Abstract
Two commercial Chimeric Antigen Receptor (CAR) T cell therapies for CD19 expressing B cell malignancies, Kymriah and Yescarta, have recently been approved by the FDA. The administration of CAR T cells is a complex endeavor involving cell manufacture, tracking and shipping of apheresis products, and management of novel and severe toxicities. At Memorial Sloan Kettering Cancer Center, we have identified eight essential tasks that define the CAR T cell workflow. In this review, we discuss practical aspects of CAR T cell program development, including clinical, administrative, and regulatory challenges for successful implementation.
Keywords: CAR T cells, cellular therapy, Chimeric Antigen Receptor, Cytokine Release Syndrome, diffuse large b cell lymphoma, acute lymphoblastic leukemia
The recent FDA approval of Chimeric Antigen Receptor (CAR) T cell therapies is the culmination of decades of scientific and clinical development1. Modern CARs were built from advances in tumor-specific cell therapy2,3, the development of ‘T-bodies’ by Zelig Eshar4,5, advances in genetic manipulation of primary human T cells6,and the design of a second generation of CARs with significantly enhanced potency7,8. Next, physician-scientists led a series of single center trials that demonstrated CAR T cell activity in relapsed and aggressive forms of non-Hodgkin’s lymphoma (NHL)9-11, adult acute lymphoblastic leukemia (ALL)12,13, pediatric ALL14,15, and chronic lymphocytic leukemia (CLL)16.
We are now faced with administrative and logistic challenge of delivering CAR therapy outside the scope of clinical trials. Attention in the scientific and lay press has led to tremendous excitement and high demand, and meeting expectations will be a challenge. Patients may assume there will be broad access to these products in a similar fashion to other chemotherapeutic agents; however, CAR T cells will only be limited to certain centers that are certified by the manufacturers, and only provided to patients who fulfill strict label requirements. The process of building cell therapy infrastructure, meeting regulatory requirements, and ultimately treating patients is a significant undertaking. This review describes our institutional experience with the development of a CAR T cell program at Memorial Sloan Kettering Cancer Center (MSKCC) in New York City.
CARs in the Clinic
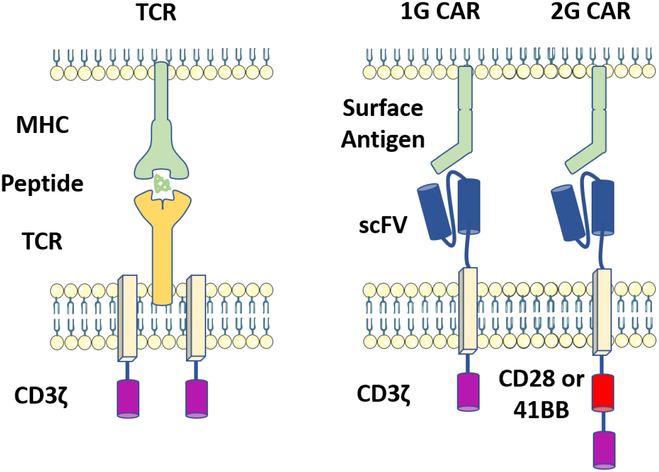
CARs are minimally composed of two domains: 1) an extracellular antibody fragment domain that recognizes tumor-associated antigens, fused via linker and transmembrane domains to 2) an internal activation signaling domain (such as CD3 zeta) that activates T cells (Figure 1). Although CARs can theoretically be designed to target any extracellular antigen, the target antigen CD19, displayed on both normal B cells and in malignant B cell lymphomas and leukemia at high levels31, has achieved the most clinical success. To enhance signaling and improve efficacy, second generation CARs7,8 additionally include a co-stimulatory signaling domain derived from CD28 17,18 or 41BB 18,19. While CD28 containing CARs expand robustly and to rapidly eradicate tumor20, second generation 4-1BB containing CARs are initially fewer in number21 but may better withstand T cell exhaustion induced by tonic CAR signaling22,23. A full assessment of the relative benefits of each approach is beyond the scope of this review, but as described below, both CD28 and 4-1BB containing CAR T cells will enter the clinic as FDA approved cellular therapies.
Figure 1: Antigen Recognition by T cells.
T cell receptors recognize intracellular antigens presented as peptides in the context of major histocompatibility complex (MHC). First generation (1G) CAR T cells target extracellular tumor-associated antigens using an antibody-derived Single Chain Variable Fragment (scFV; blue) fused to an adaptor domain (tan) and internal CD3ζ signaling domain (purple). Second Generation (2G) CAR T cells incorporate a co-stimulatory domain (red) for enhanced T cell function. Co-stimulation is commonly provided through signaling of CD28 or 4-1BB.
Tisagenlecleucel (Kymirah)
In 2012, Novartis and the University of Pennsylvania entered into a global collaboration and licensing agreement which culminated in Novartis’ tisagenlecleucel, tradename Kymriah. The ongoing ELIANA study ( NCT02435849) is the first pediatric global CAR T cell therapy trial24, while the JULIET study ( NCT02445248) is evaluating the efficacy and safety of tisagenlecleucel in adult patients with relapsed/refractory DLBCL25.
Tisagenlecleucel utilizes a murine single chain variable fragment to recognize human CD19, linked to a CD8a spacer domain (Table 1). Like the University of Pennsylvania construct on which it is based, it is a second-generation CAR that utilizes a 4-1BB costimulatory domain, which has important implications for persistence and activity, as discussed previously.
Table 1:
Characteristics of Approved CAR T Cell Products
| Company | Gilead (KITE) | Novartis |
| Product Name | Axicabtagene Ciloleucel | Tisagenlecleucel |
| Academic License | NCI | University of Pennsylvania |
| Binding Domain (All Murine ScFv) |
FMC63 | FMC63 |
| Indications | DLBCL, TFL PMBCL | Pediatric ALL <26 yrs old |
| Spacer Domain | CD28 | CD8α |
| Transmembrane Domain | CD28 | CD8α |
| Stimulatory Domain | CD28-CD3ζ | 4-1BB-CD3ζ |
| Starting Cell Population Selection | None | None |
| Final CD4/CD8 ratio | Variable | Variable |
| Ablation Technology | None | None |
| Viral Vector | Gamma retrovirus | Lentivirus |
| Outcomes | ||
| CR Rate | NHL 54% | ALL 83% |
| Grade 3-5 Cytokine Release | 30% | 30% |
| Grade 3-5 Neurotoxicity | 30% | 30% |
| Average LOS on Study | 14 days | NS |
| Projected Demand in the United States | 4000 to 6000 patients | 400-600 patients |
Interim analysis of the ELIANA trial for relapsed/refractory pediatric ALL published in June 201724 showed that 83% of patients achieved CR/CRi, with 79% survival at 12 months. CAR T cells and concomitant B-cell aplasia in peripheral blood were observed for more than one year in some patients. Approximately 8% of patients suffered manufacturing failures, and 10% were not infused due to death or adverse events.
Kymriah (Tisagenlecleucel) was recently FDA approved and carries an indication for patients up to 25 years of age with B-cell precursor ALL that is refractory or in second or later relapse. Notably, ELIANA eligibility criteria required CD19 positivity confirmed within 3 months of enrollment, excluded patients with active CNS disease or Burkitt’s lymphoma/leukemia, and required a 6-month delay from transplant.
Axicabtagene Ciloleucel (Yescarta)
Kite pharmaceuticals (Santa Monica, CA), which was acquired by Gilead, developed CAR technology licensed technology from the National Cancer Institute (NCI). Axicabtagene ciloleucel, brand name Yescarta, is being evaluated in the ZUMA-1 trial ( NCT02614066) for refractory aggressive non-Hodgkin lymphoma26 (Table 1). Like Kymriah, it utilizes a murine CD19 binding domain, but unlike Kymriah it contains both a CD28 spacer and a CD28 co-stimulatory signaling domain.
Response durability in NHL is lower than that seen in ALL26; of 111 eligible patients, ORR was 82% and CR 59%; at a median of 15.4 months, 47% of patients were still responding and 40% in CR. Overall survival at 18 months was 52%.
Patients enrolled in the trial were >18 years old and had relapsed/refractory diffuse large B cell lymphoma (DLBCL), primary mediastinal B cell lymphoma (PMBCL) or transformed follicular lymphoma (TFL). Results from the NCI suggest that this construct can be effective in ‘double-hit’ lymphomas27, suggesting CAR T cells have a role in treating aggressive disease. However, as in ALL, the patient population most likely to benefit from CAR T cell administration, and the optimal timing of therapy, are not yet known.
Building a CAR T Cell Program
The process of CAR T cell administration involves lymphocyte collection, genetic manipulation, and reinfusion. There are complex logistics involved in collecting cells in the apheresis center, shipping to the manufacturer for production, and coordinating receipt of the product.
Several organizations provide standards and regulations for this process. The Federal Drug Administration (FDA) approves and regulates products, manages adverse event reporting, and oversee a Risk Evaluation and Mitigation (REMS) process in partnership with the manufacturers. The Foundation for the Accreditation of Cellular Therapy (FACT) provides standards for handling, processing, and tracking of cell therapy products28-30. The Center for International Blood and Marrow Transplant Research functions as a data repository and conduit between centers and the FDA. Finally, manufacturers themselves inspect apheresis and storage facilities, authorize individual centers to provide therapeutics, and provide both the products themselves and mechanisms for product shipping and tracking.
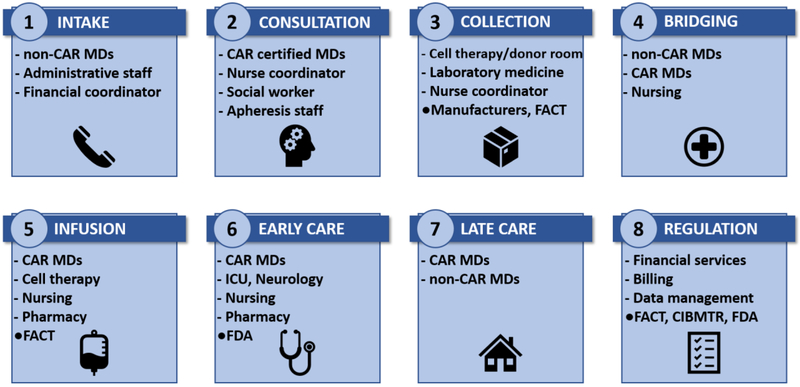
At MSKCC, we identified eight essential tasks that define the CAR T cell workflow and are required to successfully implement the delivery of commercial CAR T cell products in the clinic. We developed this workflow by leveraging the experience of both our investigational CAR T cell team (Cellular Therapeutic Center) and our adult and pediatric hematopoietic stem cell transplantation (HSCT) programs. The overall development was guided by a task force headed by a project managers and lead physicians. The task force included physician representative from leukemia, lymphoma, and bone marrow transplantation, nursing leadership, laboratory medicine, apheresis and transfusion medicine, legal services, financial services, social work and hospital administration. The eight tasks together with the personnel necessary to implement them are depicted in Figure 2.
Figure 2:
The eight essential steps required to establish a CAR T cell program and the personnel required to implement them. Key regulatory agencies involved in each step are denoted by a bullet.
Task 1: Patient Intake
Institutions must build an effective administrative infrastructure to facilitate patient referral, selection, and evaluation. This includes administrative staff to field calls and intake, nursing staff for triage and coordination, and financial and insurance advisers to ensure eligibility. While common to all medical enterprise, intake and pre-screening are particularly important for CAR T cell, as demand is likely to be high, eligibility is limited, and the availability of CAR T cells will at least initially be limited to a small number of centers.
Task 2: CAR T Cell Consultation Service:
We next developed a CAR T cell consultation service to handle patient selection and treatment. The consultation service consists of physicians, nurses, and social workers with expertise in CAR T cell therapy. Specialized physicians must be trained and registered with both the institution and manufacturers as CAR certified MDs who are authorized to prescribe CAR products and manage CAR patients.
A key task of the consultation service includes assessing patients for therapy. Appropriate candidates must have disease that will benefit from CAR T cell treatment, fit the labeling indication for the product, be well enough to receive therapy, have a caregiver, be able to stay within 2 hours of the treating institution for one month, and have insurance coverage or other financing for treatment. Disease burden at time of evaluation is a critical consideration, as patients with low burden of disease experience fewer treatment related toxicities and appear to derive more benefit from treatment13.
If investigational cell therapy products are available, patients must be triaged between the commercially available products and clinical trials. In some cases, this will be straightforward and based on product labeling; for example, a 27-year-old with ALL is not eligible for Kymriah, and an adult with Mantle Cell Lymphoma is not eligible for Yescarta. At other times, patients will be eligible for commercial CAR products, standard of care treatment, and clinical trials. Therefore, it is important to create a process by which patients and physicians can decide on the appropriate therapy.
Of particular importance is that patients continue to receive therapy that is most appropriate for their disease; thus, for example, patients with chemosensitive relapsed DLBCL should be treated with an autologous HSCT after high dose BEAM until the appropriate randomized trials of CAR T vs autologous HSCT have been performed. At our institution, candidates are reviewed at a weekly conference which brings together CAR certified MDs and disease-focused specialists.
Task 3: CAR T Cell Collection, Ordering, Shipping and Receiving
CAR T cell therapy requires an apheresis center and cell storage facility where T cells can be harvested, maintained, and easily transferred. This requires training of apheresis personnel, cell therapy technologists, administrative personnel, and nursing coordinators to process CAR products. Such centers and the products they generate are regulated by and must be registered with the FDA.
Despite concerns regarding leukopenia in these heavily pre-treated patients, minimal required collections can be successful in more than 90% of patients, with optimal yields in approximately 70% 31. There are, however, minimal requirements for initiation of collection; the Novartis product, for example, requires absolute lymphocyte counts > 500 mcL/L or absolute CD3+ T cell counts > 150 mcL/L.
Both Novartis and Kite have an extensive contracting process to establish technical and quality agreements for apheresis and cell processing facilities. The goal of these agreements is to ensure a high-quality cell product is delivered reliably to the manufacturer. Manufacturers work with the institution to define processes for tracking and verifying product identity, manipulating and storing products, and training personnel. Importantly, cell manipulation and freezing protocols must be reconciled with technical standards established by each company without significantly disrupting existing practice. This is a thorough and time-consuming process which involves on-site inspections by the manufacturer and, in certain cases, mock shipments.
Kymriah, but not Yescarta, can be collected, frozen, and stored for later use, a strategy Novartis has termed “at risk collection.” The acceptable duration of storage is validated up to 9 months per the manufacturer. Similarly, the optimal timing for collection is not known, although theoretically early collection before patients are heavily pre-treated and leukopenic may be beneficial.
Once collected, apheresis products are shipped to a commercial facility where T cells are isolated, activated, genetically modified with a CAR-encoding vector, and expanded prior to cryopreservation. Our own institutional process of clinical CAR- T manufacturing is reviewed in Wang et al32. After processing, they are shipped back to the originating institution. One critical area of focus, and a key focus of FACT standards, is ensuring chain of custody of the cell product. Staff must be trained on the software programs developed by each product manufacturer which coordinates shipping and ensures product identity.
Task 4: Bridging Strategy
Average turn-around time for initial apheresis to return of cells to the clinical site is 17 days for Yescarta33, with some products requiring 2-4 weeks before minimum cell numbers are obtained; slow expansion, insurance authorization and treatment planning can extend the bridging period to 1-2 months. While time-to-infusion has been drastically reduced, many patients have aggressive malignancies which must be carefully managed during insurance authorization, apheresis and manufacture. In selecting patients for CAR T cell therapy, clinicians must evaluate the pace of disease progression, the potential for near-term complications, and the expected date of infusion.
This process is managed in collaboration between the disease-specific primary oncologist and the CAR T cell consultation service, with nursing and administrative staff in support. Alternative therapies for patients who suffer manufacturing failures or disease complications should be considered; for example, in ZUMA-1, 101 of 111 patients ultimately received therapy26, with one manufacturing failure, one rapid disease progression, and several interval adverse events and other medical complications.
Two to seven days prior to infusion, patients receive a lymphodepleting conditioning regimen to enhance CAR T cell function and engraftment34,35. This requires the development of order sets specific to the conditioning regimen used for each commercial product.
While there are slight differences in the conditioning regimens use for different products and trials, investigators have coalesced around low-dose cyclophosphamide and fludarabine, as utilized in JULIET and ZUMA-1. The optimal conditioning regimen has not been rigorously defined, but experiences at the NCI10 and Fred Hutchinson Cancer Center36 are illustrative, with more aggressively lymphodepleting regimens associated with both increased expansion and toxicity.
Task 5: CAR T-Cell Infusion
Infusion requires the development of order sets, nursing standard of practice protocols, and documentation and verification procedures for administration. As part of FACT accreditation, electronic cell therapy orders must be developed for collection and thaw/infusion products and must be recorded in chemotherapy administration records. Therefore, pharmacy may track and register cell products and a work-flow for pharmacy involvement in product administration should be developed.
Consents must be developed for cell collection and infusion. Although specific consents are not mandated by regulatory agencies, the complex process and significant toxicity suggests a thorough patient consent process should be a central part of CAR T cell administration.
Task 6, Post Infusion Care (Day 0 to 30):
After infusion, patients are at risk for CAR T cell related adverse effects. The Cytokine Release Syndrome (CRS)12,37,38 occurs on a spectrum from low grade fever, which may not require any treatment, to fulminant multi-organ failure, which requires intensive care and aggressive immunosuppression. In some studies, approximately 10-15% of CAR T cell patients have required vasopressor support for hypotension and/or mechanical ventilation36,39. The mainstays of treatment for severe CRS include the anti-IL6R antibody Tocilizumab, high-dose steroids, and supportive care delivered in an intensive care unit40.
In addition to CRS, a syndrome of neurologic toxicity has been described41,42. Severe neurologic toxicities include encephalopathy, nerve palsies, and seizures, and have resulted in patient deaths43,44; these are often preceded by more subtle deficits in complex commands such as hand-writing, word-finding, and attention40. Information on the appropriate clinical management of CRS and other CAR T toxicities is both extensively reviewed elsewhere40,45 and is available on the package insert of approved therapies.
Subspecialists in Neurology and Intensive Care must be educated about CAR specific toxicities. We have developed management guidelines to harmonize treatment of CRS and neurotoxicity across departments and between commercial products and clinical trials. However, different products and disease indications have different rates and patterns of toxicity, and clinical judgement must be used in applying guidelines. Tisagenlecleucel induced severe CRS (Grade 3 or 4 by the UPenn toxicity criteria) in 48% of pediatric and young adult ALL patients in ELIANA24 and 28% of adult DLBCL patients in JULIET25, while ZUMA-1 reported a 13% rate (by NCI Common Terminology Criteria for Adverse Events) for axicabtagene ciloleucel in adult DLBCL.
CRS correlates with disease burden in pediatric trials46. Similarly, in adult ALL trials, severe CRS has only been observed in patients with morphologic evidence of disease at time of T cell infusion (> 5% bone marrow blasts) 13,47. Therefore, patients with a low burden of disease but a high risk of relapse may be optimal candidates for CAR therapy.
Nurses and ancillary medical personnel must also be educated about the recognition of CAR T cell specific care and toxicities, including CRS and neurotoxicity. Nursing units with experience in managing leukemia, lymphoma, and transplant patients are also well suited to handling more common issues including infection48 and transfusion. All physicians, mid-level providers, pharmacists and nurses who will interact with CAR T cell patients must undergo FDA mandated training as part of a Risk Evaluation Mitigation Strategy (REMS). This includes training which is provided by manufacturers on CAR-specific toxicity management with a knowledge assessment and the ability to audit these assessments if requested by the FDA as part of an adverse event assessment.
By FDA mandate, the inpatient pharmacy will need to stock at least two doses of Tocilizumab for each patient prior to CAR T cell administration, and ordering protocols and order sets will need to be developed to assure timely administration of both anti-IL6 therapy as well as corticosteroids and supportive medications as needed for CRS.
Each institution must undertake a thoughtful assessment of anticipated patient volumes, which depend on referral rates, relapse and treatment patterns, average length of stay, and relapse and complication rates. Although neither of the two products currently approved require an up-front hospitalization, both carry a significant risk of neutropenic fever, CRS, and neurotoxicity, resulting in an average a length-of-stay of 10-14 days. The need for inpatient admission, intensive care, and subspecialty care (such as neurology consultation) needs to be anticipated and requires planning by multiple departments.
Outpatient administration and management of patients undergoing CAR T cell therapy is feasible but requires well-staffed, dedicated outpatient units like those used to monitor outpatient autologous HCT recipients. Older patients, patients with bulky disease and patients with more comorbidities are likely to be at higher risk for complications and thus closer monitoring will be reasonable. Since a large proportion of patients will be coming from a significant distance, provisions must be made for lodging, transportation and living expenses. Patients and caregivers must be educated on CAR T toxicities and are given a manufacturer-provided wallet card describing symptoms that warrant immediate evaluation, such as fevers which can herald the onset of CRS or infection.
Task 7, Post-Infusion Care (Day 30 Onwards):
Documentation and hand-off procedures must be established to transition successfully treated patients to the outpatient setting. Order sets, standards of practice, and documentation must be developed for this purpose. The role of CAR T cell consultation and disease-specific groups must be delineated in patient monitoring and follow up. As discussed below, patients must be assessed for disease response at one month.
The use of CAR T cells in R/R B-ALL raises the question if subsequent allogeneic stem cell transplant is needed for durable remission. The University of Pennsylvania experience with adult and pediatric ALL showed long-term persistence of CAR cells, and only 9% patients went on to receive all-HSCT15. Similarly, the recently published MSKCC experience did not demonstrate a survival difference among patients with MRD negative complete response after CAR therapy who underwent transplant compared to those who did not13. Prospective randomized control trials comparing a “bridge-to-transplant” compared to CAR-only approach are not available but will be quite informative as this question is key to the successful use of this technology.
When patients do relapse, it is often due to CD19 undetectable disease with detectable CAR T cells in the peripheral blood46. Lymphoid cells can also express CD19 isoforms that do not contain CAR binding domains49. In either case, re-treatment with CD19 CAR would not have clinical activity; however, alternative targets such as CD2050 and CD2251,52 are in development.
Task 8, Financing, Regulatory and Reporting Requirements:
Administrative personnel will need to be educated on the billing and reimbursement protocols for CAR therapy. Novartis has announced a unique payment paradigm for Kymriah, which carries a nearly half-million price tag contingent upon response at one month53. CRs are thankfully quite high, however, suggesting hospitals will face the full costs for at least 4/5th of patients. Yescarta is priced at $375,000 per patient. Notably, these costs account only for the therapy itself, and do not account for hospitalization and numerous ancillary processes required for CAR T cell therapy (“parts, not labor”). Extrapolating from experience with bone marrow transplantation, management of patient hospitalization and complications is likely to contribute at least another $200,000-$500,000 depending on complication rates 54, which brings the total price tag close to one million dollars. Private payors will likely announce strict, label-specific requirements, while public payors will need to create new Diagnosis Related Groups and other funding mechanisms to accommodate cost. Regardless, institutions must be prepared to face significant downside financial risk.
From a regulatory perspective, gene therapy that is deemed “high risk,” including the use of integrating vectors, carries an FDA requirement to track patients for up to 15 years after administration. This requirement stems from potential oncogenic transformation that has been seen in several patients infused with genetically modified hematopoietic stem cells used for the treatment of primary immune deficiency55. However, oncogenic transformation in mature T cells has not been observed56,57.
Data management personnel are needed for obligatory data reporting on outcomes, particularly severe adverse events. FACT recommends that elements of CIBMTR cellular therapy data forms, including patient demographics and outcomes, are captured at regular follow-up intervals. Manufacturers have established reporting requirements for CAR-related toxicities: all incidents of CRS and neurotoxicity must be reported, and manufacturers also encourage reporting of severe and unusual toxicities. Events may be reported directly to the FDA via MedWatch or to the manufacturers.
Conclusion
Built on decades of scientific advancement and clinical development, CAR T cells are a new treatment paradigm for previously intractable malignancies. While CAR T cells were initially developed in a few academic centers, they have been approved at a time where no center has treated more than 400 patients. Each institution that initiates a CAR therapy program will face both clinical and administrative challenges, and the next decade will no doubt bring significant knowledge of the optimal use and management of these powerful tools. Despite significant financial, regulatory, and administrative barriers, we must not lose sight of the tremendous benefit that CAR T cells, skillfully deployed and thoughtfully applied, can bring to our patients.
Highlights:
Commercial Chimeric Antigen Receptor T cell products are efficacious, but the delivery of cell products requires significant planning and coordination
A consultation service composed CAR-certified physicians identifies and manages patients
Apheresis requires significant coordination with manufacturers, including site inspection and sample tracking
Toxicity management requires REMs training for all providers, and participation of ICU and neurology specialists
Regulatory standards are provided by the FDA, CIBMTR, FACT and product manufacturers
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Bibliography
- 1.Sadelain M Chimeric Antigen Receptors: A Paradigm Shift in Immunotherapy. Annu Rev Cancer Biol. 2017;1:447–466. doi: 10.1146/annurev-cancerbio-050216-034351. [DOI] [Google Scholar]
- 2.Morgan R a, Dudley ME, Wunderlich JR, et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science (80-). 2006;314(5796):126–129. doi: 10.1126/science.1129003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rosenberg SA, John R, Yang JC, et al. Treatment of Patients With Metastatic Melanoma With Autologous Tumor-Infiltrating Lymphocytes and Interleukin 2. J Natl Cancer Inst. 1994;86(15):1159–1166. [DOI] [PubMed] [Google Scholar]
- 4.Eshhar Z, Waks T, Gross G, Schindler DG. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci U S A. 1993;90(2):720–724. doi: 10.1073/pnas.90.2.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eshhar Z, Waks T, Gross G. The emergence of T-bodies/CAR T cells. Cancer J. 2014;20(2):123–126. doi: 10.1097/PPO.0000000000000027. [DOI] [PubMed] [Google Scholar]
- 6.Miller AD. Retrovirus Packaging Cells. Hum Gene Ther. 1990;14:5–14. [DOI] [PubMed] [Google Scholar]
- 7.Finney HM, Lawson AD, Bebbington CR, Weir AN. Chimeric receptors providing both primary and costimulatory signaling in T cells from a single gene product. J Immunol. 1998;161(6):2791–2797. http://www.ncbi.nlm.nih.gov/pubmed/9743337. Accessed August 15, 2017. [PubMed] [Google Scholar]
- 8.Maher J, Brentjens RJ, Gunset G, Rivière I, Sadelain M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotechnol. 2002;20(1):70–75. doi: 10.1038/nbt0102-70. [DOI] [PubMed] [Google Scholar]
- 9.Kochenderfer JN, Wilson W, Janik J, et al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood. 2010;116(20):4099–4103. doi: 10.1182/blood-2010-04-281931.The. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kochenderfer JN, Dudley ME, Kassim SH, et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J Clin Oncol. 2015;33(6):540–549. doi: 10.1200/JCO.2014.56.2025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schuster SJ, Svoboda J, Chong EA, et al. Chimeric Antigen Receptor T Cells in Refractory B-Cell Lymphomas. N Engl J Med. 2017:NEJMoa1708566. doi: 10.1056/NEJMoa1708566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Davila ML, Riviere I, Wang X, et al. Efficacy and Toxicity Management of 19-28z CAR T Cell Therapy in B Cell Acute Lymphoblastic Leukemia. Sci Transl Med. 2014;6(224):224ra25–224ra25. doi: 10.1126/scitranslmed.3008226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Park JH, Rivière I, Gonen M, et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N Engl J Med. 2018;378(5):449–459. doi: 10.1056/NEJMoa1709919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Grupp S a, Kalos M, Barrett D, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368(16):1509–1518. doi: 10.1056/NEJMoa1215134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maude SL, Laetsch TW, Buechner J, et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N Engl J Med. 2018;378(5):439–448. doi: 10.1056/NEJMoa1709866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Porter DL, Levine BL, Kalos M, Bagg A, June CH. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med. 2011;365(8):725–733. doi: 10.1056/NEJMoa1103849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hombach A, Sent D, Schneider C, Heuser C. Receptors CD28 Costimulation Is Required for Interleukin 2 Secretion and Receptor-mediated T-Cell Proliferation but Does Not Affect Receptor-mediated Target Cell. Cancer Res. 2001;61:1976–1982. http://cancerres.aacrjournals.org/content/61/5/1976.short. Accessed August 11, 2014. [PubMed] [Google Scholar]
- 18.Posey AD, Fraietta J, Lee J, Scholler J, Zhao Y, June CH. Distinct Signaling By Chimeric Antigen Receptors (CARs) Containing CD28 Signaling Domain Versus 4-1BB In Primary Human T Cells. Blood. 2013;122(21). http://www.bloodjournal.org/content/122/21/2902?sso-checked=true. Accessed August 15, 2017. [Google Scholar]
- 19.Song DG, Ye Q, Carpenito C, et al. In vivo persistence, tumor localization, and antitumor activity of CAR-engineered T cells is enhanced by costimulatory signaling through CD137 (4-1BB). Cancer Res. 2011;71(13):4617–4627. doi: 10.1158/0008-5472.CAN-ll-0422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhao Z, Condomines M, van der Stegen SJC, et al. Structural Design of Engineered Costimulation Determines Tumor Rejection Kinetics and Persistence of CAR T Cells. Cancer Cell. 2015;28(4):415–428. doi: 10.1016/j.ccell.2015.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mamonkin M, Mukherjee M, Srinivasan M, et al. Reversible Transgene Expression Reduces Fratricide and Permits 4-1BB Costimulation of CAR T Cells Directed to T-Cell Malignancies. Cancer Immunol Res. October 2017:canimm.0126.2017. doi: 10.1158/2326-6066.CIR-17-0126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Long AH, Haso WM, Shern JF, et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med. 2015;21(6):581–590. doi: 10.1038/nm.3838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kawalekar OU, O’Connor RS, Fraietta JA, et al. Distinct Signaling of Coreceptors Regulates Specific Metabolism Pathways and Impacts Memory Development in CAR T Cells. Immunity. 2016;44(2):380–390. doi: 10.1016/j.immuni.2016.01.021. [DOI] [PubMed] [Google Scholar]
- 24.Buechner J, Grupp SA, Maude SL, et al. Global Registration Trial Of Efficacy And Safety Of Ctl019 In Pediatric And Young Adult Patients With Relapsed/Refractory (R/R) Acute Lymphoblastic Leukemia (All): Update To The Interim Analysis. In: European Hematology Association Annual Meeting; 2017. https://learningcenter.ehaweb.org/eha/2017/22nd/181763/stephan.a.grupp.global.registration.trial.of.efficacy.and.safety.of.ctl019.in.html?f=m3. Accessed August 18, 2017. [Google Scholar]
- 25.Schuster SJ, Bishop MR, Tam CS, et al. Primary Analysis of Juliet: A Global, Pivotal, Phase 2 Trial of CTL019 in Adult Patients with Relapsed or Refractory Diffuse Large B-Cell Lymphoma. Am Soc Hematol Annu Meet. 2017. https://ash.confex.com/ash/2017/webprogram/Paper105399.html. Accessed December 10, 2017.
- 26.Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N Engl J Med. 2017:NEJMoa1707447. doi: 10.1056/NEJMoa1707447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kochenderfer JN, Somerville RPT, Lu T, et al. Lymphoma Remissions Caused by Anti-CD19 Chimeric Antigen Receptor T Cells Are Associated With High Serum Interleukin-15 Levels. J Clin Oncol. 2017;35(16):1803–1813. doi: 10.1200/JCO.2016.71.3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maus MV, Nikiforow S The why, what, and how of the new FACT standards for immune effector cells. J Immunother Cancer. 2017;5(1):36. doi: 10.1186/s40425-017-0239-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Foundation for the Accreditation of Cellular Therapy. Standards for Immune Effector Cells, First Edition. http://www.factwebsite.org/Standards/. Published 2017. Accessed January 11, 2017. [Google Scholar]
- 30.Therapy F for the A of C. Immune Effector Cells Accreditation Manual, First Edition. http://www.factwebsite.org/Standards/. Published 2017. Accessed January 11, 2017. [Google Scholar]
- 31.Allen ES, Stroncek DF, Ren J, et al. Autologous lymphapheresis for the production of chimeric antigen receptor T cells. Transfusion. 2017;57(5):1133–1141. doi: 10.1111/trf.14003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang X, Rivière I. Clinical manufacturing of CAR T cells: foundation of a promising therapy. Mol Ther — Oncolytics. 2016;3(16015):1–7. doi: 10.1038/mto.2016.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Locke FL, Neelapu SS, Bartlett NL, et al. Abstract CT019: Primary results from ZUMA-1: a pivotal trial of axicabtagene ciloleucel (axicel; KTE-C19) in patients with refractory aggressive non-Hodgkin lymphoma (NHL). Cancer Res. 2017;77(13 Supplement). http://cancerres.aacrjournals.org/content/77/13_Supplement/CT019. Accessed August 16, 2017. [Google Scholar]
- 34.Restifo NP, Dudley ME, Rosenberg S a. Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol. 2012;12(4):269–281. doi: 10.1038/nri3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Klebanoff C a, Khong HT, Antony P a, Palmer DC, Restifo NP. Sinks, suppressors and antigen presenters: how lymphodepletion enhances T cell-mediated tumor immunotherapy. Trends Immunol. 2005;26(2):111–117. doi: 10.1016/j.it.2004.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Turtle CJ, Hanafi L, Berger C, et al. Immunotherapy of non-Hodgkin ’ s lymphoma with a defined ratio of CD8 + and CD4 + CD19-specific chimeric antigen receptor – modified T cells. Sci Transl Med. 2016;8(355). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Brentjens R, Davila M, Riviere I, Park J, Wang X. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci Transl Med. 2013;5(177):177ra38. doi: 10.1126/scitranslmed.3005930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kochenderfer JN, Dudley ME, Feldman S a, et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood. 2012;119(12):2709–2720. doi: 10.1182/blood-2011-10-384388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kochenderfer JN, Somerville RPT, Lu T, et al. Lymphoma remissions caused by anti-CD19 chimeric antigen receptor T cells are associated with high serum interleukin-15 levels. J Clin Oncol. 2017;35(16):1803–1813. doi: 10.1200/JCO.2016.71.3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Neelapu SS, Tummala S, Kebriaei P, et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Nat Rev Clin Oncol. September 2017:nrclinonc.2017.148. doi: 10.1038/nrclinonc.2017.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Grupp SA, Porter DL, Teachey DT, et al. CD19-Redirected Chimeric Antigen Receptor T (CART19) Cells Induce a Cytokine Release Syndrome (CRS) and Induction of Treatable Macrophage Activation Syndrome (MAS) That Can Be Managed by the IL-6 Antagonist Tocilizumab (toc). Blood. 2015;120(21). http://www.bloodjournal.org/content/120/21/2604?sso-checked=true. Accessed September 16, 2017. [Google Scholar]
- 42.Maude SL, Barrett D, Teachey DT, Grupp SA. Managing cytokine release syndrome associated with novel T cell-engaging therapies. Cancer J. 2014;20(2):119–122. doi: 10.1097/PPO.0000000000000035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Maude SL, Frey N, Shaw PA, et al. Chimeric Antigen Receptor T Cells for Sustained Remissions in Leukemia. N Engl J Med. 2014;371(16):1507–1517. doi: 10.1056/NEJMoa1407222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Davila ML, Riviere I, Wang X, et al. Efficacy and Toxicity Management of 19-28z CAR T Cell Therapy in B Cell Acute Lymphoblastic Leukemia. Sci Transl Med. 2014;6(224):224ra25–224ra25. doi: 10.1126/scitranslmed.3008226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Brudno JN, Kochenderfer JN. Toxicities of chimeric antigen receptor T cells : recognition and management. Blood. 2016;127(26):3321–3331. doi: 10.1182/blood-2016-04-703751.CAR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Porter DL, Hwang W, Frey NV, et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med. 2015;7(303):303ra139. doi: 10.1126/scitranslmed.aac5415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Park Jae Hong, Riviere Isabelle, Wang Xiuyan, Purdon Terence, Sadelain Michel RJB. Impact of Disease Burden on Long-Term Outcome of 19-28z CAR Modified T Cells in Adult Patients with Relapsed, Refractory B-Cell ALL. In: American Society for Clinical Oncology Annual Meeting ; 2016:Abstract #7003. http://meetinglibrary.asco.org/record/122672/abstract. Accessed August 16, 2017. [Google Scholar]
- 48.Hill JA, Li D, Hay KA, et al. Infectious complications of CD19-targeted chimeric antigen receptor-modified T cell immunotherapy. Blood. October 2017:blood-2017-07-793760. doi: 10.1182/blood-2017-07-793760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sotillo E, Barrett DM, Black KL, et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy. Cancer Discov. 2015;5(12):1282–1295. doi: 10.1158/2159-8290.CD-15-1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Till BG, Jensen MC, Wang J, et al. antigen receptor with both CD28 and 4-1BB domains : pilot clinical trial results CD20-specific adoptive immunotherapy for lymphoma using a chimeric antigen receptor with both CD28 and 4-1BB domains : pilot clinical trial results. 2014;119(17):3940–3950. doi: 10.1182/blood-2011-10-387969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shah NN, Stetler-Stevenson M, Yuan CM, et al. Minimal Residual Disease Negative Complete Remissions Following Anti-CD22 Chimeric Antigen Receptor (CAR) in Children and Young Adults with Relapsed/Refractory Acute Lymphoblastic Leukemia (ALL). Blood. 2016;128(22). http://www.bloodjournal.org/content/128/22/650?sso-checked=true. Accessed September 18, 2017. [Google Scholar]
- 52.Fry TJ, Shah NN, Orentas RJ, et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. November 2017:nm.4441. doi: 10.1038/nm.4441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bach PB, Giralt SA, Saltz LB. FDA Approval of Tisagenlecleucel: Promise and Complexities of a $475 000 Cancer Drug. Jama. 2017;10017:0–1. doi: 10.1001/JAMA.2017.15218. [DOI] [PubMed] [Google Scholar]
- 54.Perales M, Bonafede M, Cai Q, et al. Real-World Economic Burden Associated with Transplant Related Complications. Biol Blood Marrow Transplant. 2017;23(10):1788–1794. doi: 10.1016/j.bbmt.2017.06.017. [DOI] [PubMed] [Google Scholar]
- 55.Hacein-Bey-Abina S, Von Kalle C, Schmidt M, et al. LMO2-Associated Clonal T Cell Proliferation in Two Patients after Gene Therapy for SCID-X1. Science (80-). 2003;302(5644):415–419. doi: 10.1126/science.1088547. [DOI] [PubMed] [Google Scholar]
- 56.Wang GP, Levine BL, Binder GK, et al. Analysis of lentiviral vector integration in HIV+ study subjects receiving autologous infusions of gene modified CD4+ T cells. Mol Ther. 2009;17(5):844–850. doi: 10.1038/mt.2009.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Davila ML, Sadelain M. Biology and clinical application of CAR T cells for B cell malignancies. Int J Hematol. 2016;104(1):6–17. doi: 10.1007/s12185-016-2039-6. [DOI] [PMC free article] [PubMed] [Google Scholar]