Abstract
The incidences of cancer are continuously increasing worldwide, affecting life of millions of people. Several factors associated with the internal and external environment are responsible for this deadly disease. The key internal determinants like abnormal hormonal regulation, genetic mutations and external determinants such as lifestyle and occupational factors enhances onset of cancer. From the ancient time, plants were remained as the most trusted source of medicine for the treatment of diverse disease conditions. Extensive studies have been performed for the discovery of effective anticancer agent from the plant and still it is going on. Pentacyclic triterpenoids are biologically active phytochemicals having a different range of activities such as anti-inflammatory, hepatoprotective, anti-hypertensive, antiulcerogenic and anti-tumor. These compounds generally contain ursane, oleanane, lupane and friedelane as a chief skeleton of pentacyclic triterpenoids which are generally present in higher plants. Isoprene unit, phytochemical, with good antitumor/anticancer activity is required for the biosynthesis of pentacyclic triterpenoids. Mechanisms such as cytotoxicity, DNA polymerase inhibition, regulation of apoptosis, change in signal transductions, interfere with angiogenesis and dedifferentiation, antiproliferative activity and metastasis inhibition are might be responsible for their anticancer effect. Present review spotlights diverse targets, mechanisms and pathways of pentacyclic triterpenoids responsible for anticancer effect.
Keywords: cancer, pentacyclic triterpenoids, apoptosis, anti-angiogenic, isoprene, antiproliferative
1. Introduction
Continuous increase in the cancer incidences and mortality in developed and developing countries is a serious concern worldwide. Cancer is the second major cause of mortality after the cardiovascular diseases [1]. Among the different types of cancer, the prevalence of lung cancer is more common in man and breast cancer in women [2]. Several factors contribute to the development of cancer, which may be internal such as genetic alteration, mutation in the genetic material, abnormal hormonal condition and compromised immune system; and external causes may be the environmental factors including the modern lifestyle and specific occupation conditions. The incidences of oral cancer are more in India and about among 30 to 40% of Indians are accounting for oral cancer [3].
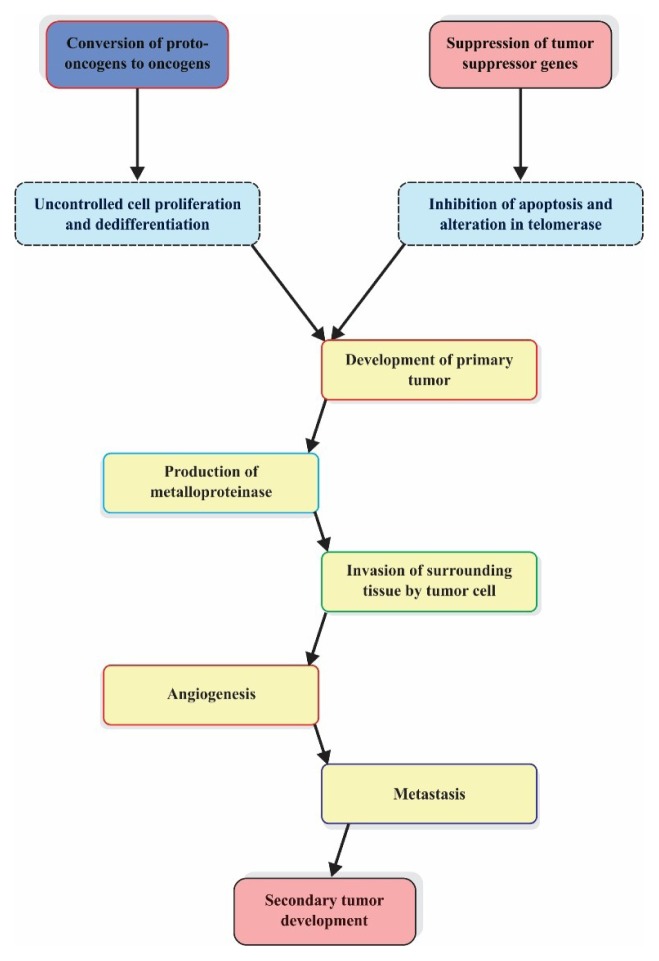
Cancer cells are characterized by abnormal, uncontrolled cell proliferation and have the ability to invade in surrounding organ tissue, called metastasis. Carcinogenesis is a complex multistep process that usually involves several genetic alterations and other epigenetic factors (Fig. 1). One or multiple mutations in host cell DNA translate normal cell into the cancer cell [2]. The mutation in the DNA could be inherited or acquired. One of the examples of acquired mutation is breast cancer where inheritance of a single defective copy of either of the tumor suppressor genes BRCA1 and BRCA2 increases chances of cancer development [4]. The turnover of cells in the body is regulated by apoptosis, a programmed cell death process and failure of apoptosis leads to abnormal cell multiplication and ultimately growth [5].
Figure 1.
Altered gene expression associated cancer development.
The use of plants as a source of medicine is as old as mankind itself. Plants remained the ultimate trusted source of medicine for disease prevention and cure from ancient time. More than 60% of anticancer drug currently used are obtained from natural sources. Most of modern drugs employed for cancer treatment were obtained from plant and possess strong ability to cure the cancer [6]. A report published by WHO showed that more than 80% population prefers natural products for their health needs. Literature survey also revealed that 60% people take herbs, vitamins, and herbal medicines to control cancer and associated complications. Use of medicinal plants can help to reduce toxic and side effects of chemotherapy and radiation treatment by reinforcing their anticancer effect [7]. It is estimated that in the last decade, approximately 30.69 billion USD plant derived drugs have been estimated in the global market [8]. Several phytochemicals produce strong anti-inflammatory and antioxidative actions mediated through Nrf2, NF-κB, and AP-1 signalling pathways [9]. Similarly, phytochemicals with anticancer potential can prevent cancer initiation, promotion and progression by interfering various intermittent mechanisms and pathways [10]. Near about 12.4 billion USD terpenes and steroids obtained from the plant were sale annually worldwide [11].
Several studies have revealed biological potential of triterpenoids such as anti-inflammatory, antisepsis, anti-fatigue, anti-tumor, anti-hyperlipidemic, antihypertension, anti-hyperglycemia, anti-ulcerogenic, anti-plasmodiac, antiviral, analgesic and immunomodulatory [12]. Asiatic acid was studied for the management of memory associated problems such as memory and dementia. Ursolic acid has been reported for antioxidant activity and protective effect against D-galactose-induced neurotoxicity [13]. The hepatoprotective effect of oleanolic acid is successfully implemented in the China and it is an over the counter oral drug to treat human for liver disorder [14, 15]. α and β amyrin were found to possess different biological activity such as anti-tumor, gastro-protective, hepatoprotective and anti-inflammatory activity [16]. Many pentacyclic triterpenoids have potential to inhibit tumor formation which mainly includes ursolic acid, betulinic acid, betulin, glycyrrhetinic and boswellic acid [17, 18]. Also glycyrrhetic acid, asiaticoside, oleanolic acid and carbenoxolone (a synthetic derivative of glycyrrhetic acid) has been successfully demonstrated clinically as marketed drugs [17].
2. Cancer epidemiology
Cancer epidemiology and associated mortality are at its highest peak. According to the American Cancer Society in the US, about 1,685,210 new cancer cases are expected to be diagnosed in 2016 [19]. It is estimated that the burden of cancer will increase to 23.6 million new cases each year by 2030. In 2008, 7.6 million deaths (around 13% of all deaths) occurred worldwide because of cancer. Although death is due to different types of cancer, but most common is lung cancer in men (16.5% of all new cases in men) and breast cancer in women (23% of all new cases in women) [20]. The American Cancer Society has estimated that in 2013 about 174,100 deaths occurred might be due to tobacco associated cancer. In 1971, 1 in 91 people was a cancer survivor and in 2012, 1 in 23 people were cancer survivor [21]. In 2002, about 4.2 million of new cancer cases among 3.2 billion of people were diagnosed in India, China, Sri Lanka, Japan, Singapore, Taiwan, Malaysia, Thailand, Philippines, Vietnam, Indonesia, Mongolia, Laos and Cambodia. These countries belong to Southeast, East and South Asia. China and India together accounting about 37% of world population and near about 3 million of new cancer cases were reported [22, 23].
Breast cancer in female and lung cancer in male is the most common types of cancer in Indian population. Cervix cancer ranks third among all types of cancer. Tobacco is a major contributing cause in all these types of cancer in men and women [22, 24]. In India, every year about 850,000 new cancer cases are being diagnosed and 580,000 deaths are occurring [2]. Most of the cancer deaths are in the age group of 30–69 years out of which 200,100 and 195,300 were men and women respectively. Most common fatal cancer types are oral (22.9% of cancer), stomach (12.6% of cancer) and lung (11.4% of cancer) in men and cervical (17.1% of cancer), stomach (14.1% of cancer) and breast (10.2% of cancer) cancer in women. According to International Agency for Research on cancer during 2008 about 635000 people died from cancer which represents about 8% of all estimated global cancer deaths. The cancer associated deaths projected to increase continuously because of increasing population and life expectancy [25, 26].
3. Pentacyclic triterpenoids
Triterpenes are the terpenes consisting of six isoprene units and have 30 carbon atoms. These are secondary metabolites of plant which are extensively present in the plant kingdom. Various plant parts such as leaves, fruit peel and stem bark contains triterpenoids [16]. In higher plant and organism isoprene (2-methylbutadiene) unit is used for the biosynthesis of terpenes and terpenoids. The basic molecular formula for terpenes is multiples of C5H8 [27].
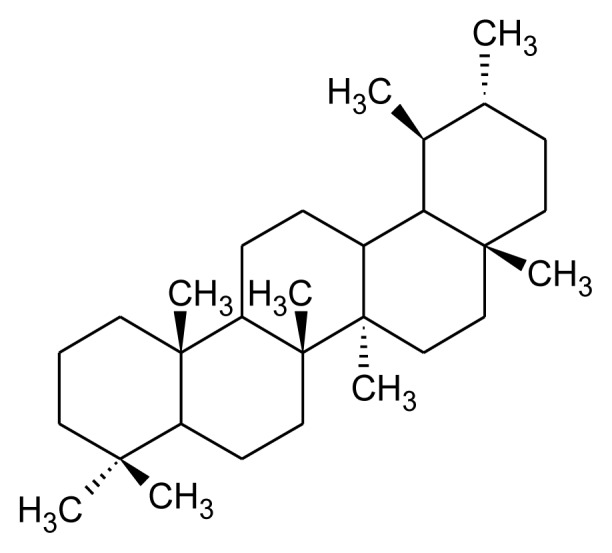
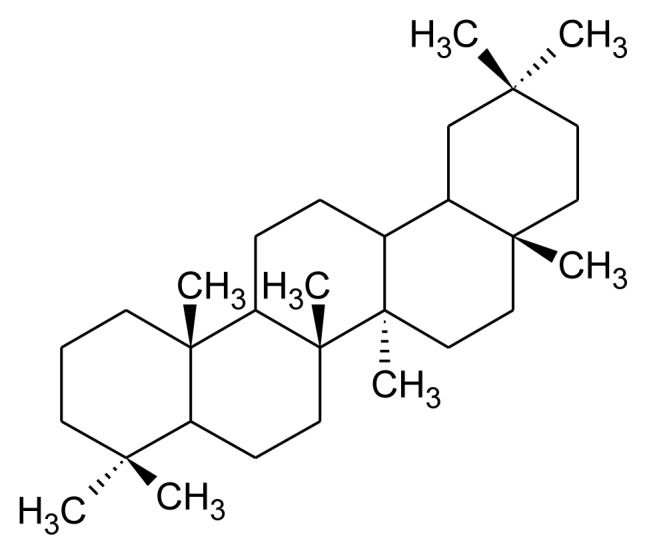
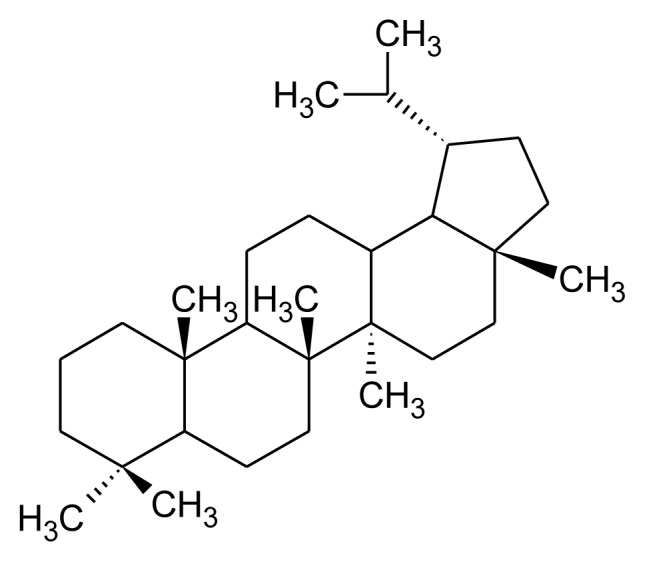
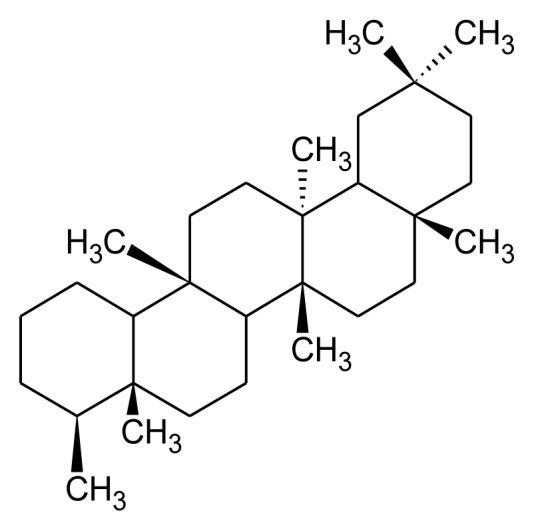
Tetracyclic and pentacyclic are two principal groups of triterpenoids. Tetracyclic triterpenoids contain methylated steroid (oleandrin, euphol and cucubitacins) whereas pentacyclic triterpenoids further differentiated into friedelane, lupane, ursane, oleane, serratane, and taraxastane category (Table 1). Higher plants such as edible plant largely distributed with ursane and oleanane. These are common and structurally diverse group of natural compounds which exhibit about 200 diverse skeletons. 6-6-6-5 tetracycles, 6-6-6-6-5 and 6-6-6-6-6 pentacycles are the most important triterpenoids [13, 18] Squalene is required for biosynthesis of commonly occurring polycyclic triterpenoids through multiple steps such as cyclization, small molecule loss and ring expansions or contraction (Figure 2). Biosynthesis of triterpenoids requisite highly specific enzymes [28–30]. Head to head condensation of two C15 units of farnesyl diphosphate also produces polycyclic triterpenoids [11]. Carbon skeleton like oleanane, ursane, lupine and dammarane-euphane are considered to be the most important triterpenoid structure [11, 13].
Table 1.
Major class of pentacyclic triterpenoid, their structure and examples.
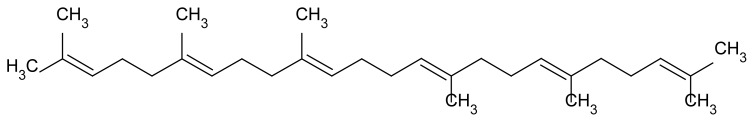
Figure 2.
Chemical structure of squalene.
Triterpenoids also exist in the form of triterpenoids-saponins, which is a combined form triterpenoid with sugar [30]. Squalene is a most common precursor required for the biosynthesis of steroid and triterpenoids. Serratene, a pentacyclic triterpenoid having central seven membered ring and seven tertiary methyl groups (21β-hydroxy-serrat-14-en-3α-oland 21 β-hydroxy-serrat-14-en-3α-yl acetate) shows anti-tumor activity [31, 32]. The pentacyclic triterpenoid ring has been reported for the inhibitory activity of topoisomerase [18].
4. Physiology of cancer
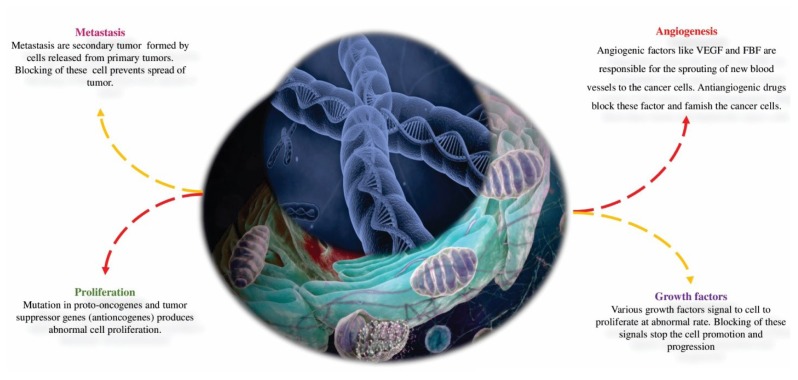
Human body made up of cells having little or no capacity to divide and proliferate like neurons and continuously dividing cells, such as bone marrow cells. Hence, it is not true that cancerous cells grow faster than normal cells because plasma cell tumor multiplies slowly whereas the cells of Burkitt’s lymphoma multiply faster [39]. Cancer cells are featured by fast multiplication or abnormal cell division and growth regulatory mechanisms (Figure 3). Normal host cell and cancer cell are distinguished by proliferation rate and cancer cells generally complement the biomolecules that are necessary for survival or proliferation, differentiation, cell death, and expression of many cell-type-specific functions. Improper regulation of controlled cell proliferation, distorted cell differentiation mechanism, deterioration of genetic and chromosomal organization, and disturbed programmed cell death pathways such as apoptosis could be responsible for the altered phenotype and formation of cancer [40, 41]. Hence, cancer or tumor formation could be controlled by controlling cell proliferation, differentiation, cell death and expression of many cell-type-specific functions (Figure 3). This mainly includes regulation of apoptic pathways and the genesis of cancer cell, expression of telomerase, tumor related blood vessels and interference with signal transduction [4, 42].
Figure 3.
Principal pathways such as angiogenesis, metastasis, proliferation and growth factors as targets for the treatment of cancer.
5. Triterpenoids as anticancer agents
Triterpenoids exhibits diverse physiological and therapeutic actions such as antiangiogenic and dedifferentiation effects that are related with anticancer actions. Triterpenoic acid, monoalcohol and diols exhibits antioxidant effects by inhibiting reactive oxidative radicals. Many previous studies demonstrated that lupane, oleanane and ursane have potent anticancer action [43]. New therapeutic strategies are continuously emerging to combat cancer, which includes modification of the tumor environment, modulation of the immune response and inhibition of angiogenesis [28]. Different cellular receptors llike hydrocarbon receptor, androgen receptor and vascular endothelial growth factor receptor can be used as a target for the treatment of cancer [44, 45]. Triterpenoids have the ability to inhibit nuclear factor-κB (NF-κB) activation and signal transduction, cell proliferation, apoptosis, angiogenesis, mitochondrial dysfunction and modulation of MDR genes and proteins [30].
5.1. Cell proliferation and apoptosis
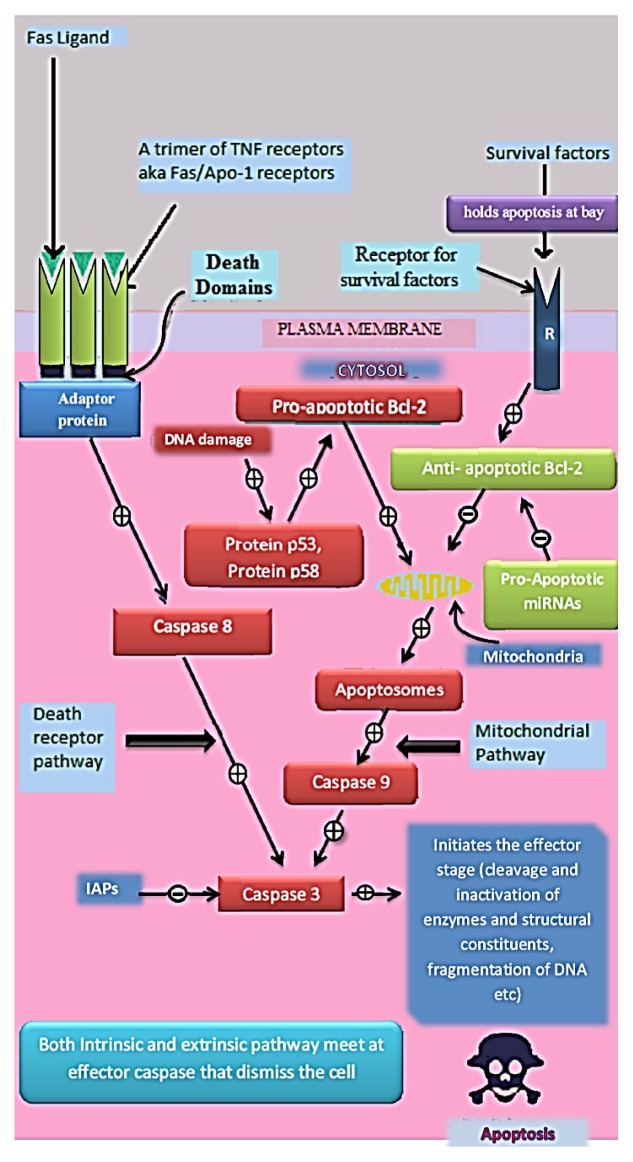
Cell proliferation is a process of increasing cell number, including cell development and cell division [46]. This intern responsible to increase in cell mass and ultimately the organism development. In certain cells, proliferation is restricted while it remains continue in some of the cells throughout lifetime. In certain situation cell proliferation becomes abnormal which further augment the tumor cell formation [40]. In the body, cell proliferation process is highly regulated by cell division and apoptosis [2]. Apoptosis is programmed cell death mechanism that destroys unwanted cells and also involved in defense mechanism of the body. It also plays a role in morphological and cellular mechanisms of cell, caspases functioning, translocation of phosphatidyl serine and DNA fragmentation [43, 47]. This complex process comprises intrinsic pathway and extrinsic pathway which are also called as mitochondrial pathway and cytoplasmic pathway or death receptor pathway, respectively (Fig. 4) [5]. In cancer, usually apoptosis pathways are irregular because of mutated genes or tumor suppressor gene or abnormal proto-oncogenes and oncogenes [4, 17].
Figure 4.
Extrinsic Pathway (Death receptor pathway) is activated when a tumor necrosis factor (TNF) receptor is stimulated by particular death ligands that recruits proteins (adaptor protein) and activate initiator caspases (caspases 8) which further activate effector caspases (caspase 3). Whereas, intrinsic pathway (mitochondrial pathway) activate other signals, such as being DNA damage which cannot be repaired, the p53 (tumor suppressor gene) protein activates a subpathway that leads to release of cytochrome c from the mitochondrion, with successive participation of the apoptosome [a complex of procaspase 9, cytochrome c and apoptic activating protease factor-1 (Apaf-1)], and activation of caspasen 3 (initiator caspases) and caspase 9.
R→ Receptor for growth factors, trophic factor and cell to cell contact factor (Continuous stimulation to this receptor is necessary for cell survival). Fas ligand→Trans membrane receptor that induce apoptosis. miRNAs→ micro RNAs that negatively regulate gene expression. p53→ tumor suppressor protein.
Triterpenes such as betulinic acid activate apoptosis by acting on mitochondrial membrane mediated through the intrinsic pathway which generates reactive oxygen species (ROS) and activate proapoptic-p38 MAPK and SAP/JNK kinases that are involved in several regulatory processes like cell proliferation, gene expression, differentiation, mitosis, cell survival, and apoptosis [17, 30, 42]. Previous in vivo and in vitro studies has been reported that in melanoma cell lines betulinic acid aggravate apoptosis and produces selective cytotoxicity to neuroectodermal tumor, including human neuroblastoma, melanoma and Ewing sarcomas [35]. In tumor cell lines like leukemia HL-60 and HeLa cells oleanolic acid encourage apoptosis [14, 48]. 18β-Glycyrrhetinic acid has the ability to induce cancer in a dose dependent manner. In rat liver mitochondria, at 10 μM concentrations it changes pro-apoptic pathways such as membrane potential loss, cytochrome c enzyme release and Ca2+ dependent apoptosis inducing mediators [42].
Caspase plays regulatory role in cell protein turnover by controlling several mechanisms like delicate protein surgery, the cleaving of selected set of the target protein and different activating and deactivating mechanisms of cell. Also they have crucial role in the process of apoptosis and about 9 different types of caspases are involved in apoptic pathway, such as death receptor pathway and mitochondrial pathway (Fig. 4) [48]. In apoptosis, TNFR acts as a death receptor which is activated by ligand mediated external stimuli for example, stimulation of TNF causes adapter protein recruitment that in turn activate caspases 8, an initiator caspase. Furthermore, activation of mitochondrial pathway can promote DNA damaging that cannot be revamped and survival factors like Bcl-2 protein involved in antiapoptic pathways are removed [4, 30, 43, 48]. Celastrol methyl ester (pristimerin), a triterpenoid obtained from Celastrus and Maytenus spp. demonstrated potent inhibitory effect on NF-kB and proteasome activity, and also triggers apoptosis in myeloma cells [30].
Triterpenoids such as betulinic acid, pomolic acid and oleanolic acid have an inhibitory effect on multidrug resistant (MDR) and sensitive leukemia cell lines [30, 49]. Also, ursolic acid found to exhibit cytotoxicity against multidrug resistant human hepatoma (R-HepG2) cells [42].
5.2. Anti-angiogenesis
Angiogenesis comprise new blood vessels formation from existing blood vessel. In the body, it requires for growth and development of new cells and eventually for wound healing [5, 43]. Angiogenesis plays major role in the vasculature, tumor generation and its proliferation. Pro-angiogenic factor like vascular endothelial growth factor (VEGF) and anti-angiogenic factor such as angiostatin control formation of new blood vessels. Imbalances between these two factors produce tumor angiogenesis which significantly contributes to malignant tissue growth [5, 50]. New capillary growth inhibition and destruction of existing tumor blood vessels consequently leads to tumor shrinkage or demise and thus, tumor growth can be restrained. Therefore, suppression of angiogenesis is a prominent approach in cancer treatment [50].
Triterpenoids selectively regulates angiogenic factors such as bFGF and VEGF, and thereby inhibiting formation of new blood vessels, cell migration and cell proliferation [43]. Betulinic acid was reported for anti-angiogenic activity mediated through damaging of mitochondrial functioning [17, 28]. Ursolic acid and oleanolic acid also exhibited similar activity on bovine aortic endothelial cells in the chick embryo chorioallantoic membrane assay [28].
5.3. Differentiation
Differentiation is defined as the extent of functional and morphological resemblance of parenchymal tumor cell to the corresponding normal cell [51]. Differentiation takes place in G0 stage of cell cycle, a resting phase where cell enters into cell cycle for proliferation process. Inhibition of this differentiation of cells, which is to be proliferated is one of the possible approaches for cancer treatment [52].
Betulinic acid induces the differentiation in normal keratinocytes by upregulating filaggrin and involucrin. Oleanolic acid also has been found to augment expression of these two markers in mice. Oleanolic acid and ursolic acid produce a cell cycle arrest when tested in human colon, esophageal and non-small cell lung cancer (NSCLC) cell lines. Boswellic acid arrests cell cycle at G1 phase by inducing p21 expression and down regulating cyclin D1 and cyclin E expressions (cell cycle regulatory proteins) [43]. Tumor suppressor protein like p53 controls the cell cycle, which is selectively elevated by pomolic acid. Pomolic acid also enhances the level of apoptosis related genes p21 (cyclin-dependent kinase inhibitor 1) and reduce the survival gene and vascular growth factors [2, 49, 53].
Many triterpenoids (lupeol, betulinic acid, oleanolic acid and ursolic acid) exerts their actions by interacting multiple targets [28]. Topoisomerases are the enzymes that involved in regulation of topology state of DNA and play a crucial role in various aspects of DNA metabolism like replication, transcription, recombination and chromosomal segregation during mitosis. Furthermore, these enzymes break and reunite one (Topoisomerase I) or both (Topoisomerase II) the strands of DNA. During cell proliferation the level of topoisomerase increases. Tumor formation can be reduced by selectively inhibiting activity of topoisomerase enzyme [18]. Betulinic, boswellic, ursolic, and oleanolic acids competitively inhibit topoisomerase I and II for topoisomerase binding sites on DNA, which in turn prevent topoisomerase-DNA cleavable complex formation and breaking of strands and avert DNA replication [54, 55].
5.4. Antioxidative effect
Reactive oxygen species (ROS) are required to perform numerous cellular functions and have both beneficial and deleterious effects. Moderate concentration of ROS is produced in the body and its production is tightly regulated by the different enzymes. Their beneficial physiological effects mainly include immune responses, whereas overproduction of ROS results in oxidative stress that causes breakdown of cell organelles and DNA, depletion of antioxidant enzymes (glutathione), decrease in host antioxidative system and results in tumor initiation and promotion [2, 56]. Certain carcinogens also cause damage to the cell specially DNA damage or induce inflammation. Chemoprotective agents protect normal cells from these attacks [57].
Triterpenoids like lupeol, oleanolic acid, betulin and ursolic acid have strong antioxidant activity [28]. Lupeol selectively increases the glutathione pool, antioxidising enzymes and antioxidants and thereby acts as a potent antioxidant. It selectively binds and modulate protein kinase that principally involved in signal transduction pathway and exerts cytotoxicity towards cancerous cell [58]. Oleanolic acid has been shown to have ROS inhibitory and ability to enhance the production of antioxidant enzymes such as catalase and thioredoxin peroxidase mediated through Nrf2 [14]. ROS generated due to exposure of ultraviolet B (UVB) radiation causes genotoxicity. Ursolic acid restrains these UVB-induced oxidative changes such as lipid peroxidation, oxidative stress and DNA damage [59]. It has been observed that ursolic acid and oleanolic acid exhibit chemopreventive effect against crypt foci induced by the carcinogen 1, 2-dimethylhydrazine (DMH). Development of crypt foci generally occurs in early stages of colorectal carcinogenesis and is considered to be an important step in tumor formation which is selectively inhibited by ursolic acid and oleanolic acid [60].
Certain triterpenoids act on multiple targets, for example ursolic acid has been found to act on target cell cycle proteins, growth factors, kinase, chemokines, cytokines, adhesion molecules, inflammatory enzymes and multiple pro-inflammatory factors. The modulation of these targets such as initiation, promotion and metastasis of cancer might be responsible for chemopreventive and therapeutic effects of ursolic acid [61].
5.5. Other targets
Several inflammatory signaling cascades such as nuclear factor-κB (NF-κB), signal transducer and activator of transcription (STAT) and cyclooxygenase-2 (COX-2) are reported to link with different stages of cancer progression and regulate tumor proliferation, survival invasion, metastasis, invasion and angiogenesis [43]. Cellular events are tightly regulated by transcription factor and nuclear factor-κB and activation of these are often associated with tumorigenesis. Ursolic acid has been found to inhibit carcinogens (tumor necrosis factor, hydrogen peroxide and cigarette smoke) that provoke NF-κB activation [61, 62]. Mutated ras oncogene is known for 90% metastasis of prostate cancer. Lupeol selectively inhibit ras protein and subsequent down regulation of MAPK (Mitogen activated protein kinase) signaling pathway and NF-κB pathway [42, 63]. AMP-activated protein kinase (AMPK) is present in all eukaryotes and senses cellular energy status. Activation of AMPK suppresses tumor cell proliferation and provokes apoptosis in the cancerous cell. Pomolic acid has an inhibitory effect on growing cancer cells, but it has been shown that in breast cancer cell line, pomolic acid restrain cell proliferation and induces apoptosis mediated through AMPK activation [49].
Telomerase is imperative noncoding sequences that prevent aberrant recombination and degradation, thus these are responsible for genome stability and cell viability. Likewise, last few nucleotides present at the end of DNA cannot be copied easily by DNA polymerase. Thus, during each round of cell division a portion of telomeres gets eroded and eventually it becomes non-functional. These telomeres are an attractive therapeutic target for cancer treatment [4, 50, 64]. Table 2 describes targets and mechanisms contributing anticancer activity of some pentacyclic triterpenoids.
Table 2.
Pentacyclic triterpenoids with anticancer activity and their mechanisms.
| Sr. No. | Pentacyclic triterpenoids | Targets and general mechanisms | References |
|---|---|---|---|
| 1. | α-Amyrin | Anti-tumor. Inhibit NF-κB, IL-1β, COX-2, CREB, ERK, PKC, P38 MAPK. |
[16], [27] |
| 2. | β-Amyrin | Anti-tumor. Inhibit NF-κB, IL-1β, COX-2, CREB, ERK, PKC, P38 MAPK. |
[16], [27] |
| 3. | Asiatic acid | Anti-cancer. Apoptosis inducer via caspase-3 activation and ROS, Growth inhibition by ↑apoptosis,S-G2/M, ↑p21/WAF1, ↓cyclin B1, ↓cyclin A, ↓Cdc2, ↓Cdc25C, ↓Bcl-2, ↑Bax, ↑ERK ½, ↑p38 |
[42, 59] |
| 4. | Betulin | Apoptosis, Anti-angiogenic, Antioxidant, Cell differentiation enhancer. Inhibit ROS generation, Fas upregulation, Caspase-8-dependent BID activation with successive inhibition of mitochondrial pathway. |
[37], [42] |
| 5. | Betulinic acid | Apoptosis inducer mediated by the activation of the mitochondrial pathway. Cytotoxic, ↑Apoptosis, Caspase activation, Inhibit cell proliferation through ↓Bcl-2, ↓cyclin D1, ↑Bax. |
[16], [35], [36], [65] |
| 6. | Boswellic acid | Apoptosis inducer. Arrest cell cycle in G1 phase through activation of caspases-8,9 and 3. Inhibit NF-kB signaling, Bind and inhibit IKKa and IKKb kinases and modulate downstream NF-κB signaling. |
[42, 66] |
| 7. | Friedelin | Suppress cell proliferation and topoisomerase | [18, 65] |
| 8. | Glycyrrhetinic acid | Induce apoptosis. Produce pro-apoptic changes of the rat liver mitochondria such as loss of membrane potential and release of cytochrome c and other apoptosis inducing factors in a Ca2+ dependent manner. Induce apoptosis through caspase-8 activation, Bcl-2 and Bcl-xL (antiapoptic) down regulation. |
[16, 27] |
| 9. | Lantadene A and B | Anti-cancer. Induces efficiently cell apoptosis by activating the caspase-3 pathway and through regulation of Bcl-2 and Bax expression. Enhances apoptosis through inhibition of tumors via down regulation of AP-1 and NF-κB (p65). |
[68 – 72] |
| 10. | Lupeol | Induces apoptosis. Suppress cell proliferation by ↑ERα protein and mRNA. Inhibits NF-kB signaling including phosphorylation of IkBa protein, DNA binding of NF-kB complex and NF-κB-dependent reporter gene activity. |
[62, 63, 73] |
| 11. | Maslinic acid | Reduces cell proliferation rates. Induce apoptosis through increasing activity of caspase 3. Triggers release of cytochrome c from mitochondria and subsequently activates the downstream apoptosis executors. Induce apoptosis through intrinsic pathway. |
[13, 74] |
| 12. | Oleanolic acid | Cytotoxicity, Inhibit angiogenesis, Inhibit DNA polymerase β. | [65] |
| 13. | Pomolic acid | Cytotoxicity, Inhibit DNA polymerase β, Activate AMPK. | [30, 49] |
| 14. | Pristimerin | Induce cell apoptosis, Inhibit cell viability. Inhibit tumor angiogenesis and tumor cell proliferation, Induce caspase dependent apoptosis. Inhibit NF-kB and proteasome activity. Inhibits NF-κB and cell cycling through VEGFR2. |
[30, 38, 75] |
| 15. | Taraxerol | Inhibitory action on cancer cell. | [76] |
| 16. | Ursolic acid | Anti-mutagenic, Anti-tumor, Inhibition of cell proliferation, Displayed cytotoxicity cancer cells by ↑apoptosis, ↓Bcl-2, PARP cleavage, GR modulation. Growth inhibitory effect by decreasing O2 consumption. Cytotoxicity, Increases NO. |
[33, 65] |
| 17. | 24-Hydroxyursolic acid | Inhibit cell proliferation. Induce cellular apoptosis by activation of poly(ADP-ribose) polymerase (PARP), caspase-3, and phosphorylation of p53 at Ser15. Activate AMP-activated protein kinase (AMPK) and mediate critical anti-cancer effects by inhibition of cyclooxygenase (COX-2) expression in HT-29 cells. Strongly induce DNA fragmentation cancer cells. |
[77] |
| 18. | 2α-Hydroxyursolic acid (corosolic acid, colosolic acid) | Suppress cell proliferation, ↓NF-κ B, ↓ proteosomal activity | [65, 78] |
6. Conclusions
A vast literature is available showing that plants are richly distributed with a diverse range of chemical compounds and can influence many physiological processes in normal and disease state. This review summarizes crucial mechanisms of pentacyclic triterpenoids accountable for anticancer effects. Literature obtained from in vitro and in vivo studies revealed that pentacyclic triterpenoid can indeed act on multiple molecular targets and cell proliferation regulatory pathways that play a pivotal role in uncontrolled cell proliferation. It mainly includes DNA polymerase inhibition, regulation of apoptosis, change in signal transductions, interfere with angiogenesis and dedifferentiation, antiproliferative activity and metastasis inhibition. Ursolic acid exerts anticancer efficacy by several mechanisms such as proliferation inhibition, enhancing apoptosis, growth receptor modulation, reducing cellular oxygen consumption and increasing cellular level of NO. Pristimerin selectively inhibits NF-kB and proteasome activity, and cell cycling through VEGFR2. Many targets and related interactions by which pentacyclic triterpenoid acts as anticancer agent has been revealed, but still a detailed investigation and clinical trials are required to clarify correct molecular mechanisms and its anticancer efficacy.
Acknowledgment
Thanks to all of the researchers and authors whose studies were used in this article.
Abbreviations
- DNA
Deoxyribonucleic acid
- BRCA1
Breast cancer gene 1
- BRCA2
Breast cancer gene 2
- WHO
World Health Organization
- Nrf2
Nuclear factor erythroid 2 [NF-E2]-related factor 2
- NF-κB
Nuclear factor kappa-light-chain-enhancer of activated B cells
- MDR
Multidrug resistance
- AP-1
Activator protein 1
- ROS
Reactive oxygen species
- MAPK
Mitogen-activated protein kinase
- SAPK
Stress-activated protein kinases
- JNK
Jun amino-terminal kinases
- HL-60
Human promyelocytic leukemia cells
- HeLa
Immortal cell line
- Bcl-2
B-cell lymphoma 2
- R-HepG2
Resistant human hepatoma
- bFGF or FGF
fibroblast growth factor
- VEGF
Vascular endothelial growth factor
- NSCLC
Non-small cell lung cancer
- ROS
Reactive oxygen species
- STAT
Signal transducer and activator of transcription
- AMPK
AMP-activated protein kinase
- R
Receptor
- Apaf-1
Apoptic activating protease factor-1
- IL-1β
Interleukin-1β
- CREB
Cyclic AMP-responsive element-binding protein 1
- ERK
Extracellular signal–regulated kinases
- PKC
Protein kinaseC
- BID
BH3interacting-domain
- S-G2/M
Cell cycle checkpoints
- ↑p21/WAF1
Cyclin-dependent kinase inhibitor 1 or CDK-interacting protein 1
- Cdc2
Cell division cycle protein 2
- Cdc25C
Cell Division Cycle 25 Homolog C
- Bax
BCL 2 associated X protein
- ERK
Extracellular signal–regulated kinases
- IKK
I kappa B kinase
- VEGFR
Vascular endothelial growth factor receptor
- GR
Growth receptor
- NO
Nitric oxide
Footnotes
This paper meets the requirements of KS X ISO 9706, ISO 9706-1994 and ANSI/NISO Z39.48-1992 (Permanence of Paper).
Conflict of interest
There is no conflict of interest and disclosures associated with the manuscript.
References
- 1.Loney T, Aw TC, Handysides DG, Ali R, Blair I, Grivna M, Shah SM, et al. An analysis of the health status of the United Arab Emirates: the ‘Big 4’public health issues. Global health action. 2013;6:1–8. doi: 10.3402/gha.v6i0.20100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dhanamani M, Devi SL, Kannan S. Ethnomedicinal plants for cancer therapy - a review. Hygeia JD Med. 2011;3:1–10. [Google Scholar]
- 3.Singh MP, Kumar V, Agarwal A, Kumar R, Bhatt ML, Misra S. Clinico-epidemiological study of oral squamous cell carcinoma: A tertiary care centre study in North India. J Oral Biol Craniofac Res. 2016;6:32–35. doi: 10.1016/j.jobcr.2015.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rang HP, Ritter JM, Flower RJ. Rang and Dale’s Pharmacology: With student consult online access: Elsevier Health Sciences. 7th edition. Churchill Livingstone; Edinburgh: 2012. [Google Scholar]
- 5.Wang FZ, Yang NN, Zhao YL, Liu QQ, Fei HR, Zhang JG. PF-04691502 triggers cell cycle arrest, apoptosis and inhibits the angiogenesis in hepatocellular carcinoma cells. Toxicol Lett. 2013;220:150–156. doi: 10.1016/j.toxlet.2013.04.018. [DOI] [PubMed] [Google Scholar]
- 6.Niedzwiecki A, Roomi MW, Kalinovsky T, Rath M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients. 2016;8:552. doi: 10.3390/nu8090552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Safarzadeh E, Shotorbani SS, Baradaran B. Herbal medicine as inducers of apoptosis in cancer treatment. Adv Pharm Bull. 2014;4:421–427. doi: 10.5681/apb.2014.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fabio GD, Romanucci V, De Marco A, Zarrelli A. Triterpenoids from Gymnema sylvestre and their pharmacological activities. Molecules. 2014;19:10956–10981. doi: 10.3390/molecules190810956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pratheeshkumar P, Sreekala C, Zhang Z, Budhraja A, Ding S, Son YO, Wang X, Hitron A, Hyun-Jung K, et al. Cancer prevention with promising natural products: mechanisms of action and molecular targets. Anticancer Agents Med Chem. 2012;12:1159–1184. doi: 10.2174/187152012803833035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shu L, Cheung KL, Khor TO, Chen C, Kong AN. Phytochemicals: cancer chemoprevention and suppression of tumor onset and metastasis. Cancer Metastasis Rev. 2010;29:483–502. doi: 10.1007/s10555-010-9239-y. [DOI] [PubMed] [Google Scholar]
- 11.Muffler K, Leipold D, Scheller MC, Haas C, Steingroewer J, Bley T, et al. Biotransformation of triterpenes. Process Biochemistry. 2011;46:1–15. doi: 10.1016/j.procbio.2010.07.015. [DOI] [Google Scholar]
- 12.Martinez A, Rivas F, Perojil A, Parra A, Garcia-Granados A, Fernandez-Vivas A. Biotransformation of oleanolic and maslinic acids by Rhizomucor miehei. Phytochemistry. 2013;94:229–237. doi: 10.1016/j.phytochem.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 13.Parmar S, Sharma T, Airao V, Bhatt R, Aghara R, Chavda S, et al. Neuropharmacological effects of triterpenoids. Phytopharmacology. 2013;4:354–372. [Google Scholar]
- 14.Pollier J, Goossens A. Oleanolic acid. Phytochemistry. 2012;77:10–15. doi: 10.1016/j.phytochem.2011.12.022. [DOI] [PubMed] [Google Scholar]
- 15.Maia JL, Lima-Júnior RC, Melo CM, David JP, David JM, Campos AR, Santos FA, Rao VS. Oleanolic acid, a pentacyclic triterpene attenuates capsaicin-induced nociception in mice: possible mechanisms. Pharmacol Res. 2006;54:282–286. doi: 10.1016/j.phrs.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 16.Moreira DLF, de-Souza GHB, Rodrigues IV, Lopes NP, de Oliveira AR. A non-michaelian behavior of the in vitro metabolism of the pentacyclic triterpene alfa and beta amyrins by employing rat liver microsomes. J Pharm Biomed Anal. 2013;84:14–19. doi: 10.1016/j.jpba.2013.05.038. [DOI] [PubMed] [Google Scholar]
- 17.Csuk R, Siewert B, Dressel C, Schafer R. Tormentic acid derivatives: synthesis and apoptic activity. Eur J Med Chem. 2012;56:237–245. doi: 10.1016/j.ejmech.2012.08.032. [DOI] [PubMed] [Google Scholar]
- 18.Mandal A, Ghosh S, Bothra AK, Nanda AK, Ghosh P. Synthesis of friedelan triterpenoid analogs with DNA topoisomerase IIα inhibitory activity and their molecular docking studies. Eur J Med Chem. 2012;54:137–143. doi: 10.1016/j.ejmech.2012.04.037. [DOI] [PubMed] [Google Scholar]
- 19.American Cancer Society. Cancer Facts & Figures 2016. Atlanta: American Cancer Society; 2016. [Google Scholar]
- 20.International Agency for Research on Cancer and Cancer Research UK. World Cancer Factsheet. Cancer Research UK; London: 2012. [Google Scholar]
- 21.American Cancer Society. Cancer Facts & Figures 2013. Atlanta: American Cancer Society; 2013. [Google Scholar]
- 22.Ali I, Wani WA, Saleem K. Cancer scenario in India with future perspectives. Cancer Ther. 2011;8:56–70. [Google Scholar]
- 23.Pittayapan P. Health Insurance for Cancer Care in Asia: Thailand. Asia Pac J Oncol Nurs. 2016;3:54. doi: 10.4103/2347-5625.178173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sreedevi A, Javed R, Dinesh A. Epidemiology of cervical cancer with special focus on India. Int J Womens Health. 2015;7:405–414. doi: 10.2147/IJWH.S50001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dsouza ND, Murthy N, Aras R. Projection of cancer incident cases for India-till 2026. Asian Pac J Cancer Prev. 2013;14:4379–4386. doi: 10.7314/APJCP.2013.14.7.4379. [DOI] [PubMed] [Google Scholar]
- 26.Dikshit R, Gupta PC, Ramasundarahettige C, Gajalakshmi V, Aleksandrowicz L, Badwe R, et al. Cancer mortality in India: a nationally representative survey. The Lancet. 2012;379:1807–1816. doi: 10.1016/S0140-6736(12)60358-4. [DOI] [PubMed] [Google Scholar]
- 27.James JT, Dubery IA. Pentacyclic triterpenoids from the medicinal herb, Centella asiatica (L.) Urban. Molecules. 2009;14:3922–3941. doi: 10.3390/molecules14103922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Laszczyk MN. Pentacyclic triterpenes of the lupane, oleanane and ursane group as tools in cancer therapy. Planta med. 2009;75:1549–1560. doi: 10.1055/s-0029-1186102. [DOI] [PubMed] [Google Scholar]
- 29.Reyes CP, Nunez MJ, Jimenez IA, Busserolles J, Alcaraz MJ, Bazzocchi IL. Activity of lupane triterpenoids from Maytenus species as inhibitors of nitric oxide and prostaglandin E 2. Bioorg Med Chem. 2006;14:1573–1579. doi: 10.1016/j.bmc.2005.10.063. [DOI] [PubMed] [Google Scholar]
- 30.Yan XJ, Gong LH, Zheng FY, Cheng KJ, Chen ZS, Shi Z. Triterpenoids as reversal agents for anticancer drug resistance treatment. Drug Discov Today. 2014;19:482–488. doi: 10.1016/j.drudis.2013.07.018. [DOI] [PubMed] [Google Scholar]
- 31.Yan J, Sun L, Zhang X, Li Z, Zhou L, Qiu M. Serratene Triterpenoids from Palhinhaea cernua var. sikkimensis. Chem Pharm Bull (Tokyo) 2009;57:1381–1384. doi: 10.1248/cpb.57.1381. [DOI] [PubMed] [Google Scholar]
- 32.Wittayalai S, Sathalalai S, Thorroad S, Worawittayanon P, Ruchirawat S, Thasana N. Lycophlegmariols A–D: Cytotoxic serratene triterpenoids from the club moss Lycopodium phlegmaria L. Phytochemistry. 2012;76:117–123. doi: 10.1016/j.phytochem.2012.01.006. [DOI] [PubMed] [Google Scholar]
- 33.Feng JH, Chen W, Zhao Y, Ju XL. Anti-tumor activity of oleanolic, ursolic and glycyrrhetinic acid. The Open Natural Products Journal. 2009;2:48–52. doi: 10.2174/1874848100902010048. [DOI] [Google Scholar]
- 34.Soldi C, Pizzolatti MG, Luiz AP, Marcon R, Meotti FC, Mioto LA, et al. Synthetic derivatives of the α-and β-amyrin triterpenes and their antinociceptive properties. Bioorg Med Chem. 2008;16:3377–3386. doi: 10.1016/j.bmc.2007.12.008. [DOI] [PubMed] [Google Scholar]
- 35.Kvasnica M, Sarek J, Klinotova E, Dzubak P, Hajduch M. Synthesis of phthalates of betulinic acid and betulin with cytotoxic activity. Bioorg Med Chem. 2005;13:3447–3454. doi: 10.1016/j.bmc.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 36.Gauthier C, Legault J, Lebrun M, Dufour P, Pichette A. Glycosidation of lupane-type triterpenoids as potent in vitro cytotoxic agents. Bioorg Med Chem. 2006;14:6713–6725. doi: 10.1016/j.bmc.2006.05.075. [DOI] [PubMed] [Google Scholar]
- 37.Patocka J. Biologically active pentacyclic triterpenes and their current medicine signification. J Appl Biomed. 2003;1:7–12. doi: 10.32725/jab.2003.002. [DOI] [Google Scholar]
- 38.Bishayee A, Ahmed S, Brankov N, Perloff M. Triterpenoids as potential agents for the chemoprevention and therapy of breast cancer. Front Biosci. 2011;16:980. doi: 10.2741/3730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li ZJ, Yao C, Liu SF, Chen L, Xi YM, Zhang W, et al. Cytotoxic effect of icaritin and its mechanisms in inducing apoptosis in human burkitt lymphoma cell line. Biomed Res Int. 2014:1–7. doi: 10.1155/2014/391512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Andreeff M, Goodrich DW, Pardee AB. Cell proliferation, differentiation, and apoptosis. In: Kufe DW, Pollock RE, Weichselbaum RR, editors. Holland-Frei Cancer Medicine. 6th edit. Hamilton (ON): BC Decker; 2003. [Google Scholar]
- 41.Sharma P, Allison JP. Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell. 2015;161:205–214. doi: 10.1016/j.cell.2015.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang SR, Fang WS. Pentacyclic triterpenoids and their saponins with apoptosis-inducing activity. Curr Top Med Chem. 2009;9:1581–1596. doi: 10.2174/156802609789909821. [DOI] [PubMed] [Google Scholar]
- 43.Janakiram NB, Indranie C, Malisetty SV, Jagan P, Steele VE, Rao CV. Chemoprevention of colon carcinogenesis by oleanolic acid and its analog in male F344 rats and modulation of COX-2 and apoptosis in human colon HT-29 cancer cells. Pharm Res. 2008;25:2151–2157. doi: 10.1007/s11095-008-9582-7. [DOI] [PubMed] [Google Scholar]
- 44.Kanda T, Yokosuka O. The androgen receptor as an emerging target in hepatocellular carcinoma. J Hepatocell Carcinoma. 2015;2:91–99. doi: 10.2147/JHC.S48956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fritz WA, Lin TM, Peterson RE. The aryl hydrocarbon receptor (AhR) inhibits vanadate-induced vascular endothelial growth factor (VEGF) production in TRAMP prostates. Carcinogenesis. 2008;29:1077–1082. doi: 10.1093/carcin/bgn069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Senese S, Lo Y, Huang D, Zangle TA, Gholkar AA, Robert L, et al. Chemical dissection of the cell cycle: probes for cell biology and anti-cancer drug development. Cell Death Dis. 2014;5:e1462. doi: 10.1038/cddis.2014.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lu JJ, Dang YY, Huang M, Xu WS, Chen XP, Wang YT. Anti-cancer properties of terpenoids isolated from Rhizoma Curcumae–A review. J Ethnopharmacol. 2012;143:406–411. doi: 10.1016/j.jep.2012.07.009. [DOI] [PubMed] [Google Scholar]
- 48.Kim SS, Won SJ, Kim NJ, Cho SD, Choi HS. 3-Oxoolean-12-en-27-oic acid isolated from Aceriphyllum rossii induces caspase-8-dependent apoptosis in human promyelocytic leukemia HL-60 cells. Biol Pharm Bull. 2009;32:91–98. doi: 10.1248/bpb.32.91. [DOI] [PubMed] [Google Scholar]
- 49.Youn SH, Lee JS, Lee MS, Cha EY, Thuong PT, Kim JR, et al. Anticancer properties of pomolic acid-induced AMP-activated protein kinase activation in MCF7 human breast cancer cells. Biol Pharm Bull. 2012;35:105–110. doi: 10.1248/bpb.35.105. [DOI] [PubMed] [Google Scholar]
- 50.Sun CC, Zhang YS, Xue X, Cheng YN, Liu HP, Zhao CR, et al. Inhibition of angiogenesis involves in anticancer activity of riccardin D, a macrocyclic bisbibenzyl, in human lung carcinoma. Eur J Pharmacol. 2011;667:136–143. doi: 10.1016/j.ejphar.2011.06.013. [DOI] [PubMed] [Google Scholar]
- 51.Harsh M. Textbook of pathology. Jaypee Brothers Medical Publishers; New Delhi: 2010. [Google Scholar]
- 52.Hu Y, Fu L. Targeting cancer stem cells: a new therapy to cure cancer patients. Am J Cancer Res. 2012;2:340–356. [PMC free article] [PubMed] [Google Scholar]
- 53.Ebner F, Schremmer-Danninger E, Rehbock J. The role of TP53 and p21 gene polymorphisms in breast cancer biology in a well specified and characterized German cohort. J Cancer Res Clin Oncol. 2010;136:1369–1375. doi: 10.1007/s00432-010-0788-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mutai C, Abatis D, Vagias C, Moreau D, Roussakis C, Roussis V. Cytotoxic lupane-type triterpenoids from Acacia mellifera. Phytochemistry. 2004;65:1159–1164. doi: 10.1016/j.phytochem.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 55.Ketron AC, Osheroff N. Phytochemicals as anticancer and chemopreventive topoisomerase II poisons. Phytochem Rev. 2013;13:19–35. doi: 10.1007/s11101-013-9291-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sun H, Fang WS, Wang WZ, Hu C. Structure-activity relationships of oleanane-and ursane-type triterpenoids. Botanical Studies. 2006;47:339–368. [Google Scholar]
- 57.Chen Y, Williams V, Filippova M, Filippov V, Duerksen-Hughes P. Viral carcinogenesis: factors inducing DNA damage and virus integration. Cancers. 2014;6:2155–2186. doi: 10.3390/cancers6042155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Nath R, Roy S, De B, Choudhury MD. Anticancer and antioxidant activity of croton: a review. Int J Pharm Pharm Sci. 2013;5:63–70. [Google Scholar]
- 59.Ramachandran S, Prasad NR. Effect of ursolic acid, a triterpenoid antioxidant, on ultraviolet-B radiation-induced cytotoxicity, lipid peroxidation and DNA damage in human lymphocytes. Chem Biol Interact. 2008;176:99–107. doi: 10.1016/j.cbi.2008.08.010. [DOI] [PubMed] [Google Scholar]
- 60.Furtado RA, Rodrigues EP, Araujo FR, Oliveira WL, Furtado MA, Castro MB, et al. Ursolic acid and oleanolic acid suppress preneoplastic lesions induced by 1, 2-dimethylhydrazine in rat colon. Toxicol Pathol. 2008;36:576–580. doi: 10.1177/0192623308317423. [DOI] [PubMed] [Google Scholar]
- 61.Shanmugam MK, Dai X, Kumar AP, Tan BK, Sethi G, Bishayee A. Ursolic acid in cancer prevention and treatment: molecular targets, pharmacokinetics and clinical studies. Biochem Pharmacol. 2013;85:1579–1587. doi: 10.1016/j.bcp.2013.03.006. [DOI] [PubMed] [Google Scholar]
- 62.Lopez-Hortas L, Perez-Larran P, Gonzalez-Munoz MJ, Falque E, Dominguez H. Recent developments on the extraction and application of ursolic acid. A review. Food Research International. 2018;103:130–149. doi: 10.1016/j.foodres.2017.10.028. [DOI] [PubMed] [Google Scholar]
- 63.Kangsamaksin T, Chaithongyot S, Wootthichairangsan C, Hanchaina R, Tangshewinsirikul C, Svasti J. Lupeol and stigmasterol suppress tumor angiogenesis and inhibit cholangiocarcinoma growth in mice via downregulation of tumor necrosis factor-α. Plos One. 2017;12:1–16. doi: 10.1371/journal.pone.0189628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Turan I, Demir S, Kilinc K, Yaman SO, Misir S, Kara H, et al. Cytotoxic effect of Rosa canina extract on human colon cancer cells through repression of telomerase expression. Journal of Pharmaceutical Analysis. 2018;8(6):394–399. doi: 10.1016/j.jpha.2017.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Thoppil RJ, Bishayee A. Terpenoids as potential chemopreventive and therapeutic agents in liver cancer. World J Hepatol. 2011;3:228–249. doi: 10.4254/wjh.v3.i9.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Salminen A, Lehtonen M, Suuronen T, Kaarniranta K, Huuskonen J. Terpenoids: natural inhibitors of NF-κB signaling with anti-inflammatory and anticancer potential. Cell Mol Life Sci. 2008;65:2979–2999. doi: 10.1007/s00018-008-8103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Satomi Y, Nishino H, Shibata S. Glycyrrhetinic acid and related compounds induce G1 arrest and apoptosis in human hepatocellular carcinoma HepG2. Anticancer Res. 2005;25:4043–4047. [PubMed] [Google Scholar]
- 68.Grace-Lynn C, Darah I, Chen Y, Latha LY, Jothy SL, Sasidharan S. In vitro antioxidant activity potential of lantadene A, a pentacyclic triterpenoid of Lantana plants. Molecules. 2012;17:11185–11198. doi: 10.3390/molecules170911185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Patel S. A weed with multiple utility: Lantana camara. Reviews in Environmental Science and Bio/Technology. 2011;10:341–351. doi: 10.1007/s11157-011-9254-7. [DOI] [Google Scholar]
- 70.Kaur J, Sharma M, Sharma P, Bansal MP. Antitumor activity of lantadenes in DMBA/TPA induced skin Tumors in mice: expression of transcription factors. Am J Biomed Sci. 2010;2:79–90. doi: 10.5099/aj100100079. [DOI] [Google Scholar]
- 71.Sharma M, Sharma P, Bansal M, Singh J. Lantadene A-induced apoptosis in human leukemia HL-60 cells. Indian J Pharmacol. 2007;39:140. doi: 10.4103/0253-7613.33433. [DOI] [Google Scholar]
- 72.Wynendaele W, Oosterom VA, Pawinski A, De Bruijn EA, Maesi RA. Angiogenesis: possibilities for therapeutic interventions. Pharm World Sci. 1998;20:225–235. doi: 10.1023/A:1008600603059. [DOI] [PubMed] [Google Scholar]
- 73.Prasad S, Kalra N, Shukla Y. Induction of apoptosis by lupeol and mango extract in mouse prostate and LNCaP cells. Nutr Cancer. 2008;60:120–130. doi: 10.1080/01635580701613772. [DOI] [PubMed] [Google Scholar]
- 74.Juan ME, Planas JM, Ruiz-Gutierrez V, Daniel H, Wenzel U. Antiproliferative and apoptosis-inducing effects of maslinic and oleanolic acids, two pentacyclic triterpenes from olives, on HT-29 colon cancer cells. Br J Nutr. 2008;100:36–43. doi: 10.1017/S0007114508882979. [DOI] [PubMed] [Google Scholar]
- 75.Mu X, Shi W, Sun L, Li H, Li H, Jiang Z, et al. Pristimerin, a triterpenoid, inhibits tumor angiogenesis by targeting VEGFR2 activation. Molecules. 2012;17:6854–6868. doi: 10.3390/molecules17066854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Swain SS, Rout KK, Chand PK. Production of triterpenoid anti-cancer compound taraxerol in Agrobacterium-transformed root cultures of butterfly pea (Clitoria ternatea L.) Appl Biochem Biotechnol. 2012;168:487–503. doi: 10.1007/s12010-012-9791-8. [DOI] [PubMed] [Google Scholar]
- 77.Khanal P, Oh WK, Thuong PT, Cho SD, Choi HS. 24-Hydroxyursolic acid from the leaves of the Diospyros kaki (Persimmon) induces apoptosis by activation of AMP-activated protein kinase. Planta Med. 2010;76:689–693. doi: 10.1055/s-0029-1240678. [DOI] [PubMed] [Google Scholar]
- 78.Neto CC. Berries and Cancer Prevention. Springer; New York: 2011. Ursolic acid and other pentacyclic triterpenoids: anticancer activities and occurrence in berries. [DOI] [Google Scholar]