Abstract
Background:
Resting heart rate is an independent risk factor for cardiovascular disease and is mainly controlled by β‐blockers (BBs). BBs are part of the optimal medical treatment for coronary artery disease (CAD), and their benefit correlates with resting heart rate (RHR) reduction.
Hypothesis:
RHR is poorly controlled in daily practice among patients with stable cardiovascular disease, and control is only achieved by some BBs.
Methods:
Observational, cross‐sectional, and multicenter study of CAD patients recruited nationwide from 20 institutions. Antecedents, risk factors, and treatments were collected. Controlled RHR was considered at <70 bpm.
Results:
The mean age of the 2897 patients included was 67.4 years (11.4%), and 75.9% were males. Patients treated with a BB (56.5%) had a lower mean age and comorbidities. The mean RHR was 69.6 bpm (12.6). A significantly lower RHR was observed in patients treated with a BB compared to the rest (67.2 vs 73.0 bpm; P<0.01), and no difference was observed in patients treated with a calciumchannel blocker (CCB). The analysis by individual agents identified that only patients treated with atenolol, bisoprolol, and metoprolol had significantly lower RHR than those not receiving a BB. No differences were observed in mean doses of each agent according to RHR control, except for verapamil. BB treatment was independently associated with RHR control (odds ratio [OR]: 2.42, 95% CI: 2.05–2.87; P<0.01), and no association was found for nondihydropyridine CCBs (OR: 0.99, 95% CI: 0.96–1.02; P = 0.38). Bisoprolol (OR: 1.56, 95% CI: 1.38–1.78; P<0.01), atenolol (OR: 2.01, 95% CI: 1.57–3.49; P<0.01), and metoprolol (OR: 1.29, 95% CI: 1.04–1618; P = 0.04) were independently associated with RHR control.
Conclusions:
RHR is poorly controlled in CAD patients, and although BBs are the most efficient therapy, in daily clinical practice RHR <70 bpm is only independently associated with atenolol, bisoprolol, or metoprolol. © 2011 Wiley Periodicals, Inc.
The TRECE registry has an unrestricted grant from Servier Laboratories, Spain. The authors have no other funding, financial relationships, or conflicts of interest to disclose.
Introduction
β‐Blocker(BB) agents are among the most effective treatments for coronary artery disease (CAD).1, 2, 3 Clinical benefit and prognosis improvement obtained by BB treatment in CAD patients correlate closely with the grade of resting heart rate (RHR) reduction.4, 5 RHR is influenced by many clinical situations,6, 7 but even taking those into consideration, RHR independently predicts the incidence of coronary events,8, 9, 10 heart failure,10, 11 and mortality.4, 8, 9, 11, 12, 13 BBs are the most effective treatment for long‐term RHR reduction,4, 5, 14, 15 and CAD patients who have RHR >70 bpm, despite BB treatment, exhibit a higher risk of coronary events, heart failure, and mortality.10 Currently available BB agents have quite different metabolic and action profiles,16, 17, 18, 19 their use in the setting of chronic CAD is weakly supported by clinical trial evidence,20, 21, 22 and doses are usually lower than recommended.23
The TRECE registry (TRatamiento de la EnfermedadCoronaria en España [Treatment of coronary artery disease in Spain]) was designed to describe the current treatments of CAD patients to identify the most frequent limitations or failures in optimal treatment achievement.24, 25 The present subanalysis aimed to describe RHR control in CAD patients under 2 major hypotheses: first, that RHR is poorly controlled in daily practice, and second, a differential effect of BB in RHR could exist.
Methods
The TRECE registry is a descriptive, cross‐sectional, and multicenter study that included practitioners nationwide. The methodology and general characteristics of the sample have been previously described.24, 25 Throughout the first 3 months of 2006, each investigator included 15 consecutive patients with CAD, therefore a total of 3000 patients constituted the initial database. After exclusion of patients with missing data, a final population of 2897 patients was analyzed.
Inclusion and Exclusion Criteria
Inclusion criteria were confirmed diagnosis of chronic stable angina, chest pain and positive treadmill test, previous diagnosis of acute coronary syndrome, myocardial infarction, or unstable angina. Patients could have multiple inclusion criteria. Exclusion criteria were rejection of the informed consent or missing data regarding the diagnosis of CAD, cardiovascular risk factors, or clinical features.
Variables Description
RHR was measured after 10 minutes of complete repose and was obtained by the electrocardiogram or by physical examination. According to previous reports, RHR <70 bpm was considered the threshold for controlled RHR.10, 26 Current guidelines recommend that RHR in patients with permanent atrial fibrillation should be between 60 to 80 bpm,27 therefore the threshold of <70 bmp was also applied in these patients. Biochemical determinations had to be obtained within 3 months of the inclusion visit and were not processed in a central laboratory. Overweight was considered if body mass index (BMI) was 25 to 29.9 kg/m2 and obesity if BMI >30.0 kg/m2. Waist circumference was measured at the upper third of the line between the last rib and the iliac crest. Abdominal obesity was considered for waist circumference >102 cm in males or >88 cm in females.
The diagnosis of atrial fibrillation was only accepted when an electrocardiogram was recorded in the inclusion visit or in previous patient reports. Chronic obstructive pulmonary disease (COPD) was recorded when it was reported in a previous diagnosis of the patients or when specific active treatment was present. The antecedent of peripheral artery disease was assessed by the presence of walking claudication or peripheral revascularization (either surgical or percutaneous). The assessment of noncardiac comorbidities was made with the Charlson index adapted for patients with CAD.28 This index includes active smoking (1 point), hypertension (1 point), stroke (2 points), diabetes (2 points), COPD (2 points), peripheral artery disease (2 points), erectile dysfunction (2 points), and serum creatinine >3 mg/dL (7 points).
All cardiovascular active treatments and the corresponding doses were registered. All β‐blocker agents were available at the time of the inclusion in Spain. Verapamil and diltiazem are the only nondihydropyridinecalcium channel blocker (CCB) currently used.
Statistical Analysis
Data management was processed using SPSS 15.0 software (SPSS, Inc., Chicago, IL). All variables had normal distribution and are presented as mean (standard deviation). One‐way analysis of variance and the χ 2 test were used to analyze statistical differences among patients; the Tukey posthoc test was performed to describe differences amonggroups. To assess the effect of possible confounding factors the presence of interactions was tested. Multivariate analysis to identify variables associated with RHR control was performed by logistic regressions, and RHR <70 bpm was introduced as the dependent variable. Results are presented as odds ratio (OR) (95% confidence interval [CI]). Calibration of the multivariate model was tested by the Hosmer‐Lemeshow statistic and the discriminative power by the area under the receiver operating characteristic (ROC) curve obtained by the analysis of the probability of the prognosticated value of the multivariate model. Statistical difference was assumed at the value of P<0.05.
Results
The mean age of the 2897 patients included was 67.4 years (11.4), and 75.9% were males. The most frequent antecedent of CAD was myocardial infarction (42.3%), followed by unstable angina (34.5%), chronic stable angina (29.9%), and non–Q‐wave myocardial infarction (18.1%). BB treatment was present in 1636 (56.5%) patients, CCB in 319 (11%), BB +CCB in 49 (1.7%) patients, and none of them in 893 (30.8%) patients. As shown in Table 1, patients treated with a BB had a lower mean age and prevalence of atrial fibrillation, COPD, or peripheral arterial disease; globally, their comorbidities were less present, as the median Charlsonindex number was statistically lower in this group. No interactions were observed between those treated with a BB and the presence of heart failure (P = 0.39), COPD (P = 0.35) or peripheral artery disease (P = 0.16), although previous myocardial infarction (P = 0.06) and atrial fibrillation (P = 0.06) obtained nearlysignificant results. Patients under BB treatment were in the group with the highest prevalence of statins, angiotensin‐converting enzyme inhibitors, and dualantiplatelet treatments (Table 2).
Table 1.
Clinical Characteristics of Patients According to the Presence of Heart Rate‐Lowering Treatments
| Total | None | BB | CCB | BB+CCB | P | |
|---|---|---|---|---|---|---|
| No.(%) | 2,897 | 893 (30.8) | 1,636 (56.5) | 319 (11.0) | 49 (1.7) | |
| Age, y(SD) | 67.4 (11.5) | 68.4 (12.2) | 66.3 (11.2) | 70.4 (10.0) | 69.4 (10.0) | <0.01a |
| Males, % | 71.5 | 71.4 | 72.3 | 67.5 | 74.5 | 0.34 |
| Percutaneous revascularization, % | 41.2 | 33.8 | 45.7 | 37.6 | 51.0 | <0.01 |
| Surgical revascularization, % | 15.7 | 13.9 | 16.6 | 15.4 | 24.5 | 0.11 |
| Hypertension, % | 68.6 | 66.3 | 69.0 | 68.7 | 91.8 | 0.01 |
| Diabetes mellitus, % | 38.8 | 37.9 | 39.0 | 37.1 | 57.4 | 0.06 |
| Dyslipidema, % | 67.4 | 60.6 | 70.4 | 69.5 | 76.6 | <0.01 |
| Current smokers, % | 10.7 | 15.3 | 8.2 | 10.7 | 12.2 | <0.01 |
| Obesity, % | 29.0 | 26.3 | 30.0 | 30.4 | 36.7 | 0.13 |
| Heart failure | 18.6 | 19.4 | 18.6 | 14.6 | 29.2 | 0.06 |
| Atrial fibrillation, % | 10.1 | 12.0 | 8.3 | 12.1 | 22.9 | <0.01 |
| Stroke, % | 9.9 | 10.3 | 9.2 | 11.1 | 18.2 | 0.17 |
| PAD, % | 15.2 | 15.4 | 13.2 | 22.2 | 32.6 | <0.01 |
| GFR <60 mL/min, % | 27.4 | 27.1 | 26.9 | 29.9 | 33.3 | 0.57 |
| COPD, % | 15.9 | 19.2 | 9.5 | 36.2 | 37.0 | <0.01 |
| Attended by cardiologist, % | 54.6 | 50.2 | 57.8 | 53.3 | 38.8 | <0.01 |
| Charlson index | 2.0 (1.0–4.0) | 3.0 (1.0–4.0) | 2.0 (1.0–3.0) | 3.0 (1.0–5.0) | 3.5 (3.0–7.0) | <0.01a |
Abbreviations: BB, β‐blocker; CCB, calcium channel blocker; CHD, coronary heart disease; COPD, chronic obstructive pulmonary disease; GFR, glomerular filtration rate; PAD, peripheral artery disease; SD, standard deviation.
Charlson index is presented as median (interquartile range).
P values are for comparison among the four groups.
Only for the comparison between BB and the rest.
Table 2.
Medical Treatments of Patients According to the β‐Blocker or Calcium Channel Blocker Treatment
| Total, % | None, % | BB, % | CCB, % | BB+CCB, % | P | |
|---|---|---|---|---|---|---|
| BB | 58.2 | 0.0 | 100 | 0.0 | 100 | <0.01 |
| Nondihydropyridine CCB | 12.7 | 0.0 | 0.0 | 100 | 100 | <0.01 |
| Dihydropyridine CCB | 17.8 | 18.3 | 20.4 | 4.4 | 10.2 | <0.01 |
| Single antiplatelet | 62.8 | 51.6 | 66.9 | 73.2 | 64.6 | <0.01 |
| Double antiplatelet | 17.8 | 7.7 | 24.5 | 11.7 | 18.8 | <0.01 |
| ACEI | 43.3 | 39.3 | 46.8 | 37.6 | 38.8 | <0.01 |
| ARB | 18.5 | 18.0 | 18.2 | 21.0 | 22.4 | 0.56 |
| Nitrates | 40.0 | 29.7 | 42.1 | 53.9 | 67.3 | <0.01 |
| Statins | 68.3 | 45.2 | 79.8 | 73.0 | 77.6 | <0.01 |
| Fibrates | 2.4 | 2.3 | 2.8 | 0.4 | 2.6 | 0.15 |
| Ezetimibe | 3.9 | 2.1 | 4.3 | 6.4 | 2.6 | <0.01 |
Abbreviations: ACEI, angiotensin‐converting enzyme inhibitor; ARB, angiotensin‐receptor blocker; BB, β‐blocker; CCB, calcium channel blocker.
P values are for the comparison among the four groups.
Mean RHR was 69.6 bpm (12.6) and was lower in men compared to women (68.8 [12.5] vs 71.8 [12.8]; P<0.01). Figure 1 depicts mean RHR according to heart rate‐lowering treatments and shows that the lowest RHR was present in the BB‐treated patients. The most frequently used BB was atenolol (43.9%), followed by bisoprolol (30.9%), carvedilol (22.1%), metoprolol (2.3%), and propranolol or nevibolol (0.8%). RHR <70 bpm was present in only 1495 (53.2%) patients and only in 61.4% of the patients treated with BBs. The analysis of RHR according to each BB agent showed that only patients treated with atenolol, bisoprolol, or metoprolol had significantly lower RHR compared to those patients not receiving a BB, whereas no significant difference was observed in patients treated with propranolol (P = 0.68) or carvedilol (P = 0.08) (Figure 2). As shown in Table 3, patients receiving atenolol or bisoprolol had a statistically higher prevalence of RHR <70 bpm. When doses of each agent were analyzed, no difference was found amongpatients with RHR >or <70 bpm, with the exception of verapamil. It is also patent that patients not receiving a BB or nondihydropyridine CCB had a higher prevalence of RHR >70 bpm.
Figure 1.
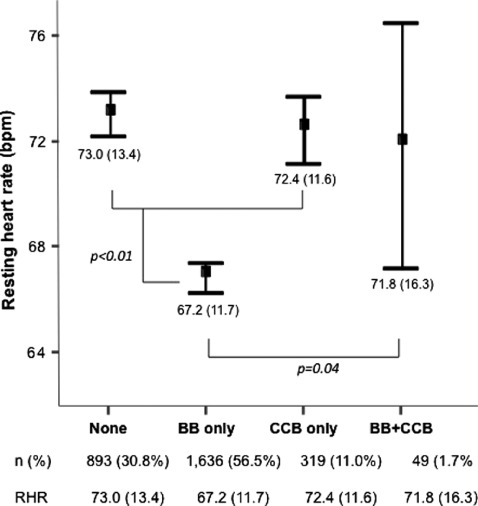
Mean resting heart rate (RHR) observed in patients treated with a β‐blocker (BB), nondihydropyridine calcium channel blocker (CCB), both, or none of them.
Figure 2.
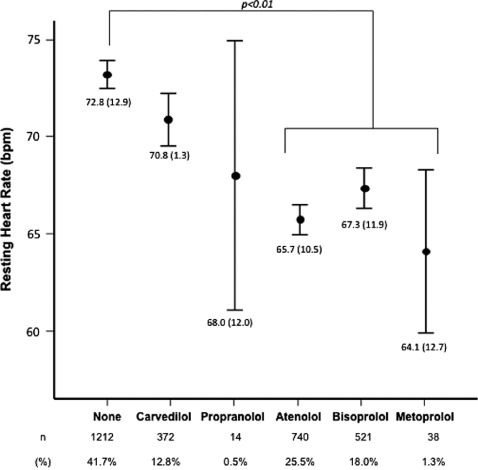
Mean resting heart rate in patients treated with each β‐blocker or none of them.
Table 3.
Mean Doses of β Blocker and Nondihydropyridine Calcium Channel Blocker Agents at the Inclusion Visit
| Agent | All | RHR<70 bpm | RHR>70 bpm | P |
|---|---|---|---|---|
| Atenolol | ||||
| Mg (SD)/d | 60.2 (26.6) | 60.5 (26.8) | 59.8 (26.1) | 0.77 |
| N (%) | 740 | 545 (73.8) | 195 (26.2) | <0.01 |
| Bisoprolol | ||||
| Mg (SD)/d | 7.6 (12.9) | 7.1 (10.0) | 8.4 (16.7) | 0.25 |
| N (%) | 521 | 361 (69.4) | 160 (30.6) | <0.01 |
| Carvedilol | ||||
| Mg (SD)/d | 21.5 (35.3) | 20.0 (17.0) | 23.0 (46.7) | 0.43 |
| N (%) | 372 | 221 (59.6) | 151 (40.4) | 0.62 |
| Metoprolol | ||||
| Mg (SD)/d | 109.0 (47.3) | 111.0 (45.7) | 105.0 (52.5) | 0.72 |
| N (%) | 38 | 27 (73.0) | 11 (27.0) | 0.13 |
| Propranolol | ||||
| Mg (SD)/d | 65.2 (44.0) | 77.5 (54.1) | 52.9 (30.4) | 0.31 |
| N (%) | 14 | 8 (57.1) | 6 (42.9) | 0.78 |
| Diltiazem | ||||
| Mg (SD)/d | 187.1 (63.2) | 183.8 (62.4) | 189.9 (64.2) | 0.40 |
| N (%) | 311 | 164 (52.7) | 147 (47.3) | <0.01 |
| Verapamil | ||||
| Mg (SD)/d | 224.4 (93.4) | 267.2 (100.8) | 202.6 (84.2) | 0.02 |
| N (%) | 56 | 22 (39.3) | 34 (60.7) | <0.01 |
| No BB or CCB | ||||
| N (%) | 893 | 435 (48.7) | 458 (51.3) | <0.01 |
Abbreviations: BB, β blocker; CCB, calcium channel blocker; RHR, resting heart rate; SD, standard deviation.
We performed a multivariate analysis, adjusted by age, gender, risk factors, previous myocardial infarction, atrial fibrillation, comorbidities, and years since coronary heart disease (CHD) onset to identify the associations of each treatment and RHR control. BB treatment was independently associated with RHR control (OR: 2.42, 95% CI: 2.05–2.87; P<0.01), and no association was found for nondihydropyridine CCBs (OR: 0.99, 95% CI: 0.96–1.02; P = 0.38). When each agent was introduced in the same model, bisoprolol (OR: 1.56, 95% CI: 1.38–1.78; P<0.01), atenolol (OR: 2.01, 95% CI: 1.57–3.49; P<0.01), and metoprolol (OR: 1.29, 95% CI: 1.04–1618; P = 0.04) were independently associated with RHR control. The multivariate analysis was accurately calibrated (P = 0.25; χ 2 = 10.2) and had discriminative power (area under the curve: 0.66, 95% CI: 0.64–0.69; P<0.01).
Discussion
The present study demonstrates that BB therapy significantly reduces RHR in CAD patients, and that atenolol, bisoprolol, and metoprolol are more effective than other BBs for RHR control in these patients. In this registry of daily clinical practice, global control of RHR was poor and was poorly controlled by CCBs. Our results highlight that the effect of a BB on RHR seems to be independent of daily doses and is related to the agent used, butthey also draw attention to the clinical issue of doseequivalence of BBs.
BB agents are undoubtedly a first‐line treatment of patients with CAD, although they only have a firm indication when ejection fraction is impaired or when the antecedent of clinically overt heart failure or myocardial infarction is present. In fact, only 56% of the patients of our study were receiving a BB, and myocardial infarction was present in more than 40% and heart failure in almost a fifth of the whole sample. These results bring to light the low use of BBs in a large sample of CHD patients and agrees with other registries,24, 25, 29, 30 although none of them analyzed RHR. We also obtained nearly‐significant interactions between BB use and the antecedent of myocardial infarction or atrial fibrillation, which could affect the association of BBs with RHR control.
The effect of BBs on RHR is usually employed to assess their effect1 and has been proposed to predict clinical prognosis.4, 5, 31, 32 In our study, patients treated with a BB had less cardiac and noncardiac comorbidities, reflecting that BBs are more frequently used in lower‐risk patients in daily practice. BB drugs are a very heterogeneous group, with highly individualized pharmacologic properties, receptor biologies, hemodynamic effects, and variable patterns of tolerability. BBs can be classified into nonselective or cardio‐selective, lipophilic or hydrophilic, short‐term or long‐term action, presence of intrinsic sympathomimetic activity, vasodilator activity through α‐1 blockers, through β‐2 agonism, or through independent mechanisms like nitric oxide agonism.14 Due to this wide variability, it is not surprising that in our study propranolol and carvedilol did not obtain as much RHR reduction as atenolol, bisoprolol, or metoprolol. Some other aspects must be taken into consideration. Carvedilol use is preferred in cases of heart failure, a condition that increases RHR itself, butrequires greater effort to achieve therapeutic doses.21 In a recent report from a registry of hypertensive patients treated with a BB, we demonstrated that the use of different BBs differs if heart failure or CAD are present.19
We did not find differences in BB doses between patients with RHR below or above 70 bpm, although the absence of dose equivalence should be addressed. Nevertheless, parameters like selectivity or vasodilator activity can be highly dosedependent, as different patients may exhibit widely variable pharmacodynamic responsiveness to the same dose of an individual drug. Moreover, there are relevant differences in pharmacokinetics and doses of currently available BB agents. Recent evidence supports that clinical benefits are more tightly associated to RHR reduction than to absolute dose.5, 33 Our results also demonstrate that currently available treatments provide a significant effect on RHR because patients whowere not receiving either BBs orCCBs had a higher prevalence of RHR >70 bpm.
Class effect has been usually admitted for BB agents,14, 22 but our results clearly demonstrate that atenolol, bisoprolol, and metoprolol are the only BB agents that significantly reduce RHR in CAD patients. The fact that the benefit of different BBs in the clinical context of heart failure or hypertension provide similar benefit34, 35 cannot be extrapolated to CAD. For example, the benefit of atenolol in postmyocardial infarction has not been proved.36 The GEMINI (The Glycemic Effects in Diabetes Mellitus: Carvedilol‐Metoprolol Comparison in Hypertensives) study was conducted to assess metabolic differences between metoprolol and carvedilol and demonstrated higher RHR reduction in metoprolol‐treated patients17; moreover, binding to a β‐adrenergic receptor is much more persistent with carvedilol than metoprolol.18 In the DELTHA registry (DisfunciónErectiL en pacientes con HTA [Erectile Dysfunction in Hypertensive patients]), nebivolol was independently associated with lower prevalence of erectile dysfunction.19 The largely debated meta‐analysis by Lindholm et al16 showed that BB treatment for hypertension was associated with lower or no cardiovascular protection compared to angiotensin‐converting enzyme, angiotensin‐receptor blockers, diuretics, or CCBs, but this effect was largely attributable to the results obtained in clinical trials with atenolol. More recently, another meta‐analysis37 addressed the negative effect of RHR reduction with BBs in patients with hypertension, especially driven by the effect of atenolol. Nevertheless, atenolol was the most commonly employed BB in our study, which might reflect the absence of concern about these data. Taking all this into considerationalong withour results concerning RHR and the dose‐independent finding, bisoprolol and metoprolol seem to be most favorable BB agents for CAD treatment.
Nondihydropyridine CCBs have more modest long‐term effect in reduction of RHR15, 38 and can also increase serum catecholamine levels when taken regularly.39 Most patients included in the present registry had chronic CAD and were under long‐term treatment, and this could explain the discouraging results of RHR control in patients treated with nondihydropyridine CCBs. Verapamil15 and diltiazem38 have demonstrated prognostic benefits in CAD patients, although they are considered a second‐line treatment after BBs.1 Moreover, short‐life CCBs have been identified as harmful in CAD patients.40 Nevertheless, a recent meta‐analysis demonstrated the beneficial effect on stroke and angina prevention, without any excess in cardiovascular events or events, of long‐acting CCB treatment.41 The growing evidence concerning RHR and our results seem to agree with that recommendation. Other therapies that reduce RHR, such as the inhibitor of sinus node I(f) current ivabradine,42 were not approved at the time of the inclusion period.
Another relevant issue is the optimal RHR for patients with permanent atrial fibrillation. A target of 60 to 80 bmp is recommended in current guidelines,27 although it seems to be highly restrictive and might be difficult to achieve in daily practice. In our study, 10% of the patients had the antecedent of atrial fibrillation, the same as the REACH (REduction of Atherothrombosis for Continued Health) registry30 and other national registries,43 which might seem low even though they represent a high‐risk subset of patients.43 Moreover, our results might emphasize a lack in knowledge of the optimal resting heart rate in these patients.
Limitations and Strengths
The main limitation of our study is the cross‐sectional design that does not allow the identification of risk factors, but shows consistent relationships between BBs and RHR. A prospective design comparing different agents might demonstrate clinical superiority of some BBs above others. To minimize the collection of a poorly representative sample, the registry was designed with nationwide coverage and recruited patients from 3 different medical specialties who were included from outpatient and hospital assistance departments. Another limitation is that the time of determination of RHR was not registered. All patients were recruited in daily medical visits, therefore most of them were assessed during morning time. Clinical characteristics of our sample are similar to published CAD registries,2, 3, 30, 43, 44 which might enhance the representativity and relevance of our results. Moreover, RHR in BB‐treated patients in our sample (67.2 bpm) was similar to RHR achieved in the placebo‐treated patients of the BEAUTIFUL (Morbidity‐Mortality Evaluation of the If Inhibitor Ivabradine in Patients With Coronary Artery Disease and Left Ventricular Dysfunction) trial10 (69.0 bpm), the first study that assessed the impact of RHR reduction in CAD patients. The low proportion of patients treated with another BB(ie, nebivolol), does not allow drawing conclusions regarding agents not well represented in the sample. Additionally, the low use of propranolol, metoprolol, or carvedilol could have underpowered our results, limiting the possibility of obtaining statistical differences. Nevertheless, clear tendencies were obtained in mean RHR that achieved statistical results compatible to current evidence. Finally, doseequivalence of different agents should also be taken under consideration because the biological and clinical effects of these agents are closely related to daily doses.
Conclusion
RHR is poorly controlled in CAD patients. Although BB‐treated patients have a lower RHR, RHR <70 bpm is only independently associated with atenolol, bisoprolol, or metoprolol. The effect of BB on RHR control was independent of mean daily dose, except for verapamil, and was related to the individual effect of each agent.
References
- 1. Fraker TD Jr, Fihn SD, Gibbons RJ, et al. Chronic Angina Focused Updated of the ACC/AHA 2002 Guidelines for the Management of Patients With Chronic Stable Angina: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines Writing Group to Develop the Focused Update of the 2002 Guidelines for the Management of Patients With Chronic Stable Angina. Circulation. 2007;116:2762–2772. [DOI] [PubMed] [Google Scholar]
- 2. Daly C, Clemens F, Lopez‐Sendon JL, et al. The impact of guideline compliant medical therapy on clinical outcome in patients with stable angina: findings from the Euro Heart Survey of stable angina. Eur Heart J. 2006;27:1298–1304. [DOI] [PubMed] [Google Scholar]
- 3. Boden WE, OŔourke RA, Teo KK, et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007;356:1503–1516. [DOI] [PubMed] [Google Scholar]
- 4. Kjekshus JK. Importance of heart rate in determining beta‐blocker efficacy in acute and long‐term acute myocardial infarction intervention trials. Am J Cardiol. 1986;57:43F–49F. [DOI] [PubMed] [Google Scholar]
- 5. Cucherat M. Quantitative relationship between resting heart rate reduction and magnitude of clinical benefits in post‐myocardial infarction: a meta‐regression of randomized clinical trials. Eur Heart J. 2007;28:3012–3019. [DOI] [PubMed] [Google Scholar]
- 6. Black A, Murray L, Cardwell C, et al. Secular trends in heart rate in young adults, 1949 to 2004: analyses of cross sectional studies. Heart. 2006;92:468–473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Martin LJ, Comuzzie AG, Sonnenberg GE, et al. Major quantitative trait locus for resting heart rate maps to a region on chromosome 4. Hypertension. 2004;43:1146–1151. [DOI] [PubMed] [Google Scholar]
- 8. Copie X, Hnatkova K, Staunton A, et al. Predictive power of increased heart rate versus depressed left ventricular ejection fraction and heart rate variability for risk stratification after myocardial infarction. Results of a two‐yearfollow‐up study. J Am Coll Cardiol. 1996;27:270–276. [DOI] [PubMed] [Google Scholar]
- 9. Dyer AR, Persky V, Stamler J, et al. Heart rate as a prognostic factor for coronary heart disease and mortality: findings in three Chicago epidemiologic studies. Am J Epidemiol. 1980;112:736–749. [DOI] [PubMed] [Google Scholar]
- 10. Fox K, Ford I, Steg PG, et al. Heart rate as a prognostic risk factor in patients with coronary artery disease and left‐ventricular systolic dysfunction (BEAUTIFUL): a subgroup analysis of a randomised controlled trial. Lancet. 2008;372:817–821. [DOI] [PubMed] [Google Scholar]
- 11. Bohm M, Swedberg K, Komajda M, et al. Heart rate as a risk factor in chronic heart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo‐controlled trial. Lancet. 2010;376:886–894. [DOI] [PubMed] [Google Scholar]
- 12. Hjalmarson A, Gilpin EA, Kjekshus J, et al. Influence of heart rate on mortality after acute myocardial infarction. Am J Cardiol. 1990;65:547–553. [DOI] [PubMed] [Google Scholar]
- 13. Jouven X, Empana JP, Escolano S, et al. Relation of heart rate at rest and long‐term (>20 years) death rate in initially healthy middle‐aged men. Am J Cardiol. 2009;103:279–283. [DOI] [PubMed] [Google Scholar]
- 14. Lopez‐Sendon J, Swedberg K, McMurray J, et al. Expert consensus document on beta‐adrenergic receptor blockers [in Spanish]. Rev Esp Cardiol. 2005;58:65–90. [DOI] [PubMed] [Google Scholar]
- 15. Pepine CJ, Handberg EM, Cooper‐Dehoff RM, et al. A calcium antagonist vs a non‐calcium antagonist hypertension treatment strategy for patients with coronary artery disease. The International Verapamil‐Trandolapril Study (INVEST): a randomized controlled trial. JAMA. 2003;290:2805–2816. [DOI] [PubMed] [Google Scholar]
- 16. Lindholm LH, Carlberg B, Samuelsson O. Should beta blockers remain first choice in the treatment of primary hypertension? A meta‐analysis. Lancet. 2005;366:1545–1553. [DOI] [PubMed] [Google Scholar]
- 17. Bakris GL, Fonseca V, Katholi RE, et al. Metabolic effects of carvedilol vs metoprolol in patients with type 2 diabetes mellitus and hypertension: a randomized controlled trial. JAMA. 2004;292:2227–2236. [DOI] [PubMed] [Google Scholar]
- 18. Kindermann M, Maack C, Schaller S, et al. Carvedilol but not metoprolol reduces beta‐adrenergic responsiveness after complete elimination from plasma in vivo. Circulation. 2004;109: 3182–3190. [DOI] [PubMed] [Google Scholar]
- 19. Cordero A, Bertomeu‐Martinez V, Mazon P, et al. Erectile dysfunction in high‐risk hypertensive patients treated with beta‐blockade agents. CardiovascTher. 2010;28:15–22. [DOI] [PubMed] [Google Scholar]
- 20. Theroux P, Taeymans Y, Morissette D, et al. A randomized study comparing propranolol and diltiazem in the treatment of unstable angina. J Am Coll Cardiol. 1985;5:717–722. [DOI] [PubMed] [Google Scholar]
- 21. Frishman WH. Carvedilol. N Engl J Med. 1998;339:1759–1765. [DOI] [PubMed] [Google Scholar]
- 22. Freemantle N, Cleland J, Young P, et al. Beta blockade after myocardial infarction: systematic review and meta regression analysis. BMJ. 1999;318:1730–1737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Goldberger JJ, Bonow RO, Cuffe M, et al. B‐blocker use following myocardial infarction: low prevalence of evidence‐based dosing. Am Heart J. 2010;160:435–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Bertomeu‐Martinez V, Cordero A, Quiles J, et al. Control of risk factors and treatment of patients with coronary heart disease: the TRECE registry. Rev Esp Cardiol. 2009;62:807–811. [DOI] [PubMed] [Google Scholar]
- 25. Cordero A, Bertomeu‐Martinez V, Mazon P, et al. Differences in medical treatment of chronic coronary heart disease patients according to medical specialities. CardiovascTher. 2009;27: 173–180. [DOI] [PubMed] [Google Scholar]
- 26. Diaz A, Bourassa MG, Guertin MC, et al. Long‐term prognostic value of resting heart rate in patients with suspected or proven coronary artery disease. Eur Heart J. 2005;26:967–974. [DOI] [PubMed] [Google Scholar]
- 27. Camm AJ, Kirchhof P, Lip GY, et al. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 2010;31:2369–2429. [DOI] [PubMed] [Google Scholar]
- 28. Sachdev M, Sun JL, Tsiatis AA, et al. The prognostic importance of comorbidity for mortality in patients with stable coronary artery disease. J Am Coll Cardiol. 2004;43:576–582. [DOI] [PubMed] [Google Scholar]
- 29. Dewilde S, Carey IM, Richards N, et al. Trends in secondary prevention of ischaemic heart disease in the UK 1994 2005: use of individual and combination treatment. Heart. 2008;94:83–88. [DOI] [PubMed] [Google Scholar]
- 30. Bhatt DL, Eagle KA, Ohman EM, et al. Comparative determinants of 4‐year cardiovascular event rates in stable outpatients at risk of or with atherothrombosis. JAMA. 2010;304:1350–1357. [DOI] [PubMed] [Google Scholar]
- 31. Cook S, Togni M, Schaub MC, et al. High heart rate: a cardiovascular risk factor? Eur Heart J. 2006;27:2387–2393. [DOI] [PubMed] [Google Scholar]
- 32. Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol. 2007;50:823–830. [DOI] [PubMed] [Google Scholar]
- 33. Wikstrand J, Hjalmarson A, Waagstein F, et al. Dose of metoprolol CR/XL and clinical outcomes in patients with heart failure: analysis of the experience in metoprolol CR/XL randomized intervention trial in chronic heart failure (MERIT‐HF). J Am Coll Cardiol. 2002;40:491–498. [DOI] [PubMed] [Google Scholar]
- 34. The Cardiac Insufficiency Bisoprolol Study II (CIBIS‐II): a randomised trial: Lancet. 1999;353:9–13. [PubMed] [Google Scholar]
- 35. Packer M, Fowler MB, Roecker EB, et al. Effect of carvedilol on the morbidity of patients with severe chronic heart failure: results of the carvedilol prospective randomized cumulative survival (COPERNICUS) study. Circulation. 2002;106: 2194–2199. [DOI] [PubMed] [Google Scholar]
- 36. Aursnes I, Osnes JB, Tvete IF, et al. Does atenolol differ from other beta‐adrenergic blockers? BMC ClinPharmacol. 2007; 7:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bangalore S, Sawhney S, Messerli FH. Relation of beta‐blocker‐induced heart rate lowering and cardioprotection in hypertension. J Am Coll Cardiol. 2008;52:1482–1489. [DOI] [PubMed] [Google Scholar]
- 38. Hansson L, Hedner T, Lund‐Johansen P, et al. Randomised trial of effects of calcium antagonists compared with diuretics and beta‐blockers on cardiovascular morbidity and mortality in hypertension: the Nordic Diltiazem (NORDIL) study. Lancet. 2000;356:359–365. [DOI] [PubMed] [Google Scholar]
- 39. Grossman E, Messerli FH. Effect of calcium antagonists on plasma norepinephrine levels, heart rate, and blood pressure. Am J Cardiol. 1997;80:1453–1458. [DOI] [PubMed] [Google Scholar]
- 40. Furberg CD, Psaty BM, Meyer JV. Nifedipine. Dose‐related increase in mortality in patients with coronary heart disease. Circulation. 1995;92:1326–1331. [DOI] [PubMed] [Google Scholar]
- 41. Bangalore S, Parkar S, Messerli FH. Long‐acting calcium antagonists in patients with coronary artery disease: a meta‐analysis. Am J Med. 2009;122:356–365. [DOI] [PubMed] [Google Scholar]
- 42. Fox K, Ford I, Steg PG, et al. Ivabradine for patients with stable coronary artery disease and left‐ventricular systolic dysfunction (BEAUTIFUL): a randomised, double‐blind, placebo‐controlled trial. Lancet. 2008;372:807–816. [DOI] [PubMed] [Google Scholar]
- 43. Rodriguez‐Manero M, Cordero A, Bertomeu‐Gonzalez V, et al. Impact of new criteria for anticoagulant treatment in atrial fibrillation [in Spanish]. Rev Esp Cardiol. 2011;64:649–653. [DOI] [PubMed] [Google Scholar]
- 44. Kotseva K, Wood D, De BG, et al. Cardiovascular prevention guidelines in daily practice: a comparison of EUROASPIRE I, II, and III surveys in eight European countries. Lancet. 2009;373: 929–940. [DOI] [PubMed] [Google Scholar]


