthy | 0.95 | [25, 29, 30] | |
| Develop F4 disease/ cirrhosis | 0.04 | [25, 29, 30] | |
| Develop HCC (without cirrhosis) | 0.004 | [29] | |
| Mortality (all cause) | 0.005 | [29] | |
| Compensated cirrhosis (F4) | |||
| Remain compensated | 0.93 | Calculated from other variables | |
| Develop varices | 0.03 | [31] | |
| Develop HCC | 0.003 | [32, 34–37] | |
| Develop other complications (inc. jaundice, ascites, HE) | 0.02 | [29, 31, 32] | |
| Mortality (all cause) | 0.02 | [31, 38, 39], expert opinion | |
| BCLC Stage 0 and A HCC | |||
| Cure (liver transplant) | 0.36 | [40] | |
| Cure (non transplant) | 0.39 | [40] | |
| Mortality (all cause) | 0.25 | [40] | |
| BCLC stage B – D HCC | |||
| Clinical stability (post TACE, RFA etc) | 0.24 | [41] | |
| Mortality (all cause) | 0.76 | [35] | |
| Varices detection in surveillance programme | |||
| Clinical stability | 0.92 | [42] | |
| Liver transplant | 0.01 | Expert opinion | |
| Mortality (all cause) | 0.07 | [33] | |
| Detection of varices after emergency presentation | |||
| Clinical stability | 0.73 | [43] | |
| Liver transplant | 0.02 | [42] | |
| Mortality (all cause) | 0.25 | [44] | |
| Mild/ Moderate ‘other’ complication | |||
| Clinical stability | 0.74 | [17] | |
| Liver transplant | 0.10 | [42] | |
| Mortality (all cause) | 0.16 | [17] | |
| Severe ‘other’ complication | |||
| Clinical stability | 0.45 | [17] | |
| Liver transplant | 0.10 | [42] | |
| Mortality (all cause) | 0.45 | [17] | |
| Severity of CLD Complication | No screening | Screening | |
| Probability of BCLC stage 0 + A HCC | 0.299 | 0.709 | [45] |
| Probability of BCLC stage B - D HCC | 0.701 | 0.291 | [45] |
| Detecting varices in surveillance programme | 0.0 | 0.60 | [42] |
| Detecting varices after emergency presentation | 100.0 | 0.40 | [42] |
| Mild/ moderate CLD ‘other’ complication | 0.527 | 0.622 | [17, 46] |
| Severe CLD ‘other’ complication | 0.473 | 0.378 | [17, 46] |
Cost data inputs
A healthcare payer perspective was adopted. Costs were derived from published resources and local costing tariffs (February 2015) (Table 3) for the UK. A 3.5% discount rate was applied. Direct healthcare costs included PCP consultations, blood tests and ultrasound scans. The cost of FIB-4 was considered to be £0, as ALT, AST and platelet tests were incorporated as ‘routine blood tests’. ELF was priced at £42, the quoted rate charged to the Camden and Islington CCG in their pathway, and fibroscan was priced at £43 [19]. In secondary care, the costs incurred related to specialist consultations, investigations including liver biopsy and expenditure related to HCC surveillance (6 monthly AFP and ultrasound) and variceal surveillance (2–3 yearly endoscopy). Finally, costs for the management of complications of CLD, including inpatient and outpatient costs, pharmacological treatment and surgical procedures including liver resection and transplant were obtained from the Royal Free London NHS Foundation Trust finance department.
Table 3.
Health care costs for patients with NAFLD/ NASH (£, 2014–2015)
| Resource Use | Unit Cost | Reference |
|---|---|---|
| Primary Care | ||
| GP consultation (per patient contact lasting 11.7 min) | £45.00 | [48] |
| Secondary care | ||
| Hepatology Consultant appointment (new) | £148.34 | Royal Free, February 2015 |
| Hepatology Consultant follow up appt | £98.63 | Royal Free, February 2015 |
| Dietician review | £57.00 | Royal Free, February 2015 |
| Investigations | ||
| Routine blood tests (inc. FBC, LFT’s, INR) | £68.06 | Royal Free, February 2015 |
| Liver aetiology panel | £147.98 | Royal Free, February 2015 |
| FIB-4 (AST/ALT/ platelets included in ‘routine blood tests’) | £0.00 | Royal Free, February 2015 |
| ELF | £42.00 | North Middlesex Hospital, February 2015, |
| Ultrasound Liver | £63.67 | Royal Free, February 2015 |
| CT Abdomen/ Liver | £80.78 | Royal Free, February 2015 |
| MRI Abdomen/ Liver | £101.00 | Royal Free, February 2015 |
| Fibroscan | £43.00 | Royal Free, February 2015 |
| Liver biopsy | £642.75 | Royal Free, February 2015 |
| Endoscopy | £264.00 | Royal Free, February 2015 |
| Surgical procedures | ||
| Liver resection | £7000 | [49] |
| Liver transplant (1st year) | £70,000 | [19, 49, 50], Royal Free, February 2015 |
The primary outcome measure was cost per case of advanced fibrosis detected - a surrogate for cost utility.
Secondary outcomes included unnecessary referral rates of patients with non-advanced disease, the severity of CLD complications, liver transplantation and mortality rates.
Results
Clinical outcome
The base case analysis for 1000 patients with NAFLD over a 1-year timeframe demonstrated 650 patients (65%) were identified as being at low risk of advanced fibrosis and remained in primary care (scenario 1, SC). Of this group, 49 patients (8%) had advanced fibrosis but remained in primary care inappropriately (false negative rate). The remaining 350 patients (35%) were referred to a specialist. After specialist investigation, 93% (324 patients) were determined to be at low risk (false positive rate) and discharged whilst 26 patients (7%) were confirmed to have advanced fibrosis (true positive) and progressed on specialist pathways.
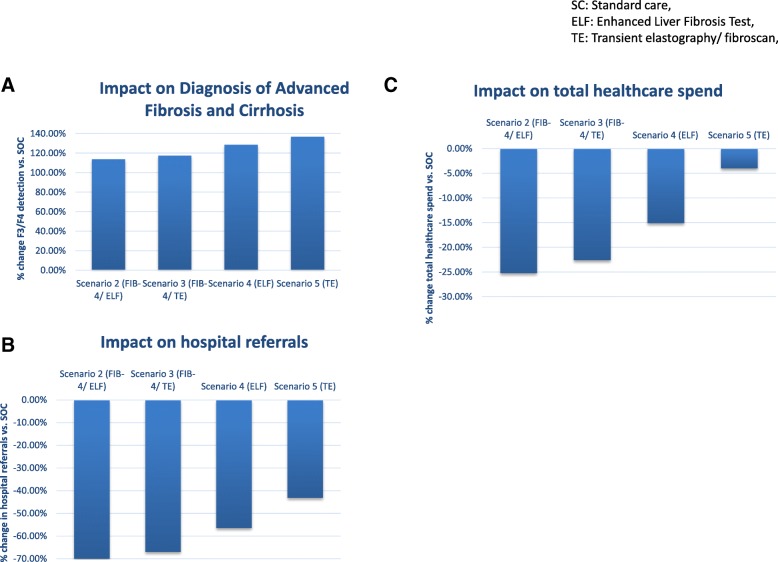
The impact of introducing non-invasive tests into primary care using FIB-4 and ELF (Scenario 2), FIB-4 and TE (Scenario 3), ELF alone (scenario 4) or TE alone (Scenario 5) was assessed (Fig. 4 and Table 4). Over the 1 year time-horizon, compared to SC these strategies reduced the relative referral rate from primary care to hospital by 70, 67, 56 and 43% for scenarios 2, 3, 4 and 5 respectively; corresponding to 245, 223, 198 and 150 fewer referrals over 1 year per 1000 patients. This reduced the need for investigation performed in secondary care. The number of patients requiring imaging in secondary care reduced by 147, 134, 118 and 60 in scenarios 2, 3, 4, and 5 respectively, whilst 25, 22, 20 and 10 fewer patients required endoscopy after 1 year per 1000 patients referred. The requirement for liver biopsy was reduced by 37, 33, 30 and 15 patients in scenarios 2, 3, 4 and 5 respectively. This translated into cost savings in secondary care investigation in the first year per 1000 patients referred of £165,530.04, £150,184.67, £133,505.60 and £68,256.85 for scenarios 2, 3, 4 and 5 respectively.
Fig. 4.
Clinical impact of risk stratification in primary care. Graphs illustrating the impact of introducing NILT into primary care on increasing identification of advanced fibrosis/ cirrhosis (a), reducing hospital referrals (b) and reducing overall healthcare spend (c), for 1000 NAFLD patients over 1 year
Table 4.
Base case analysis of introducing FIB-4, ELF and fibroscan into primary care risk stratification pathways compared to standard of care (scenario 1) after 1 year for 1000 patients with NAFLD
| Scenario 2 - FIB-4/ELF | Scenario 3 - FIB-4/ TE | Scenario 4 - SOC + ELF | Scenario 5 - SOC + TE | |
|---|---|---|---|---|
| Pathway performance: patients referred to specialist (secondary care) | ||||
|
Incremental number of referrals (stratified as ≥F3 fibrosis) (% increase vs SOC) |
−245 (− 70%) | − 222 (− 67%) | −198 (− 56%) | − 101 (25%) |
| Incremental number of ≥F3 disease referred | 30 (53%) | 31 (45%) | 34 (39%) | 36 (25%) |
| Incremental number of ≤F2 disease referred | −275 (−85%) | −253 (−78%) | − 231 (−71%) | −137 (−42%) |
| Incremental number of cirrhotics referred | 1.16 (113%) | 1.20 (116%) | 1.31 (128%) | 1.40 (136%) |
| Pathway performance: patients remain under primary care management | ||||
| Incremental number of patient stratified as ≤F2 fibrosis (Primary care management) | 245 (38%) | 222 (34%) | 198 (30%) | 101 (15%) |
| Incremental number of patients correctly identified as ≤F2 | 274 (46%) | 253 (42%) | 231 (38%) | 137 (23%) |
| Incremental number of patients incorrectly identified as ≤F2 | −30 (−61%) | −31 (−63%) | −34 (− 69%) | −36 (−74%) |
| Overall performance of pathways | ||||
| Sensitivity | 0.75 | 0.76 | 0.80 | 0.83 |
| Specificity | 0.95 | 0.92 | 0.90 | 0.80 |
| Positive Predictive Value | 0.53 | 0.45 | 0.39 | 0.25 |
| Negative Predictive Value | 0.98 | 0.98 | 0.98 | 0.98 |
| Positive Likelihood Ratio | 14.11 | 9.96 | 8.00 | 4.10 |
| Negative Likelihood ratio | 0.27 | 0.26 | 0.22 | 0.21 |
| Impact on end stage liver disease | ||||
|
BCLC Stage 0/A curable HCC (% of all HCC) |
0.06 (36%) | 0.06 (38%) | 0.07 (41%) | 0.08 (44%) |
| BCLC Stage B-D incurable HCC (% of all HCC) | −0.06 (−29%) | −0.07 (−30%) | −0.07 (−33%) | −0.08 (−35%) |
| Varices detected via surveillance programme (% of all new varices) | 0.02 (113%) | 0.02 (117%) | 0.02 (128%) | 0.03 (136%) |
|
Emergency presentation of varices (% of all new varices) |
−0.02 (−30%) | − 0.02 (−31%) | −0.02 (−34%) | −0.03 (−36%) |
| Mild/Moderate ‘other’ complications | < 0.01 (6%) | < 0.01 (6%) | < 0.01 (7%) | < 0.01 (7%) |
| Severe ‘other’ complication | < 0.01 (−9%) | < 0.01 (−9%) | < 0.01 (−10%) | < 0.01 (−10%) |
| Number of liver transplants (of all cirrhotics known to specialist) | 0.02 (32%) | 0.02 (33%) | 0.03 (36%) | 0.03 (39%) |
| Outcomes | ||||
| Mortality / 1000 NAFLD patients | −0.03 (−0.34%) | − 0.03 (− 0.35%) | −0.04 (− 0.39%) | −0.04 (− 0.41%) |
Tabulated analysis of the impact of non-invasive liver fibrosis tests for the management of patients with NAFLD (scenarios 2–5) compared to the standard of care (scenario 1) in the primary care setting
These approaches resulted in reductions in referral of patients with non-advanced liver fibrosis (deemed “unnecessary” referrals) by 85, 78, 71 and 42% (absolute reduction) in scenarios 2, 3, 4, and 5 respectively compared to scenario 1; corresponding to 275, 253, 231 and 137 reduction in inappropriate referrals from 324 patients in scenario 1 over the 1-year time horizon.
TE alone was the most clinically effective strategy in the detection of ≥F3 fibrosis. Compared to SC, introducing NILT increased the identification of patients with advanced fibrosis (true positive rate) by 30, 31, 34 and 36 patients in scenarios 2, 3, 4 and 5 respectively equating to an 114, 118, 129 and 137% improvement..
Considering cirrhosis specifically, over the 1-year timeframe, employing each of the strategies improved detection by 1 patient per 1000 population compared to the SC,. Specifically, an extra 1.2 (113%), 1.2 (116%), 1.3 (128%) and 1.4 (136%) cirrhotic patients per 1000 population were detected in scenarios 2, 3, 4 and 5 respectively over 1 year compared to SC.
The model tested the impact of earlier detection of advanced fibrosis on complications of CLD (Table 4). Patients known to have cirrhosis routinely undergo regular surveillance. The model demonstrated that the number of patients presenting with curable HCC (Stage O/A) increased from a base case of 0.17 patients per 1000 population following SC, by 0.06 patients per 1000 population in scenarios 2 and 3 and by 0.07 and 0.08 patients per 1000 population in scenarios 4 and 5 respectively. Conversely, over 1 year, incurable HCC (stage B-D), rates decreased from a base case of 0.22 patients per 1000 population following SC the same amounts equivalent to reductions of 29, 30, 33 and 35% for scenarios 2–5 respectively. Patients presenting with variceal haemorrhage reduced by 0.02 patients per 1000 population in scenarios 2, 3 and 4, and by 0.03 patients per 1000 population in scenario 5, compared to SOC (0.07 patients per 1000 population). The model predicted that variceal detection through surveillance prior to haemorrhage increased from 0.02 patients per 1000 with SC by 0.02 patients per 1000 in scenarios 2, 3, 4 and 0.03 patients per 1000 in scenario 5, permitting the instigation of primary prophylaxis of variceal haemorrhage that is associated with reduced mortality [44]. Finally, the model demonstrated that management of cirrhosis in secondary care achieved a reduction in episodes of hospitalization due to other complications of CLD including jaundice, ascites and hepatic encephalopathy from 0.03 patients per 1000 in SC by 0.01 patients per 1000 in all scenarios.
Improvements in the detection and management of cirrhosis would permit increased rates of liver transplantation by 0.02 patients per 1000 in scenarios 2 and 3 and 0.03 patients per 1000 in scenarios 4 and 5, compared to SC (0.07 patients per 1000) over 1 year. Predicted all-cause mortality reduced by 0.03 patients per 1000 in scenarios 2 and 3 and 0.04 patients per 1000 in scenarios 4 and 5 over 1 year compared to SC (9.87 patients per 1000).
Cost outcome
The healthcare costs of the competing strategies are summarised in Fig. 5 using a 1-year horizon. For 1000 patients with NAFLD undergoing PCP assessment for liver disease, the costs directly associated with NILT were £10,385 in Scenario 2, £10,632 in scenario 3, £42,000 in scenario 4 and £43,000 in scenario 5 (incorporating additional PCP appointments and blood tests).
Fig. 5.
Cost impact of risk stratification in primary care. Incremental cost expenditure and savings compared to SOC at different stages of the management pathway over a 1-year time horizon for 1000 NAFLD patients
Compared to SC (scenario 1) which cost £670,504 over 1 year, the incremental reductions in healthcare spending achieved through use of NILT in each scenario were £169 K, £152 K, 101 K and 27 K per 1000 patients in 1 year in scenarios 2, 3, 4 and 5 respectively equating to reductions of 25, 23, 15 and 4%. Using cost-per-case of advanced fibrosis as a surrogate for cost utility, all scenarios were favourable to SC (£25,543.02), with the model predicting cost-per-case of advanced fibrosis at £8.932.19, £9083.78, £9487.26 and £10,351.67 in scenarios 2, 3, 4 and 5 respectively over the 1 year timeframe.
From a commissioning perspective, a significant contributor to the immediate cost saving was the reduction in secondary care referrals. Compared to scenario 1 (£41,300 per 1000 patients over 1 year), cost-savings attributable to reduced specialist referral were £28,895, £26,216, £23,305 and £11,915 in scenarios 2, 3, 4 and 5 respectively, equating to 70, 63, 56 and 29% reductions.
A budget impact analysis (Table 5) assessed the impact of introducing the interventions nationally in the UK NHS system. Assuming the prevalence of NAFLD to be 20% in the UK population of 60 million people and a 5-year cycle of disease assessment, nationally 2.4 million people would potentially be risk stratified annually. The incremental cost to the budget holder of introducing NILT for fibrosis in NAFLD in primary care would be £24.9 M, £25.5 M, £100.8 M and £103.2 M in scenario’s 2, 3, 4 and 5 respectively. However, this would deliver savings equating to 23, 21, 14 and 4% reductions in total healthcare expenditure for each of the scenarios respectively.
Table 5.
Budget impact analysis of introducing FIB-4, ELF and fibroscan into primary care risk stratification pathways compared to standard care after 1 year for a population of 60 million patients
| Scenario 2 - FIB-4/ELF | Scenario 3 - FIB-4/ TE | Scenario 4 - SC + ELF | Scenario 5 - SC + TE | |
|---|---|---|---|---|
| Pathway performance: | ||||
| Incremental number of referrals (stratified as ≥F3 fibrosis) (% increase vs SOC) | − 587,700 (−70%) | − 533,217 (−67%) | − 474,000 (−56%) | − 242,340 (−25%) |
| Incremental number of ≥F3 disease referred | 71,640 (53%) | 74,041 (45%) | 81,000 (39%) | 86,220 (25%) |
| Incremental number of ≤F2 disease referred | − 659,340 (−85%) | − 607,258 (−78%) | − 555,021 (−71%) | − 328,560 (−42%) |
| Incremental number of cirrhotics referred | 2786 (113%) | 2880 (116%) | 3153 (128%) | 3359 (136%) |
| Incremental number of patients incorrectly identified as ≤F2 | −71,640 (−61%) | −74,041 (− 63%) | −81,000 (− 69%) | −86,220 (− 74%) |
| IMPACT ON END STAGE LIVER DISEASE | ||||
|
BCLC Stage 0/A curable HCC (% of all HCC) |
121 (36%) | 125 (38%) | 137 (41%) | 146 (44%) |
| BCLC Stage B-D incurable HCC (% of all HCC) | −121 (− 29%) | − 125 (− 30%) | −137 (− 33%) | − 146 (− 35%) |
| Varices detected via surveillance programme (% of all new varices) | 50 (113%) | 51 (117%) | 57 (128%) | 60 (136%) |
|
Emergency presentation of varices (% of all new varices) |
−50 (− 30%) | −51 (− 31%) | −57 (− 34%) | −60 (− 36%) |
| Mild/Moderate ‘other’ complications | 5 (6%) | 5 (6%) | 6 (7%) | < 6 (7%) |
| Severe ‘other’ complication requiring hospital admission | −5 (− 9%) | − 5 (− 9%) | − 6 (− 10%) | −6 (− 10%) |
| Outcomes | ||||
| Mortality / 1000 NAFLD patients | −67 (− 0.34%) | −69 (− 0.35%) | −76 (− 0.39%) | −81 (− 0.41%) |
| Budget | ||||
| Cost of tests | £24.9 M | £25.5 | £100.8 | £103.2 M |
| Total expenditure | -£406 M (−23%) | -£364 M (−21%) | -£243 M (−14%) | -£65 M (− 4%) |
Tabulated analysis of the impact of non-invasive liver fibrosis tests for the management of patients with NAFLD (scenarios 2–5) compared to the standard of care (scenario 1) in the primary care setting for a population of 60 million patients with 20% NAFLD prevelance risk stratified on a 5 year cycle
Table 6 summarises the outcomes of introducing non-invasive liver fibrosis tests in primary care for the base case.
Table 6.
Summary of outcomes resulting from introducing non-invasive liver fibrosis tests in primary care (per 1000 NAFLD patients over 1 years)
| Scenario 1 Standard Care |
Scenario 2 FIB 4 +/− ELF |
Scenario 3 FIB 4 +/− TE |
Scenario 4 ELF |
Scenario 5 TE |
|
|---|---|---|---|---|---|
| Total Referrals avoided (vs. SOC) | – | 245 | 234 | 198 | 150 |
| Cases F3/F4 detected | 26.3 | 56.1 | 57.1 | 60.0 | 62.2 |
| Cases Cirrhosis detected | 5.3 | 11.0 | 11.2 | 11.8 | 12.3 |
| Cases Cirrhosis missed | 11.3 | 5.5 | 5.3 | 4.7 | 4.2 |
| Cost saving (vs. SOC) | – | - £169,408 | - £151,816 | - £101,268 | - £26,889 |
| Cost per ≥ F3 detected | £25,543 | £8932 | £9083 | £9487 | £10,351 |
Projected outcomes over a 5 year timeframe
Analyses were performed using a five-year horizon to assess the longer-term outcomes of the pathway (Table 7).
Table 7.
Projected clinical outcomes and costs of the scenarios projected over 1 year and 5 years
| Scenario 1- SC | Scenario 2 - FIB-4/ELF | Scenario 3 - FIB-4/ TE | Scenario 4 - SC + ELF | Scenario 5 - SC + TE | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 year | 5 years | 1 year | 5 years | 1 year | 5 years | 1 Year | 5 years | 1 year | 5 years | |
| Total number of cirrhotics entered into specialist services (out of all cirrhotics) | 1.03 (34%) | 5.28 (32%) | 2.19 (74%) | 10.97 (66%) | 2.23 (75%) | 11.17 (68%) | 2.34 (79%) | 11.75 (71%) | 2.43 (82%) | 12.23 (74%) |
| Total number of cirrhotics not known to specialist services (out of all cirrhotics) | 1.96 (66%) | 11.278 (68%) | 0.78 (26%) | 5.53 (34%) | 0.74 (25%) | 5.41 (32%) | 0.63 (21%) | 4.73 (29%) | 0.54 (18%) | 4.24 (26%) |
|
Early stage complication (stage 0/A HCC, non-bleeding varices, mild ascites etc) (% of all complications) Cost |
0.22 (42%) £3.0 K | 3.52 (39%) £31.1 K | 0.31 (57%) £4.1 K | 4.62 (52%) £41.2 K | 0.31 (58%) £4.1 K | 4.66 (52%) £41.5 K | 0.32 (60%) £4.3 K | 4.77 (54%) £42.6 K | 0.33 (61%) £4.3 K | 4.87 (55%) £43.4 K |
|
Late stage complication (stage B-D HCC, bleeding varices, severe ascites etc) (% of all complications) Cost |
0.32 (58%) £12.9 K | 5.44 (61%) £141 K | 0.23 (43%) £9.2 K | 4.3 (48%) £108 K | 0.23 (42%) 9.1 K | 4.26 (48%) £107 K | 0.22 (40%) £8.8 K | 4.14 (46%) £103 K | 0.21 (39%) £8.4 K | 4.05 (45%) £101 K |
|
Liver transplant Cost |
0.07 £5.9 K | 1.05 £89.5 K | 0.10 £7.9 K | 1.16 £98.9 K | 0.10 £8.0 K | 1.16 £99.3 K | 0.10 £8.2 K | 1.17100.2 K | 0.10 £8.3 K | 1.18 £101 K |
| Mortality (%) | 9.87 0.99% | 28.56 2.86% | 9.84 0.98% | 28.18 2.82% | 9.83 0.98% | 28.17 2.82% | 9.83 0.98% | 28.13 2.81% | 9.83 0.98% | 28.10 2.81% |
| Total cost/1000 NAFLD patients | 638 K | 1.1 M | 502 K | 946 K | 522 K | 971 K | 570 K | 1.0 M | 647 K | 1.1 M |
| Cost/advanced fibrotic detected | 25.7 K | 49.9 K | 9.0 K | 19.4 K | 9.1 K | 19.4 K | 9.5 K | 19.5 K | 10.4 K | 20.5 K |
Five years after the introduction of NILT the model demonstrated clinical benefit, with increases in the detection of cirrhosis by 107, 111, 123 and 132% in scenarios 2, 3, 4 and 5 respectively equating to an extra 5.69, 5.90, 6.48 and 6.95 cases per 1000 patients tested per year.
Using a discount rate of 3.5%, compared to SC over 5 years incremental savings of £168,449.80, £142,752.51, £86.604.60 and £20,769.62 were made in scenarios 2, 3, 4, and 5 respectively.
One way sensitivity analyses
We performed a one-way sensitivity analysis on the base-case scenario using a time-frame of 1 year.
A pathway uptake sensitivity analysis tested assumptions about the proportion of patients entering the pathway (0–100%) and confirmed a linear benefit proportional to pathway uptake and reinforced that any utilisation of the pathway (i.e. > 0%) would deliver benefit in all scenarios over the 1-year timeframe.
A clinical effectiveness sensitivity analysis was performed by varying the specificity of SC for the detection of advanced fibrosis. In the base-case model, a value of 0.65 was assumed (65% specificity for the detection of advanced fibrosis). To counter the influence of this assumption, around which sparse data are published, it was varied from 0.00 to 1.00, demonstrating a significant influence on cost-effectiveness. However, the cost-benefit was only negated when the specificity of SC for the detection of advanced fibrosis exceeded 0.88, 0.86, 0.80 and 0.68 in scenarios 2, 3, 4 and 5 respectively.
Discussion
Our cost consequence analyses indicate that the use of NILT to stratify patients with NAFLD in primary care is clinically effective and cost saving. Utilizing fibroscan alone was most effective in detecting patients with advanced fibrosis, whilst employing FIB-4 and ELF delivered the greatest cost saving.
All of the scenarios using NILT in primary care permitted the earlier identification of advanced fibrosis/cirrhosis, creating opportunities to modify fibrosis progression [22, 51] and to start surveillance and treatment of varices and HCC. Modelling indicated that significant benefits could accrue from the detection of early stage curable HCC (stage 0/ A) and non-bleeding varices that can be treated with beta-blockers and band ligation that can avert emergency presentations with bleeding varices. A modest reduction in hospital admissions for other complications of CLD including jaundice, ascites and hepatic encephalopathy was demonstrated. The relatively limited impact of current care pathways on mortality, largely attributable to missed or late diagnosis of advanced fibrosis, highlights the need for new approaches to diagnosis and treatment to prevent NAFLD progression.
Of interest to commissioners, the implementation of NILT in primary care offers the potential to reduce the total number of referrals, and in particular the unnecessary referral of patients who have minimal fibrosis. Over a 1-year horizon, there was a reduction in total referrals of 70, 63, 56 and 29% in scenarios 2, 3, 4 and 5 respectively, with an 85, 78, 71 and 42% reduction in referrals of patients with non-advanced disease. Real-world data from a secondary care hepatology service found that 66% of referred patients had a baseline FIB-4 score < 1.30 indicating that these patients had a low risk of advanced fibrosis and could have avoided referral [13]. This group of patients represents an inefficient use of resources, adding pressure to overstretched outpatient specialist services and PCP healthcare budgets [52]. Introduction of the use of NILT in primary care would deliver immediate reductions in expenditure through avoidance of unnecessary referrals, unlike the cost-benefits associated with improved outcomes attributable to earlier diagnosis that accrue much later.
All scenarios were cost saving. The cost of detecting a case of advanced fibrosis using SC was £49,917.83 in scenario. This compared to £19,360.75 in scenario 2, £19,448.49 in scenario 3, £19,487.75 in scenario 4 and £20,451.35 in scenario 5. Healthcare budgets were reduced by 17, 15, 11 and 3% in scenarios 2, 3, 4 and 5 respectively, attributable to the reduction in costs associated with end stage liver disease and improved resource utilisation. The budget decrease in scenario 5 was modest as it was assumed all patients who failed fibroscan (5% of cohort) were referred to specialists.
In this analysis, we explored the impact of the use of FIB-4 and ELF to replicate a primary care pathway introduced in north London. This combination of tests was optimal for the stratification of patients in a hospital based population [53] and has subsequently been shown to be clinically effective [13] when applied in primary care where the proportion of cases of advanced fibrosis amongst cases of NAFLD is smaller.
There are limitations to the model. The model was populated with the best available published evidence. A lack of high quality data for some variables was remedied with expert opinion. The measures of performance for FIB-4, ELF and fibroscan were drawn from validations in hospital cohorts, but these estimates may be inappropriately high for primary care populations in which the prevalence of fibrosis is likely to be lower (spectrum bias) [54, 55]. The model assumes that use of ELF and fibroscan as second-tier tests has the same performance characteristics as a first-tier test. This may under-estimate the performance of the pathway. Additionally, the model was limited to FIB-4, ELF and fibroscan. Local PCP focus groups supported by consultants in Public Health, formed to aide Camden and Islington pathway development, outlined the practical need for simplicity, giving FIB-4 an advantage over the NAFLD-fibrosis score, which the PCP considered to be more challenging to obtain such as BMI, or associated with uncertainty about case definition such as diabetes. The model examines fibrosis, but not NASH, and so may underestimate disease progression. Other sources of error include analytical performance. We assumed a 5% failure rate for fibroscan but higher rates have been reported [47], whilst serum tests can be influenced by comorbidities. For the purposes of the model, liver biopsy was considered the reference test for liver fibrosis. Given the inherent inaccuracies associated with liver biopsy [56], this may have over-estimated the performance of liver biopsy. The cost estimate of liver biopsy incorporated procedural elements but not those associated with complications of the procedure. This approach may underestimate the true cost associated with liver biopsy. The clinical- and cost- efficiency of all scenarios are highly favourable, but may not reflect real-life outcomes. Not all patients with NAFLD consult their PCP, and pathway uptake by health professionals is variable. The base case is a 50-year-old man with abnormal transaminases, reflecting a screening strategy identified by the Camden and Islington steering committee as practical in real-life primary care practice. However this approach will miss cases of NAFLD with advanced fibrosis but normal LFTs. A screen-all strategy is likely to be clinically optimal, as patients with NAFLD and normal transaminases are at risk of significant fibrosis. The costing in the model is comprehensive, assuming full adherence to guidelines and protocols and thereby potentially overestimating the cost of care. We employed a probabilistic decision model as our main economic focus was on the payer perspective rather than a population health perspective, where alternative cost-effectiveness approaches using quality of life data and Markov simulations would be desirable. The lack of beta or triangular distributions and true probability sensitivity analysis limits the model. The model lacks cost/ QALY data and relies on descriptive measures including cost per case of advanced fibrosis detected. These outcome measures have no standard comparator limiting the current model. These shortcomings will be addressed in future work.
NAFLD should be considered as the hepatic manifestation of a multisystem disorder associated with widespread morbidity including cardiovascular disease, diabetes, hyperlipidaemia and cancer. While a fully comprehensive health economic model would need to take these morbidities into consideration we chose to focus on liver disease. Accommodating all NAFLD associated morbidities, their evaluation and management was beyond the scope of this study.
This study has several strengths. A comprehensive literature review was undertaken to identify estimates for clinical parameters, transition rates and costs. The study adds to the current body of evidence and our conclusions are similar to other health economic analyses [57]. A Health Technology Assessment undertaken for the National Institute for Health Research [19] concluded that use of NILT was more cost-effective than liver biopsy in detecting cases of advanced fibrosis. Tapper et al. [58] demonstrated that the use of the NAFLD fibrosis score and fibroscan in primary care yielded cost-effective results. Robust data are lacking regarding the performance of PCP’s in the identification of patients with advanced liver disease. Additionally, there is no published randomised controlled trial exploring the performance of NILT in primary care. Whilst limiting the model, the information provided by the analysis may be supplemented with the results from real-life pilot studies in due course. Harman et al. [59] have demonstrated the use of fibroscan in patients with risk factors for CLD, including diabetes, obesity and alcohol excess can increase detection of cirrhosis by 140%, similar to the modelling results in scenario 5 (132%). Our group has evaluated the pathway employing FIB-4 and ELF (Scenario 2) [13].
Conclusions
This study demonstrates that the introduction of NILT in primary care has the potential to increase the detection of cases of NAFLD with advanced fibrosis and cirrhosis, reduce unnecessary referrals to secondary care of patients at low risk of liver disease and to deliver immediate and sustained significant cost savings. The model provides compelling evidence for clinicians, commissioners and policy makers to consider the formal introduction of non-invasive liver fibrosis testing in primary care, in line with other central policy statements [4, 60, 61].
Acknowledgements
Not applicable.
Abbreviations
- ALD
Alcoholic liver disease
- ALT
Alanine aminotransferase
- AST
Aspartate aminotransferase
- BCLC
Barcelona Clinic Liver Cancer
- CLD
Chronic liver disease
- ELF
Enhanced Liver Fibrosis
- FIB-4
Fibrosis-4 score
- HCC
Hepatocellular carcinoma
- HCV
Hepatitis C Virus
- LFT
Liver function tests
- NAFLD
Non-alcoholic fatty liver disease
- NHS
National health system
- NILT
Non-invasive liver fibrosis tests
- PCP
Primary care physician
- SOC
Standard of care
Authors’ contributions
AS, SJ, AG and WR were major contributors in study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, and critical revision. RG, SM, KS, ST, JO, ET and JP were involved in acquisition of data and critical revision of the manuscript. EP contributed to analysis and interpretation of data, and critical revision of the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by an unrestricted grant from Siemens Healthineers.
The funder had no role in the design of the study, collection, analysis and interpretation of data or writing the manuscript.
Availability of data and materials
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study does not report on animal or human data. It reports theoretical modelling data. Data used for calculating costs in the modelling work were obtained from NHS sources and published literature. Some of the clinical data were drawn from an evaluation of service improvement conducted at The Royal Free London NHS Foundation Trust. The Royal Free London NHS Foundation Trust Research and Development Department judged this work to be an evaluation of a service improvement innovation, therefore this was registered for audit (EDGE ID:122031) but not subject to review by an independent ethics committee and individual patient consent was not sought. All activities were performed in accordance with the guidelines of the Helsinki Declaration.
Consent for publication
Not applicable.
Competing interests
AS, JP and WR have received speaker bureau from Siemens Healthineers; AS, SJ and WR have received support for research from Siemens Healthineers; WR is a NIHR Senior Investigator and is supported by the UCLH NIHR BRC.
EP is supported by the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) North Thames at Bart’s Health NHS Trust. The views expressed are those of the author (s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
The other authors have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ankur Srivastava, Email: Ankur.srivastava@nhs.net.
Simcha Jong, Email: s.jong@liacs.leidenuniv.nl.
Anna Gola, Email: anna.gola@ucl.ac.uk.
Ruth Gailer, Email: r.gailer@nhs.net.
Sarah Morgan, Email: sarah.morgan1@nhs.net.
Karen Sennett, Email: karen.sennett@nhs.net.
Sudeep Tanwar, Email: sudeep.tanwar@nhs.net.
Elena Pizzo, Email: e.pizzo@ucl.ac.uk.
James O’Beirne, Email: j.o'beirne@ucl.ac.uk.
Emmanuel Tsochatzis, Email: e.tsochatzis@ucl.ac.uk.
Julie Parkes, Email: jules@soton.ac.uk.
William Rosenberg, Email: w.rosenberg@ucl.ac.uk.
References
- 1.Scalone L, Fagiuoli S, Ciampichini R, Gardini I, Bruno R, Pasulo L, Lucà MG, Fusco F, Gaeta L, Del Prete A, et al. The societal burden of chronic liver diseases: results from the COME study. BMJ Open Gastroenterol. 2015;0(0):e000025. doi: 10.1136/bmjgast-2014-000025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Neff GW, Duncan CW, Schiff ER. The Current Economic Burden of Cirrhosis. Gastroenterol Hepatol. 2011;7(10):661–671. [PMC free article] [PubMed] [Google Scholar]
- 3.Office for National Statistics . Newport: Office for National Statistics. 2011. Mortality statistics: deaths registered in England and Wales (series DR), 2010. [Google Scholar]
- 4.Williams R, Aspinall R, Bellis M, Camps-Walsh G, Cramp M, Dhawan A, Ferguson J, Forton D, Foster G, Gilmore I, et al. Addressing liver disease in the UK: a blueprint for attaining excellence in health care and reducing premature mortality from lifestyle issues of excess consumption of alcohol, obesity, and viral hepatitis. Lancet (London, England) 2014;384(9958):1953–1997. doi: 10.1016/S0140-6736(14)61838-9. [DOI] [PubMed] [Google Scholar]
- 5.Armstrong MJ, Houlihan DD, Bentham L, Shaw JC, Cramb R, Olliff S, Gill PS, Neuberger JM, Lilford RJ, Newsome PN. Presence and severity of non-alcoholic fatty liver disease in a large prospective primary care cohort. J Hepatol. 2012;56(1):234–240. doi: 10.1016/j.jhep.2011.03.020. [DOI] [PubMed] [Google Scholar]
- 6.Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34(3):274–285. doi: 10.1111/j.1365-2036.2011.04724.x. [DOI] [PubMed] [Google Scholar]
- 7.Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, Mills PR, Keach JC, Lafferty HD, Stahler A, et al. Liver Fibrosis, but No Other Histologic Features, Is Associated With Long-term Outcomes of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015;149(2):389–397.e310. doi: 10.1053/j.gastro.2015.04.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ekstedt M, Hagström H, Nasr P, Fredrikson M, Stål P, Kechagias S, Hultcrantz R. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015;61(5):1547–1554. doi: 10.1002/hep.27368. [DOI] [PubMed] [Google Scholar]
- 9.Verma S, Jensen D, Hart J, Mohanty SR. Predictive value of ALT levels for non-alcoholic steatohepatitis (NASH) and advanced fibrosis in non-alcoholic fatty liver disease (NAFLD) Liver Int. 2013;33(9):1398–1405. doi: 10.1111/liv.12226. [DOI] [PubMed] [Google Scholar]
- 10.Lam B, Younossi ZM. Treatment options for nonalcoholic fatty liver disease. Ther Adv Gastroenterol. 2010;3(2):121–137. doi: 10.1177/1756283X09359964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lay CS, Tsai YT, Teg CY, Shyu WS, Guo WS, Wu KL, Lo KJ. Endoscopic variceal ligation in prophylaxis of first variceal bleeding in cirrhotic patients with high-risk esophageal varices. Hepatology. 1997;25(6):1346–1350. doi: 10.1002/hep.510250608. [DOI] [PubMed] [Google Scholar]
- 12.Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004;130(7):417–422. doi: 10.1007/s00432-004-0552-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Srivastava A, Gailer R, Tanwar S, Trembling P, Parkes J, Rodger A, Suri D, Thorburn D, Sennett K, Morgan S, et al. Prospective evaluation of a primary care referral pathway for patients with non-alcoholic fatty liver disease. J Hepatol. 2019. 10.1016/j.jhep.2019.03.033. [Epub ahead of print] [DOI] [PubMed]
- 14.Sebastiani G. Non-invasive assessment of liver fibrosis in chronic liver diseases: Implementation in clinical practice and decisional algorithms. World J Gastroenterol. 2009;15(18):2190–2203. doi: 10.3748/wjg.15.2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.National Liver Plan . BSG. BSG. 2009. A time to act: improving liver health and outcomes in liver disease. [Google Scholar]
- 16.Srivastava A, Gailer R, Tanwar S, Trembling P, Warner A, Morgan S, Sennett K, Thorburn D, Tsochatzis E, Rosenberg W. P1046 : A one year retrospective review of new patient attendances at a tertiary hepatology centre highlighting the increasing challenge of NAFLD and the need to develop clinical pathways. J Hepatol. 2015;62:S740.
- 17.Ratib S, Fleming KM, Crooks CJ, Aithal GP, West J. 1 and 5 year survival estimates for people with cirrhosis of the liver in England, 1998–2009: A large population study. J Hepatol. 2014;60(2):282–9. [DOI] [PubMed]
- 18.McPherson S, Stewart SF, Henderson E, Burt AD, Day CP. Simple non-invasive fibrosis scoring systems can reliably exclude advanced fibrosis in patients with non-alcoholic fatty liver disease. Gut. 2010;59(9):1265–1269. doi: 10.1136/gut.2010.216077. [DOI] [PubMed] [Google Scholar]
- 19.Crossan C, Tsochatzis EA, Longworth L, Gurusamy K, Davidson B, Rodriguez-Peralvarez M, Mantzoukis K, O'Brien J, Thalassinos E, Papastergiou V, et al. Cost-effectiveness of non-invasive methods for assessment and monitoring of liver fibrosis and cirrhosis in patients with chronic liver disease: systematic review and economic evaluation. Health Technol Assess (Winchester, England) 2015;19(9):1–409. doi: 10.3310/hta19090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guha IN, Parkes J, Roderick P, Chattopadhyay D, Cross R, Harris S, Kaye P, Burt AD, Ryder SD, Aithal GP, et al. Noninvasive markers of fibrosis in nonalcoholic fatty liver disease: Validating the European Liver Fibrosis Panel and exploring simple markers. Hepatology (Baltimore, Md) 2008;47(2):455–460. doi: 10.1002/hep.21984. [DOI] [PubMed] [Google Scholar]
- 21.Wong VW, Vergniol J, Wong GL, Foucher J, Chan HL, Le Bail B, Choi PC, Kowo M, Chan AW, Merrouche W, et al. Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology (Baltimore, Md) 2010;51(2):454–462. doi: 10.1002/hep.23312. [DOI] [PubMed] [Google Scholar]
- 22.Cobbold JFL, Raveendran S, Peake CM, Anstee QM, Yee MS, Thursz MR. Piloting a multidisciplinary clinic for the management of non-alcoholic fatty liver disease: initial 5-year experience. Frontline Gastroenterology. 2013;4(4):263–269. doi: 10.1136/flgastro-2013-100319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Teli MR, James OF. Burt AD, Bennett MK, Day CP. The natural history of nonalcoholic fatty liver: a follow-up study. Hepatology (Baltimore, Md) 1995;22(6):1714–1719. [PubMed] [Google Scholar]
- 24.Pais R, Charlotte F, Fedchuk L, Bedossa P, Lebray P, Poynard T, Ratziu V. A systematic review of follow-up biopsies reveals disease progression in patients with non-alcoholic fatty liver. J Hepatol. 2013;59(3):550–556. doi: 10.1016/j.jhep.2013.04.027. [DOI] [PubMed] [Google Scholar]
- 25.Ekstedt M, Franzen LE, Mathiesen UL, Thorelius L, Holmqvist M, Bodemar G, Kechagias S. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology (Baltimore, Md) 2006;44(4):865–873. doi: 10.1002/hep.21327. [DOI] [PubMed] [Google Scholar]
- 26.Adams LA, Lymp JF, St. Sauver J, Sanderson SO, Lindor KD, Feldstein A, Angulo P. The Natural History of Nonalcoholic Fatty Liver Disease: A Population-Based Cohort Study. Gastroenterology. 2005;129(1):113–121. doi: 10.1053/j.gastro.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 27.Jepsen P, Vilstrup H, Mellemkjaer L, Thulstrup AM, Olsen JH, Baron JA, Sorensen HT. Prognosis of patients with a diagnosis of fatty liver--a registry-based cohort study. Hepato-gastroenterology. 2003;50(54):2101–2104. [PubMed] [Google Scholar]
- 28.Pagadala MR, McCullough A. The Relevance of Liver Histology to Predict Clinically Meaningful Outcomes in NASH. Clin Liver Dis. 2012;16(3):487–504. doi: 10.1016/j.cld.2012.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bhala N, Angulo P, van der Poorten D, Lee E, Hui JM, Saracco G, Adams LA, Charatcharoenwitthaya P, Topping JH, Bugianesi E, et al. The natural history of nonalcoholic fatty liver disease with advanced fibrosis or cirrhosis: an international collaborative study. Hepatology (Baltimore, Md) 2011;54(4):1208–1216. doi: 10.1002/hep.24491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Adams LA, Sanderson S, Lindor KD, Angulo P. The histological course of nonalcoholic fatty liver disease: a longitudinal study of 103 patients with sequential liver biopsies. J Hepatol. 2005;42(1):132–138. doi: 10.1016/j.jhep.2004.09.012. [DOI] [PubMed] [Google Scholar]
- 31.Hui JM, Kench JG, Chitturi S, Sud A, Farrell GC, Byth K, Hall P, Khan M, George J. Long-term outcomes of cirrhosis in nonalcoholic steatohepatitis compared with hepatitis C. Hepatology (Baltimore, Md) 2003;38(2):420–427. doi: 10.1053/jhep.2003.50320. [DOI] [PubMed] [Google Scholar]
- 32.Sanyal AJ, Banas C, Sargeant C, Luketic VA, Sterling RK, Stravitz RT, Shiffman ML, Heuman D, Coterrell A, Fisher RA, et al. Similarities and differences in outcomes of cirrhosis due to nonalcoholic steatohepatitis and hepatitis C. Hepatology (Baltimore, Md) 2006;43(4):682–689. doi: 10.1002/hep.21103. [DOI] [PubMed] [Google Scholar]
- 33.Fleming KM, Aithal GP, Card TR, West J. The rate of decompensation and clinical progression of disease in people with cirrhosis: a cohort study. Aliment Pharmacol Ther. 2010;32(11–12):1343–1350. doi: 10.1111/j.1365-2036.2010.04473.x. [DOI] [PubMed] [Google Scholar]
- 34.Ascha MS, Hanouneh IA, Lopez R, Tamimi TA-R, Feldstein AF, Zein NN. The incidence and risk factors of hepatocellular carcinoma in patients with nonalcoholic steatohepatitis. Hepatology (Baltimore, Md) 2010;51(6):1972–1978. doi: 10.1002/hep.23527. [DOI] [PubMed] [Google Scholar]
- 35.Singal AG, Conjeevaram HS, Volk ML, Fu S, Fontana RJ, Askari F, Su GL, Lok AS, Marrero JA. Effectiveness of Hepatocellular Carcinoma Surveillance in Patients with Cirrhosis. Cancer Epidemiol Biomark Prev. 2012;21(5):793–799. doi: 10.1158/1055-9965.EPI-11-1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Younossi ZM, Otgonsuren M, Henry L, Venkatesan C, Mishra A, Erario M, Hunt S. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology. 2015;62(6):1723–1730. doi: 10.1002/hep.28123. [DOI] [PubMed] [Google Scholar]
- 37.Estes C, Anstee QM, Arias-Loste MT, Bantel H, Bellentani S, Caballeria J, Colombo M, Craxi A, Crespo J, Day CP, et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016-2030. J Hepatol. 2018;69(4):896–904. doi: 10.1016/j.jhep.2018.05.036. [DOI] [PubMed] [Google Scholar]
- 38.Matteoni CA, Younossi ZM, Gramlich T, Boparai N, Liu YC, McCullough AJ. Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology. 1999;116(6):1413–1419. doi: 10.1016/s0016-5085(99)70506-8. [DOI] [PubMed] [Google Scholar]
- 39.D’Amico G. Natural History and Stages of Cirrhosis. New York: Springer New York; 2014. [Google Scholar]
- 40.Reddy SK, Steel JL, Chen H-W, DeMateo DJ, Cardinal J, Behari J, Humar A, Marsh JW, Geller DA, Tsung A. Outcomes of curative treatment for hepatocellular cancer in nonalcoholic steatohepatitis versus hepatitis C and alcoholic liver disease. Hepatology (Baltimore, Md) 2012;55(6):1809–1819. doi: 10.1002/hep.25536. [DOI] [PubMed] [Google Scholar]
- 41.Pons F, Varela M, Llovet JM. Staging systems in hepatocellular carcinoma. HPB. 2005;7(1):35–41. doi: 10.1080/13651820410024058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Spiegel BM, Esrailian E, Eisen G. The budget impact of endoscopic screening for esophageal varices in cirrhosis. Gastrointest Endosc. 2007;66(4):679–692. doi: 10.1016/j.gie.2007.02.048. [DOI] [PubMed] [Google Scholar]
- 43.Bosch J, Garcia-Pagan JC. Prevention of variceal rebleeding. Lancet (London, England) 2003;361(9361):952–954. doi: 10.1016/S0140-6736(03)12778-X. [DOI] [PubMed] [Google Scholar]
- 44.Garcia-Tsao G, Sanyal AJ, Grace ND, Carey W. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology (Baltimore, Md) 2007;46(3):922–938. doi: 10.1002/hep.21907. [DOI] [PubMed] [Google Scholar]
- 45.Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med. 2014;11(4):e1001624. doi: 10.1371/journal.pmed.1001624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Samonakis DN, Koulentaki M, Coucoutsi C, Augoustaki A, Baritaki C, Digenakis E, Papiamonis N, Fragaki M, Matrella E, Tzardi M, et al. Clinical outcomes of compensated and decompensated cirrhosis: A long term study. World J Hepatol. 2014;6(7):504–512. doi: 10.4254/wjh.v6.i7.504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Castera L, Foucher J, Bernard PH, Carvalho F, Allaix D, Merrouche W, Couzigou P, de Ledinghen V. Pitfalls of liver stiffness measurement: a 5-year prospective study of 13,369 examinations. Hepatology (Baltimore, Md) 2010;51(3):828–835. doi: 10.1002/hep.23425. [DOI] [PubMed] [Google Scholar]
- 48.Curtis LA. Unit Costs of Health & Social Care 2013. Personal Social Service Research Unit. 2013. [Google Scholar]
- 49.Thompson Coon J, Rogers G, Hewson P, Wright D, Anderson R, Cramp M, Jackson S, Ryder S, Price A, Stein K. Surveillance of cirrhosis for hepatocellular carcinoma: systematic review and economic analysis. Health Technol Assess (Winchester, England) 2007;11(34):1–206. doi: 10.3310/hta11340. [DOI] [PubMed] [Google Scholar]
- 50.Longworth L, Young T, Buxton MJ, Ratcliffe J, Neuberger J, Burroughs A, Bryan S. Midterm cost-effectiveness of the liver transplantation program of England and Wales for three disease groups. Liver Transpl. 2003;9(12):1295–1307. doi: 10.1016/j.lts.2003.09.012. [DOI] [PubMed] [Google Scholar]
- 51.Dyson JK, Anstee QM, McPherson S. Non-alcoholic fatty liver disease: a practical approach to treatment. Frontline Gastroenterol. 2014;5(4):277–286. doi: 10.1136/flgastro-2013-100404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mehrotra A, Forrest CB, Lin CY. Dropping the Baton: Specialty Referrals in the United States. Milbank Q. 2011;89(1):39–68. doi: 10.1111/j.1468-0009.2011.00619.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tanwar STP, Hogan B, Thorburn D, Guha I, Parkes I, Kaye P, Burt A, Ryder S, Aithal G, Day C, Rosenberg W. An algorithm combining direct and indirect liver fibrosis tests enhances diagnostic performance for the detection of advanced fibrosis (F3-4) in NAFLD minimising the need for liver biopsy. Hepatology (Baltimore, Md) 2012;56:264A. [Google Scholar]
- 54.Schmidt RL, Factor RE. Understanding sources of bias in diagnostic accuracy studies. Arch Pathol Lab Med. 2013;137(4):558–565. doi: 10.5858/arpa.2012-0198-RA. [DOI] [PubMed] [Google Scholar]
- 55.Willis BH. Spectrum bias--why clinicians need to be cautious when applying diagnostic test studies. Fam Pract. 2008;25(5):390–396. doi: 10.1093/fampra/cmn051. [DOI] [PubMed] [Google Scholar]
- 56.Ratziu V, Charlotte F, Heurtier A, Gombert S, Giral P, Bruckert E, Grimaldi A, Capron F, Poynard T. Sampling Variability of Liver Biopsy in Nonalcoholic Fatty Liver Disease. Gastroenterology. 2005;128(7):1898–1906. doi: 10.1053/j.gastro.2005.03.084. [DOI] [PubMed] [Google Scholar]
- 57.Mahady SE, Wong G, Craig JC, George J. Pioglitazone and vitamin E for nonalcoholic steatohepatitis: A cost utility analysis. Hepatology (Baltimore, Md) 2012;56(6):2172–2179. doi: 10.1002/hep.25887. [DOI] [PubMed] [Google Scholar]
- 58.Tapper EB, Hunink MG, Afdhal NH, Lai M, Sengupta N. Cost-Effectiveness Analysis: Risk Stratification of Nonalcoholic Fatty Liver Disease (NAFLD) by the Primary Care Physician Using the NAFLD Fibrosis Score. PLoS One. 2016;11(2):e0147237. doi: 10.1371/journal.pone.0147237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Harman DJ, Ryder SD, James MW, Jelpke M, Ottey DS, Wilkes EA, Card TR, Aithal GP, Guha IN. Direct targeting of risk factors significantly increases the detection of liver cirrhosis in primary care: a cross-sectional diagnostic study utilising transient elastography. BMJ Open. 2015;5(4):e007516. doi: 10.1136/bmjopen-2014-007516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Non-alcoholic fatty liver disease (NAFLD): assessment and management; NICE guidelines (NG49). In National Institute for Health and Care Excellence: National Institute for Health and Care Excellence. 2016. http//www.niceorg.uk/guidance/ng49. [PubMed]
- 61.Newsome PN, Cramb R, Davison SM, Dillon JF, Foulerton M, Godfrey EM, Hall R, Harrower U, Hudson M, Langford A, et al. Guidelines on the management of abnormal liver blood tests. Gut. 2018;67(1):6–19. doi: 10.1136/gutjnl-2017-314924. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.