Abstract
Background
Although lung protection with low tidal volume and limited plateau pressure (Pplat) improves survival in acute respiratory distress syndrome patients (ARDS), the best way to set positive end-expiratory pressure (PEEP) is still debated.
Methods
This study aimed to compare two strategies using individual PEEP based on a maximum Pplat (28–30 cmH2O, the Express group) or on keeping end-expiratory transpulmonary pressure positive (0–5 cmH2O, PLexpi group). We estimated alveolar recruitment (Vrec), end-expiratory lung volume and alveolar distension based on elastance-related end-inspiratory transpulmonary pressure (PL,EL).
Results
Nineteen patients with moderate to severe ARDS (PaO2/FiO2 < 150 mmHg) were included with a baseline PEEP of 7.0 ± 1.8 cmH2O and a PaO2/FiO2 of 91.2 ± 31.2 mmHg. PEEP and oxygenation increased significantly from baseline with both protocols; PEEP Express group was 14.2 ± 3.6 cmH2O versus 16.7 ± 5.9 cmH2O in PLexpi group. No patient had the same PEEP with the two protocols. Vrec was higher with the latter protocol (299 [0 to 875] vs. 222 [47 to 483] ml, p = 0.049) and correlated with improved oxygenation (R2 = 0.45, p = 0.002). Two and seven patients in the Express and PL,expi groups, respectively, had PL,EL > 25 cmH2O.
Conclusions
There is a great heterogeneity of PLexpi when Pplat is used to titrate PEEP but with limited risk of over-distension. A PEEP titration for a moderate positive level of PLexpi might slightly improve alveolar recruitment and oxygenation but increases the risk of over-distension in one-third of patients.
Keywords: ARDS, PEEP titration, Monitoring, Transpulmonary pressure
Background
Mechanical ventilation for acute respiratory distress syndrome (ARDS) may lead to ventilation-induced lung injury [1]. A lung protective ventilation strategy, with low tidal volume (VT), limited plateau pressure and positive end-expiratory pressure (PEEP), aims to improve survival [2, 3]. Different protocols have been proposed to set PEEP in order to avoid alveolar collapse with limited end-inspiratory distension of the lungs [4]. Some of these strategies use a table of PEEP values which depend on inspired fraction of oxygen (FiO2), while others are based on individual respiratory mechanics. The Express protocol, developed by Mercat et al., consists of attaining airway plateau pressure (Pplat) up to 28–30 cmH2O with a fixed VT of 6 ml kg−1 predicted body weight [5]. These authors reported a significant reduction in morbidity but not mortality. Because airway pressure is an oversimplified surrogate for lung stress in patients with abnormal chest wall elastance, it could be relevant to assess lung distending pressure estimated from transpulmonary pressure (PL). This later, in static airway conditions, can be estimated by measuring pleural via esophageal pressure [6]. This estimation can be affected by elastic recoil of the balloon, of the esophagus, esophageal muscular tone and pressures transmitted from the heart beat and mediastinum [7]. The relationship between esophageal and pleural pressure, and its measurement in ARDS patients with an important anteroposterior gradient in the supine position, requires the acceptance of several assumptions [8]. However, a recent study directly measured pleural pressure in pigs and human cadavers and found that esophageal pressure accurately reflects pleural pressure close to the balloon, corresponding to dependent lung regions to mid-chest [7]. Thus, collapse and trauma from recurrent alveolar collapse and re-opening can be related to end-expiratory PL. [9, 10] Elastance-derived calculation of relative end-inspiratory PL (PL,EL) is close to direct measurement of pleural transpulmonary end-inspiratory pressure in the non-dependent lung region and might therefore give some information about alveolar distension in the non-dependent lung, more at risk for over-distension [11]. Different protocols have been proposed for setting PEEP according to PL. The EPVent 1 and 2 trials titrated PEEP by measuring pleural pressure to achieve a positive end-expiratory transpulmonary pressure (PLexpi) between 0 to 10 cmH2O according to a sliding scale based on FiO2 [12, 13] Grasso et al. used PL,EL to increase PEEP in severe ARDS until PL,EL = 25 cmH2O [14]. Some patients improved their oxygenation significantly and avoided extracorporeal membrane oxygenation.
The aim of this study was to compare estimated alveolar recruitment (Vrec) with end-expiratory lung volume (EELV) measurement, and alveolar distension with measurement of PL,EL, during individual PEEP titration using two different targets: Pplat (28–30 cmH2O) or positive PLexpi (0–5 cmH2O).
Methods
Study design and participants
This multicenter, prospective crossover physiological study was conducted in severe ARDS patients admitted to three French intensive care units in Bordeaux (Robert Picqué Military Teaching Hospital; North Bordeaux Aquitaine Clinic; Thoracic Intensive Care Unit, Bordeaux University Hospital) between 2016 and 2017. All patients had recent (within a week) bilateral opacities not fully explained by cardiac failure or fluid overload with moderate to severe hypoxemia defined by their PaO2/FiO2 below 150 with 5–8 cmH2O of PEEP and required volume-controlled mechanical ventilation [15]. The ventilator used was a carescape R860 (General Electrics, Madison WI, USA).
Exclusion criteria included esophageal disease, pulmonary leakage (major bronchopleural fistula, pneumothorax), severe coagulopathy, solid organ transplantation (hepatic, pulmonary) and refusal to participate.
Experimental protocol
Sedation was achieved with a midazolam-sufentanil infusion to obtain a bispectral index between 40 and 60. Subjects recieved cisatracurium to obtain myorelaxation, monitoring was Train Of Four of ulnar, 1 or 2 twitches out of 4 was considered appropriate. All subjects were placed in a 30° head up position. A validated nasogastric tube with an esophageal balloon-catheter (Nutrivent™; Sidam, Modena, Italy) was inserted to estimate pleural pressure [16]. The balloon was filled with 4 ml of air. The correct position of the Nutrivent tube was confirmed by an end-expiratory occlusion maneuver with four chest compressions and four ΔPes/ΔPaw ratio measurements as described previously and with thoracic radiography (radio-opaque markers) [6, 17]. At baseline, patients were ventilated with a VT of 6 ml kg−1 predicted body weight and a PEEP between 5–8 cmH2O (PEEPbaseline). Twenty minutes later, PEEP was titrated according to the Express or PLexpi protocols in a randomized order. For the Express protocol, PEEP was titrated in order to obtain a Pplat between 28–30 cmH2O. For the PLexpi protocol, PEEP was titrated in order to obtain a PLexpi between 0–5 cmH2O. PEEP level according to each protocol was maintained for 20 min before recording all respiratory parameters and blood withdrawal for blood gas analysis. PEEP was returned to 0 cmH2O between each protocol during less than 30 s. For hemodynamic assessment, respiratory variation of the arterial pulse pressure and the response to a passive leg raising test were used before PEEP titration, with an echocardiography. If positive, a fluid challenge was performed to avoid hypovolemia. After PEEP titration, echocardiography was done to assess right heart function and the occurrence of septal dyskinesia.
Measurement of variables
End-inspiratory and -expiratory airway and esophageal pressures were measured during a 5 s pause of the ventilator; VT were monitored continuously. EELV was measured using the nitrogen washin/washout technique; FiO2 variation was 10% and the average of washin EELV and washout EELV for each PEEP levels was recorded.
Variables were calculated using the following equations:
Absolute inspiratory transpulmonary pressure (PL,es) = Pplat − end-inspiratory esophageal pressure;
Elastance-related transpulmonary pressure (PL,EL) = Pplat × (lung elastance/respiratory system elastance) [18];
PLexpi = total PEEP − end-expiratory esophageal pressure;
Airway driving pressure (DPaw) = Pplat − total PEEP;
Transpulmonary driving pressure (DPL) = DPaw − (end-inspiratory − end-expiratory esophageal pressure);
Elastance-related driving pressure = DPaw × (lung elastance/respiratory system elastance).
Respiratory system elastance = (Pplat − total PEEP)/VT;
Lung elastance = DPL/VT;
Respiratory system elastance = Lung elastance + Chest wall elastance.
The following were also measured:
Estimated recruitment volume (Vrec in ml) = (EELV at high PEEP − EELV at low PEEP) − ((VT/(Pplat − low PEEP) × (high PEEP − low PEEP)). [19]
Statistical analysis
No statistical power calculation was conducted prior to the study; the sample size was based on our previous studies with this design of physiological crossover study with pairing. Data are expressed, respectively, as mean ± standard deviation (SD) and median [interquartile range] for variables normally and non-normally distributed. The outliers were evaluated, but no action was necessary. The categorical data were expressed as numbers (percentage of patients). Comparison of variables between three settings was performed by using one-way repeated measures analysis of variance (ANOVA) followed by post hoc Tukey’s test for multiple comparisons. Comparison between categorical variables was performed using the Chi-squared test. Correlations used Spearman’s test. All statistical tests were two-tailed, and a p value of less than 0.05 was considered significant. All statistical analysis was performed using NCSS2007 software (Statistical Solutions Ltd, Cork, Ireland) and Prism 6 (GraphPad Software, La Jolla, CA, USA).
Results
Nineteen patients were included; they were all enrolled in the study less than 48 h after intubation. The baseline characteristics of these patients are summarized in Table 1, and there were no missing data.
Table 1.
Baseline characteristics of the patients (n = 19)
| Characteristic | |
|---|---|
| Male, n (%) | 13 (68.4) |
| Age (years) | 72 ± 10 |
| Body mass index (kg/m2) | 28 ± 6 |
| SAPS II score | 65 ± 15 |
| Etiology of ARDS, n (%) | |
| Pneumonia/aspiration | 16 (84.2) |
| Sepsis | 2 (10.5) |
| Pancreatitis | 1 (5.3) |
| Organ failure at baseline (SOFA), n (%) | |
| Hemodynamic | 18 (94.7) |
| Renal | 9 (47.4) |
| Hepatic | 0 (0) |
| Hematological | 2 (10.5) |
| Arterial blood gas | |
| PaO2/FiO2 ratio | 92 ± 31 |
| FiO2 (%) | 80 ± 21 |
| pH | 7.31 ± 0.11 |
| PaCO2 (mmHg) | 45 ± 10 |
| HCO3- (mmol/l) | 22.4 ± 4.0 |
| Base excess | − 3.4 ± 5.1 |
| Lactates (mmol/l) | 1.6 ± 1.0 |
| Hemodynamic variables | |
| Heart rate (beats/min) | 99 ± 27 |
| Systolic arterial pressure (mmHg) | 127 ± 23 |
| Diastolic arterial pressure (mmHg) | 59 ± 12 |
| Mean arterial pressure (mmHg) | 82 ± 13 |
| Respiratory mechanics | |
| Minute ventilation (L/min) | 9.6 ± 1.6 |
| Tidal volume (ml/kg PBW) | 6.1 ± 0.4 |
| EELV (ml) | 1319 ± 626 |
| Aspect of ARDS, n (%) | |
| Patchy | 6 (31.6) |
| Diffuse | 10 (52.6) |
| Focal | 3 (15.8) |
| Mortality at Hospital discharge | 10/19 (53%) |
Results are expressed as number (%), or mean ± standard deviation
SAPS 2: Simplified Acute Physiology Score 2; ARDS: acute respiratory distress syndrome; PBW: predicted body weight. SOFA: sepsis-related organ failure assessment; EELV: end-expiratory lung volume (ml)
PEEP levels, oxygenation and alveolar recruitment
Respiratory mechanics according to each PEEP setting are summarized in Table 2. In comparison with PEEPbaseline, the Express and PLexpi protocols significantly increased PEEP and Pplat without any change in driving pressure (Table 2). These changes were associated with a significant improvement in oxygenation. Median PEEP value was not significantly different between the Express and PLexpi protocols. However, analysis of individual PEEP data according to each protocol shows that no patient had the same PEEP (Fig. 1) with a median of absolute difference of 5.0 [4.0–8.0] cmH2O and 13 patients had higher PEEP with PLexpi protocol.
Table 2.
Measurements of respiratory function and hemodynamics (n = 19)
| Protocols | PEEPbaseline | Express protocol | P Lexpi | p value |
|---|---|---|---|---|
| PEEP (cmH2O) | 7.0 ± 1.8 | 14.2 ± 3.6* | 16.7 (5.9)* | < 0.0001 |
| Pplat (cmH2O) | 20.8 ± 4.0 | 28.8 ± 2.0 * | 33.9 ± 10.6* | < 0.0001 |
| PL,es (cmH2O) | 7.0 ± 5.9 | 11.9 ± 6.2* | 15.5 ± 8.5* | 0.0013 |
| PL,EL (cmH2O) | 15.3 ± 4.9) | 20.5 ± 4.7* | 24.3 ± 11.4* | 0.0025 |
| PLexpi (cmH2O) | − 2.6 ± 5.2 | 1.4 ± 5.1* | 3.3 ± 1.6* | < 0.0001 |
| EELV (ml) | 1546 ± 634 | 2067 ± 924* | 2287 ± 945* | 0.001 |
| DPaw (cmH2O) | 13.0 ± 3.9 | 14.2 ± 5.0 | 16.4 ± 7.8 | 0.17 |
| DPL (cmH2O) | 9.9 ± 4.4 | 10.6 ± 5.6 | 12.3 ± 8.3 | 0.20 |
| DPL,EL (cmH2O) | 7.5 ± 4.3 | 8.1 ± 5.6 | 9.5 ± 8.1 | 0.30 |
| Crs (ml/cmH2O) | 33.3 ± 15.8 | 30.0 ± 10.7 | 28.3 ± 13.2 | 0.17 |
| Ecw (cmH2O/l) | 8.7 ± 2.7 | 9.6 ± 3.4* | 10.9 ± 4.3* | 0.03 |
| EL (cmH2O/l) | 26.0 ± 11.9 | 28.0 ± 15.9 | 33.2 ± 25.1 | 0.25 |
| FiO2 (%) | 80.0 ± 21.1 | 80.6 ± 21.2 | 81.1 ± 21.6 | 0.46 |
| PaO2/FiO2 | 91.2 ± 31.2 | 134.0 ± 67.2* | 152.7 ± 80.1* | 0.01 |
| pH | 7.31 ± 0.11 | 7.30 ± 0.11 | 7.31 ± 0.12 | 0.08 |
| PaCO2 (mmHg) | 45.2 ± 10.4 | 46.5 ± 9.6 | 45.3 ± 11.0 | 0.26 |
| MAP (mmHg) | 82.0 ± 13.4 | 74.7 ± 12.9 | 75.7 ± 12.0 | 0.06 |
| Heart rate (beats/min) | 99 ± 27 | 102 ± 26 | 107 ± 28 | 0.19 |
| Lactates (mmol/l) | 1.6 ± 0.9 | 1.5 ± 0.8 | 1.5 ± 0.8 | 0.27 |
Results are expressed as mean ± standard deviation
Pplat: plateau pressure; PLexpi: end-expiratory transpulmonary pressure; DPL: transpulmonary driving pressure; PL,EL: relative end-expiratory pressure; PL,es: absolute inspiratory transpulmonary pressure; DPaw: airway driving pressure; DPL: transpulmonary driving pressure, DPL,EL: transpulmonary elastance-related driving pressure; EL: lung elastance; EELV: end-expiratory lung volume; Ecw: elastance chest wall; Crs: compliance respiratory system; MAP: mean arterial pressure. p value refers to repeated measures ANOVA. *p < 0.05 of Express and PLexpi groups versus baseline group. §p < 0.05 of Express versus PLexpi groups
Fig. 1.
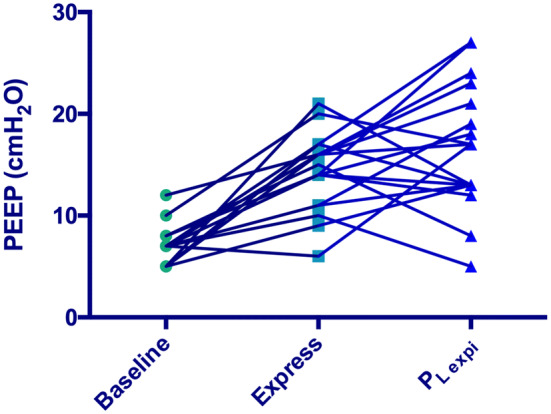
Individual PEEP levels according to the Express or PLexpi protocol. PEEP increased from baseline but is individually different for almost all patients with each protocol Express or PLexpi
EELV variation from baseline was significantly higher with PLexpi protocol 60 (58) % vs 36 (28) % with Express protocol, p = 0.025. Estimated Vrec was significantly higher with the PLexpi protocol than with Express, 298 [0 to 845] vs 222 [47 to 483] ml, respectively. Vrec and PaO2/FiO2 ratio changes were significantly correlated with the PLexpi protocol (R2 = 0.45, p = 0.002). Arterial blood gases were not significantly different between the Express and PLexpi protocols.
Expiratory transpulmonary pressure
PLexpi increased with both protocols (Table 2, Fig. 2).
Fig. 2.
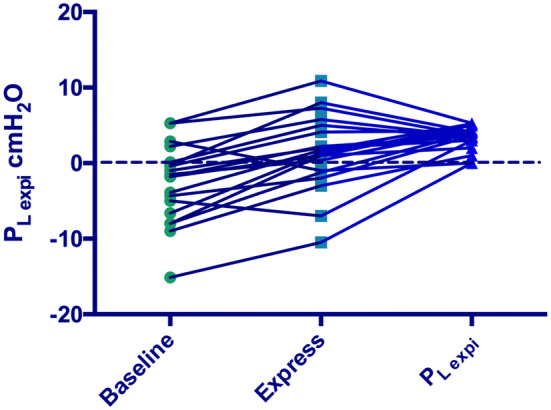
Individual PLexpi levels according to baseline, Express and PLexpi protocols. PLexpi = positive end-expiratory transpulmonary pressure. Dash line represents the limit of 0 cmH2O; more patients had negative PLexpi with the Express protocol
With the Express protocol, six patients had negative PLexpi − 2.5 [− 7.8 to − 1.15] cmH2O with a median PEEP of 14.0 [9.7 to 14.5] cmH2O. These six patients had a significant lower respiratory system compliance (25.2 [15.8 to 29.0] vs. 30.7 [25.6 to 39.2] ml/cmH2O, p = 0.027), and a significant lower EELV (1240 [835 to 2118] vs. 2019 [1700 to 3045] ml, p = 0.040). When these patients received the PLexpi protocol, median PEEP increased to 24.0 [18.0 to 26.5] cmH2O, Pplat was 40.2 [33.1 to 51.0] cmH2O, and PaO2/FiO2 increased by +31 [11 to 206] % (p = 0.062).
Inspiratory transpulmonary pressure
With the Express protocol, mean PL,EL was 20 cmH2O, and PL,es was significantly lower than PL,EL with a mean of difference of 8.5 cmH2O (p < 0.0001) (Table 2). PL,EL increased from baseline with the Express protocol (Table 2), one patient was > 25 cmH2O, and the rest were below (Fig. 3), but 2 patients had PL,es > 20 cmH2O.
Fig. 3.
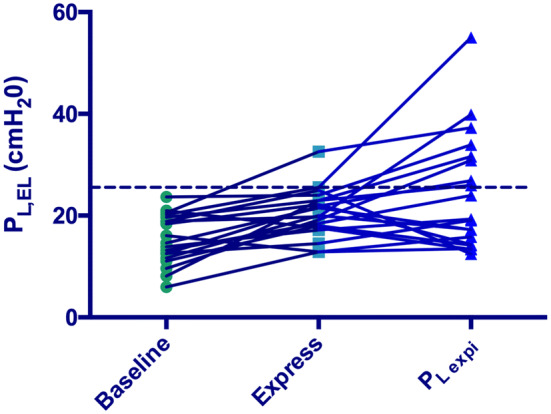
Individual PL,EL with baseline, Express and PLexpi protocols. PL,EL = elastance-derived calculation of relative end-inspiratory transpulmonary pressure. Dash line represents the limit of 25 cmH2O; more patients had PL,EL above 25 cmH2O with the PLexpi protocol
With the PLexpi protocol, mean PL,EL was 24 cmH2O, and PL,es was significantly lower than PL,EL with a mean of difference of 8.7 cmH2O (p < 0.0001) (Table 2). PL,EL increased from baseline with the PLexpi protocol (Table 2), seven patients had PL,EL > 25 cmH2O (Fig. 3), and four of them had PL,es > 20 cmH2O.
With the PLexpi protocol, patients with PL,EL > 25 cmH2O had pH of 7.25 [7.17 to 7.42] with significantly lower respiratory system compliance, higher DP and PEEP, than those with PL,EL ≤ 25 cmH2O (17 [15 to 22] ml/cmH2O, 23 [18 to 25] cmH2O and 21[17 to 27] cmH2O, respectively).
Airway, transpulmonary and elastance-related driving pressures were not different between groups (Table 2).
Elastance
In comparison with baseline, respiratory system elastance and lung elastance were not significantly modified by the increase in PEEP with the Express protocol; only chest wall elastance was statistically different (Table 2). PEEP increase from baseline with the Express protocol was significantly correlated with lung elastance (R2 = 0.38, p = 0.008). Respiratory system and lung elastance were similar between Express and PLexpi protocols.
Complications
No episodes of pneumothorax were detected by chest X-Ray after each protocol implementation. Hemodynamic assessment was done before PEEP titration, and fluid was given when required. Cardiac frequency and mean arterial pressure remained relatively stable with each PEEP protocol (Table 2). Fifteen patients underwent transthoracic echocardiography (four had no transthoracic echocardiography window), and none of them had right heart failure with septal dyskinesia after PEEP titration.
Discussion
This study demonstrates heterogeneity of end-expiratory or end-inspiratory transpulmonary pressures during PEEP titration with each method in severe ARDS. PEEP titration with a target of positive PLexpi will suggest individually different levels of PEEP than with Express protocol in a large majority of patients (higher or lower PEEP). These two protocols are not interchangeable, as described previously with two other protocols based on expiratory PL [20, 21].
The goal of increasing PEEP from baseline with a specific titration protocol using PL is to improve alveolar recruitment and oxygenation with limited hyperinflation and morbi-mortality [4]. ARDS patients have a great variation in lung damage with a gravitational vertical gradient of alveolar injuries determining dependent and non-dependent regions [22]. Pleural pressure also has a vertical gradient in the supine position [23]. Interestingly, PL,EL is close to direct measurement of pleural transpulmonary end-inspiratory pressure in the non-dependent lung region and might therefore limit the risk of ventilator-induced lung injury by over-distension [11]. Our main results demonstrate that this risk is probably reduced when PEEP is titrated according to Pplat (28–30 cmH2O) instead of a moderate positive PLexpi. (0–5 cmH2O).
Oxygenation and alveolar recruitment, according to PLexpi
In our study, when compared to baseline, the Express and PLexpi protocols increased PEEP, Vrec and oxygenation at the same time significantly. These two protocols based on respiratory mechanic proposed high PEEP level which might explain the small differences in recruitment. In the 2 EPVent trials, the difference in the results might come from the comparator group with empirical PEEP-FiO2 strategies, resulting in significantly different PLexpi on protocol between groups. PLexpi protocol proposed individually a different level of PEEP than Express protocol. The corresponding increase of PEEP with PLexpi protocol, when compared from baseline, was correlated to oxygenation. Patients with protective ventilation who respond to increased PEEP by improved oxygenation might have a lower risk of death [3]. The PLexpi protocol had a better alveolar recruitment than Express protocol with a median PLexpi of 3.6 cmH2O. Indeed, increasing PEEP in order to have a positive PLexpi of around 4.5 cmH2O can significantly reduce the risk of atelectasis in the dependent lung, whereas a PLexpi of 0 cmH2O might not be enough [23].
With the Express protocol, six patients had negative PLexpi, which meant that esophageal pressure was greater than airway pressure. This may occur when pressures in the thorax and abdomen are pathologically elevated [24]. These patients had lower respiratory system compliance and EELV. With the PLexpi protocol, these patients received higher levels of PEEP and a trend toward increased oxygenation. This protocol might improve alveolar recruitment and oxygenation, but over-distension has to be controlled at the same time [4].
Over-distension, according to PL,EL
In order to limit over-distension, Pplat should remain < 32 cmH2O and DPaw < 15 cmH2O [2, 25]. PL,EL can also help to limit over-distension in animal study based on CT scan [26], and a limit of 25 cmH2O has been proposed in ARDS patients [13]. We found that DPaw remained unchanged and within a safer range below 15 cmH2O with all protocols [25]. DPaw and DPL were highly correlated, and the mean difference was similar to previous data (approximately 4 cmH2O) [27]. The EPVent trial 2 proposed stopping the PEEP increase if PL,es was > 20 cmH2O [13]. With the PLexpi protocol, four patients had PL,es > 20 cmH2O, but seven patients had PL,EL > 25 cmH2O. This can be explained by a significant difference between these two assessments of end-inspiratory transpulmonary pressure as described previously [9, 23]. With the Express protocol, PEEP was titrated on the basis of Pplat, with the expectation that it closely approximated PL. In patients with normal chest wall elastance, Pplat is a reasonable surrogate for PL and with Express protocol, we found a median PL,EL of 20 cmH2O which is lower than the target/limit of 25 cmH2O proposed by Grasso et al. [11]; this limit was crossed by only one patient in our study.
With the PLexpi protocol, Vrec was slightly higher, but some patients had PL,EL > 25 cmH2O; these patients had lower respiratory system compliance with higher DP and respiratory acidosis. Thus, for some patients having both moderate positive PLexpi and limited PL,EL is not possible, and moderate positive PLexpi between 0–5 cmH2O does not prevent PL,EL being > 25 cmH2O. In these patients, PL,EL may need to be reduced and VT might be decreased below 6 ml kg−1 PBW in order to benefit from PEEP-induced alveolar recruitment with less distension and DP. The use of the prone position could also be interesting [28], but assessment of pleural pressure by esophageal pressure in prone position requires further investigations.
Elastance
Lung elastance in our patients was similar to previous studies [13, 26]. When chest wall elastance is high, Pplat may be much higher than PL. Indeed, a substantial portion of Pplat is dissipated in distending the chest wall. As the chest wall becomes stiffer, the proportion of Pplat that distends the lung (PL) decreases progressively [29]. Chest wall elastance was within the same range as other data in ARDS patients [12, 13]. In the study of Grasso et al., patients improved by PEEP had a higher chest wall elastance with a mean of 17 cmH2O/l. Chest wall elastance can be elevated in patients with acute respiratory failure for various reasons [30]. Increases in chest wall elastance can occur as a result of intra-abdominal hypertension, pleural effusion, massive ascites, thoracic trauma and edema of the intra-thoracic and intra-abdominal tissues as a result of fluid resuscitation [7]. We found a significant increase of Ecw with Plexpi protocol. This has already been described by Mezidi et al. and might be explained by an upward shift of the chest wall pressure–volume curve with more EELV [31].
Severity and complications
In our cohort, patients were severely ill with high Simplified Acute Physiology Scores II (SAPS II) and PaO2/FiO2 ratio at baseline was lower than in most studies including the EPVent trials [12, 13], Express study [5] and ARMA study [2]. Titration of PEEP was achieved with the PLexpi and Express protocols in all patients without hemodynamic impairment.
Limits and perspectives
There are several limitations. First of all, it is a small physiological study of moderate to severe ARDS patients with the risk of underpowered statistical analysis. We assessed alveolar recruitment and the risk of over-distension only with bedside respiratory mechanics and not with a CT scan that is the gold standard. This study did not assess alveolar inflammation, and we did not assess any outcome according to a specific PEEP titration protocol. The majority of the patients had a direct insult of the lung with pneumoniae or aspiration so it was not possible to compare respiratory mechanic between pulmonary and extrapulmonary ARDS [32]. All patients had a risk of derecruitment with a short ventilation with no PEEP between each protocol. Some of these limits need further investigations.
Conclusions
Our study demonstrates that, in comparison with a protocol using Pplat to titrate PEEP, a positive level of PLexpi with PEEP might slightly improve alveolar recruitment and oxygenation but also increases PL,EL above 25 cmH2O and the risk of over-distension in the dependent lung in one-third of patients.
Acknowledgements
The authors would like to thank Erwan Floch, PharmD (Newmed publishing services) and Pr Laurent Brochard for reviewing the manuscript.
Authors’ contributions
MB, NF, HR were involved in study concept and design; MB, NF, DTV, TS, AG, BF, HR contributed to acquisition of data; MB, NF, TB, HR were involved in analysis and interpretation of data; MB, HR, AO helped in drafting of the manuscript. All authors read and approved the final manuscript.
Funding
Support was provided solely from institutional sources.
Availability of data and materials
If requested.
Ethics approval and consent to participate
The study was approved by the French South-West Ethical Committee on November 2015 (CPP: 2015/113 by Chairperson Dr. Berdai), and written informed consent was obtained from all patients in accordance with current regulations.
Consent for publication
From all authors.
Competing interests
The authors declare that they have no competing interests.
Prior presentation
This study was presented in ESICM Lives Forum 2018 in Madrid as best abstract.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126–2136. doi: 10.1056/NEJMra1208707. [DOI] [PubMed] [Google Scholar]
- 2.Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med 2000;342:1301–8. [DOI] [PubMed]
- 3.Goligher EC, Kavanagh BP, Rubenfeld GD, Adhikari NKJ, Pinto R, Fan E, et al. Oxygenation response to positive end-expiratory pressure predicts mortality in acute respiratory distress syndrome. A secondary analysis of the LOVS and ExPress trials. Am J Respir Crit Care Med. 2014;190:70–76. doi: 10.1164/rccm.201404-0688OC. [DOI] [PubMed] [Google Scholar]
- 4.Rouby J-J, Brochard L. Tidal recruitment and overinflation in acute respiratory distress syndrome: yin and yang. Am J Respir Crit Care Med. 2007;175:104–106. doi: 10.1164/rccm.200610-1564ED. [DOI] [PubMed] [Google Scholar]
- 5.Mercat A, Richard J-CM, Vielle B, Jaber S, Osman D, Diehl J-L, et al. Expiratory Pressure (Express) Study Group: positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299:646–655. doi: 10.1001/jama.299.6.646. [DOI] [PubMed] [Google Scholar]
- 6.Akoumianaki E, Maggiore SM, Valenza F, Bellani G, Jubran A, Loring SH, et al. PLUG Working Group (Acute Respiratory Failure Section of the European Society of Intensive Care Medicine): the application of esophageal pressure measurement in patients with respiratory failure. Am J Respir Crit Care Med. 2014;189:520–531. doi: 10.1164/rccm.201312-2193CI. [DOI] [PubMed] [Google Scholar]
- 7.Mauri T, Yoshida T, Bellani G, Goligher EC, Carteaux G, Rittayamai N, et al. PLeUral pressure working Group (PLUG—Acute Respiratory Failure section of the European Society of Intensive Care Medicine): esophageal and transpulmonary pressure in the clinical setting: meaning, usefulness and perspectives. Intensive Care Med. 2016;42:1360–1373. doi: 10.1007/s00134-016-4400-x. [DOI] [PubMed] [Google Scholar]
- 8.Sahetya SK, Brower RG. The promises and problems of transpulmonary pressure measurements in acute respiratory distress syndrome. Curr Opin Crit Care. 2016;22:7–13. doi: 10.1097/MCC.0000000000000268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Talmor D, Sarge T, O’Donnell CR, Ritz R, Malhotra A, Lisbon A, et al. Esophageal and transpulmonary pressures in acute respiratory failure. Crit Care Med. 2006;34:1389–1394. doi: 10.1097/01.CCM.0000215515.49001.A2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chiumello D, Cressoni M, Colombo A, Babini G, Brioni M, Crimella F, et al. The assessment of transpulmonary pressure in mechanically ventilated ARDS patients. Intensive Care Med. 2014;40:1670–1678. doi: 10.1007/s00134-014-3415-4. [DOI] [PubMed] [Google Scholar]
- 11.Yoshida T, Amato MBP, Grieco DL, Chen L, Lima CAS, Roldan R, et al. Esophageal manometry and regional transpulmonary pressure in lung injury. Am J Respir Crit Care Med. 2018 doi: 10.1164/rccm.201709-1806oc. [DOI] [PubMed] [Google Scholar]
- 12.Talmor D, Sarge T, Malhotra A, O’Donnell CR, Ritz R, Lisbon A, et al. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med. 2008;359:2095–2104. doi: 10.1056/NEJMoa0708638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Beitler JR, Sarge T, Banner-Goodspeed VM, Gong MN, Cook D, Novack V, et al. EPVent-2 study group: effect of titrating positive end-expiratory pressure (PEEP) with an esophageal pressure-guided strategy vs an empirical high PEEP-Fio2 strategy on death and days free from mechanical ventilation among patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2019;321:846–857. doi: 10.1001/jama.2019.0555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Grasso S, Terragni P, Birocco A, Urbino R, Del Sorbo L, Filippini C, et al. ECMO criteria for influenza A (H1N1)-associated ARDS: role of transpulmonary pressure. Intensive Care Med. 2012;38:395–403. doi: 10.1007/s00134-012-2490-7. [DOI] [PubMed] [Google Scholar]
- 15.ARDS Definition Task Force. Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 16.Chiumello D, Gallazzi E, Marino A, Berto V, Mietto C, Cesana B, et al. A validation study of a new nasogastric polyfunctional catheter. Intensive Care Med. 2011;37:791–795. doi: 10.1007/s00134-011-2178-4. [DOI] [PubMed] [Google Scholar]
- 17.Chiumello D, Consonni D, Coppola S, Froio S, Crimella F, Colombo A. The occlusion tests and end-expiratory esophageal pressure: measurements and comparison in controlled and assisted ventilation. Ann Intensive Care. 2016;6:13. doi: 10.1186/s13613-016-0112-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gattinoni L, Chiumello D, Carlesso E, Valenza F. Bench-to-bedside review: chest wall elastance in acute lung injury/acute respiratory distress syndrome patients. Crit Care Lond Engl. 2004;8:350–355. doi: 10.1186/cc2854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dellamonica J, Lerolle N, Sargentini C, Beduneau G, Di Marco F, Mercat A, et al. PEEP-induced changes in lung volume in acute respiratory distress syndrome. Two methods to estimate alveolar recruitment. Intensive Care Med. 2011;37:1595–1604. doi: 10.1007/s00134-011-2333-y. [DOI] [PubMed] [Google Scholar]
- 20.Gulati G, Novero A, Loring SH, Talmor D. Pleural pressure and optimal positive end-expiratory pressure based on esophageal pressure versus chest wall elastance: incompatible results*. Crit Care Med. 2013;41:1951–1957. doi: 10.1097/CCM.0b013e31828a3de5. [DOI] [PubMed] [Google Scholar]
- 21.Chiumello D, Cressoni M, Carlesso E, Caspani ML, Marino A, Gallazzi E, et al. Bedside selection of positive end-expiratory pressure in mild, moderate, and severe acute respiratory distress syndrome. Crit Care Med. 2014;42:252–264. doi: 10.1097/CCM.0b013e3182a6384f. [DOI] [PubMed] [Google Scholar]
- 22.Pelosi P, D’Andrea L, Vitale G, Pesenti A, Gattinoni L. Vertical gradient of regional lung inflation in adult respiratory distress syndrome. Am J Respir Crit Care Med. 1994;149:8–13. doi: 10.1164/ajrccm.149.1.8111603. [DOI] [PubMed] [Google Scholar]
- 23.Cherniack RM, Farhi LE, Armstrong BW, Proctor DF. A comparison of esophageal and intrapleural pressure in man. J Appl Physiol. 1955;8:203–211. doi: 10.1152/jappl.1955.8.2.203. [DOI] [PubMed] [Google Scholar]
- 24.Loring SH, O’Donnell CR, Behazin N, Malhotra A, Sarge T, Ritz R, et al. Esophageal pressures in acute lung injury: do they represent artifact or useful information about transpulmonary pressure, chest wall mechanics, and lung stress? J Appl Physiol Bethesda Md. 1985;2010(108):515–522. doi: 10.1152/japplphysiol.00835.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Amato MBP, Meade MO, Slutsky AS, Brochard L, Costa ELV, Schoenfeld DA, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372:747–755. doi: 10.1056/NEJMsa1410639. [DOI] [PubMed] [Google Scholar]
- 26.Staffieri F, Stripoli T, De Monte V, Crovace A, Sacchi M, De Michele M, et al. Physiological effects of an open lung ventilatory strategy titrated on elastance-derived end-inspiratory transpulmonary pressure: study in a pig model*. Crit Care Med. 2012;40:2124–2131. doi: 10.1097/CCM.0b013e31824e1b65. [DOI] [PubMed] [Google Scholar]
- 27.Baedorf Kassis E, Loring SH, Talmor D. Mortality and pulmonary mechanics in relation to respiratory system and transpulmonary driving pressures in ARDS. Intensive Care Med. 2016;42:1206–1213. doi: 10.1007/s00134-016-4403-7. [DOI] [PubMed] [Google Scholar]
- 28.Richard J-CM, Marini JJ. Transpulmonary pressure as a surrogate of plateau pressure for lung protective strategy: not perfect but more physiologic. Intensive Care Med. 2012;38:339–341. doi: 10.1007/s00134-012-2492-5. [DOI] [PubMed] [Google Scholar]
- 29.Riad Z, Mezidi M, Subtil F, Louis B, Guérin C. Short-term effects of the prone positioning maneuver on lung and chest wall mechanics in ARDS patients. Am J Respir Crit Care Med. 2017 doi: 10.1164/rccm.201709-1853le. [DOI] [PubMed] [Google Scholar]
- 30.Jardin F, Genevray B, Brun-Ney D, Bourdarias JP. Influence of lung and chest wall compliances on transmission of airway pressure to the pleural space in critically ill patients. Chest. 1985;88:653–658. doi: 10.1378/chest.88.5.653. [DOI] [PubMed] [Google Scholar]
- 31.Mezidi M, Parrilla FJ, Yonis H, Riad Z, Böhm SH, Waldmann AD, Richard JC, Lissonde F, Tapponnier R, Baboi L, Mancebo J, Guérin C. Effects of positive end-expiratory pressure strategy in supine and prone position on lung and chest wall mechanics in acute respiratory distress syndrome. Ann Intensive Care. 2018;8:86. doi: 10.1186/s13613-018-0434-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Coppola S, Froio S, Marino A, Brioni M, Cesana BM, Cressoni M, Gattinoni L, Chiumello D. Respiratory Mechanics, Lung Recruitability, and Gas Exchange in Pulmonary and Extrapulmonary Acute Respiratory Distress Syndrome. Crit Care Med. 2019;47:792–799. doi: 10.1097/CCM.0000000000003715. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
If requested.


