Abstract
Post-amputation phantom limb pain (PLP) is highly prevalent and very difficult to treat. The high-prevalence, high-pain intensity levels, and decreased quality of life associated with PLP compel us to explore novel avenues to prevent, manage, and reverse this chronic pain condition. This narrative review focuses on recent advances in the treatment of PLP and reviews evidence of mechanism-based treatments from randomized controlled trials published over the past 5 years. We review recent evidence for the efficacy of targeted muscle reinnervation, repetitive transcranial magnetic stimulation, imaginal phantom limb exercises, mirror therapy, virtual and augmented reality, and eye movement desensitization and reprocessing therapy. The results indicate that not one of the above treatments is consistently better than a control condition. The challenge remains that there is little level 1 evidence of efficacy for PLP treatments and most treatment trials are underpowered (small sample sizes). The lack of efficacy likely speaks to the multiple mechanisms that contribute to PLP both between and within individuals who have sustained an amputation. Research approaches are called for to classify patients according to shared factors and evaluate treatment efficacy within classes. Subgroup analyses examining sex effects are recommended given the clear differences between males and females in pain mechanisms and outcomes. Use of novel data analytical approaches such as growth mixture modeling for multivariate latent classes may help to identify sub-clusters of patients with common outcome trajectories over time.
Keywords: phantom limb pain, cortical reorganization, referred pain, neuropathic pain, central sensitization, assessment, treatment
Effective treatment of phantom limb pain (PLP) is a central issue that continues to confront amputees and their clinicians. The majority of amputees report PLP at some point after limb amputation, and lifetime prevalence estimates are between 50 and 80% 1– 3. The pain is typically neuropathic in origin and referred to the missing limb with qualities of sensation such as throbbing, “pins and needles”, shooting, stabbing, and burning. PLP is usually reported within the first week after amputation and generally decreases in severity and frequency over time in most individuals 4. It is accompanied by a variety of secondary effects, including depression, impairments in everyday activities, and decreased quality of life 5. Over the past 50 years, researchers have explored how PLP can be treated via medication, surgery, therapy, and numerous other approaches. More than 25 treatments for PLP are currently available yet not one is widely accepted or clearly superior to others 6, 7. This likely speaks to the multiple mechanisms that contribute to PLP. Treatments typically target a single proposed mechanism, yet amputees can have PLP that arises from multiple mechanisms. Consequently, not one intervention has been found to be consistently effective.
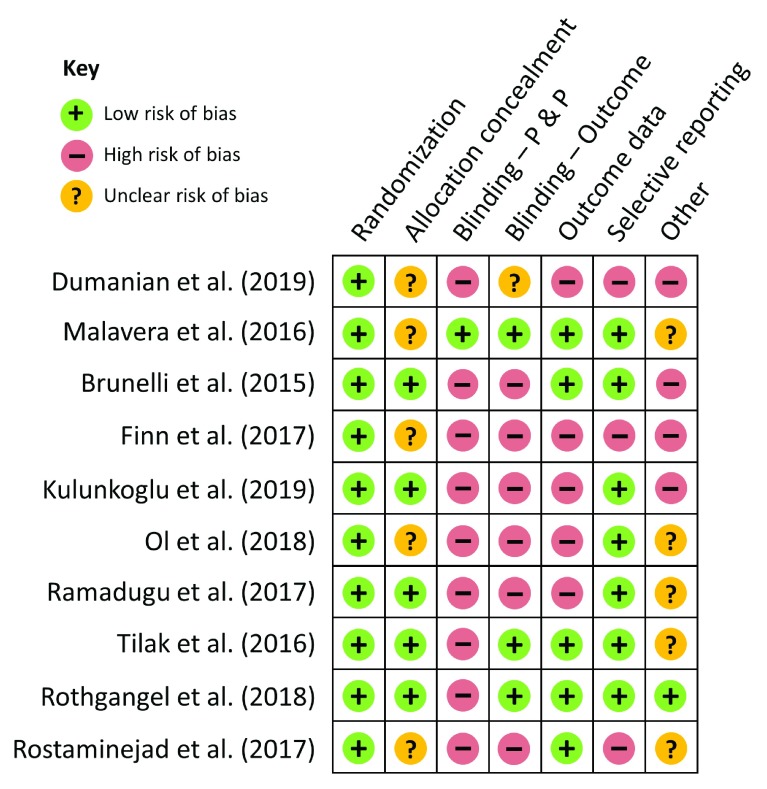
This article presents a narrative review of randomized controlled trials (RCTs) evaluating the efficacy of PLP interventions published over the past 5 years. In the following sections, we review evidence for the efficacy of targeted muscle reinnervation (TMR), repetitive transcranial magnetic stimulation (rTMS), imaginal phantom limb exercises, mirror therapy (MT), virtual and augmented reality, and eye movement desensitization and reprocessing (EMDR) therapy. The Cochrane Collaboration’s tool 8 is used to assess risk of bias for the RCTs included in this review. Each included RCT was evaluated according to seven criteria assessing selection, performance, detection, attrition, reporting, and other biases. Table 1 lists the main features of the included articles along with their associated global risk-of-bias rating. The Supplementary Tables 1 to 10 present the detailed risk-of-bias assessments for each included trial for interested readers. Figure 1 summarizes the risk-of-bias assessments across the seven criteria for each included RCT.
Table 1. Summary of the literature reviewed in this article investigating the treatment of phantom limb pain.
| Authors, year,
and sample size |
Site (and
percentage) of amputation |
Reason(s) for
amputation and percentages |
Mean time
(range) since amputation |
Treatment
groups |
Treatment
duration |
Main outcome
measure(s) |
Assessments | Findings | Risk of bias |
|---|---|---|---|---|---|---|---|---|---|
| Dumanian
et al. 9 (2019) (n = 28) |
UE = 13%
LE = 87% |
Trauma = 90%
Infection = 10% |
(Less than 1
year to more than 10 years) |
1. Targeted
muscle reinnervation 2. Standard neuroma surgery |
N/A | Change in NRS worst
pain from baseline to 12 months post-operatively for PLP and residual limb pain |
Baseline
3 months 6 months 9 months 12 months |
No significant between-
group differences in worst PLP or residual limb pain 1 year post- surgery |
High risk |
| Malavera
et al. 16 (2016) (n = 54) |
LE = 100% | Trauma = 100% | 7.8 years | 1. Active rTMS
2. Sham rTMS |
20 stimuli of
6 seconds each (54- second intervals), 5 days per week for 2 weeks |
PLP intensity measured
via a visual analogue scale (VAS) |
Baseline
15 days post- rTMS 30 days post- rTMS |
No significant between-
group differences in PLP scores at either follow-up time |
Low risk |
| Brunelli
et al. 19 (2015) (n = 40) |
AK = 73%
BK = 27% |
Dysvascular = 70%
Other = 30% |
458
days |
1. SAIPAN
protocol 2. Standard treatment |
~1 hour two
times per week for 4 weeks |
1. PLP intensity, rate,
duration, and bother measured via Prosthesis Evaluation Questionnaire (PEQ) and Brief Pain Inventory (BPI) 2. Phantom limb symptom (PLS) intensity, rate, and bother measured via PEQ |
Baseline
1 month 2 months |
Significant group
differences in PLP rate, duration, and bother from PEQ at 2-month follow-up only. No significant differences with BPI. Significant group differences in PLS rate, intensity, and bother at 2-month follow-up only. |
High risk |
| Finn
et al. 20 (2017) (n = 15) |
AE = 40%
BE = 60% |
Trauma = 100% | 4.5 months
(0.55–24 months) |
1. MT
2. Control (covered mirror) 3. Control (mental visualization) |
15 minutes
5 days per week for 4 weeks |
PLP intensity measured
via VAS |
Baseline
4 weeks |
No between-group
comparisons reported for main outcome measure |
High risk |
| Anaforoğlu
Külünkoğlu et al. 21 (2019) (n = 40) |
BK = 100% | Trauma = 100% | 13.25
months (3–53 months) |
1. MT
2. Phantom exercise (PE) |
15 minutes
of MT daily at home for 4 weeks PE group performed exercises daily with 15 repetitions |
1. PLP intensity measured
via VAS 2. Quality of life evaluated via SF-36 3. Psychological status measured using BDI |
Baseline
4 weeks 3 months 6 months |
Significant group
difference in VAS PLP severity, BDI scores, and PF, SF, MH, and V subscales of the SF-36 in favour of the MT group at all follow-up times |
High risk |
| Ol
et al.
22
(2018) (n = 45) |
BK = 100% | Trauma = 100% | (15–32
years) |
1. MT
2. Tactile 3. Combination (mirror and tactile therapy) |
5 minutes
every morning and night for 4 weeks |
PLP intensity measured
via VAS |
Baseline
5 weeks 3 months after end of treatment |
No significant between-
group differences in PLP scores at either follow-up time |
High risk |
| Ramadugu
et al. 23 (2017) (n = 60) |
AE = 8%
BE = 8% AK = 34% BK = 50% |
NR | NR | 1. MT
2. Control (covered mirror) |
15 minutes
every day for 4 weeks |
PLP intensity measured
via VAS and the short form of the McGill Pain Questionnaire |
Baseline
4 weeks 8 weeks 12 weeks 16 weeks (20 weeks, control only) |
No between-group
comparisons reported for main outcome measures |
High risk |
| Tilak
et al.
24
(2016) (n = 26) |
UE = 26%
LE = 74% |
NR | 45 days | 1. MT
2. Contralateral transcutaneous electrical nerve stimulation |
20 minutes
every day for 4 days |
PLP intensity measured
via VAS and Universal Pain Score (UPS) |
Baseline
4 days |
No significant between-
group differences in PLP intensity at the end of treatment |
High risk |
| Rothgangel
et al. 25 (2018) (n = 75) |
AK = 61%
K = 7% BK = 32% |
Trauma = 32%
Dysvascular = 40% Tumor = 14% Other = 14% |
~35 months | 1. MT followed
by teletreatment using augmented reality 2. MT followed by self-delivered MT 3. Sensomotor exercises to the intact limb followed by self-delivered exercises |
At least 10
30-minute sessions across 4 weeks followed by 6 weeks of self- delivered treatment |
PLP frequency, duration,
and intensity (measured via NRS) |
Baseline
4 weeks 10 weeks 6 months |
No significant between-
group differences at 4- or 10-week follow-up. At 6-month follow-up, there were significant between- group differences on PLP duration only, in favor of MT followed by self-delivered MT. |
High risk |
| Rostaminejad
et al. 26 (2017) (n = 60) |
AK = 40%
BK = 60% |
Diabetes = 45%
Trauma = 50% Cancer = 5% |
(2–38 months) | 1. EMDR
2. Routine care |
12 one-hour
sessions over 1 month |
PLP intensity measured
via the subjective units of distress scale and pain rating scale |
Baseline
1 month 24 months |
No between-group
comparisons reported for main outcome measures |
High risk |
Results from interventions using targeted muscle reinnervation, repetitive transcranial magnetic stimulation (rTMS), mirror therapy (MT), augmented reality, and eye movement desensitization and repossessing (EMDR) therapy are summarized above. For each study, the site, reason, and mean time since amputation are shown. Treatment groups and duration as well as main outcome measures, times of assessment, and major findings are included. The final column describes the article’s global risk-of-bias rating as assessed by the Cochrane Collaboration’s tool 8. UE and LE refer to upper and lower extremity amputations, respectively. AK, BK, AE, and BE refer to amputations performed above (A) and below (B) the knee (K) and elbow (E) joint. NR denotes information that was not reported by the author. NRS refers to the numeric 0–10 rating scale. BDI, Beck Depression Inventory; MH, mental health; N/A, not applicable; PF, physical functioning; PLP, phantom limb pain; SAIPAN, Santa Lucia Alleviation Intervention for Phantom in Amputees’ Neurorehabilitation; SF, social functioning; SF-36, 36-Item Short Form Survey; V, vitality.
Figure 1. Risk-of-bias 8 assessments for studies presented in the review of recent randomized controlled trials exploring treatment of phantom limb pain.
Randomization: randomization sequence generation; Blinding – P & P: blinding of participants and personnel; Blinding – Outcome: blinding of outcome assessment; Outcome data: incomplete outcome data.
Targeted muscle reinnervation
Proposed mechanisms underlying PLP have traditionally been classified according to the level of the nervous system: peripheral or central. Peripheral mechanisms include activation of residual limb neuromas by mechanical stimulation, circulating catecholamines, pro-inflammatory immunological factors, and other pronociceptive neurochemicals. TMR is a relatively recent technique that involves surgically re-routing and coapting the distal aspect of a transected nerve to a motor nerve that innervates an adjacent muscle. Over time, the coapted residual nerve and motor nerve fasten together and the new combined nerve reinnervates the muscle 9. The mechanism by which TMR is believed to reduce PLP is not entirely clear, yet researchers have suggested restoration of physiological continuity and function 10, neuroma prevention 11, and upstream effects on cortical reorganization 9. Recent research has suggested that, though originally proposed to facilitate use and control of myoelectric prostheses, TMR may also be an effective way to reduce PLP 10– 13.
A recent RCT examined the efficacy of TMR for PLP among 28 unilateral and bilateral upper or lower limb amputees with chronic PLP (30 limbs treated) 9. Time since amputation ranged from less than 1 year to more than 10 years. TMR was compared to standard treatment involving excising the neuroma and burying the remaining nerve into neighboring muscle. Worst, best, and current levels of phantom and residual limb pain in the past 24 hours were assessed pre-operatively and post-operatively at 3-month intervals for 1 year. Secondary outcomes, including pain behaviour, pain intensity, and pain interference assessments of the Patient-Reported Outcomes Measurement Information System (PROMIS), were also completed at these assessments. One year after surgery, significant between-group differences were not found in worst PLP intensity, worst residual limb pain intensity, or the three PROMIS pain scales 9. However, an analysis using all available data (regardless of whether the last assessment was at the 1-year time point) showed that worst PLP change scores at the final assessment were significantly greater in TMR than standard treatment, indicating greater reduction in PLP for the former group 9. Higher baseline pain scores for the intervention group may explain this significant result. Taken together, the results do not support the efficacy of TMR for PLP. Future research should compare TMR with a less invasive but putatively equally efficacious treatment such as non-invasive brain stimulation 14.
Repetitive transcranial magnetic stimulation
Maladaptive cortical re-organization is one of the central neural mechanisms thought to underlie PLP. It has been proposed that, after amputation, cortical areas that once represented the amputated extremity become reoccupied by adjacent zones in the primary somatosensory and motor cortex corresponding to other body parts 6, 15. Imagined movement of the phantom extremity is accompanied by brain activity in regions corresponding to not only the lost extremity but also the adjacent body part. PLP has been shown to be more intense among individuals for whom a greater degree of maladaptive cortical remapping has taken place 6. rTMS has been explored for its potential in preventing maladaptive sensorimotor cortical remapping and in reducing PLP 16. Targeting the somatosensory and motor cortex using a magnetic pulse emitted by the rTMS coil has been proposed to activate descending inhibitory pathways to the thalamus, thereby modulating subsequent ascending nociceptive signals and reducing PLP 16.
rTMS was evaluated in an RCT of 54 unilateral trauma-related lower limb amputees 16. The mean time since amputation was 7.8 years. Participants were randomly assigned to receive active rTMS or sham rTMS for 20 minutes five times per week over 2 weeks. The control condition was exposed to a sham coil that did not emit a magnetic pulse or induce a tactile sensation on the scalp. For the active rTMS group, the authors targeted the hand area of motor cortex contralateral to the amputated leg, citing evidence of efficacy for this stimulation site in past studies regardless of anatomic location of the pain 17, 18. PLP was evaluated daily for 1 week before the start of treatment and 15 and 30 days after the last treatment. Levels of depression and anxiety were also measured at each of these time points. Between-group differences in PLP scores and levels of depression and anxiety were not significant at either follow-up time 16. However, PLP scores at both follow-up times were significantly lower than at baseline for the active group but not the sham group. Fifteen days after treatment, there was a significantly greater percentage reduction in PLP in the group that received active versus sham rTMS; however, this was no longer significant 30 days after treatment. rTMS of the motor cortex does not appear to reduce PLP in lower extremity amputees to a greater extent than a sham control condition when the hand area, rather than the foot area, is stimulated. Future studies may prove more effective by matching the rTMS stimulation site in motor cortex contralateral to the extremity amputated.
Imaginal phantom limb exercises
Cortical reorganization has also been proposed to occur as a result of mental imagery, including engaging in phantom limb exercises (that is, active imaginal efforts to move the phantom), under the assumption that the neural pathways involved in performing actual movements are activated when using one’s imagination to move the phantom extremity. Initial studies of phantom limb exercises show promise in reducing PLP 27, 28.
In a 2015 study, unilateral lower limb amputees were randomly assigned to one of two groups to receive progressive muscle relaxation, mental imagery, and phantom exercises (n = 27) or residual limb exercises (n = 24) 19. The mean reported time since amputation was 458 days, and the majority of amputations were due to diabetes and peripheral vascular disease. The treatment group received a 50-minute combined training session in progressive muscle relaxation, mental imagery, and phantom exercises twice per week for 4 weeks. Phantom exercises involved imagining moving the phantom limb and then attempting to perform these movements. The control group received the same amount of treatment involving exercising their residual limb. In addition, both groups participated in a rehabilitation program involving occupational therapy and prosthesis training, which took place twice per day for 4 weeks. Items from the Prosthesis Evaluation Questionnaire (PEQ) and Brief Pain Inventory (BPI) measuring pain and bodily sensations were collected at baseline, at the end of treatment, and 1 month after treatment. Owing to participant attrition, only 20 participants from each group were included in the final analysis. At the end of treatment, the two groups did not differ significantly on either the PEQ or BPI items 19. At the 1-month follow-up, the treatment group had significantly lower pain intensity scores on the BPI worst and average pain items and significantly lower scores on the PLP rate, intensity, and bother items of the PEQ compared with the control group 19. Although preliminary evidence seems promising, the authors did not adjust the type 1 error rate for multiple comparisons. Moreover, the absence of between-group differences in PEQ and BPI outcomes at the end of treatment is puzzling and raises the possibility that some factor unrelated to treatment accounts for the significant effects at the 1-month follow-up. Furthermore, the researchers did not measure the amputees’ self-report of their ability to perform the phantom exercises or the extent to which they engaged in these movements. This is an important factor to measure given that self-reported motor control is a predictor of PLP severity 29. More research is needed on the effects of combined progressive muscle relaxation, mental imagery, and phantom exercises using larger sample sizes and better measures.
Mirror therapy
It has been suggested that PLP may be especially difficult to treat because of the absence of tactile and visual feedback from the limb 30. The role of the visual and tactile modality is especially important since they provide important information involving exteroceptive sensibility. Lower limb amputees frequently report that it was not until they looked under the bed sheets and reached out to touch the limb that they realized it had been cut off. When there is a discrepancy or contradiction between incoming information from different modalities or when a state of uncertainty exists based upon somatosensory input alone, additional information is sought via these modalities, which usually determine the perceptual experience. Amputation not only results in the loss of afferent input/feedback from the amputated limb but also produces a loss of visual and tactile information related to the limb. The central influences that normally inhibit pain may be further reduced by the absence of information from these external sources that might otherwise confirm or disconfirm the perception of pain arising from the periphery (for example, a phantom limb in a painful position or a “crawling” sensation on the skin) 30. Thus, some forms of PLP may arise, in part, from a mechanism involving a release from inhibitory control (that is, disinhibition).
Self-touch of a painful area can gate pain signals from reaching the brain, therefore minimizing the pain experience 31. Looking at one’s own body has also been shown to reduce pain intensity and neural responses to painful stimuli compared with viewing a neutral object 32. Research on reducing PLP has focused on restoring this lack of sensory feedback. MT is a long-standing treatment for PLP 33 and is thought to reduce PLP by restoring normal somatosensory and visual inputs to associated brain structures, although the precise mechanisms by which this occurs are not well understood 20. A recent neuroimaging study of lower limb amputees with PLP found enhanced responsiveness to viewing images of feet (but not hands) in the foot area of sensorimotor cortex contralateral to the amputated limb as well as in posterior parietal cortex 34. Both PLP intensity and the increased visual responsiveness were abolished after 4 weeks of MT. These results are consistent with the “PLP as disinhibition” hypothesis 30 described above whereby seeing the limb (via MT) re-establishes, in somatosensory and parietal structures, the normal inhibitory control processes which were lost because of amputation (that is, visual deafferentation). The restoration, via the visual modality, of inhibitory control over cells in these and other brain regions reduces abnormal brain activity, which contributed to increased levels of PLP, and thereby reduces pain.
Five RCTs examining the efficacy of MT for PLP have been published over the past 5 years 20, 21– 24. Sample sizes range from a total of 15 20 to 60 23. Two studies 21, 22 recruited unilateral lower extremity amputees only (n = 85), one study 20 recruited unilateral upper limb amputees only (n = 15), and the remaining two studies 23, 24 recruited both unilateral lower (n = 60) and unilateral upper (n = 17) extremity amputees. Time since amputation ranged from less than 1 month 20 to 32 years 22; one study 23 did not report time since amputation. MT was compared with sensorimotor exercises with or without a covered mirror 20, 21, 23, tactile therapy 22, or contralateral transcutaneous electrical nerve stimulation 24, a treatment that has been shown to be effective for PLP 35– 37. Parameters of the MT intervention ranged from a low of a single 20-minute session daily for 4 consecutive days 24 to 5-minute sessions twice per day for 4 weeks 22 to a high of one 15-minute session daily for 4 weeks 20, 21, 23. Participants were instructed to move both the intact and phantom limb synchronously during MT while viewing the reflected image of the intact limb (that is, the phantom) in the mirror 20, 21– 23. One study did not specify what participants were instructed to do regarding phantom limb exercises 24. PLP was assessed at various times, including pre-treatment baseline and immediately post-treatment for all studies and up to 3 22, 4 23, or 6 21 months post-treatment. Of the five studies, only one showed significantly lower PLP intensity scores in favour of MT up to 6 months after treatment 21. The remaining studies either did not report a between-group test of PLP intensity 20, 23 or failed to show a significant benefit of MT on any measure of PLP at the end of treatment 22, 24. Taken together, the results of the most recent studies evaluating the efficacy of MT for PLP are not promising. Overall, MT does not appear to reduce PLP to a greater degree than control or other known treatments.
Virtual and augmented reality
Virtual and augmented reality interventions have recently emerged as novel approaches to treating PLP. Virtual reality involves completely immersing an individual in a virtual world, whereas augmented reality adds digital elements, such as the missing limb, to a real environment. These interventions represent a “high-tech” alternative to traditional MT 38 as they allow amputees to move their intact and phantom limbs independently while seeing their phantom limb integrated into, and interacting with, the surrounding setting 39. They also represent a more engaging form of treatment which may increase adherence 39. Although virtual and augmented reality interventions have gained popularity, quality evidence does not exist to support its efficacy 40. Existing studies are typically underpowered and lack comparison groups.
In spite of the excitement and popularity surrounding virtual and augmented reality for treatment of PLP, only one RCT using augmented reality has been published in the past 5 years 25. Seventy-five unilateral lower limb amputees with a median time since amputation of about 3.5 years were randomly assigned to one of three interventions. The first group completed 4 weeks of MT followed by 6 weeks of teletreatment involving augmented reality and digital exercise programs. The second group underwent 4 weeks of MT followed by 6 weeks of self-delivered MT (traditional MT group). The third group received 4 weeks of sensorimotor exercises to the unamputated limb and 6 weeks of self-delivered exercises (control group). Each group received at least 10 30-minute sessions of their respective intervention across the initial 4 weeks. Participants assigned to the MT intervention were instructed to perform exercises using their intact limb in front of a mirror. Only once they perceived voluntary and pain-free movements of their phantom limb were they asked to engage in phantom exercises. PLP ratings of intensity, frequency, and duration were collected at baseline and 4, 10, and 24 weeks later. At the 4- and 10-week follow-ups, the three groups did not differ significantly on any of the PLP measures. At the 6-month follow-up, the duration of PLP episodes was significantly shorter in the traditional MT group in comparison with the control and teletreatment groups. In contrast, 6-month average PLP intensity and 6-month PLP frequency did not differ between the groups; moreover, not one of the three PLP outcome measures showed a significant between-group difference immediately after treatment (4 weeks) or at the 10-week follow-up. This raises the possibility that the 6-month outcome was due to factors other than traditional MT. The results do not favour MT or augmented reality as a viable treatment for PLP. Further research is needed to determine whether virtual and augmented reality are effective in the treatment of PLP.
Eye movement desensitization and reprocessing
Researchers have also explored treatments that target psychological mechanisms maintaining PLP, such as painful and traumatic memories. One such intervention is EMDR therapy. EMDR is thought to reduce PLP by emotionally processing “painful memories” that are proposed to maintain PLP 26. EMDR therapy was compared with a control condition in an RCT of 60 unilateral lower limb amputees 26. The reason for amputation was trauma (50%), diabetes-related complications (45%), or cancer (5%). Time since amputation ranged from 2 to 38 months. The control group received routine care while the experimental group underwent 12 one-hour sessions of EMDR administered by trained psychologists over 1 month. Average PLP ratings were collected at baseline, at the end of treatment, and 24 months later. The authors did not report the results of a between-group analysis at any point in time. PLP intensity in the EMDR group was significantly lower than baseline after treatment and at the 24-month follow-up. In contrast, PLP intensity in the control group remained consistently high across the study period. Further studies are needed to determine whether EMDR effectively reduces PLP.
Summary and Conclusions
The results of this review do not support the efficacy of any of the treatments described for PLP, including TMR, rTMS, imaginal phantom limb exercises, MT, augmented reality, or EMDR therapy. The multiple mechanisms underlying PLP have made it difficult to treat, and one specific treatment that targets multiple mechanisms of PLP has yet to evolve. Moreover, the published literature does not assess the putative mechanism(s) causing PLP in individuals recruited into clinical trials and so it is not surprising that, on average, PLP in the treatment group does not differ from that in the control group. This may explain why many of the treatments available are ineffective. Many of the studies reviewed have small sample sizes with short follow-up periods ( Table 1). Little has changed in the more than 20 years since the recommendation for a rational approach to assessment and management of PLP 41, 42. The field continues to lack a mechanism-based method of classifying amputees.
Recommendations for future studies
This review of RCTs conducted over the past 5 years has not demonstrated consistent evidence for a given intervention. The data in Supplementary Tables 1 to 10 and Figure 1 show that the risk of bias is high for most of the studies included in this review, thus raising questions about the studies’ internal validity and quality. These data highlight the need to develop guidelines on how to improve future PLP treatment research. We suggest that in addition to improving the methodological quality of studies by adhering to the most recent Consolidated Standards of Reporting Trials (CONSORT) statement (http://www.consort-statement.org), the following methodological improvements are required: larger sample sizes, long-term follow-ups, and limiting inclusion criteria for any given study to minimize participant heterogeneity. We recommend limiting recruitment for RCTs to upper or lower extremity amputations, trauma-related or vascular disease-related amputations, and short or long time since amputation. Moreover, given the importance of sex differences in the field of pain 43, 44, we strongly recommend that subgroup analyses look separately at female and male amputees. Finally, the use of average pain scores as the best measure of treatment efficacy has been criticized on empirical and theoretical grounds 45. Adopting novel data analytical approaches such as growth mixture modeling for multivariate latent classes may help to identify sub-clusters of patients with common outcome trajectories over time.
Acknowledgements
AA is supported by a scholarship from the Fonds de la Recherche pour la Santé du Québec. JK is supported by a Canadian Institutes of Health Research Canada Research Chair in Health Psychology.
Editorial Note on the Review Process
F1000 Faculty Reviews are commissioned from members of the prestigious F1000 Faculty and are edited as a service to readers. In order to make these reviews as comprehensive and accessible as possible, the referees provide input before publication and only the final, revised version is published. The referees who approved the final version are listed with their names and affiliations but without their reports on earlier versions (any comments will already have been addressed in the published version).
The referees who approved this article are:
Jack W Tsao, Department of Neurology, University of Tennessee Health Science Center, Memphis, USA
Tamar R Makin, Institute of Cognitive Neuroscience, University College London, London, UK
Funding Statement
This work was supported by funding from JK’s Canada Research Chair in Health Psychology.
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
[version 1; peer review: 2 approved]
Supplementary material
Supplementary Tables 1–10References
- 1. Ephraim PL, Wegener ST, MacKenzie EJ, et al. : Phantom pain, residual limb pain, and back pain in amputees: results of a national survey. Arch Phys Med Rehabil. 2005;86(10):1910–9. 10.1016/j.apmr.2005.03.031 [DOI] [PubMed] [Google Scholar]
- 2. Richardson C, Glenn S, Nurmikko T, et al. : Incidence of phantom phenomena including phantom limb pain 6 months after major lower limb amputation in patients with peripheral vascular disease. Clin J Pain. 2006;22(4):353–8. 10.1097/01.ajp.0000177793.01415.bd [DOI] [PubMed] [Google Scholar]
- 3. Kooijman CM, Dijkstra PU, Geertzen JH, et al. : Phantom pain and phantom sensations in upper limb amputees: an epidemiological study. Pain. 2000;87(1):33–41. 10.1016/S0304-3959(00)00264-5 [DOI] [PubMed] [Google Scholar]
- 4. Bosmans JC, Geertzen JH, Post WJ, et al. : Factors associated with phantom limb pain: a 31/2-year prospective study. Clin Rehabil. 2010;24(5):444–53. 10.1177/0269215509360645 [DOI] [PubMed] [Google Scholar]
- 5. Horne CE, Paul J: Pain Support for Adults with a Diabetes-Related Lower Limb Amputation: an Empirical Phenomenology Study. Pain Manag Nurs. 2019;20(3):270–5. 10.1016/j.pmn.2018.09.007 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 6. Flor H: Phantom-limb pain: characteristics, causes, and treatment. Lancet Neurol. 2002;1(3):182–9. 10.1016/S1474-4422(02)00074-1 [DOI] [PubMed] [Google Scholar]
- 7. Subedi B, Grossberg GT: Phantom limb pain: mechanisms and treatment approaches. Pain Res Treat. 2011;2011: 864605. 10.1155/2011/864605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Higgins JP, Altman DG, Gøtzsche PC, et al. : The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. 10.1136/bmj.d5928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Dumanian GA, Potter BK, Mioton LM, et al. : Targeted Muscle Reinnervation Treats Neuroma and Phantom Pain in Major Limb Amputees: A Randomized Clinical Trial. Ann Surg. 2019;270(2):238–246. 10.1097/SLA.0000000000003088 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 10. Bowen JB, Wee CE, Kalik J, et al. : Targeted Muscle Reinnervation to Improve Pain, Prosthetic Tolerance, and Bioprosthetic Outcomes in the Amputee. Adv Wound Care (New Rochelle). 2017;6(8):261–7. 10.1089/wound.2016.0717 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 11. Souza JM, Cheesborough JE, Ko JH, et al. : Targeted muscle reinnervation: a novel approach to postamputation neuroma pain. Clin Orthop Relat Res. 2014;472(10):2984–90. 10.1007/s11999-014-3528-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Valerio IL, Dumanian GA, Jordan SW, et al. : Preemptive Treatment of Phantom and Residual Limb Pain with Targeted Muscle Reinnervation at the Time of Major Limb Amputation. J Am Coll Surg. 2019;228(3):217–26. 10.1016/j.jamcollsurg.2018.12.015 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 13. Bowen JB, Ruter D, Wee C, et al. : Targeted Muscle Reinnervation Technique in Below-Knee Amputation. Plast Reconstr Surg. 2019;143(1):309–12. 10.1097/PRS.0000000000005133 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 14. Kikkert S, Mezue M, O'Shea J, et al. : Neural basis of induced phantom limb pain relief. Ann Neurol. 2019;85(1):59–73. 10.1002/ana.25371 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 15. Katz J: Psychophysiological contributions to phantom limbs. Can J Psychiatry. 1992;37(5):282–98. 10.1177/070674379203700502 [DOI] [PubMed] [Google Scholar]
- 16. Malavera A, Silva FA, Fregni F, et al. : Repetitive Transcranial Magnetic Stimulation for Phantom Limb Pain in Land Mine Victims: A Double-Blinded, Randomized, Sham-Controlled Trial. J Pain. 2016;17(8):911–8. 10.1016/j.jpain.2016.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 17. Lefaucheur JP: Pain. In Handbook of Clinical Neurology M.H. A. M. Lozano, Editor.2013;116:423–440. 10.1016/B978-0-444-53497-2.00035-8 [DOI] [PubMed] [Google Scholar]
- 18. Lefaucheur JP, Drouot X, Ménard-Lefaucheur I, et al. : Motor cortex rTMS restores defective intracortical inhibition in chronic neuropathic pain. Neurology. 2006;67(9):1568–74. 10.1212/01.wnl.0000242731.10074.3c [DOI] [PubMed] [Google Scholar]
- 19. Brunelli S, Morone G, Iosa M, et al. : Efficacy of progressive muscle relaxation, mental imagery, and phantom exercise training on phantom limb: a randomized controlled trial. Arch Phys Med Rehabil. 2015;96(2):181–7. 10.1016/j.apmr.2014.09.035 [DOI] [PubMed] [Google Scholar]
- 20. Finn SB, Perry BN, Clasing JE, et al. : A Randomized, Controlled Trial of Mirror Therapy for Upper Extremity Phantom Limb Pain in Male Amputees. Front Neurol. 2017;8:267. 10.3389/fneur.2017.00267 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 21. Anaforoğlu Külünkoğlu B, Erbahçeci F, Alkan A: A comparison of the effects of mirror therapy and phantom exercises on phantom limb pain. Turk J Med Sci. 2019;49(1):101–9. 10.3906/sag-1712-166 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 22. Ol HS, Van Heng Y, Danielsson L, et al. : Mirror therapy for phantom limb and stump pain: a randomized controlled clinical trial in landmine amputees in Cambodia. Scand J Pain. 2018;18(4):603–10. 10.1515/sjpain-2018-0042 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 23. Ramadugu S, Nagabushnam SC, Katuwal N, et al. : Intervention for phantom limb pain: A randomized single crossover study of mirror therapy. Indian J Psychiatry. 2017;59(4):457–64. [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 24. Tilak M, Isaac SA, Fletcher J, et al. : Mirror Therapy and Transcutaneous Electrical Nerve Stimulation for Management of Phantom Limb Pain in Amputees - A Single Blinded Randomized Controlled Trial. Physiother Res Int. 2016;21(2):109–15. 10.1002/pri.1626 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 25. Rothgangel A, Braun S, Winkens B, et al. : Traditional and augmented reality mirror therapy for patients with chronic phantom limb pain (PACT study): Results of a three-group, multicentre single-blind randomized controlled trial. Clin Rehabil. 2018;32(12):1591–608. 10.1177/0269215518785948 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 26. Rostaminejad A, Behnammoghadam M, Rostaminejad M, et al. : Efficacy of eye movement desensitization and reprocessing on the phantom limb pain of patients with amputations within a 24-month follow-up. Int J Rehabil Res. 2017;40(3):209–14. 10.1097/MRR.0000000000000227 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 27. Moseley GL: Graded motor imagery for pathologic pain: a randomized controlled trial. Neurology. 2006;67(12):2129–34. 10.1212/01.wnl.0000249112.56935.32 [DOI] [PubMed] [Google Scholar]
- 28. Beaumont G, Mercier C, Michon PE, et al. : Decreasing phantom limb pain through observation of action and imagery: a case series. Pain Med. 2011;12(2):289–99. 10.1111/j.1526-4637.2010.01048.x [DOI] [PubMed] [Google Scholar]
- 29. Kikkert S, Mezue M, Henderson Slater D, et al. : Motor correlates of phantom limb pain. Cortex. 2017;95:29–36. 10.1016/j.cortex.2017.07.015 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 30. Katz J: The reality of phantom limbs. Motiv Emot. 1993;17(3):147–79. 10.1007/BF00992218 [DOI] [Google Scholar]
- 31. Kammers MP, de Vignemont F, Haggard P: Cooling the thermal grill illusion through self-touch. Curr Biol. 2010;20(20):1819–22. 10.1016/j.cub.2010.08.038 [DOI] [PubMed] [Google Scholar]
- 32. Longo MR, Betti V, Aglioti SM, et al. : Visually Induced Analgesia: Seeing the Body Reduces Pain. J Neurosci. 2009;29(39):12125–30. 10.1523/JNEUROSCI.3072-09.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 33. Ramachandran VS, Rogers-Ramachandran D, Cobb S: Touching the phantom limb. Nature. 1995;377(6549):489–90. 10.1038/377489a0 [DOI] [PubMed] [Google Scholar]
- 34. Chan AW, Bilger E, Griffin S, et al. : Visual responsiveness in sensorimotor cortex is increased following amputation and reduced after mirror therapy. Neuroimage Clin. 2019;23:101882. 10.1016/j.nicl.2019.101882 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
- 35. Carabelli RA, Kellerman WC: Phantom limb pain: Relief by application of TENS to contralateral extremity. Arch Phys Med Rehabil. 1985;66(7):466–7. [PubMed] [Google Scholar]
- 36. Giuffrida O, Simpson L, Halligan PW: Contralateral Stimulation, Using TENS, of Phantom Limb Pain: Two Confirmatory Cases. Pain Med. 2010;11(1):133–41. 10.1111/j.1526-4637.2009.00705.x [DOI] [PubMed] [Google Scholar]
- 37. Katz J, France C, Melzack R: An association between phantom limb sensations and stump skin conductance during transcutaneous electrical nerve stimulation (TENS) applied to the contralateral leg: A case study. Pain. 1989;36(3):367–77. 10.1016/0304-3959(89)90098-5 [DOI] [PubMed] [Google Scholar]
- 38. Giummarra M: Augmented reality for treatment of phantom limb pain-are we there yet? Lancet. 2016;388(10062):2844–5. 10.1016/S0140-6736(16)32416-3 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 39. Ortiz-Catalan M, Guðmundsdóttir RA, Kristoffersen MB, et al. : Phantom motor execution facilitated by machine learning and augmented reality as treatment for phantom limb pain: A single group, clinical trial in patients with chronic intractable phantom limb pain. Lancet. 2016;388(10062):2885–94. 10.1016/S0140-6736(16)31598-7 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 40. Dunn J, Yeo E, Moghaddampour P, et al. : Virtual and augmented reality in the treatment of phantom limb pain: A literature review. NeuroRehabilitation. 2017;40(4):595–601. 10.3233/NRE-171447 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 41. Sherman RA: Phantom limb pain. Mechanism-based management. Clin Podiatr Med Surg. 1994;11(1):85–106. [PubMed] [Google Scholar]
- 42. Sherman RA, Jones DC, Marbach JJ: Mechanism-based assessment and management. Phantom pain. 1997;149–166. 10.1007/978-1-4757-6169-6_9 [DOI] [Google Scholar]
- 43. Rosen S, Ham B, Mogil JS: Sex differences in neuroimmunity and pain. J Neurosci Res. 2017;95(1–2):500–8. 10.1002/jnr.23831 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 44. Dance A: Why the sexes don’t feel pain the same way. Nature. 2019;567(7749):448–50. 10.1038/d41586-019-00895-3 [DOI] [PubMed] [Google Scholar]; F1000 Recommendation
- 45. Pagé MG, Romero Escobar EM, Ware MA, et al. : Predicting treatment outcomes of pain patients attending tertiary multidisciplinary pain treatment centers: A pain trajectory approach. Can J Pain. 2017;1(1):61–74. 10.1080/24740527.2017.1325715 [DOI] [PMC free article] [PubMed] [Google Scholar]; F1000 Recommendation
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.