Abstract
People at risk for coronary heart disease are often at risk for nonalcoholic fatty liver disease (NAFLD). The association of modest wine consumption with NAFLD has not been studied and the recommendation of wine for patients at risk for both diseases is controversial. The aim is to test the hypothesis that modest wine consumption is associated with decreased prevalence of NAFLD. We included Third National Health and Nutrition Examination Survey participants who either reported no alcohol consumption or preferentially drinking wine with total alcohol consumption up to 10 g per day. Suspected NAFLD was based on unexplained serum alanine aminotransferase (ALT) elevation over the cut point of the reference laboratory (ALT > 43) and the cut point based on the 95th percentile of healthy subjects (ALT > 30 for men; ALT > 19 for women). Multivariate analysis was adjusted for age, gender, race, neighborhood, income, education, caffeine intake, and physical activity. A total of 7,211 nondrinkers and 945 modest wine drinkers comprised the study sample. Based on the reference laboratory cut point, suspected NAFLD was observed in 3.2% of nondrinkers and 0.4% of modest wine drinkers. The adjusted odds ratio was 0.15 (95% confidence interval, 0.05-0.49). Using the healthy subject cut point, suspected NAFLD was observed in 14.3% of nondrinkers and 8.6% of wine drinkers. The adjusted odds ratio was 0.51 (95% confidence interval, 0.33-0.79).
Conclusion:
Modest wine consumption is associated with reduced prevalence of suspected NAFLD. The current study supports the safety of one glass of wine per day for cardioprotection in patients at risk for both coronary heart disease and NAFLD. (HEPATOLOGY 2008;47:1947-1954.)
The beneficial effects of wine and wine-derived chemicals have received a great deal of recent attention. Moderate wine consumption has been associated with reduced risk of coronary heart disease (CHD).1 Therefore, some physicians recommend moderate wine consumption to patients with adverse cardiovascular risk factors.2 However, these are the same individuals at increased risk for developing nonalcoholic fatty liver disease (NAFLD).3,4 NAFLD is closely related to metabolic syndrome and therefore shares many risk factors with CHD. Although the threshold for alcoholic liver disease is thought to be ingestion of 20 g to 30 g of alcohol per day for men and 10 g to 30 g of alcohol per day for women,5,6 the threshold may be lower for overweight subjects who are at risk for both CHD and NAFLD.7 The threshold for patients with pre-existing liver disease is unclear. The usual recommendation for patients with NAFLD is to strictly avoid alcohol. Despite the potential for liver injury, there is indirect evidence to suggest that modest wine and alcohol may by protective against liver injury. In an animal model, the wine polyphenol, resveratrol, reduces steatosis in mice fed with a high calorie diet.8 Wine ameliorates many risk factors common to NAFLD and CHD such as insulin resistance and dyslipidemia.9-11 These studies raise the possibility that wine may actually be protective against NAFLD. In the absence of concrete evidence, any recommendation regarding wine consumption in people at risk for both CHD and NAFLD remains controversial. Therefore the current study aimed to test the hypothesis that modest wine consumption is associated with a decreased prevalence of NAFLD.
Patients and Methods
Study Sample.
Subjects were participants in the Third National Health and Nutrition Examination Survey (NHANES III). NHANES III was a cross-sectional study conducted in the United States from 1988 to 1994. The study used a stratified, multistage, probability cluster sampling design to obtain a representative sample of the civilian, noninstitutionalized population.12 This study was approved by the institutional review board of The University of California, San Diego, San Diego, CA.
We included participants age 21 years and older, ethnically white, black, or Mexican American. Although fatty liver is common in older teenagers,13 we chose a minimum age of 21 years because it is the legal drinking age in the United States. We included participants who either reported no alcohol consumption during the past month, or reported drinking wine, beer, or liquor up to one alcoholic beverage per day. We did not include higher level of alcohol consumption because higher levels of alcohol consumption have already been reported to be harmful in overweight and obese subjects.7
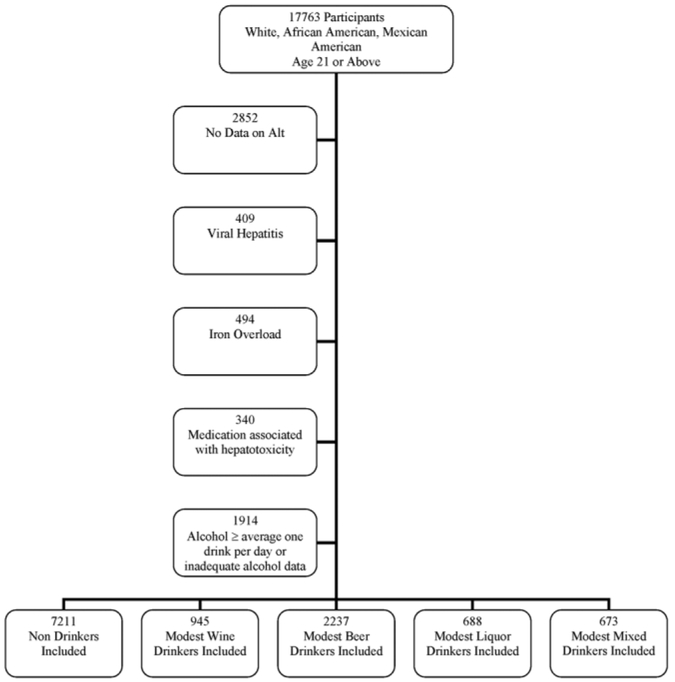
The primary disease under investigation was suspected NAFLD, which was defined using unexplained serum alanine aminotransferase (ALT) elevation. The use of ALT with exclusion of causes other than NAFLD is justified by several lines of reasoning. Among patients who are referred to a hepatologist and undergo a liver biopsy for unexplained ALT elevation, up to 90% will have a clinicopathological diagnosis of NAFLD.14-16 As opposed to a clinical hepatology referral hepatology population, in a population-based study such as NHANES, unexplained ALT elevations may be even more likely to be due to NAFLD.17 Therefore, we excluded subjects without data on ALT and those subjects with identifiable conditions other than NAFLD to account for ALT elevation. These conditions include excessive alcohol consumption (>1 alcoholic beverage per day), viral hepatitis (positive hepatitis B surface antigen, positive hepatitis C antibody), iron overload (transferitin saturation >50%), and use of medications associated with hepatotoxicity (androgens, antivirals medications, antifungals medications, nitrofuratoin, phenytoin, sulfonamides, trazadone, or tetracycline). After exclusion of these conditions, unexplained ALT elevation was strongly associated with adiposity and other features of the metabolic syndrome, and thus consistent with NAFLD.18 Moreover, those forms of chronic liver disease not tested are sufficiently uncommon that they were unlikely to contribute significantly to the cause of ALT elevation.19 (Refer to the inclusion and exclusion flowchart in Fig. 1.)
Fig. 1.
Inclusion and exclusion flow chart.
Suspected NAFLD.
Given the lack of a single uniform cut point for ALT elevation we chose to use both the higher cut point of the NHANES III reference laboratory (ALT > 43 U/L for both genders, referred to in this work as the laboratory cut point) and the widely used gender-specific lower cut point based on the 95th percentile of healthy subjects (ALT > 30 for men, ALT >19 for woman, referred to in this work as the healthy cut point).12,20 In addition to the bivariate cut points, the log-transformed ALT was also analyzed as a continuous variable.
Modest Wine, Beer, and Liquor Drinkers.
The primary exposure of interest was self-report of modest wine drinking versus no alcohol consumption. To distinguish wine from alcohol more generally, the association of modest beer and liquor drinking were also studied. The alcohol history was obtained by a trained interviewer in a private room to ensure confidentiality. A nondrinker was a person that reported no wine, beer, or liquor per month. A beverage preference (for example, wine, beer, or liquor) was defined as the type of alcohol whose consumption accounted for the majority of the total alcohol intake.21 Participants who did not have a preferred alcoholic beverage were defined as mixed drinkers. Modesty was defined as alcohol consumption up to an average of one drink per day. One drink is defined as 12 ounces of beer, 4 ounces of wine, or 1 ounce of liquor.7
Social, Demographic, and Lifestyle Confounders.
Social, demographic, and lifestyle confounders were considered to include age, gender, race, poverty income ratio, neighborhood population density, years of education, caffeine intake, and activity level.12 In NHANES III the prevalence of suspected NAFLD did not have a linear relationship with age. Therefore, age was adjusted using a categorical variable (21 to 40, 41 to 60, and >60 years). Neighborhood population density was based on U.S. Department of Agriculture Rural/Urban continuum codes. Participants were also asked if they considered themselves more active, less active, or the same as same-age sex peers. Participants were asked about their monthly consumption of caffeine-containing beverages. Total caffeine in-take was calculated as (regular coffee X 136 mg per cup + regular tea X 64 mg per cup + soda X 46 mg per can or bottle)/30.22 The caffeine intake was not normally distributed and therefore we used the loge-transformed caffeine intake for analysis.
Metabolic Syndrome Features.
NAFLD is closely associated with the metabolic syndrome23,24 and wine has been shown to ameliorate many metabolic syndrome features.9-11,25,26 Therefore, the following list of metabolic syndrome features were studied as we explored the potential protective mechanisms of modest wine drinking: body mass index (BMI) (weight [kg]/height [m]2), waist circumference, waist-to-hip ratio (WHR), systolic blood pressure (SBP), diastolic (DBP) blood pressure, diabetes, triglyceride level, high density lipoprotein (HDL) level, and insulin sensitivity. Participants were considered to have diabetes if they reported having a diagnosis of diabetes or if they had glycosylated hemoglobin (hemoglobin A1c) above 6.5%.27 Triglyceride level and insulin resistance were only considered only in subjects with >8 hours of fasting prior to phlebotomy. Triglyceride level was not normally distributed and therefore the loge-transformed triglyceride level was used for analysis. Insulin resistance was measured as quantitative insulin sensitivity check index (QUICKI = 1/[log(fasting insulin [lU/mL]) + log(fasting glucose [mg/dL])].28
Statistical Methods.
NHANES III was based on a complex, multistage, stratified, clustered probability sampling that could not be treated as a simple random sample. The SUDAAN 9.0 module (Research Triangle Institute, Research Triangle Park, NC) was used to incorporate primary sampling unit (sdppsu6), strata (sdpstra6), and weighting (wtpfhx6) into the analysis.12
Logistic regression was used to assess the association between reported nondrinkers, modest wine drinkers, modest beer drinkers, and modest liquor drinkers with suspected NAFLD. The outcome variable was suspected NAFLD based on each of the two ALT cut points. Our first multivariate model aimed to adjust for potential confounders and included the entire social, demographic, and lifestyle confounders listed above. Effect modification of modest wine drinking with age, gender, race, and BMI was tested using product variables (for example, modest wine drinking X age). Linear regression was used to estimate the association of modest wine drinking with loge(ALT) as a continuous variable.
Subjects identified with pre-existing conditions known to be aggravated by alcohol might be less likely to consume alcohol. Subjects may also stop drinking alcohol because of illness related to alcohol. To reduce potential selection bias, a separate analysis excluded nondrinkers who reported a lifetime cumulative intake of more than 12 alcoholic drinks, and excluded subjects who reported having a diagnosis of diabetes, gallbladder, liver, or pancreatic disease, and subjects counseled by their physician to cut down on alcohol due to hypertension.
To explore the potential protective mechanism of modest wine drinking, metabolic syndrome features were included in the second and third multivariate model. The mechanism of modest wine drinking was considered to be independent of metabolic syndrome features if the odds ratio (OR) of wine drinking remained unchanged after correction for these metabolic syndrome features.22 Our second multivariate model included all the variables in the first multivariate model with the addition of BMI, WHR, SBP, DBP, HDL, and diabetes status. Our third multivariate included all the variables in the second multivariate model with the addition of triglyceride level and QUICKI, which were only available in participants with >8 hours of fasting prior to phlebotomy (n = 2,840).
Results
Study Sample.
The study sample included 7,211 nondrinkers, 945 modest wine drinkers, 2,237 modest beer drinkers, 688 modest liquor drinkers, and 673 modest mixed drinkers. Refer to Fig. 1 for the inclusion and exclusion flowchart. Table 1 illustrates the social, demographic, and lifestyle characteristics of the four groups. As compared to nondrinkers, modest wine drinkers were younger, more likely to be non-Hispanic white and female, to reside in a rural area, to have more years of education and a higher income, and to consider themselves more active than their same age-matched and sex-matched peers.
Table 1.
Social, Demographic, and Behavioral Characteristics
| Non-Drinker (n = 7211) (%) |
Modest Wine Drinker (n = 945) (%) |
Modest Beer Drinker (n = 2237) (%) |
Modest Liquor Drinker (n = 688) (%) |
Modest Mixed Drinker (n = 673) (%) |
P value | |
|---|---|---|---|---|---|---|
| Age (years) | 50.1 (18.1) | 45.7(15.9) | 39.2 (14.1) | 46.5 (16.4) | 41.5 (14.5) | <0.001 |
| 21-40 | 2348 (42.5) | 410 (44.8) | 1270 (63.8) | 237 (38.3) | 332 (55.0) | <0.001 |
| 41-60 | 1805 (31.3) | 254 (34.5) | 548 (25.9) | 215 (39.3) | 207 (31.8) | |
| >61 | 3058 (31.8) | 281 (20.7) | 417 (10.3) | 236 (22.4) | 134 (13.3) | |
| Gender | <0.001 | |||||
| Male | 2553 (37.1) | 230 (23.8) | 1457 (65.9) | 309 (43.1) | 319 (49.4) | |
| Female | 4658 (62.9) | 715 (76.2) | 780 (34.1) | 379 (56.9) | 354 (50.6) | |
| Race | <0.001 | |||||
| White | 3172 (81.2) | 572 (90.6) | 792 (81.0) | 417 (90.5) | 332 (86.0) | |
| Black | 1997 (12.8) | 201 (6.6) | 655 (11.7) | 167 (7.2) | 221 (11.0) | |
| Hispanic | 2042 (6.0) | 172 (2.8) | 790 (7.4) | 102 (2.3) | 120 (2.99) | |
| Neighborhood Population Density | ||||||
| Rural | 3053 (39.0) | 561 (60.7) | 1139 (44.7) | 343 (47.4) | 407 (61.1) | <0.001 |
| Urban | 4158 (61.0) | 384 (39.3) | 1098 (55.3) | 345 (52.6) | 266 (38.9) | |
| Poverty Income Ratio | 2.73 (1.74) | 3.86 (1.94) | 3.28 (1.9) | 3.94(2.1) | 3.85 (1.82) | <0.001 |
| Education | 11.7 (3.3) | 13.6 (2.68) | 12.8 (3.0) | 13.1 (2.5) | 13.9 (2.58) | <0.001 |
| Activities compared to same age sex peers | <0.001 | |||||
| More Active | 2047 (29.7) | 349 (39.0) | 745 (36.7) | 274 (41.4) | 239 (39.5) | |
| Same | 3308 (47.0) | 395 (41.3) | 1027 (45.2) | 251 (35.0) | 287 (41.5) | |
| Less Active | 1702 (23.3) | 186 (19.7) | 421 (18.1) | 145 (23.7) | 138 (19.0) | |
| Caffeine Intake | 228 (310) | 220 (239) | 228 (246) | 221 (260) | 243 (313) | 0.90 |
Categorical variables shown as actual number (weighted frequency). Continuous variables shown as point estimate (standard deviation). Analysis of variance was used to compare continuous variables and chi square was used to compare categorical variables.
Prevalence of Suspected NAFLD in Nondrinkers and Modest Alcohol Drinkers.
Based on the laboratory cut point, suspected NAFLD was observed in 3.2% of nondrinkers, 0.4% of modest wine drinkers, 3.5% of modest beer drinkers, 2.3% of modest liquor drinkers, and 2.6% of modest mixed drinkers. Using the healthy cut point, suspected NAFLD was observed in 14.3% of nondrinkers, 8.6% of modest wine drinkers, 12.4% of modest beer drinkers, 14.9% of modest liquor drinkers, and 10.3% of modest mixed drinkers. Table 2 lists the prevalence and adjusted OR of suspected NAFLD in each drinking category. Compared to nondrinkers, only modest wine drinkers were consistently associated with a lower prevalence of suspected NAFLD across both cut points. After correction for all social, demographic, and behavioral characteristics (multivariate model 1) (Table 3), the adjusted OR for suspected NAFLD was 0.15 (95% confidence interval [CI], 0.05-0.49) based on the laboratory cut point and 0.51 (95% CI, 0.33-0.79) based on the healthy cut point. There were no significant interactions between wine drinking and age (P = 0.97), gender (P =0.99), race (P = 0.84), or BMI (P = 0.80). When treated as a continuous variable, loge(ALT) was significantly lower in modest wine drinkers than in nondrinkers (univariate analysis P = 0.006, multivariate analysis P = 0.001).
Table 2.
Prevalence of Suspected NAFLD in Nondrinkers and Various Types of Modest Drinkers
| ALT >43 |
ALT >30 for Men ALT > 19 for Women |
||||
|---|---|---|---|---|---|
| Drinking Categories | n | Prevalence (%) | OR (95% CI)* | Prevalence (%) | OR (95% CI)* |
| Non-Drinker | 7211 | 3.2 | 1.00 | 14.3 | 1.00 |
| Modest Wine Drinker | 945 | 0.4 | 0.16 (0.05-0.50) | 8.6 | 0.51 (0.33-0.80) |
| Modest Beer Drinker | 2237 | 3.5 | 0.66 (0.38-1.15) | 12.4 | 0.73 (0.52-1.01) |
| Modest Liquor Drinker | 688 | 2.3 | 0.74 (0.31-1.75) | 14.9 | 1.01 (0.66-1.56) |
| Modest Mixed Drinker | 673 | 2.6 | 0.68 (0.32-1.45) | 10.3 | 0.62 (0.41-0.92) |
Abbreviations: n, number of participants; OR, odds ratio; CI, confidence interval.
Multivariate odds adjusted for age, gender, race, income, education, neighborhood population density, caffeine consumption, and physical activity.
Table 3.
Crude and Adjusted Odds Ratios of Suspected NAFLD in Nondrinkers and Modest Wine Drinkers
| ALT >43 OR (95% CI) |
ALT >30 for Men ALT >19 for Women OR (95% CI) |
|
|---|---|---|
| Univariate | 0.13 (0.05-0.38) | 0.56 (0.38-0.84) |
| Multivariate model 1* | 0.15 (0.05-0.49) | 0.51 (0.33-0.79) |
| Multivariate model 2† | 0.20 (0.06-0.68) | 0.60 (0.39-0.92) |
| Multivariate model 3‡ | 0.22(0.06-0.85) | 0.63 (0.40-0.99) |
Abbreviations: OR, odds ratio; CI, confidence interval.
Multivariate Model 1 adjusted for age, gender, race, income, education, neighborhood population density, caffeine consumption, and physical activity.
Multivariate Model 2 adjusted for age, gender, race, income, education, neighborhood population density, caffeine consumption, physical activity, BMI, WHR, SBP, DBP, HDL, and diabetes.
Multivariate Model 3 adjusted for age, gender, race, income, education, neighborhood population density, caffeine consumption, physical activity, BMI, WHR, SBP, DBP, HDL, diabetes, TG, and QUICKI.
In a separate, more restrictive, analysis that excluded nondrinkers who reported more than 12 alcoholic drinks in their lifetime, and excluded subjects who were told by their doctors that they had diabetes, gallbladder, liver, or pancreatic conditions, or to decrease their alcohol consumption due to hypertension (non-drinkers n = 1,841, modest wine drinkers n = 903), the protective association of modest wine drinking remained unchanged (laboratory cut point adjusted OR 0.14, 95% CI, 0.03-0.73; healthy cut point adjusted OR 0.51, 95% CI, 0.30-0.85).
When the four groups of modest drinkers were pooled together, the prevalence of suspected NAFLD was 2.4% using the laboratory cut point and 11.6% using the healthy cut-point. After adjusting for social, demographic, and behavioral characteristics, the adjusted OR for suspected NAFLD was 0.59 (95% CI, 0.36-0.96) based on the laboratory cut point and 0.70 (95% CI, 0.53-0.93) based on the healthy cut point.
Metabolic Syndrome Features as Potential Protective Mechanisms.
Compared to nondrinkers, wine drinkers had lower BMI, WHR, SBP, DBP, triglyceride level, and higher insulin sensitivity. Wine drinkers also had a significantly lower prevalence of diabetes and metabolic syndrome. For men, but not for women, modest wine drinkers had a smaller waist circumference and higher HDL (Table 4). As reported,29 high BMI, WHR, waist circumference, triglyceride level, and DBP, and low HDL and QUICKI, as well as diabetes and metabolic syndrome, were significantly associated with suspected NAFLD. After controlling for the features of the metabolic syndrome in multivariate models 2 and 3, the adjusted OR of wine drinking remained significant and unchanged (Table 3).
Table 4.
Biological Features of Nondrinkers and Various Types of Modest Drinkers
| Non-Drinker | Modest Wine Drinkers |
Modest Beer Drinkers |
Modest Liquor Drinkers |
Modest Mixed Drinker |
P value | |
|---|---|---|---|---|---|---|
| BMI | 27.5 (6.2) | 26.1 (5.2) | 26.0 (4.8) | 27.1 (6.5) | 26.0 (4.8) | <0.001 |
| Waist | ||||||
| Men | 98.8 (14.1) | 96.8 (12.0) | 94.3 (12.4) | 98.7 (12.3) | 95.5 (9.9) | <0.001 |
| Women | 92.0 (15.5) | 86.2 (13.4) | 92.0 (13.5) | 86.2 (17.2) | 85.3 (13.3) | <0.001 |
| Waist-hip ratio | ||||||
| Men | 0.975 (0.073) | 0.955 (0.066) | 0.946 (0.072) | 0.974 (0.065) | 0.951 (0.0633) | <0.001 |
| Women | 0.882 (0.081) | 0.845 (0.078) | 0.846 (0.099) | 0.858 (0.086) | 0.839 (0.079) | <0.001 |
| SBP | 124.8 (19.2) | 119.7 (17.2) | 120.0 (15.4) | 122.4 (17.0) | 119.2 (15.3) | <0.001 |
| DBP | 74.0 (10.0) | 72.7 (8.8) | 74.4 (9.9) | 74.8 (9.7) | 73.8 (9.5) | 0.008 |
| TG | 149 (127) | 116 (82) | 126 (96) | 132 (123) | 125.0 (128) | <0.001 |
| HDL | ||||||
| Men | 41.2 (10.6) | 45.5 (12.6) | 43.8 (11.1) | 44.0 (11.4) | 47.5 (13.2) | <0.001 |
| Women | 52.8 (14.6) | 58.1 (14.2) | 55.9 (15.9) | 57.0 (18.4) | 57.3 (14.8) | <0.001 |
| QUICKI < 0.339 (%) | 2285/4160 (48.2) | 249/584 (33.0) | 606/1398 (36.0) | 207/427 (43.6) | 178/404 (41.0) | <0.001 |
| Diabetes (%) | 1169/7211 (11.5) | 50/945 (4.3) | 159/2237 (4.4) | 58/688 (7.4) | 39/671 (4.3) | <0.001 |
Categorical variables shown as actual number/denominator (weighted frequency). Continuous variables shown as point estimate (standard deviation). Analysis of variance was used to compare continuous variables and chi-square was used to compare categorical variables.
Prevalence of Suspected NAFLD in Modest Wine Drinkers Compared to Other Types of Modest Alcohol Drinkers.
Based on the laboratory cut point, both modest beer drinkers and modest liquor drinkers were significantly associated with a higher prevalence of suspected NAFLD compared to modest wine drinkers. After adjusting for all social, demographic, and behavioral characteristics, modest beer drinkers, modest liquor drinkers, and modest mixed drinkers compared to modest wine drinkers had 4.18 (95% CI, 1.42-12.33) times, 4.77 (95% CI, 1.34-16.90) times, and 4.32 (95% CI, 1.39-13.42) times the odds of having suspected NAFLD, respectively. Based on the healthy cut point, the association was only significant between modest liquor drinkers and modest wine drinkers (adjusted OR 1.96, 95% CI, 1.17-3.30).
Discussion
This cross-sectional population-based study showed that self-report of wine consumption up to one serving per day compared to no alcohol use is associated with a lower prevalence of suspected NAFLD. Both the higher NHANES laboratory reference cut point and the lower gender-specific cut point based on the 95th percentile of healthy subjects were used. After adjusting for social, demographic, and lifestyle differences, multivariate analysis showed that modest wine drinking was independently associated with lower prevalence of suspected NAFLD. Subjects identified with pre-existing conditions known to be aggravated by alcohol might be less likely to consume alcohol. Subjects may also stop drinking alcohol due to health problems from alcohol. Exclusion of these subjects did not affect the result. Although modest wine drinkers had fewer metabolic characteristics associated with NAFLD, these metabolic characteristics do not appear to explain the potential protective mechanism of modest wine drinking.
NAFLD is the most common liver disease in the United States. As many as one-third of adults in the United States may have fatty liver based on magnetic resonance screening30 and living donor liver biopsy.31 It has been demonstrated that at least 5% of adults with NAFLD will develop cirrhosis.32 The current study suggests that modest wine drinking is associated with a decreased prevalence of NAFLD. The cross-sectional design of the current study cannot address whether this observation is causative or associative, and whether wine is preventive or therapeutic. Nevertheless, this study presents a paradigm shift that modest wine consumption may not only be safe from a liver perspective but may actually decrease the prevalence of NAFLD. These data may be considered preliminary evidence supporting prospective studies of modest wine drinking in the prevention and treatment of NAFLD.
NAFLD shares many risk factors with CHD and therefore people at risk for NAFLD are likely to be at risk for CHD.3,33 From a cardiology and primary care perspective, the recommendation of alcohol use for cardioprotection in patients who are at risk for CHD, and thus at risk for NAFLD as well, is controversial because excessive alcohol intake may lead to liver injury. The current study suggests that modest wine consumption may be actually be protective against NAFLD and therefore supports the use of up to one glass of wine per day for cardioprotection in people at risk for both CHD and NAFLD.
Interaction with age and BMI must be considered in the study of alcohol’s association with liver injury. Ruhl and Everhart7 showed that overweight subjects have a lower threshold for alcohol-induced liver injury than normal weight subjects. Suzuki et al.34 showed that the influence of alcohol on liver injury varies with age. The current study specifically focused on wine, rather than all alcohol drinking, and at a lower level than the studies by Ruhl and Everhart7 or Suzuki et al.34 The protective association of modest wine drinking was not different across age and BMI. This finding is important because middle-age and overweight people may benefit the most from the cardioprotective effect and the potential liver protective effect of modest wine drinking.
Our study adds to the body of evidence that supports the liver protective role of modest wine drinking. The Copenhagen cohort35 of 30,630 individuals followed for 417,325 person-years demonstrated that participants who drank one to seven servings of wine per week had the lowest risk of developing cirrhosis. A prospective Japanese study34 following 326 male government workers for 5 years found that the incidence of hypertransaminasemia was lower in subjects who drank 140 g to 280 g of alcohol per week compared to subjects who drank none or up to one drink per week. Although subjects predisposed36 to alcoholic liver disease (for example, previous history of hypertransaminasemia or alcoholic liver disease) were excluded from the study, the finding did suggest a potential protective role of alcohol. In a small study of morbidly obese subjects undergoing bariatric surgery,37 any alcohol consumption compared to no alcohol was associated with decreased prevalence of nonalcoholic steatohepatitis, the histologically severe form of NAFLD. However, these patients are not representative of the general population and alcohol amount, frequency, and type were not clearly de-lineated.
Both alcohol and nonalcohol components of wine may each be responsible for the lower prevalence of some of the metabolic syndrome features in the current study. Alcohol consumption has been reported to prevent diabetes and improve insulin resistance.9,38,39 Nonalcohol components of wine have been shown to lower serum triglyceride levels.40,41 Both alcohol42,43 and nonalcohol wine components40,41 have been shown to increase serum HDL levels. Wine, but not other alcohol, has been shown to be inversely associated with central obesity.44 Our study confirms that modest wine drinking is associated with a decreased prevalence of these metabolic syndrome features.
Despite the inverse association between modest wine drinking and metabolic syndrome features, the improvement in metabolic profile does not account for the liver protective association of modest wine drinking. In multivariate analysis, correcting for all of those biological variables did not attenuate the OR, suggesting that the potential protective mechanism of modest wine drinking is independent of these biological features.22 Furthermore, both modest beer and liquor consumption were also associated with improvement in metabolic syndrome features but they were not associated with lower prevalence of suspected NAFLD. Although modest alcohol drinkers pooled together as a group were associated with lower prevalence of suspected NAFLD, it was modest wine drinkers that accounted for the association. We postulate that the protective property of modest wine drinking, if present, is mediated through its nonalcohol rather than its alcohol components. The exact protective mechanisms cannot readily be measured in the NHANES III database.
The current study has a number of important methodological characteristics that allow accurate assessment of the association between modest wine drinking and suspected NAFLD. Notably, the use of NHANES makes the findings generalizable to the U.S. population. As such, the use of unexplained ALT elevation as a surrogate of NAFLD is imperfect but appropriate for a large population-based study. Without excluding alternative diagnosis, in the Dallas Heart Study30 over half of participants with abnormal ALT had fatty liver based on magnetic resonance spectroscopy while nearly three-quarters of participants with normal ALT did not have fatty liver. After excluding alternative causes of elevated ALT and/or fatty liver, population-based studies in Israel,45 Japan,46 and Taiwan47 showed that the positive predictive value (60%-90%) and negative predictive value (70%-90%) of ALT were even stronger. Importantly, the current study made use of strict exclusion criteria to exclude excessive alcohol, viral hepatitis, iron overload, and hepatotoxic medications. When similar exclusions were applied in case series of unexplained ALT elevation, liver biopsy revealed a positive predictive value of 65%-90%.14-16 Together, these data support the contention that the majority of participants in the current study were accurately classified as having or not having suspected NAFLD. Because the specific positive and negative predictive values will vary depending on the ALT cut point being used, two well-recognized cut points were used in this study. While the possibility remains that the protective effect of wine applies to other forms of liver injury, it is most likely attributable to effects on fatty liver. Although imperfect, any misclassification bias based on the ALT should affect wine drinkers and nondrinkers equally and therefore would only falsely diminish the size of the association observed. The use of standardized methodology with trained interviewers was important for the accuracy of the data but cannot fully eliminate errors in the self-reported amount and type of alcohol consumed. To adjust for confounders, social, demographic, and lifestyle covariates were entered into the multivariate analysis and the protective association of modest wine drinking remained unchanged. This minimized, although did not completely exclude, the possibility that self-report of modest wine drinking was a surrogate marker of another unmeasured lifestyle factor. Finally, the cross-sectional design cannot demonstrate the safety of modest wine consumption in subjects with pre-existing NAFLD. The preventive and therapeutic potential of modest wine drinking should be further explored by prospective cohort and experimental studies.
The current study presents a paradigm shift that modest wine consumption may not only be safe from a liver perspective but may actually decrease the prevalence of NAFLD. This can be considered initial evidence to support future prospective studies with either wine or nonalcohol wine components for prevention or treatment of NAFLD. In patients at risk for both CHD and NAFLD, up to one serving of wine per day for cardioprotection is likely safe from a liver perspective, although wine consumption for cardioprotection remains an important decision at the discretion of the individual and the physician. Finally, we must emphasize that the current study does not support the use of wine or alcohol at more than one serving per day. The adverse consequence of excessive alcohol intake has been well established from previous studies.5 Patients at risk for developing alcohol abuse should not be advised to consume wine.
Acknowledgment:
We thank Dr. Vijay Shah (Mayo Clinic) for reviewing the manuscript.
Supported in part by the National Institutes of Health (NIH) National Research Service Award (NRSA) grant T32 DK07202, and by the NIH National Center for Research Resources grant M01 RR000827 for the General Clinical Research Center at the University of California, San Diego (UCSD).
Abbreviations:
- ALT
serum alanine aminotransferase
- BMI
body mass index
- CHD
coronary heart disease
- CI
confidence interval
- DBP
diastolic blood pressure
- HDL
high-density lipoprotein
- NAFLD
nonalcoholic fatty liver disease
- NHANES III
Third National Health and Nutrition Examination Survey
- OR
odds ratio
- SBP
systolic blood pressure
- WHR
waist-to-hip ratio
- QUICKI
quantitative insulin sensitivity check index
Footnotes
Potential conflict of interest: The funders did not participate in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. The authors have no financial conflicts of interest.
References
- 1.Rimm EB, Klatsky A, Grobbee D, Stampfer MJ. Review of moderate alcohol consumption and reduced risk of coronary heart disease: is the effect due to beer, wine, or spirits. BMJ 1996;312:731–736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goldberg IJ, Mosca L, Piano MR, Fisher EA. AHA science advisory: wine and your heart: a science advisory for healthcare professionals from the Nutrition Committee, Council on Epidemiology and Prevention, and Council on Cardiovascular Nursing of the American Heart Association. Circulation 2001;103:472–475. [DOI] [PubMed] [Google Scholar]
- 3.Ioannou GN, Weiss NS, Boyko EJ, Mozaffarian D, Lee SP. Elevated serum alanine aminotransferase activity and calculated risk of coronary heart disease in the United States. HEPATOLOGY 2006;43:1145–1151. [DOI] [PubMed] [Google Scholar]
- 4.Ruhl CE, Everhart JE. Determinants of the association of overweight with elevated serum alanine aminotransferase activity in the United States. Gastroenterology 2003;124:71–79. [DOI] [PubMed] [Google Scholar]
- 5.Thun MJ, Peto R, Lopez AD, Monaco JH, Henley SJ, Heath CW Jr, et al. Alcohol consumption and mortality among middle-aged and elderly U.S. adults. N Engl J Med 1997;337:1705–1714. [DOI] [PubMed] [Google Scholar]
- 6.Becker U, Deis A, Sorensen TI, Gronbaek M, Borch-Johnsen K, Muller CF, et al. Prediction of risk of liver disease by alcohol intake, sex, and age: a prospective population study. HEPATOLOGY 1996;23:1025–1029. [DOI] [PubMed] [Google Scholar]
- 7.Ruhl CE, Everhart JE. Joint effects of body weight and alcohol on elevated serum alanine aminotransferase in the United States population. Clin Gastroenterol Hepatol 2005;3:1260–1268. [DOI] [PubMed] [Google Scholar]
- 8.Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006;444:337–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Howard AA, Arnsten JH, Gourevitch MN. Effect of alcohol consumption on diabetes mellitus: a systematic review. Ann Intern Med 2004;140:211–219. [DOI] [PubMed] [Google Scholar]
- 10.Davies MJ, Baer DJ, Judd JT, Brown ED, Campbell WS, Taylor PR. Effects of moderate alcohol intake on fasting insulin and glucose concentrations and insulin sensitivity in postmenopausal women: a randomized controlled trial. JAMA 2002;287:2559–2562. [DOI] [PubMed] [Google Scholar]
- 11.Schulze MB, Hoffmann K, Manson JE, Willett WC, Meigs JB, Weikert C, et al. Dietary pattern, inflammation, and incidence of type 2 diabetes in women. Am J Clin Nutr 2005;82:675–684; quiz 714-675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Plan and Operation of the Third National Health and Nutrition Examination Survey, 1988-94. Hyattsville, MD: National Center for Health Statistics; 1994. [PubMed] [Google Scholar]
- 13.Schwimmer JB, Deutsch R, Kahen T, Lavine JE, Stanley C, Behling C. Prevalence of fatty liver in children and adolescents. Pediatrics 2006;118: 1388–1393. [DOI] [PubMed] [Google Scholar]
- 14.Daniel S, Ben-Menachem T, Vasudevan G, Ma CK, Blumenkehl M. Prospective evaluation of unexplained chronic liver transaminase abnormalities in asymptomatic and symptomatic patients. Am J Gastroenterol 1999; 94:3010–3014. [DOI] [PubMed] [Google Scholar]
- 15.Skelly MM, James PD, Ryder SD. Findings on liver biopsy to investigate abnormal liver function tests in the absence of diagnostic serology. J Hepatol 2001;35:195–199. [DOI] [PubMed] [Google Scholar]
- 16.Ratziu V, Giral P, Charlotte F, Bruckert E, Thibault V, Theodorou I, et al. Liver fibrosis in overweight patients. Gastroenterology 2000;118:1117–1123. [DOI] [PubMed] [Google Scholar]
- 17.Clark JM, Diehl AM. Defining nonalcoholic fatty liver disease: implications for epidemiologic studies. Gastroenterology 2003;124:248–250. [DOI] [PubMed] [Google Scholar]
- 18.Clark JM, Brancati FL, Diehl AM. The prevalence and etiology of elevated aminotransferase levels in the United States. Am J Gastroenterol 2003;98: 960–967. [DOI] [PubMed] [Google Scholar]
- 19.Yu AS, Keeffe EB. Elevated AST or ALT to nonalcoholic fatty liver disease: accurate predictor of disease prevalence? Am J Gastroenterol 2003;98:955–956. [DOI] [PubMed] [Google Scholar]
- 20.Prati D, Taioli E, Zanella A, Della Torre E, Butelli S, Del Vecchio E, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med 2002;137:1–10. [DOI] [PubMed] [Google Scholar]
- 21.Gronbaek M, Jensen MK, Johansen D, Sorensen TI, Becker U. Intake of beer, wine and spirits and risk of heavy drinking and alcoholic cirrhosis. Biol Res 2004;37:195–200. [DOI] [PubMed] [Google Scholar]
- 22.Ruhl CE, Everhart JE. Coffee and caffeine consumption reduce the risk of elevated serum alanine aminotransferase activity in the United States. Gastroenterology 2005;128:24–32. [DOI] [PubMed] [Google Scholar]
- 23.Hamaguchi M, Kojima T, Takeda N, Nakagawa T, Taniguchi H, Fujii K, et al. The metabolic syndrome as a predictor of nonalcoholic fatty liver disease. Ann Intern Med 2005;143:722–728. [DOI] [PubMed] [Google Scholar]
- 24.Neuschwander-Tetri BA. Nonalcoholic steatohepatitis and the metabolic syndrome. Am J Med Sci 2005;330:326–335. [DOI] [PubMed] [Google Scholar]
- 25.Estruch R, Sacanella E, Badia E, Antunez E, Nicolas JM, Fernandez-Sola J, et al. Different effects of red wine and gin consumption on inflammatory biomarkers of atherosclerosis: a prospective randomized crossover trial. Effects of wine on inflammatory markers. Atherosclerosis 2004;175:117–123. [DOI] [PubMed] [Google Scholar]
- 26.Frankel EN, Kanner J, German JB, Parks E, Kinsella JE. Inhibition of oxidation of human low-density lipoprotein by phenolic substances in red wine. Lancet 1993;341:454–457. [DOI] [PubMed] [Google Scholar]
- 27.Woerle HJ, Pimenta WP, Meyer C, Gosmanov NR, Szoke E, Szombathy T, et al. Diagnostic and therapeutic implications of relationships between fasting, 2-hour postchallenge plasma glucose and hemoglobin a1c values. Arch Intern Med 2004;164:1627–1632. [DOI] [PubMed] [Google Scholar]
- 28.Hrebicek J, Janout V, Malincikova J, Horakova D, Cizek L. Detection of insulin resistance by simple quantitative insulin sensitivity check index QUICKI for epidemiological assessment and prevention. J Clin Endocrinol Metab 2002;87:144–147. [DOI] [PubMed] [Google Scholar]
- 29.Freiberg MS, Cabral HJ, Heeren TC, Vasan RS, Curtis Ellison R. Alcohol consumption and the prevalence of the Metabolic Syndrome in the US.: a cross-sectional analysis of data from the Third National Health and Nutrition Examination Survey. Diabetes Care 2004;27:2954–2959. [DOI] [PubMed] [Google Scholar]
- 30.Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. HEPATOLOGY 2004;40:1387–1395. [DOI] [PubMed] [Google Scholar]
- 31.Ryan CK, Johnson LA, Germin BI, Marcos A. One hundred consecutive hepatic biopsies in the workup of living donors for right lobe liver transplantation. Liver Transpl 2002;8:1114–1122. [DOI] [PubMed] [Google Scholar]
- 32.Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A, et al. The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology 2005;129:113–121. [DOI] [PubMed] [Google Scholar]
- 33.Ekstedt M, Franzen LE, Mathiesen UL, Thorelius L, Holmqvist M, Bodemar G, et al. Long-term follow-up of patients with NAFLD and elevated liver enzymes. HEPATOLOGY 2006;44:865–873. [DOI] [PubMed] [Google Scholar]
- 34.Suzuki A, Angulo P, St Sauver J, Muto A, Okada T, Lindor K. Light to moderate alcohol consumption is associated with lower frequency of hypertransaminasemia. Am J Gastroenterol 2007;102:1912–1919. [DOI] [PubMed] [Google Scholar]
- 35.Becker U, Gronbaek M, Johansen D, Sorensen TI. Lower risk for alcohol-induced cirrhosis in wine drinkers. HEPATOLOGY 2002;35:868–875. [DOI] [PubMed] [Google Scholar]
- 36.Lieber CS. Alcoholic fatty liver: its pathogenesis and mechanism of progression to inflammation and fibrosis. Alcohol 2004;34:9–19. [DOI] [PubMed] [Google Scholar]
- 37.Dixon JB, Bhathal PS, O’Brien PE. Nonalcoholic fatty liver disease: predictors of nonalcoholic steatohepatitis and liver fibrosis in the severely obese. Gastroenterology 2001;121:91–100. [DOI] [PubMed] [Google Scholar]
- 38.Wannamethee SG, Camargo CA Jr, Manson JE, Willett WC, Rimm EB. Alcohol drinking patterns and risk of type 2 diabetes mellitus among younger women. Arch Intern Med 2003;163:1329–1336. [DOI] [PubMed] [Google Scholar]
- 39.Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG, et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med 2001;345:790–797. [DOI] [PubMed] [Google Scholar]
- 40.Castilla P, Echarri R, Davalos A, Cerrato F, Ortega H, Teruel JL, et al. Concentrated red grape juice exerts antioxidant, hypolipidemic, and anti-inflammatory effects in both hemodialysis patients and healthy subjects. Am J Clin Nutr 2006;84:252–262. [DOI] [PubMed] [Google Scholar]
- 41.Zern TL, Wood RJ, Greene C, West KL, Liu Y, Aggarwal D, et al. Grape polyphenols exert a cardioprotective effect in pre- and postmenopausal women by lowering plasma lipids and reducing oxidative stress. J Nutr 2005;135:1911–1917. [DOI] [PubMed] [Google Scholar]
- 42.Kabagambe EK, Baylin A, Ruiz-Narvaez E, Rimm EB, Campos H. Alcohol intake, drinking patterns, and risk of nonfatal acute myocardial infarction in Costa Rica. Am J Clin Nutr 2005;82:1336–1345. [DOI] [PubMed] [Google Scholar]
- 43.Linn S, Carroll M, Johnson C, Fulwood R, Kalsbeek W, Briefel R. High-density lipoprotein cholesterol and alcohol consumption in US white and black adults: data from NHANES II. Am J Public Health 1993;83:811–816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Halkjaer J, Tjonneland A, Thomsen BL, Overvad K, Sorensen TI. Intake of macronutrients as predictors of 5-y changes in waist circumference. Am J Clin Nutr 2006;84:789–797. [DOI] [PubMed] [Google Scholar]
- 45.Zelber-Sagi S, Nitzan-Kaluski D, Halpern Z, Oren R. Prevalence of primary non-alcoholic fatty liver disease in a population-based study and its association with biochemical and anthropometric measures. Liver Int 2006;26:856–863. [DOI] [PubMed] [Google Scholar]
- 46.Jimba S, Nakagami T, Takahashi M, Wakamatsu T, Hirota Y, Iwamoto Y, et al. Prevalence of non-alcoholic fatty liver disease and its association with impaired glucose metabolism in Japanese adults. Diabet Med 2005;22: 1141–1145. [DOI] [PubMed] [Google Scholar]
- 47.Chen CH, Huang MH, Yang JC, Nien CK, Yang CC, Yeh YH, et al. Prevalence and risk factors of nonalcoholic fatty liver disease in an adult population of Taiwan: metabolic significance of nonalcoholic fatty liver disease in nonobese adults. J Clin Gastroenterol 2006;40: 745–752. [DOI] [PubMed] [Google Scholar]