Abstract
Purpose
Inguinal lymph node metastasis (LNM) is one of the most significant prognostic factors for patients with penile cancer. This study aimed to identify potential predictors of inguinal LNM.
Patients and methods
A comprehensive search of the PubMed, Embase, and Cochrane Library databases for studies that reported predictors of inguinal LNM in penile cancer was performed. Finally, we selected 42 eligible studies with 4,802 patients. Accumulative analyses of odds ratios (ORs) and corresponding 95% confidence intervals (CIs) were performed. All analyses were performed by using Review Manager software version 5.3.
Results
Among the 4,802 patients, 1,706 (36%) were diagnosed with inguinal LNM. Predictors of LNM included two categories: tumor-associated biomarkers and invasive clinicopathologic characteristics. Biomarker-specific predictors: the program death ligand 1 (PD-L1) overexpression (OR=2.55, p=0.002), higher neutrophil-to-lymphocyte ratio (NLR) (OR=4.22, p=0.010), higher C-reactive protein (CRP) (OR=4.78, p<0.001), squamous cell carcinoma antigen (SCC-Ag) overexpression (OR=8.52, p<0.001), P53 protein overexpression (OR=3.57, p<0.001). Clinicopathological predictors: positive clinical lymph node (cN+) (OR=5.86, p<0.001), high-risk histopathological subtype (OR=14.63, p<0.001) and intermediate-risk subtype (OR=3.37, p<0.001), vertical growth pattern (OR=1.97, p=0.020), higher stage (AJCC: OR=3.66, p<0.001; UICC: OR=2.43, p<0.001), higher tumor grade (OR=3.37, p<0.001), tumor size (>3 cm) (OR=2.00, p=0.002), LVI (OR=3.37, p<0.001), invasion depth (>5 mm) (OR=2.58, p=0.002), nerve invasion (OR=2.84, p<0.001), corpora cavernosum invasion (OR=2.22, p<0.001), corpus spongiosum invasion (OR=1.73, p=0.002) and urethra invasion (OR=1.81, p=0.030).
Conclusion
Current meta-analysis conclusively identified valuable predictors of inguinal LNM for patients with penile cancer. However, high-quality studies are warranted to further validate our conclusions. The intrinsic link between these predictors needs to be further investigated to create an accurate mathematical prediction model for LNM.
Keywords: inguinal lymph node metastasis, penile cancer, predictor, meta-analysis
Introduction
Penile cancer is a rare malignant tumor, which results in significant physiological and psychological effects on patients.1 Inguinal lymph nodes are the first site of metastasis in penile carcinoma.2 The presence of inguinal lymph node metastasis (LNM) is one of the most significant prognostic factors for patients with penile cancer.3 Therefore, optimal management of inguinal lymph nodes is crucial for long-term survival after local treatment of the primary tumor.4
The best management of clinically negative nodes (cN0) is controversial because of the approximately 25% likelihood of micrometastatic disease,5,6 and complete detection of micro-metastases by current imaging techniques is difficult.7 Close surveillance, dynamic sentinel node biopsy, and modified lymphadenectomy were recommended in the treatment of patients with cN0 over the past decades, aiming to decrease the complications caused by radical inguinal lymph node dissection (ILND).4,8 But these methods remained a notable risk of missing micro-metastatic disease.9,10 Comparing these methods, prophylactic lymphadenectomy has the best overall survival benefit from early resection of occult metastasis.11,12 However, concurrent complications related to lymph drainage and wound healing are relatively high despite surgical modifications.13–15 Therefore, accurate inguinal LNM prediction could pinpoint patients who are the best candidates for inguinal lymphadenectomy, which could not only achieve the best survival rate for patients with occult metastasis but also avoid unnecessary treatment for patients with a low risk of developing LNM.
Given the rarity of penile cancer, prior studies that attempted to identify predictors of LNM had several methodological limitations, such as small series, single-center, and lack of randomized controlled trials. Moreover, the results of these studies were discrepant. Several predictive models had been developed to stratify the risk of developing LNM, such as Solsona risk groups and Hungerhuber risk stratification.16,17 Unfortunately, further validation denied the accuracy of these tools.18 Furthermore, the prognostic value of several potential biomarkers, such as PD-L1, P53 protein, NLR, CRP, SCC-Ag, Ki-67, and HPV DNA, had not been conclusively established. Hence, we performed this comprehensive meta-analysis to determine the significant predictors of LNM in penile cancer.
Methods
This meta-analysis was conducted according to the preferred reporting items for systematic review and meta-analyses (PRISMA) statement and was registered with PROSPERO (https://www.crd.york.ac.uk/PROSPERO ID: CRD42018107232).19 It was approved by the institutional review board before initiation. The need for ethical standard approval or informed consent was waived due to the nature of the research design.
Search strategy
In accordance with the PRISMA guidelines, a literature search was performed in January 2019 using PubMed, Embase, and the Cochrane Library. Search terms used included the following: (((penile cancer) or (penile tumor) or (penile neoplasm) or (penile squamous cell carcinoma))) AND ((inguinal lymph node metastasis) or (lymph node metastases) or (nodal metastasis) or (inguinal node metastasis) or (inguinal lymphadenopathy) or (inguinal lymphadenectasis) or (inguinal lymph node involvement)) and ((predictors) or (predictive factor)). All studies on this topic were reviewed, and related references of original studies were identified by manual search.
Inclusion and exclusion criteria
The PICOS (Population, Intervention, Comparator, Outcome, and Study design) principle was employed to define study eligibility. Studies that compared penile cancer patients who were pathologically diagnosed with LNM (P) after local treatment of primary tumor (I) to patients without LNM (C) to determine clinicopathologic predictors or biomarkers of inguinal LNM (O) using logistic regression analyses or providing original statistical data (S) were considered relevant to this systematic review and meta-analysis. Eligible studies were selected based on the following: 1) precise definition of potential predictors; 2) sufficient data: odds ratios (ORs) with 95% confidence interval (CIs) or credible original statistical data that could be used to calculate ORs and 95% CIs; 3) pathologically confirmed LNM; 4) moderate or high methodological quality studies according to the Newcastle–Ottawa scale;20 and 5) English studies with human subjects. Between two studies with similar research populations, study with larger sample size was chosen. All overlapping studies with different predictors were included. Relevant researches in the form of case reports, reviews, case series, editorials, or letters were excluded.
Data extraction
Data of identified studies were extracted by two independent reviewers (J.H. and J.B.C.). Discrepancies were resolved during a consensus meeting with a senior reviewer (X.Z.). The following information was extracted: author, year, country, sample size, LNM predictors, and follow-up period. We extracted the ORs with their 95% CIs directly if available in the article. Otherwise, we extracted the original statistical data to calculate the ORs and 95% CIs.
Quality of data assessment and risk of bias evaluation
Two independent reviewers evaluated the quality of the included studies using the Newcastle–Ottawa Quality Assessment Scale, which was designed to assess the quality of observational studies. A star system including nine scoring items was adapted to grade each study. A total score of 8 to 9 was defined as a high-quality study; 6 to 7, intermediate quality. Moreover, we evaluated the publication bias by visual inspection of funnel plots. We also performed a sensitivity analysis using the leave-one-out cross-validation to assess the stability of the present meta-analysis results.
Definition of predictors
Definitions of several predictors were recorded as previously published.21 Clinically positive inguinal lymph nodes (cN+) were defined as those that are palpable or visible with imaging examinations. Histological grade was divided into three groups: G1 (well-differentiated), G2 (moderately differentiated), and G3 (poorly differentiated). There were two TNM systems, including the American Joint Commission on Cancer (AJCC) and the Union for International Cancer Control (UICC). Both of them had several versions. We defined T2 and greater stage as higher stage. Growth pattern was classified as superficial or vertical; Invasion depth was measured from the intact basement membrane at the edge of the primary tumor to the deepest infiltrating tumor cell. LVI was defined as the presence of cancer embolus in the lymphatic or vascular lumen that was detected by immunohistochemical staining. Histopathological subtypes were classified as low risk (verrucous, papillary, and warty), intermediate risk (usual SCCs and mixed forms), and high risk (basaloid, sarcomatoid, adenosquamous, and poorly differentiated types) according to the European Association of Urology (EAU) guidelines.4 PD-L1, Ki-67, P53 protein, and HPV virus were measured in tumor. SCC-Ag, NLR, and CRP were measured in serum.
Statistical analysis
We performed this meta-analysis to identify potential predictors of LNM by pooling the predictor effect and its standard error, which was calculated from the available ORs and their 95% CIs or from original statistical data. The inverse variance method was adapted to evaluate the cumulative effects of these potential predictors. Statistical heterogeneity among the included studies was assessed using the Cochrane Q test and I2 statistic (I2<25%: no heterogeneity; I2=25–50%: moderate heterogeneity; I2>50%: large heterogeneity). The value of I2 indicated the degree of heterogeneity. A random-effects model was used when there was a large heterogeneity; otherwise, the fixed-effects model was used. The level of statistical significance was set at 0.05. The meta-analyses were performed using Review Manager (RevMan) software version 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen).
Results
Study population
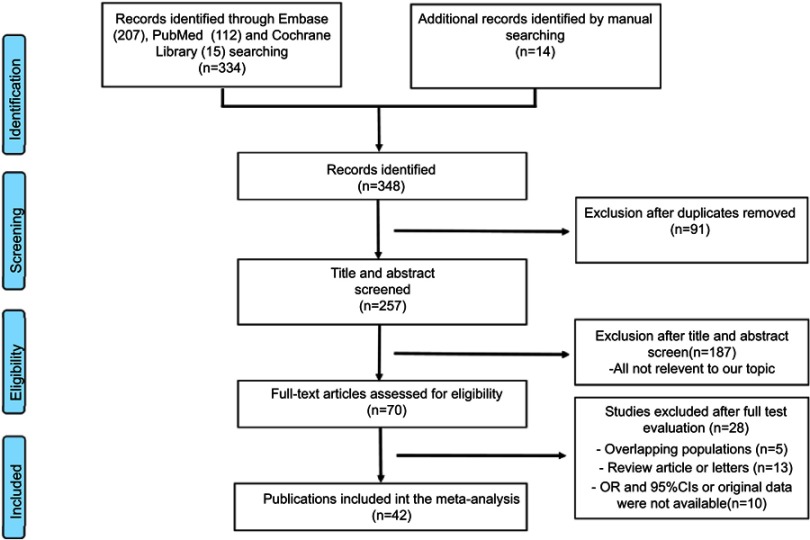
Finally, we selected 42 eligible studies.5,21–61 Among the included 4,802 patients, 1,706 (36%) were diagnosed with LNM. Figure 1 shows the study selection process. These studies were performed in the following geographical regions: Europe (n=12), South America (n=8), North America (n=10), Africa (n=1), Australia (n=1), and Asia (n=10). The characteristics of the included studies are provided in Table 1. The pooled results of the predictors are provided in Table 2. Original data are summarized in Tables S1–3. The symmetrical funnel plots showed in Figures S1 and S2 revealed low publication bias for these predictors.
Figure 1.
PRISMA flow chart.
Table 1.
Characteristics of included studies
| Studies (year) | Country | No. of patients | Median age, years (range) | No. of LNM | Median follow-up, Mo (range) | Included predictors | NOS score |
|---|---|---|---|---|---|---|---|
| Alkatout et al 201121 | Germany | 72 | 64 (34–91) | 34 | 35 (0–142) | Clinical lymph node, stage, grade, growth pattern, LVI | 8 |
| Azizi et al 201824 | America | 68 | 65 (53–69) | 45 | 36 (19–90) | Clinical lymph node, LVI, NLR | 8 |
| Bhagat et al 201025 | India | 53 | 50 (27–74) | 22 | 19 (9–65) | Age, clinical lymph node, stage, grade, LVI, invasion depth |
6 |
| Chalya et al 201526 | Tanzania | 236 | 47 (21–78) | 154 | 22 (3–61) | Grade, histopathological type, tumor size, LVI, urethra invasion | 6 |
| Chen et al 201227 | China | 56 | 53.42 (NR) | 14 | 27 (17–43) | Stage, grade | 6 |
| Cubilla et al 200128 | America | 61 | NR | 20 | NR | Histopathological type | 6 |
| Dai et al 200629 | China | 72 | 51 (27–81) | 23 | NR | Stage, grade, histopathological type | 7 |
| Emerson et al 200130 | America | 20 | 63 (40–81) | 8 | 28 (4–99) | Stage, grade, histopathological type | 6 |
| Ficarra et al 200531 | Italy | 175 | 62 (34–91) | 71 | NR | Age, clinical lymph node, stage, grade, tumor size, growth pattern, LVI, vascular invasion, lymphatic invasion, corpora cavernosa invasion, corpus spongiosum invasion, urethra invasion | 9 |
| Fonseca et al 201332 | Brazil | 82 | 58 (22–91) | 46 | 20 (1–71) | Stage, grade, LVI, nerve invasion, HPV infection | 8 |
| Ghazal et al 201323 | Germany | 51 | 63 (33–88) | 16 | 27 (NR) | Stage, grade | 8 |
| Graafland et al 201033 | Netherlands | 342 | 65 (26–96) | 68 | 32 (3–91) | Grade, corpora cavernosa invasion, corpus spongiosum invasion, LVI, urethra invasion | 9 |
| Guimares et al 200735 | Brazil | 125 | NR | 55 | 33 (1–453) | Age, race, clinical lymph node, stage, grade, LVI, corpora cavernosa invasion, Ki-67 | 8 |
| Guimares et al 200934 | Brazil | 333 | NR | 81 | 100 (1–453) | Histopathological type | 7 |
| Gunia et al 201236 | Germany | 92 | NR | 19 | NR | P53 | 6 |
| Hall et al 199837 | America | 46 | NR | 14 | NR | Stage, grade, invasion depth | 6 |
| Harmaya et al 201738 | Indonesia | 50 | NR | 25 | NR | Age, stage, grade, vascular invasion | 6 |
| Kroon et al 200840 | Netherlands | 56 | NR | 32 | NR | Stage, grade, vascular invasion | 7 |
| Lopes et al 199644 | Brazil | 145 | 53 (26–79) | 76 | 33 (1–453) | Age, race, clinical lymph node, stage, grade, lymphatic invasion, invasion depth, corpora cavernosa invasion, corpus spongiosum invasion, urethra invasion, HPV infection |
8 |
| Lopes et al 200243 | Brazil | 82 | 52 (27–77) | 42 | 89 (1–453) | P53 | 7 |
| Mannweiler et al 201345 | Austria | 72 | NR | 8 | 47 (5–265) | Clinical lymph node, growth pattern, lymphatic invasion, urethra invasion, invasion depth, HPV infection | 8 |
| Ornellas et al 200846 | Brazil | 196 | 57 (25–98) | 70 | 74 (1–93) | Stage, grade, invasion depth, LVI, corpora cavernosa invasion, corpus spongiosum invasion, urethra invasion, nerve invasion | 8 |
| Protzel et al 200747 | Germany | 28 | 69 (35–89) | 16 | 46 (2–105) | Clinical lymph node, stage, grade, Ki-67, HPV infection | 7 |
| Qu et al 201848 | Canada | 380 | 66 (29–99) | 63 | 54 (0–131) | Age, stage, grade, vascular invasion, histopathological type | 9 |
| Slaton et al 200150 | America | 48 | 51 (26–81) | 18 | 73 (23–154) | Stage, grade, invasion depth, vascular invasion | 8 |
| Termini et al 201552 | Brazil | 125 | NR | 44 | NR | Age, clinical lymph node, stage, grade, invasion depth, vascular invasion, corpora cavernosa invasion, corpus spongiosum invasion, urethra invasion, nerve invasion, HPV infection | 6 |
| Theodorescu et al 199653 | America | 42 | 62 (22–84) | 26 | 42 (1–168) | Age, grade, tumor size | 7 |
| Velazquez et al 200856 | Paraguay | 134 | 55 (24–82) | 66 | NR | Grade, nerve invasion, | 5 |
| Wang et al 20185 | China | 198 | 53 (20–84) | 96 | NR | Age, clinical lymph node, stage, grade, invasion depth, LVI, nerve invasion, histopathological type | 9 |
| Winters et al 201657 | America | 461 | 62 (52–71) | 111 | NR | Stage, grade, LVI | 9 |
| Zargar-Shoshtari et al 201559 | America | 57 | 60 (53–73) | 31 | 22 (NR) | Race, stage, grade, LVI, HPV infection, P53 | 7 |
| Li et al 201642 | China | 124 | 50 (25–86) | 60 | NR | CRP, SCC-Ag | 7 |
| Steffens et al 201322 | Germany | 79 | 65.2 (33–92) | 16 | 23 (NR) | CRP | 7 |
| Kasuga et al 201651 | Japan | 41 | 69 (68.5±11.8) | 9 | 34.7 (2.3–271.7) | NLR | 6 |
| Hungerhuber et al 200739 | Germany | 24 | NR | 16 | NR | SCC-Ag | 6 |
| Touloupidis et al 200754 | Greece | 16 | NR | 7 | 48 (24–84) | SCC-Ag | 6 |
| Laniado et al 200341 | British | 11 | NR | 7 | 36 (NR) | SCC-Ag | 6 |
| Deng et al 201658 | China | 116 | 53 (24–86) | 42 | NR | PD-L1 | 8 |
| Udager et al 201655 | America | 37 | NR | 11 | NR | PD-L1 | 7 |
| Ottenhof et al 201849 | Netherlands | 213 | NR | 68 | 100.7 (69.4–119.7) | PD-L1 | 8 |
| Zhu et al 200761 | China | 73 | 55 (27–75) | 30 | NR | Stage, grade, Ki-67, | 8 |
| Zhu et al 201060 | China | 110 | 54 (20–75) | 26 | NR | LVI, P53 | 8 |
Abbreviations: LNM, lymph node metastasis; NOS, Newcastle–Ottawa quality assessment scale; LVI, lymphovascular invasion; HPV, human papillomavirus; NR, not reported; PD-L1, program death ligand 1; SCC-Ag, squamous cell carcinoma antigen; NLR, neutrophil-to-lymphocyte ratio; CRP, C-reactive protein.
Table 2.
Pooled results of predictors for LNM
| Predictors | No. of studies | No. of patients (pre/non-pre) | Pooled OR [95%CI] | p | I2 (%) | Effects model |
|---|---|---|---|---|---|---|
| Tumor size | 4 | NR | 2.00 [1.29–3.10] | 0.002 | 0 | Fixed |
| Vertical growth pattern | 3 | 149/149 | 1.97 [1.13–3.43] | 0.02 | 0 | Fixed |
| Histopathological typea | 6 | 63/178 | 14.63 [6.40–33.42] | <0.001 | 0 | Fixed |
| Histopathological typeb | 6 | 799/178 | 3.37 [1.97–5.74] | <0.001 | 0 | Fixed |
| Higher tumor stagec | 8 | 757/431 | 3.66 [2.47–5.42] | <0.001 | 50 | Fixed |
| Higher tumor staged | 13 | 800/371 | 2.43 [1.80–3.26] | <0.001 | 0 | Fixed |
| Higher tumor grade | 25 | 1652/1028 | 3.37 [2.38–4.78] | <0.001 | 59 | Random |
| Lymphovascular invasion | 18 | 490/1638 | 3.37 [2.72–4.16] | <0.001 | 0 | Fixed |
| Invasion depth | 6 | 408/144 | 2.58 [1.42–4.64] | 0.002 | 38 | Fixed |
| Corpora cavernosa invasion | 6 | 385/630 | 2.22 [1.63–3.04] | <0.001 | 0 | Fixed |
| Corpus spongiosum invasion | 5 | 430/370 | 1.73 [1.22–2.46] | <0.001 | 0 | Fixed |
| Urethra invasion | 7 | 204/763 | 1.81 [1.07–3.05] | 0.03 | 59 | Random |
| Nerve invasion | 7 | 196/670 | 2.84 [1.99–4.04] | <0.001 | 7 | Fixed |
| PD-L1 | 3 | NR | 2.55 [1.40–4.64] | 0.002 | 2 | Fixed |
| P53 | 4 | 111/165 | 3.57 [1.93–6.62] | <0.001 | 0 | Fixed |
| SCC-Ag | 4 | 85/90 | 8.52 [4.09–17.78] | <0.001 | 0 | Fixed |
| CRP | 2 | 69/119 | 4.78 [2.48–9.20] | <0.001 | 0 | Fixed |
| NLR | 2 | 58/51 | 4.22 [1.36–13.09] | 0.01 | 0 | Fixed |
| Ki-67 | 3 | 112/106 | 2.70 [0.81–9.05] | 0.11 | 55 | Random |
| Race | 3 | 72/255 | 0.92 [0.52–1.63] | 0.77 | 0 | Fixed |
| Age | 9 | NR | 0.99 [0.95–1.03] | 0.65 | 29 | Fixed |
| Positive HPV infection | 6 | 208/285 | 0.85 [0.58–1.25] | 0.41 | 0 | Fixed |
Notes: aHigh-low risk group and bIntermediate-low risk group. cAJCC TNM stage system and dUICC TNM stage system.
Abbreviations: LNM, lymph node metastasis; OR: odds ratio; CI, confidence intervals; I2, the heterogeneity between studies; pre, predictors; NR, not reported; PD-L1, program death ligand 1; SCC-Ag, squamous cell carcinoma antigen; NLR, neutrophil-to-lymphocyte ratio; CRP, C-reactive protein.
Biomarker-specific predictors for LNM
Immune-related biomarkers: PD-L1, CRP, and NLR
Immune-related biomarkers as a predictor of LNM were reported in seven studies. For these predictors, heterogeneity was not observed. Cumulative analysis of these homogeneous data revealed that PD-L1 overexpression in tumor cells (OR 2.55, 95% CI 1.40–4.64; p=0.002), higher NLR in serum (OR 4.22, 95% CI 1.36–13.09; p=0.010) and higher level of CRP in serum (OR 4.78, 95% CI 2.48–9.20; p<0.001) are significant predictors of LNM (Figure 2A).
Figure 2.

Forest plots of biomarker-specific predictors. (A) Immune-related predictors. (B) SCC-Ag. (C) P53 protein. (D) Ki-67. (E) HPV infection.
SCC-Ag and P53 protein
SCC-Ag overexpression as a predictor was reported in four studies. There was no heterogeneity between these studies (I2=0%; p=0.88). Cumulative analysis of these data revealed that it (OR 8.52, 95% CI 4.09–17.78; p<0.001) is a significant predictor of LNM (Figure 2B). Similarly, there was no heterogeneity between four studies which reported the correlation between P53 and LNM (I2=0%; p=0.68). Cumulative analysis of these homogeneous data revealed that P53 overexpression (OR 3.57, 95% CI 1.93–6.62; p<0.001) is a significant predictor of LNM (Figure 2C).
Ki-67 and HPV infection status
Ki-67 overexpression as a predictor of LNM was reported in three studies. Heterogeneity was observed between the studies (I2=55%; p=0.11). Cumulative analysis of these data revealed that Ki-67 overexpression (OR 2.70, 95% CI 0.81–9.05; p=0.110) is not a significant predictor of LNM (Figure 2D). Six studies reported the association between HPV infection and LNM, in which 208 (42%) and 285 (58%) patients had positive and negative HPV infection, respectively. No heterogeneity was noted between the studies (I2=0%; p=0.49); Cumulative analysis of these homogeneous data demonstrated that HPV infection (OR 0.85, 95% CI 0.58–1.25; p=0.410) is not a significant predictor of LNM (Figure 2E).
Clinicopathological predictors for LNM
Histopathological type and growth pattern
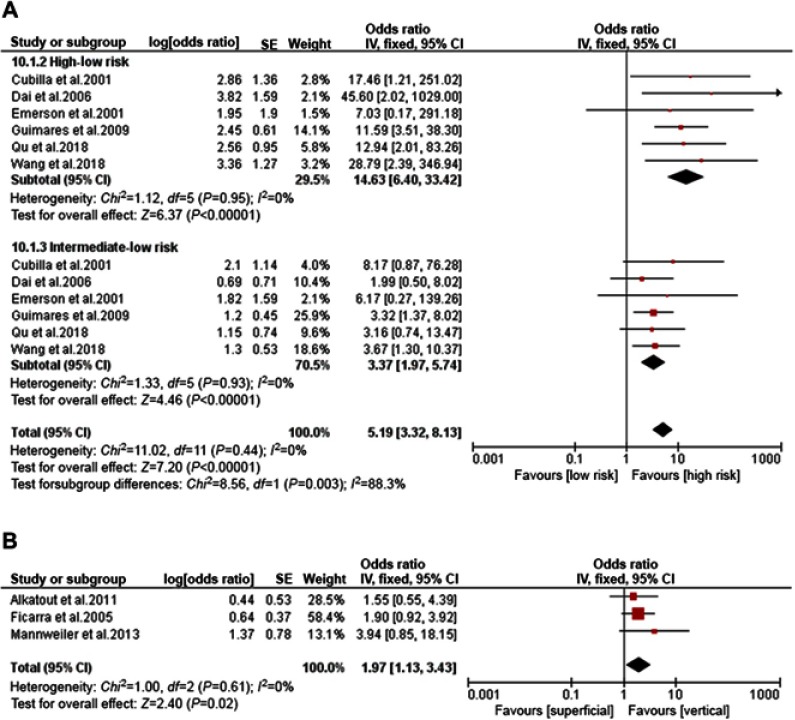
Histopathological type as a predictor of LNM was reported in six studies; no heterogeneity between the studies was noted (I2=0%). Accumulative analysis of these homogeneous data revealed that high-risk type (OR 14.63, 95% CI 6.40–33.42; p<0.001) and intermediate-risk type (OR 3.37, 95% CI 1.97–5.74; p<0.001) are significant predictors of LNM compared with low-risk type (Figure 3A). Three studies reported growth pattern-related LNM risk, which included 398 patients (vertical pattern 149 vs superficial pattern 149). Cumulative analysis of homogeneous data demonstrated that vertical growth pattern (OR 1.97, 95% CI 1.13–3.43; p=0.020) is a significant predictor of LNM (Figure 3B).
Figure 3.
Forest plots of histopathological type , growth pattern. . (A) Histopathological type. (B) Growth pattern.
Tumor stage: AJCC and UICC TNM stage system
Eight studies adapted the AJCC TNM system. We performed a subgroup analysis based on different versions (1997, 2002, 2010, and 2016). Heterogeneity of each subgroup was acceptable. Pooled results of all subgroups demonstrated that higher stage was a significant predictor of LNM (Figure 4). Thirteen studies adapted the UICC TNM system. Similarly, pooled results of subgroups demonstrated the same conclusion.
Figure 4.

Forest plots of different TNM systems. (A) American Joint Commission on Cancer (AJCC) TNM stage system. (B) Union for International Cancer Control (UICC) TNM stage system.
Tumor grade and tumor size
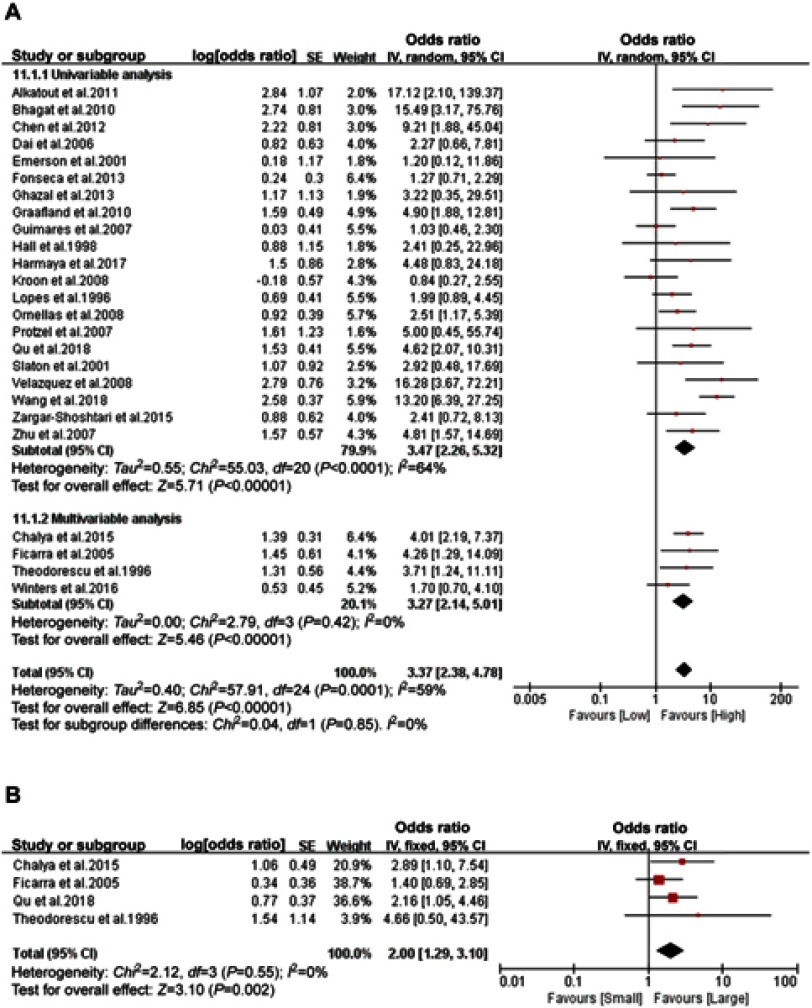
Among the 2,680 patients from 25 studies with ORs for tumor grade-related LNM risk, 1,652 (62%) and 1,028 (38%) had high-grade (G2, G3) and low-grade (G1) penile cancer, respectively. Accumulative analysis of available ORs demonstrated that high tumor grade (univariable subgroup: OR 3.47, 95% CI 2.26–5.32; p<0.001; multivariable subgroup: OR 3.27, 95% CI 2.14–5.01; p<0.001) is a significant predictor of LNM (Figure 5A). Heterogeneity between the studies in univariable subgroup was significant (I2=64%; p<0.001); tumor size as a predictor of LNM was reported in four studies. They used 3 cm as the cutoff value. No heterogeneity between the studies (I2=0%; p=0.55) was found, and cumulative analysis of homogeneous data revealed that tumor size (>3 cm) (OR 2.00, 95% CI 1.29–3.10; p=0.002) is a significant predictor of LNM (Figure 5B).
Figure 5.
Forest plots of tumor grade and size. (A) Tumor grade. (B) Tumor size.
Lymphovascular invasion, invasion depth, and nerve invasion
Among the 2,128 patients from 18 studies with ORs for LVI–related LNM risk, 490 (23%) and 1,638 (77%) had positive LVI and negative LVI, respectively. No between-study heterogeneity was observed in the two subgroups. Accumulative analysis of available ORs revealed that LVI (univariable subgroup: OR 4.44, 95% CI 3.12–6.31; p<0.001; multivariable subgroup: OR 2.88, 95% CI 2.20–3.75; p<0.001) is a significant predictor of LNM (Figure 6A). Invasion depth as a predictor of LNM was reported in six studies. They used 5 mm as the cutoff value. Moderate heterogeneity was observed between the studies (I2=38%; p=0.15); Cumulative analysis of the ORs revealed that invasion depth (>5 mm) (OR 2.58, 95% CI 1.42–4.69; p=0.002) is a significant predictor of LNM (Figure 6B). Nerve invasion as a predictor of LNM was reported in seven studies, in which 196 (23%) and 670 (77%) patients had positive and negative nerve invasion, respectively. No heterogeneity was noted between the studies (I2=7%; p=0.37). Cumulative analysis of these homogeneous data revealed that nerve invasion (OR 2.84, 95% CI 1.99–4.04; p<0.001) is a significant predictor of LNM (Figure 6C).
Figure 6.

Forest plots of lymphovascular invasion, invasion depth and nerve invasion. (A) Lymphovascular invasion (LVI). (B) Invasion depth. (C) Nerve invasion.
Cavernosum invasion, urethra invasion, age, and race
Six studies reported the association between corpora cavernosum invasion and LNM. The data were homogeneous (I2=0%; p=0.900) and cumulative results demonstrated that corpora cavernosum invasion (OR 2.22, 95% CI 1.63–3.04; p<0.001) is a significant predictor of LNM (Figure 7A). Similarly, cumulative analysis revealed that corpus spongiosum invasion (OR 1.73, 95% CI 1.22–2.46; p=0.002) is also a significant predictor of LNM (Figure 7B). Seven studies reported the association between urethra invasion and LNM. Pooled results demonstrated that urethra invasion (OR 1.81, 95% CI 1.07–3.05; p=0.030) is a significant predictor of LNM (Figure 7C). Heterogeneity was observed between included studies (I2=59%; p=0.02); Nine studies reported the association between age and LNM. Heterogeneity was moderate (I2=29%; p=0.19); Cumulative analysis of available ORs demonstrated that age (OR 0.99, 95% CI 0.95–1.03; p=0.65) was not a significant predictor of LNM (Figure 7D). Three studies reported the race-related LNM risk. Accumulative analysis of these homogeneous data revealed that race (OR 0.92, 95% CI 0.52–1.63; p=0.77) was not a significant predictor of LNM (Figure 7E).
Figure 7.

Forest plots of corpora cavernosa invasion, corpus spongiosum invasion, urethra invasion, age and race. (A) Corpora cavernosa invasion. (B) Corpus spongiosum invasion. (C) Urethra invasion. (D) Age. (E) Race.
Discussion
The effectiveness of current guidelines in managing inguinal lymph nodes of patients with penile cancer had been challenged by a prospective study.62 This prospective study revealed that over 80% of the patients, who were categorized as intermediate or high risk for developing LNM according to the current guidelines, accepted unnecessary prophylactic ILND. This inaccuracy may be caused by the small number of predictors included in the current guidelines. In addition, ILND exhibits serious surgical complications. To pinpoint patients who are the best candidates for receiving radical ILND, we conducted this comprehensive meta-analysis. Consequently, we identified numerous valuable predictors which could be classified into two categories, including tumor-associated biomarkers and invasive clinicopathologic characteristics. To our knowledge, this meta-analysis is the first to combine comprehensive and detailed evidence on extensive potential predictors of LNM for patients with penile cancer.
It is well established that inflammation contributes a lot to the initiation and progression of cancers.63 Several immune-related predictors, such as NLR, CRP, and PD-L1, were conclusively identified in this meta-analysis. NLR combining of neutrophilia and lymphopenia represents systemic inflammatory response and immune response. It is an independent predictor of poor prognosis for several solid cancers including castration-resistant prostate cancer, cervical adenocarcinoma, lung cancer, and esophageal carcinoma.64–67 Similarly, a study enrolling 84 consecutive penile cancer patients investigated the association between NLR with pathologic LNM and prognosis.24 This study demonstrated that NLR was an independent predictor of overall survival. In addition, patients with an elevated NLR were related to higher risk of pathologic LNM, although this relationship was not significant in adjusted analysis. CRP is an acute-phase protein produced almost exclusively by the liver. Its prognostic value for patients with penile cancer has been demonstrated.22,42 According to the I2 statistics, there was no heterogeneity for these biomarkers between included studies. Furthermore, both NLR and CRP are measured easily and economically in clinical practice. These features may facilitate the clinical application of these two biomarkers.
Another important and therapeutic potential biomarker is PD-L1, a transmembrane protein with the ability to suppress host immune system. It is a critical component of tumor-specific immune resistance mechanisms.68 Cancer immunotherapy by targeting PD-L1 can improve overall survival for patients with advanced cancer.69 However, evidence on cancer immunotherapy for patients with penile cancer is limited. One target of this meta-analysis was to investigate the association between PDL-1 with inguinal LNM and prognosis in populations with penile cancer. We revealed that higher PD-L1 expression in tumor cells was related to higher risk of LNM and poorer prognosis. This conclusion, to some extent, provided a theoretical basis for the application of targeted anti-PD-L1 immunotherapy in penile cancer patients.
SCC-Ag, a tumor-associated glycoprotein, is bound up with early recurrence of cervical cancer.70 However, its relationship with early recurrence and risk of LNM in patients with penile cancer has not been summarized. Touloupidis et al revealed elevated SCC-Ag level predicted LNM and distant metastasis.54 Laniado et al demonstrated elevated SCC-Ag level had a high specificity (100%) and an intermediate sensitivity (57%) for prediction of LNM.41 However, Hungerhuber et al found that it was just related to tumor burden rather than LNM.39 Based on an accumulate meta-analysis of these discrepant data, we conclusively demonstrated that elevated SCC-Ag was a predictor of LNM. However, due to the small sample size of prior studies, the clinical application of SCC-Ag to predict LNM should be further validated by more large-scale researches.
For these biochemical predictors, given the limited number of included studies, we are unable to perform subgroup analysis based on different cut-off points, different antibodies, or different measurement methods. This accounted for the cross-study heterogeneity. Meanwhile, there are other immune biomarkers, such as CD8, CD163, and so on. However, we cannot include them in this meta-analysis because of insufficient data. This may be a direction of our future research.
Two TNM stage systems consisting of AJCC and UICC were adapted for penile cancer in this meta-analysis and they are constantly updated. Therefore, we performed a subgroup analysis based on different TNM versions. No matter which version we analyzed, we found that patients with higher stage disease were at higher LNM risk. In addition, we further analyzed all components of these TNM systems as well as other common pathological characters. Conclusively, the LNM risk will increase significantly as long as there is any invasive pathological character, such as LVI, corpora cavernosum invasion, corpus spongiosum invasion, urethra invasion, nerve invasion, larger tumor size, and deeper tumor invasion.
A large-scale study performed by Wang et al showed that LNM rates ranged from 5% to 100% between different risk stratified histological subtypes.5 This huge risk difference highlighted the importance of a conclusive histological risk stratification. Therefore, we divided patients into three risk groups (high-risk, intermediate-risk, and low-risk groups) and compared the LNM risk between each group. Results suggested that this risk stratification was viable. Patients with high-risk histological subtype (basaloid, sarcomatoid, adenosquamous, and poorly differentiated types) were at obviously higher LNM risk.
For patients with cN+, the LNM risk ranged from 54% to 85% (mean 62%) in these included studies. Results of all individual studies were statistically significant. For tumor grade, we performed subgroups analysis based on multivariable and univariable analysis. Both groups demonstrated that higher grade was related to higher LNM risk. Similarly, such a positive correlation was also revealed between tumor vertical growth pattern and LNM risk. However, we did not find any correlation between LNM risk and age, race or HPV infection.
This study has several limitations. First, we failed to analyze some important factors, such as the tumor site, tumor multifocality, tumor cell keratinization, and koilocytosis. Second, some studies only provided original statistical data rather than direct ORs. Third, several methodological drawbacks of the included studies were noted, such as the small series, single-center and retrospective nature of these studies. Fourth, the number of studies focusing on biomarkers was small, and some other important biomarkers were not included due to insufficient data. Given these drawbacks, high-quality studies are warranted to further validate our conclusion. In addition, future studies should explore the intrinsic links between these predictors and then create an accurate and comprehensive mathematical predictive model for LNM by integrating multiple predictors.
Conclusion
We identified valuable predictors of LNM in penile cancer patients, such as tumor-associated biomarkers (NLR, CRP, PD-L1, SCC-Ag, and P53 protein) and invasive clinicopathologic characteristics (higher stage, LVI, cavernosum invasion, urethra invasion, nerve invasion, deeper invasion, cN+, larger tumor size, higher grade, vertical growth pattern, and high- and intermediate-risk histopathological subtype).
Acknowledgments
This work was supported by the National key R&D Program of China (2016YFC0902603), the National Natural Science Foundation of China [81572523, 81700665], and the Hunan Province Funds for Distinguished Young Scientists of China [2016JJ1026].
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Yu C, Hequn C, Longfei L, et al. Sexual function after partial penectomy: a prospectively study from China. Sci Rep. 2016;6:21862. doi: 10.1038/srep21862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Leijte JA, Valdes Olmos RA, Nieweg OE, Horenblas S. Anatomical mapping of lymphatic drainage in penile carcinoma with SPECT-CT: implications for the extent of inguinal lymph node dissection. Eur Urol. 2008;54:885–890. doi: 10.1016/j.eururo.2008.04.094 [DOI] [PubMed] [Google Scholar]
- 3.Wen S, Ren W, Xue B, et al. Prognostic factors in patients with penile cancer after surgical management. World J Urol. 2018;36:435–440. doi: 10.1007/s00345-017-2167-5 [DOI] [PubMed] [Google Scholar]
- 4.Hakenberg OW, Comperat EM, Minhas S, Necchi A, Protzel C, Watkin N. EAU guidelines on penile cancer: 2014 update. Eur Urol. 2015;67:142–150. doi: 10.1016/j.eururo.2014.10.017 [DOI] [PubMed] [Google Scholar]
- 5.Wang JY, Gao MZ, Yu DX, et al. Histological subtype is a significant predictor for inguinal lymph node metastasis in patients with penile squamous cell carcinoma. Asian J Androl. 2018;20:265–269. doi: 10.4103/aja.aja_60_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wespes E. The management of regional lymph nodes in patients with penile carcinoma and reliability of sentinel node biopsy. Eur Urol. 2007;52:15–16. discussion 20-1. doi: 10.1016/j.eururo.2007.02.045 [DOI] [PubMed] [Google Scholar]
- 7.Schlenker B, Scher B, Tiling R, et al. Detection of inguinal lymph node involvement in penile squamous cell carcinoma by 18F-fluorodeoxyglucose PET/CT: a prospective single-center study. Urol Oncol. 2012;30:55–59. doi: 10.1016/j.urolonc.2009.10.012 [DOI] [PubMed] [Google Scholar]
- 8.Lam W, Alnajjar HM, La-Touche S, et al. Dynamic sentinel lymph node biopsy in patients with invasive squamous cell carcinoma of the penis: a prospective study of the long-term outcome of 500 inguinal basins assessed at a single institution. Eur Urol. 2013;63:657–663. doi: 10.1016/j.eururo.2012.10.035 [DOI] [PubMed] [Google Scholar]
- 9.Kirrander P, Andren O, Windahl T. Dynamic sentinel node biopsy in penile cancer: initial experiences at a Swedish referral centre. BJU Int. 2013;111:E48–53. doi: 10.1111/j.1464-410X.2012.11437.x [DOI] [PubMed] [Google Scholar]
- 10.Leijte JA, Kirrander P, Antonini N, Windahl T, Horenblas S. Recurrence patterns of squamous cell carcinoma of the penis: recommendations for follow-up based on a two-centre analysis of 700 patients. Eur Urol. 2008;54:161–168. doi: 10.1016/j.eururo.2008.04.016 [DOI] [PubMed] [Google Scholar]
- 11.Kroon BK, Horenblas S, Lont AP, Tanis PJ, Gallee MP, Nieweg OE. Patients with penile carcinoma benefit from immediate resection of clinically occult lymph node metastases. J Urol. 2005;173:816–819. doi: 10.1097/01.ju.0000154565.37397.4d [DOI] [PubMed] [Google Scholar]
- 12.Kulkarni JN, Kamat MR. Prophylactic bilateral groin node dissection versus prophylactic radiotherapy and surveillance in patients with N0 and N1-2A carcinoma of the penis. Eur Urol. 1994;26:123–128. [DOI] [PubMed] [Google Scholar]
- 13.Cui Y, Chen H, Liu L, et al. Saphenous vein sparing during laparoscopic bilateral inguinal lymphadenectomy for penile carcinoma patients. Int Urol Nephrol. 2016;48:363–366. doi: 10.1007/s11255-015-1182-y [DOI] [PubMed] [Google Scholar]
- 14.Yuan JB, Chen MF, Qi L, et al. Preservation of the saphenous vein during laparoendoscopic single-site inguinal lymphadenectomy: comparison with the conventional laparoscopic technique. BJU Int. 2015;115:613–618. doi: 10.1111/bju.12838 [DOI] [PubMed] [Google Scholar]
- 15.Yuan P, Zhao C, Liu Z, et al. Comparative study of video endoscopic inguinal lymphadenectomy through a hypogastric vs leg subcutaneous approach for penile cancer. J Endourol. 2018;32:66–72. doi: 10.1089/end.2017.0455 [DOI] [PubMed] [Google Scholar]
- 16.Hungerhuber E, Schlenker B, Karl A, et al. Risk stratification in penile carcinoma: 25-year experience with surgical inguinal lymph node staging. Urology. 2006;68:621–625. doi: 10.1016/j.urology.2006.03.052 [DOI] [PubMed] [Google Scholar]
- 17.Solsona E, Iborra I, Rubio J, Casanova JL, Ricos JV, Calabuig C. Prospective validation of the association of local tumor stage and grade as a predictive factor for occult lymph node micrometastasis in patients with penile carcinoma and clinically negative inguinal lymph nodes. J Urol. 2001;165:1506–1509. [PubMed] [Google Scholar]
- 18.Chaux A. Risk group systems for penile cancer management: a study of 203 patients with invasive squamous cell carcinoma. Urology. 2015;86:790–796. doi: 10.1016/j.urology.2015.03.050 [DOI] [PubMed] [Google Scholar]
- 19.Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8:336–341. doi: 10.1016/j.ijsu.2010.02.007 [DOI] [PubMed] [Google Scholar]
- 20.Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in metaanalysis. Available from: www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed December12, 2016.
- 21.Alkatout I, Naumann CM, Hedderich J, et al. Squamous cell carcinoma of the penis: predicting nodal metastases by histologic grade, pattern of invasion and clinical examination. Urol Oncol. 2011;29:774–781. doi: 10.1016/j.urolonc.2009.10.014 [DOI] [PubMed] [Google Scholar]
- 22.Steffens S, Al Ghazal A, Steinestel J, et al. High CRP-values predict poor survival in patients with penile cancer. BMC Cancer. 2013;13:223. doi: 10.1186/1471-2407-13-223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Al Ghazal A, Steffens S, Steinestel J, et al. Elevated C-reactive protein values predict nodal metastasis in patients with penile cancer. BMC Urol. 2013;13:53. doi: 10.1186/1471-2490-13-53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Azizi M, Peyton CC, Boulware DC, et al. Prognostic value of neutrophil-to-lymphocyte ratio in penile squamous cell carcinoma patients undergoing inguinal lymph node dissection. Can Urol Assoc J. 2018. doi: 10.1016/j.euf.2018.06.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bhagat SK, Gopalakrishnan G, Kekre NS, et al. Factors predicting inguinal node metastasis in squamous cell cancer of penis. World J Urol. 2010;28:93–98. doi: 10.1007/s00345-009-0421-1 [DOI] [PubMed] [Google Scholar]
- 26.Chalya PL, Rambau PF, Masalu N, Simbila S. Ten-year surgical experiences with penile cancer at a tertiary care hospital in northwestern Tanzania: a retrospective study of 236 patients. World J Surg Oncol. 2015;13:71. doi: 10.1186/s12957-015-0482-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen X, Li X, Garcia MM, et al. Prognostic factors in Chinese patients with penile invasive squamous cell carcinoma. J Androl. 2012;33:1276–1281. doi: 10.2164/jandrol.112.016378 [DOI] [PubMed] [Google Scholar]
- 28.Cubilla AL, Reuter V, Velazquez E, Piris A, Saito S, Young RH. Histologic classification of penile carcinoma and its relation to outcome in 61 patients with primary resection. Int J Surg Pathol. 2001;9:111–120. doi: 10.1177/106689690100900204 [DOI] [PubMed] [Google Scholar]
- 29.Dai B, Ye DW, Kong YY, Yao XD, Zhang HL, Shen YJ. Predicting regional lymph node metastasis in Chinese patients with penile squamous cell carcinoma: the role of histopathological classification, tumor stage and depth of invasion. J Urol. 2006;176:1431–1435. discussion 5. doi: 10.1016/j.juro.2006.06.024 [DOI] [PubMed] [Google Scholar]
- 30.Emerson RE, Ulbright TM, Eble JN, Geary WA, Eckert GJ, Cheng L. Predicting cancer progression in patients with penile squamous cell carcinoma: the importance of depth of invasion and vascular invasion. Mod Pathol. 2001;14:963–968. doi: 10.1038/modpathol.3880419 [DOI] [PubMed] [Google Scholar]
- 31.Ficarra V, Zattoni F, Cunico SC, et al. Lymphatic and vascular embolizations are independent predictive variables of inguinal lymph node involvement in patients with squamous cell carcinoma of the penis: gruppo Uro-Oncologico del Nord Est (Northeast Uro-Oncological Group) penile cancer data base data. Cancer. 2005;103:2507–2516. doi: 10.1002/cncr.21076 [DOI] [PubMed] [Google Scholar]
- 32.Fonseca AG, Soares FA, Burbano RR, Silvestre RV, Pinto LO. Human papilloma virus: prevalence, distribution and predictive value to lymphatic metastasis in penile carcinoma. International Braz J Urol. 2013;39:542–550. doi: 10.1590/S1677-5538.IBJU.2013.04.12 [DOI] [PubMed] [Google Scholar]
- 33.Graafland NM, Lam W, Leijte JA, et al. Prognostic factors for occult inguinal lymph node involvement in penile carcinoma and assessment of the high-risk EAU subgroup: a two-institution analysis of 342 clinically node-negative patients. Eur Urol. 2010;58:742–747. doi: 10.1016/j.eururo.2010.08.015 [DOI] [PubMed] [Google Scholar]
- 34.Guimaraes GC, Cunha IW, Soares FA, et al. Penile squamous cell carcinoma clinicopathological features, nodal metastasis and outcome in 333 cases. J Urol. discussion 34, 2009;182:528–534. doi: 10.1016/j.juro.2009.04.028 [DOI] [PubMed] [Google Scholar]
- 35.Guimaraes GC, Leal ML, Campos RS, et al. Do proliferating cell nuclear antigen and MIB-1/Ki-67 have prognostic value in penile squamous cell carcinoma? Urology. 2007;70:137–142. doi: 10.1016/j.urology.2007.03.003 [DOI] [PubMed] [Google Scholar]
- 36.Gunia S, Kakies C, Erbersdobler A, Hakenberg OW, Koch S, May M. Expression of p53, p21 and cyclin D1 in penile cancer: p53 predicts poor prognosis. J Clin Pathol. 2012;65:232–236. doi: 10.1136/jclinpath-2011-200429 [DOI] [PubMed] [Google Scholar]
- 37.Hall MC, Sanders JS, Vuitch F, Ramirez E, Pettaway CA. Deoxyribonucleic acid flow cytometry and traditional pathologic variables in invasive penile carcinoma: assessment of prognostic significance. Urology. 1998;52:111–116. [DOI] [PubMed] [Google Scholar]
- 38.Harmaya A, Yudiana I, Oka A, Djatisoesanto W. Predictive factors of inguinal lymph node metastasis in men with penile cancer at sanglah hospital, Denpasar, Bali. Jurnal Bedah Nasional. 2017;1:39–46%V. [Google Scholar]
- 39.Hungerhuber E, Schlenker B, Schneede P, Stief CG, Karl A. Squamous cell carcinoma antigen correlates with tumor burden but lacks prognostic potential for occult lymph node metastases in penile cancer. Urology. 2007;70:975–979. doi: 10.1016/j.urology.2007.07.013 [DOI] [PubMed] [Google Scholar]
- 40.Kroon BK, Leijte JA, van Boven H, et al. Microarray gene-expression profiling to predict lymph node metastasis in penile carcinoma. BJU Int. 2008;102:510–515. doi: 10.1111/j.1464-410X.2008.07697.x [DOI] [PubMed] [Google Scholar]
- 41.Laniado ME, Lowdell C, Mitchell H, Christmas TJ. Squamous cell carcinoma antigen: a role in the early identification of nodal metastases in men with squamous cell carcinoma of the penis. BJU Int. 2003;92:248–250. [DOI] [PubMed] [Google Scholar]
- 42.Li ZS, Yao K, Li YH, et al. Clinical significance of preoperative C-reactive protein and squamous cell carcinoma antigen levels in patients with penile squamous cell carcinoma. BJU Int. 2016;118:272–278. doi: 10.1111/bju.13379 [DOI] [PubMed] [Google Scholar]
- 43.Lopes A, Bezerra AL, Pinto CA, Serrano SV, de Mell OC, Villa LL. p53 as a new prognostic factor for lymph node metastasis in penile carcinoma: analysis of 82 patients treated with amputation and bilateral lymphadenectomy. J Urol. 2002;168:81–86. [PubMed] [Google Scholar]
- 44.Lopes A, Hidalgo GS, Kowalski LP, Torloni H, Rossi BM, Fonseca FP. Prognostic factors in carcinoma of the penis: multivariate analysis of 145 patients treated with amputation and lymphadenectomy. J Urol. 1996;156:1637–1642. [DOI] [PubMed] [Google Scholar]
- 45.Mannweiler S, Sygulla S, Tsybrovskyy O, Razmara Y, Pummer K, Regauer S. Clear-cell differentiation and lymphatic invasion, but not the revised TNM classification, predict lymph node metastases in pT1 penile cancer: a clinicopathologic study of 76 patients from a low incidence area. Urol Oncol. 2013;31:1378–1385. doi: 10.1016/j.urolonc.2012.01.017 [DOI] [PubMed] [Google Scholar]
- 46.Ornellas AA, Nobrega BL, Wei Kin Chin E, Wisnescky A, Da Silva PC, de Santos Schwindt AB. Prognostic factors in invasive squamous cell carcinoma of the penis: analysis of 196 patients treated at the Brazilian National Cancer Institute. J Urol. 2008;180:1354–1359. doi: 10.1016/j.juro.2008.06.028 [DOI] [PubMed] [Google Scholar]
- 47.Protzel C, Knoedel J, Zimmermann U, Woenckhaus C, Poetsch M, Giebel J. Expression of proliferation marker Ki67 correlates to occurrence of metastasis and prognosis, histological subtypes and HPV DNA detection in penile carcinomas. Histol Histopathol. 2007;22:1197–1204. doi: 10.14670/HH-22.1197 [DOI] [PubMed] [Google Scholar]
- 48.Qu XM, Siemens DR, Louie AV, Yip D, Mahmud A. Validation of predictors for lymph node status in penile cancer: results from a population-based cohort. Can Urol Assoc J. 2018;12:119–125. doi: 10.5489/cuaj.4711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ottenhof SR, Djajadiningrat RS, Thygesen HH, et al. The prognostic value of immune factors in the tumor microenvironment of penile squamous cell carcinoma. Front Immunol. 2018;9:1253. doi: 10.3389/fimmu.2018.01253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Slaton JW, Morgenstern N, Levy DA, et al. Tumor stage, vascular invasion and the percentage of poorly differentiated cancer: independent prognosticators for inguinal lymph node metastasis in penile squamous cancer. J Urol. 2001;165:1138–1142. [PubMed] [Google Scholar]
- 51.Kasuga J, Kawahara T, Takamoto D, et al. Increased neutrophil-to-lymphocyte ratio is associated with disease-specific mortality in patients with penile cancer. BMC Cancer. 2016;16:396. doi: 10.1186/s12885-016-2443-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Termini L, Fregnani JH, Boccardo E, et al. SOD2 immunoexpression predicts lymph node metastasis in penile cancer. BMC Clin Pathol. 2015;15:3. doi: 10.1186/s12907-015-0003-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Theodorescu D, Russo P, Zhang ZF, Morash C, Fair WR. Outcomes of initial surveillance of invasive squamous cell carcinoma of the penis and negative nodes. J Urol. 1996;155:1626–1631. [PubMed] [Google Scholar]
- 54.Touloupidis S, Zisimopoulos A, Giannakopoulos S, Papatsoris AG, Kalaitzis C, Thanos A. Clinical usage of the squamous cell carcinoma antigen in patients with penile cancer. Int J Urol. 2007;14:174–176. doi: 10.1111/j.1442-2042.2007.01694.x [DOI] [PubMed] [Google Scholar]
- 55.Udager AM, Liu TY, Skala SL, et al. Frequent PD-L1 expression in primary and metastatic penile squamous cell carcinoma: potential opportunities for immunotherapeutic approaches. Ann Oncol. 2016;27:1706–1712. doi: 10.1093/annonc/mdw216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Velazquez EF, Ayala G, Liu H, et al. Histologic grade and perineural invasion are more important than tumor thickness as predictor of nodal metastasis in penile squamous cell carcinoma invading 5 to 10 mm. Am J Surg Pathol. 2008;32:974–979. doi: 10.1097/PAS.0b013e3181641365 [DOI] [PubMed] [Google Scholar]
- 57.Winters BR, Mossanen M, Holt SK, Lin DW, Wright JL. Predictors of nodal upstaging in clinical node negative patients with penile carcinoma: a national cancer database analysis. Urology. 2016;96:29–34. doi: 10.1016/j.urology.2016.06.033 [DOI] [PubMed] [Google Scholar]
- 58.Deng C, Li Z, Guo S, et al. Tumor PD-L1 expression is correlated with increased TILs and poor prognosis in penile squamous cell carcinoma. Oncoimmunology. 2017;6:e1269047. doi: 10.1080/2162402X.2016.1269047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zargar-Shoshtari K, Spiess PE, Berglund AE, et al. Clinical significance of p53 and p16ink4a status in a contemporary north american penile carcinoma cohort. Clin Genitourin Cancer. 2016;14:346–351. doi: 10.1016/j.clgc.2015.12.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhu Y, Zhang HL, Yao XD, et al. Development and evaluation of a nomogram to predict inguinal lymph node metastasis in patients with penile cancer and clinically negative lymph nodes. J Urol. 2010;184:539–545. doi: 10.1016/j.juro.2010.03.145 [DOI] [PubMed] [Google Scholar]
- 61.Zhu Y, Zhou XY, Yao XD, Dai B, Ye DW. The prognostic significance of p53, Ki-67, epithelial cadherin and matrix metalloproteinase-9 in penile squamous cell carcinoma treated with surgery. BJU Int. 2007;100:204–208. doi: 10.1111/j.1464-410X.2007.06908.x [DOI] [PubMed] [Google Scholar]
- 62.Hegarty PK, Kayes O, Freeman A, Christopher N, Ralph DJ, Minhas S. A prospective study of 100 cases of penile cancer managed according to European Association of Urology guidelines. BJU Int. 2006;98:526–531. doi: 10.1111/j.1464-410X.2006.06296.x [DOI] [PubMed] [Google Scholar]
- 63.Ohaegbulam KC, Assal A, Lazar-Molnar E, Yao Y, Zang X. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol Med. 2015;21(1):24–33. doi: 10.1016/j.molmed.2014.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang Z, Peng S, Xie H, et al. Neutrophil-lymphocyte ratio is a predictor of prognosis in patients with castration-resistant prostate cancer: a meta-analysis. Cancer Manag Res. 2018;10:3599–3610. doi: 10.2147/CMAR.S159105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jonska-Gmyrek J, Gmyrek L, Zolciak-Siwinska A, Kowalska M, Fuksiewicz M, Kotowicz B. Pretreatment neutrophil to lymphocyte and platelet to lymphocyte ratios as predictive factors for the survival of cervical adenocarcinoma patients. Cancer Manag Res. 2018;10:6029–6038. doi: 10.2147/CMAR.S178745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Song X, Chen D, Yuan M, Wang H, Wang Z. Total lymphocyte count, neutrophil-lymphocyte ratio, and platelet-lymphocyte ratio as prognostic factors in advanced non-small cell lung cancer with chemoradiotherapy. Cancer Manag Res. 2018;10:6677–6683. doi: 10.2147/CMAR.S188578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sun Y, Zhang L. The clinical use of pretreatment NLR, PLR, and LMR in patients with esophageal squamous cell carcinoma: evidence from a meta-analysis. Cancer Manag Res. 2018;10:6167–6179. doi: 10.2147/CMAR.S171035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity. 2007;27:111–122. doi: 10.1016/j.immuni.2007.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu B, Song Y, Liu D. Recent development in clinical applications of PD-1 and PD-L1 antibodies for cancer immunotherapy. J Hematol Oncol. 2017;10:174. doi: 10.1186/s13045-017-0541-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Salvatici M, Achilarre MT, Sandri MT, Boveri S, Vanna Z, Landoni F. Squamous cell carcinoma antigen (SCC-Ag) during follow-up of cervical cancer patients: role in the early diagnosis of recurrence. Gynecol Oncol. 2016;142:115–119. doi: 10.1016/j.ygyno.2016.04.029 [DOI] [PubMed] [Google Scholar]