Abstract
Polysialic acid (PSA) is a large and highly negatively charged glycan that plays crucial roles in nervous system development and function in the adult. It has been suggested to facilitate cell migration, neurite outgrowth, and synaptic plasticity because its hydration volume could enhance flexibility of cell interactions. Evidence for receptors of PSA has so far been elusive. We now identified histone H1 as binding partner of PSA via a single-chain variable fragment antibody using an anti-idiotypic approach. Histone H1 directly binds to PSA as shown by ELISA. Surface biotinylation of cultured cerebellar neurons indicated an extracellular localization of histone H1. Immunostaining of live cerebellar neurons and Schwann cells confirmed that an extracellular pool of histone H1 colocalizes with PSA at the cell surface. Histone H1 was also detected in detergent-insoluble synaptosomal membrane subfractions and postsynaptic densities. When applied in vitro, histone H1 stimulated neuritogenesis, process formation and proliferation of Schwann cells, and migration of neural precursor cells via a PSA-dependent mechanism, further indicating that histone H1 is active extracellularly. These in vitro observations suggested an important functional role for the interaction between histone H1 and PSA not only for nervous system development but also for regeneration in the adult. Indeed, histone H1 improved functional recovery, axon regrowth, and precision of reinnervation of the motor branch in adult mice with femoral nerve injury. Our findings encourage investigations on the therapeutic potential of histone H1 in humans.
Introduction
Polysialic acid (PSA)—a long, linear polymer composed entirely of negatively charged sialic acid (5′-N-acetylneuraminic acid) in an unusual α2,8 linkage—belongs to a class of functionally important anionic glycans (for review, see Rutishauser, 2008). Because of its high negative charge, PSA is highly hydrated and has therefore been suggested to attenuate cellular interactions and increase cell motility (Mühlenhoff et al., 1998). In the mammalian brain, PSA is predominantly or probably exclusively attached to the protein backbone of the neural cell adhesion molecule (NCAM). So far, only neuropilin-2 has been identified in human dendritic cells as another carrier of PSA in mammals (Curreli et al., 2007). It remains to be shown, however, whether carriers other than NCAM express PSA in the brain.
PSA is associated with morphogenetic changes during developmental processes, such as cell migration, axonal growth, neuritic branching, and synaptogenesis, and persists in the adult brain mainly in structures that display a high degree of functional plasticity (Kleene and Schachner, 2004; Rutishauser, 2008). During embryonic development, PSA is expressed abundantly by Schwann cells and axons in the peripheral nervous system. After peripheral nerve injury in adult rodents, PSA is upregulated in axons, Schwann cells, and skeletal muscle fibers (Carratù et al., 1993; Olsen et al., 1995; Mehanna et al., 2009). In vitro and in vivo functional assays using PSA mimetics and/or the bacterial homolog of PSA, colominic acid, indicate that PSA enhances myelination and regeneration of peripheral nerves (Jungnickel et al., 2009; Mehanna et al., 2009) and promotes spinal cord regeneration of mice (Papastefanaki et al., 2007; Marino et al., 2009; Mehanna et al., 2010).
The search for PSA binding partners has been hampered by low affinity of this glycan with protein receptors. It has been suggested that PSA interacts with brain-derived neurotrophic factor (BDNF) (Muller et al., 2000), heparan sulfate proteoglycans (Storms and Rutishauser, 1998), AMPA receptors (Vaithianathan et al., 2004), NR2B subunit-containing NMDA receptors (Hammond et al., 2006), and the gonadal hormone estradiol (Garcia-Segura et al., 1995). However, evidence for a direct interaction has so far been only provided for BDNF (Kanato et al., 2008). Here, we identified histone H1 as a novel interaction partner of PSA using the unconventional anti-idiotype approach and showed direct binding of histone H1 to PSA.
Histone H1 was discovered as a classical nuclear protein that is involved as a linker histone in the organization of nucleosomes that mediate DNA packing (Doenecke et al., 1997). It is also present extranuclearly (Parseghian and Luhrs, 2006) and even extracellularly at the surface of several cell types (Watson et al., 1995; Brix et al., 1998; Henriquez et al., 2002).
Here, we show that histone H1 is present extracellularly at the cell surface of cerebellar neurons and Schwann cells and that extracellular histone H1 has a beneficial effect on neural precursor cell migration, neurite outgrowth, as well as Schwann cell proliferation and process elongation. Increased functional recovery and preferential motoneuron reinnervation after peripheral nerve injury elicited by histone H1 indicates its importance in regeneration in vivo.
Materials and Methods
Antibodies and reagents.
Rabbit polyclonal antibody against human histone H1 (Santa Cruz Biotechnology), mouse monoclonal antibody AE-4 against histone H1 (Millipore), horseradish peroxidase (HRP)-conjugated NeutrAvidin (Thermo Fisher Scientific), HNK-1 antibody 412 (Kruse et al., 1984), and mouse antibody G3G4 against 5-bromo-2′-deoxyuridine (BrdU) (Developmental Studies Hybridoma Bank) were used. Mouse monoclonal IgG2a antibody 735 against PSA (Frosch et al., 1985) and endoglycosidase N were kind gifts from Dr. Rita Gerardy-Schahn (Zentrum Biochemie, Zelluläre Chemie, Medizinische Hochschule, Hannover, Germany). Secondary antibodies were purchased from Dianova. Histone H1 (14-155), H2A (14-493), H2B (14-491), H3 (14-411), and H4 (14-412) were purchased from Millipore, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), colominic acid (average molecular weight: ∼12 kDa), chondroitin sulfate A (average molecular weight: ∼20 kDa) and C (average molecular weight: ∼63 kDa), and heparin (average molecular weight: ∼3 kDa) were from Sigma-Aldrich.
Propagation of phage library.
The “Griffin.1” phage scFv library (Griffiths et al., 1994) consisting of ∼106 individual synthetic human scFv antibodies cloned into the phagemid pHEN2 was a kind gift from Dr. Greg Winter (Medical Research Council Center for Protein Engineering, Cambridge, UK). The suppressor Escherichia coli strain TG1 (K12, supE, hsdD5/F′, thi, Δ(lac− pro), traD36, proA+B+, laclq, lacZΔM15) was infected with the Griffin.1 phage scFv library and grown in 2TY medium (16 g/L tryptone, 10 g/L yeast extract, 5 g/L NaCl) supplemented with 100 μg/ml ampicillin and 1% glucose. When reaching exponential growth (OD600 = 0.5), 2 × 1011 M13K07 helper phages (GE Healthcare) were applied to 25 ml cultures (corresponding to ∼1 × 1010 bacteria). After incubation for 30 min at 37°C, the infected bacteria were centrifuged for 10 min at 3300 × g, and the pellet was incubated in 2YT medium supplemented with 100 μg/ml ampicillin and 25 μg/ml kanamycin overnight at 30°C. The bacteria were pelleted at 10,800 × g for 10 min at 4°C, and 0.2 vol of polyethylene glycol (PEG)/NaCl (20% PEG6000 in 2.5 m NaCl) was added to the resulting supernatant containing the phages. After incubation on ice for 1 h, phages were pelleted by centrifugation at 10,800 × g for 30 min, and the resulting pellet was resuspended in 40 ml of H2O and 8 ml of PEG/NaCl and incubated for 20 min, all at 4°C. After centrifugation at 10,800 × g for 30 min, the phage pellet was resuspended in 5 ml of PBS (8.1 mm Na2HPO4, 1.74 mm NaH2PO4, 0.15 m NaCl, pH 7.5).
Selection of PSA-mimicking anti-idiotypic scFv antibodies.
The PSA IgG2a antibody, diluted in 4 ml of PBS (100 μg/ml), was incubated in immunotubes (Maxisorb; Nalgene Nunc International) overnight at 4°C. After three washes with PBS, the tubes were blocked with 2% skim milk (Frema Reform, DE-VAU-GE) in PBS (MPBS) for 2 h at room temperature. For preabsorption of unspecifically binding phages, an immunotube was blocked with 2% MPBS and incubated with 4 ml of PBS containing ∼4 × 1013 phages, 2 mg of mouse serum (Sigma-Aldrich) and 24 μg of mouse IgG2a (Cymbus Biotechnology) for 1 h at room temperature. The preincubated phage suspension was transferred to the immunotube coated with PSA antibody and incubated at room temperature for 30 min on a head-over-top rotator and 1.5 h without agitation. After incubation, the tube was washed 20 times with PBS containing 0.1% Tween 20, followed by 20 washes with PBS at room temperature. Bound phages were eluted at room temperature with 1 ml of 100 mm triethylamine and immediately neutralized with 0.5 ml of 1 m Tris/HCl, pH 7.4. The eluted phages were used to infect exponentially growing E. coli TG1 cells, and infected bacteria were grown overnight at 30°C on TYE plates (15 g/L agar, 10 g/L yeast extract, 8 g/L NaCl) containing 100 μg/ml ampicillin and 1% glucose. After four rounds of selection, individual clones were analyzed by phage ELISA.
Phage ELISA.
The binding specificity of phages that expressed potential PSA-mimicking anti-idiotypic scFv antibodies was tested by phage ELISA. Microtiter plates (Maxisorb; Nalgene Nunc International) were coated overnight at 4°C with 50 μl of purified PSA antibody in PBS (100 μg/ml) per well. All following steps were performed at room temperature. The plates were blocked with 2% MPBS. As negative controls, 50 μl of nonimmune mouse serum (500 μg/ml) and nonimmune IgG2a (25 μg/ml) were used. After blocking with 2% MPBS for 1 h at room temperature, plates were incubated with bacterial supernatants containing phages that expressed potential PSA-mimicking anti-idiotypic scFv antibodies. After six washes with PBS, bound phages were detected using a HRP-conjugated monoclonal antibody against M13 phage (GE Healthcare) and 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid) as substrate. Absorption at 405 nm was measured.
DNA sequencing and truncation of the scFv linker region.
The sequences of phage clones that expressed potential PSA-mimicking anti-idiotypic scFv antibodies were determined with the primers LMB3 (5′-CAG GAA ACA GCT ATG AC-3′) and fdSeq1 (5′-GAA TTT TCT GTA TGA GG-3′) using a model 377 DNA Sequencer from Applied Biosystems. For truncation of the linker region from 14 aa (SSGGGGSGGGGSGG) to 5 aa (SSGGG), pHEN2-derived plasmid DNA deriving from phages that expressed potential PSA-mimicking anti-idiotypic scFv antibodies were subjected to PCR using the primers 5′-CAG CCG GCC ATG GCC CAG GTG-3′ and 5′-GAG TCA CCG TGC ACT GCC TCC ACC ACT CGA GAC GGT GAC CAG GGT-3′. The PCR product and the pHEN2 phage-derived plasmids were cut with NcoI and ApaL1 and ligated.
Production, purification, and FPLC analysis of scFv antibodies.
To produce soluble scFv antibodies, the nonsuppressor strain HB2151 [K12, ara, thi/ F′, proA+B+, Δ(lac− pro), laclq, lacZΔM15] was infected with phages that encode PSA-mimicking anti-idiotype scFv antibodies. An amber stop codon in the pHEN2 sequence between the scFv gene and the M13 minor coat protein III gene allowed the expression of his- and myc-tagged scFv antibodies. Infected bacteria were grown at 37°C in LB medium containing 0.1% glucose and 100 μg/ml ampicillin. At OD600 0.8–0.9, production of scFv antibodies was induced by addition of isopropyl-β-d-thiogalactopyranoside at a final concentration of 1 mm. After an overnight incubation at 30°C, culture supernatants were concentrated by ultrafiltration and applied to Ni-NTA agarose beads (QIAGEN) that had been equilibrated with 50 mm phosphate buffer, pH 7.5, 300 mm NaCl, and 10 mm imidazole. After an overnight incubation at 4°C, the beads were centrifuged at 2500 × g for 5 min and washed with 50 mm phosphate buffer, pH 7.5, 300 mm NaCl, and 20 mm imidazole. The scFv antibodies were eluted by incubating beads with 50 mm phosphate buffer, pH 7.5, 300 mm NaCl, and 250 mm imidazole for 30 min at 4°C, and the eluted scFv antibodies were concentrated by centrifugation in Centricon-5 tubes (Millipore) and diluted in PBS.
To separate monomeric forms of scFv antibodies from dimeric or oligomeric forms, purified scFv antibodies were subjected to FPLC using a Superdex-75 column (diameter, 16 mm; length, 40 cm) and a BioCAD system (Applied Biosystems) at a flow rate of 1 ml/min and detection at 280 nm.
ELISA using scFv antibodies.
The specificity of the potential PSA-mimicking anti-idiotypic scFv antibodies was tested by competition ELISA in 96-well microtiter plates (Nalgene Nunc International) coated with 100 μl of PSA antibody (100 μg/ml PBS) overnight at 4°C. All following steps were performed at room temperature. After blocking the plates with 2% MPBS, purified scFv antibodies supplemented with 2% skimmed milk were added to the plates and incubated for 1.5 h. For competition, colominic acid, chondroitin sulfate A and C, or heparin were preincubated with the coated antibody before addition of scFv antibodies. After washing three times with 0.05% Tween 20 in PBS and three times with PBS, anti-myc tag antibody (Santa Cruz Biotechnology) was added and incubated for 1 h to detect binding of the myc-tagged scFv antibodies. The wells were washed again and incubated with HRP-conjugated secondary antibody for 1 h. Binding was detected using 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid) as substrate and measuring absorption at 405 nm.
Surface plasmon resonance.
As an alternative method to test the specificity of the potential PSA-mimicking anti-idiotypic scFv antibodies, surface plasmon resonance was performed. The PSA antibody was immobilized on a CM5 sensor chip (BIAcore). Purified anti-idiotypic antibodies in PBS were injected and sensorgrams were recorded using a BIAcore 3000 instrument. For competition, 5 μm scFv antibody was mixed with the competitor before injection.
Preparation of crude membrane fraction, synaptosomes, membrane-associated proteins, and postsynaptic densities.
Brains from postnatal (7 d of age) or adult (12–16 weeks of age) C57BL/6J mice were homogenized in homogenization buffer [0.32 m sucrose, 1 mm CaCl2, 1 mm MgCl2, 5 mm Tris/HCl, pH 7.5, and protease inhibitor (Roche)]. All steps were performed on ice or at 4°C. The homogenate was centrifuged for 10 min at 17,000 × g. The pellet was resuspended in homogenization buffer, applied to a 1.2, 1.0, and 0.8 m sucrose step gradient, and centrifuged for 2 h at 100,000 × g. The material from the 1.0/1.2 m sucrose interface was collected, diluted, and pelleted by centrifugation for 20 min at 17,000 × g. The pellet enriched in synaptosomal membranes was resuspended in PBS containing 1% Triton X-100 and incubated for 30 min. This fraction was taken as a detergent-solubilized synaptosomal membrane subfraction.
For the preparation of membrane-associated proteins, brains were homogenized in 4 ml of TNE buffer (25 mm Tris-HCl, pH 7.5, 150 mm NaCl, 5 mm EDTA). The homogenates were centrifuged at 1000 × g for 10 min, and the resulting supernatant was centrifuged at 100,000 × g for 30 min. The pellet was resuspended in 2 ml of TNE buffer and adjusted to 150 mm NaHCO3, pH 11.5. After incubation for 30 min, the sample was subjected to centrifugation at 100,000 × g for 30 min. The resulting supernatant was taken as a subfraction enriched in membrane-associated proteins.
Postsynaptic densities (PSDs) were prepared according to a described protocol (Cohen et al., 1977).
Affinity chromatography with PSA-mimicking anti-idiotypic scFv antibodies.
The fraction containing membrane-associated proteins was first run over a Sepharose 4B column (GE Healthcare) and then over a column with 3.2 mg of purified scFv antibodies immobilized on 2 ml of CNBr-activated Sepharose 4B (GE Healthcare) overnight at 4°C at a flow rate of 0.2 ml/min. After washing the column with 15 ml of PBS (0.3 ml/min), bound proteins were eluted with 200 mm glycine/500 mm NaCl, pH 2.7 (0.3 ml/min). The eluted fractions were collected and neutralized immediately with 1 m Tris/HCl, pH 8.8. After ultrafiltration using Centricon-5 tubes (Millipore), the proteins were analyzed by SDS-PAGE, silver staining, and mass spectrometry.
Western blot analysis, silver staining, mass spectrometry, and cell surface biotinylation.
Western blot analysis, silver staining, mass spectrometry, and cell surface biotinylation were performed as described in detail (Makhina et al., 2009).
ELISA with histone H1.
Binding of histone H1 to PSA was analyzed by ELISA. The 384-well flat-bottom microtiter plates (Corning) were coated in triplicate with 25 μl of histone H1 or GAPDH (10 μg/ml in PBS) or PBS alone at 4°C overnight. All following steps were performed at room temperature. After blocking with 1% BSA for 1 h, the wells were washed three times with PBS containing 0.05% Tween 20 (PBST). Wells were incubated with increasing concentrations of soluble colominic acid for 1 h. After washing five times with PBST and two times with PBS, 25 μl of PSA antibody (5 μg/ml) were added to each well. After incubation for 1 h, the plates were washed six times with PBST and two times with PBS. The wells were incubated with HRP-conjugated secondary mouse antibody for 1 h and washed five times with PBST and two times with PBS. Binding was visualized by incubating wells with 50 μl of 0.5 mg/ml o-phenylenediamine dihydrochloride (Thermo Fisher Scientific) for 2–5 min. The reaction was stopped by addition of 50 μl of 2.5 m H2SO4 and quantified using an ELISA reader at 490 nm and the software KCjunior (Bio-Tek).
Analysis of neurite outgrowth from cultured cerebellar neurons and analysis of process length and proliferation of cultured Schwann cells.
Primary cultures of cerebellar neurons or Schwann cells were prepared from cerebellum or dorsal root ganglia of 7-d-old C57BL/6J mice as described previously (Mehanna et al., 2009). In brief, 48-well plates were coated with 0.01% poly-l-lysine (PLL) overnight at 4°C, washed twice with water, air-dried, and coated overnight with histone H1 or laminin at a concentration of 10 μg/ml at 4°C. The plates were washed twice with PBS, and Schwann cells or cerebellar neurons were seeded at a density of 5 × 104 or 1 × 105 cells per well in 250 μl of corresponding serum-free culture medium. For antibody blocking experiments, PSA antibody or a rabbit polyclonal antibody against histone H1 were added to the wells at a concentration of 40 or 10 μg/ml 1 h after cells had been seeded. Similarly, soluble histone H1, soluble colominic acid, and/or endoglycosidase N were added to the culture at a concentration of 10, 10, or 4.8 μg/ml, respectively. After maintaining for 24 h at 37°C, cells were fixed with 2.5% glutaraldehyde and stained with 1% methylene blue/toluidine blue in 1% borax.
Morphological quantification of neurite or process lengths was done as described previously (Mehanna et al., 2009). Schwann cell processes and neurites of cerebellar neurons with a length of at least one cell body diameter were counted, and total neurite or process length per cell was determined by counting 50 cells in each of two wells per experiment. Schwann cell proliferation assay was performed as described previously (Mehanna et al., 2009), and ∼1000 cells for each experimental value were counted.
Immunostaining of live Schwann cells and cerebellar neurons.
Schwann cells or cerebellar neurons were seeded at a density of 2 × 105 on 12 mm glass coverslips pretreated with 0.01% PLL. After 20 h at 37°C, cells were washed three times with the corresponding serum-free culture medium containing 1% BSA and incubated with the antibodies against PSA and histone H1 for 20 min on ice. Cells were then washed gently with serum-containing medium and incubated with secondary antibodies coupled to fluorescent dyes Cy3 or Cy2 for 20 min at room temperature in the dark. After washing with serum-free culture medium, cells were fixed with 4% formaldehyde for 15 min at room temperature, washed three times with PBS, stained with 4′,6′-diamidino-2-phenylindole (DAPI) for 2 min, and finally washed three times with PBS, and then mounted with Fluoromount-G. Images were taken with an Olympus Fluoview 1000 microscope (Olympus).
Preparation, immunostaining, and cell migration of neural precursor cells.
Mouse embryos (embryonic day 13.5) were decapitated and ganglionic eminences were collected, dissected, transferred to serum-free media, and mechanically dissociated by pipetting up and down with three fire-polished Pasteur pipettes with sequentially smaller diameters (Dihné et al., 2003). After dissociation, the neurospheres were centrifuged at 200 × g for 1 min at room temperature, resuspended with 0.5 ml of Accutase (Invitrogen), and incubated at 37°C for 10 min. The 0.5 ml of neural stem cell medium was added and pipetted up and down for 15–20 times. After centrifugation at 200 × g for 5 min at room temperature, neurospheres were resuspended in neurosphere formation medium [DMEM/F12 (1:1) without l-glutamine, 1× B27 supplement, 0.02% sodium bicarbonate, 0.5 m HEPES, 16.6 mm glucose, 100 mm glutamine, 20 ng/ml epidermal growth factor (EGF) and FGF-2] and maintained in culture for 4–5 d. For immunostaining, cultured neurospheres were allowed to settle for 10 min, washed three times in precooled PBS, and fixed with 4% formaldehyde in filtered 0.4 m cacodylate buffer, pH 7.3, at 4°C for 1 h and dehydrated with 30% sucrose in cacodylate buffer at 4°C for 4 h. The neurospheres were embedded in tissue-tag, frozen at −30°C, and sectioned at 14 μm on a cryostat (Leica CM 3050; Leica Instruments). Sections were incubated with 10% goat serum and 0.1% Triton X-100 at room temperature for 1 h and then with antibodies against PSA and histone H1 overnight at 4°C. After washing three times with PBS, the sections were incubated with secondary antibodies coupled to the fluorescent dyes Cy3 or Cy2 for 1 h at room temperature in the dark, washed three times with PBS, incubated with bisbenzimide (Sigma-Aldrich) for staining of nuclei, and mounted with Fluoromount-G after additional washes in PBS. Images were taken with an Olympus Fluoview 1000 microscope.
To determine the migration of neural stem cells from neurospheres (Decker et al., 2000), neurospheres were kept in culture for 10–15 d. Nonadherent neurospheres were seeded in 10 μl droplets on glass coverslips coated with 0.02% PLL and laminin or histone H1 and maintained at 37°C for 20 min. The medium was adjusted to 1 ml per well with neurosphere formation medium without FGF-2 and EGF containing colominic acid and/or histone H1. After 24 h, migration was assessed by measurement of the distance from the edge of the neurosphere to all cells having migrated out of the neurosphere using an Axiovert microscope and the AxioVision image analysis system 4.6 (Zeiss).
Analysis of femoral nerve regeneration and motor function.
Femoral injury and analysis of regeneration were performed according to Simova et al. (2006). Analysis of femoral nerve regeneration and motor function was performed as described in detail previously (Mehanna et al., 2009), with the exception that the polyethylene tubes (3 mm length, 0.58 mm inner diameter; BD Biosciences) were filled either with PBS containing scaffold peptide that forms a gel matrix support (0.5% PuraMatrix Peptide Hydrogel, 3D; BD Biosciences), or PBS/scaffold peptide supplemented with histone H1 (100 μg/ml). Retrograde labeling and evaluation of motoneuron number and soma size and analysis of degree of remyelination was performed as described in detail by Mehanna et al. (2009). The degree of myelination was estimated by calculating the g ratio (axon-to-fiber diameter). Therefore, ultrathin sections of nerves were staining with toluidine blue after fixation, and the mean orthogonal diameters of the axon (inside the myelin sheath) and of the nerve fiber (including the myelin sheath) were measured.
Results
Identification of an anti-idiotypic scFv antibody mimicking polysialic acid
To identify novel PSA binding partners we used a PSA-mimicking anti-idiotypic scFv antibody for immunoaffinity chromatography with detergent-solubilized fractions of mouse brain tissue. First, we isolated scFv antibodies that bind to a PSA antibody by screening a phage scFv library for binding to the immobilized PSA antibody. After four rounds of selection, 10 clones were isolated. Four of these clones contained the sequence LNDGPVTSA; five clones, the sequence GLPIDS; and one clone, the sequence SPFES in the CDR3 region of the VH region of the scFv antibody. All clones consisted of a VH and VL chain deriving from the VH1 (DP-3) and VL1 (1-d) antibody gene family.
Next, we checked by ELISA and surface plasmon resonance whether binding of the anti-idiotypic scFv antibodies to the PSA antibody mimicked binding of PSA. Since the yield of scFv antibody from the clone with the sequence SPFES in the CDR3 region was extremely low, this clone was not used further. As analyzed by ELISA, binding of the scFv antibody with the sequence GLPIDS to substrate-coated PSA antibody was inhibited in a concentration-dependent manner by the bacterial homolog of PSA, colominic acid. The fact that competition for binding with colominic acid is not complete may be attributed to nonspecific binding of the scFv antibody of the antibody. Other negatively charged polymers such as chondroitin sulfate A, chondroitin sulfate C, or heparin showed no inhibition of the binding (Fig. 1A). In addition, the scFv antibody did not bind to the HNK-1 antibody, which was used as a negative control antibody (data not shown). As assayed by surface plasmon resonance, the scFv antibody with the sequence GLPIDS bound in a concentration-dependent manner to the immobilized PSA antibody (Fig. 1B). This binding was also inhibited in a concentration-dependent manner by colominic acid (Fig. 1C). The scFv antibody with the sequence LNDGPVTSA showed identical results in ELISA and surface plasmon resonance (data not shown). These results verify that the scFv antibodies with the sequence GLPIDS and LNDGPVTSA, which share the consensus motif P[VI][TD]S, are PSA-mimicking scFv antibodies.
Figure 1.

Anti-idiotypic scFv antibodies directed against a PSA antibody mimic PSA. A, Substrate-coated PSA antibody was preincubated with different concentrations of colominic acid (closed circles), chondroitin sulfate A (closed triangles), chondroitin sulfate C (open triangles), or heparin (closed rectangles) and incubated with purified scFv antibody with the sequence GLPIDS. Binding of the scFv antibody to the PSA antibody was determined by ELISA and binding in the presence of competitor was related to the binding in the absence of competitor, which was set to 100%. Mean values ± SD from three independent experiments are shown. B, C, Surface plasmon resonance experiments were performed using immobilized PSA antibody and different concentrations (0.01, 0.05, 0.1, 0.5, and 1 μm) of scFv antibody with the sequence GLPIDS for injection (B) or with a constant amount of scFv antibody (5 μm) and different concentrations of colominic acid (0, 1, 10, and 100 μm) for injection or without scFv and 100 μm colominic acid (100 μm/no scFv) (C). Binding of scFv was monitored by measuring the response signal in arbitrary units (RU). A representative experiment is shown.
To identify PSA binding partners, ELISA was performed using soluble purified scFv antibodies and different subfractions from mouse brain as substrate coats. No significant binding of scFv antibodies was observed (data not shown), possibly because of a low affinity of the scFv antibodies. We therefore increased the avidity and thus the functional affinity of the scFv antibodies. Since truncation of the linker region between the VH and VL domains of the scFv antibody results in enhanced dimerization and/or oligomerization of scFv monomers and thus in an increased avidity (Holliger et al., 1993), we truncated the linker region (Fig. 2A). FPLC analysis of the scFv antibodies without and with linker truncation showed that the nontruncated scFv antibody with the sequence GLPIDS existed predominantly as a monomer with low amounts of dimer, whereas the truncated scFv antibody existed only as a dimer (Fig. 2B). After linker truncation, the scFv with the sequence LNDGPVTSA also existed only as a dimer (data not shown). The scFv antibodies with truncated linkers were analyzed by ELISA and surface plasmon resonance for binding to substrate-coated PSA antibody. By ELISA, the linker truncated scFv antibody with the sequence GLPIDS showed a concentration-dependent and saturable binding (Fig. 2C). This binding was inhibited by colominic acid in a concentration-dependent manner, whereas heparin, chondroitin sulfate A, or chondroitin sulfate C did not inhibit (Fig. 2D). By surface plasmon resonance, binding of the linker truncated scFv antibody with the sequence GLPIDS to immobilized PSA antibody was inhibited by colominic acid in a concentration-dependent manner (Fig. 2E). The linker truncated scFv antibody with the sequence LNDGPVTSA showed identical results in ELISA and surface plasmon resonance (data not shown). Interpretation of sensorgrams by a classical 1:1 binding model did not lead to convincing results reflecting the intrinsic property of scFv to exist as a mixture of monomer and dimer. However, a plot of equilibrium response versus concentration allowed us to estimate a range for the apparent affinity of the scFv (0.5–5 μm) that is well in accordance with published data on the binding of colominic acid to the PSA antibody (Evans et al., 1995). The linker truncated scFv antibodies were then applied to substrate-coated brain subfractions. The linker truncated scFv antibody with the sequence GLPIDS strongly bound to soluble proteins and detergent-solubilized membrane proteins from brains of early postnatal and adult mice, whereas the linker truncated scFv antibody with the sequence LNDGPVTSA showed only weak binding to soluble brain proteins and no binding to membrane proteins (Fig. 3A).
Figure 2.
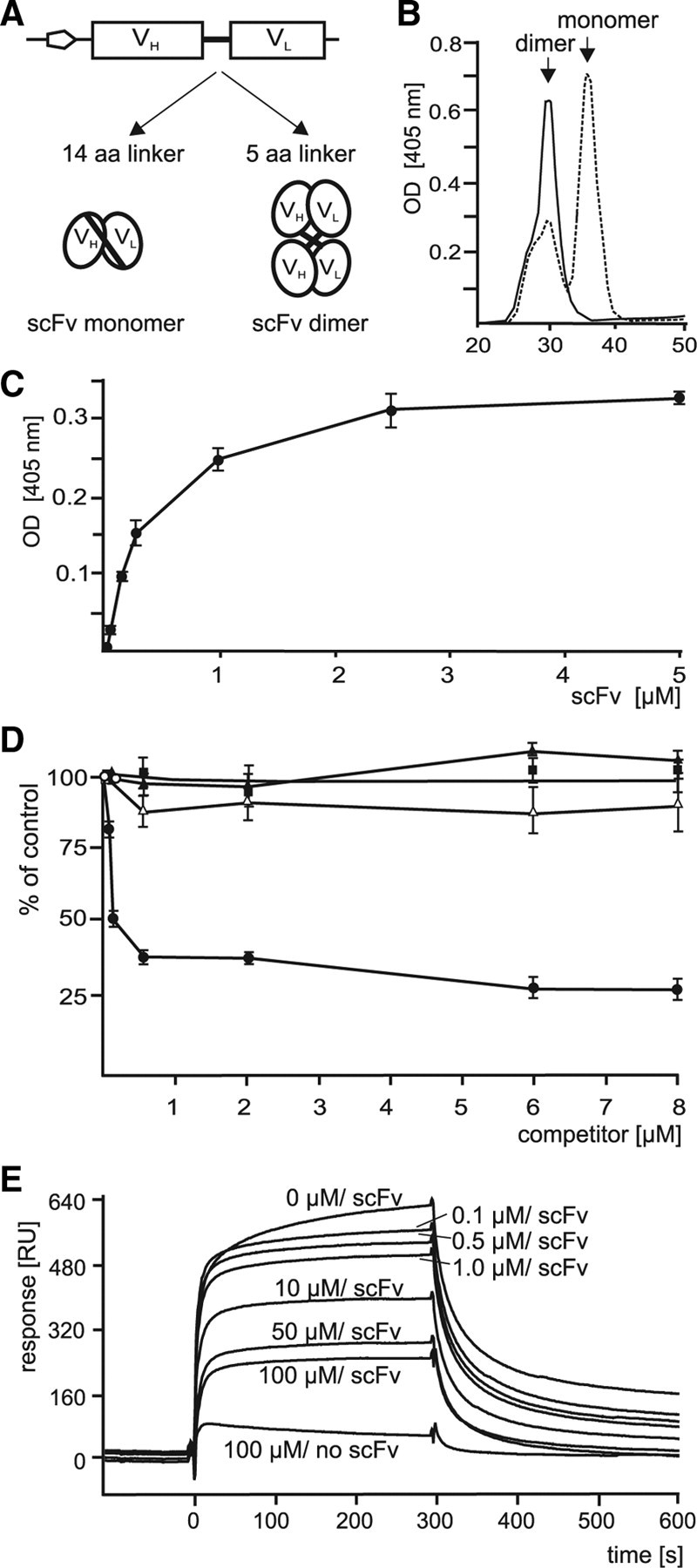
Affinity of PSA-mimicking scFv antibodies to a PSA antibody after linker truncation. A, The scFv antibody contains a linker of 14 aa (14 aa linker) between the VH and VL chains before truncation and 5 aa (5 aa linker) after truncation, which should result in increased formation of scFv antibody dimers. B, Purified scFv antibody with nontruncated (dashed line) or with truncated linker region (solid line) were separated by FPLC. The retention times of the monomeric and dimeric forms are indicated. C, Increasing concentrations of scFv antibody with truncated linker region were used in ELISA with substrate-coated PSA antibody. D, In the competition ELISA, different concentrations of colominic acid (closed circles), chondroitin sulfate A (closed triangles), chondroitin sulfate C (open triangles), or heparin (closed rectangles) and a constant amount of scFv antibody with truncated linker region were incubated with substrate-coated PSA antibody. C, D, Mean values ± SD from three independent experiments are shown for the binding of scFv antibody to the PSA antibody relative to the binding in the absence of competitor, which was set to 100%. E, Surface plasmon resonance experiments were performed using immobilized PSA antibody and different concentrations of colominic acid (0, 0.1, 0.5, 1.0, 10, 50, and 100 μm) together with a constant amount of scFv antibody (5 μm) with truncated linker region or without scFv and 100 μm colominic acid (100 μm/no scFv) for injection. Binding of scFv antibody in a representative experiment is shown. B–E, Results obtained for the scFv antibody with the sequence GLPIDS are shown.
Figure 3.
Identification of histone H1 as extracellular membrane-attached PSA binding partner using a PSA-mimicking scFv antibody for affinity chromatography. A, Detergent-solubilized membrane (memb) or soluble (sol) brain proteins isolated from 7-d-old (7 d) or 2-month-old (adult) mice were used as substrate coat and incubated without (control) or with either scFv antibody with the sequence GLPIDS or LNDGPVTSA and truncated linker region. Mean values ± SD from three independent experiments are shown for the binding of the scFv antibodies to soluble or membrane proteins. B, Purified scFv antibody with the sequence GLPIDS was immobilized and used for affinity chromatography with membrane-attached brain proteins obtained by alkaline treatment of membranes. Proteins binding to the scFv antibody were eluted and subjected to silver staining together with purified scFv antibody as a control. The position of the 30 kDa protein band that was analyzed by mass spectrometry and identified as histone H1 is indicated by an arrow. C, Histone H1, H2A, H2B, H3, and H4 as well as GAPDH as negative control were substrate-coated and incubated with increasing concentrations of colominic acid. The binding of colominic acid was detected by ELISA using PSA antibody and corresponding HRP-conjugated secondary antibodies. Mean values ± SD from three independent experiments performed in triplicate are shown. D, Triton X-100-soluble (Tx sol), Triton X-100-insoluble membrane proteins (Tx insol), and PSDs were subjected to Western blot analysis using histone H1-specific polyclonal rabbit antibody. E, Cultured live cerebellar neurons were incubated with biotinylation reagent. Biotinylated surface proteins (surface) were precipitated by streptavidin beads and subjected to Western blot analysis using a monoclonal histone H1 antibody, a calreticulin antibody, or HRP-conjugated streptavidin (SA). Total cell lysate (lysate) and post-pull-down supernatant (post-PD) were used as controls. The biotinylated histone H1 form with apparent molecular weights of ∼60 kDa (black arrowhead) and ∼30 kDa (gray arrowhead) are indicated.
Identification of histone H1 as synaptic membrane-associated PSA binding partner using a PSA-mimicking scFv antibody
The purified scFv antibody with the truncated linker and the sequence GLPIDS reacting with brain proteins was taken for affinity chromatography using different brain subfractions. By silver staining, a protein of ∼30 kDa was detected in an eluate deriving from a subfraction enriched in membrane-associated proteins that were released from the membranes by alkaline treatment (Fig. 3B). The ∼30 kDa band was analyzed by electrospray ionization–tandem mass spectrometry (MS/MS). After tryptic digestion of the band, two of the detected peptides could be assigned to histone H1. The MS/MS spectrum of a 1228.6936 Da and a 923.4724 Da precursor mass (detected as doubly charged ion at m/z = 614.8 and 554.2) matched the tryptic peptide TSGPPVSELITK and ALAAAGYDVEK of mouse histone H1, respectively. The result of the chromatography suggested that membrane-associated histone H1 binds to PSA.
To verify the direct interaction between histone H1 and PSA, an ELISA was performed with purified colominic acid using purified histone H1 from calf thymus as substrate and GAPDH as a negative control substrate. In parallel, we also analyzed whether other histones, namely H2A, H2B, H3, or H4, bound to PSA. PSA directly interacted with histone H1 in a concentration-dependent and saturable manner, whereas it did not interact with GAPDH or with any of the other histones (Fig. 3C).
To confirm that histone H1 is tightly associated with the plasma membrane, the distribution of histone H1 in brain membrane subfractions was analyzed on detergent treatment. By Western blot analysis, a histone H1-specific antibody reacted with a ∼30 kDa protein only in Triton X-100-insoluble postsynaptic densities or in the Triton X-100-insoluble, but not in the detergent-soluble fraction of the subfraction enriched in synaptic membranes (Fig. 3D), indicating that histone H1 is tightly associated with synaptic membranes in detergent-insoluble membrane microdomains such as PSDs.
Since PSA is exposed at the cell surface, we investigated whether histone H1 is present extracellularly. To test this, live cerebellar neurons in culture were cell surface biotinylated and biotinylated proteins were isolated by streptavidin beads. In Western blot analysis with a histone H1 antibody, histone H1 was mainly detectable as a protein with an apparent molecular weight of ∼60 kDa in the eluate containing biotinylated proteins, whereas only minor amounts of a ∼30 kDa protein were detectable in the eluate (Fig. 3E). Histone H1 was detectable only as a ∼30 kDa protein in the cell lysate and in the post-pull-down supernatant containing nonbiotinylated proteins (Fig. 3E). Probing the Western blots with HRP-conjugated streptavidin showed biotinylated proteins only in the eluate (Fig. 3E) demonstrating that the cell surface biotinylation and the isolation of biotinylated proteins had been successful. The intracellular protein calreticulin was detectable by Western blot analysis in cell lysate and post-pull-down supernatant, but not in the eluate of biotinylated proteins (Fig. 3E), indicating the intactness of the cells during cell surface biotinylation and serving as a control. The results of the cell surface biotinylation thus indicate that histone H1 indeed is present at the cell surface as extracellular protein. In addition, because of the molecular weights of the cell surface-biotinylated histone H1, which is mainly ∼60 kDa, and the intracellular nonbiotinylated histone H1, which is ∼30 kDa, we assume that extracellular histone H1 mainly exists as a dimer.
To further corroborate the notion that histone H1 interacts with PSA at the cell surface, we immunocytochemically colabeled histone H1 and PSA at the surface of cultured live cerebellar neurons and Schwann cells using a polyclonal histone H1 antibody and a monoclonal PSA antibody. Confocal microscopic analysis showed histone H1 and PSA immunoreactivity both at the surface of neuronal cell bodies and along neurites (Fig. 4A) as well as processes of Schwann cells (Fig. 4B). The superimposed images revealed a significant portion of histone H1 colocalizing with PSA (Fig. 4A,B).
Figure 4.

Colocalization of histone H1 and PSA at the surface of live cerebellar neurons and Schwann cells. A, Cultured live cerebellar neurons (A) or Schwann cells (B) were immunostained with rabbit polyclonal histone H1 antibody, mouse monoclonal PSA antibody, and the corresponding fluorescent dye-labeled secondary antibodies. After fixation and permeabilization of cells, nuclei were stained with DAPI. Superimpositions of histone H1 and PSA staining show partial colocalization (seen in yellow) at the surface of cell bodies and processes. Scale bars, 10 μm.
Histone H1 alters PSA-induced neurite outgrowth of cerebellar neurons
Next, we investigated whether the interaction between PSA and histone H1 has functional consequences. First, we tested whether histone H1 influences neurite outgrowth. Cerebellar neurons were grown on substrate-coated PLL or PLL with histone H1 or as control with laminin or histone H2A, H2B, H3, and H4. In parallel, neurons were grown on PLL in the absence or presence of soluble histone H1 and/or soluble PSA or as control in the presence of histone H2A, H2B, H3, or H4. Neurite outgrowth on substrate-coated histone H1 was enhanced by approximately threefold when compared with PLL and nearly reached values obtained for the positive control substrate laminin (Fig. 5A,B). Similarly, neurite outgrowth on PLL was increased with soluble histone H1 and soluble PSA (Fig. 5A,C), indicating that soluble PSA and histone H1 both trigger neurite outgrowth. Moreover, a concentration-dependent promotion of neurite outgrowth by both substrate-coated and soluble histone H1 was observed (Fig. 5C). However, in the concomitant presence of soluble histone H1 and PSA, neurite outgrowth was not increased and was similar to that observed in the absence of both histone H1 and PSA (Fig. 5A,C), indicating that soluble histone H1 and PSA had interacted and were no longer able to trigger neurite outgrowth. These results indicate that the interaction between histone H1 and PSA is involved in neurite outgrowth. Unexpectedly, substrate-coated and soluble histone H2A, H2B, and H3 also promoted neurite outgrowth (Fig. 5B,C), whereas H4 had no effect on neurite outgrowth as coated (Fig. 5B) or soluble (data not shown) substrate. To analyze whether neurite outgrowth induced by histone H1 or the other histones depends on PSA at the surface of cerebellar neurons, we measured neurite lengths of neurons treated with the PSA degrading endoglycosidase N or with the PSA-specific antibody. On the control substrates PLL or laminin, neurite outgrowth in the presence of PSA antibody or endoglycosidase N was not altered compared with that observed in the absence of antibody and enzyme (Fig. 5D). In addition, the enhanced neurite outgrowth in the presence of soluble histone H2A, H2B, or H3 was also not inhibited either by the PSA-specific antibody or by the endoglycosidase N treatment (Fig. 5D), indicating an PSA-independent enhancement of neurite outgrowth by these histones. In contrast, the enhanced neurite outgrowth in the presence of soluble histone H1 was reduced nearly to that observed on PLL in the presence of either the PSA-specific antibody or the PSA-degrading enzyme (Fig. 5D). Thus, PSA at the surface of neurons mediates histone H1-induced neurite outgrowth.
Figure 5.
Histone H1 promotes neurite outgrowth of cerebellar neurons in a PSA-dependent manner. Primary cultures of cerebellar neurons were grown on substrate-coated PLL or PLL with 10 μg/ml histone H1, H2A, H2B, H3, or H4, or laminin (lam), on PLL with increasing amounts of histone H1 or on PLL in the presence of increasing amounts of soluble histone H1 or on PLL in the presence of 10 μg/ml soluble PSA and histone H1 (A, C), on PLL or laminin in the absence and presence of PSA-specific antibody (735) or endoglycosidase N (endo) and/or histone H1, H2A, H2B, H3, or H4 (D), or on substrate-coated PLL or laminin in the absence or presence of soluble PSA and/or histone H1 antibody (αH1) (E). A, Images of representative neuron on substrate-coated PLL in the absence or presence of PSA and/or soluble histone H1, on substrate-coated histone H1 or on control substrate laminin are shown. B–E, The total length of neurites per cerebellar neuron was determined. The asterisks, double asterisks, and triple asterisks denote p < 0.05, p < 0.01, and p < 0.001 obtained by the two-tailed t test, respectively, from n = ∼100 neurons in three independent experiments.
To investigate whether endogenous extracellular histone H1 is involved in PSA-mediated neurite outgrowth, neurite outgrowth was determined in the absence or presence of soluble PSA and/or a polyclonal histone H1 antibody with the expectation that the antibody binds to endogenous histone H1 and blocks its effect on neurite outgrowth. Addition of soluble histone H1 antibody indeed blocked neurite outgrowth promotion by soluble PSA, whereas the antibody had no significant effect on neurite outgrowth in the absence of PSA (Fig. 5E). Surprisingly, laminin-induced neurite outgrowth was also reduced in the presence of the histone H1 antibody independently of the presence of PSA (Fig. 5E). Since neurite outgrowth enhanced by laminin was not affected in the presence of the PSA-specific antibody or endoglycosidase N (Fig. 5C), it is likely that laminin stimulates neurite outgrowth in a histone H1-dependent, but PSA-independent manner.
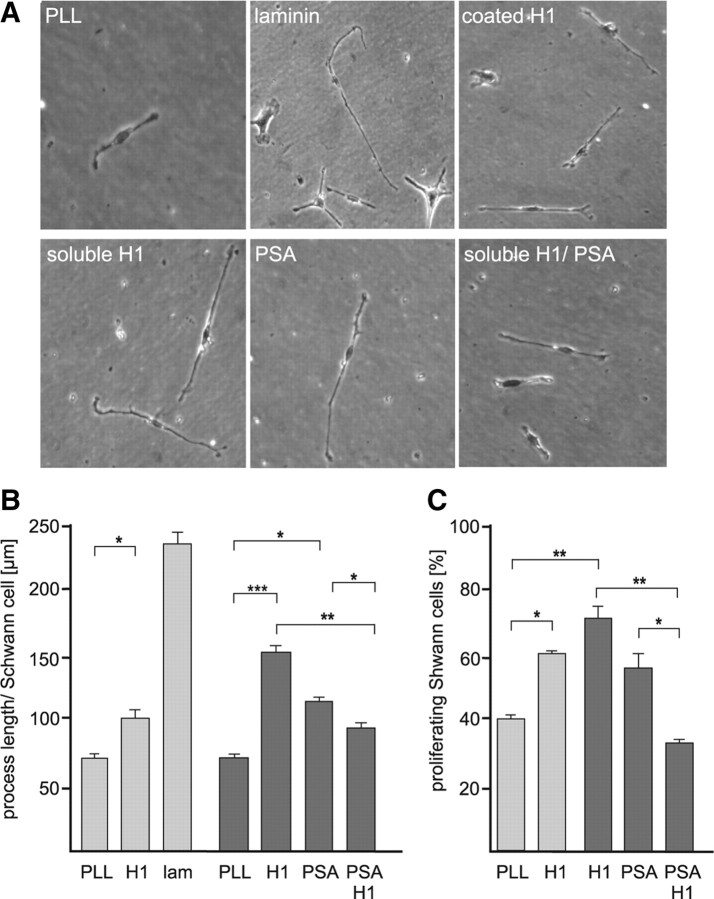
Histone H1 and PSA influence process formation and proliferation of Schwann cells
Since we showed that histone H1 and PSA colocalize at the Schwann cell surface and since a PSA-mimicking peptide and overexpression of PSA by Schwann cells improve myelination by Schwann cells in vivo (Papastefanaki et al., 2007; Mehanna et al., 2009), we tested the effects of histone H1 on Schwann cell process formation. Schwann cells isolated from dorsal root ganglia of early postnatal mice were cultured on substrate-coated PLL, laminin, histone H1, or PLL in the presence of soluble histone H1 protein and/or soluble PSA. Morphometric analysis of Schwann cell process length revealed an increase of process length on substrate-coated histone H1 relative to PLL substrate and also in the presence of soluble PSA or soluble histone H1 (Fig. 6A,B). The concomitant presence of soluble PSA and histone H1 led to a neutralization of the promotion seen with PSA and histone H1 when applied individually (Fig. 6A,B), indicating that the exogenously added histone H1 and PSA likely bound to each other and were no longer able to promote process extension.
Figure 6.
Histone H1 enhances process elongation and proliferation of Schwann cells in a PSA-dependent manner. Schwann cells were grown on substrate-coated PLL, histone H1 (H1), or laminin (lam) or on substrate-coated PLL in the presence of soluble PSA and/or histone H1 (A, B). A, Images of representative Schwann cells on substrate-coated PLL in the absence or presence of PSA and/or soluble histone H1, on substrate-coated histone H1 or laminin are shown. The process length per Schwann cell (B) and the number of BrdU-labeled proliferating Schwann cells (C) were determined. B, C, The asterisks, double asterisks, and triple asterisks denote p < 0.05, p < 0.01, and p < 0.001 obtained by the two-tailed t test, respectively, from n = ∼100 (B) or n = ∼1000 (C) Schwann cells in three independent experiments.
We also investigated the effect of histone H1 and PSA on Schwann cell proliferation by analysis of BrdU incorporation and found that Schwann cell proliferation was enhanced by substrate-coated histone H1 and in the presence of soluble histone H1 and soluble PSA, when compared with proliferation in the absence of PSA and histone H1, respectively (Fig. 6C). The mixture of soluble histone H1 and soluble PSA decreased the enhanced proliferation seen with histone H1 or PSA to levels observed on the PLL substrate (Fig. 6C). These results show that PSA and histone H1 are involved in proliferation and process formation of Schwann cells.
Histone H1 and PSA alter migration of neural progenitor cells
Previous studies have suggested that PSA is required for migration of neural progenitor cells (Hu et al., 1996; Chazal et al., 2000; Hu, 2000; Patrzykat et al., 2001; Angata et al., 2007). Based on these studies, we next investigated whether histone H1 affects neural progenitor cell migration. Beforehand, we investigated by immunostaining whether neural progenitor cells in neurospheres express histone H1 and PSA and observed that these cells not only express both histone H1 and PSA at their cell surface but also that both protein partially colocalize (Fig. 7A). For analysis of migration, the neurospheres were maintained on substrate-coated histone H1 and in the presence of soluble histone H1 or soluble PSA. The number of cells migrating from the neurospheres and the distance between migrating cells and the edge of the neurospheres were determined and used as criteria for migration. On PLL, the number of cells that had migrated out of the neurospheres was low (Fig. 7B). Migration of neural progenitor cells was enhanced when either soluble PSA or soluble histone H1 were applied, whereas a mixture of PSA and histone H1 only slightly affected migration (Fig. 7B). Compared with the positive control substrate laminin, substrate-coated histone H1 enhanced migration almost equally well (Fig. 7B). These findings suggest that both histone H1 and PSA are important for neural progenitor cell migration in vitro.
Figure 7.

Histone H1 and PSA are expressed by neurospheres and affect migration of neural progenitor cells. A, Slices of neurospheres were stained with histone H1- and PSA-specific antibodies followed by DAPI staining. Superimpositions of histone H1 and PSA staining show partial colocalization (seen in yellow) at the surface of neural precursor cells. Scale bar, 10 μm. B, Neurospheres were seeded onto substrate-coated PLL (PLL), histone H1 (H1), or laminin (lam) or on substrate-coated PLL in the presence of soluble PSA and/or histone H1. Micrographs displaying neurospheres (borders are indicated by dashed lines) with cells migrating out of the neurospheres in the absence (PLL) or presence of soluble PSA (PSA), soluble histone H1 (H1), or both soluble PSA and soluble histone H1 (PSA/H1) (left panel) and the quantification of the total distance reached by all migrating cells measured from the border of the neurosphere (right panel) are shown. Mean values ± SD from three independent experiments are shown. The asterisks and double asterisks denote p < 0.05 and p < 0.01, respectively, obtained by the two-tailed t test from 20 neurospheres per group from three independent experiments.
Histone H1 promotes functional recovery after femoral nerve injury in adult mice
Since PSA enhances regeneration after injury of the peripheral nervous system of adult mice (Mehanna et al., 2009) and since we have observed that histone H1 has beneficial effects on Schwann cells and neurons in vitro, we addressed the question whether histone H1 enhances regeneration after femoral nerve injury. The function of the quadriceps muscle was evaluated during a 4 month period after injury after application of histone H1 or PBS. Femoral nerve injury in mice induces changes in gait that can be quantitatively evaluated, for instance by the foot–base angle (FBA) (Fig. 8A–D,I) and the heels–tail angle (HTA) (Fig. 8E–H,J) during beam walking (Fig. 8A,H). Changes in gait are caused by impaired extensor function of the quadriceps muscle leading to abnormal external rotation of the ankle and high heel position at defined gait cycle phases. We used these parameters to evaluate the effect of histone H1 on locomotor recovery. One week after injury, the degree of functional impairment, as evaluated by the increase of the foot–base angle (Fig. 8B) and decrease of the heels–tail angle (Fig. 8F) compared with the preoperative values (Fig. 8A,E) was found to be similar between groups of mice treated with histone H1 and PBS. When compared with the PBS control groups (Fig. 8C,G,I,J), better functional improvement was observed in histone H1-treated mice at 2 (Fig. 8I,J), 3 (Fig. 8I,J), and 4 (Fig. 8D,H–J) months after injury. Analysis of the protraction length ratio, which is another parameter for assessment of functional recovery measuring the ability of a mouse to extend the hindlimb to catch an object with its hindpaws, was not different between the two groups. However, there was a tendency for better recovery of hindlimb extension after application of histone H1 compared with PBS (Fig. 8K). Superior functional recovery after histone H1 treatment was apparent when the stance recovery index was calculated for both angles on an individual animal basis (Fig. 8L). Stance recovery index is a measure of the individual degree of recovery and reflects the degree of postoperative normalization of the quadriceps extensor function during ground locomotion. The recovery index of animals treated with histone H1 reached values at 3 and 4 months that were remarkably the same as the preoperative values, which are set to 100%, indicating complete functional recovery. At the same time, recovery in the control group after 3 and 4 months was only 62 and 76%, respectively. Thus, application of histone H1 treatment leads to a most superior functional outcome in peripheral nerve regeneration.
Figure 8.
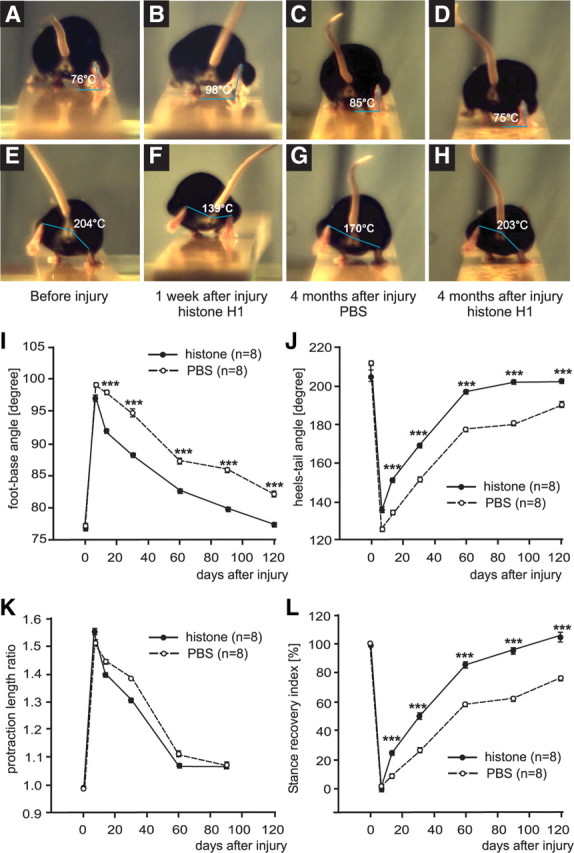
Analysis of motor function after femoral nerve injury and application of histone H1. FBA (A–D) and HTA (E–H) were determined by analyzing single video frames from recordings of beam walking of mice before injury (A, E), 1 week (B, F) and 4 months (C, D, G, H) after injury and application of histone H1 (B, D, F, H) or PBS as vehicle solution (C, G) to the lesion site. I–L, Time course of motor recovery after application of histone H1 or PBS are shown as mean values ± SEM of foot–base angle (I) and heels–tail angle (J), protraction length ratio (K), and stance recovery index (L) at different time points after injury and application of histone H1 or PBS. Preinjury values are plotted for day 0 and for other time points indicated in the graphs. Numbers of animals studied per group are indicated in brackets. The asterisks indicate significant differences (p < 0.05, one-way ANOVA with Tukey's post hoc test) between the histone H1-treated group and the PBS control group at the given time points.
Histone H1 promotes survival, extent, and precision of axonal regrowth, and reinnervation of motoneurons
To search for structural correlates of the superior functional recovery after application of histone H1, retrograde labeling of regenerated motoneurons was performed in animals after analyzing functional recovery. The number of motoneurons backlabeled through the motor (quadriceps) nerve branch that represent correctly projected regrown motoneurons was remarkably increased in animals to which histone H1 had been applied relative to PBS-treated animals (Fig. 9A). The number of backlabeled motoneurons from the sensory (saphenous) branch representing incorrectly projecting neurons was considerably decreased in animals treated with histone H1 compared with PBS-treated animals (Fig. 9A). The small numbers of motoneurons backlabeled through both femoral nerve branches were similar in histone H1- or PBS-treated mice (Fig. 9A). Although not reaching the level of statistical significance, a noticeable increase in the total number of labeled neurons was observed in histone H1-treated mice relative to PBS-treated mice (Fig. 9A). Since numbers of retrogradely labeled motoneurons reflect the extent of motoneuron survival (de la Cruz et al., 1994; Waters et al., 1998; Asahara et al., 1999, Guseva et al., 2009), we conclude that treatment with histone H1 reduces motoneuron death, a characteristic feature of the femoral nerve injury paradigm in mice (Simova et al., 2006). In addition, histone H1 application increased the number of regenerated motoneurons projecting to the appropriate motor nerve branch and thus increased preferential motor reinnervation, which is a characteristic feature of femoral nerve regeneration (Brushart, 1988; Al-Majed et al., 2000; Franz et al., 2005; Vrbova et al., 2009). In conclusion, our results show that better functional recovery in histone H1-treated mice is attributable to better motoneuron regeneration, since extent and precision of motor reinnervation was improved compared with control mice.
Figure 9.
Analysis of regrowth of motoneuron axons after femoral nerve injury. Four months after injury and application of histone H1 or vehicle (PBS), animals were subjected to retrograde labeling of motoneurons. A, Mean numbers + SEM of motoneurons labeled through the motor branch representing correctly projecting neurons (correct), through the sensory branch representing incorrectly projecting neurons (incorrect), through both branches (mixed), and the sum of labeled neurons (total number) are shown. B, Morphometric analysis of soma size of regenerated motoneurons was performed. Mean values + SEM of soma area of correctly and incorrectly projecting motoneurons after application of histone H1 or PBS are shown. A, B, Significant differences between the groups of eight animals are indicated by asterisks (*p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Tukey's post hoc test). C, Analysis of myelinated nerve fibers in regenerated and intact nerves. Shown are normalized frequency distributions of g ratios (axon/fiber diameter) in the motor nerve branch of noninjured mice (intact) and mice treated with histone H1 (histone) or vehicle (PBS) 4 months after injury.
Since a significant correlation between recovery index and motoneuron size was found 3 months after femoral nerve injury (Simova et al., 2006; Ahlborn et al., 2007), we measured soma size of motoneurons regrowing their axons correctly and found a significant increase in soma size of these motoneurons in histone H1- versus PBS-treated animals. No difference in soma size of incorrectly projecting motoneurons was observed among the groups (Fig. 9B). Thus, better functional recovery of histone H1-treated mice can be attributed to larger motoneuron size, which is an indicator of a better functional state of regenerated motoneurons (Simova et al., 2006; Ahlborn et al., 2007).
We also analyzed regenerated nerves with regard to the degree of remyelination as evaluated by the g ratio (axon- to fiber-diameter ratio). The frequency distributions of the g ratio for axons of histone H1- or PBS-treated injured nerves were similar and shifted to higher values when compared with values for intact axons (Fig. 9C), indicating that histone H1 does not enhance remyelination or myelin thickness of injured nerves compared with those that were treated with PBS only.
Discussion
In the present study, we isolated PSA-mimicking anti-idiotypic scFv antibodies by screening a scFv phage library with a PSA antibody and identified histone H1 as a novel interaction partner of PSA using the PSA-mimicking scFv antibody with the sequence GLPIDS in the CDR3 region of its VH chain for immunoaffinity chromatography of membrane-associated brain proteins.
ELISA experiments showed that histone H1 directly interacts with PSA, whereas other histones, namely histone H2A, H2B, H3, and H4, do not bind to PSA. This uniqueness of histone H1 to interact with PSA is further underscored by neurite outgrowth experiments showing that only histone H1 promotes neurite outgrowth in a PSA-dependent manner, whereas neurite outgrowth induced by the other histones or by laminin is PSA-independent. Interestingly, laminin-induced neurite outgrowth appears to depend on histone H1 since histone H1 antibody inhibits the laminin-induced neurite outgrowth. However, data from ELISA (data not shown) indicated that laminin does not directly bind to histone H1. The mechanism by which histone H1 and the other histones trigger PSA-dependent or PSA-independent neuronal responses such as neurite outgrowth are not yet known and will require additional investigations.
Immunostaining of live cerebellar neurons, Schwann cells, and neural progenitor cells indicated that extracellular histone H1 and PSA colocalize. Cell surface biotinylation of cerebellar neurons confirmed the extracellular localization of histone H1. Furthermore, the results of the in vitro functional tests (e.g., neurite outgrowth from cerebellar neurons, Schwann cell process formation and proliferation, as well as migration of neural progenitor cells) indicated that extracellular histone H1 interacts with PSA at the cell surface and that this interaction induces the observed effects of histone H1 on the investigated neural cells. Indications for the unusual extracellular location of histone H1, which was first identified as a nuclear protein, have been found in several other studies: histone H1 has been observed at the cell surface of cultured mouse cortical neurons (Bolton and Perry, 1997), human monocytes (Holers and Kotzin, 1985), activated human peripheral blood lymphocytes (Watson et al., 1995), cultured T-cells (Watson et al., 1995, 1999), a macrophage cell line (Brix et al.,1998), and skeletal muscle cells (Henriquez et al., 2002). So far, the mechanism(s) by which histone H1 crosses the membrane are not known. Since penetration of extracellular histones through the plasma membrane into the cell has been demonstrated (Hariton-Gazal et al., 2003), it is conceivable that a similar mechanism may exist for the export of histone H1.
Extracellular histone H1 has been shown to bind to cell surface heparan sulfate proteoglycans (Watson et al., 1999) and lipopolysaccharide (Hampton et al., 1988; Bolton and Perry, 1997) and apparently binds to amyloid precursor protein (Potempska et al., 1993) and associates with β-amyloid in amyloid plaques (Duce et al., 2006). We identified PSA as another novel binding partner of extracellular histone H1 and showed that the effects of histone H1 on neural cells, such as neurite outgrowth, are mediated by PSA. For instance, application of the PSA antibody or enzymatic degradation of PSA abolished the enhanced neurite outgrowth from cerebellar neurons. It is worthwhile to mention that, like histone H1, PSA also binds to heparan sulfate proteoglycans (Storms and Rutishauser 1998) and that the heparan sulfate-dependent interaction between heparan sulfate proteoglycans, in particular perlecan, and extracellular histone H1 plays an important role in skeletal muscle development and regeneration (Henriquez et al., 2002).
In our study, in vivo experiments showed that histone H1 improves regeneration of injured femoral nerve of mice. Administration of histone H1 to lesioned femoral nerves promotes survival of motoneurons, axonal regrowth, precision of reinnervation, and better regeneration of the injured femoral nerve, leading to a remarkable complete functional recovery of locomotion after injury. We have observed similar positive effects on recovery after femoral nerve lesion in mice for PSA using PSA-mimicking peptides (Mehanna et al., 2009). The time course of improvement is similar for histone H1 and PSA-mimicking peptides with positive effects becoming apparent later (i.e., at 2–3 months after injury and treatment). Since the local availability of a protein is most likely limited to hours or at best days after application because of its degradation, the question arises how a single application of histone H1 to the lesion site produces this late-appearing and long-lasting conducive effect of this protein. We propose that histone H1 modulates early cellular and molecular responses to injury such that subsequent regeneration processes are positively influenced over weeks. Interaction of histone H1 with its cell surface receptors (e.g., PSA-NCAM) may trigger signal transduction events that alter gene expression in Schwann cells and/or neurons resulting, for example, in inhibition of the expression of factors involved in neurodegeneration and/or in induction or enhancement of the expression of factors, such as cell surface receptors, growth factors, or metabolic enzymes that generate long-lasting beneficial and positive effects on neuronal survival and neurite regrowth and thus on functional recovery and regeneration. Since histone H1 can penetrate through the plasma membrane (Hariton-Gazal et al., 2003), it is conceivable that extracellular histone H1 directly affects gene regulation after translocation into the cell and import into the nucleus. It is also conceivable that PSA may facilitate or enhance the translocation of histone H1 through the plasma membrane. Based on our in vitro data of enhanced proliferation and process elongation of Schwann cells, we propose that histone H1 stimulates an early response of these cells to injury resulting in priming the injured femoral nerve at the lesion site to regenerate. Recently, it has been shown that the communication between Schwann cells and regrowing motoneurons plays a crucial in regeneration (Vrbova et al., 2009). Since neurite outgrowth in vitro was promoted by histone H1, we consider it likely that regeneration of the injured nerve elicited by histone H1 is, at least in part, because of enhanced axonal regrowth in vivo. Indeed, retrograde labeling of motoneurons demonstrated enhanced axonal regrowth and precision of appropriate reinnervation of the correct branch of the femoral nerve when compared with controls. It is interesting, in this respect, that brief low-frequency electrical stimulation of the injured femoral nerve, which also leads to long-lasting enhancement of axonal regrowth (Al-Majed et al., 2000; Brushart et al., 2005) and functional recovery (Ahlborn et al., 2007), is associated with an upregulation in BDNF expression, which is a PSA binding partner (Kanato et al., 2008) that promotes neuronal survival and differentiation during development (for instance, see Sofroniew et al., 2001) and that is required for myelination and regeneration of injured sciatic nerve (Zhang et al., 2000).
The diverse and, importantly, long-lasting effects of the histone H1 treatment suggest its involvement in basic signaling mechanisms that are most likely PSA-dependent, but may be also PSA-independent. PSA, which is N-glycosidically linked to NCAM, acts as a positive modulator of NCAM functions by inhibiting the cis-interactions of NCAM and thus making more NCAM available for trans-interactions (Doherty et al., 1990; Lüthl et al., 1994; Doherty and Walsh, 1996; Muller et al., 1996) with, for instance, L1 and proteoglycans. It has been proposed that the PSA-mimicking peptide, which improves regeneration and myelination of injured femoral nerve but influences neither motoneuron regrowth nor preferential reinnervation (Mehanna et al., 2009), interferes with inhibitory interactions between PSA-NCAM and its receptor(s) and thereby could affect signaling pathways and lead to the observed in vivo effects. Fibroblast growth factor receptors (FGFRs), which are involved in NCAM signaling (Kiselyov et al., 2003), are likely mediators of PSA-induced signal pathways that affect myelination, since FGFRs are expressed on Schwann cells (Grothe et al., 2001) and are involved in myelination (Ogata et al., 2004). Furthermore, levels of FGFRs are increased in Schwann cells after sciatic nerve injury (Grothe et al., 2001) and mice deficient in FGFR-3 showed reduced myelin thickness compared with wild-type controls (Jungnickel et al., 2004). Since myelination was not improved by application of histone H1, whereas preferential reinnervation and axonal regrowth was enhanced, we assume that binding of histone H1 to PSA-NCAM activates other signal pathways than the PSA-mimicking peptide. These yet-unknown PSA-dependent or PSA-independent signal transduction pathways or mechanisms could trigger axonal regrowth and correct reinnervation, but not remyelination. However, the interaction of histone H1 with PSA binding partners, such as heparan sulfate proteoglycans, which promote axonal growth and synaptic plasticity in a PSA-dependent manner (Storms and Rutishauser, 1998; Dityatev et al., 2004), may modulate the interaction between PSA and other binding molecules, such as BDNF and, thus, may influence PSA-induced signal transduction pathways.
The functions of histone H1 on different neural cell types and its interaction with different binding partners show that it plays a central role in modulating and integrating various cellular functions associated with cell survival, proliferation, and regeneration in a PSA-dependent or possibly PSA-independent manner. Our results reveal that histone H1 has beneficial effects in regeneration in the mouse peripheral nervous system and indicate a promising therapeutic potential for treatment of peripheral nerve injuries in humans.
Footnotes
M.S. is supported by the New Jersey Commission for Spinal Cord Research and is a consultant at the Center for Neuroscience at Shantou University Medical College, China. We are grateful to Drs. Alexandre Podtelejnikov and Matthias Mann (Protein Bioinformatics, Odense, Denmark) for protein sequencing, Dr. Ali Mehanna for help with the surgery, Emanuela Szpotowicz for excellent technical assistance, Dr. Nina Kurschat for the PSA-mimicking peptide, and Dr. Rita Gerardy-Schahn (Medizinische Hochschule Hannover, Hannover, Germany) for the PSA antibody and endoglycosidase N. The University Medical Center Hamburg–Eppendorf has filed a patent application for certain technology described in this paper.
References
- Ahlborn P, Schachner M, Irintchev A. One hour electrical stimulation accelerates functional recovery after femoral nerve repair. Exp Neurol. 2007;208:137–144. doi: 10.1016/j.expneurol.2007.08.005. [DOI] [PubMed] [Google Scholar]
- Al-Majed AA, Neumann CM, Brushart TM, Gordon T. Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J Neurosci. 2000;20:2602–2608. doi: 10.1523/JNEUROSCI.20-07-02602.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angata K, Huckaby V, Ranscht B, Terskikh A, Marth JD, Fukuda M. Polysialic acid-directed migration and differentiation of neural precursors are essential for mouse brain development. Mol Cell Biol. 2007;27:6659–6668. doi: 10.1128/MCB.00205-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asahara T, Lin M, Kumazawa Y, Takeo K, Akamine T, Nishimura Y, Kayahara T, Yamamoto T. Long-term observation on the changes of somatotopy in the facial nucleus after nerve suture in the cat: morphological studies using retrograde labeling. Brain Res Bull. 1999;49:195–202. doi: 10.1016/s0361-9230(99)00041-6. [DOI] [PubMed] [Google Scholar]
- Bolton SJ, Perry VH. Histone H1; a neuronal protein that binds bacterial lipopolysaccharide. J Neurocytol. 1997;26:823–831. doi: 10.1023/a:1018574600961. [DOI] [PubMed] [Google Scholar]
- Brix K, Summa W, Lottspeich F, Herzog V. Extracellularly occurring histone H1 mediates the binding of thyroglobulin to the cell surface of mouse macrophages. J Clin Invest. 1998;102:283–293. doi: 10.1172/JCI1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brushart TM. Preferential reinnervation of motor nerves by regenerating motor axons. J Neurosci. 1988;8:1026–1031. doi: 10.1523/JNEUROSCI.08-03-01026.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brushart TM, Jari R, Verge V, Rohde C, Gordon T. Electrical stimulation restores the specificity of sensory axon regeneration. Exp Neurol. 2005;194:221–229. doi: 10.1016/j.expneurol.2005.02.007. [DOI] [PubMed] [Google Scholar]
- Carratù MR, Labate M, De Santis S, Lacomba C, Cuomo V. Up-regulation of polysialic acid in peripheral myelinated axons of rat chronically exposed to 2,5-hexanedione. Pharmacol Toxicol. 1993;72:236–239. doi: 10.1111/j.1600-0773.1993.tb01642.x. [DOI] [PubMed] [Google Scholar]
- Chazal G, Durbec P, Jankovski A, Rougon G, Cremer H. Consequences of neural cell adhesion molecule deficiency on cell migration in the rostral migratory stream of the mouse. J Neurosci. 2000;20:1446–1457. doi: 10.1523/JNEUROSCI.20-04-01446.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen RS, Blomberg F, Berzins K, Siekevitz P. The structure of postsynaptic densities isolated from dog cerebral cortex. J Cell Biol. 1977;74:181–203. doi: 10.1083/jcb.74.1.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curreli S, Arany Z, Gerardy-Schahn R, Mann D, Stamatos NM. Polysialylated neuropilin-2 is expressed on the surface of human dendritic cells and modulates dendritic cell-T lymphocyte interactions. J Biol Chem. 2007;282:30346–30356. doi: 10.1074/jbc.M702965200. [DOI] [PubMed] [Google Scholar]
- Decker L, Avellana-Adalid V, Nait-Oumesmar B, Durbec P, Baron-Van Evercooren A. Oligodendrocyte precursor migration and differentiation: combined effects of PSA residues, growth factors, and substrates. Mol Cell Neurosci. 2000;16:422–439. doi: 10.1006/mcne.2000.0885. [DOI] [PubMed] [Google Scholar]
- de la Cruz RR, Pastor AM, Delgado-García JM. Effects of target depletion on adult mammalian central neurons: morphological correlates. Neuroscience. 1994;58:59–79. doi: 10.1016/0306-4522(94)90156-2. [DOI] [PubMed] [Google Scholar]
- Dihné M, Bernreuther C, Sibbe M, Paulus W, Schachner M. A new role for the cell adhesion molecule L1 in neural precursor cell proliferation, differentiation, and transmitter-specific subtype generation. J Neurosci. 2003;23:6638–6650. doi: 10.1523/JNEUROSCI.23-16-06638.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dityatev A, Dityateva G, Sytnyk V, Delling M, Toni N, Nikonenko I, Muller D, Schachner M. Polysialylated neural cell adhesion molecule promotes remodeling and formation of hippocampal synapses. J Neurosci. 2004;24:9372–9382. doi: 10.1523/JNEUROSCI.1702-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doenecke D, Albig W, Bode C, Drabent B, Franke K, Gavenis K, Witt O. Histones: genetic diversity and tissue-specific gene expression. Histochem Cell Biol. 1997;107:1–10. doi: 10.1007/s004180050083. [DOI] [PubMed] [Google Scholar]
- Doherty P, Walsh FS. CAM-FGF receptor interactions: a model for axonal growth. Mol Cell Neurosci. 1996;8:99–111. doi: 10.1006/mcne.1996.0049. [DOI] [PubMed] [Google Scholar]
- Doherty P, Cohen J, Walsh FS. Neurite outgrowth in response to transfected N-CAM changes during development and is modulated by polysialic acid. Neuron. 1990;5:209–219. doi: 10.1016/0896-6273(90)90310-c. [DOI] [PubMed] [Google Scholar]
- Duce JA, Smith DP, Blake RE, Crouch PJ, Li QX, Masters CL, Trounce IA. Linker histone H1 binds to disease associated amyloid-like fibrils. J Mol Biol. 2006;361:493–505. doi: 10.1016/j.jmb.2006.06.038. [DOI] [PubMed] [Google Scholar]
- Evans SV, Sigurskjold BW, Jennings HJ, Brisson JR, To R, Tse WC, Altman E, Frosch M, Weisgerber C, Kratzin HD. Evidence for the extended helical nature of polysaccharide epitopes. The 2.8 Å resolution structure and thermodynamics of ligand binding of an antigen binding fragment specific for alpha-(2→8)-polysialic acid. Biochemistry. 1995;34:6737–6744. doi: 10.1021/bi00020a019. [DOI] [PubMed] [Google Scholar]
- Franz CK, Rutishauser U, Rafuse VF. Polysialylated neural cell adhesion molecule is necessary for selective targeting of regenerating motor neurons. J Neurosci. 2005;25:2081–2091. doi: 10.1523/JNEUROSCI.4880-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frosch M, Görgen I, Boulnois GJ, Timmis KN, Bitter-Suermann D. NZB mouse system for production of monoclonal antibodies to weak bacterial antigens: isolation of an IgG antibody to the polysaccharide capsules of Escherichia coli K1 and group B meningococci. Proc Natl Acad Sci U S A. 1985;82:1194–1198. doi: 10.1073/pnas.82.4.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Segura LM, Cañas B, Parducz A, Rougon G, Theodosis D, Naftolin F, Torres-Aleman I. Estradiol promotion of changes in the morphology of astroglia growing in culture depends on the expression of polysialic acid of neural membranes. Glia. 1995;13:209–216. doi: 10.1002/glia.440130307. [DOI] [PubMed] [Google Scholar]
- Griffiths AD, Williams SC, Hartley O, Tomlinson IM, Waterhouse P, Crosby WL, Kontermann RE, Jones PT, Low NM, Allison TJ. Isolation of high affinity human antibodies directly from large synthetic repertoires. EMBO J. 1994;13:3245–3260. doi: 10.1002/j.1460-2075.1994.tb06626.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grothe C, Meisinger C, Claus P. In vivo expression and localization of the fibroblast growth factor system in the intact and lesioned rat peripheral nerve and spinal ganglia. J Comp Neurol. 2001;434:342–357. doi: 10.1002/cne.1181. [DOI] [PubMed] [Google Scholar]
- Guseva D, Angelov DN, Irintchev A, Schachner M. Ablation of adhesion molecule L1 in mice favours Schwann cell proliferation and functional recovery after peripheral nerve injury. Brain. 2009;132:2180–2195. doi: 10.1093/brain/awp160. [DOI] [PubMed] [Google Scholar]
- Hammond MS, Sims C, Parameshwaran K, Suppiramaniam V, Schachner M, Dityatev A. Neural cell adhesion molecule-associated polysialic acid inhibits NR2B-containing N-methyl-d-aspartate receptors and prevents glutamate-induced cell death. J Biol Chem. 2006;281:34859–34869. doi: 10.1074/jbc.M602568200. [DOI] [PubMed] [Google Scholar]
- Hampton RY, Golenbock DT, Raetz CR. Lipid A binding sites in membranes of macrophage tumor cells. J Biol Chem. 1988;263:14802–14807. [PubMed] [Google Scholar]
- Hariton-Gazal E, Rosenbluh J, Graessmann A, Gilon C, Loyter A. Direct translocation of histone molecules across cell membranes. J Cell Sci. 2003;116:4577–4586. doi: 10.1242/jcs.00757. [DOI] [PubMed] [Google Scholar]
- Henriquez JP, Casar JC, Fuentealba L, Carey DJ, Brandan E. Extracellular matrix histone H1 binds to perlecan, is present in regenerating skeletal muscle and stimulates myoblast proliferation. J Cell Sci. 2002;115:2041–2051. doi: 10.1242/jcs.115.10.2041. [DOI] [PubMed] [Google Scholar]
- Holers VM, Kotzin BL. Human peripheral blood monocytes display surface antigens recognized by monoclonal antinuclear antibodies. J Clin Invest. 1985;76:991–998. doi: 10.1172/JCI112100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holliger P, Prospero T, Winter G. “Diabodies”: small bivalent and bispecific antibody fragments. Proc Natl Acad Sci U S A. 1993;90:6444–6448. doi: 10.1073/pnas.90.14.6444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu H. Polysialic acid regulates chain formation by migrating olfactory interneuron precursors. J Neurosci Res. 2000;61:480–492. doi: 10.1002/1097-4547(20000901)61:5<480::AID-JNR2>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- Hu H, Tomasiewicz H, Magnuson T, Rutishauser U. The role of polysialic acid in migration of olfactory bulb interneuron precursors in the subventricular zone. Neuron. 1996;16:735–743. doi: 10.1016/s0896-6273(00)80094-x. [DOI] [PubMed] [Google Scholar]
- Jungnickel J, Gransalke K, Timmer M, Grothe C. Fibroblast growth factor receptor 3 signaling regulates injury-related effects in the peripheral nervous system. Mol Cell Neurosci. 2004;25:21–29. doi: 10.1016/j.mcn.2003.09.014. [DOI] [PubMed] [Google Scholar]
- Jungnickel J, Brämer C, Bronzlik P, Lipokatic-Takacs E, Weinhold B, Gerardy-Schahn R, Grothe C. Level and localization of polysialic acid is critical for early peripheral nerve regeneration. Mol Cell Neurosci. 2009;40:374–381. doi: 10.1016/j.mcn.2008.12.003. [DOI] [PubMed] [Google Scholar]
- Kanato Y, Kitajima K, Sato C. Direct binding of polysialic acid to a brain-derived neurotrophic factor depends on the degree of polymerization. Glycobiology. 2008;18:1044–1053. doi: 10.1093/glycob/cwn084. [DOI] [PubMed] [Google Scholar]
- Kiselyov VV, Skladchikova G, Hinsby AM, Jensen PH, Kulahin N, Soroka V, Pedersen N, Tsetlin V, Poulsen FM, Berezin V, Bock E. Structural basis for a direct interaction between FGFR1 and NCAM and evidence for a regulatory role of ATP. Structure. 2003;11:691–701. doi: 10.1016/s0969-2126(03)00096-0. [DOI] [PubMed] [Google Scholar]
- Kleene R, Schachner M. Glycans and neural cell interactions. Nat Rev Neurosci. 2004;5:195–208. doi: 10.1038/nrn1349. [DOI] [PubMed] [Google Scholar]
- Kruse J, Mailhammer R, Wernecke H, Faissner A, Sommer I, Goridis C, Schachner M. Neural cell adhesion molecules and myelin-associated glycoprotein share a common carbohydrate moiety recognized by monoclonal antibodies L2 and HNK-1. Nature. 1984;311:153–155. doi: 10.1038/311153a0. [DOI] [PubMed] [Google Scholar]
- Lüthl A, Laurent JP, Figurov A, Muller D, Schachner M. Hippocampal long-term potentiation and neural cell adhesion molecules L1 and NCAM. Nature. 1994;372:777–779. doi: 10.1038/372777a0. [DOI] [PubMed] [Google Scholar]
- Makhina T, Loers G, Schulze C, Ueberle B, Schachner M, Kleene R. Extracellular GAPDH binds to L1 and enhances neurite outgrowth. Mol Cell Neurosci. 2009;41:206–218. doi: 10.1016/j.mcn.2009.02.010. [DOI] [PubMed] [Google Scholar]
- Marino P, Norreel JC, Schachner M, Rougon G, Amoureux MC. A polysialic acid mimetic peptide promotes functional recovery in a mouse model of spinal cord injury. Exp Neurol. 2009;219:163–174. doi: 10.1016/j.expneurol.2009.05.009. [DOI] [PubMed] [Google Scholar]
- Mehanna A, Mishra B, Kurschat N, Schulze C, Bian S, Loers G, Irintchev A, Schachner M. Polysialic acid glycomimetics promote myelination and functional recovery after peripheral nerve injury in mice. Brain. 2009;132:1449–1462. doi: 10.1093/brain/awp128. [DOI] [PubMed] [Google Scholar]
- Mehanna A, Jakovcevski I, Acar A, Xiao M, Loers G, Rougon G, Irintchev A, Schachner M. Polysialic acid glycomimetic promotes functional recovery and plasticity after spinal cord injury in mice. Mol Ther. 2010;18:34–43. doi: 10.1038/mt.2009.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mühlenhoff M, Eckhardt M, Gerardy-Schahn R. Polysialic acid: three-dimensional structure, biosynthesis and function. Curr Opin Struct Biol. 1998;8:558–564. doi: 10.1016/s0959-440x(98)80144-9. [DOI] [PubMed] [Google Scholar]
- Muller D, Wang C, Skibo G, Toni N, Cremer H, Calaora V, Rougon G, Kiss JZ. PSA-NCAM is required for activity-induced synaptic plasticity. Neuron. 1996;17:413–422. doi: 10.1016/s0896-6273(00)80174-9. [DOI] [PubMed] [Google Scholar]
- Muller D, Djebbara-Hannas Z, Jourdain P, Vutskits L, Durbec P, Rougon G, Kiss JZ. Brain-derived neurotrophic factor restores long-term potentiation in polysialic acid-neural cell adhesion molecule-deficient hippocampus. Proc Natl Acad Sci U S A. 2000;97:4315–4320. doi: 10.1073/pnas.070022697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogata T, Iijima S, Hoshikawa S, Miura T, Yamamoto S, Oda H, Nakamura K, Tanaka S. Opposing extracellular signal-regulated kinase and Akt pathways control Schwann cell myelination. J Neurosci. 2004;24:6724–6732. doi: 10.1523/JNEUROSCI.5520-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen M, Zuber C, Roth J, Linnemann D, Bock E. The ability to re-express polysialylated NCAM in soleus muscle after denervation is reduced in aged rats compared to young adult rats. Int J Dev Neurosci. 1995;13:97–104. doi: 10.1016/0736-5748(95)00003-y. [DOI] [PubMed] [Google Scholar]
- Papastefanaki F, Chen J, Lavdas AA, Thomaidou D, Schachner M, Matsas R. Grafts of Schwann cells engineered to express PSA-NCAM promote functional recovery after spinal cord injury. Brain. 2007;130:2159–2174. doi: 10.1093/brain/awm155. [DOI] [PubMed] [Google Scholar]
- Parseghian MH, Luhrs KA. Beyond the walls of the nucleus: the role of histones in cellular signaling and innate immunity. Biochem Cell Biol. 2006;84:589–604. doi: 10.1139/o06-082. [DOI] [PubMed] [Google Scholar]
- Patrzykat A, Zhang L, Mendoza V, Iwama GK, Hancock RE. Synergy of histone-derived peptides of coho salmon with lysozyme and flounder pleurocidin. Antimicrob Agents Chemother. 2001;45:1337–1342. doi: 10.1128/AAC.45.5.1337-1342.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potempska A, Ramakrishna N, Wisniewski HM, Miller DL. Interaction between the beta-amyloid peptide precursor and histones. Arch Biochem Biophys. 1993;304:448–453. doi: 10.1006/abbi.1993.1374. [DOI] [PubMed] [Google Scholar]
- Rutishauser U. Polysialic acid in the plasticity of the developing and adult vertebrate nervous system. Nat Rev Neurosci. 2008;9:26–35. doi: 10.1038/nrn2285. [DOI] [PubMed] [Google Scholar]
- Simova O, Irintchev A, Mehanna A, Liu J, Dihné M, Bächle D, Sewald N, Loers G, Schachner M. Carbohydrate mimics promote functional recovery after peripheral nerve repair. Ann Neurol. 2006;60:430–437. doi: 10.1002/ana.20948. [DOI] [PubMed] [Google Scholar]
- Sofroniew MV, Howe CL, Mobley WC. Nerve growth factor signaling, neuroprotection, and neural repair. Annu Rev Neurosci. 2001;24:1217–1281. doi: 10.1146/annurev.neuro.24.1.1217. [DOI] [PubMed] [Google Scholar]
- Storms SD, Rutishauser U. A role for polysialic acid in neural cell adhesion molecule heterophilic binding to proteoglycans. J Biol Chem. 1998;273:27124–27129. doi: 10.1074/jbc.273.42.27124. [DOI] [PubMed] [Google Scholar]
- Vaithianathan T, Matthias K, Bahr B, Schachner M, Suppiramaniam V, Dityatev A, Steinhaüser C. Neural cell adhesion molecule-associated polysialic acid potentiates AMPA receptor currents. J Biol Chem. 2004;279:47975–47984. doi: 10.1074/jbc.M407138200. [DOI] [PubMed] [Google Scholar]
- Vrbova G, Mehra N, Shanmuganathan H, Tyreman N, Schachner M, Gordon T. Chemical communication between regenerating motor axons and Schwann cells in the growth pathway. Eur J Neurosci. 2009;30:366–375. doi: 10.1111/j.1460-9568.2009.06847.x. [DOI] [PubMed] [Google Scholar]
- Waters HJ, Barnett G, O'Hanlon GM, Lowrie MB. Motoneuron survival after neonatal peroneal nerve injury in the rat—evidence for the sparing effect of reciprocal inhibition. Exp Neurol. 1998;152:95–100. doi: 10.1006/exnr.1998.6820. [DOI] [PubMed] [Google Scholar]
- Watson K, Edwards RJ, Shaunak S, Parmelee DC, Sarraf C, Gooderham NJ, Davies DS. Extra-nuclear location of histones in activated human peripheral blood lymphocytes and cultured T-cells. Biochem Pharmacol. 1995;50:299–309. doi: 10.1016/0006-2952(95)00142-m. [DOI] [PubMed] [Google Scholar]
- Watson K, Gooderham NJ, Davies DS, Edwards RJ. Nucleosomes bind to cell surface proteoglycans. J Biol Chem. 1999;274:21707–21713. doi: 10.1074/jbc.274.31.21707. [DOI] [PubMed] [Google Scholar]
- Zhang JY, Luo XG, Xian CJ, Liu ZH, Zhou XF. Endogenous BDNF is required for myelination and regeneration of injured sciatic nerve in rodents. Eur J Neurosci. 2000;12:4171–4180. [PubMed] [Google Scholar]