This article describes the clinic‐pathological and molecular features of two young male patients, who presented with locally advanced and metastatic colorectal cancer. These cases show the importance of the special care and molecular diagnostic procedures needed for young patients.
Abstract
Colorectal cancer (CRC) is rare in young patients without a confirmed family history of cancer. Reports of an increased prevalence of POLD1/POLE mutations in young patients with colorectal cancer have raised awareness and support routine genetic testing for patients with early‐onset tumors. In cases of CRC without proven MMR‐germline mutation, molecular analyses are warranted to confirm or rule out other familial CRC syndromes. This article describes the cases of two young male patients, who presented with locally advanced and metastatic CRC, and reports the results of the germline mutational analyses done for both patients. These cases demonstrate the importance of special care and molecular diagnostic procedures for young patients with CRC.
Key Points.
Patients with colorectal cancer who are younger than 50 years at initial diagnosis (early onset) should routinely undergo genetic testing.
Early‐ and very‐early‐onset patients (younger than 40 years) with absence of microsatellite instability should be considered for tumor mutation burden testing and/or DNA polymerase proofreading mutation.
The mutational signature of HSP110 within mismatch repair deficiency‐related tumors may help to identify patients likely to benefit from 5‐fluorouracil‐based chemotherapy.
Intensified, maintained, and specific surveillance may help to reduce secondary tumor progression.
Introduction
Colorectal cancer (CRC) is clinically and molecularly heterogeneous. Attempts to stratify patients into subtypes have resulted in the description of four “consensus molecular subtypes” (CMSs) [1], now known to be already detectable in colorectal premalignancy [1], [2]: (a) CMS1, characterized by hypermutation, marked levels of BRAFV600E mutation, and high immunogenicity; (b) CMS2, the canonical subtype with WNT/MYC pathway activation; (c) CMS3, with KRAS mutations and metabolic dysregulation; and (d) CMS4, a mesenchymal, stroma‐rich subtype [1], [2].
Based on this classification, CRC are either hypermutated or ultramutated (∼16%) or nonhypermutated (∼84%) [3]. High tumor mutation burden is traditionally associated with mismatch repair deficiency (MMR‐D; i.e., CMS1) and a correspondingly high level of microsatellite instability (MSI) in coding regions of MSI target genes [4]. MMR‐D may be sporadic or inherited as part of Lynch syndrome (LS), mostly arising as a consequence of germline mutations in one of the MMR genes [5], [6].
Somatic or germline mutations in the DNA polymerases (POLD1 and POLE) have been identified as a second pathogenic mechanism, contributing to an ultramutated tumor phenotype because of a multitude of base substitution mutations [7]. The estimated frequency of DNA polymerase mutations in CRC is around 1%–3% [8] but with substantial influence on tumor development and/or progression [9]. A growing number of reports have increased awareness of a prevalence of POLD1 and POLE mutations in young male patients [9], [10].
Patients with early‐onset tumors should routinely undergo genetic testing [6]. In cases without MMR germline mutation, molecular analyses are warranted to confirm or deny other familial CRC syndromes, such as familial adenomatous polyposis and MUTYH‐associated polyposis. Herein, we describe the clinicopathological and molecular features of two young male patients, aged 33 and 36 years at diagnosis, who presented with a locally advanced and metastatic CRC. The cases demonstrate the importance of special care and molecular diagnostic procedures for young patients.
Clinical Cases
Patient #1
A 33‐year‐old male patient presented with pain in the right lower abdomen. Appendicitis was suspected clinically, and the patient underwent an ileocecal wedge resection. Histopathologically, adenocarcinoma of the coecum and appendix including perforation in the appendix region was diagnosed. Because subsequent tumor staging by computed tomography (CT) revealed no distant metastases, oncologic resection was completed (right hemicolectomy plus peritonectomy). On pathological examination, the surgical specimen was found to harbor the remaining tumor in the ascending colon, infiltrating pericolic tissue, as well as metastases in regional lymph nodes [G3 pT4a(m) pN2b(21/48) L1 V1 Pn1 R0]. Absence of MLH1 and PMS2 by immunohistochemistry (IHC) and high‐degree MSI (BAT25, BAT26, Cat25, D2S123, and D17S250) confirmed MMR‐D. Additional testing showed that the patient's tumor had no evidence of MLH1 promoter methylation and no somatic BRAFV600E mutations. Genetic testing was initiated.
One month after surgery the patient started adjuvant treatment (FOLFOX4) according to recommendations for Union for International Cancer Control stage IIIC CRC. Because of recurrent neutropenia, dose reductions were necessary. After cycle 10, the patient reported recurrent pain in the right groin. The CT scan showed a peritoneal tumor in the right pelvis of 3 cm diameter, infiltrating the ileum and abdominal wall. The tumor was resected in toto. Histopathological examination proved a metastatic CRC deposit. The patient was without evidence of tumor in close follow‐up 11 months after initial diagnosis.
The patient's family history identified gastric cancer in a maternal grandfather who died of the disease (aged 68 years).
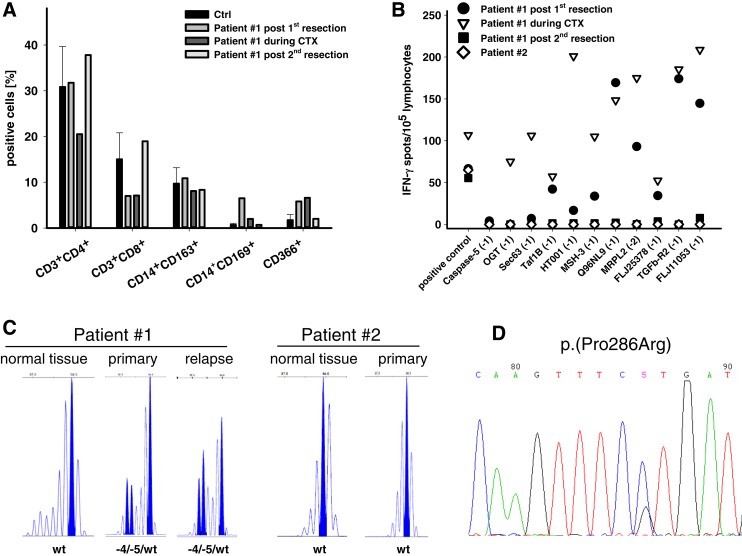
The high immunogenicity of MMR‐D‐related diseases, along with the observation of a host's MMR‐D‐specific immune surveillance [11], [12], prompted us to perform immune monitoring at different intervals (Fig. 1A). Of note, the percentage of immunosuppressive CD14+CD169+ monocytes and CD366+ T cells decreased only upon removal of the metastatic deposit. On the other hand, amounts of CD3+CD8+ cells increased. Although this interesting finding must be interpreted cautiously, surveys on immune status during or after therapy could—upon validation in a larger patient cohort—potentially act as a surrogate marker. In support of this, MSI‐specific immune responses (enzyme‐linked immonospot assay; Fig. 1B) increased shortly after chemotherapy but declined after the second resection. This additional finding is based on a study in which peptide‐specific T‐cell responses were found to be higher in patients with MMR‐D CRC compared with healthy mutation carriers [11].
Figure 1.
Immune‐based and molecular pathological analyses. (A): Flow cytometric phenotyping for assessment of immune status from patient #1 before (after first surgery), during chemotherapy, and after second surgical intervention in comparison with healthy control. (B): IFN‐γ ELISpot. Peripheral blood mononuclear cells were seeded in anti‐IFN‐γ enzyme‐linked immunospot (ELISpot) plates and cultured overnight in the presence of different microsatellite instability (MSI)‐derived peptides [27]. Finally, bound antibody was visualized by BCIP/NBT; spots were counted using an ELISpot reader. Presented are the numbers of IFN‐γ‐secreting cells per 100,000 effector cells corrected for background levels counted in the absence of target cells. (C): MSI analysis of HSP110 was done by polymerase chain reaction (PCR) amplification of target sequence using HSP110‐specific primer sequences spanning T17 repeat. Fluorescently labeled DNA fragments were analyzed on an ABI 3500 Genetic Analyzer. Normal adjacent tissue from patients served as controls. Instability was scored if novel peaks were obtained compared with controls or if the ratio of peak areas of corresponding peaks in tumor samples and stable controls revealed values ≤ 0.5 or ≥ 2. (D): Mutational analysis of the POLE gene (exons 9–14) was done by PCR amplification of the target exons and subsequently analyzed by Sanger sequencing on an ABI 3500 Genetic Analyzer.
Abbreviations: Ctrl, healthy control; CTX, chemotherapy; IFN‐γ, interferon γ.
Patient #2
The 36‐year‐old male patient presented with colic‐type pain, nausea, and vomiting. The clinical workup by abdominal ultrasound and CT scan of thorax and abdomen identified a circular tumor at the right colonic flexure and multiple bilobar hepatic metastases. Colonoscopy and biopsy led to the diagnosis of a colonic adenocarcinoma. Therapy was planned in a multidisciplinary tumor board. Because of symptoms of stenosis, laparoscopic right hemicolectomy was performed first (week 1). Tumor growth into the pericolic tissue, but absence of nodal metastases, was demonstrated by histopathology [G2 pT3 pN0(0/19) L0 V2 R2 pM1]. Because MMR expression was completely retained by IHC and no evidence of MSI was detected by polymerase chain reaction, LS was deemed unlikely. Complete resection of liver metastases was reported histopathologically after associating liver partition and portal vein ligation for staged hepatectomy 4 weeks later (week 5). Shortly after surgery (week 9), multiple new liver metastases were detected, as well as ascites and pleural effusions. Chemotherapy with mFOLFOX6 was initiated (week 13). During the first two cycles there was rapid improvement of the patient's overall condition. Regression of liver metastases, ascites, and pleural effusion was confirmed radiologically after five cycles. Chemotherapy was continued for additional five cycles, with the addition of bevacizumab from cycle 7. Subsequent staging examinations showed further tumor regression with only small residual liver metastases (week 33). Because of polyneuropathy symptoms, the therapeutic protocol was modified to bevacizumab plus 5‐fluorouracil (5‐FU) and leucovorin. After two cycles the patient was lost to follow‐up (week 37).
Family history revealed that this patient's father had undergone regular endoscopic treatment for gastrointestinal polyps. An aunt was diagnosed with CRC (aged 65 years), and an uncle was diagnosed with unspecified cancer (aged 40 years).
Molecular Tumor Board
Genotyping Results and Interpretation of the Molecular Results
CRC arises either sporadically or in the setting of inherited syndromes. In addition to the most prevalent LS and the familial adenomatous polyposis, some other rare syndromes contribute to early‐onset CRC [13], [14]. To identify potential driver mutations and thus make a precise diagnosis, germline mutational analysis was done for both patients with the Illumina EBM Panel (paired‐end mode; National Center for Tumor Diseases, Dresden, Germany; coverage, 10×): APC, ATM, BMPR1A, BRCA1/2, BRIP1, CHEK2, CDH1, EPCAM, EXO1, GALNT12, MLH1/3, MSH2/6, MUTYH, NBN, NF1, NTHL1, PALB2, PMS1/2, POLD1/E, PRSS1, PTEN, RAD51C/D, SMAD, SPINK1, STK11, TP53.
Unexpectedly, germline MMR gene mutation could not be demonstrated in patient #1, putting him into the group of “suspected LS” [15]. However, a heterozygous germline mutation in the tumor suppressor gene BRCA2 [c.9190G > T, p.(Asp3064Tyr)] was identified. Although CRC does not fall into the typical cancer spectrum [16], and this particular mutation is predicted to be neutral (https://databases.lovd.nl), BRCA2 is the more important gene for men (vs. BRCA1 in women), linked to a wide spectrum of cancers, among them breast, pancreatic, and prostate cancer [17]. One may thus speculate that there were some indirect effects of the identified mutation, with potential higher vulnerability for somatic MMR‐D. A recent large‐cohort‐based study identified BRCA1/2 mutations in 1% of patients with CRC and suspected LS [18]. Another study reported mutations in high‐ or moderate‐penetrance genes not traditionally associated with CRC among 450 patients with early‐onset CRC and described two cases with BRCA1 as well as four cases with BRCA2 mutations [13]. These data (a) support findings on a direct link between CRC risk and BRCA1/2 mutations and (b) show that BRCA2 mutations predispose to a wider diversity of cancer than predicted. And, vice versa, the fact that BRCA2 is a frequent MSI target gene [19] argues in favor of an interplay between BRCA2 and MMR‐D.
Recently, HSP110 was proposed as a molecular biomarker to be tested prior to chemotherapy [20], [21]. Roughly 30% of all MMR‐D cases harbor truncating mutations in the chaperone HSP110 gene. These mutations result from large deletions within a noncoding T17 repeat, leading to exon 9 skipping (HSP110ΔE9) and production of a nonfunctioning protein [21]. MMR‐D tumors with this mutation respond to treatment with 5‐FU and oxaliplatin [20]. In patient #1, the tumor harbored a truncating version of HSP110 (wt/‐4/‐5; Fig. 1C), which was also confirmed at relapse (Fig. 1C).
In patient #2, no germline mutations were detected by the gene panel. Genomic profiling of the primary tumor also revealed no mutations in classical hotspot genes such as KRAS, NRAS, BRAF, and PIK3CA. Extended mutational analyses covering the DNA polymerases demonstrated a somatic POLE P286R mutation p.(Pro286Arg) in the primary tumor and metastasis (Fig. 1D). This mutation, located along the DNA‐binding pocket, has been identified as a recurrent somatic hotspot mutation [22] and is predicted to have a direct effect on the proofreading function [7]. Therefore, the p.(Pro286Arg) mutation is very likely a driver for tumor initiation and progression and may be at the base of aggressive tumor growth.
Functional and Clinical Significance of the Specific Mutation in the Particular Cancer
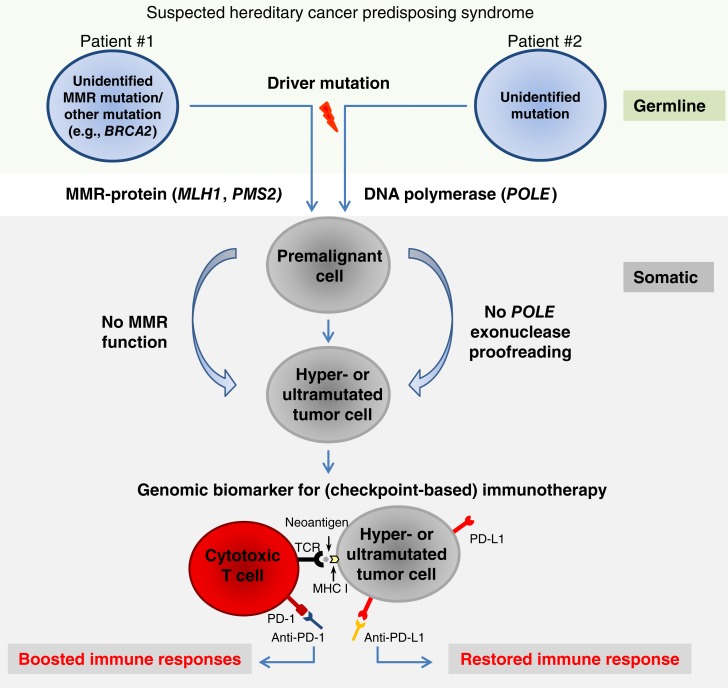
The two cases described here have some parallels with potential consequences for therapy decision and follow‐up (Fig. 2). Patient #1 has suspected LS, with somatically confirmed MMR‐D but a heterozygous germline mutation in BRCA2 [p.(Asp3064Tyr)]. BRCA2 is one of the most crucial DNA repair genes [23]. Also, few studies associate breast cancer with LS [24], and no guidelines yet recommend intensified screening [24]. About 6% of all CRC cases are classified as suspected LS [15]; that is, no germline mutation in the causative MMR genes is detected during routine diagnostics. When only considering early‐onset CRC, numbers of patients with suspected LS are even higher. Overall cancer risks in patients and their relatives are largely unknown because of heterogeneity in their clinical presentation. For first‐degree relatives, an intermediate risk is described [15]. Implementation of optimal screening strategies according to underlying genetic alterations—well balanced between over‐ and under‐screening—is likely to improve the number of cases identified (Fig. 3).
Figure 2.
Molecular mechanisms of hypermutation and its applicability as a genomic biomarker for therapy decision. Depending on the germline mutation and additional somatic mutations, immunotherapeutic concepts aim to restore (e.g., in the case of POLE mutations) or reactivate (e.g., in the case of mismatch repair deficiency) preformed (T‐cell‐) mediated immune responses to control tumor disease and eventually prevent or delay relapse.
Abbreviations: MHC, major histocompatibility complex; MMR, mismatch repair; PD‐1, programmed cell death 1; PD‐L1, programmed cell death ligand 1; TCR, T‐cell receptor.
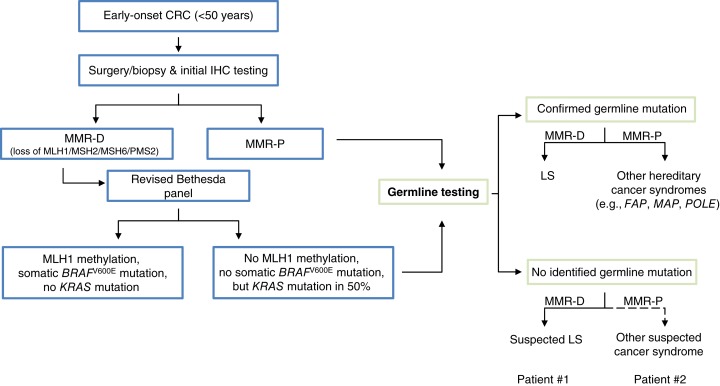
Figure 3.
Strategies to diagnose hereditary cancer syndromes based on current clinical practice. A proposed algorithm for diagnostic procedures is displayed along with intensified and maintained surveillance for risk stratification of each individual patient.
Abbreviations: CRC, colorectal cancer; FAP, familial adenomatous polyposis; IHC, immunohistochemistry; LS, Lynch syndrome; MAP, MUTYH‐associated polyposis; MMR‐D, mismatch repair deficiency; MMR‐P, mismatch repair proficiency; POL, DNA polymerase.
Similarly, POLE‐mutant CRC is characterized by hypermutation. Hodel et al. described loss of a single POLE proofreading allele to be sufficient for driving these tumors [10]. The p.(Pro286Arg) mutation in patient #2 is among the more frequent of the pathogenic mutations described, inducing a strong mutator phenotype that is comparable to complete MMR‐D.
Potential Strategies to Target the Pathway and Implications for Clinical Practice
MMR‐D is considered a marker for drug response and resistance, such as good response toward immune‐checkpoint inhibition and its recent U.S. Food and Drug Administration approval as therapy after standard treatment. On the other hand, responsiveness toward drugs, such as 5‐FU and oxaliplatin, is described differentially [25]. To address drug response molecularly, a large clinical trial is examining the predictive value of HSP110ΔE9 (NCT02458664). Although it is not yet a standard procedure in clinical management, HSP110ΔE9 testing may become valuable for adjuvant chemotherapy decisions.
For POLE‐mutant CRC, classification into one of the CMS remains an open question. Although MMR‐D tumors fall into the CMS1 group (i.e., immunogenic [3]), assignment to this group was not formally shown for POLE‐mutant tumors. However, the well‐known hypermutated status and results from studies on POLE‐mutant‐derived major histocompatibility complex I peptides point toward high immunogenicity [26], similar to MMR‐D tumors [27]. This makes them likely candidates for CMS1 type.
Like high‐degree MSI, POLE mutations in CRC may have important consequences for therapy: although these tumors may be susceptible to immunotherapy, their response to standard chemotherapy is not clear [26]. Patient #2’s tumor rapidly progressed after surgery but responded well to 5‐FU‐based chemotherapy. This individual observation is obviously far from representative. Nevertheless, we would like to emphasize that this interesting finding warrants further investigation in larger patient cohorts.
Finally, we conclude that there are strong arguments for including POLE mutational analyses into clinical testing of patients with early‐ or very‐early‐onset CRC. This may help to refine risk stratification and forward development of specific therapeutic strategies.
Patient Updates
Patient #1
During close follow‐up, the CT scan 4 months after last resection showed progression in mesenterial and retroperitoneal lymph nodes. Therefore, chemotherapy with FOLFIRI plus bevacizumab (because KRAS G13D mutation was present) was started.
Patient #2
This patient presented again at the oncology department about 7 months after last contact. He was in very good clinical condition with no signs of tumor manifestations on clinical examination. Because of lack of symptoms, he had not sought any medical contact in the meantime, nor had he received any chemotherapy. CT scan (week 72) showed slow progression of liver metastases as well as some new lesions but no tumor manifestations at other sites. 5‐FU‐based chemotherapy (FOLFIRI) plus bevacizumab was reinitiated.
Acknowledgments
The authors thank the Department of Surgical Research and the Department of Clinical Genetics, Universitätsklinikum of the Technische Universität Dresden, Dresden, Germany, for performing germline mutational analysis. This work was supported by a grant from the German Research Foundation to C.M. (grant MA5799/2‐1).
Author Contributions
Conception/design: Claudia Maletzki
Provision of study material or patients: Maja Hühns, Ingrid Bauer, Larissa Henze
Collection and/or assembly of data: Claudia Maletzki
Data analysis and interpretation: Claudia Maletzki, Maja Hühns, Ingrid Bauer
Manuscript writing: Claudia Maletzki, Larissa Henze
Final approval of manuscript: Friedrich Prall, Christian Junghanss
Disclosures
The authors indicated no financial relationships.
References
- 1.Komor MA, Bosch LJ, Bounova G et al. Consensus molecular subtype classification of colorectal adenomas. J Pathol 2018;246:266–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang K, Willis JA, Reumers J et al. Colorectal premalignancy is associated with consensus molecular subtypes 1 and 2. Ann Oncol 2018;29:2061–2067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Muller MF, Ibrahim AEK, Arends MJ. Molecular pathological classification of colorectal cancer. Virchows Arch 2016;469:125–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kloor M, Von Knebel Doeberitz M. The immune biology of microsatellite‐unstable cancer. Trends in Cancer 2016;2:121–133. [DOI] [PubMed] [Google Scholar]
- 5.Pritchard CC, Grady WM. Colorectal cancer molecular biology moves into clinical practice. Gut 2011;60:116–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pérez‐Carbonell L, Ruiz‐Ponte C, Guarinos C et al. Comparison between universal molecular screening for Lynch syndrome and revised Bethesda guidelines in a large population‐based cohort of patients with colorectal cancer. Gut 2011;61:865–872. [DOI] [PubMed] [Google Scholar]
- 7.Castellucci E, He T, Goldstein DY et al. DNA polymerase ε deficiency leading to an ultramutator phenotype: A novel clinically relevant entity. The Oncologist 2017;22:497–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nebot‐Bral L, Brandao D, Verlingue L et al. Hypermutated tumours in the era of immunotherapy: The paradigm of personalised medicine. Eur J Cancer 2017;84:290–303. [DOI] [PubMed] [Google Scholar]
- 9.Temko D, Tomlinson IPM, Severini S et al. The effects of mutational processes and selection on driver mutations across cancer types. Nat Commun 2018;9:1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hodel KP, De Borja R, Henninger EE et al. Explosive mutation accumulation triggered by heterozygous human Pol ε proofreading‐deficiency is driven by suppression of mismatch repair. Elife 2018;7:1–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schwitalle Y, Kloor M, Eiermann S et al. Immune response against frameshift‐induced neopeptides in HNPCC patients and healthy HNPCC mutation carriers. Gastroenterology 2008;134:988–997. [DOI] [PubMed] [Google Scholar]
- 12.Saeterdal I, Bjorheim J, Lislerud K et al. Frameshift‐mutation‐derived peptides as tumor‐specific antigens in inherited and spontaneous colorectal cancer. Proc Natl Acad Sci 2001;98:13255–13260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pearlman R, Frankel WL, Swanson B et al. Prevalence and spectrum of germline cancer susceptibility gene mutations among patients with early‐onset colorectal cancer. JAMA Oncol 2017;3:464–471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fearon ER. Molecular genetics of colorectal cancer. Annu Rev Pathol 2011;6:479–507. [DOI] [PubMed] [Google Scholar]
- 15.Buchanan DD, Rosty C, Clendenning M et al. Clinical problems of colorectal cancer and endometrial cancer cases with unknown cause of tumor mismatch repair deficiency (suspected Lynch syndrome). Appl Clin Genet 2014;7:183–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Risch HA, McLaughlin JR, Cole DEC et al. Population BRCA1 and BRCA2 mutation frequencies and cancer penetrances: A kin‐cohort study in Ontario, Canada. J Natl Cancer Inst 2006;98:1694–1706. [DOI] [PubMed] [Google Scholar]
- 17.Liede A, Karlan BY, Narod SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: A review of the literature. J Clin Oncol 2004;22:735–742. [DOI] [PubMed] [Google Scholar]
- 18.Yurgelun MB, Kulke MH, Fuchs CS et al. Cancer susceptibility gene mutations in individuals with colorectal cancer. J Clin Oncol 2017;35:1086–1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zaanan A, Meunier K, Sangar F. Microsatellite instability in colorectal cancer: From molecular oncogenic mechanisms to clinical implications. Cell Oncol (Dordr) 2011;34:155–176. [DOI] [PubMed] [Google Scholar]
- 20.Collura A, Lagrange A, Svrcek M et al. Patients with colorectal tumors with microsatellite instability and large deletions in HSP110 T17 have improved response to 5‐fluorouracil‐based chemotherapy. Gastroenterology 2014;146:401–411.e1. [DOI] [PubMed] [Google Scholar]
- 21.Dorard C, De Thonel A, Collura A et al. Expression of a mutant HSP110 sensitizes colorectal cancer cells to chemotherapy and improves disease prognosis. Nat Med 2011;17:1283–1289. [DOI] [PubMed] [Google Scholar]
- 22.Shinbrot E, Henninger EE, Weinhold N et al. Exonuclease mutations in DNA polymerase epsilon reveal replication strand specific mutation patterns and human origins of replication. Genome Res 2014;24:1740–1750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nejadtaghi M, Jafari H, Farrokhi E et al. Familial colorectal cancer type X (FCCTX) and the correlation with various genes ‐ A systematic review. Curr Probl Cancer 2017;41:388–397. [DOI] [PubMed] [Google Scholar]
- 24.Espenschied CR, LaDuca H, Li S et al. Multigene panel testing provides a new perspective on Lynch syndrome. J Clin Oncol 2017;35:2568–2575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guastadisegni C, Colafranceschi M, Ottini L et al. Microsatellite instability as a marker of prognosis and response to therapy: A meta‐analysis of colorectal cancer survival data. Eur J Cancer 2010;46:2788–2798. [DOI] [PubMed] [Google Scholar]
- 26.Domingo E, Freeman‐Mills L, Rayner E et al. Somatic POLE proofreading domain mutation, immune response, and prognosis in colorectal cancer: A retrospective, pooled biomarker study. Lancet Gastroenterol Hepatol 2016;1:207–216. [DOI] [PubMed] [Google Scholar]
- 27.Schwitalle Y, Linnebacher M, Ripberger E et al. Immunogenic peptides generated by frameshift mutations in DNA mismatch repair‐deficient cancer cells. Cancer Immun 2004;4:14. [PubMed] [Google Scholar]