Abstract
Translocation of 78-kDa glucose-regulated protein (GRP78) from endoplasmic reticulum (ER) to plasma membrane represents a paradigm shift beyond its traditional function as an ER chaperone protein. Cell surface GRP78 (csGRP78) exerts novel signaling functions, and mechanisms underlying its cell surface expression are just emerging. Acquired tamoxifen resistance of breast cancer cells is accompanied with elevated level of csGRP78. Therefore, the tamoxifen-resistant MCF7 breast cancer cells (MCF7-LR) represents a clinically relevant model to study mechanisms of csGRP78 expression. We discovered that a proline-rich region (PRR) containing three consecutive prolines close to the COOH-terminus of GRP78 is important for its ability to form a complex with the partner protein, CD44v, as demonstrated by in vitro glutathione S-transferase pull-down assay. Proline to alanine mutations at the PRR compromised GRP78 expression level on the cell surface as evidenced by purification of biotinylated cell surface proteins. Reconstitution of MCF7-LR cells with the PRR mutant after knockdown of endogenous GRP78 diminished the capacity of GRP78 to stimulate STAT3 activation. The enforced expression of a short peptide bearing the PRR region of GRP78 led to reduction of CD44v and Cyclin D1 protein levels as well as cell viability, accompanied with increase in apoptotic signaling including cleaved Caspase-3 and PARP. These findings suggest that the COOH-terminal PRR of GRP78 is critical for its interaction with CD44v as well as its cell surface expression, and enforced expression of the short peptide bearing the PRR region may provide a new approach to lower the viability of tamoxifen-resistant breast cancer cells.
Abbreviations: GRP78, 78-kDa glucose-regulated protein; ER, endoplasmic reticulum; csGRP78, cell surface GRP78; MCF7-LR, tamoxifen-resistant MCF7; PRR, proline-rich region; CD44v, CD44 containing variant exon 3 to 10; vHA, HA-tagged CD44 containing variant exon 3 to 10
Introduction
Breast cancer is among the leading causes of cancer deaths in women [1]. Estrogen receptor–positive breast cancer accounts for about 70% of breast cancer patients [2]; thus, the estrogen receptor antagonist tamoxifen is the most widely used adjuvant hormonal therapy. However, tamoxifen resistance is responsible for the relapse of about one-third of these patients [3]. MCF7 is one of the best studied estrogen receptor–positive breast cancer cell lines. We recently discovered that 78-kDa glucose-regulated protein (GRP78, also referred to as BiP or HSPA5) was upregulated intracellularly and on the cell surface of tamoxifen-resistant MCF7 breast cancer cells (MCF7-LR) compared to the parental nonresistant MCF7-L cells [4]. Thus, these cells provide a clinically relevant model for the investigation of how GRP78 relocalizes from the endoplasmic reticulum (ER) to the cell surface.
GRP78 belongs to heat-shock protein-70 (HSP70) family, and it is a major chaperone protein that facilitates protein folding and quality control in the ER [5]. It also regulates ER stress by its interaction with the unfolded protein response (UPR) stress sensors in the ER [5], [6], [7]. GRP78 consists of a NH2-terminal ER signal sequence, an ATPase domain, a substrate binding domain (SBD), and a COOH-terminal KDEL motif, where it facilitates the retrieval of GRP78 by the KDEL receptor from Golgi back to ER [8]. GRP78 is also observed on the cell surface of various cancer cells, and its cell surface expression could be further enhanced by stress conditions [4], [9], [10]. Cell surface GRP78 (csGRP78) regulates novel cell signaling pathways beyond its traditional protein foldase activity in the ER [4], [9], [11]. Importantly, csGRP78 is preferentially expressed in cancer cells and minimally in normal cells, making it an attractive target for cancer therapeutics, including breast cancer [7], [12], [13], [14], [15], [16], [17]. Evidence is emerging on how GRP78 relocalizes from the ER to the cell surface. First, recent studies revealed that csGRP78 largely exists as a peripheral protein at the plasma membrane, where it associates with transmembrane or GPI-anchored proteins for its anchor on the cell surface [10]. Second, GRP78 harboring a mutation in the T453D substrate binding domain exhibited about 80% reduction of its cell surface expression in HeLa cervical cancer cells, suggesting substrate binding function of GRP78 was required for its cell surface expression [10]. Third, it has been reported that the GRP78 co-chaperone MTJ-1, a transmembrane protein and also referred to as DnaJC1 (DnaJ homolog subfamily C member 1), was required for cell surface expression of GRP78 in mouse macrophages [18]. However, GRP78 harboring R197H DnaJ binding mutation [19] showed similar level of cell surface expression as wild-type GRP78 in HeLa cells [10]. Additionally, MTJ-1 was not detected on the cell surface of hepatoma and other cell lines which express csGRP78 [20]. Collectively, these results suggest that the requirement of MTJ-1 or the human homolog, HTJ-1, for csGRP78 expression is likely to be cell context-dependent, and this issue remains an open question.
Since current studies support the notion that cell surface expression of GRP78 is dependent on its capacity to form complex with transmembrane proteins, GPI-anchored proteins, co-chaperones, and likely folding substrates, we speculate that CD44, a transmembrane cell surface protein abundantly expressed in tamoxifen-resistant MCF7 cells [21], [22], may be a folding substrate of GRP78 and, in such capacity, contribute to the cell surface expression of GRP78 in the context of these resistant cells. CD44 is a highly heterogeneous single-pass type I transmembrane glycoprotein, and it is regulated by posttranslational modifications and alternative splicing [23], [24]. It is a major cell surface receptor of hyaluronan [25] and serves as a co-receptor of FGF2, HGF, VEGF, and osteopontin [23]. The expression of CD44 variant isoforms enhanced the lung colonization of breast cancer stem-like cells [24], and CD44v3-10 but not CD44v8-10 or CD44s was correlated with poor prognosis of breast cancer patients [26]. In this study, we determined that GRP78 forms complex with CD44v3-10 through the region containing PPP polyproline sequence localized close to the COOH-terminus of GRP78 and further discovered that mutation of this PPP polyproline sequence impedes GRP78 expression on the cell surface in the MCF7-LR cells. Enforced expression of an expression plasmid containing secretory signal peptide and 20 amino acids spanning the PRR reduced CD44v protein level and led to increase in apoptotic markers. Our studies uncovered critical functions of the COOH-terminal proline-rich region of GRP78 in its cell surface expression and may have clinical implications in lowering the viability of tamoxifen-resistant breast cancer.
Materials and Methods
Cell Culture
Tamoxifen-resistant MCF7 breast cancer cells were a kind gift from Dr. Rachel Schiff (Baylor College of Medicine, TX) and cultured as previously described [21] in phenol-red free RPMI 1640 medium containing 5% charcoal-stripped fetal bovine serum, 2.5 μg/ml fungizone, 200 mM glutamine, 10 IU/ml penicillin, and 10 μg/ml streptomycin supplemented with 100 nM 4-hydroxy tamoxifen. The cells were authenticated by STR DNA profiling analysis at the reagent core facility in the USC Norris Comprehensive Cancer Center. Only mycoplasma-negative cells were used.
Antibodies
We used the following primary antibodies for the proteins listed: GRP78 from Dr. Parkash S. Gill at USC (MAb159), CD44v3 from Thermo Fisher Scientific (BMS144, Waltham, MA), glutathione S-transferase (GST) tag from Santa Cruz Biotechnology, Inc. (sc-138, Dallas, TX), HA tag from Santa Cruz Biotechnology, Inc. (sc-805, Dallas, TX), FLAG M2 from Sigma-Aldrich (F1804, St. Louis, MO), Annexin II from BD Biosciences (610068, San Jose, CA), β-actin from Sigma-Aldrich (A5316, St. Louis, MO), Phospho-STAT3(Y705) from Cell Signaling Technology (9145, Danvers, MA), STAT3 from BD Biosciences (610190, San Jose, CA), Cyclin D1 from Santa Cruz Biotechnology, Inc. (sc-753, Dallas, TX), cleaved Caspase-3 from Cell Signaling Technology (9661, Danvers, MA), and cleaved PARP from Cell Signaling Technology (5625, Danvers, MA). Secondary antibodies were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX), LI-COR Biosciences (Lincoln, NE), and Enzo Life Sciences (Farmingdale, NY).
Plasmids and Cloning
The GST-tagged full-length human GRP78 (FL, a.a. 19-654) and truncated mutants including NH2-terminal half (N, a.a. 19-407), COOH-terminal half (C, a.a. 413-654), ΔKDEL (a.a. 19-650), ΔC11 (a.a. 19-643), ΔC17 (a.a. 19-637), ΔC73 (a.a. 19-581), and C73 (a.a. 582-654) were inserted in-frame into pGEX-4T-1 expression plasmid (GE Healthcare, Chicago, IL) at BamHI and XhoI sites. The FLAG-tagged full-length human GRP78 bearing a COOH-terminal PPP to AAA (a.a. 640-642) mutation was generated by PCR amplification of the GRP78 coding sequencing from FLAG-tagged human GRP78 (wild-type) expression plasmid using the reverse primer containing modified sequence. The PCR product was inserted in-frame into pcDNA3 expression vector (Thermo Scientific, Waltham, MA) at BamHI and XhoI sites. The hemagglutinin (HA)-tagged CD44v3-10 (CD44v-HA) expression plasmid was generated by PCR amplification from cDNA library of MCF7-LR cells using the reverse primer containing HA tag sequence, and the PCR product was inserted in-frame into pcDNA3 expression vector (Thermo Scientific, Waltham, MA) at KpnI and ECoRI sites. All constructs were verified by sequencing. We used the following primers: CD44v3-10-HA 5′-CGGGGTACCATGGACAAGTTTTGGTGGCACGCAGCCT-3′ and 5′-CCGGAATTCTTAAGCGTAATCTGGAACATCGTATGGGTACACCCCAATCTTCATGTCCACATTCT-3′; GST-GRP78 (FL) 5′-CGCGGATCCATGGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTACAACTCATCTTTTTCTGCT-3′; GST-GRP78 (N) 5′-CGCGGATCCATGGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTAACCAGAGAGCACACCAGC-3′; GST-GRP78 (C) 5′-CGCGGATCCATGGACCTGGTACTGCTTGATGTA-3′ and 5′-CCGCTCGAGCTACAACTCATCTTTTTCTGCT-3′; GST-GRP78 (ΔKDEL) 5′-CGCGGATCCGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTATTCTGCTGTATCCTCTTCACCAG-3′; GST-GRP78 (ΔC11) 5′-CGCGGATCCGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTAAGTTGGGGGAGGGCCTGCA-3′; GST-GRP78 (ΔC17) 5′-CGCGGATCCGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTAACTTCCATAGAGTTTGCTGATAATTGG-3′; GST-GRP78 (ΔC73) 5′-CGCGGATCCGAGGAGGAGGACAAGAAGGAGGA-3′ and 5′-CCGCTCGAGCTACTTTTCTTTATCTCCAATCTGATTC-3′; GST-GRP78 (C73) 5′-CGCGGATCCCTGGGAGGTAAACTTTCCTCTG-3′ and 5′-CCGCTCGAGCTACAACTCATCTTTTTCTGCTG-3′; and FLAG-GRP78 (AAA mutant) 5′-CGCGGATCCATGAAGCTCTCCCTGGTGGC-3′ and 5′-CCGCTCGAGCTACAACTCATCTTTTTCTGCTGTATCCTCTTCACCAGTTGCGGCAGCGCCTGCACTTCCATAGAGTTTGCTGA-3′.
The expression plasmids for small peptides containing a previously described [27] secretory and sorting sequence of α-melanocyte-stimulating hormone at NH2-terminus were generated by insertion of the annealed synthetic DNA oligonucleotides into pcDNA3 expression plasmid at BamHI and ECoRI sites. All constructs were verified by sequencing. For the plasmid containing wild-type sequence (P), we used the following oligonucleotides: 5′-GATCCATGCCGAGATCGTGCTGCAGCCGCTCGGGGGCCCTGTTGCTGGCCTTGCTGCTTCAGGCCTCCATGGAAGTGCGTGGCTGGTGCCTGGAGAGCAGCCAGTGTCAGGACCTCACCACGGAAAGCAACCTGCTGGAGTGCATCCGGGCCTGCAAGCCCCGCGAGGGCAAGCGCATCAGCAAACTCTATGGAAGTGCAGGCCCTCCCCCAACTGGTGAAGAGGATACAGCAGAATAAG-3′ and 5′-AATTCTTATTCTGCTGTATCCTCTTCACCAGTTGGGGGAGGGCCTGCACTTCCATAGAGTTTGCTGATGCGCTTGCCCTCGCGGGGCTTGCAGGCCCGGATGCACTCCAGCAGGTTGCTTTCCGTGGTGAGGTCCTGACACTGGCTGCTCTCCAGGCACCAGCCACGCACTTCCATGGAGGCCTGAAGCAGCAAGGCCAGCAACAGGGCCCCCGAGCGGCTGCAGCACGATCTCGGCATG-3′; for the plasmid containing mutant sequence (mP), we used the following oligonucleotides: 5′-GATCCATGCCGAGATCGTGCTGCAGCCGCTCGGGGGCCCTGTTGCTGGCCTTGCTGCTTCAGGCCTCCATGGAAGTGCGTGGCTGGTGCCTGGAGAGCAGCCAGTGTCAGGACCTCACCACGGAAAGCAACCTGCTGGAGTGCATCCGGGCCTGCAAGCCCCGCGAGGGCAAGCGCATCAGCAAACTCTATGGAAGTGCAGGCGCTGCCGCAACTGGTGAAGAGGATACAGCAGAATAAG-3′ and 5′-AATTCTTATTCTGCTGTATCCTCTTCACCAGTTGCGGCAGCGCCTGCACTTCCATAGAGTTTGCTGATGCGCTTGCCCTCGCGGGGCTTGCAGGCCCGGATGCACTCCAGCAGGTTGCTTTCCGTGGTGAGGTCCTGACACTGGCTGCTCTCCAGGCACCAGCCACGCACTTCCATGGAGGCCTGAAGCAGCAAGGCCAGCAACAGGGCCCCCGAGCGGCTGCAGCACGATCTCGGCATG-3′.
siRNA Knockdown and GRP78 Overexpression
Cells were transfected with Lipofectamine RNAiMAX Reagent (Thermo Fisher Scientific, Waltham, MA) containing siRNA (Dharmacon Inc., Lafayette, CO) to the final concentration of 60 pM by reverse transfection. Twenty-four hours post–siRNA transfection, plasmids containing FLAG-tagged wild-type or mutant GRP78 were transfected into cells using BioT reagent (Bioland Scientific, Paramount, CA). Culture media were replaced 5 hours post–BioT transfection with fresh media containing 60 pM siRNA. We used the following siRNAs: siGrp78_3′ untranslated region (UTR) 5′-CUUAAGUCUCGAAUGUAAUdTdT-3′; sictrl: 5′-GAGAUCGUAUAGCAACGGUdTdT-3′.
Plasmid Transfection
Plasmids were transfected into subconfluent cells with BioT reagent (Bioland Scientific, Paramount, CA) according to the manufacturer's instruction. Culture media were replaced 5 hours posttransfection, and cells were harvested 48 hours posttransfection for further analyses.
Western Blotting
Cells were lysed by radioimmunoprecipitation (RIPA) lysis buffer containing 50 mM Tris–HCl, pH 7.5, 150 mM NaCl, 0.5% sodium deoxycholate, 1% NP-40, 0.1% SDS, and a protease and phosphatase inhibitor cocktail (Roche, Basel, Switzerland). Whole cell lysates were cleared by centrifugation at 4°C and 13,000 rpm for 15 minutes. Proteins were analyzed by 10% SDS-PAGE and then transferred at 4°C overnight on nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA). Membranes were blocked by Tris-buffered saline containing 0.05% Tween-20 (TBST) and 5% nonfat dry milk at room temperature for 1 hour followed by incubation with primary antibody at 4°C overnight. Membranes were washed three times with TBST and then incubated with fluorescent IRDye-labeled antibodies or HRP-conjugated secondary antibodies. Fluorescent IRDye signal was detected by Odyssey (LI-COR Biosciences, Lincoln, NE). HRP signal was visualized by an ECL chemiluminescent substrate (Thermo Fisher Scientific, Waltham, MA) and then quantified with Image Lab software (Bio-Rad Laboratories, Hercules, CA).
Immunofluorescence and Confocal Microscopy
For detection of endogenous GRP78 and CD44 containing v3 exon intracellularly in MCF7-LR cells, cells were grown for 48 hours to subconfluence on sterile coverslips. Coverslips were sequentially coated with 50 μg/ml poly-L-lysine in ultrapure water (Sigma-Aldrich, St. Louis, MO) at RT for 1 hour and then 100 μg/ml collagen I from rat tail (Corning Inc., Corning, NY) in 0.02% acetic acid at RT for 2 hours. The cells were fixed in 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA) in Dulbecco's phosphate-buffered saline (DPBS) at RT for 10 minutes and then permeabilized by 0.3% saponin in PBS at RT for 15 minutes. Then the cells were incubated with blocking buffer containing 4% BSA and 0.01% saponin in PBS at RT for 3 hours. The primary antibody against GRP78 (MAb159, a kind gift from Dr. Parkash S. Gill at USC) was incubated with the cells at 4°C overnight in blocking buffer, followed by staining with the AlexaFluor-568 secondary antibody (Thermo Scientific, Waltham, MA) at 37°C for 40 minutes. Then the cells were treated with M.O.M. Mouse Ig Blocking Reagent (Vector Laboratories, Inc., Burlingame, CA) at 37°C for 3 hours to block mouse immunoglobulin from the primary antibody against GRP78. The cells were then incubated with the primary antibody against CD44 variable exon 3 at 4°C overnight in blocking buffer, followed by staining with the AlexaFluor-647 secondary antibody (Thermo Scientific, Waltham, MA) at 37°C for 40 minutes. Each step was followed by four washes in PBS containing 0.01% saponin. Coverslips were rinsed once with ultrapure water (Sigma-Aldrich, St. Louis, MO) before mounting with Vectashield anti-fade medium containing DAPI (Vector Laboratories, Inc., Burlingame, CA). Z-stack images were obtained on a Leica TCS SP8 confocal microscope equipped with a 63×/1.4 NA oil DIC objective lens and Leica Application Suite 10 Software (Leica Microsystems, Wetzlar, Germany).
For detection of endogenous GRP78 and CD44 containing v3 exon on the cell surface, MCF7-LR cells were grown for 48 hours to subconfluence on sterile coverslips. Coverslips were coated as described above. The cells were fixed in 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA) in DPBS at RT for 10 minutes and then incubated with blocking buffer containing 4% BSA in PBS at RT for 1 hour. The primary antibody against GRP78 (MAb159) was incubated with the cells at 4°C overnight in blocking buffer, followed by staining with AlexaFluor-594 secondary antibody (Thermo Scientific, Waltham, MA) at RT for 1 hour. Then, the cells were treated with M.O.M. Mouse Ig Blocking Reagent (Vector Laboratories, Inc., Burlingame, CA) at RT for 2 hours. Then the cells were incubated with the primary antibody against CD44 variable exon 3 (Thermo Scientific, Waltham, MA) at 4°C overnight in blocking buffer, followed by staining with AlexaFluor-488 secondary antibody (Thermo Scientific, Waltham, MA) at RT for 1 hour. Coverslips were washed four times following each step and rinsed once with ultrapure water (Sigma-Aldrich, St. Louis, MO) before mounting with Vectashield anti-fade medium containing DAPI (Vector Laboratories, Inc., Burlingame, CA). Z-stack images were obtained on a Zeiss LSM 510 confocal microscope equipped with a Plan-Apochromat 100×/1.4 NA oil DIC objective lens and LSM 510 version 4.2 SP1 acquisition software (Carl Zeiss, Oberkochen, Germany). Confocal images were analyzed using Adobe Photoshop CS2 software (Adobe Inc., San Jose, CA) with the same settings for all conditions under comparison.
Purification of GST-tagged Recombinant Proteins
Plasmids containing GST-tagged full-length GRP78 or deletion mutants were transformed into E. coli (BL21). The expression of the GST-fusion proteins was induced with 4 mM isopropyl-β-D-thiogalactoside when the optical density (OD600) of bacterial broth culture reached 0.5. Bacteria were then incubated at 37°C and 200 rpm for 4 hours to allow the expression of recombinant proteins. Cells were then lysed in TBS containing 50 mM Tris-Cl, pH 7.5, 150 mM NaCl, 1 mg/ml lysozyme, 1% Triton X-100, and protease and phosphatase inhibitor cocktails (Thermo Scientific, Waltham, MA). Bacterial cells were then sonicated for 4 minutes with 20 seconds on and 20 seconds off, followed by centrifugation at 4°C and 11,500 rpm for 1 hour. Supernatant was collected and incubated with Glutathione-Sepharose 4B beads (GE Healthcare, Chicago, IL) at 4°C for 12 hours. Recombinant GST-tagged protein was eluted with freshly prepared reduced glutathione (10 mM, Sigma-Aldrich, St. Louis, MO) at 4°C for 12 hours. The solution containing recombinant proteins was then buffer-exchanged to TBS using protein concentrators (Pall Corporation, Port Washington, NY). Recombinant proteins in TBS containing 15% glycerol were snap-frozen in liquid nitrogen and then stored at −80°C.
In Vitro GST Pull-Down Assay
Recombinant GST-tagged proteins were coupled to Glutathione-Sepharose 4B beads (GE Healthcare, Chicago, IL) at 4°C for 4 hours. Then, the beads were incubated with 1 mg whole cell lysate collected from 293T cells transiently expressing HA-tagged CD44v3-10 at 4°C overnight in IP lysis buffer (Thermo Fisher Scientific, Waltham, MA; 25 mM Tris–HCl, pH 7.4, 150 mM NaCl, 1 mM EDTA, 5% glycerol, 1% NP-40). The beads were then washed six times with IP lysis buffer, and the bound proteins were eluted from the beads with equal volume of 2× SDS sample buffer.
Purification of Cell Surface Proteins
Experiments were performed according to previously described protocol [10]. Briefly, cell surface proteins were biotinylated with 0.5 mg/ml EZ-Link Sulfo-NHS-SS-Biotin (Thermo Fisher Scientific, Waltham, MA) at 4°C for 30 minutes, and excessive biotin was quenched by four washes with glycine (100 mM) in PBS at 4°C. Cells were then lysed with RIPA lysis buffer (50 mM Tris–HCl, pH 7.5, 150 mM NaCl, 0.5% sodium deoxycholate, 1% NP-40, 0.1% SDS, and a protease and phosphatase inhibitor cocktail). The biotinylated cell surface proteins were captured on high-capacity NeutrAvidin agarose resin (Thermo Fisher Scientific, Waltham, MA).
WST-1 Viability Assay
Cell viability was assessed with the WST-1 reagent (Roche, Indianapolis, IN). Briefly, 24 hours posttransfection in six-well culture plate, 3000 cells per well were reseeded into 96-well culture plates with 100 μl culture medium per well. Then, in another 24 hours, the cell viability was measured by incubating each plate with 10 μl per well of WST-1 substrate for 3 hours, and then the plates were read at a wavelength of 450 nm with a reference wavelength of 655 nm.
Statistical Analysis
Data are presented as means ± SEM from three biological repeats. P values were calculated via two-tailed unpaired Student's t test. Statistical significance was represented as *P ≤ .05, **P < .01, and ***P < .001.
Results
Colocalization of GRP78 with CD44v Both in the ER Compartment and on the Cell Surface
In permeabilized MCF7-LR cells, immunofluorescent staining and confocal microscopy showed that endogenous GRP78 was expressed abundantly in the perinuclear as expected for its ER protein folding function (Figure 1A). As a transmembrane protein, CD44 is synthesized in the ER and traffics to the cell surface. CD44v, a variant isoform of CD44 containing variable exon 3 to 10 that is abundantly expressed in the MCF7-LR cells, was detected in the cytosolic and perinuclear region as well as inside the nucleus. Colocalization of GRP78 and CD44v was detected in the perinuclear region (Figure 1A). Since GRP78 is a major chaperone protein in the ER, it is likely that CD44v is a folding substrate of GRP78. In nonpermeabilized cells, GRP78 and CD44v were detected on the cell surface and showed substantial colocalization (Figure 1B). These results suggest that GRP78 and CD44v colocalize both in the ER compartment and on the cell surface.
Figure 1.
GRP78 colocalizes with CD44v in MCF7-LR breast cancer cells. (A) Immunofluorescence and confocal images showing the distribution and colocalization of GRP78 (red) and CD44v (green) in the saponin-permeabilized MCF7-LR breast cancer cells. GRP78 and CD44v were detected by MAb159 and anti-CD44v3 antibodies, respectively. The mouse-on-mouse (M.O.M.) control staining was performed with the same protocol as double-stained cells but lacking the primary antibody targeting CD44v. No primary control staining was conducted without the primary antibodies but with secondary antibodies. The nuclei were stained by DAPI in blue. Thickness of single immunofluorescent image section: 0.3 μm. Signals were visualized with 63×/1.4 NA objective on a Leica TCS SP8 confocal microscope. Scale bars, 20 μm. (B) Immunofluorescence and confocal images showing the distribution and colocalization of GRP78 (red) and CD44v (green) on the cell surface of nonpermeabilized MCF7-LR breast cancer cells. GRP78 and CD44v were detected by MAb159 and anti-CD44v3 antibodies, respectively. The M.O.M. control staining was performed with the same protocol as double-stained cells but lacking primary antibody targeting CD44v. No primary control staining was conducted without the primary antibodies but with secondary antibodies. The nuclei were stained by DAPI in blue. DIC: differential interference contrast. Thickness of single immunofluorescent image section: 0.8 μm. Cell peripheries were outlined with black lines. Signals were visualized with 100×/1.4 NA objective on an LSM 510 confocal microscope. Scale bars, 20 μm.
Requirement of the COOH-terminal PRR of GRP78 for Forming Complex with CD44v In Vitro
We next characterized the region of GRP78 binding to CD44v. A schematic drawing of the identified ER signal, ATPase domain, substrate binding domain, and the KDEL Golgi-to-ER retrieval motif of GRP78 is shown in Figure 2A. A panel of GST-tagged GRP78 deletion mutants was created (Figure 2A). We purified the recombinant GST-tagged GRP78 full-length (FL) and deletion mutants from E. coli (BL21) and then incubated them with whole cell lysates containing transiently expressed HA-tagged CD44v (vHA, Figure 2B). The immunoblots of these GST pull-down assays revealed that GRP78 binds CD44v via the regions localized in its COOH-terminal half region (Figure 2C). The binding of FL, C (a.a. 413-654), and ΔKDEL mutants to vHA was comparable when the amount of binding was normalized to the level of input GST-tagged proteins. Interestingly, an increased complex formation was observed upon deletion of the last 11 amino acids of GRP78 (ΔC11, a.a. 19-643), suggesting that this deletion likely exposed this region for more effective binding and/or forming a more stable complex. The deletion of the last 17 amino acids of GRP78 (ΔC17, a.a. 19-637) reduced its capacity to form complex with vHA. The deletion of the last 73 amino acids of GRP78 (ΔC73, a.a. 19-581) abolished binding to vHA. These results suggest that the amino acids between 638 and 643 of GRP78 are essential for forming complex with vHA. We then explored if the COOH-terminal region of GRP78 devoid of the substrate binding domain was sufficient for complex formation with vHA. Surprisingly, the last 73 amino acids of GRP78 alone (C73) could not form complex with vHA (Figure 2D). Collectively, these results indicate that a.a. 638 to 643 of GRP78 are essential but not sufficient for GRP78 to form complex with CD44v. A closer look at the amino acid sequence between a.a. 638 and 643 of GRP78 revealed a PRR containing three consecutive prolines between a.a. 640 and 642 localized outside the SBD. A computational three-dimentional protein structure of human GRP78 modeled by I-TASSER [28], [29], [30] showed that these three consecutive prolines created a turn in a less structured region of the last 27 amino acids of GRP78 (C27) as depicted in Figure 2E.
Figure 2.
The COOH-terminal proline-rich region of GRP78 is essential for forming complex with CD44v in vitro. (A) Schematic representation of the human GST-tagged GRP78 wild-type and deletion mutants cloned into pGEX-4T-1 backbone vector. a.a., amino acids. FL, a.a. 19-654; N, a.a. 19-407; C, a.a. 413-654; ΔKDEL, a.a. 19-650; ΔC11, a.a. 19-643; ΔC17, a.a. 19-637; ΔC73, a.a. 19-581; C73, a.a. 582-654. The locations of the ER signal, ATPase domain, substrate binding domain, proline-rich region, and KDEL motif of GRP78 are depicted on top. (B) Schematic representation of the expression construct of HA-tagged human CD44 containing variable exon 3 to 10. EC, extracellular; TM, transmembrane; IC, intracellular. (C-D) Western blot analysis of samples from in vitro GST pull-down assay. GST or GST-tagged GRP78 wild-type and mutant proteins purified from E. coli (BL21) were incubated with 293T whole cell lysate containing overexpressed CD44v-HA (vHA). (E) Upper panel: I-TASSER model of full-length human GRP78 protein. ATPase domain is in light blue. SBD is in orange. The last 27 amino acids close to the COOH-terminus (C27) are lighted in rainbow color and by dashed circle. Arrow indicates the PPP polyproline amino acids (red). Lower panel: The enlarged C27 area shows the polyproline amino acids (a.a. 640-642; red) and the adjacent protein structure.
The Importance of the COOH-terminal PRR of GRP78 for Its Cell Surface Expression and Modulation of STAT3 Signaling
Proline-rich region plays important roles in mediating protein-protein interactions, and the specificity of interactions can be finely tuned by the proline-rich sequence [31]. On the cell surface, GRP78 largely exists as a peripheral protein [10] and associates with GPI-anchored [11], [32], [33] or transmembrane partner proteins [18], [34], [35]. Since the PRR is essential for GRP78 to form complex with CD44v, we hypothesized that the COOH-terminal PRR of GRP78 may be critical for its cell surface expression through its ability to form complex with partner proteins such as CD44v. To test this hypothesis, we created expression plasmids in pcDNA3 backbone vector (v) for FLAG-tagged human GRP78 (F-GRP78) wild-type (WT) and mutant harboring a PPP to AAA mutation (AAA) at a.a. 640-642 (Figure 3A). We then purified cell surface proteins of MCF7-LR cells transfected with v, WT, or AAA plasmids by cell surface biotinylation and high-capacity avidin pull-down, and we found that the AAA mutant showed about 65% decrease of cell surface expression compared to the WT (Figure 3, B and C). These results indicate that the COOH-terminal polyproline sequence of GRP78 plays a previously unidentified role in mediating its cell surface expression in tamoxifen-resistant breast cancer cells.
Figure 3.
The COOH-terminal polyproline sequence of GRP78 is critical for its cell surface expression and modulates STAT3 signaling in MCF7-LR breast cancer cells. (A) Schematic representation of FLAG-tagged human GRP78 (F-GRP78) wild-type (WT) and mutant harboring a PPP to AAA mutation (AAA). a.a., amino acids. SBD, substrate binding domain. (B) Western blot analysis of MCF7-LR cells transfected with FLAG-tagged GRP78 WT, AAA, or pcDNA3 backbone vector (v). Annexin II and β-actin are loading controls for cell surface fraction and whole cell lysate, respectively. (C) Quantitative analysis of cell surface (cs) F-GRP78 levels. Data represent means ± SEM from three biological repeats. **P < .01 by Student's t test. (D) Schematic illustration of siRNA designed to target 3′ UTR of Grp78. WT and AAA expression plasmids do not contain 3′ UTR. (E) Western blot analysis of MCF7-LR cells co-transfected with v, WT, or AAA and control (sictrl) or Grp78 siRNA targeting 3′ UTR (siGrp78, 3′ UTR). Numbers below the β-actin bands represent individual experimental conditions. (F) Quantification of the ratio of pSTAT3 (Y705) and STAT3 of each experimental condition as shown in panel E. Data represent means ± SEM from three biological repeats. *P ≤ .05 by Student's t test.
We next explored the signaling function of this polyproline sequence by transfecting the v, WT, and AAA mutant into MCF7-LR cells. As a proof of principle, we focused on the activation of signal transducer and activator of transcription 3 (STAT3) through phosphorylation at Tyr705 (pY705) because it is a common signaling pathway of CD44 [36], [37] and GRP78 [38] and plays important roles in cell proliferation and migration in breast cancer. GRP78 is abundantly expressed in MCF7-LR cells; thus, we co-transfected the plasmids for transient expression with the siRNA targeting the 3′ UTR of endogenous Grp78 mRNA (Figure 3D). We observed about 30% reduction of STAT3 activation upon knockdown of GRP78 and an increase of STAT3 activation when WT GRP78 was transiently expressed, and this stimulation was blunted when AAA was introduced (Figure 3, E and F). This result suggests that the polyproline sequence close to the COOH-terminus of GRP78 is a novel regulator of STAT3 activation in tamoxifen-resistant breast cancer cells.
Conservation of the COOH-terminal Polyproline Sequence of GRP78 in Higher Eukaryotic Organisms
A closer examination at the amino acid sequence of human GRP78 revealed that another consensus proline-rich region (a.a. 487-496) containing a classical xPPxP (IPPAP, a.a. 487-491) motif and a PQ sequence (a.a. 495 and 496) preceded the PPP polyproline motif (a.a. 640-642). The three-dimensional localizations of these two proline-rich regions were mapped to the corresponding sites on the computational GRP78 protein structure (a.a. 413-654) modeled by I-TASSER, and the result showed that these two sequences were both localized to more unstructured regions of GRP78 protein (Figure 4A). The proline residue, P495, localized between a.a. 487 and 496, was reported to facilitate the substrate binding function of GRP78 [39]; however, the function of the PPP sequence close to the COOH-terminus of GRP78 is largely unknown.
Figure 4.
Comparisons of the proline-rich regions of GRP78. (A) I-TASSER model of human GRP78 (a.a. 413-654) demonstrating the three-dimentional localizations of PPP and IPPAP/PQ proline-rich sequences. The classical IPPAP motif was highlighted in magenta, the PQ amino acids in blue, and the COOH-terminal PPP polyproline amino acids in red. (B) Results of multiple sequence alignment of GRP78 COOH-terminal proline-rich regions from a broad range of species using MEGA-X software with polyproline residues in red. DnaK is a bacterial homolog of GRP78. Asterisk indicates conserved residue among all species. Dots show conserved residues with human (Homo sapiens) GRP78. Dashes indicate gaps. (C) Results of multiple sequence alignment of GRP78 IPPAP/PQ proline-rich regions from a broad range of species using MEGA-X software with the proline residues localized at the classical IPPAP motif in magenta and the proline residue localized at the PQ amino acids in blue. DnaK is a bacterial homolog of GRP78. Asterisks indicate conserved residues among all species. Dots show conserved residues with human GRP78. (D) The COOH-terminal amino acid sequences of other human (H. sapiens) heat shock proteins with polyproline residues in red.
Multiple sequence alignment provides insights into the evolutionary, structural, and functional relationships among the amino acid sequences. We applied Clustal analysis using MEGA-X software [40] to compare the proline-rich sequences of GRP78 homologs from a diverse set of species (Figure 4, B and C). Interestingly, we found that the GRP78 COOH-terminal PPP polyproline sequence was conserved among Homo sapiens, Rattus norvegicus, Gallus gallus, Xenopus laevis, and Stylophora pistillata. Although Latimeria chalumnae, Priapulus caudatus, and Octopus bimaculoides did not show triproline sequence, they exhibited two consecutive proline residues. However, Saccharomyces cerevisiae and GRP78 homolog DnaK in Escherichia coli did not show proline residues in their COOH-termini (Figure 4B). These results suggest that the presence of GRP78 COOH-terminal polyproline sequence is likely a unique feature in higher eukaryotic organisms. On the other hand, the IPPAP/PQ proline-rich region was highly conserved among all species, with Octopus bimaculoides and GRP78 homolog DnaK in Escherichia coli exhibiting single amino acid variances (Figure 4C). We then asked whether the PPP polyproline sequence is unique to GRP78 or common to heat shock family proteins. Therefore, we examined the COOH-terminal amino acid sequences of human heat shock 70 (HSP70) and 90 (HSP90) family chaperones (Figure 4D). We did not observe PPP polyproline sequence in these heat shock family proteins. Of note, HSC70, HSP90α, and HSP90β showed two consecutive prolines. This result indicates that the COOH-terminal PPP polyproline sequence is unique in GRP78 among the listed heat-shock proteins in human.
Promotion of Apoptosis in Tamoxifen-resistant Breast Cancer by Enforced Expression of a Short Peptide Bearing the PRR of GRP78
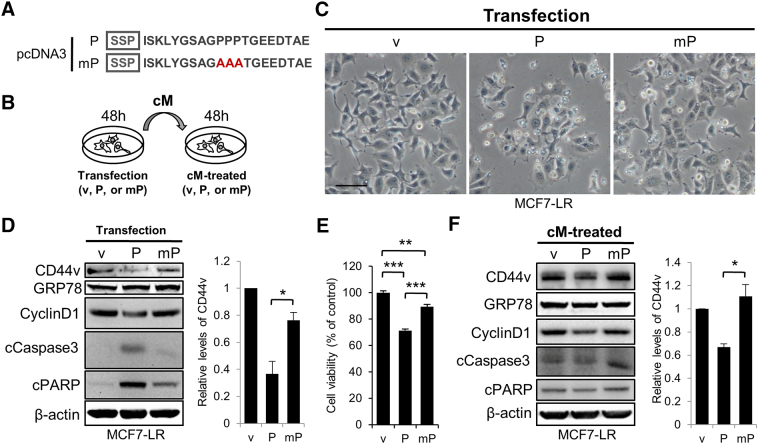
Overexpression of GRP78 in tumor lesions from breast cancer patients has been associated with the development of therapeutic resistance, and knockdown of GRP78 can resensitize tamoxifen-resistant breast cancer cells to tamoxifen [41]. Thus, we investigated the feasibility of utilization of small peptide encoding the COOH-terminal PRR of GRP78 to target potential protein-protein interactions mediated by the PRR of GRP78 in tamoxifen-resistant breast cancer cells. We first constructed expression plasmids in a pcDNA3 backbone vector (v) containing wild-type (P) or mutant (mP) sequence encoding 20 amino acids spanning the COOH-terminal polyproline sequence (a.a. 631-650) of GRP78. These sequences were preceded by a previously described secretory and sorting peptide of α-melanocyte-stimulating hormone (SSP) [27] (Figure 5A). We selected this region because it is localized in between SBD and KDEL motif, and it preserves some conformational and sequence information in addition to the PPP sequence, as previous studies have shown that the flanking region of core proline-rich sequence also substantially contributed to the specificity and affinity of PRR-mediated protein-protein interactions [42], [43]. The inclusion of the sorting and secretory sequences predicted that the peptides would be localized to the ER as well as secreted into the medium. We then analyzed the MCF7-LR cells transfected with the plasmids as well as the cells applied with the conditional media (cM) obtained from the culture media of transfected cells as depicted in Figure 5B. Notably, more cells became rounded and detached from the culture surface when they were transfected with the plasmid containing wild-type PRR sequence (P) compared to the cells transfected with v and mP plasmids (Figure 5C). Further analyses of the cell lysates from transfected cells by SDS-PAGE and immunoblotting revealed that the cells transfected with the plasmid containing wild-type PRR sequence (P) exhibited a significant reduction of CD44v level as well as a reduction of proliferation marker Cyclin D1. Additionally, they demonstrated an increase in apoptotic markers cleaved Caspase-3 and PARP compared to the v and mP controls (Figure 5D). In agreement with this result, cells transfected with the plasmid containing wild-type PRR sequence (P) displayed a decrease of cell viability as shown by WST-1 assay (Figure 5E). These results further demonstrate the functional importance of the COOH-terminal PRR of GRP78.
Figure 5.
Enforced expression of short peptide encoding the proline-rich region of GRP78 reduced cell viability and promoted apoptosis in MCF7-LR breast cancer cells. (A) Schematic illustration of expression plasmids containing wild-type or mutant sequences encoding 20 amino acids spanning the proline-rich region of GRP78 (a.a. 640-642). The sequences were preceded by a secretory and sorting peptide sequence of α-melanocyte-stimulating hormone (SSP). The plasmids were constructed in pcDNA3 backbone vector (v). P, the plasmid containing wild-type proline-rich sequence. mP, the plasmid containing PPP to AAA mutation. (B) Schematic representation of experimental procedures. cM, conditional media. h, hours. (C) Bright-field images of MCF7-LR cells with the enforced expression of v, P, or mP. Images were photographed 48 hours post transfection. Scale bar, 50 μm. (D) Left: Western blot analysis of whole cell lysates from MCF7-LR cells transfected with v, P, or mP. Cells lysates were collected 48 hours posttransfection. CD44v and GRP78 were detected by anti-CD44v3 and MAb159 antibodies, respectively. β-Actin is loading control. Right: Quantification of CD44v levels compared to the control. Data represent means ± SEM from three biological repeats. *P ≤ .05 by Student's t test. (E) Quantitative analysis of cell viability by WST-1 assay in MCF7-LR cells overexpressing v, P, or mP. Cells were analyzed 48 hours posttransfection. Data represent means ± SEM from four experimental repeats. **P < .01 ***P < .001 by Student's t test. (F) Left: Western blot analysis of whole cell lysates from MCF7-LR cells treated with cM collected from MCF7-LR cells transfected with v, P, or mP. Conditional media were collected 48 hours posttransfection and then applied to new cells for another 48 hours. CD44v and GRP78 were detected by anti-CD44v3 and MAb159 antibodies, respectively. β-Actin is loading control. Right: Quantification of CD44v levels compared to the control. Data represent means ± SEM from three biological repeats. *P ≤ .05 by Student's t test.
To test the functionality of the secreted peptides, we treated MCF7-LR cells with the cM harvested from the culture media of transfected cells (Figure 5F). Similar to cells transfected with the plasmids, cells treated with the cM collected from the cells transfected with the plasmid containing wild-type PRR sequence (P), but not the mP sequence, showed a significant reduction of CD44v level and a decrease in proliferation marker Cyclin D1 (Figure 5F). However, these reductions did not lead to change of cell morphology (data not shown) and increase in cell apoptotic markers (Figure 5F). This was likely because the forced expression would continuously supply the small peptides, while the amount in the collected conditional media is limiting.
Discussion
Tamoxifen resistance presents a major challenge for the treatment of estrogen receptor–positive breast cancer as that is associated with disease relapse and a poor prognosis. Therefore, it is important to identify molecular pathways that can be used to target tamoxifen-resistant breast cancer cells. Our previous studies revealed that in human tamoxifen-resistant breast cancer cell line MCF7-LR, the molecular chaperone GRP78 was elevated both intracellularly and on the cell surface [4]. This same cell line also expressed a very high level of CD44v. Interestingly, both GRP78 and CD44 have been documented to play important roles in mediating endocrine resistance in breast cancer [21], [22], [44], [45]. In this study, while we explored the physical and functional relationship between GRP78 and CD44v in the MCF7-LR cells, we uncovered several novel findings that advance our understanding on the requirement of GRP78 to express on the cell surface and how CD44v level and signaling are modulated in these cells.
Through confocal microscopy, we observed substantial colocalizations of GRP78 and CD44v in the intracellular compartments and on the cell surface of MCF7-LR cells. Their cytosolic colocalizations are presumably in the ER since GRP78 is a major ER resident protein, and this suggests that CD44v is a folding substrate of GRP78. Interestingly, we found that most of the cultured cells showed nuclear CD44v. Because we used the antibody against the extracellular variable exon 3 of CD44v for the immunofluorescent staining, it is likely that it is the full-length CD44v in the nuclei. All CD44 isoforms contain a bipartite nuclear localization signal (NLS) at their cytosolic COOH-terminal region [46], and the full-length CD44 was reported internalized upon ligand binding. The internalized full-length CD44 formed complex with STAT3 in the cytosol and facilitated the acetylation and activation of STAT3, and then the complex traveled together into the nucleus and binding to the promoter of Cyclin D1, leading to elevated expression of Cyclin D1 [47]. The same study [47] also showed that the nuclear CD44 bound to the 5′ sequence of the Grp78 allele, and it remains to be determined whether nuclear CD44 can directly facilitate the expression of Grp78 at the transcriptional level.
GRP78 has well-established roles as a chaperone protein that facilitates substrate folding through its SBD domain (a.a. 413-627) and the subsequent releasing of the substrate through the ATP hydrolysis by the ATPase domain (a.a. 19-407). Despite these advances, the function of the COOH-terminal sequence between a.a. 628 and 650 preceding the KDEL Golgi-to-ER retrieval motif is largely unknown, although antibodies against the COOH-terminal region of GRP78 have shown efficacy in suppressing cancer growth and metastasis [14], [20]. Here, we uncovered that the GRP78 COOH-terminal proline-rich region containing three consecutive prolines (a.a. 640-642) is essential for complex formation with CD44v as demonstrated by the in vitro GST-binding assay. However, such capacity is not sufficient and likely also dependent on the substrate binding function of GRP78 as we have found that the last 73 amino acids alone are unable to form complex with CD44v, whereas the subdomain spanning the COOH-terminal half from a.a. 413 to 654 is sufficient. Importantly, we identified that this PPP polyproline sequence is critical for GRP78 expression on the cell surface of MCF7-LR cells. Since GRP78 colocalized with CD44v both intracellularly and on the cell surface, it is tempting to speculate that the cell surface transmembrane protein CD44v forms complex with GRP78 in the ER and facilitates GRP78 cell surface translocation.
Proline-rich sequence is an important mediator of protein-protein interactions [31]. A previous genome-wide study [48] showed that the frequencies of PPP and PPG motifs were low in prokaryotes and drastically increased with the complexity of eukaryotic organisms. Here, we found that the COOH-terminal PPP polyproline sequence is uniquely presented in the GRP78 of higher eukaryotic organisms but not the yeast (S. cerevisiae) and E. coli as shown from the result of multiple sequence alignment. This implies that the PPP polyproline sequence of GRP78 may have functions specific to higher eukaryotes. Interestingly, the result of multiple sequence alignments suggests that the PPP sequence is evolved from a D/E rich region. Proline is nonpolar and is the only cyclic amino acid. It is often found in turns or loops or at the end of α-helix. Its amino acid property is very different from the acidic amino acid aspartic acid (D) and glutamic acid (E). This further supports the idea of functional divergence of the PPP polyproline sequence during evolution. Additionally, the PPP polyproline sequence is localized in a region with a lot more sequence variance compared to the region bearing IPPAP/PQ sequences within the substrate binding domain among the analyzed species. In future studies, it will be interesting to determine whether the PPP sequence/region may bear functions independent of the traditional view of GRP78 as a foldase.
GRP78 and CD44v have both been reported to mediate STAT3 signaling in breast cancer cells [38], [49], and STAT3 signaling is also important in mediating tamoxifen resistance in breast cancer cells [36]. We found that this polyproline sequence is essential for STAT3 activation mediated by enforced expression of GRP78, providing the first evidence that the COOH-terminal polyproline sequence is a previously unidentified modulator of STAT3 activation. In testing the functional consequence of enforced expression of the COOH-terminal peptide of GRP78 (a.a. 631-650) in MCF7L-R cells, we discovered that enforced expression of the wild-type peptide but not the mutant bearing AAA mutation to the PPP motif caused dramatic rounding of the cells leading to detachment, associating with substantial decrease in CD44v level, increase of apoptotic markers, and reduction in cell viability. This raises the interesting possibility that enforced expression of this GRP78 COOH-terminal peptide may disrupt the interaction of GRP78 with CD44 as well as other unidentified binding/folding candidate proteins critical for promoting tamoxifen resistance, thus representing a novel approach to combat endocrine resistance in breast cancer. Taken together, we identified that the COOH-terminal polyproline sequence of GRP78 is a new evolutionary feature gained by higher eukaryotic organisms, and our biochemical and cellular studies supported the perspective that this sequence harbors previously unidentified signaling functions in regulating cell surface expression of GRP78 and STAT3 activation in tamoxifen-resistant breast cancer cells. The basic and clinical implications of these findings warrant vigorous investigation in the future.
Acknowledgements
We thank Rachel Schiff (Baylor College of Medicine, TX) for the gift of MCF7-LR breast cancer cells, Parkash S. Gill for the gift of GRP78 antibody, and Daisy Flores Rangel and Dat Ha for helpful discussions. We thank the Cell and Tissue Imaging Core of the USC Research Center for Liver Diseases for assistance of confocal microscopy.
Footnotes
Funding: This work was supported by the National Institutes of Health USA (ASL: NIH-5R01CA027607 and USC Research Center for Liver Diseases: NIH-5P30DK048522).
Declaration of interest: none.
Author contributions: C.-C. Tseng conceived initial idea for the project. C.-C. Tseng, P. Zhang, and A. S. Lee conceived and designed the experiments. C.-C. Tseng, P. Zhang, and A. S. Lee wrote the manuscript. C.-C. Tseng and P. Zhang performed experiments.
Contributor Information
Chun-Chih Tseng, Email: tsengchu@usc.edu.
Pu Zhang, Email: puzhang@usc.edu.
Amy S. Lee, Email: amylee@usc.edu.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. doi: 10.3322/caac.21442. [DOI] [PubMed] [Google Scholar]
- 2.Harvey JM, Clark GM, Osborne CK, Allred DC. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol. 1999;17(5):1474–1481. doi: 10.1200/JCO.1999.17.5.1474. [DOI] [PubMed] [Google Scholar]
- 3.Davies C, Godwin J, Gray R, Clarke M, Cutter D, Darby S, McGale P, Pan HC, Taylor C. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378(9793):771–784. doi: 10.1016/S0140-6736(11)60993-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang Y, Tseng CC, Tsai YL, Fu X, Schiff R, Lee AS. Cancer cells resistant to therapy promote cell surface relocalization of GRP78 which complexes with PI3K and enhances PI(3,4,5)P3 production. PLoS One. 2013;8(11) doi: 10.1371/journal.pone.0080071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ni M, Lee AS. ER chaperones in mammalian development and human diseases. FEBS Lett. 2007;581:3641–3651. doi: 10.1016/j.febslet.2007.04.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luo B, Lee AS. The critical roles of endoplasmic reticulum chaperones and unfolded protein response in tumorigenesis and anticancer therapies. Oncogene. 2013;32(7):805–818. doi: 10.1038/onc.2012.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee AS. Glucose-regulated proteins in cancer: molecular mechanisms and therapeutic potential. Nat Rev Cancer. 2014;14(4):263–276. doi: 10.1038/nrc3701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Munro S, Pelham HR. An Hsp70-like protein in the ER: identity with the 78 kd glucose-regulated protein and immunoglobulin heavy chain binding protein. Cell. 1986;46:291–300. doi: 10.1016/0092-8674(86)90746-4. [DOI] [PubMed] [Google Scholar]
- 9.Ni M, Zhang Y, Lee AS. Beyond the endoplasmic reticulum: atypical GRP78 in cell viability, signalling and therapeutic targeting. Biochem J. 2011;434(2):181–188. doi: 10.1042/BJ20101569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tsai YL, Zhang Y, Tseng CC, Stanciauskas R, Pinaud F, Lee AS. Characterization and mechanism of stress-induced translocation of 78-kilodalton glucose-regulated protein (GRP78) to the cell surface. J Biol Chem. 2015;290(13):8049–8064. doi: 10.1074/jbc.M114.618736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsai YL, Ha DP, Zhao H, Carlos AJ, Wei S, Pun TK, Wu K, Zandi E, Kelly K. Endoplasmic reticulum stress activates SRC, relocating chaperones to the cell surface where GRP78/CD109 blocks TGF-beta signaling. Proc Natl Acad Sci U S A. 2018;115(18):E4245–E4254. doi: 10.1073/pnas.1714866115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Arap MA, Lahdenranta J, Mintz PJ, Hajitou A, Sarkis AS, Arap W, Pasqualini R. Cell surface expression of the stress response chaperone GRP78 enables tumor targeting by circulating ligands. Cancer Cell. 2004;6(3):275–284. doi: 10.1016/j.ccr.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 13.Sato M, Yao VJ, Arap W, Pasqualini R. GRP78 signaling hub. a receptor for targeted tumor therapy Adv Genet. 2010;69:97–114. doi: 10.1016/S0065-2660(10)69006-2. [DOI] [PubMed] [Google Scholar]
- 14.Liu R, Li X, Gao W, Zhou Y, Wey S, Mitra SK, Krasnoperov V, Dong D, Liu S. Monoclonal antibody against cell surface GRP78 as a novel agent in suppressing PI3K/AKT signaling, tumor growth, and metastasis. Clin Cancer Res. 2013;19(24):6802–6811. doi: 10.1158/1078-0432.CCR-13-1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dobroff AS, D'Angelo S, Eckhardt BL, Ferrara F, Staquicini DI, Cardo-Vila M, Staquicini FI, Nunes DN, Kim K. Towards a transcriptome-based theranostic platform for unfavorable breast cancer phenotypes. Proc Natl Acad Sci U S A. 2016;113(45):12780–12785. doi: 10.1073/pnas.1615288113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bakewell SJ, Rangel DF, Ha DP, Sethuraman J, Crouse R, Hadley E, Costich TL, Zhou X, Nichols P. Suppression of stress induction of the 78-kilodalton glucose regulated protein (GRP78) in cancer by IT-139, an anti-tumor ruthenium small molecule inhibitor. Oncotarget. 2018;9(51):29698–29714. doi: 10.18632/oncotarget.25679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.D'Angelo S, Staquicini FI, Ferrara F, Staquicini DI, Sharma G, Tarleton CA, Nguyen H, Naranjo LA, Sidman RL. Selection of phage-displayed accessible recombinant targeted antibodies (SPARTA): methodology and applications. JCI Insight. 2018;3(9) doi: 10.1172/jci.insight.98305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Misra UK, Gonzalez-Gronow M, Gawdi G, Pizzo SV. The role of MTJ-1 in cell surface translocation of GRP78, a receptor for alpha 2-macroglobulin-dependent signaling. J Immunol. 2005;174(4):2092–2097. doi: 10.4049/jimmunol.174.4.2092. [DOI] [PubMed] [Google Scholar]
- 19.Awad W, Estrada I, Shen Y, Hendershot LM. BiP mutants that are unable to interact with endoplasmic reticulum DnaJ proteins provide insights into interdomain interactions in BiP. Proc Natl Acad Sci U S A. 2008;105(4):1164–1169. doi: 10.1073/pnas.0702132105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ray R, de Ridder GG, Eu JP, Paton AW, Paton JC, Pizzo SV. The Escherichia coli subtilase cytotoxin A subunit specifically cleaves cell-surface GRP78 protein and abolishes COOH-terminal-dependent signaling. J Biol Chem. 2012;287(39):32755–32769. doi: 10.1074/jbc.M112.399808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hiscox S, Baruha B, Smith C, Bellerby R, Goddard L, Jordan N, Poghosyan Z, Nicholson RI, Barrett-Lee P. Overexpression of CD44 accompanies acquired tamoxifen resistance in MCF7 cells and augments their sensitivity to the stromal factors, heregulin and hyaluronan. BMC Cancer. 2012;12:458. doi: 10.1186/1471-2407-12-458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bellerby R, Smith C, Kyme S, Gee J, Gunthert U, Green A, Rakha E, Barrett-Lee P, Hiscox S. Overexpression of specific CD44 isoforms is associated with aggressive cell features in acquired endocrine resistance. Front Oncol. 2016;6:145. doi: 10.3389/fonc.2016.00145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zoller M. CD44: can a cancer-initiating cell profit from an abundantly expressed molecule? Nat Rev Cancer. 2011;11(4):254–267. doi: 10.1038/nrc3023. [DOI] [PubMed] [Google Scholar]
- 24.Yae T, Tsuchihashi K, Ishimoto T, Motohara T, Yoshikawa M, Yoshida GJ, Wada T, Masuko T, Mogushi K. Alternative splicing of CD44 mRNA by ESRP1 enhances lung colonization of metastatic cancer cell. Nat Commun. 2012;3:883. doi: 10.1038/ncomms1892. [DOI] [PubMed] [Google Scholar]
- 25.Ghatak S, Hascall VC, Markwald RR, Misra S. Stromal hyaluronan interaction with epithelial CD44 variants promotes prostate cancer invasiveness by augmenting expression and function of hepatocyte growth factor and androgen receptor. J Biol Chem. 2010;285(26):19821–19832. doi: 10.1074/jbc.M110.104273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu J, Li G, Zhang P, Zhuang X, Hu G. A CD44v(+) subpopulation of breast cancer stem-like cells with enhanced lung metastasis capacity. Cell Death Dis. 2017;8(3):e2679. doi: 10.1038/cddis.2017.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yin P, Luby TM, Chen H, Etemad-Moghadam B, Lee D, Aziz N, Ramstedt U, Hedley ML. Generation of expression constructs that secrete bioactive alphaMSH and their use in the treatment of experimental autoimmune encephalomyelitis. Gene Ther. 2003;10(4):348–355. doi: 10.1038/sj.gt.3301902. [DOI] [PubMed] [Google Scholar]
- 28.Roy A, Kucukural A, Zhang Y. I-TASSER: a unified platform for automated protein structure and function prediction. Nat Protoc. 2010;5(4):725–738. doi: 10.1038/nprot.2010.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang J, Yan R, Roy A, Xu D, Poisson J, Zhang Y. The I-TASSER Suite: protein structure and function prediction. Nat Methods. 2015;12(1):7–8. doi: 10.1038/nmeth.3213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang Y. I-TASSER server for protein 3D structure prediction. BMC Bioinformatics. 2008;9:40. doi: 10.1186/1471-2105-9-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kay BK, Williamson MP, Sudol M. The importance of being proline: the interaction of proline-rich motifs in signaling proteins with their cognate domains. FASEB J. 2000;14(2):231–241. [PubMed] [Google Scholar]
- 32.Kouznetsova VL, Hu H, Teigen K, Zanetti M, Tsigelny IF. Cripto stabilizes GRP78 on the cell membrane. Protein Sci. 2018;27(3):653–661. doi: 10.1002/pro.3358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Philippova M, Ivanov D, Joshi MB, Kyriakakis E, Rupp K, Afonyushkin T, Bochkov V, Erne P, Resink TJ. Identification of proteins associating with glycosylphosphatidylinositol-anchored T-cadherin on the surface of vascular endothelial cells: role for Grp78/BiP in T-cadherin–dependent cell survival. Mol Cell Biol. 2008;28(12):4004–4017. doi: 10.1128/MCB.00157-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Triantafilou M, Fradelizi D, Triantafilou K. Major histocompatibility class one molecule associates with glucose regulated protein (GRP) 78 on the cell surface. Hum Immunol. 2001;62:764–770. doi: 10.1016/s0198-8859(01)00269-5. [DOI] [PubMed] [Google Scholar]
- 35.Bhattacharjee G, Ahamed J, Pedersen B, El-Sheikh A, Mackman N, Ruf W, Liu C, Edgington TS. Regulation of tissue factor–mediated initiation of the coagulation cascade by cell surface grp78. Arterioscler Thromb Vasc Biol. 2005;25(8):1737–1743. doi: 10.1161/01.ATV.0000173419.31242.56. [DOI] [PubMed] [Google Scholar]
- 36.Wang X, Wang G, Zhao Y, Liu X, Ding Q, Shi J, Ding Y, Wang S. STAT3 mediates resistance of CD44(+)CD24(−/low) breast cancer stem cells to tamoxifen in vitro. J Biomed Res. 2012;26(5):325–335. doi: 10.7555/JBR.26.20110050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xu H, Tian Y, Yuan X, Wu H, Liu Q, Pestell RG, Wu K. The role of CD44 in epithelial-mesenchymal transition and cancer development. Onco Targets Ther. 2015;8:3783–3792. doi: 10.2147/OTT.S95470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yao X, Liu H, Zhang X, Zhang L, Li X, Wang C, Sun S. Cell surface GRP78 accelerated breast cancer cell proliferation and migration by activating STAT3. PLoS One. 2015;10(5) doi: 10.1371/journal.pone.0125634. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 39.Shen J, Snapp EL, Lippincott-Schwartz J, Prywes R. Stable binding of ATF6 to BiP in the endoplasmic reticulum stress response. Mol Cell Biol. 2005;25(3):921–932. doi: 10.1128/MCB.25.3.921-932.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018;35(6):1547–1549. doi: 10.1093/molbev/msy096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cook KL, Clarke R. Role of GRP78 in promoting therapeutic-resistant breast cancer. Future Med Chem. 2015;7(12):1529–1534. doi: 10.4155/fmc.15.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Holtzman JH, Woronowicz K, Golemi-Kotra D, Schepartz A. Miniature protein ligands for EVH1 domains: interplay between affinity, specificity, and cell motility. Biochemistry. 2007;46(47):13541–13553. doi: 10.1021/bi700975f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ball LJ, Kuhne R, Hoffmann B, Hafner A, Schmieder P, Volkmer-Engert R, Hof M, Wahl M, Schneider-Mergener J. Dual epitope recognition by the VASP EVH1 domain modulates polyproline ligand specificity and binding affinity. EMBO J. 2000;19(18):4903–4914. doi: 10.1093/emboj/19.18.4903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fu Y, Li J, Lee AS. GRP78/BiP inhibits endoplasmic reticulum BIK and protects human breast cancer cells against estrogen starvation-induced apoptosis. Cancer Res. 2007;67(8):3734–3740. doi: 10.1158/0008-5472.CAN-06-4594. [DOI] [PubMed] [Google Scholar]
- 45.Cook KL, Shajahan AN, Warri A, Jin L, Hilakivi-Clarke LA, Clarke R. Glucose-regulated protein 78 controls cross-talk between apoptosis and autophagy to determine antiestrogen responsiveness. Cancer Res. 2012;72(13):3337–3349. doi: 10.1158/0008-5472.CAN-12-0269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Janiszewska M, De Vito C, Le Bitoux MA, Fusco C, Stamenkovic I. Transportin regulates nuclear import of CD44. J Biol Chem. 2010;285(40):30548–30557. doi: 10.1074/jbc.M109.075838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee JL, Wang MJ, Chen JY. Acetylation and activation of STAT3 mediated by nuclear translocation of CD44. J Cell Biol. 2009;185(6):949–957. doi: 10.1083/jcb.200812060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mandal A, Mandal S, Park MH. Genome-wide analyses and functional classification of proline repeat-rich proteins: potential role of eIF5A in eukaryotic evolution. PLoS One. 2014;9(11) doi: 10.1371/journal.pone.0111800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chung SS, Aroh C, Vadgama JV. Constitutive activation of STAT3 signaling regulates hTERT and promotes stem cell-like traits in human breast cancer cells. PLoS One. 2013;8(12) doi: 10.1371/journal.pone.0083971. [DOI] [PMC free article] [PubMed] [Google Scholar]