Abstract
Objectives
To examine the patterns of fat mass gain in pregnancy and fat loss in the early postpartum period relative to women’s pre-pregnancy body mass index (BMI) and by adherence to Institute of Medicine’s gestational weight gain (GWG) recommendations.
Design
Prospective cohort study with three to four study visits.
Setting
This study is a part of the prospective longitudinal birth cohort, ‘The Alberta Pregnancy Outcomes and Nutrition Study’ (APrON) that recruited pregnant women from the cities of Edmonton and Calgary in Alberta.
Participants
1820 pregnant women were recruited and followed through their pregnancy and at 3 months postpartum.
Outcome measures
Body weight and skinfold thicknesses were measured during pregnancy and early postpartum in women. Body density was calculated from sum of skinfold thickness (biceps, triceps, subscapula and suprailiac), and total fat mass accretion during pregnancy was calculated using Van Raaij’s equations and at postpartum using Siri’s equation. Differences in total fat mass gain, fat mass loss and fat retention according to pre-pregnancy BMI categories and GWG categories were tested using two-way analysis of variance and post hoc comparisons.
Results
Most women (64%) had a normal pre-pregnancy BMI, and overall 49% women exceeded the GWG recommendations. Obese women gained significantly less total fat mass, had lower fat mass loss and had lower postpartum fat retention than normal-weight women (p<0.05). Women with excessive GWG gained higher total fat mass and had higher postpartum fat mass retention (p<0.03) than women who met the GWG recommendations. Total GWG was positively correlated with total fat gain (r=0.61, p<0.01) and total fat retention (r=0.31, p<0.05).
Conclusion
Excessive GWG is the significant risk factor for higher fat mass accretion during pregnancy and higher postpartum fat retention, irrespective of pre-pregnancy BMI.
Keywords: pre-pregnancy BMI, gestational weight gain, body composition, skinfold thickness, fat mass
Strengths and limitations of this study.
The prospective nature of this study allowed for longitudinal measurement and analysis of maternal body fat during pregnancy and early postpartum in relation to the recent Institute of Medicine’s gestational weight gain recommendations.
Anthropometric measurements were used to assess fat mass accretion and distribution in this study; trained research staff performed all the data collection to minimise measurement errors.
We used self-reported highest weight to calculate gestational weight gain; however, a comparison of this measure to the highest weight measured during prenatal clinic visits showed no significant differences.
Owing to the small sample size of underweight women in this study, most analyses excluded these women.
Introduction
The prevalence rates of overweight and obesity among women of childbearing years have increased dramatically in the past four decades,1 and recent reports of high gestational weight gain (GWG) accompanied with women not returning to their pre-pregnancy weight may further exacerbate this problem.2 3 The Institute of Medicine’s (IOM) recommendations for GWG and results from InterGrowth 214 have focused attention on appropriate GWG for optimal pregnancy outcomes3 5 and reducing the risks for chronic diseases in maternal and offspring’s later life.6 While body weight is an important indicator of pregnancy outcomes,3 5 maternal fat mass is a stronger predictor of long-term maternal health.7
Normal physiological adaptations to pregnancy require accumulation of subcutaneous fat to meet fetal energy demands for growth and development,8 increased maternal energy demands9 and to support lactation.10 However, changes in adiposity during pregnancy and postpartum have not been well-documented. Most studies are limited to small sample sizes in cross-sectional studies conducted either in pregnancy or in postpartum, but not both.11–13 This study determined the patterns of fat mass changes in pregnancy and early postpartum in a group of women with low-risk pregnancies who were participating in a prospective cohort study in Alberta, Canada. Fat gain, loss and retention were examined relative to women’s pre-pregnancy body mass index (BMI) and by adherence to IOM’s GWG recommendations3 to better understand how these factors contribute to changes in fat mass during and after pregnancy.
Methods
Study design and population
Pregnant women <27 weeks’ gestation, >16 years of age and able to read and write in English were enrolled in a prospective longitudinal cohort, the Alberta Pregnancy Outcomes and Nutrition (APrON) study (n=1820), between 2009 and 2012.14 Detailed descriptions of participants and the study are published elsewhere.14 15 Data were collected at two to three study visits during pregnancy, spaced to coincide with each trimester and one follow-up visit at approximately 3 months postpartum.
Procedures
Women completed questionnaires detailing their sociodemographic information and medical history prior to and during pregnancy. At each study visit, weight and height were measured with light clothing to the nearest 0.01 kg (Healthometer Professional 752 KL, Pelstar LLC, Illinois, USA) and 0.1 cm (Charder HM200P Portstad Portable Stadiometer, USA), respectively, by trained staff.
Pre-pregnancy weight and the highest weight during pregnancy were self-reported. Women were classified as underweight (<18.5), normal (18.5–24.9), overweight (<25.0–29) or obese (≥30) according to their pre-pregnancy BMI.16 GWG was calculated as the difference between pre-pregnancy body weight and the highest weight during pregnancy. Women with different pre-pregnancy BMI were categorised based on whether they were ‘Below’, ‘Met’ or ‘Exceeded’ IOM’s BMI-specific GWG recommendations.3
Waist circumference was measured to the nearest 0.1 cm using a non-elastic tape placed according to standard physical landmarks17 in women whose first study visit occurred at ≤16 weeks’ gestation and all who attended the postpartum visit.18 Subcutaneous fat was measured as skinfold thickness (SFT) using callipers at each study visit at the biceps, triceps, subscapular, suprailiac and mid-thigh sites (Lange skinfold callipers, Beta Technologies, Inc., Cambridge, Maryland, USA); all measures were taken in triplicate on the participant’s right side, and the mean value was used.17 The same research assistant completed SFT measurements on 68% of participants. Inter-rater reliability, expressed as the coefficient of variation between study personnel (CV%), was determined by repeating the SF measurements on every 50th participant and was between 5.1% and 2.8% for all sites. Acceptable CV% for intra-rater error of 12% have been reported by other investigators.19 20
The sum of biceps, triceps, subscapula and suprailiac SF was used as a proxy for body density21 and was used to calculate total fat mass using a pregnancy-specific equation22 in pregnancy and an equation for non-pregnant women at postpartum.23 The fat mass measured at each clinic visit was used to calculate outcome measures as follows: the rate of fat mass gain in early pregnancy (second trimester–first trimester/number of intervening weeks), late pregnancy (third trimester–second trimester/number of intervening weeks), total fat mass gained in pregnancy (third trimester–first trimester), fat mass loss (postpartum–third trimester) and fat retention at postpartum (postpartum visit–first trimester). Fat mass distribution during pregnancy and at postpartum was studied by examining changes in waist circumference and in SFT at each site measured. The effects of maternal breastfeeding practices (exclusive breast feeding (breast fed only for ≥3 months), mixed feeding (fed breast milk at breast or expressed breastmilk and formula) or exclusive formula feeding (only formula fed for ≥3 months)) on postpartum fat retention were also examined.
Statistical analysis
Differences in SFT at different anatomical sites according to maternal pre-pregnancy BMI were tested using one-way analysis of variance (ANOVA) followed by Tukey’s test for multiple comparisons. Differences in total fat mass gained during pregnancy, fat mass loss between third trimester to postpartum and fat retention according to pre-pregnancy BMI categories and GWG categories were examined using two-way ANOVA and post hoc comparisons. A multilinear regression analysis was performed to test the association between GWG as a continuous variable and total fat gain, after adjusting for pre-pregnancy BMI. The effects of maternal pre-pregnancy BMI on fat mass accretion during pregnancy were analysed using a multivariate mixed model adjusted for maternal age, parity, marital status, ethnicity, family income, GWG categories and time of study visit (trimesters 1, 2, 3). A separate linear regression model was used to determine the association between GWG as a continuous variable and total fat mass accretion. A multivariable linear regression analysis was completed to determine the association between breastfeeding practices and fat retention at the time of the postpartum visit. Data were adjusted for pre-pregnancy BMI and GWG, and the interaction between breastfeeding and pre-pregnancy BMI was tested.
Data were analysed using R statistical package (V.3.2.4, R Foundation for Statistical Computing, Vienna, Austria). A p value of <0.05 was considered statistically significant. Results are presented as mean±SD unless otherwise stated.
Patient and public involvement
No study participants or public were involved in setting the research question or the outcome measures nor were they involved in developing plans for recruitment, design or implementation of the study. Participants were not involved in any aspects of data analysis, interpretation of results or manuscript preparation. There are no plans currently to disseminate the results of the research to study participants or the relevant patient community.
Results
A total of 2212 women were recruited in the APrON study and 1820 were included in these analyses. Women with missing pre-pregnancy BMI (n=266), preterm deliveries at <37 weeks (n=140) and those who gave birth to twins (n=24) were excluded. Four hundred and thirty-five women were enrolled in their first trimester and 1385 were enrolled in their second trimester. Baseline characteristics of women excluded from this study were not different from those of women included in further analyses (online supplementary table 1). Most women (64.1%) had a normal pre-pregnancy BMI and 49.3% exceeded the GWG recommendations.3 Participants were predominantly Caucasian (80.7%), married/common-law (95.7%), university graduates (68.3%) and had high family incomes (55.1% ≥$70 000/year). At the postpartum visit (~3 months after delivery), very few women exclusively formula-fed their infants (n=14), while 48% exclusively breast fed their infants and 51% women used mixed feeding practices (table 1). No interaction effect was observed between pre-pregnancy BMI and GWG, with respect to fat mass accretion during pregnancy, fat loss or fat retention. Underweight women were excluded from most analyses due to small sample size.
Table 1.
Anthropometric and sociodemographic characteristics of women enrolled in the Alberta Pregnancy Outcomes and Nutrition Study (APrON) by pre-pregnancy BMI categories
| Characteristic | n* | Underweight | Normal | Overweight | Obese | P value |
| Pre-pregnancy weight (kg)† | 1820 | 49.17±5.09‡ | 59.72±6.48§ | 74.51 ± 6.77¶ | 93.78±13.46** | <0.001 |
| Height (cm)† | 1820 | 167.03±7.32 | 165.40±6.36 | 165.81±6.05 | 165.01±7.41 | 0.10 |
| Pre-pregnancy BMI (kg/m2)† | 1820 | 17.59±0.92‡ | 21.80±1.66§ | 27.06 ± 1.42¶ | 34.39±4.17** | <0.001 |
| Pre-pregnancy BMI (kg/m2) (%) | 1820 | 3.8 | 64.1 | 21.5 | 10.6 | |
| Body weight (kg)† | ||||||
| Trimester 1 | 486 | 51.20±4.31‡ | 61.79±7.31§ | 76.10 ± 7.37¶ | 94.58±13.27** | <0.001 |
| Trimester 2 | 1718 | 57.56±12.58‡ | 64.68±7.44§ | 79.07 ± 7.83¶ | 96.68±13.96** | <0.001 |
| Trimester 3 | 1590 | 62.32±7.04‡ | 72.19±8.11§ | 86.81 ± 8.33¶ | 103.26±13.90** | <0.001 |
| Postpartum | 1477 | 55.06±6.98‡ | 64.23±8.03§ | 79.37 ± 8.86¶ | 95.90±14.04** | <0.001 |
| Gestational weight gain (%) as per 2009 IOM GWG guidelines | ||||||
| Below | 272 | 17.2 | 22.0 | 6.1 | 14.3 | <0.001 |
| Met | 508 | 55.2 | 37.6 | 23.2 | 16.1 | |
| Exceeded | 761 | 27.6 | 40.4 | 70.7 | 69.6 | |
| Age (years)† | ||||||
| 17 to 30 | 785 | 26.48±3.35 | 27.34±2.73 | 27.23±2.79 | 26.89±2.91 | 0.17 |
| 31 to 45 | 980 | 33.36±2.61 | 34.23±2.78 | 34.65±2.96 | 34.06±2.59 | 0.07 |
| Parity (%) | ||||||
| 0 | 959 | 56.3 | 57.5 | 47.2 | 48.9 | <0.01 |
| 1 | 631 | 31.3 | 34.1 | 39.3 | 40.3 | |
| 2 + | 175 | 12.4 | 8.4 | 13.5 | 10.8 | |
| Marital status (%) | ||||||
| Married | 1691 | 89.2 | 96.1 | 95.8 | 95.2 | 0.09 |
| Unmarried | 76 | 10.8 | 3.9 | 4.2 | 4.8 | |
| Ethnicity (%) | ||||||
| Caucasian | 1424 | 68.8 | 79.9 | 82.5 | 86.6 | 0.01 |
| Other | 340 | 31.2 | 20.1 | 17.5 | 13.4 | |
| Family income as per Statistics Canada 2011 (%) | ||||||
| Low (≤$69 999) | 393 | 34.9 | 21.0 | 22.9 | 26.3 | 0.001 |
| Medium ($70 000–$99 999) | 392 | 17.5 | 20.9 | 23.7 | 30.6 | |
| High (≥$100 000) | 965 | 47.6 | 58.1 | 53.4 | 43.1 | |
| Maternal education (%) | ||||||
| Less than high school diploma | 50 | 9.5 | 2.6 | 2.7 | 2.7 | <0.001 |
| High school/diploma/certificate | 504 | 25.4 | 24.7 | 34.1 | 44.3 | |
| University degree/postgraduate degree | 1198 | 65.1 | 72.7 | 63.2 | 53.0 | |
| Gestational age (weeks)† | ||||||
| Trimester 1 | 494 | 11.85±2.47 | 11.17±2.54 | 10.73±2.38 | 10.66±2.39 | 0.14 |
| Trimester 2 | 1768 | 19.42±3.53 | 18.99±3.38 | 18.97±3.23 | 18.57±3.22 | 0.27 |
| Trimester 3 | 1749 | 32.32±1.10 | 32.41±1.30 | 32.35±1.33 | 32.44±1.15 | 0.77 |
| Delivery | 1775 | 39.43±1.22‡ | 39.69±1.12‡ | 39.65±1.19‡ | 39.37±1.20‡ | <0.01 |
| Postpartum age (weeks)† | 1642 | 12.79±2.09 | 12.80±2.25 | 12.80±2.41 | 12.63±1.72 | 0.84 |
| Breast feeding (0–3 months) (%)†† | ||||||
| Exclusive breast feeding | 730 | 55.2 | 52.3 | 42.4 | 33.1 | 0.07 |
| Mixed feeding | 768 | 44.8 | 46.8 | 57.0 | 65.0 | |
| Exclusive formula feeding | 14 | 0 | 0.9 | 0.6 | 1.9 | |
*n=1820 (underweight=69, normal=1166, overweight=391, obese=194); sample sizes within a particular characteristic may not total n=1820 due to missing responses.
†Values are reported as mean±SD.
‡,§,¶,**Values with different superscripts are different from each other within a row; data were analysed using one-way ANOVA and post hoc estimation by Tukey’s multiple comparisons test.
††Data were analysed using chi-squared test.
bmjopen-2018-026908supp001.pdf (188KB, pdf)
Fat mass distribution by pre-pregnancy BMI categories
The average SFT measurements for biceps, triceps, subscapular, suprailiac and thigh sites by BMI categories for all study visits are presented in online supplementary table 2.
Rate of gain in SFT during pregnancy and early postpartum
The triceps, biceps and thigh SFT increased in early pregnancy similarly across all BMI categories. In contrast, subscapula and suprailiac SFT increased more slowly in overweight and obese women than in those with a normal BMI (p<0.01) (table 2). The suprailiac SFT continued to increase at a slower rate in late pregnancy in overweight and obese women compared with women with normal BMI. The triceps and thigh SFT also increased at a slower rate in late pregnancy in obese compared with women with a normal BMI (p=0.02) (table 2).
Table 2.
Rate of gain in skinfold thickness during pregnancy according to pre-pregnancy BMI categories
| Skinfold site |
Pre-pregnancy BMI |
Early pregnancy Trimester 2–Trimester 1 (mm/week) |
Late pregnancy Trimester 3–Trimester 2 (mm/week) |
||||
| n | Mean±SD | P value | n | Mean±SD | P value | ||
| Triceps* | Underweight | NA | NS | 51 | 0.06±0.30†‡ | 0.009 | |
| Normal | 234 | 0.12±0.42 | 988 | 0.08±0.37† | |||
| Overweight | 81 | 0.18±0.64 | 326 | 0.06±0.41† ‡ | |||
| Obese | 53 | 0.10±0.84 | 164 | −0.03±0.47‡ | |||
| Subscapula* | Underweight | NA | 0.001 | 51 | 0.17±0.24 | NS | |
| Normal | 217 | 0.18±0.36† | 970 | 0.20±0.36 | |||
| Overweight | 84 | 0.04±0.60†‡ | 328 | 0.18±0.45 | |||
| Obese | 50 | −0.09±0.59‡ | 158 | 0.16±0.47 | |||
| Suprailiac* | Underweight | NA | <0.01 | 52 | 0.26±0.46† ‡ | 0.02 | |
| Normal | 233 | 0.33±0.65† | 978 | 0.24±0.57† | |||
| Overweight | 79 | 0.04±0.88‡ | 323 | 0.14±0.58 ‡ | |||
| Obese | 51 | −0.24±0.88‡ | 160 | 0.11±0.60 ‡ | |||
| Thigh* | Underweight | NA | NS | 53 | 0.25±0.52† ‡ | 0.02 | |
| Normal | 228 | 0.15±0.64 | 967 | 0.27±0.55† | |||
| Overweight | 77 | 0.05±1.05 | 314 | 0.28±0.63† | |||
| Obese | 47 | 0.14±0.75 | 154 | 0.12±0.59 ‡ | |||
*One-way ANOVA and post hoc estimation by Tukey’s multiple comparisons test.
†‡Values with different symbols within a column are significantly different from each other, the p values for significance are presented in adjacent the columns.
ANOVA, analysis of variance; BMI, body mass index; NA, not applicable, due to small sample size (n=6); NS, statistically not significant.
Changes in SFT during pregnancy and early postpartum
The total gains in triceps, biceps and thigh SFT during pregnancy were similar among women in different BMI categories. Gains in subscapular and suprailiac SFT were lower in obese than normal weight women (p<0.01) while suprailiac SFT gain was lower (p<0.01), but subscapular gain was similar in overweight women compared with normal weight women (table 3).
Table 3.
Changes in skinfold thickness during pregnancy and early postpartum by pre-pregnancy BMI categories
| Skinfold site |
Pre-pregnancy BMI |
Total gain Trimester 3–Trimester 1 (mm) |
Loss Postpartum–Trimester 3 (mm) |
Retention Postpartum–Trimester 1 (mm) |
||||||
| n | Mean±SD | P value* | n | Mean±SD (n) |
P value* | n | Mean±SD | P value* | ||
| Biceps | Underweight | NA | NS | 50 | −1.02±3.36 | NS | NA | NS | ||
| Normal | 229 | 0.81±2.83 | 924 | −0.83±3.63 | 224 | 0.60±3.35 | ||||
| Overweight | 66 | −0.22±4.67 | 285 | −0.34±5.33 | 60 | 0.03±5.87 | ||||
| Obese | 49 | −0.54±7.73 | 150 | 0.03±6.06 | 53 | −1.18±8.29 | ||||
| Triceps | Underweight | NA | NS | 48 | −0.85±4.53 | NS | NA | NS | ||
| Normal | 242 | 1.62±4.39 | 936 | −0.38±4.73 | 236 | 1.96±4.66 | ||||
| Overweight | 77 | 1.65±5.61 | 297 | −0.98±5.65 | 72 | 1.76±6.26 | ||||
| Obese | 51 | 1.21±7.42 | 152 | −0.14±6.59 | 54 | 0.48±8.11 | ||||
| Subscapula | Underweight | NA | 0.002 | 48 | −1.84±3.77†§ | 0.01 | NA | NS | ||
| Normal | 226 | 3.82±3.58† | 920 | −1.21±4.44† | 221 | 3.14±4.74 | ||||
| Overweight | 80 | 3.26±5.54† ‡ | 298 | −0.21±5.53 ‡§ | 74 | 2.73±6.59 | ||||
| Obese | 47 | 1.26±5.93‡ | 148 | 0.70±5.84 ‡ | 19 | 1.47±7.08 | ||||
| Suprailiac | Underweight | NA | <0.01 | 49 | −6.74±5.54† | <0.01 | NA | 0.04 | ||
| Normal | 236 | 5.48±6.16† | 926 | −4.23±6.99† | 235 | 1.46±6.77† | ||||
| Overweight | 74 | 2.14±6.91 ‡ | 294 | −2.59±7.18 ‡ | 70 | 0.88±7.15† ‡ | ||||
| Obese | 47 | −0.01±10.38 ‡ | 148 | 0.01 ± 8.60§ | 52 | −1.47±9.36 ‡ | ||||
| Thigh | Underweight | NA | NS | 50 | −1.35±6.60 | NS | NA | NS | ||
| Normal | 237 | 4.49±6.50 | 922 | −2.17±6.83 | 231 | 3.03±6.92 | ||||
| Overweight | 75 | 2.78±9.15 | 287 | −2.48±8.29 | 70 | 0.52±9.79 | ||||
| Obese | 49 | 2.44±7.58 | 143 | −1.64±7.50 | 53 | 1.48±7.98 | ||||
*One-way ANOVA and post hoc estimation by Tukey’s multiple comparisons test.
ANOVA, analysis of variance; BMI, body mass index; NA, not applicable, due to small sample size (n=7); NS, statistically not significant.
The SFT loss between third trimester and postpartum was similar at the biceps, triceps and thigh skinfold sites for women in all BMI groups; however, compared with normal weight women, women in the overweight and obese BMI classes lost lower amounts of SFT at the subscapula and suprailiac sites (p=0.01) (table 3).
SFT retention (ie, the difference between first trimester and postpartum) was similar at the biceps, triceps, subscapula and thigh sites for all women, but higher among normal weight than obese women at the suprailiac site (p=0.04) (table 3).
Waist circumference
Waist circumferences varied between all pre-pregnancy BMI groups at ≤16 weeks’ gestation and postpartum (p<0.01) (data not shown). Although changes between the first measurement at ≤16 weeks’ gestation and postpartum were similar among all women (normal: mean increase=1.98±4.84 cm, n=238; overweight: mean increase=3.02±4.97 cm, n=75; obese: mean increase=1.70±7.03 cm, n=54, p>0.05), normal and overweight women, respectively, had significant increases in their waist circumference between early pregnancy and postpartum (p<0.001).
Changes in fat mass during pregnancy
Women in the overweight and obese BMI groups had higher fat mass than normal or underweight women (p<0.001) at all study visits (online supplementary table 3). Those who exceeded GWG recommendations had higher fat mass than women who met or gained below GWG recommendations (p<0.05) at all study visits during pregnancy and women who met GWG recommendations had higher fat mass than those who gained below recommendations at trimesters 2 and 3 (p<0.05) (online supplementary table 3). Among the obese women, the rate of fat mass accretion in early (p=0.001) and late pregnancy (p=0.001) was significantly slower compared with normal weight women (table 4). In comparison with women who exceeded GWG recommendations, women who gained below GWG recommendations also had a lower rate of fat mass accretion during late pregnancy (p=0.001) (table 4). A comparison of rate of fat gain in late pregnancy versus early pregnancy showed that all women irrespective of BMI and GWG categories gained higher rates of fat mass accretion in late pregnancy than early pregnancy (mean±SD=0.072±0.09 kg/week, p=0.0002, n=1714).
Table 4.
Rate of gain in fat mass during pregnancy according to pre-pregnancy BMI and GWG categories
| Characteristic | Early pregnancy Trimester 2–Trimester 1 (kg/week) |
Late pregnancy Trimester 3–Trimester 2 (kg/week) |
||
| Mean±SD (n) |
P value | Mean±SD (n) | P value | |
| Pre-pregnancy BMI | ||||
| Underweight | NA | 0.01 | 0.19±0.16* (49) |
<0.001 |
| Normal | 0.18±0.19* (194) |
0.22±0.18* (923) |
||
| Overweight | 0.13±0.28* (59) |
0.23±0.22* (294) |
||
| Obese | 0.03±0.29† (100) |
0.15±0.24† (144) |
||
| Gestational weight gain | ||||
| Below | 0.06±0.26* (45) |
0.001 | 0.12±0.15* (217) |
<0.001 |
| Met | 0.11±0.26*† (71) |
0.18±0.18† (418) |
||
| Exceeded | 0.20±0.23† (120) |
0.27 ± 0.21‡ (628) |
||
*,†,‡Values with different superscripts are different from each other within a column. Data were analysed using one-way ANOVA.
ANOVA, analysis of variance; BMI, body mass index; GWG, gestational weight gain; NA, not applicable, due to small sample size.
Total fat mass gain during pregnancy was similar between normal and overweight women (table 5), while obese women gained less fat mass than either of these other two groups (p=0.01) (table 5).
Table 5.
Changes in fat mass during pregnancy and early postpartum by pre-pregnancy BMI and GWG categories
| Total gain Trimester 3–Trimester 1 (kg) |
Loss Postpartum–Trimester 3 (kg) |
Retention Postpartum–Trimester 1 (kg) |
||||||||
| n | Mean±SD | P value | n | Mean±SD | P value | n | Mean±SD | P value | ||
| Pre-pregnancy* BMI |
Underweight | NA | 0.01 | 46 | −2.49±2.60† | 0.003 | NA | <0.05 | ||
| Normal | 199 | 4.18±2.36† | 878 | −2.35±2.59† | 199 | 2.22±2.94† | ||||
| Overweight | 56 | 3.93±2.99† | 268 | −2.05±3.38† | 54 | 2.57±3.92† | ||||
| Obese | 42 | 2.20±3.87‡ | 135 | −0.87±3.79‡ | 48 | 0.79±4.53‡ | ||||
| GWG categories* |
Below | 50 | 2.48±1.94† | <0.001 | 44 | 1.02±2.42† | 0.002 | 44 | 1.01±2.42† | 0.028 |
| Met | 80 | 2.77±2.59† | 71 | 1.50±3.42‡ | 77 | 1.51±3.42† | ||||
| Exceeded | 128 | 5.05±2.64‡ | 120 | 3.07±3.35‡ | 134 | 3.07±3.35‡ | ||||
| Interactions * Pre-pregnancy BMI and GWG |
NS | 0.55 | 0.74 | |||||||
*Two-way ANOVA and post hoc estimation by Tukey’s multiple comparisons test.
†,‡Values with different superscripts are different from each other within a column.
ANOVA, analysis of variance; BMI, body mass index; GWG, gestational weight gain; NA, not applicable, due to small sample size (n=6); NS, statistically not significant.
Women with excessive GWG gained more total fat mass during pregnancy than either women who gained below or met the GWG recommendations (p<0.001) (table 5). Total GWG was positively associated with total fat mass gained in pregnancy in all groups (normal: r=0.63, p<0.001; overweight: r=0.50, p<0.001; obese: r=0.50, p<0.001).
Results from the multiple linear regression analysis indicated that a 1 kg increase in GWG was associated with an average of 0.54 kg increase in total fat gain, after adjusting for pre-pregnancy BMI categories (figure 1). There was no significant difference in the relationship between total fat gain and GWG among different BMI categories.
Figure 1.
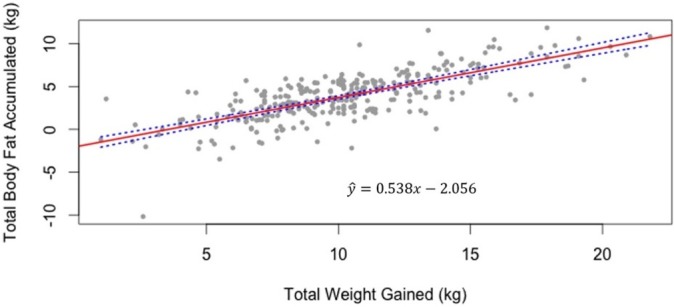
Association between total body fat accumulated and total weight gained during pregnancy (n=297 including women from the normal, overweight and obese body mass index categories). *Note: Underweight women have been excluded due to small sample size (n=6).
The multivariate analysis demonstrated that time of study visit, pre-pregnancy BMI and GWG were significantly associated with fat mass accretion during pregnancy. Irrespective of BMI and GWG categories, all women showed an incremental gain in fat mass at each subsequent trimester visit compared with the previous trimester (second trimester: mean difference=1.08 kg, 95% CI=0.81 to 1.35, p<0.001; third trimester: mean difference=3.89 kg, 95% CI=3.62 to 4.16, p<0.001). Higher pre-pregnancy BMI was associated with greater fat mass. When compared with normal weight women, overweight and obese women had higher fat mass (overweight: mean difference over pregnancy=7.56 kg, 95% CI=6.93 to 8.19, p<0.001; obese: mean difference=17.44 kg, 95% CI=16.64 to 18.24, p<0.001) and underweight women had lower fat mass (mean difference=−5.53 kg, 95% CI=−6.90 to −4.16, p<0.001). Women with GWG above recommendations gained significantly more fat mass than women who met (met: mean difference=−2.02 kg, 95% CI=−2.57 to −1.47, p<0.001) or gained below recommendations (below: mean difference=−4.00 kg, 95% CI=−4.69 to −3.31, p<0.001).
Fat loss between pregnancy and early postpartum
Fat mass loss between third trimester and postpartum was similar between women in the underweight, normal and overweight categories (table 5), while obese women lost significantly less fat mass than those in the other BMI groups (p=0.003). Fat mass loss between the third trimester and postpartum was similar among women who met or exceeded the GWG recommendations; however, women who gained below the GWG recommendations lost significantly less fat mass (p=0.002) than women who exceeded the recommendations.
Fat retention at early postpartum
Results from the two-way ANOVA indicated that fat mass retention at postpartum (postpartum–first trimester) was similar between women in the normal and overweight BMI categories (table 5). However, in comparison with women in the normal and overweight BMI categories, obese women retained significantly less fat mass at postpartum (p<0.05), adjusting for GWG guideline adherence.
When pre-pregnancy BMI category was adjusted, fat mass retention at postpartum was not significantly different among women who gained below or met the GWG recommendations; however, women who exceeded the guidelines retained significantly more fat mass than the latter groups (p=0.03) (table 5). There was a positive correlation (normal: r=0.31, p<0.001; overweight: r=0.31, p=0.02; obese: r=0.49, p<0.001) between total GWG and total fat retention at postpartum.
Since women in this cohort had similar breastfeeding practices irrespective of their pre-pregnancy BMI, we hypothesised that the effects of breastfeeding on fat retention would not differ among these groups. As anticipated, there were no significant differences in fat retention by breastfeeding practices at an average of 3-month postpartum (β=0.01, 95% CI=–0.12 to 0.23, p=0.40). No interactions were observed between breastfeeding and pre-pregnancy BMI (online supplementary table 4).
Discussion
This prospective cohort study of pregnant women showed that maternal fat mass distribution and accretion during pregnancy are influenced by maternal pre-pregnancy BMI. Also, exceeding GWG was associated with higher fat mass accretion during pregnancy and greater fat mass retention at postpartum compared with meeting the GWG recommendations.
Comparison with other studies
In our study, we found that women with an obese BMI had different patterns of fat mass distribution, accretion and retention than women from other BMI categories. In particular, obese women had lower rates of fat mass accretion during early and late pregnancy and an overall smaller total fat mass gain during pregnancy. They also had lower rates of SFT gain at the subscapula and suprailiac sites in early pregnancy than normal women. These observations concur with those of a previous study, in which women with higher pre-pregnancy BMI experienced slower gains in SFT during early pregnancy.24 Our results further reveal that, in comparison with normal-weight women, obese women had slower rates of gain in SFT at the triceps, suprailiac and thigh sites during late pregnancy, as well as smaller total increases in subscapula and suprailiac SFT during pregnancy.
Mechanisms underlying these differences in fat accretion among women starting pregnancy with different BMIs are unclear but could have to do with differences in insulin sensitivity as women enter pregnancy. In early pregnancy, women normally experience increased insulin sensitivity, which facilitates greater lipogenesis and fat storage in preparation for maternal and fetal energy demands. Those starting pregnancy with larger fat stores and lower insulin sensitivity may experience a smaller increment in fat stores at this particular time.25 Similar trends were observed in Argentinian women, where participants with a normal pre-pregnancy BMI gained significantly more at the biceps, triceps and subscapula skinfold sites than overweight and obese women between 16 and 36 weeks of pregnancy.26 Interestingly, overweight and obese women in our study had different trajectories for SFT, while overweight and normal women had similar rates of increase at all skinfold sites except at the suprailiac. Further, our results demonstrate that irrespective of BMI or GWG categories all women showed an increased gain in fat mass in the second and third trimesters, gaining significantly higher amount of fat compared with their trimester 1 fat mass. Future studies examining changes in metabolic and lifestyle changes during late pregnancy can shed light on this important aspect of weight gain. Interventions to help women make healthier choices in diet and lifestyle could also mediate reducing fat accretion in this period.
With respect to fat mass retention at postpartum, obese women in this study retained less fat mass than women from other BMI categories. The mean fat mass loss between third trimester and postpartum was also lowest for obese women. Differences in fat retention were not affected by variation in breastfeeding practices among women from the different pre-pregnancy BMI groups. Previous studies suggest that women with higher pre-pregnancy BMI tend to lose peripheral fat more easily than central fat postpartum.8 Our results also revealed that the overweight and obese women had smaller reductions in SFT from the suprailiac and subscapula sites at postpartum compared with normal weight women. Additionally, the average waist circumference increased at postpartum for overweight and normal weight women. Further, it should be noted that in comparison with normal-weight women, women with an overweight or obesity pre-pregnancy BMI had higher total fat mass and SFT (at all body sites) during pregnancy and postpartum. Thus, the combined effects of existing higher fat mass, coupled with greater fat mass accretion in the truncal regions and decreased mobilisation of abdominal body fat stores, as seen more especially in overweight women, could exacerbate the central adiposity present prior to pregnancy. Moreover, the physiological milieu of pregnancy, coupled with lifestyle behaviours promoting weight gain, may predispose normal-weight women to gain more fat mass25 in the truncal and abdominal regions, as observed in the present study.
This study also illustrated that GWG was positively associated with total fat mass accretion, irrespective of pre-pregnancy BMI categories. Women who exceeded GWG recommendations had greater fat mass accretion during pregnancy than women who either met or gained below GWG recommendations. Similar results were observed in two other studies27 28 where a positive correlation was observed between GWG and change in fat mass (r=0.87),27 (r=0.76).28 Our findings of a positive correlation between total GWG and postpartum fat retention (normal: r=0.31; overweight: r=0.31; obese: r=0.49) support the findings from other groups who measured fat retention at 27 weeks’ postpartum (r=0.59),28 suggesting that this pattern of fat retention may remain for some time.
Previous research has revealed that maternal biological and sociodemographic factors such as age, parity, ethnicity and income status contribute to the variability in gestational weight gain29 30; however, sparse evidence is available regarding the influence of these factors on fat mass accretion during pregnancy. In a growing multicultural population with diverse lifestyle behaviours and increasing prevalence of chronic diseases, studying the influences of sociodemographic factors on the variability and composition of weight gain is important. Future studies should examine these variables in addition to pre-pregnancy BMI and GWG.
Strengths and limitations of this study
The strengths of this study include that it sheds light on body fat changes in present day women from a developed country and highlights its association with reference to the recent GWG recommendations. In this study, anthropometric measurements were used to assess fat mass accretion and distribution. This technique is safe in pregnancy, relatively inexpensive and easy to apply in longitudinal studies, and thus provides an important estimate of body fatness in a large group of pregnant women. Nevertheless, these techniques are subject to measurement errors that must be considered but were minimised by having data collected by trained research staff. Intraobserver error was within acceptable limits. We used self-reported highest weight to calculate GWG; however, a comparison of this measure to the highest weight measured at the prenatal visits extracted from chart reviews showed no significant difference between the two measurements for women included in our analysis (median difference in clinical measured and self-reported weight is 0.46 kg, IQR=2.56, n=1054). This difference in weight was negligible as the highest body weight measured at prenatal clinic visit was 79.82 kg.
Conclusion and implications
In conclusion, women with higher GWG are likely to have higher fat mass accretion during pregnancy and higher postpartum fat retention, irrespective of pre-pregnancy BMI. Furthermore, women with an overweight pre-pregnancy BMI gained similar amounts of fat mass as normal-weight women, which could be an important factor in raising their risk of continued overweight or obesity. Overweight women also lose fat at a slower rate in the subscapula and suprailiac sites and have larger waist circumference at postpartum, further exacerbating the existing central adiposity. Although obese women gained less fat mass during pregnancy, they lost smaller amounts at postpartum than did normal or overweight women, which may be indicative of a modified metabolic milieu or different behavioural approaches to limit their pregnancy-related weight gain to achieve recommendations. Lastly, normal-weight women in our study retained higher fat mass in the suprailiac and waist regions at postpartum than at the beginning of their pregnancy, which may make them susceptible to increased central adiposity. Further examination of the effects of sociodemographic factors and lifestyle changes, such as diet and exercise, on body composition during pregnancy would aid in identifying plausible causes for variations in fat mass accretion and retention according to pre-pregnancy BMI.
Supplementary Material
Acknowledgments
We extend our sincere thanks to all members of the Alberta Pregnancy Outcomes and Nutrition study team and the study participants. The authors acknowledge Sarah Loehr, Lubna Anis, Dayna-Lynn Dymianiw, Roycent Tumbare, Ala Qabaja, Ye Shen and student volunteers who contributed to data collection, database development, data entry and cleaning.
Footnotes
Contributors: FS conducted the research and drafted the manuscript; LS conducted the data cleaning and statistical analysis under the supervision of YY and LK; RCB and LJMC are collaborators on the APrON Study and designed the protocol for data collection. YY, RCB and LK obtained funding for analysis of data presented in this study. All authors critically revised the manuscript and provided important intellectual content, approved the final version submitted for publication and agreed to act as guarantors of the work.
Funding: This research is made possible because of the generous supporters of Lois Hole Hospital for Women through the Women and Children’s Health Research Institute and funding from Alberta Innovates as a Collaborative Research and Innovation Opportunity grant.
Competing interests: None declared.
Ethics approval: Ethics approval for this study was obtained from the Health Research Ethics Boards at the University of Alberta (Pro 00002954) and the University of Calgary (E22101).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: No additional data available.
Collaborators: The APrON Study Team : Nicole Letourneau; Bonnie J Kaplan; Catherine J Field; Deborah Dewey; Rhonda C Bell; Francois Bernier; Marja Cantell; Linda Casey; Misha Eliasziw; Anna Farmer; Lisa Gagnon; Gerry Giesbrecht; Laki Goonewardene; Libbe Kooistra; Donna Mance; Jon Martin; Linda McCargar; Maeve O’Brien; Victor Pop; Nalini Singhal; Brenda Leung. ENRICH Team: Rhonda C Bell; Paula Robson; Maria Mayan; Terri Miller; Ellen Toth; Richard Oster; Kim Raine; Dolly Bondarianzadeh; IIona Csizmadi; Venu Jain; Kara Narenberg; Carla Prado; Arya Sharma; Sheile Tyminski; Yan Yuan; Kristi Adamo; Elizabeth Adams; Mary Barker; Michelle Berlin; Sally Bowman; Hilde Brekke; Wemdy Lawrence; Christine Olson; Sara Wilson-Wolfe; Sue Woodbury.
Patient consent for publication: Not required.
Contributor Information
The APrON Study Team and ENRICH:
Nicole Letourneau, Bonnie J Kaplan, Catherine J Field, Deborah Dewey, Rhonda C Bell, Francois Bernier, Marja Cantell, Linda Casey, Misha Eliasziw, Anna Farmer, Lisa Gagnon, Gerry Giesbrecht, Laki Goonewardene, Libbe Kooistra, Donna Mance, Jon Martin, Linda Mccargar, Maeve O’brien, Victor Pop, Nalini Singhal, Brenda Leung, Rhonda Bell, Paula Robson, Maria Mayan, Terri Miller, Ellen Toth, Richard Oster, Kim Raine, Dolly Bondarianzadeh, Iiona Csizmadi, Venu Jain, Kara Narenberg, Carla Prado, Arya Sharma, Sheile Tyminski, Yan Yuan, Kristi Adamo, Elizabeth Adams, Mary Barker, Michelle Berlin, Sally Bowman, Hilde Brekke, Wemdy Lawrence, Christine Olson, Sara Wilson-Wolfe, and Sue Woodbury
Collaborators: The APrON Study Team and ENRICH
References
- 1. NCD Risk Factor Collaboration (NCD-RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19·2 million participants. Lancet 2016;387:1377–96. 10.1016/S0140-6736(16)30054-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Jarman M, Yuan Y, Pakseresht M, et al. . Patterns and trajectories of gestational weight gain: a prospective cohort study. CMAJ Open 2016. 4:E338–45. 10.9778/cmajo.20150132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Institute of Medicine. Weight Gain During Pregnancy: Reexamining the Guidelines. 2009. http://www.nap.edu/openbook.php?record_id=12584 (Retrieved on 13 Apr 2018). [PubMed]
- 4. Cheikh Ismail L, Bishop DC, Pang R, et al. . Gestational weight gain standards based on women enrolled in the Fetal Growth Longitudinal Study of the INTERGROWTH-21st Project: a prospective longitudinal cohort study. BMJ 2016;352:i555 10.1136/bmj.i555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Health Canada. Prenatal Nutrition Guidelines for Health Professionals-Gestational Weight Gain. 2010. http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/pubs/guide-prenatal-eng.pdf.
- 6. Viswanathan M, Siega-Riz AM, Moos MK, et al. . Outcomes of maternal weight gain. Evid Rep Technol Assess 2008;168:1–223. [PMC free article] [PubMed] [Google Scholar]
- 7. Leddy MA, Power ML, Schulkin J. The impact of maternal obesity on maternal and fetal health. Rev Obstet Gynecol 2008;1:170–8. Fall. [PMC free article] [PubMed] [Google Scholar]
- 8. Sidebottom AC, Brown JE, Jacobs DR. Pregnancy-related changes in body fat. Eur J Obstet Gynecol Reprod Biol 2001;94:216–23. 10.1016/S0301-2115(00)00329-8 [DOI] [PubMed] [Google Scholar]
- 9. Bobrow KL, Quigley MA, Green J, et al. . Persistent effects of women’s parity and breastfeeding patterns on their body mass index: results from the Million Women Study. Int J Obes 2013;37:712–7. 10.1038/ijo.2012.76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Maple-Brown LJ, Roman NM, Thomas A, et al. . Perinatal factors relating to changes in maternal body fat in late gestation. J Perinatol 2013;33:934–8. 10.1038/jp.2013.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Jean N, Somers VK, Sochor O, et al. . Normal-weight obesity: implications for cardiovascular health. Curr Atheroscler Rep 2014;16:464 10.1007/s11883-014-0464-7 [DOI] [PubMed] [Google Scholar]
- 12. Larciprete G, Valensise H, Vasapollo B, et al. . Body composition during normal pregnancy: reference ranges. Acta Diabetol 2003;40(Suppl 1):s225–32. 10.1007/s00592-003-0072-4 [DOI] [PubMed] [Google Scholar]
- 13. Sommer C, Jenum AK, Waage CW, et al. . Ethnic differences in BMI, subcutaneous fat, and serum leptin levels during and after pregnancy and risk of gestational diabetes. Eur J Endocrinol 2015;172:649–56. 10.1530/EJE-15-0060 [DOI] [PubMed] [Google Scholar]
- 14. Kaplan BJ, Giesbrecht GF, Leung BM, et al. . The Alberta Pregnancy Outcomes and Nutrition (APrON) cohort study: rationale and methods. Matern Child Nutr 2014;10:44–60. 10.1111/j.1740-8709.2012.00433.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Begum F, Colman I, McCargar LJ, et al. . Gestational weight gain and early postpartum weight retention in a prospective cohort of Alberta women. J Obstet Gynaecol Can 2012;34:637–47. 10.1016/S1701-2163(16)35316-6 [DOI] [PubMed] [Google Scholar]
- 16. Health Canada. Canadian Guidelines for Body Weight Classification in Adults. 2003. http://www.hc-sc.gc.ca/fn-an/nutrition/weights-poids/guide-ld-adult/weight_book_tc-livres_des_poids_tm-eng.php (Retrieved on 13 Apr 2018). [DOI] [PMC free article] [PubMed]
- 17. Lohman TG, Roche AF, R M. Anthropometric standardization reference manual Champaign. Ill: Human Kinetics Books, 1988:1–177. [Google Scholar]
- 18. Lean ME, Han TS, Seidell JC. Impairment of health and quality of life in people with large waist circumference. Lancet 1998;351:853–6. 10.1016/S0140-6736(97)10004-6 [DOI] [PubMed] [Google Scholar]
- 19. Klipstein-Grobusch K, Georg T, Boeing H. Interviewer variability in anthropometric measurements and estimates of body composition. Int J Epidemiol 1997;26(Suppl 1):174S–80. 10.1093/ije/26.suppl_1.S174 [DOI] [PubMed] [Google Scholar]
- 20. Fuller NJ, Jebb SA, Goldberg GR, et al. . Inter-observer variability in the measurement of body composition. Eur J Clin Nutr 1991;45:43–9. [PubMed] [Google Scholar]
- 21. Durnin JV, Womersley J. Body fat assessed from total body density and its estimation from skinfold thickness: measurements on 481 men and women aged from 16 to 72 years. Br J Nutr 1974;32:77–97. 10.1079/BJN19740060 [DOI] [PubMed] [Google Scholar]
- 22. van Raaij JM, Peek ME, Vermaat-Miedema SH, et al. . New equations for estimating body fat mass in pregnancy from body density or total body water. Am J Clin Nutr 1988;48:24–9. 10.1093/ajcn/48.1.24 [DOI] [PubMed] [Google Scholar]
- 23. Siri WE. The gross composition of the body. Adv Biol Med Phys 1956;4:239–80. [DOI] [PubMed] [Google Scholar]
- 24. Widen EM, Gallagher D. Body composition changes in pregnancy: measurement, predictors and outcomes. Eur J Clin Nutr 2014;68:643–52. 10.1038/ejcn.2014.40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lain KY, Catalano PM. Metabolic changes in pregnancy. Clin Obstet Gynecol 2007;50:938–48. 10.1097/GRF.0b013e31815a5494 [DOI] [PubMed] [Google Scholar]
- 26. López LB, Calvo EB, Poy MS, et al. . Changes in skinfolds and mid-upper arm circumference during pregnancy in Argentine women. Matern Child Nutr 2011;7:253–62. 10.1111/j.1740-8709.2009.00237.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Berggren EK, Groh-Wargo S, Presley L, et al. . Maternal fat, but not lean, mass is increased among overweight/obese women with excess gestational weight gain. Am J Obstet Gynecol 2016;214:745.e1–5. 10.1016/j.ajog.2015.12.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Butte NF, Ellis KJ, Wong WW, et al. . Composition of gestational weight gain impacts maternal fat retention and infant birth weight. Am J Obstet Gynecol 2003;189:1423–32. 10.1067/S0002-9378(03)00596-9 [DOI] [PubMed] [Google Scholar]
- 29. Lowell H, Miller DC. Weight gain during pregnancy: adherence to Health Canada’s guidelines. Health Rep 2010;21:31–6. [PubMed] [Google Scholar]
- 30. Pike IL. Age, reproductive history, seasonality, and maternal body composition during pregnancy for nomadic Turkana of Kenya. Am J Hum Biol 1999;11:658–72. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2018-026908supp001.pdf (188KB, pdf)


