Summary
Thiamine (vitamin B1, VB1) can act as a plant defence trigger, or priming agent, leading to a rapid counterattack on pathogen invasion. In this study, the priming effect of thiamine on rice (O ryza sativa cv. Nipponbare) and its activity against root‐knot nematode (M eloidogyne graminicola) infection were evaluated. Thiamine treatment and subsequent nematode inoculation activated hydrogen peroxide (H2O 2) accumulation and lignin deposition in plant roots, and this correlated with enhanced transcription of OsPAL 1 and OsC4H, two genes involved in the phenylpropanoid pathway. The number of nematodes in rice roots was slightly but significantly reduced, and the development of the nematodes was delayed, whereas no direct toxic effects of VB1 on nematode viability and infectivity were observed. The combined application of thiamine with l‐2‐aminooxy‐3‐phenylpropionic acid (AOPP), an inhibitor of phenylalanine ammonia‐lyase (PAL), significantly hampered the VB1‐priming capacity. These findings indicate that thiamine‐induced priming in rice involves H2O 2 and phenylpropanoid‐mediated lignin production, which hampers nematode infection. Further cellular and molecular studies on the mechanism of thiamine‐induced defence will be useful for the development of novel nematode control strategies.
Keywords: callose, hydrogen peroxide, induced defence, lignin, Meloidogyne graminicola, priming, thiamine
Introduction
The root‐knot nematode (RKN) Meloidogyne graminicola causes significant yield reductions in aerobic, lowland and deepwater rice in South‐East Asia, southern Africa and America (Kyndt et al., 2014). The symptoms include knot‐ or gall‐like formations on the roots, caused mainly by the development of ‘giant cells’. These induced giant cells act as feeding sites and display transfer cell‐like features (Kyndt et al., 2013). The control of RKN using conventional methods is difficult because of its wide host range, ability to survive in soil and weeds, and the low inherent level of resistance in rice against this nematode. Instead of the use of nematicides, priming could be a more environmentally friendly control method (Conrath et al., 2006).
Priming is a unique physiological state that enables plant cells to respond to a biotic or abiotic stress in a more rapid and robust manner than non‐primed cells, and this state can be induced by the application of low levels of so‐called ‘priming agents’ (Conrath et al., 2006). Priming is one of the most efficient types of induced resistance because the metabolic investment of the plant is reduced compared with constitutive defence activation. On priming, defence responses are strongly activated when plants are challenged by pathogen or insect invasion. The priming induced by some chemical activators, such as β‐aminobutyric acid (BABA) and benzo‐(1,2,3)‐thiadiazole‐7‐carbothioic acid s‐methylester (BTH), is capable of inducing rapid and effective defence responses to impede various invading pathogens, such as Pseudomonas syringae (Ahn et al., 2007), Alternaria brassicicola and Plectosphaerella cucumerina (Ton and Mauch‐Mani, 2004).
Thiamine (vitamin B1, VB1) is a water‐soluble B‐complex vitamin that is produced in plants and microbes (Burrows et al., 2000). Studies have verified that thiamine is a systemic, broad‐spectrum, long‐lasting resistance inducer in multiple plant species (Ahn et al., 2005, 2007; Jung and Kim, 2003). Spraying of thiamine on rice leaves, followed by infection by the blast fungus Magnaporthe grisea, induced stronger and more rapid pathogenesis‐related (PR) gene expression in comparison with the non‐treated control, and also enhanced the activity of protein kinase C (Ahn et al., 2005). Ahn et al. (2007) further verified that thiamine‐induced priming in Arabidopsis against P. syringae is dependent on hydrogen peroxide (H2O2) and requires intact Nonexpressor of Pathogenesis‐Related Protein 1 (NPR1). Seed treatment with thiamine induced rapid lipoxygenase (LOX) gene expression and resulted in significant disease protection against downy mildew disease in pearl millet (Pushpalatha et al., 2011). Although thiamine has been found to induce resistance to different pathogens, the molecular mechanisms underlying this have not been completely elucidated (Asensi‐Fabado and Munné‐Bosch, 2010), and nematode infection has not been tested previously.
The aim of this study was to determine the priming capacity and molecular mechanisms involved in thiamine‐induced defence in rice against M. graminicola. The results presented here demonstrate that thiamine can reduce nematode infection. Furthermore, we demonstrate that this phenomenon involves priming of H2O2 production and lignification in rice roots.
Results
Thiamine has no direct negative impact on the viability and infectivity of M . graminicola
To evaluate whether thiamine has a direct negative effect on the viability of RKNs, the nematodes were soaked in different concentrations of thiamine. At doses ranging from 0.6 to 10 mm thiamine, the percentage of dead nematodes in the thiamine‐incubated group (6.8% ± 0.6%) was not significantly different from that in the water‐incubated control group (6.3% ± 0.4%) (Fig. 1a). These data show that thiamine has no direct toxic effect on M. graminicola up to 72 h at the investigated doses.
Figure 1.

Effect of thiamine soaking on nematode viability and infectivity. (a) The percentage of dead juveniles after 24 h and 72 h of incubation in various concentrations of thiamine and water (control). (b) Infectivity and development of thiamine‐incubated and water‐incubated M eloidogyne graminicola in rice roots. (c) thiamine‐incubated (VB1) and water‐incubated nematodes were inoculated on plants and photographed at 7 days pos‐inoculation (dpi) and (d) 14 dpi. The whole experiment was repeated three times, and there were six individual plants in each replicate. Data presented are the means ± standard error (SE) of six replicates.
To verify whether thiamine has a direct effect on the general infectivity of the nematodes, thiamine‐incubated and water‐incubated nematodes were inoculated on plants and their development in the roots was recorded at different time points after inoculation (Fig. 1b). At 7 days post‐inoculation (dpi), most nematodes had developed into third‐stage juveniles (J3s). No significant difference was observed between the numbers of J3s in the thiamine‐incubated and water‐incubated groups (Fig. 1b,c). At 14 dpi, most nematodes had developed into adult females and the number of females in the thiamine‐incubated group was similar to that in the water‐incubated group (Fig. 1b,d). Taken together, these data show that thiamine has no direct negative impact on the viability and infectivity of M. graminicola.
Thiamine protects rice from M . graminicola infection without restricting the growth of the plant
In a preliminary experiment, different concentrations of thiamine were applied to rice plants and the disease severity was assessed. Rice roots were inoculated with M. graminicola 1 d after drenching the roots in thiamine, and the numbers of galls and nematodes were counted at 14 dpi. Based on the observation that 2.5 mm thiamine is effective in inducing the defence of rice against M. graminicola (Fig. S1, see Supporting Information), this concentration was chosen for further studies. Compared with control plants, pretreatment with 2.5 mm thiamine resulted in a significantly smaller number of nematodes and root galls per plant at 14 dpi (Fig. 2a). In addition, a slight delay in development was observed for the nematodes in thiamine‐treated roots. At 14 dpi, the ratio of adult females in thiamine‐treated plants was significantly lower (86.2% ± 3.7%) than in control plants (95.6% ± 4.1%) (Fig. 2a,b). However, the ratio of the fourth‐stage juveniles (J4s) in thiamine‐treated plants (13.0% ± 1.4%) was significantly higher than that in water‐treated plants (4.7% ± 1.2%). No significant difference in J3 nematodes was observed between these two treatments. These data demonstrate that thiamine treatment 1 day before inoculation not only hinders the penetration of M. graminicola, but also delays nematode development, indicating that thiamine can prime rice for induced defence against M. graminicola. The thiamine treatment did not cause any visible negative alterations in plant growth based on an evaluation of plant height and fresh weight (Fig. 2c). In thiamine‐treated, nematode‐inoculated plants, the height and weight of the shoot and whole plant were significantly higher than in non‐treated, but inoculated, plants.
Figure 2.

Effect of 2.5 mM thiamine drenching on the plant and nematode infection. Number of nematodes (a) and root galls (b) in different developmental stages in thiamine‐drenched and non‐drenched Nipponbare roots were counted at 14 days post‐inoculation (dpi). (c) Roots were dyed with acid fuchsin and nematodes in roots were observed under a Leica stereomicroscope. Plant heigth (d) and fresh weigth (e) of the plants at 14 dpi. Mg, M eloidogyne graminicola. The bars in the different graphs represent the means ± SE of the data from three independent biological replicates, each containing six plants. Asterisks indicate significant differences (Duncan's multiple range test with P ≤ 0.05).
Thiamine treatment does not change rice root attractiveness to nematodes and does not visibly affect giant cell development
J2s in the soil are attracted to the root tips by molecular signals originating from the plant roots. To evaluate whether thiamine treatment influences the attractiveness of rice roots, we examined the number of attracted nematodes around the root tips of plants treated with thiamine or water (Fig. 3a). At 9 h after inoculation, the number of nematodes in a 5‐mm diameter around the root tips was counted. The number of nematodes attracted to thiamine‐treated root tips was not significantly different from that of control roots (Fig. 3b).
Figure 3.

Effect of thiamine (VB1) on the behaviour of M eloidogyne graminicola and microscopic observation of giant cells. (a) Attraction of M . graminicola towards the rice root tip after drenching with 2.5 mm thiamine or water. (b) Nematodes within 5 mm from the root tip were counted at 9 h after inoculation. Data presented are the means ± standard error (SE) of six replicates. (c) Giant cells (*) in thiamine‐treated rice root galls and control root galls were stained with toluidine blue and observed under an Olympus BX 51 (Berchem, Belgium) microscope with a ColorView III camera at 7 dpi.
Microscopic analysis of the nematode feeding sites inside the galls at 7 dpi revealed that the giant cells in both thiamine‐treated plants and control plants were enlarged, with dense cytoplasm, large vacuoles, multiple nuclei and thickened cell walls, and no obvious differences in these morphological features were observed (Fig. 3c).
Thiamine treatment does not affect callose deposition in nematode‐induced galls
Callose is known to be induced as a plant resistance response after exposure to a range of biotic and abiotic stresses (Hamiduzzaman et al., 2005). Therefore, the expression of a callose synthase‐encoding gene, OsGSL1, was investigated using quantitative reverse transcription‐polymerase chain reaction (qRT‐PCR) in both thiamine‐primed and control plants (Fig. S2, see Supporting Information). The expression level of OsGSL1 was significantly upregulated in thiamine‐treated plants at 6 h post‐inoculation (hpi) (Time 1), but not at later time points (Time 2, Time 3) (Fig. 4a).
Figure 4.
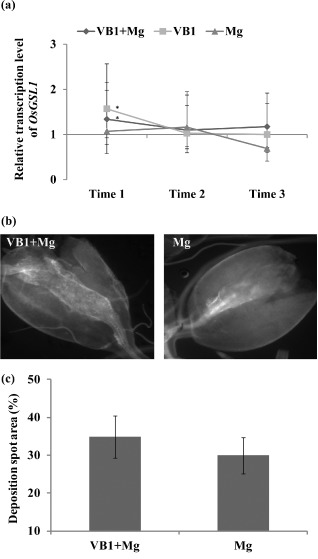
Quantitative reverse transcription‐polymerase chain reaction (qRT‐PCR) analysis of expression of the callose synthesis gene OsGSL 1 and microscopic observation of callose deposition. (a) The relative transcript levels of the callose synthesis gene (OsGSL 1) were analysed using qRT‐PCR at 6, 24 and 72 h post‐inoculation (hpi). Gene expression levels were normalized using three internal reference genes, OsEXP, OsEif5C and OsEXP narsai. Data are shown as relative transcript levels normalized to the control roots (expression level set at 1). The bars represent the mean expression levels ± standard error (SE) from two independent biological replicates and three technical replicates, each containing a pool of six plants. Asterisks indicate significant differential expression (Duncan's multiple range test with P ≤ 0.05). Mg, M eloidogyne graminicola. (b) Callose deposition in root galls at 7 days post‐inoculation (dpi) was examined under UV light using a Nikon (Amsterdam, The Netherlands) Eclipse Ti‐E epifluorescence microscope. (c) Quantification of callose deposition was performed using Image J software. Data presented are the means ± SE of two independent experiments, each performed using 10 galls.
Microscopic observations revealed no significant difference in callose deposition in the galls of thiamine‐treated plants in comparison with non‐treated plants at 7 dpi (Fig. 4b). Quantitative analysis further confirmed these results. The average area of the deposition spot in the galls of thiamine‐treated plants was similar to that of non‐treated plants. These data show that, although the callose synthase gene may be slightly induced at early time points after thiamine treatment, this does not lead to callose accumulation inside the galls. Therefore, callose deposition is not the major driver of thiamine‐induced priming in rice.
Thiamine primes the plant for induced H2O 2 generation on nematode infection
Based on the peroxidation role of thiamine in Arabidopsis against Pseudomonas syringae (Ahn et al., 2007), it was investigated whether thiamine is responsible for priming the generation of reactive oxygen species (ROS) in the rice–M. graminicola interaction (Fig. S2). Thiamine alone did not trigger an H2O2 response in rice roots as no significant differences in H2O2 levels were observed 30 h after thiamine treatment compared with control roots (Time 1) (Fig. 5a). However, on nematode challenge, thiamine‐treated plants showed induced H2O2 accumulation compared with non‐treated plants by 203.5% ± 7.2% at 24 hpi (Time 2) and 196.5% ± 8.1% at 72 hpi (Time 3). The expression level of OsRbohB, an NADPH oxidase gene known to be involved in the plant immune response (Wong et al., 2007), was investigated using qRT‐PCR. After an initial downregulation at 6 hpi (Time 1) after thiamine treatment, the gene showed an induced expression at the two later time points, and the highest expression was detected in thiamine‐treated plants that were inoculated (Fig. 5b).
Figure 5.

Quantification of hydrogen peroxide (H2O 2) and relative expression analysis of the H2O 2 synthesis gene OsRbohB. (a) The H2O 2 content per gram of roots was measured using a CLARIOstar (BMG Labtech, Ortenberg, Germany) Microplate Reader at 390 nm. The bars represent the means ± standard error (SE) of four replicates, each containing a pool of six roots. Different letters indicate significant differences (Duncan's multiple range test with P ≤ 0.05). Mg, M eloidogyne graminicola. (b) The relative transcript levels of the H2O 2 synthesis gene (OsRbohB) were analysed using quantitative reverse transcription polymerase chain reaction (qRT‐PCR) at 6, 24 and 72 h post‐inoculation (hpi). Gene expression levels were normalized using three internal reference genes, i.e. OsEXP, OsEif5C and OsEXP narsai. Data are shown as relative transcript levels in comparison with the control roots (expression level set at 1). The bars represent the mean expression levels ± SE from two independent biological replicates and three technical replicates, each containing a pool of six plants. Asterisks indicate significant differential expression (P ≤ 0.05).
Thiamine‐induced defence in rice against M . graminicola is mediated by activation of the phenylpropanoid pathway
Phenylalanine ammonia‐lyase (PAL) is an important enzyme in the phenylpropanoid pathway that catalyses the deamination of phenylalanine to trans‐cinnamic acid, a precursor of the lignin and flavonoid biosynthetic pathways (Kim and Hwang, 2014). Further downstream, phenylpropanoid metabolism provides plants with thousands of compounds that are used as immunity signals (salicylic acid, SA), structural cell components (lignin, suberin and other cell wall‐associated phenolics), pigments (flavonoids, anthocyanins) and toxins (coumarins and furanocoumarins). To establish whether the PAL‐mediated pathway participates in thiamine‐induced priming against RKNs in rice, the expression levels of OsPAL1, OsC4H and OsCAD6, key markers of the phenylpropanoid pathway, and OsWRKY45, a key marker of the SA response pathway, were studied (Fig. 6). qRT‐PCR revealed significant differences in OsPAL1 expression between thiamine‐treated plants and control plants at 24 hpi (Time 2) and 72 hpi (Time 3) (Fig. 6a). Higher transcript levels of OsWRKY45 were observed in thiamine‐treated plants compared with control plants only at 6 hpi (Time 1). OsC4H transcript accumulation was significantly higher in thiamine‐treated plants compared with control plants at 24 hpi (Time 2). However, the transcript level of another phenylpropanoid biosynthesis gene, OsCAD6, was decreased in thiamine‐treated plants compared with control plants at all investigated time points, especially at 24 hpi (Time 2).
Figure 6.

Quantitative reverse transcription polymerase chain reaction (RT‐PCR) analysis of the defence‐related genes in the phenylpropanoid pathway and the effect of an inhibitor of phenylpropanoid biosynthesis on nematode infection. (a) The relative expression levels of OsPAL 1, OsC4H, OsCAD 6 and OsWRKY 45, which are involved in lignin biosynthesis, were analysed using qRT‐PCR at 6, 24 and 72 h post‐inoculation (hpi). Gene expression levels were normalized using three internal reference genes, OsEXP, OsEif5C and OsEXP narsai. Data are shown as relative transcript levels in comparison with the control roots (expression level set at unity). The bars represent the mean expression levels ± standard error (SE) from two independent biological replicates and three technical replicates, each containing a pool of six plants. Asterisks indicate significant differential expression (P ≤ 0.05). VB1, thiamine drench alone; Mg, M eloidogyne graminicola inoculation alone; VB1 + Mg, thiamine drench plus M . graminicola inoculation; control, water drench and non‐inoculation. WRKY, a transcription factor with the conserved amino acid sequence WRKYGQK at its N‐terminal domain; PAL 1, phenylalanine ammonia‐lyase; C4H, cinnamate 4‐hydroxylase; CAD 6, cinnamyl alcohol dehydrogenase. (b) Effect of an inhibitor (l‐2‐aminooxy‐3‐phenylpropionic acid, AOPP) of phenylpropanoid biosynthesis on nematode infection. AOPP (100 μm) was combined in a drench with or without 2.5 mm thiamine on 2‐week‐old rice roots, 1 day before inoculation. Nematodes in the roots were counted at 14 days post‐inoculation (dpi). The bars represent the means of the data from two independent biological replicates, each containing six plants. Different letters indicate significant differences (Duncan's multiple range test with P ≤ 0.05). (c) The lignin content in the cell wall residue (CWR) of roots of rice was determined using the Acetyl Bromide (AcBr) assay. Measurements were conducted 1 day after thiamine treatment (VB1 + Nipponbare) or water treatment (Nipponbare). The bars represent the means ± SE of the lignin content of six plants. Different letters indicate significant differences.
To further investigate the role of the phenylpropanoid pathway in thiamine‐induced defence in the rice–M. graminicola interaction, an inhibitor of phenylpropanoid biosynthesis, l‐2‐aminooxy‐3‐phenylpropionic acid (AOPP), was applied to rice plants at 24 h before nematode inoculation. Although no significant difference was observed between the AOPP‐treated and control plants, AOPP treatment in combination with thiamine strongly reduced the effect of thiamine‐mediated defence against M. graminicola (Fig. 6b). These results indicate that the biosynthesis of phenylpropanoids is involved in thiamine‐induced defence against M. graminicola.
One of the products of the phenylpropanoid pathway is lignin, which confers mechanical strength to plant secondary cell walls and thus contributes to basal defence against pathogens, including plant‐parasitic nematodes (Wuyts et al., 2007). It is well known that monolignols are efficiently polymerized by peroxidase with H2O2 consumption, leading to lignin formation (Pauwels et al., 2008). We hypothesized that the increased phenylpropanoid and H2O2 generation in thiamine‐treated plants could lead to increased lignin production. In non‐inoculated plants, no significant differences in lignin levels were measured in thiamine‐treated versus non‐treated plants (Fig. 6c). However, significantly higher lignin levels were measured at 24 hpi in thiamine‐treated plants that were inoculated, in comparison with those that were inoculated but not treated, indicating that thiamine can induce cell wall lignification after pathogen challenge.
Discussion
Little is known about the molecular mechanism underlying thiamine‐induced defence against RKNs in agronomically important crops, such as rice. In the present study, we have demonstrated that application of thiamine protects rice from attack by the RKN M. graminicola. Moreover, this thiamine‐induced defence is associated with priming of the host tissue for accelerated activation of a multifaceted defence response, including enhanced PAL expression and the generation of H2O2 and lignin on nematode attack.
H2O2 formation has been linked to thiamine‐induced defence in various plant pathosystems (Ahn et al., 2005, 2007; Bahuguna et al., 2012). It has been demonstrated recently that thiamine can alleviate the inhibition of Arabidopsis ROS generation by Sclerotinia‐secreted oxalate (Zhou et al., 2013). Our study confirms that thiamine alone does not induce H2O2 accumulation. However, a rapid accumulation of H2O2 was observed in the roots of thiamine‐treated plants after nematode inoculation, suggesting a priming effect mediated by thiamine on the nematode‐induced oxidative burst. It is well established that increased production of H2O2 leads to polymerization of monolignols by peroxidase and subsequent lignin formation (Boerjan et al., 2003). Because lignin deposition reinforces cell walls and makes them less accessible to cell wall‐degrading enzymes secreted by the migrating nematodes, lignin is considered to function as a physical barrier against nematode attack (Gheysen and Jones, 2006). Arabidopsis containing elevated levels of syringyl lignin significantly reduced the development of the RKN M. incognita (Wuyts et al., 2006). Higher lignin levels were observed in the wild banana Musa acuminata, which is resistant to the burrowing nematode Radopholus similis, in comparison with susceptible banana (Wuyts et al., 2007). In compatible and incompatible tomato plants, Reactive Oxygen Species were visualized to be accumulated in nematode‐penetrated cells, and H2O2 was detected to be accumulated especially in the cells neighbouring the hypersensitive reaction (HR) (Melillo et al., 2006). In Arabidopsis, a direct requirement of NADPH oxidases for lignin polymerization in the endodermis was observed. Scaffolding of NADPH oxidases to the downstream targets of ROS was speculated to be a widespread mechanism to ensure subcellular precision of ROS action within the extracellular matrix (Lee et al., 2013). In our study, higher H2O2 induced by thiamine may be correlated with a higher level of lignin in the nematode‐infected plants. Similar results have been reported by Taheri and Tarighi (2010) for riboflavin‐treated rice plants infected with Rhizoctonia solani. The ability for a more rapid generation of H2O2 and lignin on infection may be part of priming the plant defence.
Lignin is formed through the phenylpropanoid pathway, which leads to the production of a wide range of phenolic compounds, such as flavonoids, plant hormones, phytoalexins and lignin (Kim and Hwang, 2014). PAL1 and cinnamoyl 4‐hydroxylase (C4H) function as marker genes in the phenylpropanoid pathway, and their expression changes in response to biotic and abiotic stresses, such as pathogens, UV irradiation and low temperature. These genes were repressed in giant cells induced by M. javanica in tomato, as well as M. graminicola in rice (Kyndt et al., 2012). In Arabidopsis, with increased levels of syringyl lignin, the reproduction of M. incognita in transgenic and mutant plants was lower than in wild‐type plants, whereas, in tobacco, life cycle progression was stimulated with reduced levels of syringyl lignin (Wuyts et al., 2006). In addition, ROS production is required to induce jasmonic acid (JA) accumulation in Arabidopsis during the early stage of cell wall damage. However, ROS derived from the NADPH oxidase form a negative feedback loop, which, in turn, seems to influence lignin accumulation during the second stage (Denness et al., 2011). In the present study, the transcript levels of OsPAL1 and OsC4H were upregulated in thiamine‐treated plants. Our observation of the decreased priming effect of thiamine in AOPP‐treated plants further confirms the key role of the phenylpropanoid pathway in activating defence in rice. These observations are consistent with a number of recent findings showing that the phenylpropanoid pathway is involved in the induction of resistance in pathogen–plant interactions. Using BABA as the inducer of resistance to downy mildew (Plasmopara viticola) in grapevine, Hamiduzzaman et al. (2005) showed that this was associated with the primed deposition of callose and phenylpropanoid‐derived phenolics. After application of methyl jasmonate (MeJA) on Arabidopsis thaliana, Pauwels et al. (2008) measured an increase in lignin and higher expression of the genes involved in phenylpropanoid and monolignol biosynthesis. Fujimoto et al. (2015) found that sclareol induced resistance in Arabidopsis against M. incognita, and that this was correlated with higher transcript levels of the lignin biosynthetic genes PAL1, C4H and cinnamoyl‐CoA reductase (CCR2).
Callose is an amorphous, high‐molecular‐weight β‐1,3‐glucan that is deposited in the form of papillae as a defence response on infection by pathogens (Ali et al., 2013; Hamiduzzaman et al., 2005). Our results showed that thiamine treatment did not increase significantly callose deposition following root inoculation with nematodes. However, treatment with another defence‐inducing agent, BABA, did induce strong callose deposition in rice plants on RKN infection (Ji et al., 2015) and in other plant pathosystems (Ton and Mauch‐Mani, 2004). These results indicate that priming mechanisms can vary among different chemicals.
After the successful establishment of a feeding site, RKNs take up nutrients and water from giant cells. In cotton cultivars resistant to M. incognita, Mota et al. (2013) observed underdeveloped giant cells with a degenerated cytoplasmic content. No apparent morphological differences in the giant cells developing in the thiamine‐treated roots versus the control roots were observed in our study, despite a reduced number of nematodes and delayed nematode development in thiamine‐treated plants. We therefore speculate that thiamine‐activated lignin synthesis hinders nematode penetration at the early stages of infection and later suppresses the nutrient supply to giant cells, delaying nematode development.
In conclusion, this study demonstrates that thiamine can induce resistance against M. graminicola in rice, and that this thiamine‐induced defence is associated with priming of the host for H2O2 accumulation, activation of the phenylpropanoid pathway and lignin accumulation on nematode attack. Further cellular and molecular studies on the defence responses activated by thiamine against M. graminicola or other nematodes will not only improve our knowledge of the mechanism of vitamin‐induced defence, but will also be useful in the development of novel nematode control strategies.
Experimental Procedures
Chemicals and plant materials
Thiamine was purchased from Sigma‐Aldrich (Ghent, Belgium) and dissolved in distilled water. All experiments were performed using a highly susceptible rice cultivar (O. sativa cv. Nipponbare) grown in sand and synthetic absorbent polymer (SAP) (Reversat et al., 1999), at 26°C under a 16h/8h light regime as described in Ji et al. (2015). Each plant was watered twice per week with 20 mL of Hoagland solution. Two‐week‐old plants were used for nematode inoculation. All infection experiments were performed three times independently with six replicate individuals per treatment.
Nematode culture and extraction
Meloidogyne graminicola was maintained on Nipponbare in potting soil under the same conditions as described above. Four weeks after inoculation, infected roots were cut into 1‐cm pieces, and nematodes were extracted using a modified flotation–sieving method (Byrd et al., 1966). The nematode suspension was washed through nested 100‐μm and 25‐μm sieves 48 h later. The second‐stage juveniles (J2s) were resuspended in distilled water and counted under a stereomicroscope.
Evaluation of direct effects of thiamine on nematode viability and infectivity
Approximately 200 J2s were placed into a 3.5‐cm‐diameter well in a six‐well culture plate containing 1 mL of 0, 0.6, 1.2, 2.5, 5.0 or 10.0 mm thiamine. After incubation for 24 h and 72 h, several drops of 1 M NaOH were added to the solution. Nematodes that responded to NaOH by changing their body shape within 3 min were considered to be alive, whereas straight nematodes that failed to respond were classified as dead (Chen and Dickson, 2000). The alive/dead nematodes were counted under a stereomicroscope (LeicaS8APO, Leica Microsystems, Diegem, Belgium). Each treatment included six replicates, and the whole experiment was performed three times.
To determine the direct effect of thiamine on the infectivity of the nematodes, the nematodes were incubated in 2.5 mm thiamine solution or water for 72 h. Then, 200 J2s were inoculated on 2‐week‐old rice plants in SAP. At 3 dpi, the plants were transplanted into a hydroponic system containing Hoagland solution for another 11 days. At 7 and 14 dpi, the root samples were cleared for 5 min in 0.6% NaClO and boiled for 3 min in 0.8% acetic acid and 0.013% acid fuchsin (Bybd et al., 1983; Nahar et al., 2011). After destaining in 4% acidified glycerol, the nematodes inside the roots were visualized using a stereomicroscope. The ratio of the number of nematodes in each developmental stage to the total number of nematodes was calculated using Microsoft (Redmon, Washington, USA) Excel 6.0 software.
Attraction tests
Attraction bioassays were performed at room temperature (22 ± 3 °C) according to Williamson et al. (2009). Briefly, 23 g Pluronic F‐127 powder (Sigma‐Aldrich, Belgium) was added to 100 mL of sterile water at 4 °C and allowed to dissolve whilst stirring for 24 h. The roots of 2‐week‐old rice plants were soil drenched with 2.5 mm thiamine or water. One day later, a 1‐cm‐long root tip was cut and placed into the well of a six‐well culture plate containing 1 mL Pluronic gel and approximately 200 J2s. Quantification of the attracted nematodes was performed using a Leica stereomicroscope with a DFC400 camera at 9 hpi. The whole experiment was performed three times, with six replicates.
Priming experiments
Each 2‐week‐old rice plant in SAP was root drenched with 20 mL of 1.2–10 mm thiamine or the same volume of water, 1 day before inoculation. Then, each plant was inoculated with 200 J2s. At 14 dpi, the root length, shoot length and fresh weight of the plants were measured, and the root samples were stained using acid fuchsin, as described above. Nematodes at different developmental stages were counted using a Leica stereomicroscope.
To test whether the resistance induced by thiamine depends on the accumulation of phenylpropanoids, an inhibitor of phenylpropanoid biosynthesis, 100 μm of AOPP, was applied to the roots in combination with or without 2.5 mm thiamine. One day later, 200 J2s were inoculated on each plant, and the number of nematodes in the roots was counted at 14 dpi as described above. The whole experiment was performed three times, with six individual plants in each replicate.
Microscopic observation of giant cells
Giant cells from root galls were collected at 7 dpi and were microscopically analysed according to Ji et al. (2013). The experiment was performed twice, and 10 galls from six plants in each treatment were sectioned and observed under the microscope.
Callose detection
Stained root galls were examined for callose deposition as described by Ji et al. (2015). Quantification of callose was performed using Image J software. Ten galls from each treatment were observed and analysed.
Quantitative analysis of H2O 2 and lignin
The in planta accumulation of H2O2 was determined according to Velikova et al. (2000). One day before inoculation, each 2‐week‐old plant was drenched with 20 mL of 2.5 mm thiamine or water. Each sample consisted of approximately 0.1 g fresh roots, collected from a pool of six plants. Root samples were collected at 6, 24 and 72 hpi with M. graminicola (Fig. S2), and the samples were processed according to Ji et al. (2015). The experiment was performed twice, each time with four replicate samples.
The in planta accumulation of lignin was quantified as described by Ji et al. (2015). Two‐week‐old plants were drenched with 20 mL of 2.5 mm thiamine or water, 1 day before inoculation. Fresh roots were collected before inoculation and 1 day after inoculation. Each root sample was collected from a pool of six individual plants. Each experiment was performed twice using four replicate samples.
RNA extraction, reverse transcription and qRT‐PCR analysis
Root tissues were collected at 6, 24 and 72 hpi. Control samples were collected at the same time (Fig. S2). Each sample consisted of a pool of six individual plants. For each treatment, two independent biological replicates were sampled. RNA was extracted using the NucleoSpin kit (Macherey‐Nagel, Düren, Germany), and cDNA was synthesized using the SuperScript® II Reverse Transcriptase Kit (Invitrogen, Karlsruhe, Germany), as described by Kyndt et al. (2012). The primer sequences for the amplification of the defence‐related genes used in the qRT‐PCR analysis are listed in Table 1. All qRT‐PCRs were performed in triplicate. The PCR conditions consisted of an initial denaturation at 95 °C for 5 min, followed by 40 cycles of denaturation at 95 °C for 25 s, annealing at 58 °C for 40 s and extension at 72 °C for 25 s. The data obtained from qRT‐PCR were analysed using the software Rest 2009. Gene expression levels were normalized using data from three internal reference genes (Table 1). The relative expression level of each gene is shown as the fold change in the transcript level compared with the expression level in the untreated and uninfected control plants (set at an expression level of 1).
Table 1.
Overview of the primers used in this study for quantitative reverse transcription polymerase chain reaction (qRT‐PCR), showing GenBank accession/locus numbers (MSU7.0) of the reference and target genes
| Genes | GenBank accession or locus number | Primer sequences (5′→3′) | Function |
|---|---|---|---|
| OsEif5C | LOC_Os11g21990 |
F: CACGTTACGGTGACACCTTTT R: GACGCTCTCCTTCTTCCTCAG |
Reference |
| OsEXP | LOC_Os03g27010 |
F: TGTGAGCAGCTTCTCGTTTG R: TGTTGTTGCCTGTGAGATCG |
Reference |
| OsEXPnarsai | LOC_Os07g02340 |
F: AGGAACATGGAGAAGAACAAGG R: CAGAGGTGGTGCAGATGAAA |
Reference |
| OsWRKY45 | Os05g0323900 |
F: AATTCGGTGGTCGTCAAGAA R: AAGTAGGCCTTTGGGTGCTT |
Salicylic acid response |
| OsPAL1 | X16099.1 |
F: TGTGCGTGCTTCTGCTGCTG R: AGGGTGTTGATGCGCACGAG |
Phenylpropanoid biosynthesis |
| OsC4H | NM_001061725 |
F: CAGACTGGTGAGATCCGGTG R: TTCCCCATTCGATCGACCAC |
Phenylpropanoid biosynthesis |
| OsCAD6 | NM_001058825 |
F: TCGGTAAGAGGACGGTGAGT R: TGTCGATGTCCCAGGTGATG |
Phenylpropanoid biosynthesis |
| OsGSL1 | AP001389 |
F: TGAGGACCTGCCACGATT R: CACGCTGATTGCGAACAT |
Callose production |
| OsRbohB | NM001049555.1 |
F: CTGGACAGGACCAAGAGCAG R: ATCTTGAACGGAGCAGCACA |
H2O2 production |
Statistical analysis
The means and standard errors of the data obtained from the in vitro bioassay, infection experiments, callose detection and lignin quantification were subjected to statistical analysis using SPSS version 21. The significant differences (P ≤ 0.05) between the treatments were determined according to Duncan's multiple range test.
Supporting information
Fig. S1 Effects of different concentrations of thiamine on Meloidogyne graminicola. Root knots and nematodes in thiamine‐incubated (VB1) and water‐incubated rice roots were counted at 14 days post‐inoculation (dpi). The bars represent the means ± standard error (SE) of the data from two independent biological replicates, each containing six plants. Different letters indicate significant differences (Duncan's t‐test with P ≤ 0.05).
Fig. S2 Scheme of the treatments and sampling time points used in the quantitative reverse transcription‐polymerase chain reaction (qRT‐PCR) analyses and H2O2 measurements in this study. Open circles, time points for treatments. Filled circles, time points for thiamine treatment and nematode inoculation. Time 1 (four samples): (1) roots treated with 2.5 mm thiamine (vitamin B1, VB1) for 30 h (VB1); (2) roots treated with 2.5 mm VB1 for 30 h and inoculated with Meloidogyne graminicola for 6 h (VB1 + Mg); (3) roots treated with water for 30 h and inoculated with M. graminicola for 6 h (Mg); (4) control roots treated with water for 30 h. Time 2 (four samples): (1) roots treated with 2.5 mm VB1 for 48 h (VB1); (2) roots treated with 2.5 mm VB1 for 48 h and inoculated with M. graminicola for 24 h (VB1 + Mg); (3) roots treated with water for 48 h and inoculated with M. graminicola for 24 h (Mg); (4) control roots treated with water for 48 h. Time 3 (four samples): (1) roots treated with 2.5 mm VB1 for 96 h (VB1); (2) roots treated with 2.5 mm VB1 for 96 h and inoculated with M. graminicola for 72 h (VB1 + Mg); (3) roots treated with water for 96 h and inoculated with M. graminicola for 72 h (Mg); (4) control roots treated with water for 96 h.
Acknowledgements
We acknowledge the financial support of GOA 01 GB3013, the National Basic Research Program of China (2013CB127502) and the Special Project for Public Agriculture Research and Development of the Ministry of Agriculture (201103018). TK was supported by a FWO postdoctoral fellowship. WKH was funded by the China Scholarship Council. We are grateful to Dr Kristof De Schutter for his assistance in the callose observations and H2O2 measurements.
References
- Ahn, I.P. , Kim, S. and Lee, Y.H. (2005) Vitamin B1 functions as an activator of plant disease resistance. Plant Physiol. 138, 1505–1515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn, I.P. , Kim, S. , Lee, Y.H. and Suh, S.C. (2007) Vitamin B1‐induced priming is dependent on hydrogen peroxide and the NPR1 gene in Arabidopsis. Plant Physiol. 143, 838–848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali, M.A. , Abbas, A. , Kreil, D.P. and Bohlmann, H. (2013) Overexpression of the transcription factor RAP2.6 leads to enhanced callose deposition in syncytia and enhanced resistance against the beet cyst nematode Heterodera schachtii in Arabidopsis roots. BMC Plant Biol. 13, 47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asensi‐Fabado, M.A. and Munné‐Bosch, S. (2010) Vitamins in plants: occurrence, biosynthesis and antioxidant function. Trends Plant Sci. 15, 582–592. [DOI] [PubMed] [Google Scholar]
- Bahuguna, R.N. , Joshi, R. , Shukla, A. , Pandey, M. and Kumar, J. (2012) Thiamine primed defense provides reliable alternative to systemic fungicide carbendazim against sheath blight disease in rice (Oryza sativa L). Plant Physiol. Biochem. 57, 159–167. [DOI] [PubMed] [Google Scholar]
- Boerjan, W. , Ralph, J. and Baucher, M. (2003) Lignin biosynthesis. Annu. Rev. Plant Biol. 54, 519–546. [DOI] [PubMed] [Google Scholar]
- Burrows, R.J. , Byrne, K.L. and Meacock, P.A. (2000) Isolation and characterization of Saccharomyces cerevisiae mutants with derepressed thiamine gene expression. Yeast, 16, 1497–1508. [DOI] [PubMed] [Google Scholar]
- Bybd, D.W. Jr. , Kirkpatrick, T. and Barker, K.R. (1983) An improved technique for clearing and staining plant tissues for detection of nematodes. J. Nematol. 15, 142–143. [PMC free article] [PubMed] [Google Scholar]
- Byrd, D.W. , Nusbaum, C.J. and Barker, K.R. (1966) A rapid flotation‐sieving technique for extracting nematodes from soil. Plant Dis. Rep. 50, 954–957. [Google Scholar]
- Chen, S.Y. and Dickson, D.W. (2000) A technique for determining live second‐stage juveniles of Heterodera glycines . J. Nematol. 32, 117–121. [PMC free article] [PubMed] [Google Scholar]
- Conrath, U. , Beckers, G.J. , Flors, V. , García‐Agustín, P. , Jakab, G. , Mauch, F. , Newman, M.A. , Pieterse, C.M. , Poinssot, B. , Pozo, M.J. , Pugin, A. , Schaffrath, U. , Ton, J. , Wendehenne, D. , Zimmerli, L. , Mauch‐Mani, B. and Prime‐A‐Plant Group (2006) Priming: getting ready for battle. Mol. Plant–Microbe Interact. 19, 1062–1071. [DOI] [PubMed] [Google Scholar]
- Denness, L. , McKenna, J.F. , Segonzac, C. , Wormit, A. , Madhou, P. , Bennett, M. , Mansfield, J. , Zipfel, C. and Hamann, T. (2011) Cell wall damage‐induced lignin biosynthesis is regulated by a reactive oxygen species‐ and jasmonic acid‐dependent process in Arabidopsis. Plant Physiol. 156 (3), 1364–1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto, T. , Mizukubo, T. , Abe, H. and Seo, S. (2015) Sclareol induces plant resistance to root‐knot nematode partially through ethylene‐dependent enhancement of lignin accumulation. Mol. Plant–Microbe Interact. 28, 398–407. [DOI] [PubMed] [Google Scholar]
- Gheysen, G. and Jones, J. (2006) Molecular aspects of plant–nematode interactions In: Plant Nematology (Perry R.N. and Moens M., eds), pp. 234–254, Wallingford, Oxfordshire: CAB International. [Google Scholar]
- Hamiduzzaman, M.M. , Jakab, G. , Barnavon, L. , Neuhaus, J.M. and Mauch‐Mani, B. (2005) beta‐Aminobutyric acid‐induced resistance against downy mildew in grapevine acts through the potentiation of callose formation and jasmonic acid signaling. Mol. Plant–Microbe Interact. 18, 819–829. [DOI] [PubMed] [Google Scholar]
- Ji, H.L. , Gheysen, G. , Denil, S. , Lindsey, K. , Topping, J.F. , Nahar, K. , Haegeman, A. , De Vos, W.H. , Trooskens, G. , Van Criekinge, W. , De Meyeter, T. and Kyndt, T. (2013) Transcriptional analysis through RNA sequencing of giant cells induced by Meloidogyne graminicola in rice roots. J. Exp. Bot. 64, 3885–3898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji, H.L. , Kyndt, T. , He, W. , Vanholme, B. and Gheysen, G. (2015) β‐Aminobutyric acid‐induced resistance against root‐knot nematodes in rice is based on increased basal defence. Mol. Plant–Microbe Interact. 28, 519–533. Available at 10.1094/MPMI-09-14-0260-R. [DOI] [PubMed] [Google Scholar]
- Jung, I.L. and Kim, I.G. (2003) Thiamine protects against paraquat‐induced damage: scavenging activity of reactive oxygen species. Environ. Toxicol. Pharmacol. 15, 19–26. [DOI] [PubMed] [Google Scholar]
- Kim, D.S. and Hwang, B.K. (2014) An important role of the pepper phenylalanine ammonia‐lyase gene (PAL1) in salicylic acid‐dependent signalling of the defence response to microbial pathogens. J. Exp. Bot. 65, 2295–2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyndt, T. , Nahar, K. , Haegeman, A. , De Vleesschauwer, D. , Höfte, M. and Gheysen, G. (2012) Comparing systemic defence‐related gene expression changes upon migratory and sedentary nematode attack in rice. Plant Biol. (Stuttg.) 14 (Suppl. 1), 73–82. [DOI] [PubMed] [Google Scholar]
- Kyndt, T. , Vieira, P. , Gheysen, G. and de Almeida‐Engler, J. (2013) Nematode feeding sites: unique organs in plant roots. Planta, 238, 807–818. [DOI] [PubMed] [Google Scholar]
- Kyndt, T. , Fernandez, D. and Gheysen, G. (2014) Plant‐parasitic nematode infections in rice: molecular and cellular insights. Annu. Rev. Phytopathol. 52, 135–153. [DOI] [PubMed] [Google Scholar]
- Lee, Y. , Rubio, M.C. , Alassimone, J. and Geldner, N. (2013) A mechanism for localized lignin deposition in the endodermis. Cell, 153, 402–412. [DOI] [PubMed] [Google Scholar]
- Melillo, M.T. , Leonetti, P. , Bongiovanni, M. , Castagnone‐Sereno, P. and Bleve‐Zacheo, T. (2006) Modulation of reactive oxygen species activities and H2O2 accumulation during compatible and incompatible tomato–root‐knot nematode interactions. New Phytol. 170, 501–512. [DOI] [PubMed] [Google Scholar]
- Mota, F.C. , Alves, G.C.S. , Giband, M. , Gomes, A.C.M.M. , Sousa, F.R. , Mattos, V.S. , Barbosa, V.H.S. , Barroso, P.A.V. , Nicole, M. , Peixoto, J.R. , Rocha, M.R. and Carneiro, R.M.D.G. (2013) New sources of resistance to Meloidogyne incognita race 3 in wild cotton accessions and histological characterization of the defence mechanisms. Plant Pathol. 62, 1173–1183. [Google Scholar]
- Nahar, K. , Kyndt, T. , De Vleesschauwer, D. , Höfte, M. and Gheysen, G. (2011) The jasmonate pathway is a key player in systemically induced defense against root knot nematodes in rice. Plant Physiol. 157, 305–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauwels, L. , Morreel, K. , De Witte, E. , Lammertyn, F. , Van Montagu, M. , Boerjan, W. , Inzé, D. and Goossens, A. (2008) Mapping methyl jasmonate‐mediated transcriptional reprogramming of metabolism and cell cycle progression in cultured Arabidopsis cells. Proc. Natl. Acad. Sci. USA, 105, 1380–1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pushpalatha, H.G. , Sudisha, J. , Geetha, N.P. , Amruthesh, K.N. and Shekar Shetty, H. (2011) Thiamine seed treatment enhances LOX expression, promotes growth and induces downy mildew disease resistance in pearl millet. Biol. Plant. 55, 522–527. [Google Scholar]
- Reversat, G. , Boyer, J. , Pando‐Bahuon, A. and Sannier, C. (1999) Use of a mixture of sand and water‐absorbent synthetic polymer as substrate for the xenic culturing of plant‐parasitic nematodes in the laboratory. Nematology, 1, 209–212. [Google Scholar]
- Taheri, P. and Tarighi, S. (2010) Riboflavin induces resistance in rice against Rhizoctonia solani via jasmonate‐mediated priming of phenylpropanoid pathway. J. Plant Physiol. 167, 201–208. [DOI] [PubMed] [Google Scholar]
- Ton, J. and Mauch‐Mani, B. (2004) Beta‐amino‐butyric acid‐induced resistance against necrotrophic pathogens is based on ABA‐dependent priming for callose. Plant J. 38, 119–130. [DOI] [PubMed] [Google Scholar]
- Velikova, V. , Yordanov, I. and Edreva, A. (2000) Oxidative stress and some antioxidant systems in acid rain‐treated bean plants. Plant Sci. 151, 59–66. [Google Scholar]
- Williamson, V. , Wang, C. and Lower, S. (2009) Application of Pluronic gel to the study of root‐knot nematode behaviour. Nematology, 11, 453–464. [Google Scholar]
- Wong, H.L. , Pinontoan, R. , Hayashi, K. , Tabata, R. , Yaeno, T. , Hasegawa, K. , Kojima, C. , Yoshioka, H. , Iba, K. , Kawasaki, T. and Shimamoto, K. (2007) Regulation of rice NADPH oxidase by binding of Rac GTPase to its N‐terminal extension. Plant Cell, 19, 4022–4034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuyts, N. , Lognay, G. , Swennen, R. and De Waele, D. (2006) Nematode infection and reproduction in transgenic and mutant Arabidopsis and tobacco with an altered phenylpropanoid metabolism. J. Exp. Bot. 57, 2825–2835. [DOI] [PubMed] [Google Scholar]
- Wuyts, N. , Lognay, G. , Verscheure, M. , Marlier, M. , De Waele, D. and Swennen, R. (2007) Potential physical and chemical barriers to infection by the burrowing nematode Radopholus similis in roots of susceptible and resistant banana (Musa spp.). Plant Pathol. 56, 878–890. [Google Scholar]
- Zhou, J. , Sun, A. and Xing, D. (2013) Modulation of cellular redox status by thiamine‐activated NADPH oxidase confers Arabidopsis resistance to Sclerotinia sclerotiorum . J. Exp. Bot. 64, 3261–3272. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig. S1 Effects of different concentrations of thiamine on Meloidogyne graminicola. Root knots and nematodes in thiamine‐incubated (VB1) and water‐incubated rice roots were counted at 14 days post‐inoculation (dpi). The bars represent the means ± standard error (SE) of the data from two independent biological replicates, each containing six plants. Different letters indicate significant differences (Duncan's t‐test with P ≤ 0.05).
Fig. S2 Scheme of the treatments and sampling time points used in the quantitative reverse transcription‐polymerase chain reaction (qRT‐PCR) analyses and H2O2 measurements in this study. Open circles, time points for treatments. Filled circles, time points for thiamine treatment and nematode inoculation. Time 1 (four samples): (1) roots treated with 2.5 mm thiamine (vitamin B1, VB1) for 30 h (VB1); (2) roots treated with 2.5 mm VB1 for 30 h and inoculated with Meloidogyne graminicola for 6 h (VB1 + Mg); (3) roots treated with water for 30 h and inoculated with M. graminicola for 6 h (Mg); (4) control roots treated with water for 30 h. Time 2 (four samples): (1) roots treated with 2.5 mm VB1 for 48 h (VB1); (2) roots treated with 2.5 mm VB1 for 48 h and inoculated with M. graminicola for 24 h (VB1 + Mg); (3) roots treated with water for 48 h and inoculated with M. graminicola for 24 h (Mg); (4) control roots treated with water for 48 h. Time 3 (four samples): (1) roots treated with 2.5 mm VB1 for 96 h (VB1); (2) roots treated with 2.5 mm VB1 for 96 h and inoculated with M. graminicola for 72 h (VB1 + Mg); (3) roots treated with water for 96 h and inoculated with M. graminicola for 72 h (Mg); (4) control roots treated with water for 96 h.


