Abstract
Background:
The last few years have detailed a number of surgical materials and techniques to stimulate guided bone regeneration (GBR). Polypropylene has been used as a mechanical barrier, intentionally designed to be exposed to the oral environment, isolating the regeneration area, and allowing the blood clot to remain protected in a confined space while pluripotent mesenchymal cells regenerate the alveolar bone tissue.
Aim:
Due to the lack of studies on polypropylene barriers (PB) (Bone Heal– Bone heal ind. e Com. LTDA – São Paulo, Brazil), this study aimed to evaluate the histological repair process of critical defects (7 mm) made in the rodent cranial vaults comparing its efficacy in GBR and modified GBR.
Materials and Methods:
A total of 30 rats were divided into three groups. The control group consisted of 10 rats, wounds covered with just a blood clot. The second group consisted of an exposed/uncoated PB at the edges of the wound, and it was removed after 3 days. The third group used submerged PB with coaptation of the wound edges, and it was not removed. Five animals of each group were euthanized at 30 and 90 days postoperative and submitted to microscopic analysis and histomorphometric evaluation.
Results:
Modified GBR (membrane exposed to the oral medium) provided earlier tissue organization at 30 days; however, the third group presented better bone neoformation at 90 days.
Conclusion:
Modified GBR provided earlier tissue organization compared with the control group, as well as promoting improved bone neoformation, while regeneration with the submerged membrane presented better bone neoformation in the long term.
Key words: Bone regeneration, guided bone regeneration, polypropylene
INTRODUCTION
Significant physiological dimensional changes are to be expected following tooth extraction. Several studies have sought to understand and to quantify this remodeling process. Approximately 3.87 mm of loss in thickness and 1.67–2.03 mm in height occur within the first 3 months.[1] This loss may be related to reabsorption of the alveolar bone itself [2] or trauma resulting in vascular damage and an acute inflammatory reaction, which are considered mediators of bone resorption.[3] Factors, such as alveolus depth, mucosal thickness, systemic-metabolic conditions, and functional load, may affect this process.[4,5]
Alveolar preservation procedures have the potential to reduce such vertical and horizontal bone alterations, without completely interrupting them.[6] In a recent meta-analysis, compared to untreated alveoli submitted to preservation, treated areas are on average 1.31–1.54 mm thicker and 0.91–1.12 mm in higher, with better results obtained through guided bone regeneration (GBR), using nonabsorbable barriers without grafts, or through bioactive materials.[7]
GBR techniques are based on the concept of osteopromotion, in which, a barrier prevents the migration of epithelial cells into the alveolus, promoting cell exclusion, and preventing them from interfering with the repair process.[8,9]
Nonabsorbable barriers, such as expanded polytetrafluoroethylene, have a long history of success in GBR, particularly in periodontics. Its porosity allows bacterial colonization of the surface when exposed to the oral environment, therefore, one should consider its indication when a submerged membrane is required, while accepting the risk of failure should there be early exposure.[10] This represents a limitation for alveoli since the coaptation of borders at this site requires manipulation and advance of the flap without tension, a technique that is not always accessible to less-skilled professionals.
The use of a membrane exposed to the oral medium is defined as a modified GBR technique since conceptually when using this treatment modality; the membrane must remain submerged for at least 6 weeks.[11]
The fundamental characteristics of regenerative barriers are related to properties such as biocompatibility, cell exclusion, integration into host tissues, clinical handling, and the ability to occupy the space.[9,12] The polypropylene barrier (PB) meets all of these requirements and has recently emerged as a nonabsorbable option for GBR in alveoli. PB allows exposure to the oral environment through its smooth and impermeable surface while preventing bacterial colonization of the barrier.[13,14,15,16,17,18]
Thus, the objective of this study was to evaluate the efficacy of a polypropylene membrane in GBR and modified GBR in the treatment of critical defects of rodent cranial vaults, using histomorphometry.
MATERIALS AND METHODS
This study was approved by the Research Ethics Committee (protocol no. 2015-009). All procedures were carried out in accordance with the Ethical Principles of the National Animal Experimentation Council.
Thirty Wistar line, male, adult rats (Rattus norvegicus albinus), with a mean weight of 300 g, were obtained. The animals were housed in individual cages at a controlled temperature of 22°C, with a 12-h light cycle, and free access to water and industrialized feed.
A critical bone defect of approximately 7 mm was performed at the center of each skull under general anesthesia using a 0.2 ml/100 mg anesthetic solution with 50 mg/ml Ketamine (DOPALEN®) and 20 mg/ml Xylazine (ANASEDAN®) at a ratio of 2:1, injected intraperitoneal. After epilation and antisepsis with iodopovidone, a 2.5-cm incision was made in the skin, extending from the nasofrontal region to the occipital protuberance. The tissues were dissected laterally and osteotomy performed with a 7-mm diameter trephine drill, rotating at approximately 900 RPM under constant irrigation until reaching the internal cranial surfaces.
The animals were then divided into three experimental groups with 15 rats in each group [Figure 1] as follows:
Figure 1.
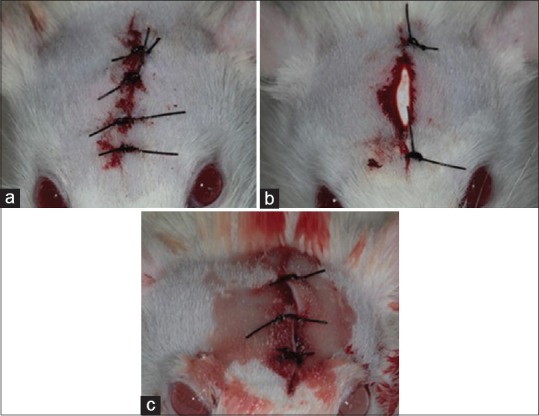
Postoperative aspect of the different experimental groups. (a) Group 1 - (Control) filling of bone defect with clot and suture; (b) Group 2 - polypropylene barrier with the defect partially exposed, removed after 3 days; (c) Group 3 - polypropylene barrier with coaptation of the edges of the wound, leaving the barrier submerged
Group 1: Confection of the bone defect and suturing
Group 2: Preparation of the bone defect, positioning of the PB (bone heal) with skin suturing using Nylon 4-0 (Ethicon), and leaving the barrier exposed. It was removed after 3 days
Group 3: Confection of the bone defect, positioning of the polypropylene membrane, and skin suture using Nylon 4-0, with wound closure by the first intention. It was not removed.
Twenty-five drops of 500 mg/ml sodium dipyrone were prescribed for analgesia. The barriers used in Group 2 were removed after 3 days, while those of Group 3 were kept submerged until euthanasia.
The animals were euthanized at 30 and 90 days postoperative by anesthetic overdose using intraperitoneal 90–150 mg/kg of sodium thiopental associated with 10 mg/ml Lidocaine. After animal sacrifice, the samples were fixed in 10% formaldehyde and processed by decalcification followed by dehydration and diaphanization in paraffin sections at a thickness of 5 μm. Each section was stained with H and E, placed on a glass slide, and submitted to microscopic analysis.
Two-way analysis of variance and Tukey's test were performed to compare the effect of GBR with time, as well as the interaction between these two factors. Statistics were performed using the SPSS 23 program (SPSS Inc., Chicago, IL, USA), adopting a 5% of significance level.
RESULTS
Postoperative repair progressed well, complication-free, in all animals. There were no clinical signs of postoperative infection observed in any of the surgical sites.
Regarding histological analysis, at 30 days, the bone defects produced in the cranial vaults were observed to be filled with connective tissue components at different phases of the organization. Overall, the control group presented a lower connective tissue thickness when compared to the groups with and without coaptation [Figure 2].
Figure 2.
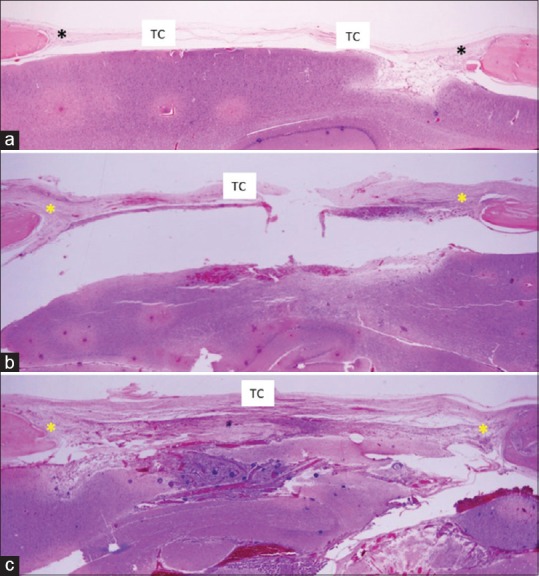
Histological section of 30-day experimental groups: panoramic image (H and E, ×2.5). (a) Control group showing presence of bony defect filled by a thin layer of connective tissue, mainly in the central region; (b) Group without coaptation: bony defect filled by a thicker layer of connective tissue, mainly within the central region; (c) Coaptation group: bony defect filled by connective tissue with greater organization in the central region. *Bony defect; TC – Connective tissue
Discrete bone neoformation, at the marginal region of the bony defect, was observed in all groups, with a slightly larger increase in the coaptation group. However, new bone formation toward the center of each defect was minimal in all samples. Within this region, the greatest difference was related to the organization and thickness of the connective tissue, with better organization and depth observed in the groups with and without coaptation. With the exception of the control group, the presence of a large number of fibroblasts and capillary neoformation was noted.
At 90 days, in the panoramic view of the three groups, the bony defect maintained different degrees of bone neoformation, with the coaptation group showing greater bone neoformation. In the images at a higher magnification, one may observe discrete bone neoformation at the margins of the bony defect in the control group, with the presence of organized and developed connective tissue filling this defect. In the noncoaptation group, neoformation was more evident particularly when considering bone development toward the center of the defect. The connective tissue presented a fibrous characteristic with areas of greater vascularization. In the coaptation group, the new bone formation was more intense, with an organized and well-vascularized connective tissue [Figure 3].
Figure 3.
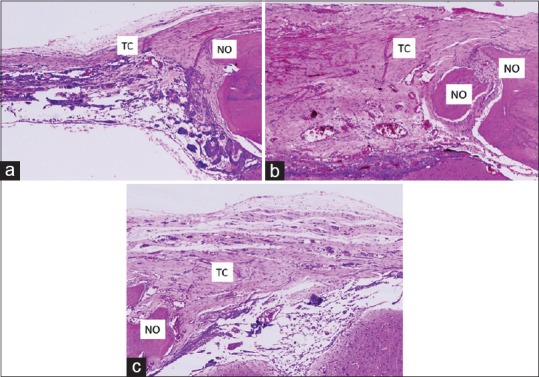
Histological section of experimental groups at 90 days: enlarged image (H and E, ×40). (a) Control group: discrete bone neoformation from the margins of bony defect, with the presence of connective tissue; (b) Noncoaptation group: bone neoformation toward the center of the bone defect, with the presence of fibrous connective tissue; (c) Coaptation group: bone neoformation toward the center of the bony defect, with the presence of organized and vascularized connective tissue. NO – Neoform bone; TC – Connective tissue
For the histometric evaluation, individual comparison of the slides was performed considering the margins of the osteotomy and the projection of bone neoformation toward the center of the cavity.
Two-way analysis of variance revealed no significant interaction between GBR and time (P = 0.118), suggesting that should there be a difference between GBR techniques, it could occur at any time interval, while differences between time intervals would occur regardless of the technique used.
Comparing the presence of new bone formation with the GBR technique used two-way analysis of variance (P < 0.001), and Tukey's test revealed that when wound coaptation was performed following PB positioning, the percentage of newly formed bone was statistically superior to that obtained without coaptation. This group did not show a significant difference compared to the control group, in which, no PB was used [Table 1]. After 30 days, the coaptation group had 94.0% and 125.6% more newly formed bone in relation to the control and noncoaptation groups, respectively. After 90 days, the new bone formation in the coaptation group was 89.9% and 67.5% higher in the control and noncoaptation groups, respectively.
Table 1.
Mean and standard deviations, in percentage, of newly formed bone in cranial vault defects, in rats, following polypropylene barrier positioning, according to the guided regeneration technique used and time
| Technique | Time (days) |
Mean | |
|---|---|---|---|
| 30 | 90 | ||
| Control | 5.0 (1.2) | 17.9 (1.1) | 11.5 (6.9) A |
| Without coaptation | 4.3 (1.3) | 20.3 (1.7) | 12.3 (8.6) A |
| With coaptation | 9.7 (3.8) | 34.0 (6.0) | 21.9 (13.7) B |
| Mean | 6.3 (3.3) a | 24.1 (8.1) b | – |
SD in brackets. Means followed by different upper case letters indicate a significant difference between techniques, regardless of time. Mean followed by different lowercase letters indicate a significant difference between times, regardless of technique. SD – Standard deviation
Two-way analysis of variance also suggested that new bone formation was significantly affected by time since the surgical procedure (P > 0.001), regardless of the technique used (with coaptation, without coaptation, or control), with significantly higher values observed after 90 days [Table 1]. After 90 days, the quantity of new bone formation was, on average, 3.8 times higher than at 30 days.
DISCUSSION
Cranial bone defects in rats represent the standard experimental model for studies evaluating bone repair processes.[19] By classical definition, a critical size defect is the smallest size tissue defect that will not completely heal over the natural lifetime of an animal. Parietal bone defects greater than 5 mm are usually incapable of spontaneous regeneration. Therefore, regeneration approaches allow for assessment of the repair potential of different materials and techniques.[20] For this, 7-mm bone defects were standardized for this study.
A series of studies [21,22,23] has evaluated several biomaterials to fill these defects, with no single biomaterial able to stimulate complete repair. In an experimental study evaluating the effect of GBR, on this type of defect, observed that the presence of a membrane was paramount for complete regeneration of the defect. The groups containing resorbable collagen membranes, independent of the biomaterial used, presented significantly improved results than those containing only the biomaterial.[8]
GBR techniques are based on the presence of a physical barrier, which prevents the migration of epithelial cells into the alveolus; hence, promoting cell deletion.[9] The PB meets all of these requirements and has emerged as a nonabsorbable GBR choice in alveoli since it intentionally allows exposure to the oral environment with a smooth, impermeable surface that prevents bacterial colonization of the barrier.[14] Its use as a mesh in the treatment of hernias, as well as for other defects of the fascia, has already proved its biocompatibility, as well as its capacity to provide mechanical support for the soft tissues. The PB also allows the stabilization of a protected clot within the alveolus using a physical support framework for the soft tissues.
The PB has demonstrated good results in alveolar preservation procedures, as described in a series of case reports [13,17] and experimental studies.[16,18] Its true role in GBR and modified GBR, mainly in alveoli, depends on experimental trials and controlled clinical studies aiming to understand its effectiveness in the process of bony defect repair.
Regarding histology, the presence of a larger quantity of connective tissue in the group in which the PB was applied, regardless of period evaluated, seems to reflect a condition described clinically when using PB, which is the thickening of the tissue biotype around the alveolar ridge. The connective tissue that filled the bony defect was thick, organized, and with intense fibroblastic and cellular neoformation. This degree of organization occurs due to the maintenance of the biological space caused by the use of a physical membrane.
The initial areas of bone neoformation observed and demonstrated an accelerated differentiation and maturation of the osteoid tissue. The maintenance of the submerged barrier resulted in improved bone neoformation, both quantitatively and qualitatively.
For cases involving nonresorbable barriers, a period of prolonged application of membranes in GBR is related to maturation of new bone formation, with the exception of the presence of infection. In a study based on histochemical techniques, Ohnishi et al. evaluated the effect of removal of nonresorbable barriers at different time periods and their influence on bone neoformation in GBR in rat maxilla. Active bone neoformation and corticalization were observed after 1–2 weeks and after 4 weeks, respectively. The authors also confirmed that after the 4th week, the continued presence of the membrane did not cause greater maturation or corticalization of the new-formed bone, however, increased atrophy was shown. Therefore, one should consider this moment as ideal for removal of the barrier. In this study, the association between neoformation and atrophy was not observed after 30 days.
Regarding histometric analysis, the percentage of newly formed bone in the group with wound coaptation after 90 days was higher when compared to the control group. However, the difference between the control group and the group with membrane exposure did not show any significant differences, demonstrating that exposure interferes with the repair pattern.
In this study, as well as that Machtei and Souza et al. showed that sites with submerged membranes presented more bone formation than sites in which membranes became exposed, and this finding was not only statistically significant but had a clinical significance.
CONCLUSION
Modified bone regeneration provided earlier tissue organization compared to the control group and promoted increased bone neoformation, while regeneration with submerged membranes presented greater bone neoformation in the long term.
Financial support and sponsorship
This was a self-funded study.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Van der Weijden F, Dell'Acqua F, Slot DE. Alveolar bone dimensional changes of post-extraction sockets in humans: A systematic review. J Clin Periodontol. 2009;36:1048–58. doi: 10.1111/j.1600-051X.2009.01482.x. [DOI] [PubMed] [Google Scholar]
- 2.Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32:212–8. doi: 10.1111/j.1600-051X.2005.00642.x. [DOI] [PubMed] [Google Scholar]
- 3.Jamjoom A, Cohen RE. Grafts for ridge preservation. J Funct Biomater. 2015;6:833–48. doi: 10.3390/jfb6030833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Atwood DA. Some clinical factors related to rate of resorption of residual ridges 1962. J Prosthet Dent. 2001;86:119–25. doi: 10.1067/mpr.2001.117609. [DOI] [PubMed] [Google Scholar]
- 5.Darby I, Chen ST, Buser D. Ridge preservation techniques for implant therapy. Int J Oral Maxillofac Implants. 2009;24(Suppl):260–71. [PubMed] [Google Scholar]
- 6.Horváth A, Mardas N, Mezzomo LA, Needleman IG, Donos N. Alveolar ridge preservation. A systematic review. Clin Oral Investig. 2013;17:341–63. doi: 10.1007/s00784-012-0758-5. [DOI] [PubMed] [Google Scholar]
- 7.Willenbacher M, Al-Nawas B, Berres M, Kämmerer PW, Schiegnitz E. The effects of alveolar ridge preservation: A meta-analysis. Clin Implant Dent Relat Res. 2016;18:1248–68. doi: 10.1111/cid.12364. [DOI] [PubMed] [Google Scholar]
- 8.Donos N, Lang NP, Karoussis IK, Bosshardt D, Tonetti M, Kostopoulos L, et al. Effect of GBR in combination with deproteinized bovine bone mineral and/or enamel matrix proteins on the healing of critical-size defects. Clin Oral Implants Res. 2004;15:101–11. doi: 10.1111/j.1600-0501.2004.00986.x. [DOI] [PubMed] [Google Scholar]
- 9.Retzepi M, Donos N. Guided bone regeneration: Biological principle and therapeutic applications. Clin Oral Implants Res. 2010;21:567–76. doi: 10.1111/j.1600-0501.2010.01922.x. [DOI] [PubMed] [Google Scholar]
- 10.Ronda M, Rebaudi A, Torelli L, Stacchi C. Expanded vs. dense polytetrafluoroethylene membranes in vertical ridge augmentation around dental implants: A prospective randomized controlled clinical trial. Clin Oral Implants Res. 2014;25:859–66. doi: 10.1111/clr.12157. [DOI] [PubMed] [Google Scholar]
- 11.Dahlin C, Linde A, Gottlow J, Nyman S. Healing of bone defects by guided tissue regeneration. Plast Reconstr Surg. 1988;81:672–6. doi: 10.1097/00006534-198805000-00004. [DOI] [PubMed] [Google Scholar]
- 12.Karring T, Nyman S, Gottlow J, Laurell L. Development of the biological concept of guided tissue regeneration – Animal and human studies. Periodontol 2000. 1993;1:26–35. [PubMed] [Google Scholar]
- 13.Salomão M, Alvarez FK, Siqueira J. Guided bone regeneration after tooth extraction using membrane exposed to the oral environment. ImplantNews. 2010;7:753–9. [Google Scholar]
- 14.Salomão M, Siqueira JT. Guiaded bone regeneration with a barrier exposed to the oral environment after exodontia. Case report. Rev Bras Implant. 2010;1:5–7. [Google Scholar]
- 15.Salomão S, Siqueira JT. Use of polypropylene barrier in post-extraction. Report of three clinical cases. Rev Bras Implant. 2009;15:12–6. [Google Scholar]
- 16.De Lucca L. Histological study of guided bone regeneration with polypropylene membrane in calvaria of rabbits. 74f. (Dissertation) - São Paulo - University of Santo Amaro. 2010 [Google Scholar]
- 17.Renda MO, Amaral AP, Soares AS, Castro CF, Tuji FM. Guided bone regeneration with polypropylene barrier intentionally exposed to the oral environment – Clinical case report. Int J Clin Dent. 2016;8:7–14. [Google Scholar]
- 18.Nesi H, Oliveira MT, Molina GO. Comparison of the use of membranes in conjunctive infiltration in alveoli freshly extracted teeth. Revista Brasileira de Odontologia. 2013;70:136–41. [Google Scholar]
- 19.Maciel J, Momesso GA, Ramalho-Ferreira G, Consolaro RB, Perri de Carvalho PS, Faverani LP, et al. Bone healing evaluation in critical-size defects treated with xenogenous bone plus porcine collagen. Implant Dent. 2017;26:296–302. doi: 10.1097/ID.0000000000000572. [DOI] [PubMed] [Google Scholar]
- 20.Gómez E, Martín M, Arias J, Carceller F. Clinical applications of Norian SRS (calcium phosphate cement) in craniofacial reconstruction in children: Our experience at hospital La Paz since 2001. J Oral Maxillofac Surg. 2005;63:8–14. doi: 10.1016/j.joms.2004.09.008. [DOI] [PubMed] [Google Scholar]
- 21.Park JW, Jang JH, Bae SR, An CH, Suh JY. Bone formation with various bone graft substitutes in critical-sized rat calvarial defect. Clin Oral Implants Res. 2009;20:372–8. doi: 10.1111/j.1600-0501.2008.01602.x. [DOI] [PubMed] [Google Scholar]
- 22.Al-Askar M, Javed F, Al-Hezaimi K, Al-Hamdan KS, Ramalingam S, Aldahmash A, et al. Guided bone regeneration in standardized calvarial defects in rats using bio-oss and β-tricalcium phosphate with adjunct platelet-derived growth factor therapy: A Real-time in vivo microcomputed tomographic, biomechanical, and histologic analysis. Int J Periodontics Restorative Dent. 2016;36(Suppl):s61–73. doi: 10.11607/prd.2265. [DOI] [PubMed] [Google Scholar]
- 23.Ohnishi H, Fujii N, Futami T, Taguchi N, Kusakari H, Maeda T, et al. A histochemical investigation of the bone formation process by guided bone regeneration in rat jaws. Effect of PTFE membrane application periods on newly formed bone. J Periodontol. 2000;71:341–52. doi: 10.1902/jop.2000.71.3.341. [DOI] [PubMed] [Google Scholar]


