Abstract
Background
Cancer has been considered a serious global health problem and a leading cause of morbidity and mortality worldwide. Despite recent advances in cancer therapy, treatments of advance stage cancers are mostly ineffective resulting in poor survival of patients. Recent evidences suggest that multipotent human mesenchymal stem cells (hMSCs) play important roles in growth and metastasis of several cancers by enhancing their engraftment and inducing tumor neovascularization. However, the effect of hMSCs on cancer cells is still controversial because there are also evidences demonstrating that hMSCs inhibited growth and metastasis of some cancers.
Methods
In this study, we investigated the effects of bioactive molecules released from bone marrow and gestational tissue-derived hMSCs on the proliferation of various human cancer cells, including C3A, HT29, A549, Saos-2, and U251. We also characterized the hMSC-derived factors that inhibit cancer cell proliferation by protein fractionation and mass spectrometry analysis.
Results
We herein make a direct comparison and show that the effects of hMSCs on cancer cell proliferation and migration depend on both hMSC sources and cancer cell types and cancer-derived bioactive molecules did not affect the cancer suppressive capacity of hMSCs. Moreover, hMSCs use distinct combination of bioactive molecules to suppress the proliferation of human hepatoblastoma and colorectal cancer cells. Using protein fractionation and mass spectrometry analysis, we have identified several novel hMSC-derived factors that might be able to suppress cancer cell proliferation.
Conclusion
We believe that the procedure developed in this study could be used to discover other therapeutically useful molecules released by various hMSC sources for a future in vivo study.
1. Background
Cancer has been considered a serious global health problem and a leading cause of morbidity and mortality worldwide. While an early diagnosed cancer can be cured by surgery or radiotherapy, patients with an advance stage of cancer can only be treated with chemotherapeutic agents or immunotherapy. Despite significant improvements during the past decades, the effectiveness of those treatments, especially in patients with solid tumors, is limited resulting in the poor survival rate of those patients.
Several recent evidences suggest that human mesenchymal stem cells (hMSCs) play important roles in growth and metastasis of various cancer cells and affect their responses to chemotherapeutic agents. hMSCs are multipotent stem/progenitor cells that exist in various tissues, such as bone marrow, adipose tissue, umbilical cord, placenta, and chorion [1–5]. Due to their ability to produce and release bioactive molecules that have various therapeutic potentials, hMSCs have been considered potential cell sources for many clinical applications [6, 7].
Previous studies show that MSCs enhanced the engraftment rate of breast cancer, ovarian cancer, melanoma, glioma, and colon cancer cells in animal models. Some studies also demonstrate that MSCs could migrate from circulation into cancer tissues and become cancer-associated fibroblasts (CAFs) and pericytes. Those MSC-derived CAFs and pericytes then released several proangiogenic factors that induce tumor neovascularization leading to the rapid tumor growth and metastasis [8–16]. The immunomodulatory property of MSCs is also believed to promote tumor growth by reducing immune reaction against tumor cells [17].
Despite those evidences, there are other studies demonstrating that MSCs inhibited growth and metastasis of several cancers, including colon cancer, hepatoma, and melanoma [18–20]. Those conflicting results possibly arise from the variability of both MSC sources and cancer cell types used in those studies.
Although bone marrow-derived hMSCs (BM-hMSCs) have been the standard source of hMSCs for most research and clinical applications, their harvest requires an invasive procedure and their number declines with age [21, 22]. Therefore, gestational tissue-derived hMSCs which can easily be obtained in large quantity by a non-invasive procedure have been considered more suitable sources of hMSCs for clinical applications. However, types of bioactive molecules that are released from gestational tissue-derived hMSCs and their effects on the properties of cancer cells have yet to be characterized. Therefore, the present study is aimed at comparing the effects of several gestational tissue-derived hMSCs, including placenta-derived hMSCs (hAMSCs), chorion-derived hMSCs (CH-hMSCs), and umbilical cord-derived hMSCs (UC-hMSCs), on the proliferative capacity of five distinct human cancer cells (hepatoblastoma cell C3A, colon adenocarcinoma cell HT29, lung adenocarcinoma cell A549, osteosarcoma cell Saos2, and glioma cell U251) with that of bone marrow-derived hMSCs (BM-hMSCs) using an in vitro model. We also identified the hMSC-derived factors that inhibit cancer cell proliferation by using protein fractionation and mass spectrometry analysis.
2. Methods
2.1. Subjects
This study was approved by the ethical committee for human researches, Faculty of Medicine, Thammasat University, which was in accordance with the Declaration of Helsinki, the Belmont Report, and ICH-GCP. Human bone marrow samples were obtained from three healthy volunteers. The human gestational tissues of normal pregnancies (umbilical cord, placenta, and chorion) were obtained from three healthy women after labor. All donors gave written informed consent.
2.2. Isolation and Culture of hMSCs
hMSCs were isolated and cultured as described in our previous study [23]. Bone marrow-derived hMSCs were isolated using Ficoll-Hypaque (Robbins Scientific Corporation, USA) density gradient centrifugation and cultured in Dulbecco's modified Eagle medium (DMEM) (GIBCO™, Invitrogen Corporation, USA) supplemented with 10% (v/v) fetal bovine serum (FBS) (Lonza, USA), 100 U/ml penicillin, and 100 μg/ml streptomycin. The cell suspensions at a density of 2 × 105 cells/cm2 were cultured in a 25 cm2 culture flask (Corning, USA). For gestational tissue-derived hMSC isolation, umbilical cord, fetal side of the placenta, and chorion (chorionic membrane) were manually separated and cut into small pieces and incubated with 0.25% (w/v) trypsin-EDTA (GIBCO™, Invitrogen Corporation, USA) for 30 minutes at 37°C. The pieces were washed twice with PBS, resuspended in DMEM+10% (v/v) FBS and cultured in a 25 cm2 culture flask (Corning, USA). Cells were cultured at 37°C, and the medium was changed every 3 days. Cells were passaged when they reached 80% confluence. The morphological features of hMSCs were observed and photographed under an inverted microscope (Nikon Eclipse Ts2R, Japan).
2.3. Culture of Human Cancer Cells
Hepatoblastoma cell C3A, osteosarcoma cell Saos2, lung adenocarcinoma cell A549, and glioma cell U251 were cultured in DMEM (GIBCO™, Invitrogen Corporation, USA) supplemented with 10% (v/v) fetal bovine serum (FBS) (Lonza, USA) while colon adenocarcinoma cell HT29 was cultured in DMEM/F12 supplemented with 10% (v/v) FBS (Lonza, USA). Cells were cultured at 37°C, and the medium was changed every 3 days. Cells were passaged when they reached 80% confluence.
2.4. Characterization of Cultured hMSCs by Flow Cytometry
The cells were harvested and immediately processed for flow cytometric analysis as described in our previous study [23]. Briefly, hMSCs (passages 3rd-5th) were washed twice with PBS. 4 × 105 hMSCs were then resuspended in 50 μl PBS, incubated with 10 μl fluorochrome-labeled mouse anti-human monoclonal antibodies: anti-CD45-FITC (BD Pharmingen, USA), anti-CD34-PE (Biolegend, USA), anti-CD90-FITC (AbD Serotec, USA), anti-CD73-PE (BD Pharmingen, USA), and anti-CD105-PE (Miltenyi Biotec, Germany) for 30 minutes at 4°C in the dark. After being incubated with the antibodies, cell pellets were washed twice with PBS and fixed with 1% (w/v) paraformaldehyde in PBS. Flow cytometry was performed by FACSCalibur™ Flow cytometer using CellQuest™ software (Becton Dickinson, USA).
2.5. Osteogenic and Adipogenic Differentiation of hMSCs
hMSCs (passages 3rd-5th) were used to assess their adipogenic and osteogenic differentiation potentials as described in our previous study [23]. For adipogenic differentiation, 5 × 104 hMSCs were cultured in NH AdipoDiff® Medium (Miltenyi Biotec, Germany). The medium was changed every 3 days. After 3 weeks of growth in adipogenic medium, the cells were washed twice with PBS, fixed with 10% formaldehyde for 15 minutes, and rinsed with distilled water. Then, the cells were incubated with Oil Red O solution [0.5% (w/v) in isopropanol] for 20 minutes at room temperature and observed under an inverted microscope (Nikon Eclipse Ts2R, Japan).
For osteogenic differentiation, 5 × 104 hMSCs were cultured in NH OsteoDiff® Medium (Miltenyi Biotec, Germany). The medium was changed every 3 days. After 3 weeks of growth in osteogenic medium, the cells were fixed with 4% paraformaldehyde, incubated with 40 mM Alizarin Red S solution (Sigma Aldrich, USA) for 20 minutes at room temperature, and observed under an inverted microscope (Nikon Eclipse Ts2R, Japan).
2.6. Preparation and Fractionation of hMSC-Conditioned Media
hMSC-conditioned media were prepared as described in our previous study [23]. 7 × 105 hMSCs (passages 3rd-5th) were cultured in DMEM+10% FBS for 24 hours. Then, the cells were washed twice with 10 ml sterile PBS and incubated with 15 ml serum-free medium (SFM) which is MesenCult™-ACF Plus Medium (STEMCELL technologies, USA) for further 24 hours. To prepare cancer-associated hMSC-conditioned media, 5 × 104 hMSCs were co-cultured with 5 × 104 human cancer cells using a transwell culture system for 7 days. After co-culture, the transwell inserts containing human cancer cells were removed; the hMSCs were washed twice with 5 ml sterile PBS and incubated with 600 μl SFM for further 24 hours.
After incubation, the conditioned media were collected, centrifuged at 2000 rpm for 10 minutes at 4°C, and filtered through a 0.45 μm syringe filter (Corning, USA). The filtered conditioned medium was then fractionated into 5 distinct fractions according to the molecular weight of their protein composition using ultraspin columns with molecular weight cutoff (MWCO) at 100 kDa (Pall Corporation, USA), 50 kDa (Merck, Germany), 30 kDa (Pall Corporation, USA), and 10 kDa (Pall Corporation, USA) as described in our previous study [23]. To fractionate hMSC-conditioned medium, the medium was transferred to 100 kDa ultraspin columns and centrifuged at 4500 rpm for 15 minutes at 4°C. After centrifugation, the fraction of hMSC-conditioned medium retained in the column was collected while the flow through was transferred to the 50 kDa ultraspin columns for further centrifugation. By repeating this procedure with the 30 kDa and 10 kDa columns, the hMSC-conditioned medium was successfully fractionated into 100 kDa, 50 kDa, 30 kDa, 10 kDa, and less than 10 kDa (<10 kDa) fractions.
2.7. Effects of hMSC-Derived Bioactive Molecules on the Proliferation and Migration of Cancer Cells
For proliferation assay, the number of cancer cells was monitored by the real-time cell analysis (RTCA) of a cell culture system as described in our previous study [23]. Briefly, 2.5 × 104 cancer cells were added into an individual well of culture E-Plate (Roche Applied Science, USA) containing 100 μl normal or cancer-associated hMSC-conditioned media. The E-Plates were incubated at 37°C and monitored on the RTCA system by the xCELLigence real-time cell analyzer (Roche Applied Science, USA) at 5-minute time intervals for 7 days. The number of cancer cells in each well was continuously monitored throughout the entire culture period and reported as the cell index. Cancer cells cultured in DMEM+10% FBS served as controls.
For migration assay, hMSC-conditioned media were used to induce migration of hepatoblastoma C3A cells through 8 μm transwell (Costar, Corning, USA) as described in our previous study [24]. 1 × 106 C3A cells were seeded in the transwell inserts which were placed into wells of the 24-well plate (Costar, Corning, USA) containing 600 μl of various hMSC-conditioned media. After 6 hours of culture, the numbers of C3A cells that migrate to the other side of the transwell's membrane were determined by hematoxylin staining. C3A cells cultured in C3A-conditioned medium served as controls.
2.8. Migratory Ability of hMSCs toward Cancer Cells
Cell migration ability was assessed using a 24-well transwell chamber (Corning, USA) with a 8 μm pore polyester membrane insert as described in our previous study [23, 24]. Briefly, 5 × 104 C3A or HT29 cells were plated into the lower chamber of transwell containing 600 μl DMEM+10% FBS and incubated at 37°C for 24 hours to allow cell attachment. After that, the medium was changed to 600 μl DMEM supplemented with 2% (v/v) FBS and the cells were cultured for further 24 hours. The following day, 4 × 104 hMSCs were seeded into the upper chamber of the transwell inserts and cultured for further 6 hours. At the end of culture, cells that migrate to the other side of the transwell membrane were stained with hematoxylin and counted from at least ten randomly selected microscopic fields. The hMSCs cultured in transwell whose lower chamber contained cell-free DMEM+2% FBS served as controls.
2.9. Mass Spectrometry Analysis and Protein Identification
Mass spectrometry analysis was performed as described in our previous study [24]. The proteins were digested by trypsin and analyzed by ESI ion trap mass spectrometry. Identification and quantification of each protein were performed by DeCyderMS differential analysis software 2.0 (GE Healthcare, USA) and MASCOT search engine software (Matrix Science, UK) based on NCBInr human protein databases. The identified proteins were then categorized into non-secretory, classical secretory, and non-classical secretory proteins by SignalP and SecretomeP software (Center for Biological Sequence Analysis; CBS, DK). Finally, the secretory proteins were further categorized by PANTHER and UniPortKB software into separated groups according to their functions.
2.10. Statistical Analysis
Data were presented as the mean ± standard error of the mean (SEM). The Mann-Whitney U test was used to assess the significance of differences between observed data. P < 0.05 was considered to be statistically significant.
3. Results
3.1. Characteristics of hMSCs Derived from Bone Marrow and Gestational Tissues
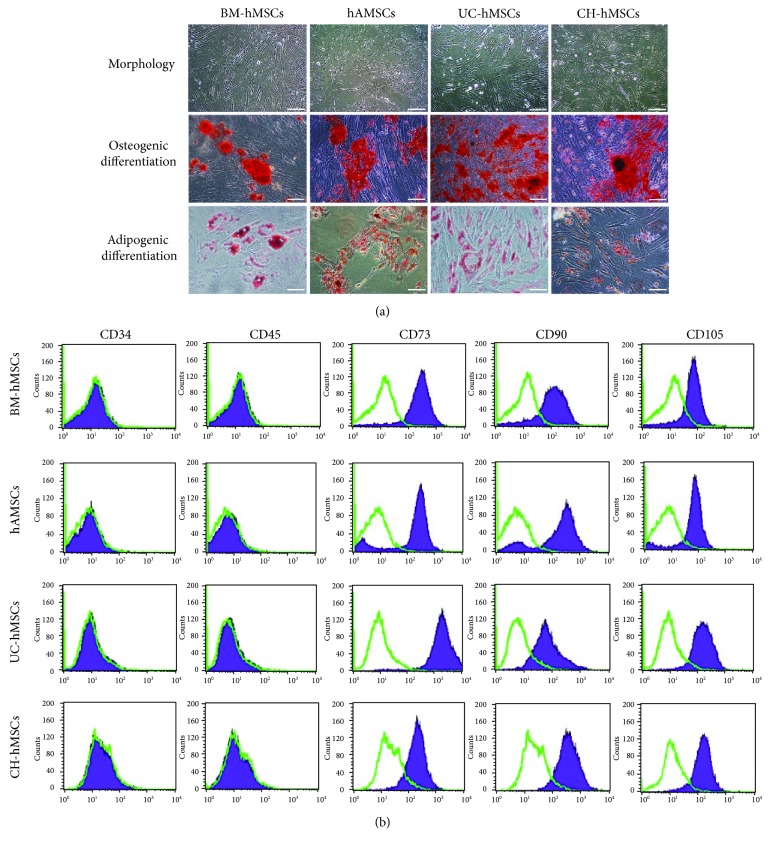
hMSCs derived from gestational tissues, including the fetal side of the placenta (hAMSCs), umbilical cord (UC-hMSCs), and chorion (CH-hMSCs), exhibited similar characteristics to those of bone marrow-derived hMSCs (BM-hMSCs). The gestational tissue-derived hMSCs displayed fibroblast-like morphology and could differentiate to adipocytes and osteocytes as demonstrated by Oil-Red O and Alizarin Red S staining, respectively (Figure 1(a)). Moreover, the gestational tissue-derived hMSCs also expressed typical hMSC surface markers (positive for CD73, CD90, and CD105 and negative for hematopoietic markers CD34 and CD45; Figure 1(b)).
Figure 1.
Characteristics of bone marrow- and gestational tissue-derived hMSCs. (a) Morphology, adipogenic, and osteogenic differentiation of bone marrow- and gestational tissue-derived hMSCs (scale bar = 200 μm). (b) Immunophenotypes of bone marrow- and gestational tissue-derived hMSCs as determined by flow cytometry.
3.2. Effect of hMSC-Derived Bioactive Molecules on Cancer Cell Proliferation
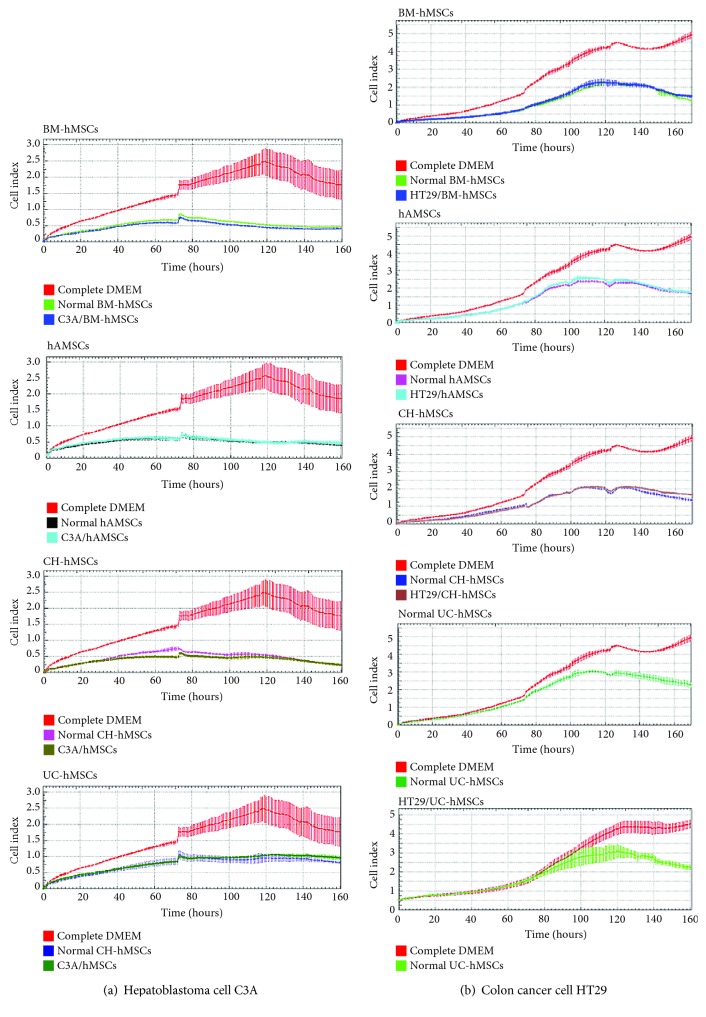
To study the effect of hMSC-derived bioactive molecules on cancer cell proliferation, the conditioned media derived from various hMSC sources were used to culture 5 human cancer cell lines, C3A, HT29, Saos2, A549, and U251. The condition media derived from all normal hMSC sources significantly reduced the proliferation of both hepatoblastoma cell C3A and colon adenocarcinoma cell HT29 (Figures 2(a) and 2(b)). The inhibitory effect of bone marrow- and gestational tissue-derived hMSCs on C3A and HT29 cell proliferation was observed at approximately 10 hours after culture and was maintained for the entire culture period (Figures 2(a) and 2(b)).
Figure 2.
Effects of hMSC-derived bioactive molecules on the proliferation of C3A and HT29 cell. Graphs show the growth kinetics of hepatoblastoma cell C3A (a) and colon adenocarcinoma cell HT29 (b), which were cultured in conditioned medium derived from various normal and cancer-associated hMSC-conditioned media for 7 days. The growth kinetics of cancer cells were determined by the xCELLigence real-time cell analyzer and were reported as the cell index against culture time. C3A cells cultured in DMEM supplemented with 10% (v/v) FBS and HT29 cells cultured in DMEM/F12 supplemented with 10% (v/v) FBS serve as controls. Data are presented as the mean ± SEM of three independent experiments.
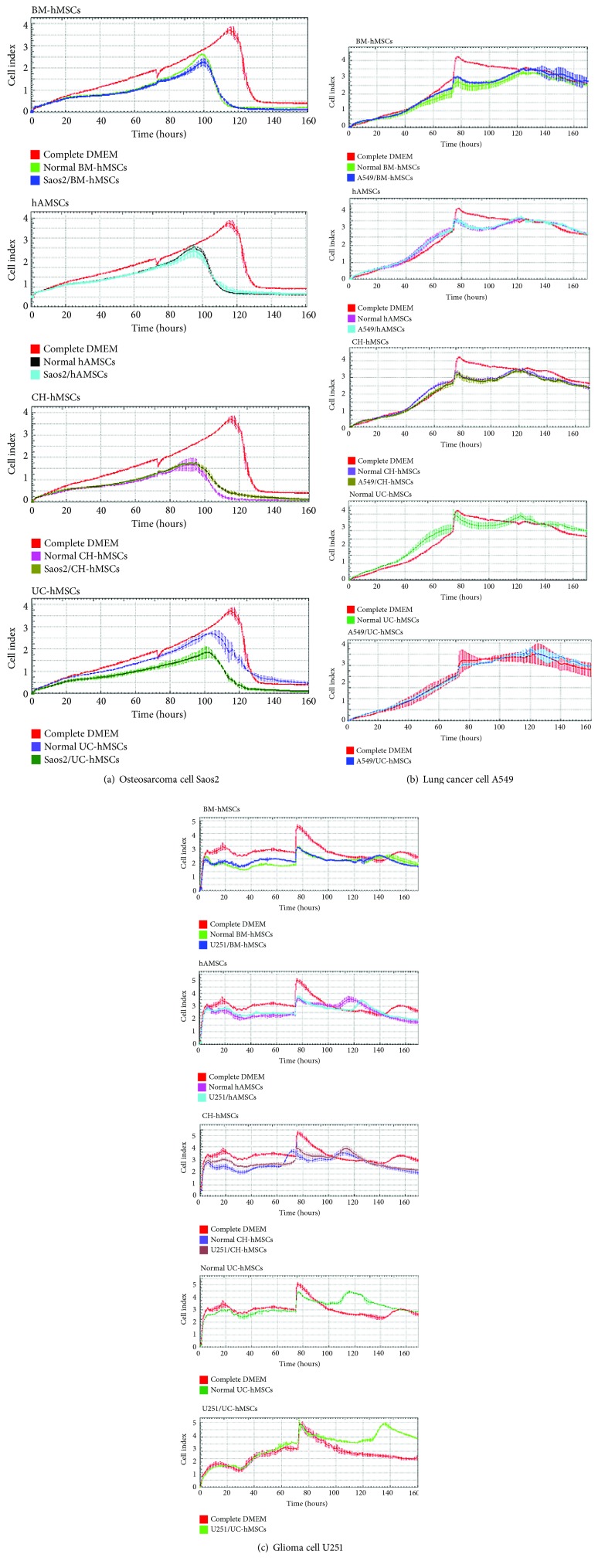
Although bone marrow- and gestational tissue-derived hMSCs decreased the proliferation of osteosarcoma cell Saos2, their inhibitory effects on Saos2 cells appeared to be lesser in degree and later in onset compared with their effects on C3A and HT29 cells (Figure 3(a)). In contrast to C3A, HT29, and Saos2 cells, all hMSCs failed to inhibit the proliferation of lung adenocarcinoma cell A549 and glioma cell U251 (Figures 3(b) and 3(c)).
Figure 3.
Effects of hMSC-derived bioactive molecules on the proliferation of Saos2, A549, and U251 cells. Graphs show the growth kinetics of osteosarcoma cell Saos2 (a), lung adenocarcinoma cell A549 (b), and glioma cell U251 (c), which were cultured in conditioned medium derived from various normal and cancer-associated hMSC-conditioned media for 7 days. The growth kinetics of cancer cells were determined by the xCELLigence real-time cell analyzer and were reported as the cell index against culture time. Saos2, A549 and U251 cells cultured in DMEM supplemented with 10% (v/v) FBS serve as controls. Data are presented as the mean ± SEM of three independent experiments.
To determine whether cancer-derived bioactive molecules can change the cancer suppressive ability of hMSCs, bone marrow- and gestational tissue-derived hMSCs were co-cultured with various cancer cells using transwell culture system for 7 days before the conditioned media derived from those hMSCs (called “cancer-associated hMSC-conditioned media”) were collected and used for cancer cell proliferation assay. The results showed that the cancer-associated hMSC-conditioned media exhibited the same levels of cancer suppressive capacity compared with their normal hMSC counterparts (Figures 2 and 3).
Those results demonstrate that the bioactive molecules secreted from bone marrow- and gestational tissue-derived hMSCs exerted different effects on distinct human cancer cells and cancer-derived bioactive molecules did not affect the cancer suppressive capacity of hMSCs. According to this, only C3A and HT29, whose proliferation was clearly suppressed by hMSC-derived bioactive molecules, were chosen for the further study.
3.3. Effect of Fractionated hMSC Secretome on C3A and HT29 Cell Proliferation
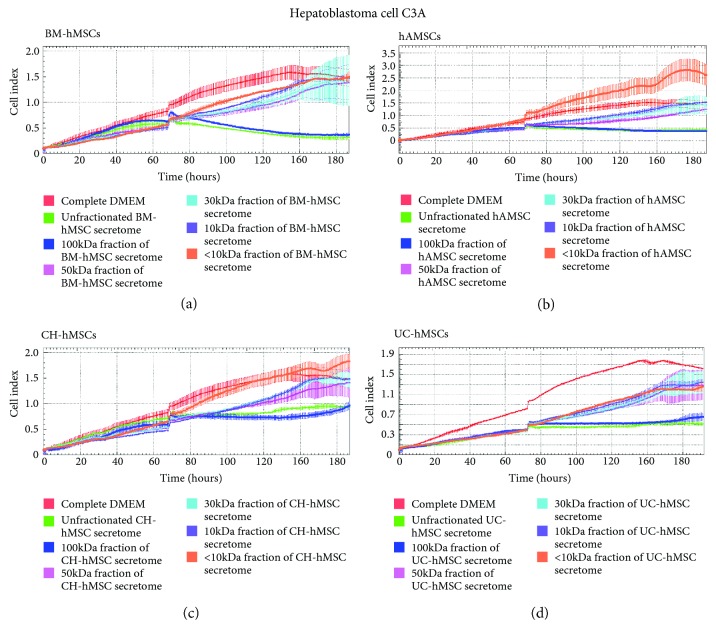
To further characterize the hMSC-derived factors that inhibited C3A and HT29 cell proliferation, the conditioned media derived from normal BM-hMSCs, hAMSCs, CH-hMSCs, and UC-hMSCs were fractionated to 5 distinct fractions according to the molecular weight of their protein compositions. The effects of each hMSC-conditioned media fraction on C3A and HT29 cell proliferation were then determined and compared with their unfractionated hMSC-conditioned media.
The results showed that only the 100 kDa fraction of BM-hMSC-, hAMSC-, CH-hMSC-, and UC-hMSC-conditioned media reduced the proliferation of C3A cells to the same levels as those cultured in the unfractionated hMSC-conditioned media (Figure 4(a)–(d)). Although the 50 kDa, 30 kDa, and 10 kDa fraction of BM-hMSC-, hAMSC-, CH-hMSC-, and UC-hMSC-conditioned media also reduced the proliferation of C3A cells in comparison to controls; their inhibitory effects were much lesser than the 100 kDa fraction and unfractionated hMSC-condition media (Figure 4(a)–(d)).
Figure 4.
Effects of various fractions of hMSC secretomes on the proliferation of C3A cells. Graphs show the growth kinetics of hepatoblastoma cell C3A which were cultured in the 100 kDa, 50 kDa, 30 kDa, 10 kDa, and less than 10 kDa (<10 kDa) fraction of the conditioned media derived from BM-hMSCs (a), hAMSCs (b), CH-hMSCs (c), and UC-hMSCs (d) for 7 days. The growth kinetics of C3A cells were determined by the xCELLigence real-time cell analyzer and were reported as the cell index against culture time. C3A cells cultured in DMEM supplemented with 10% (v/v) FBS serve as negative controls while C3A cells cultured in unfractionated hMSC-conditioned media serve as positive controls. Data are presented as the mean ± SEM of three independent experiments.
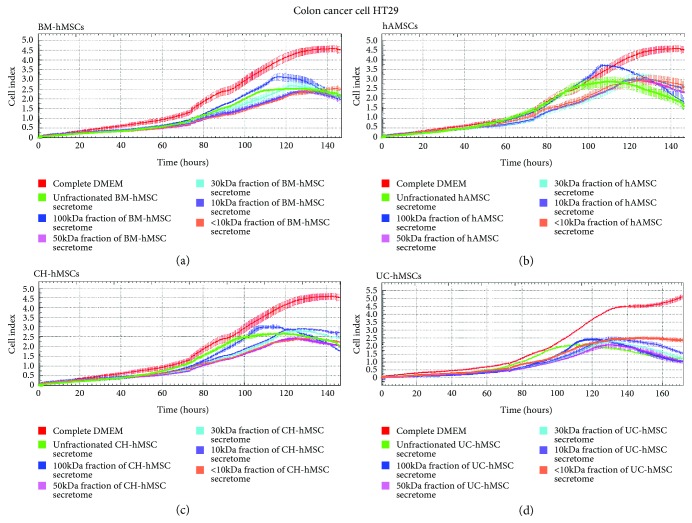
While the inhibitory effects of the 100 kDa fraction of BM-hMSC-, hAMSC-, CH-hMSC-, and UC-hMSC-conditioned media on C3A cell proliferation were greatest (Figure 4(a)–(d)), their ability to suppress HT29 cell proliferation were the lowest in comparison to other fractions (Figure 5(a)–(d)). It appeared that the ability to suppress HT29 cell proliferation of 50 kDa, 30 kDa, 10 kDa, and <10 kDa fraction of BM-hMSC-, hAMSC-, CH-hMSC-, and UC-hMSC-conditioned media was greater than the 100 kDa fraction (Figure 5(a)–(d)). The results clearly indicate that the hMSC-derived factors which inhibit C3A cell proliferation were distinct from those that inhibit HT29 cell proliferation.
Figure 5.
Effects of various fractions of hMSC secretomes on the proliferation of HT29 cells. Graphs show the growth kinetics of colon adenocarcinoma cell HT29 which were cultured in the 100 kDa, 50 kDa, 30 kDa, 10 kDa, and less than 10 kDa (<10 kDa) fraction of the conditioned media derived from BM-hMSCs (a), hAMSCs (b), CH-hMSCs (c), and UC-hMSCs (d) for 7 days. The growth kinetics of HT29 cells were determined by the xCELLigence real-time cell analyzer and were reported as the cell index against culture time. HT29 cells cultured in DMEM/F12 supplemented with 10% (v/v) FBS serve as negative controls while HT29 cells cultured in unfractionated hMSC-conditioned media serve as positive controls. Data are presented as the mean ± SEM of three independent experiments.
3.4. Effect of hMSC-Derived Bioactive Molecules on C3A Cell Migration
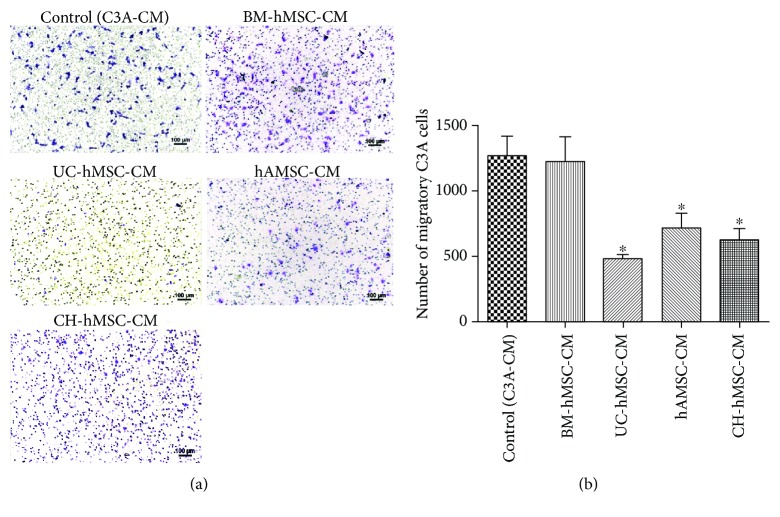
To study the effect of hMSC-derived bioactive molecules on C3A cell migration, the conditioned media derived from various hMSC sources were used to induce C3A cell migration through 8 μm transwells. Interestingly, the bioactive molecules secreted from UC-hMSCs, hAMSCs, and CH-hMSCs induced a significantly lower number of migratory C3A cells (471 ± 31 cells, 709 ± 123 cells, and 632 ± 83 cells, respectively) in comparison to controls (1268 ± 143 cells, P < 0.05) (Figures 6(a) and 6(b)). In contrast, the number of migratory C3A cells that were induced by bioactive molecules released from BM-hMSCs (1227 ± 174 cells) was not different from controls (1268 ± 143 cells) (Figures 6(a) and 6(b)). The results suggest that while the bioactive molecules secreted from UC-hMSCs, hAMSCs, and CH-hMSCs suppressed C3A cell migration, the BM-hMSC-derived bioactive molecules did not have this effect.
Figure 6.
The effect of hMSC-derived bioactive molecules on C3A cell migration. (a) C3A cells which migrated to the other side of transwell membrane after being induced by various hMSC-conditioned media as determined by hematoxylin staining. Scale bar: 100 μm. C3A cells cultured with C3A-conditioned medium serve as controls. (b) Graph shows the number of migratory C3A cells after induction with various hMSC-conditioned media. Data are presented as the mean ± SEM of three independent experiments. One-way ANOVA was used to assess the significance of differences between observed data. ∗ P < 0.05 vs. controls.
3.5. Migratory Ability of hMSCs toward C3A and HT29 Cells
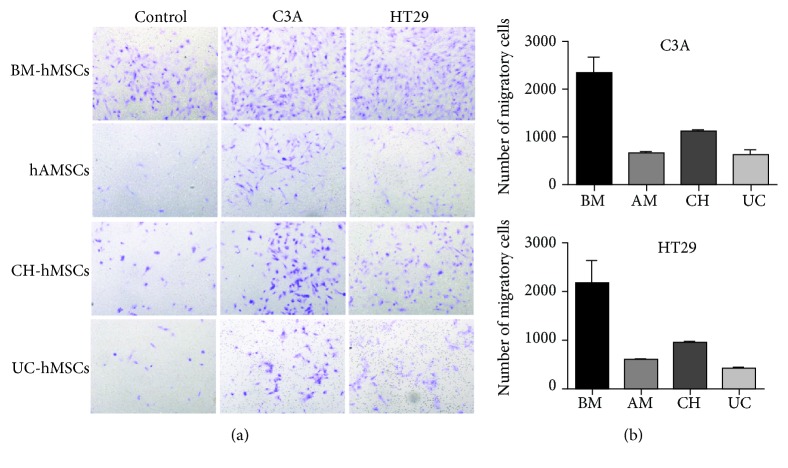
To study the migratory ability of bone marrow- and gestational tissue-derived hMSCs toward C3A and HT29 cells, BM-hMSCs, hAMSCs, CH-hMSCs, and UC-hMSCs were co-cultured with C3A or HT29 cells using the transwell culture system. The numbers of hMSCs that migrated toward C3A or HT29 were then determined after 6 hours of co-culture.
The results showed that distinct sources of hMSCs exhibited different migratory abilities toward C3A and HT29 cells. BM-hMSCs have greater migratory ability toward C3A cells (2311 ± 348 cells) than CH-hMSCs (1124 ± 28 cells), hAMSCs (660 ± 21 cells), and UC-hMSCs (625 ± 81 cells) (Figures 7(a) and 7(b)). Similar to C3A, BM-hMSCs also exhibited greater migratory ability toward HT29 cells (2161 ± 434 cells) than CH-hMSCs (946.5 ± 41 cells), hAMSCs (592 ± 34 cells), and UC-hMSCs (415 ± 4 cells) (Figures 7(a) and 7(b)).
Figure 7.
Migratory ability of hMSCs toward C3A and HT29 cells. (a) Hematoxylin-stained hMSCs which migrated to the other side of transwell membrane in response to bioactive molecules released from C3A and HT29 cells. hMSCs cultured in transwells containing DMEM supplemented with 2% (v/v) FBS serve as controls. (b) The number of hMSCs which migrated in response to bioactive molecules released from C3A and HT29 cells. Data are presented as the mean ± SEM of three independent experiments.
3.6. Identification of Proteins Presented in the 100 kDa Fraction of hAMSC-Conditioned Medium
To identify hMSC-derived factors that were able to suppress C3A cell proliferation, we chose the 100 kDa fraction of hAMSC-conditioned medium, which was shown to suppress the proliferation of C3A cells, for mass spectrometry analysis.
We chose hAMSC-conditioned medium for further analysis based on two reasons. Firstly, despite BM-hMSCs exhibiting similar levels of cancer suppressive effect, their proliferative capacity in culture is lesser than hAMSCs and could not be expanded for more than 10 passages. Because the preparation of hMSC-derived bioactive factors for future clinical applications required a large number of cultured hMSCs, we believe that hAMSCs which can be isolated from placental tissues in large quantity and can be expanded for a longer period of time than BM-hMSCs will be more suitable for this purpose. Secondly, our previous study demonstrates that hAMSCs release other therapeutically useful factors that induce migration of endothelial progenitor cells which play important roles in tissue neovascularization while other gestational tissue-derived hMSCs, such as UC-hMSCs, did not [23, 24].
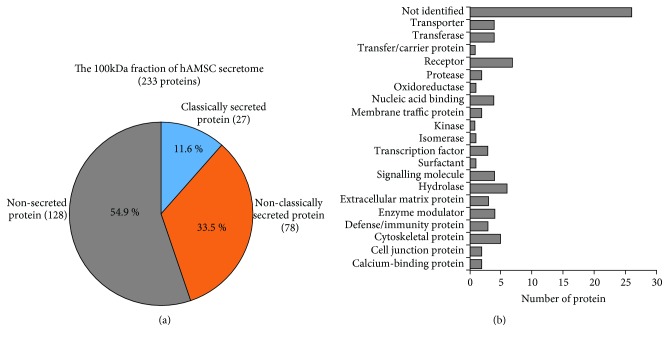
The results showed that the 100 kDa fraction of hAMSC-conditioned medium consisted of 233 proteins, of which 128 were non-secreted proteins, 78 were non-classically secreted proteins, and 27 were classically secreted proteins (Figure 8(a)). To identify hMSC-derived bioactive molecules that suppress C3A cell proliferation, 128 non-secreted proteins that might be released from dead hAMSCs during the preparation of the conditioned medium were excluded while the rest of the proteins were categorized based on their functions (Figure 8(b)). Lists of bioactive molecules identified in the 100 kDa fraction of hAMSC secretome are provided in supplementary table 1.
Figure 8.
Characterization of the 100 kDa fraction of hAMSC secretome. (a) Pie chart illustrates 233 proteins presented in the 100 kDa fraction of hAMSC secretome as being categorized into non-secreted protein, classically secreted protein, and non-classically secreted protein. (b) Graph demonstrates secretory proteins presented in the 100 kDa fraction of hAMSC secretome as being categorized into groups according to their functions.
4. Discussion
hMSCs have been considered the promising sources for cell therapy. The therapeutic potentials of hMSCs depend largely on their ability to secrete a broad range of bioactive molecules that affect various processes, such as immune response, cell proliferation, and neovascularization [25]. This is in agreement with our pervious study which demonstrates that the hAMSCs and BM-hMSCs release proangiogenic factors, such as PDGF-β, IGF-1, and SDF-1, which increase migration, invasion, and vessel-forming capacity of endothelial progenitor cells (EPCs) [23]. EPCs have been shown to play important roles in tumor neovascularization [26–28], and hence, our previous study suggests that hMSCs might indirectly affect tumor growth by regulating EPC functions.
Apart from affecting EPC functions, several previous studies show that hMSCs enhanced engraftment of several cancers, including breast cancer, ovarian cancer, melanoma, glioma, and colon cancer cells [9, 10, 12, 13, 16, 17, 29–32]. They have also been shown to be the source of cancer-associated fibroblasts (CAFs) that enhance tumor growth and metastasis by secreting various bioactive molecules that induce tumor neovascularization and inhibit immune reaction against cancer cells [8–17]. Contrary to those evidences, however, several other studies showed that hMSC inhibited growth and metastasis of several cancers, including colon cancer, hepatocellular carcinoma, and melanoma in animal models [19, 20, 33]. Those conflicting results possibly arise from the variability in both hMSC sources and cancer cell types used in those studies.
Indeed, the present study showed that the effects of hMSC-derived bioactive molecules on cancer cell proliferation depend on both hMSC sources and cancer cell types. Some particular cancer cells, such as hepatoblastoma cell C3A and colon adenocarcinoma cell HT29, were very sensitive to the suppressive effects of hMSCs while the proliferation of lung adenocarcinoma cell A549 was not affected by hMSC-derived bioactive molecules. The insensitivity of A549 to hMSC-derived factors is in agreement with the previous study which showed that Wharton's jelly-derived hMSC (WJ-hMSC) secretome and human amniotic membrane protein extract (hAMPE) did not affect the proliferation of A549 cells [34]. Also in agreement with the previous study showing that amniotic membrane-derived hMSCs (AM-hMSCs) suppressed the proliferation of Saos2 cells [35], we observed the inhibitory effects of both bone marrow and gestational tissue-derived hMSCs on Saos2 cell proliferation. However, the inhibitory effects of hMSCs on Saos2 cells appeared to be lesser in degree and later in onset compared with their effects on C3A and HT29 cells.
When compared among distinct hMSC sources, hAMSCs and CH-hMSCs inhibited cancer cell proliferation at the level similar to that of BM-hMSCs, which is the standard source of hMSCs for clinical applications. Interestingly, while BM-hMSCs have greater migratory ability toward cancer cells than gestational tissue-derived hMSCs, their ability to suppress hepatoblastoma cell migration was lower than those of gestational tissue-derived hMSCs.
Although several previous studies showed that cancer-derived bioactive molecules could induce hMSCs to release bioactive molecules that promote cancer cell proliferation [36–38], we found that cancer-derived bioactive molecules, regardless of both hMSC sources and cancer cell types, did not affect the cancer suppressive capacity of hMSCs.
The hMSC-derived factors that inhibit the proliferation of hepatoblastoma cell C3A were distinct from those that inhibited the proliferation of colon adenocarcinoma cell HT29. The hMSC-derived factors that suppress the proliferation of C3A cells, regardless of hMSC sources, were clearly enriched in the 100 kDa fraction of hMSC-conditioned media while hMSC-derived factors that suppress HT29 proliferation, also regardless of the hMSC sources, were enriched in the 50 kDa, 30 kDa, and 10 kDa fractions of hMSC-conditioned media. The results suggest that all hMSCs might suppress the proliferation of C3A and HT29 cells by secreting the same combination of bioactive molecules (in which the bioactive molecules that suppress C3A cell proliferation were mostly large proteins whose molecular weights exceed 100 kDa while the bioactive molecules that suppress HT29 proliferation were consisted of several proteins that have distinct molecular weights).
To identify hMSC-derived factors that were able to suppress C3A cell proliferation, we chose the 100 kDa fraction of hAMSC-conditioned medium, which was shown to be enriched for factors that suppress the proliferation of C3A cells, for mass spectrometry analysis. The results revealed that the 100 kDa fraction of hAMSC secretome consisted of at least 105 secreted bioactive molecules that belong to various groups of proteins, such as extracellular matrix, proteinase, and cytokines together with novel proteins whose functions have not been identified. Among the identified bioactive molecules are proteins that have previously been shown to suppress the proliferation of various cancer cells, including HEAT repeat-containing protein 1 (HEATR1) [39]; leprecan-like protein (LEPREL) [40, 41]; NK1 transcription factor-related protein 2 (NK1.2) [42]; small inducible cytokine subfamily E, member 1 (SCYE1 or AIMP1) [43, 44]; sushi domain-containing protein 2 (SUSD2) [45]; arylsulfatase B (ARSB) [46]; and type IV collagen [47] (Supplementary table 1).
Compared with ELISA array and western blot analysis that has been used to identify limited a number of hMSC-derived factors in the previous studies [48–50], the mass spectrometry analysis used in our study could discover complete protein composition of hAMSC secretome in an unbiased manner. As a result, we have identified several factors which have not been described in the previous literatures. However, due to the large number of bioactive molecules presented in the 100 kDa fraction of hAMSC-conditioned medium, the further fractionation of hMSC-conditioned media into smaller subfractions is required for the identification of the exact hMSC-derived factors that suppress cancer cell proliferation.
5. Conclusion
We herein report that the effects of hMSCs on cancer cell proliferation depend on both hMSC sources and cancer cell types. We also demonstrated that cancer-derived bioactive molecules did not affect the cancer suppressive capacity of hMSCs. Moreover, hMSCs might use a distinct combination of bioactive molecules to suppress the proliferation of human hepatoblastoma and colorectal cancer cells. We believe that the in vitro system developed in this study could be used to discover other therapeutically useful molecules released from hMSCs for the future in vivo study.
Acknowledgments
This research project was funded by a grant from Thammasat University.
Abbreviations
- BM-hMSCs:
Bone marrow-derived human mesenchymal stem cells
- CH-hMSCs:
Chorion-derived human mesenchymal stem cells
- hAMSCs:
Placenta-derived human mesenchymal stem cells
- UC-hMSCs:
Umbilical cord-derived human mesenchymal stem cell
- Complete DMEM:
DMEM supplemented with 10% (v/v) fetal bovine serum
- Normal hMSCs:
Conditioned media derived from hMSCs
- C3A/hMSCs:
Conditioned media derived from hMSCs co-cultured with C3A cells for 7 days
- HT29/hMSCs:
Conditioned media derived from hMSCs co-cultured with HT29 cells for 7 days
- Saos2/hMSCs:
Conditioned media derived from hMSCs co-cultured with Saos2 cells for 7 days
- A549/hMSCs:
Conditioned media derived from hMSCs co-cultured with A549 cells for 7 days
- U251/hMSCs:
Conditioned media derived from hMSCs co-cultured with U251 cells for 7 days.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Additional Points
Novelty and Impact Statements. We herein demonstrate that the effect of hMSCs on cancer cell proliferation and migration depends on both hMSC sources and cancer cell types. We also showed that cancer-derived bioactive molecules did not affect the cancer suppressive capacity of hMSCs and each hMSC use distinct combination of bioactive molecules to suppress the proliferation of human hepatoblastoma and colorectal cancer cells. The procedure developed in this study could be used to discover other therapeutically useful molecules produced by hMSCs for future research and clinical uses.
Ethical Approval
This study was approved by the ethical committee for human researches, Faculty of Medicine Thammasat University, which was in accordance with the Declaration of Helsinki, the Belmont Report, CIOMS Guidelines, and ICH-GCP. Human bone marrow samples were obtained from healthy volunteers after giving written informed consent. The gestational tissues (umbilical cord, placenta, and chorion) were obtained from healthy newborns after receiving written informed consents from their mothers.
Disclosure
There were no commercial organizations or funding bodies associated with data collection and analysis or with this study manuscript.
Conflicts of Interest
The authors declare no potential conflicts of interest.
Authors' Contributions
P.K. designed the experiments, analyzed the data, supervised the study, and wrote and finalized the manuscript. N.P. and W.K. performed the experiments and analyzed the data. S.M., C.T., and D.T. analyzed the data. S.R. performed proteomic analysis and analyzed the data. All authors read and approved the final manuscript.
Supplementary Materials
List of secreted proteins in the 100 kDa fraction of hAMSC secretome as identified by mass spectrometry analysis in Figures 7(a) and 7(b).
References
- 1.Pittenger M. F., Mackay A. M., Beck S. C., et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 2.Rahavi H., Hashemi S. M., Soleimani M., Mohammadi J., Tajik N. Adipose tissue-derived mesenchymal stem cells exert in vitro immunomodulatory and beta cell protective functions in streptozotocin-induced diabetic mice model. Journal Diabetes Research. 2015;2015, article 878535:10. doi: 10.1155/2015/878535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vellasamy S., Sandrasaigaran P., Vidyadaran S., Abdullah M., George E., Ramasamy R. Mesenchymal stem cells of human placenta and umbilical cord suppress T-cell proliferation at G0 phase of cell cycle. Cell Biology International. 2013;37(3):250–256. doi: 10.1002/cbin.10033. [DOI] [PubMed] [Google Scholar]
- 4.Wang H. S., Hung S. C., Peng S. T., et al. Mesenchymal stem cells in the Wharton's jelly of the human umbilical cord. Stem Cells. 2004;22(7):1330–1337. doi: 10.1634/stemcells.2004-0013. [DOI] [PubMed] [Google Scholar]
- 5.Ziadlou R., Shahhoseini M., Safari F., Sayahpour F. A., Nemati S., Eslaminejad M. B. Comparative analysis of neural differentiation potential in human mesenchymal stem cells derived from chorion and adult bone marrow. Cell and Tissue Research. 2015;362(2):367–377. doi: 10.1007/s00441-015-2210-z. [DOI] [PubMed] [Google Scholar]
- 6.Nauta A. J., Fibbe W. E. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007;110(10):3499–3506. doi: 10.1182/blood-2007-02-069716. [DOI] [PubMed] [Google Scholar]
- 7.Tao H., Han Z., Han Z. C., Li Z. Proangiogenic features of mesenchymal stem cells and their therapeutic applications. Stem Cells International. 2016;2016:11. doi: 10.1155/2016/1314709.1314709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bagley R. G., Weber W., Rouleau C., et al. Human mesenchymal stem cells from bone marrow express tumor endothelial and stromal markers. International Journal of Oncology. 2009;34(3):619–627. doi: 10.3892/ijo_00000187. [DOI] [PubMed] [Google Scholar]
- 9.Bexell D., Gunnarsson S., Tormin A., et al. Bone marrow multipotent mesenchymal stroma cells act as pericyte-like migratory vehicles in experimental gliomas. Molecular Therapy. 2009;17(1):183–190. doi: 10.1038/mt.2008.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Galiè M., Konstantinidou G., Peroni D., et al. Mesenchymal stem cells share molecular signature with mesenchymal tumor cells and favor early tumor growth in syngeneic mice. Oncogene. 2008;27(18):2542–2551. doi: 10.1038/sj.onc.1210920. [DOI] [PubMed] [Google Scholar]
- 11.Mishra P. J., Mishra P. J., Humeniuk R., et al. Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells. Cancer Research. 2008;68(11):4331–4339. doi: 10.1158/0008-5472.CAN-08-0943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shinagawa K., Kitadai Y., Tanaka M., et al. Mesenchymal stem cells enhance growth and metastasis of colon cancer. International Journal of Cancer. 2010;127(10):2323–2333. doi: 10.1002/ijc.25440. [DOI] [PubMed] [Google Scholar]
- 13.Spaeth E. L., Dembinski J. L., Sasser A. K., et al. Mesenchymal stem cell transition to tumor-associated fibroblasts contributes to fibrovascular network expansion and tumor progression. PLoS One. 2009;4(4, article e4992) doi: 10.1371/journal.pone.0004992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu Y., Chen L., Scott P. G., Tredget E. E. Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells. 2007;25(10):2648–2659. doi: 10.1634/stemcells.2007-0226. [DOI] [PubMed] [Google Scholar]
- 15.Wu Y., Wang J., Scott P. G., Tredget E. E. Bone marrow-derived stem cells in wound healing: a review. Wound Repair and Regeneration. 2007;15(Supplement 1):S18–S26. doi: 10.1111/j.1524-475X.2007.00221.x. [DOI] [PubMed] [Google Scholar]
- 16.Zhu W., Xu W., Jiang R., et al. Mesenchymal stem cells derived from bone marrow favor tumor cell growth in vivo. Experimental and Molecular Pathology. 2006;80(3):267–274. doi: 10.1016/j.yexmp.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Djouad F., Plence P., Bony C., et al. Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood. 2003;102(10):3837–3844. doi: 10.1182/blood-2003-04-1193. [DOI] [PubMed] [Google Scholar]
- 18.Maestroni G. J. M., Hertens E., Galli P. Factor(s) from nonmacrophage bone marrow stromal cells inhibit Lewis lung carcinoma and B16 melanoma growth in mice. Cellular and Molecular Life Sciences (CMLS) 1999;55(4):663–667. doi: 10.1007/s000180050322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ohlsson L. B., Varas L., Kjellman C., Edvardsen K., Lindvall M. Mesenchymal progenitor cell-mediated inhibition of tumor growth in vivo and in vitro in gelatin matrix. Experimental and Molecular Pathology. 2003;75(3):248–255. doi: 10.1016/j.yexmp.2003.06.001. [DOI] [PubMed] [Google Scholar]
- 20.Qiao L., Xu Z., Zhao T., et al. Suppression of tumorigenesis by human mesenchymal stem cells in a hepatoma model. Cell Research. 2008;18(4):500–507. doi: 10.1038/cr.2008.40. [DOI] [PubMed] [Google Scholar]
- 21.Choudhery M. S., Khan M., Mahmood R., Mehmood A., Khan S. N., Riazuddin S. Bone marrow derived mesenchymal stem cells from aged mice have reduced wound healing, angiogenesis, proliferation and anti-apoptosis capabilities. Cell Biology International. 2012;36(8):747–753. doi: 10.1042/CBI20110183. [DOI] [PubMed] [Google Scholar]
- 22.Kretlow J. D., Jin Y. Q., Liu W., et al. Donor age and cell passage affects differentiation potential of murine bone marrow-derived stem cells. BMC Cell Biology. 2008;9(1):p. 60. doi: 10.1186/1471-2121-9-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kamprom W., Kheolamai P., U-Pratya Y., et al. Effects of mesenchymal stem cell-derived cytokines on the functional properties of endothelial progenitor cells. European Journal of Cell Biology. 2016;95(3-5):153–163. doi: 10.1016/j.ejcb.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 24.Kamprom W., Kheolamai P., U-Pratya Y., et al. Endothelial progenitor cell migration-enhancing factors in the secretome of placental-derived mesenchymal stem cells. Stem Cells International. 2016;2016:13. doi: 10.1155/2016/2514326.2514326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Uccelli A., Moretta L., Pistoia V. Mesenchymal stem cells in health and disease. Nature Reviews Immunology. 2008;8(9):726–736. doi: 10.1038/nri2395. [DOI] [PubMed] [Google Scholar]
- 26.Hassan S., Ferrario C., Saragovi U., et al. The influence of tumor-host interactions in the stromal cell-derived factor-1/CXCR4 ligand/receptor axis in determining metastatic risk in breast cancer. The American Journal of Pathology. 2009;175(1):66–73. doi: 10.2353/ajpath.2009.080948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jin F., Brockmeier U., Otterbach F., Metzen E. New insight into the SDF-1/CXCR4 axis in a breast carcinoma model: hypoxia-induced endothelial SDF-1 and tumor cell CXCR4 are required for tumor cell intravasation. Molecular Cancer Research. 2012;10(8):1021–1031. doi: 10.1158/1541-7786.MCR-11-0498. [DOI] [PubMed] [Google Scholar]
- 28.Kaplan R. N., Riba R. D., Zacharoulis S., et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature. 2005;438(7069):820–827. doi: 10.1038/nature04186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Karnoub A. E., Dash A. B., Vo A. P., et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449(7162):557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- 30.Muehlberg F. L., Song Y. H., Krohn A., et al. Tissue-resident stem cells promote breast cancer growth and metastasis. Carcinogenesis. 2009;30(4):589–597. doi: 10.1093/carcin/bgp036. [DOI] [PubMed] [Google Scholar]
- 31.Rhodes L. V., Muir S. E., Elliott S., et al. Adult human mesenchymal stem cells enhance breast tumorigenesis and promote hormone independence. Breast Cancer Research and Treatment. 2010;121(2):293–300. doi: 10.1007/s10549-009-0458-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yu J. M., Jun E. S., Bae Y. C., Jung J. S. Mesenchymal stem cells derived from human adipose tissues favor tumor cell growth in vivo. Stem Cells and Development. 2008;17(3):463–474. doi: 10.1089/scd.2007.0181. [DOI] [PubMed] [Google Scholar]
- 33.Miao Z., Jin J., Chen L., et al. Isolation of mesenchymal stem cells from human placenta: comparison with human bone marrow mesenchymal stem cells. Cell Biology International. 2006;30(9):681–687. doi: 10.1016/j.cellbi.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 34.Hendijani F., Javanmard S. H., Rafiee L., Sadeghi-Aliabadi H. Effect of human Wharton's jelly mesenchymal stem cell secretome on proliferation, apoptosis and drug resistance of lung cancer cells. Research in Pharmaceutical Sciences. 2015;10(2):134–142. [PMC free article] [PubMed] [Google Scholar]
- 35.Magatti M., Munari S., Vertua E., Parolini O. Amniotic membrane-derived cells inhibit proliferation of cancer cell lines by inducing cell cycle arrest. Journal of Cellular and Molecular Medicine. 2012;16(9):2208–2218. doi: 10.1111/j.1582-4934.2012.01531.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Direkze N. C., Hodivala-Dilke K., Jeffery R., et al. Bone marrow contribution to tumor-associated myofibroblasts and fibroblasts. Cancer Research. 2004;64(23):8492–8495. doi: 10.1158/0008-5472.CAN-04-1708. [DOI] [PubMed] [Google Scholar]
- 37.Kidd S., Spaeth E., Watson K., et al. Origins of the tumor microenvironment: quantitative assessment of adipose-derived and bone marrow-derived stroma. PLoS One. 2012;7(2, article e30563) doi: 10.1371/journal.pone.0030563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Quante M., Tu S. P., Tomita H., et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell. 2011;19(2):257–272. doi: 10.1016/j.ccr.2011.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu T., Fang Y., Zhang H., et al. HEATR1 negatively regulates Akt to help sensitize pancreatic cancer cells to chemotherapy. Cancer Research. 2016;76(3):572–581. doi: 10.1158/0008-5472.CAN-15-0671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shah R., Smith P., Purdie C., et al. The prolyl 3-hydroxylases P3H2 and P3H3 are novel targets for epigenetic silencing in breast cancer. British Journal of Cancer. 2009;100(10):1687–1696. doi: 10.1038/sj.bjc.6605042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang J., Xu X., Liu Z., et al. LEPREL1 expression in human hepatocellular carcinoma and its suppressor role on cell proliferation. Gastroenterology Research and Practice. 2013;2013:7. doi: 10.1155/2013/109759.109759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen H., Liu W., Zhong L., et al. NKX2-2 suppresses osteosarcoma metastasis and proliferation by downregulating multiple target genes. Journal of Cancer. 2018;9(17):3067–3077. doi: 10.7150/jca.26382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Han J. M., Myung H., Kim S. Antitumor activity and pharmacokinetic properties of ARS-interacting multi-functional protein 1 (AIMP1/p43) Cancer Letters. 2010;287(2):157–164. doi: 10.1016/j.canlet.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 44.Lee Y. S., Han J. M., Kang T., Park Y. I., Kim H. M., Kim S. Antitumor activity of the novel human cytokine AIMP1 in an in vivo tumor model. Molecules and Cells. 2006;21(2):213–217. [PubMed] [Google Scholar]
- 45.Pan W., Cheng Y., Zhang H., et al. CSBF/C10orf99, a novel potential cytokine, inhibits colon cancer cell growth through inducing G1 arrest. Scientific Reports. 2015;4(1, article 6812) doi: 10.1038/srep06812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bhattacharyya S., Feferman L., Terai K., Dudek A. Z., Tobacman J. K. Decline in arylsulfatase B leads to increased invasiveness of melanoma cells. Oncotarget. 2017;8(3):4169–4180. doi: 10.18632/oncotarget.13751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Maeshima Y., Colorado P. C., Torre A., et al. Distinct antitumor properties of a type IV collagen domain derived from basement membrane. The Journal of Biological Chemistry. 2000;275(28):21340–21348. doi: 10.1074/jbc.M001956200. [DOI] [PubMed] [Google Scholar]
- 48.Danieli P., Malpasso G., Ciuffreda M. C., et al. Conditioned medium from human amniotic mesenchymal stromal cells limits infarct size and enhances angiogenesis. Stem Cells Translational Medicine. 2015;4(5):448–458. doi: 10.5966/sctm.2014-0253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Konig J., Weiss G., Rossi D., et al. Placental mesenchymal stromal cells derived from blood vessels or avascular tissues: what is the better choice to support endothelial cell function? Stem Cells and Development. 2015;24(1):115–131. doi: 10.1089/scd.2014.0115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kuchroo P., Dave V., Vijayan A., Viswanathan C., Ghosh D. Paracrine factors secreted by umbilical cord-derived mesenchymal stem cells induce angiogenesis in vitro by a VEGF-independent pathway. Stem Cells and Development. 2015;24(4):437–450. doi: 10.1089/scd.2014.0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
List of secreted proteins in the 100 kDa fraction of hAMSC secretome as identified by mass spectrometry analysis in Figures 7(a) and 7(b).
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.