Abstract
An efficient phase I trial is a crucial step in developing a new drug in a safe and timely manner. The main objective of a phase I trial is to determine the maximum tolerated dose in order to recommend the dose for a phase II trial. There are many designs that are implemented in phase I trials. Rule-based designs such as the traditional 3 + 3 method and rolling six design are easy to implement and assess for safety using a conservative approach. Model-based designs such as the continual reassessment method and the time-to-event continual reassessment method use mathematical models to increase the precision of dose estimation. The advantages and shortcomings of these designs, along with other designs, are reviewed.
Keywords: clinical trials, phase I, trial design.
Clinical trials are typically prospective research studies comparing the safety and efficacy of a new drug in human beings. When conducted carefully and properly, clinical trials are the safest and fastest approach to identifying whether an intervention is effective for a certain disease. In phase I trials, the goal is to explore toxicity (the level of harmful side effects from a drug in humans) and determine the maximum tolerated dose (MTD) in a small number of patients. The MTD is then applied in phase II trials, which are conducted to assess efficacy while still assessing safety. If there is sufficient efficacy in the experimental arm, the intervention moves on to a phase III trial. These trials are often large, randomized, controlled trials that involve a few hundred to several thousand patients. The goal is to understand the effectiveness of the experimental intervention compared with standard of care, while continuing to assess possible adverse reactions. If the intervention is approved for consumer sale, postmarketing phase IV trials continue. These trials are mostly observational and compare the experimental intervention with similar interventions or standard of care; assess the impact on quality of life; and determine the cost-benefit of the intervention. Here, we will focus on the first of the 4 phases of clinical trials.
The purpose of a phase I clinical trial is to determine safety and tolerable toxicity of an experimental intervention. In oncology, these experimental interventions are often first explored in patients with advanced cancer who failed standard treatments and have limited standard options remaining. A phase I trial may be the first trial of an intervention in humans or may evaluate an approved medication for a new indication. These trials also explore pharmacology and interactions with food and drugs.
The primary objective of most phase I trials is to determine the MTD, defined as the highest dose level at which ≤33% of patients experience dose-limiting toxicity (DLT).1 In Europe and Japan, the MTD is the lowest dose level at which ≥ 33% of patients experience a DLT.2 The DLTs are the toxic effects attributable to the intervention that are measured intolerable, thus limiting the dose escalation.3 Importantly, the DLT is specifically defined in the protocol prior to the beginning of the trial. The DLT is determined using rule-based designs or model-based designs, which are summarized in Table 1. Along with the DLT, all components of the study pertaining to safety, study enrollment criteria, risks associated with the disease or condition, prior knowledge of toxicities, stopping rules, adverse-event reporting, and toxicity monitoring should be clearly specified in the protocol. Furthermore, an important assumption of a phase I clinical trials is that higher dosing leads to a higher probability of efficacy prior to introducing DLTs and MTDs.
Table 1.
Summary of rule-based and model-based designs
| Rule-based designs | Advantages | Disadvantages |
|---|---|---|
| 3+3 | Emphasis on safety; easy to implement; no software is needed. | Longer trial duration; large proportion of patients treated at subtherapeutic doses. |
| Traditional best-of-five |
Easy to implement and safety is emphasized; dhortens trial duration. | Higher proportion of patientstreated at subtherapeutic doses. |
| Rolling six design | Reduces trial length. | Patients are treated at doses below the MTD. |
| Accelerated titration design | Therapeutic doses administered more frequently; patients are treated more rapidly. | Possibility of masking cumulative or delayed toxicities; intrapatient dose escalation data are difficult to interpret. |
| Model-based designs | ||
| Continual reassessment method | Rapid dose escalation; all available patient data are utilized. | Computationally difficult; data must be frequently updated. |
| Time-to-event continual reassessment method | Shortens trial duration; evaluates late-onset toxicities. | Potentially exposes a large number of patients to toxic doses. |
| Seamless design | Toxicity and efficacy outcomes are used for ongoing adaptive dose finding while assessing for safety. | Requires prior Bayesian posterior probabilities. |
Dose-toxicity Curve
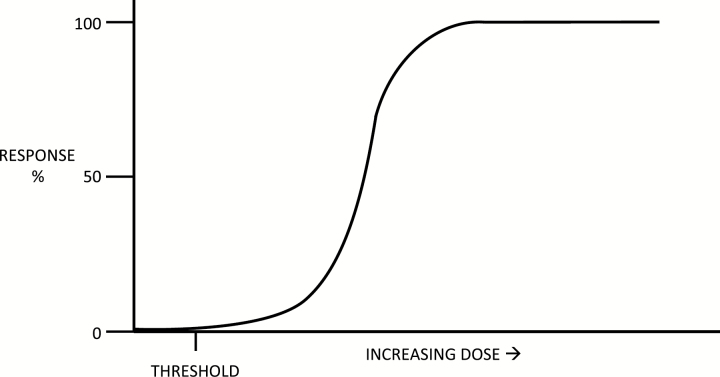
For cytotoxic drugs, it is assumed that efficacy and toxicity increase with dosage.2 Using toxicity as a primary endpoint, the dose-toxicity curve demonstrates the monotonic increasing (entirely nondecreasing) relationship between dosage and the probability of experiencing toxicity. An important point on the dose-toxicity curve is the dose known as the threshold at which no negative effects are experienced from exposure to the intervention (see Fig. 1). A sharp increase in the slope of the curve indicates higher risks of toxic responses as the dose increases. A slope near zero would indicate that as the dose increases, the adverse effects are not as prominent.
Fig. 1.
Dose-toxicity curve. The curve demonstrates the monotonic increasing relationship between dosage and the probability of experiencing a toxicity. The threshold on the curve indicates the point in which no negative effects are experienced from exposure to the intervention.
3 + 3 Design
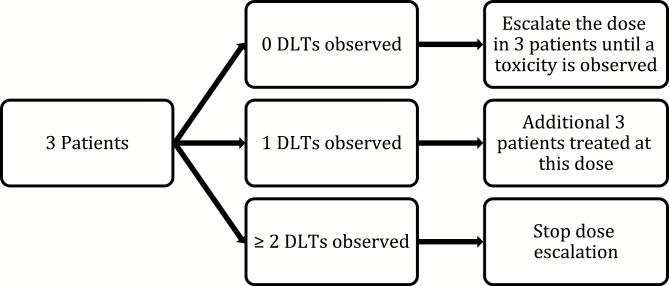
The MTD is most frequently assessed using the 3 + 3 design. In fact, a review of 1235 phase I trials published from 1991 to 2006 found 98.4% used the 3 + 3 design.4 The conventional 3 + 3 design is a dose-escalating trial in which 3 patients are administered an initial dose that is usually defined conservatively from animal trials.5 The standard approach to determining the initial dose is to use 1/10 of the dose associated with 10% lethality in mice. In phase IU.S. trials, the initial dose is 1/3 of the dose associated with 10% lethality in a beagle dog.6 If there are no DLTs observed in these 3 patients, then the dose is increased by a level prespecified in the protocol. This prespecified level is escalated using a modified Fibonacci sequence in which the dose increments decrease as the dose increases.4 The Fibonacci sequence is a sequence of numbers where the subsequent number equals the sum of the previous two numbers: 1, 2, 3, 5, 8, 13, 21, 34 and so forth. A modified Fibonacci sequence is used in phase I trials to increase a particular dose, while decreasing the amount of escalation (100%, 67%, 50%, 40%, and 33% for all subsequent dosing levels).7 The researcher will continue escalating the dose in the patients per dose level until toxicity is observed. If only 1 of the 3 patients experience toxicity, then an additional 3 subjects will be treated at this same dose. If a DLT is observed in 2 or more of the 3 patients, then dose escalation will stop (see Fig. 2). From this data, the MTD is the dose at which the trial was stopped, the previous dose, or a fraction of the stopping dose and becomes the recommended phase II dose (RP2D).
Fig. 2.
3 + 3 design. Dose escalation occurs if there are no DLTs. If there is only 1 DLT that occurs, an additional 3 subjects will be treated at this same dose. Dose escalation stops if a DLT is observed in 2 or more patients.
An advantage of the traditional 3 + 3 design is that it clearly puts patient safety first. An additional benefit of using this traditional design is the ease of implementation where the termination of the trial and the dose escalation are predetermined and no software is necessary.1 Although the 3 + 3 design is easy to implement, the disadvantages are that the trial length can be longer and it is usually performed on very few patients with respect to the RP2D. This leads to a potentially large proportion of patients who are treated at subtherapeutic doses. Due to this disadvantage, the estimate of the MTD is biased and the RP2D is often too low.8
Traditional Best-of-five Design
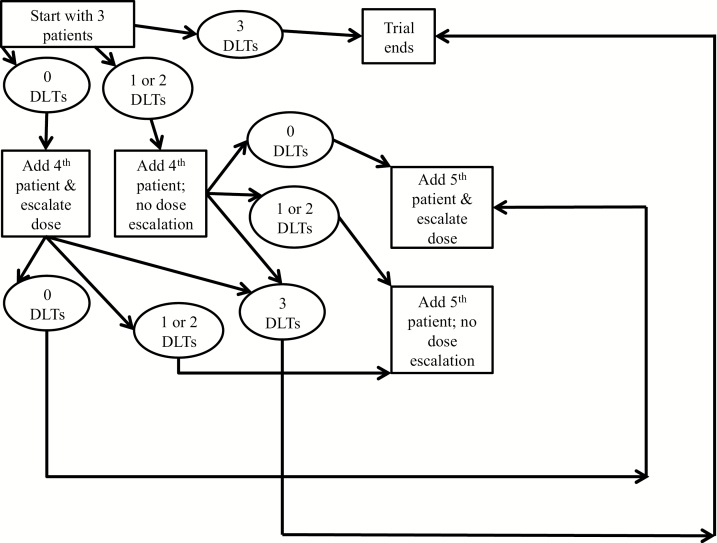
Storer9 describes an alternative design that is similar but more aggressive than the 3 + 3, known as the traditional best-of-five design, also known as the 3 + 1 + 1 design. The traditional best-of-five design differs from 3 + 3 by adding a fourth patient if there are 1 or 2 DLTs in the first 3 patients. If there are 2 DLTs in the first 4 patients, a fifth patient is added. The trial will end if 3 or more DLTs are observed. Dose escalation occurs if no DLTs are observed in the first 3 patients, if no or one DLT is observed in the first 4 patients, or if 2 or fewer DLTs are observed in the total 5 patients (see Fig. 3). The advantages of the traditional best-of-five design are similar to the 3 + 3 design with the addition of reducing trial duration. A disadvantage of this design is an even higher proportion of patients are being treated at subtherapeutic doses in comparison to the 3 + 3 design.
Fig. 3.
Traditional best-of-five design. Dose escalation occurs when no more than 0 out of 3, 1 out of 4, or 2 out of 5 patients experience a DLT. When 3 or more DLTs are observed, the trial ends.
Rolling Six Design
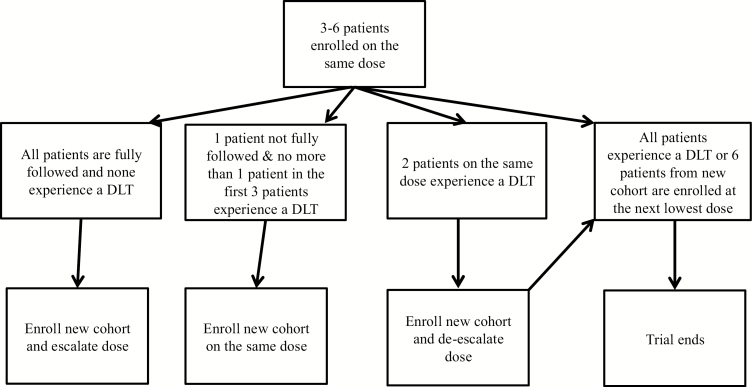
An alternative design, similar to the traditional 3 + 3, known as the rolling six design (RSD), has been adopted in phase I children’s oncology trials as a method of both decreasing suspension of patient accrual and the duration of the trial.10 In the RSD, based on data available from time of enrollment, up to 6 patients can be enrolled at a time and accrual is only stopped when awaiting data from those 6.1 The RSD is similar to the 3 + 3 design in that the trial begins by enrolling 3 patients on the same dose. The RSD differs by allowing 3 to 6 patients to enroll on the same dosage without knowing the DLT status of the patients assigned on that same dose level.12 If all patients are fully followed and do not experience a toxicity, then a new cohort of patients are enrolled at the next highest dose. If all 3 are fully followed and there is only 1 DLT or if 1 patient has not been fully followed and no more than 1 of the first 3 patients has experienced a DLT, a subsequent patient may be enrolled onto the same dose. However, if 2 patients on the same dose experience a DLT, a new cohort of patients are enrolled at the next lowest dose (see Fig. 4). The MTD is defined when 6 patients from a new cohort have entered at the next lowest dose.13 The trial is also suspended if all patients from the initial cohort experience a DLT. The aggressive nature of this design reduces the trial length by decreasing the number of patients who are turned away due to accrual suspension.12 A disadvantage of the RSD is patients are treated at doses below the MTD.
Fig. 4.
Rolling six design. The trial ends when 6 patients from a new cohort have entered at the next lowest dose or if all patients from the initial cohort experience a DLT.
Accelerated Titration Design
To address the issue of potentially administering subtherapeutic doses to patients, Simon et al14 discuss a variation of the traditional 3 + 3 design called the accelerated titration design (ATD). In the ATD, intrapatient dose escalation occurs by 40% for patients continuing with the study if there are no DLTs at the current dose.15 If a low-grade toxicity (predefined in the protocol) is observed, the dose remains constant for that patient’s next cycle. If a DLT is observed, then that patient is either taken off study or the dose is de-escalated. The rules for dose escalation and de-escalation are typically predetermined in the protocol based on DLT and a moderate level of toxicity.16 Typically, the MTD is determined in the ATD by using the conventional 3 + 3 escalation rule.2 For example, the 3 + 3 design is implemented after the accelerated phase. The accelerated phase is when only 1 patient is included per dose level while leaving the opportunity for clinicians to allow intrapatient dose escalation to occur. After the accelerated phase, the 3 + 3 design is used as a stopping rule. In addition to delivering therapeutic doses to patients, this design will also ensure that patients are treated more rapidly. The shortcomings of this design are the possibility of masking cumulative or delayed toxicities and that intra-patient dose escalation data is difficult to interpret.
Continual Reassessment Method
Using Bayesian statistical models in phase I clinical trials allows researchers to find a prespecified DLT using toxicity data from all patients. The traditional frequentist approach to clinical trials draws conclusions based only on the data collected during the current trial. The Bayesian approach (as opposed to the frequentist approach) is to draw conclusions based on the combination of a statistical model and data that were available before the trial began. As such, Bayesian models are more difficult to compute as they require the prior distribution (the data available before conducting the trial) from preclinical data or from experts who have experience with similar drugs, if they exist.2
O’Quigley et al17 discussed the first Bayesian model in phase I trial designs called the continual reassessment method (CRM) in which all patients are treated one at a time at the dose predicted to be closest to the MTD. To determine the MTD, various stopping rules have been proposed. The conservative approach to determine the MTD is when 6 patients have already been assigned to the same dose.3 An alternative proposal is to determine the MTD by reaching a prespecified probability of a DLT that is usually in the 40% to 50% range. Subsequent patient dosing is based on toxicity outcomes of all previously treated patients on the trial by modeling the relationship between dose and toxicity at each stage of the trial.18
The first CRM has been modified several times because it could expose patients to high levels of toxicities if the model is incorrect. Korn et al3 recommend treating the initial patient on the lowest starting dose based on animal toxicity trial data as well as treating numerous patients on the same dosage. The CRM has also been modified by allowing dose escalation to occur 1 prespecified level at a time18 and by not allowing dose escalation to occur for future patients if the previous patient experienced a DLT.19 The modifications of the CRM result in a more rapid dose escalation and utilize all available data from all patients. The disadvantage is that the model can be computationally difficult as data from every patient or cohort must be frequently updated.
Time-to-event Continual Reassessment Method
Cheung and Chappell20 designed an extension of the CRM that evaluates late-onset toxicities while reducing trial duration called the time-to-event continual reassessment method (TITE-CRM). The TITE-CRM allows patient accrual in a staggered manner without needing to follow up on DLTs for previously treated patients. It is advantageous by allowing a shorter trial duration; however, the model is bound by the assumption that the hazard of toxicity remains constant over time.2 The disadvantage of this design is its potential to expose a large number of patients to toxic doses, when patient accrual is fast and late-onset toxicities are present.
Molecularly Targeted Agents
Given the potential harmful effects on normal tissues, cytotoxic treatments are typically evaluated using toxicity endpoints. Unlike cytotoxic treatments, molecularly targeted agents focus on specific abnormalities in particular cancer cells with the intent to limit off-target effects on normal tissues. Using toxicity as the only endpoint for determining the RP2D may be inappropriate for molecularly targeted agents. A researcher might consider using the pharmacologic effects of the body on the drug (pharmacokinetics) as a primary endpoint. For instance, pharmacokinetics could be assessed by observing how long it takes for a drug to be absorbed in the body. In order to obtain a RP2D that is close to the optimal active dose, it is necessary to establish a dose range by combining toxicity data.21 Booth et al22 have proposed an algorithm that combines toxicity data and defines the upper limit of a dose range using toxicity data and the lower limit using pharmacokinetic data. They also recommend that levels be created within a phase I trial by expanding cohorts and/or creating separate dose ranges.22 No specific trial designs have been established for molecularly targeted agents that do not have a proven relevant target.2
Seamless Design
In an effort to reduce trial costs and duration, a proposed seamless design combines phase I and phase II trials and evaluates both the toxicity and efficacy of drug combinations in 1 trial.23 The first phase typically employees the 3 + 3 approach to find the MTD (although other approaches may be used), and the data from this phase is subsequently used in the second phase to assess efficacy. Toxicity and efficacy results are assessed throughout the trials and the most effective drug combination is selected.24 This design could be particularly attractive for using a known drug for a new indication, where toxicity is well established. The advantage of this design is that the toxicity and efficacy outcomes are used for ongoing adaptive dose finding while assessing for safety. The disadvantage of this design is when different endpoints are used at different stages, applying standard statistical methodologies to assess for treatment effects and sample size calculations is computationally difficult.25
Conclusion
Determining an appropriate phase I clinical trial design can be a complex decision for a clinician that requires the consideration of multiple factors (eg, trial length, dose escalation, patient population, etc.). To better guide a clinician on selecting the best designs in a particular setting, all preclinical and clinical data should be examined. For instance, if preclinical data indicate an uncertainty about drug tolerability, a more conservative approach such as the 3 + 3 or the traditional best-of-five would be the most appropriate design. However, if preclinical data indicate an expected low level of toxicity in patients, ATD or the CRM would be the most appropriate decision. Given that pediatric phase I trials are only conducted following the completion of adult trials, the RSD appears to be the most appropriate because the design emphasizes shortening trial duration.10 A clinician should keep in mind that there are advantages and disadvantages when using these designs. A recent simulation-based comparison showed that the 3 + 3 and the RSD are at risk for treating a significant proportion of patients at a dose lower than the MTD.12 It was also found that the duration of the 3 + 3 was higher compared to the RSD10,11 and the CRM,12 but the duration of the RSD was higher than the CRM.20
Clinicians interested in using time-to-event (eg, survival) endpoints should consider the TITE-CRM. In comparison to the RSD, the TITE-CRM can treat all available patients (RSD cannot), identifies the MTD more accurately, and patients are less susceptible to unacceptable toxic doses.13 For trials that combine both phase I and II, the seamless design should be considered in order to expedite drug development. By accelerating the process of phase I and II trials into one trial, trial duration and costs can be reduced. The seamless design would also be appropriate when evaluating an approved drug for a new indication because prior data could potentially establish a new MTD and efficacy can be assessed in a fast and efficient manner.
Although model-based designs provide a more rapid dose escalation and take into consideration unanticipated events, they are not as easy to implement and there is always a possibility that dosage may be miscalculated. Rule-based designs are easier to implement and are considered safest. Additionally, longer trial duration could also be potentially unsafe for patients awaiting dose escalation or de-escalation. As software advances, further investigation needs to be conducted in order to bridge the gaps between rule-based and model-based phase I designs.
Funding
National Cancer Institute-funded: Research grant (R01 CA163687).
Conflict of interest statement. None declared.
References
- 1. Storer BE. Design and analysis of phase I clinical trials. Biometrics. 1989;925–937. [PubMed] [Google Scholar]
- 2. Le Tourneau C, Lee JJ, Siu LL. Dose escalation methods in phase I cancer clinical trials. J Natl Cancer Inst. 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Korn EL, Midthune D, Chen TT, Rubinstein LV, Christian MC, Simon RM. A comparison of two phase I trial designs. Stat Med. 1994;13(18):1799–1806. [DOI] [PubMed] [Google Scholar]
- 4. Rogatko A, Schoeneck D, Jonas W, Tighiouart M, Khuri FR, Porter A. Translation of innovative designs into phase I trials. J Clin Oncol. 2007;25(31):4982–4986. [DOI] [PubMed] [Google Scholar]
- 5. Arbuck SG. Workshop on phase I study design Ninth NCI/EORTC New Drug Development Symposium, Amsterdam. Ann Oncol. 1996;7(6):567–573. [DOI] [PubMed] [Google Scholar]
- 6. Rubinstein LV, Simon RM. Phase I clinical trial design. in: Budman DR, Calvert AH, Rowinsky EK.(eds) Handbook of Anticancer Drug Development, Elsevier: Amsterdam; 2003. [Google Scholar]
- 7. Omura GA. Modified fibonacci search. J Clin Oncol. 2003;21(16):3177–3177. [DOI] [PubMed] [Google Scholar]
- 8. Hansen AR, Graham DM, Pond GR, Siu LL. Phase 1 trial design: is 3+ 3 the best?. Cancer Control. 2014;21(3). [DOI] [PubMed] [Google Scholar]
- 9. Storer BE. An evaluation of phase I clinical trial designs in the continuous dose–response setting. Stat Med. 2001;20(16):2399–2408. [DOI] [PubMed] [Google Scholar]
- 10. Skolnik JM, Barrett JS, Jayaraman B, Patel D, Adamson PC. Shortening the timeline of pediatric phase I trials: the rolling six design. J Clin Oncol. 2008;26(2):190–195. [DOI] [PubMed] [Google Scholar]
- 11. Hartford C, Volchenboum SL, Cohn SL. 3+ 3≠(Rolling) 6. J Clin Oncol. 2008;26(2):170–171. [DOI] [PubMed] [Google Scholar]
- 12. Onar-Thomas A, Xiong Z. A simulation-based comparison of the traditional method, rolling-6 design and a frequentist version of the continual reassessment method with special attention to trial duration in pediatric phase I oncology trials. Contemp Clin Trials. 2010;31(3):259–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhao L, Lee J, Mody R, Braun TM. The superiority of the time-to-event continual reassessment method to the rolling six design in pediatric oncology phase I trials. Clin Trials. 2011;8(4):361–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Simon R, Rubinstein L, Arbuck SG, Christian MC, Freidlin B, Collins J. Accelerated titration designs for phase I clinical trials in oncology. J Natl Cancer Inst. 1997;89(15):1138–1147. [DOI] [PubMed] [Google Scholar]
- 15. Ivy SP, Siu LL, Garrett-Mayer E, Rubinstein L. Approaches to phase 1 clinical trial design focused on safety, efficiency, and selected patient populations: a report from the clinical trial design task force of the national cancer institute investigational drug steering committee. Clin Cancer Res. 2010;16(6):1726–1736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dancey J, Freidlin B, Rubinstein L. Accelerated titration designs. In: Chevret S, editor. Statistical methods for dose-finding experiments.Chichester, West Sussex (England); Hoboken (NJ): Wiley Press; 2006:91–114. [Google Scholar]
- 17. O’Quigley J, Pepe M, Fisher L. Continual reassessment method: a practical design for phase 1 clinical trials in cancer. Biometrics. 1990;33–48. [PubMed] [Google Scholar]
- 18. Polley MYC. Practical modifications to the time-to-event continual reassessment method for phase I cancer trials with fast patient accrual and late-onset toxicities. Stat Med. 2011;30(17):2130–2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Faries D. Practical modifications of the continual reassessment method for phase I cancer clinical trials. J Biopharm Stat. 1994;4(2):147–164. [DOI] [PubMed] [Google Scholar]
- 20. Cheung YK, Chappell R. Sequential designs for phase I clinical trials with late-onset toxicities. Biometrics. 2000;56(4):1177–1182. [DOI] [PubMed] [Google Scholar]
- 21. Postel-Vinay S, Arkenau HT, Olmos D, et al. Clinical benefit in phase-I trials of novel molecularly targeted agents: does dose matter?. Br J Cancer 2009;100(9), 1373–1378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Booth CM, Calvert AH, Giaccone G, Lobbezoo MW, Seymour LK, Eisenhauer EA. Endpoints and other considerations in phase I studies of targeted anticancer therapy: recommendations from the task force on methodology for the development of innovative cancer therapies (MDICT). Eur J Cancer. 2008;44(1):19–24. [DOI] [PubMed] [Google Scholar]
- 23. Huang X, Biswas S, Oki Y, et al. A parallel phase I/II clinical trial design for combination therapies. Biometrics. 2007;63:429–36. [DOI] [PubMed] [Google Scholar]
- 24. Zang Y, Lee JJ. Adaptive clinical trial designs in oncology. Chin Clin Oncol. 2014;3(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chow SC, Chang M. Adaptive design methods in clinical trials-a review. Orphanet J Rare Dis. 2008;3(11):169–90. [DOI] [PMC free article] [PubMed] [Google Scholar]