Abstract
Background
Despite the availability of several biomarkers, the diagnosis of periprosthetic joint infection (PJI) continues to be challenging. Serum D-dimer assessment is a widely available test that detects fibrinolytic activities and has been reported as an inflammatory biomarker. However, quite a few articles have reported the diagnostic efficiency of D-dimer for PJI.
Methods
This prospective study enrolled patients who had undergone total joint arthroplasty, were suspected of PJI, and also prepared for revision arthroplasty. PJI was defined using the Musculoskeletal Infection Society criteria. In all patients, serum D-dimer level, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) level were measured preoperatively. We then compared the diagnostic efficiency of these three biomarkers.
Results
The median D-dimer level was significantly higher (p < 0.001) for the patients with PJI than for the patients with aseptic failure. With a sensitivity of 80.77% (95% CI, 65.62 to 95.92%) and a specificity of 79.63% (95% CI, 68.89 to 90.37%), the diagnostic efficiency of D-dimer did not outperform serum CRP (with a sensitivity of 84.61% and specificity of 64.81%) and ESR (with a sensitivity of 73.08% and specificity of 90.47%).
Conclusions
Serum D-dimer as a marker for the diagnosis of PJI still requires more large-scale and detailed clinical trials.
Keywords: Periprosthetic joint infection, Biomarkers, D-dimer, Erythrocyte sedimentation rate, C-reactive protein
Background
Periprosthetic joint infection (PJI) after total hip arthroplasty (THA) or total knee arthroplasty (TKA) is one of the most dreadful complications and it has extremely negative effects on the physical, emotional, social, and economic aspects of a patient’s life [1, 2]. Currently, an absolute test for the diagnosis of PJI does not exist, compelling clinicians to rely on a combination of synovial fluid tests and serological markers [3]. Due to the lack of an absolute test, the Musculoskeletal Infection Society (MSIS) introduced a set of diagnostic criteria for PJI.
The MSIS guidelines include two major and six minor diagnostic criteria, with the latter involving measurements of serum C-reactive protein (CRP) level, erythrocyte sedimentation rate (ESR), synovial fluid white blood cell (WBC) count, and neutrophil differential, culture, and leukocyte esterase testing. Although numerous serological markers for PJI have been evaluated in the past, including interleukin 6 (IL-6), erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) have been generally used as a screening test for infection because of their simplicity and cost-effectiveness [4, 5]. However, they have low sensitivity and specificity and these may increase under several conditions in addition to infection [6, 7]. A recently introduced test of synovial fluid biomarker, namely alpha-defensin, has been reported to diagnose PJI with high sensitivity and specificity, but its optimal threshold for the diagnosis of PJI remains unknown and also it is expensive [8]. The combined measurement of synovial fluid alpha-defensin and CRP for the diagnosis of PJI demonstrated a sensitivity of 97% and a specificity of 100% [9]. However, it frequently occurs when either an inadequate amount of fluid is available to perform all tests or, worse, no fluid is retrieved from the joint [10]. Obtaining synovial fluid is invasive and painful to patients. In addition, there is a theoretical, yet real, concern for the introduction of infection into the joint and, in difficult aspirations, especially in the hip, contamination of the aspirated fluid may occur, leading to false-positive results [11].
The issues mentioned above highlight the requirement for a reliable serological test that can help to diagnose PJI. D-dimers are fibrin degradation products formed as a result of fibrin clot dissolution by plasmin. D-dimer assessment was traditionally used as a screening test for detecting venous thromboembolism (VTE) but was largely abandoned because of its poor accuracy [12]. More recently, assessment of serum D-dimer has gained attention for its role in predicting poor outcome in sepsis and bacteremia [13].
Therefore, in this study, we were interested in whether patients with PJI have a higher level of circulating D-dimer and whether D-dimer could be indicative of infection in patients suspected of PJI.
Methods and materials
Ethical approval for this prospective study was obtained from the Ethics Committee of our hospital. From April 2017 to August 2018, patients who had undergone primary total joint arthroplasty (TJA) and were then suspected of PJI were included in our study. Excluded were patients who have taken preoperative pharmacologic deep vein thrombosis (DVT) prophylaxis or suspicious DVT, with inflammatory arthritis such as rheumatoid arthritis, hematoma, a history of recent trauma or dislocation (within 2 weeks), visible ecchymosis, or a history of hypercoagulation disorder. Patients were divided into PJI group and non-PJI group based on the MSIS criteria. PJI is present when one of the major criteria exists or four out of six minor criteria exist: (1) elevated serum CRP and ESR (CRP > 10mg/d, ESR > 30 mm/h), (2) elevated synovial fluid white blood cell (WBC) count (> 3000 cells/μL), (3) elevated synovial fluid polymorphonuclear neutrophil percentage (PMN > 65%), (4) presence of purulence in the affected joint, (5) isolation of a microorganism in one periprosthetic tissue or fluid culture, and (6) > 5 neutrophils per high-powered field in 5 high-power fields observed from histologic analysis of periprosthetic tissue at × 400 magnification.
We recorded patients’ sex and age and the involved joint. Moreover, concurrent antibiotic treatment (excluding a single dose of prophylactic perioperative antibiotic) and isolated organisms were noted for all of the patients. Venous blood samples were obtained preoperatively on the day before surgery, which were then analyzed for serum D-dimer, ESR, and CRP. The D-dimer level in was assessed using an immunoturbidimetric assay on a STA-R analyzer (Diagnostica Stago).
Statistical analysis
Descriptive statistics were used to report all of the laboratory values. Comparison of ESR, CRP, and D-dimer between preoperative and postoperative values was performed using a paired t test. The optimal threshold for D-dimer as diagnostic of PJI was determined by the Youden J statistic (J = sensitivity + specificity − 1) based on its correspondence with the diagnosis of PJI. The sensitivity and specificity of the diagnostic tests were calculated along with their 95% confidence intervals (CIs). All statistical analyses were performed using GraphPad Prism (version 7.0a; GraphPad Software).
Results
Based on the MSIS criteria, 26 patients (14 knee replacement and 12 hip replacement) were diagnosed as the PJI group, while the other 54 patients (33 knee replacement and 21 hip replacement) were defined as the non-PJI group. In the PJI group, 7 were males and the other 19 were females, with age ranging from 41 to 78 (65.42 ± 10.8) and body mass index (BMI) of 20.20~34.74 kg/m2 (25.07 ± 5.32 kg/m2). Among the 54 uninfected patients, there were 25 males and 29 females, with age ranging from 42 to 76 (59.76 + 12.53) and BMI of 19.06~32.68 kg/m2 (22.87 ± 3.77 kg/m2). There was no statistical difference between these two groups in terms of age, gender, and BMI (p > 0.05).
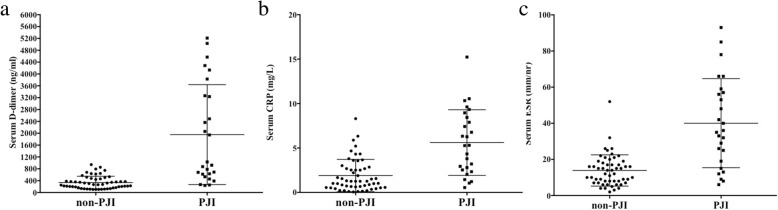
Before surgery, serum D-dimer level was significantly higher in patients with PJI; the mean D-dimer level was 1953.35 ng/mL (238 to 5211 ng/mL) in the PJI group, compared with 336.50 ng/mL (103 to 942 ng/mL) in the non-PJI group (p < 0.001) (Fig. 1a). The median ESR and CRP values were also significantly higher among the patients with PJI. The median ESR was 40.04 mm/h (6 to 93 mm/h) in the PJI group, compared with 13.87 mm/h (2 to 52 mm/h) in the non-PJI group (p < 0.001) (Fig. 1b). The mean CRP was 5.61 mg/L (0.50 to 15.23 mg/L) in the PJI group, compared with 1.9 mg/L (0.55 to 8.30 mg/L) in the non-PJI group (p < 0.001) (Fig. 1c).
Fig. 1.
Serum D-dimer (a), CRP (b), and ESR (c) significantly increased in PJI group, compared with the non-PJI group (p < 0.001)
Compared with the MSIS criteria, the serum D-dimer test demonstrated a sensitivity of 80.77% (95% CI, 65.62 to 95.92%) and a specificity of 79.63% (95% CI, 68.89 to 90.37%), for diagnosing PJI. Assessments of serum CRP level and ESR demonstrated a sensitivity of 84.61% (95% CI, 70.74 to 98.48%) and 73.08% (95% CI, 56.03 to 90.13%) and a specificity of 64.81% (95% CI, 52.41 to 74.81%) and 90.47% (95% CI, 82.74 to 98.47%), respectively (Table 1). With Youden’s index, 756 ng/ml was determined as the optimal threshold value for serum D-dimer for the diagnosis of PJI.
Table 1.
Performance of serum tests for diagnosing PJI
| D-dimer | CRP | ESR | |
|---|---|---|---|
| True positive | 21 | 22 | 19 |
| False positive | 11 | 19 | 5 |
| False negative | 5 | 4 | 7 |
| True negative | 43 | 35 | 49 |
| Sensitivity (95%CI) | 80.77% (65.62–95.92%) | 84.61% (70.74–98.48%) | 73.08% (56.03–90.13%) |
| Specificity (95%CI) | 79.63% (68.89 90.37%) | 64.81% (52.41–74.81%) | 90.47% (82.74–98.47%) |
| AUC (95%CI) | 0.890 (0.814–0.966) | 0.831 (0.737–0.926) | 0.838 (0.732–0.944) |
PPV positive predictive value, NPV negative predictive value, +LR positive likelihood ratio, −LR negative likelihood ratio
Discussion
Serum D-dimer is a fibrin degradation product released into the blood following the fibrin clot breakdown by plasmin and has been used for screening patients for venous thromboembolism (VTE) [14]. In recent years, evidence has emerged to suggest that D-dimer levels are likely to rise in the setting of systemic inflammation and infection, especially in the joint [13, 15, 16]. A persistent inflammatory response could contribute to a hypercoagulable state, possibly via cytokine-induced activation of the endothelium or by induction of monocytes to express tissue factor [17]. D-dimer and other fibrin degradation products may influence inflammatory and acute-phase responses by promoting neutrophil and monocyte activation [18]. In addition, it has been demonstrated that D-dimer levels increased in patients with rheumatoid arthritis (RA) and it might be considered as a biomarker for diagnosis of RA [19, 20]. Activation of the coagulation and fibrinolytic cascades in the joint and in the circulation could be observed in both inflammatory and degenerative joint diseases [21]. So et al. [22] demonstrated that within the joint, inflammatory mechanisms leading to the activation of the coagulation pathway and the increased amounts of thrombin-activated fibrinolysis inhibitor (TAFI) in RA might explain why fibrin formation is so prominent when compared with other joint diseases. The increased fibrinolytic activity and generation of byproducts such as D-dimer are considered to localize in the infecting organisms or inflammatory cells, thus preventing them from triggering systemic damage [23, 24]. The byproduct of this fibrinolytic activity also “leaks” into the circulation and can thus be measured [25]. Recently, D-dimer has shown its promise as a biomarker for the diagnosis of PJI, as well as timing of reimplantation [26].
From our study, we compared the diagnostic efficiency of serum D-dimer with CRP and ESR for PJI diagnosis. The sensitivity and specificity of D-dimer fall between the sensitivity and specificity of serum CRP and ESR through the evaluation of 80 patients suspected of PJI. In a prospective study conducted by Shahi et al. [26], they reported that serum D-dimer outperformed both ESR and serum CRP, with a sensitivity of 89.5% and specificity of 92.8%. They also believed that “elevated” D-dimer at the time of reimplantation could predict the infection, which caused subsequent failure. While for those false-positive results, they considered it as the infection caused by slow-growing organisms that did not elicit physiological inflammation and failed to meet the MSIS criteria for PJI. In another study conducted by Lee et al. [27], D-dimer was proved as effective in early detection of PJI if combined with levels of ESR and CRP. However, this study used the data of primary surgery and did not further compare the difference between postoperative infections with those who were not infected.
There are several limitations in our current study. Firstly, the number of patients included in this study was still limited due to the different hospital stays of patients. For some patients who were eligible for other inclusion criteria, we could not obtain the serum results of CRP, ESR, and D-dimer on the third or fifth day after surgery. Secondly, more postoperative data should be acquired to further validate the rise and fall tendency of these three tests.
Conclusions
In conclusion, we found that serum D-dimer assessment might be comparable with serum CRP and ESR for diagnosis of PJI. In addition, the rise and fall of serum D-dimer is more rapid than serum CRP and ESR postoperatively. This test might be used effectively in diagnosing early postoperative infection and this progress might be considered as a baseline for early diagnosis of infection. Further validation work is still required to reproduce these findings and confirm the relative test performance of D-dimer versus other more established serum markers.
Acknowledgements
None.
Abbreviations
- C-CRP
Reactive protein
- CIs
Confidence intervals
- ESR
Erythrocyte sedimentation rate
- MSIS
Musculoskeletal Infection Society
- PJI
Periprosthetic joint infection
- TAFI
Thrombin-activated fibrinolysis inhibitor
- THA
Total hip arthroplasty
- TKA
Total knee arthroplasty
- VTE
Venous thromboembolism
- WBC
White blood cell
Authors’ contributions
LX and SL carried out the studies, participated in collecting data, and drafted the manuscript. LX, MD, and SL performed the statistical analysis and participated in its design. MD and LX helped to draft the manuscript. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Ethical approval for this prospective study was obtained from the Ethics Committee of our hospital. Participants have provided their written informed consent to participate in this study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Longjiang Xiong, Email: xlj95196@163.com.
Siyun Li, Email: 653230771@qq.com.
Min Dai, Email: mindai2018@yeah.net.
References
- 1.Parvizi J, Cavanaugh PK, Diaz-Ledezma C. Periprosthetic knee infection: ten strategies that work. Knee Surg Relat Res. 2013;25(4):155–164. doi: 10.5792/ksrr.2013.25.4.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Parvizi J, Shohat N, Gehrke T. Prevention of periprosthetic joint infection: new guidelines. Bone Joint J. 2017;99-B(4 Supple B):3–10. doi: 10.1302/0301-620X.99B4.BJJ-2016-1212.R1. [DOI] [PubMed] [Google Scholar]
- 3.Parvizi J, Della Valle CJ. AAOS Clinical Practice Guideline: diagnosis and treatment of periprosthetic joint infections of the hip and knee. J Am Acad Orthop Surg. 2010;18(12):771–772. doi: 10.5435/00124635-201012000-00007. [DOI] [PubMed] [Google Scholar]
- 4.Deirmengian C, Kardos K, Kilmartin P, et al. Diagnosing periprosthetic joint infection: has the era of the biomarker arrived? Clin Orthop Relat Res. 2014;472(11):3254–3262. doi: 10.1007/s11999-014-3543-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kim TW, Kim DH, Oh WS, et al. Analysis of the causes of elevated C-reactive protein level in the early postoperative period after primary total knee arthroplasty. J Arthroplast. 2016;31(9):1990–1996. doi: 10.1016/j.arth.2016.02.037. [DOI] [PubMed] [Google Scholar]
- 6.Berbari E, Mabry T, Tsaras G, et al. Inflammatory blood laboratory levels as markers of prosthetic joint infection: a systematic review and meta-analysis. J Bone Joint Surg Am. 2010;92(11):2102–2109. doi: 10.2106/JBJS.I.01199. [DOI] [PubMed] [Google Scholar]
- 7.McArthur BA, Abdel MP, Taunton MJ, et al. Seronegative infections in hip and knee arthroplasty: periprosthetic infections with normal erythrocyte sedimentation rate and C-reactive protein level. Bone Joint J. 2015;97-B(7):939–944. doi: 10.1302/0301-620X.97B7.35500. [DOI] [PubMed] [Google Scholar]
- 8.Wyatt MC, Beswick AD, Kunutsor SK, et al. The alpha-defensin immunoassay and leukocyte esterase colorimetric strip test for the diagnosis of periprosthetic infection: a systematic review and meta-analysis. J Bone Joint Surg Am. 2016;98(12):992–1000. doi: 10.2106/JBJS.15.01142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Deirmengian C, Kardos K, Kilmartin P, et al. Combined measurement of synovial fluid alpha-defensin and C-reactive protein levels: highly accurate for diagnosing periprosthetic joint infection. J Bone Joint Surg Am. 2014;96(17):1439–1445. doi: 10.2106/JBJS.M.01316. [DOI] [PubMed] [Google Scholar]
- 10.Poss R, Thornhill TS, Ewald FC, et al. Factors influencing the incidence and outcome of infection following total joint arthroplasty. Clin Orthop Relat Res. 1984;(182):117–26. [PubMed]
- 11.Ali F, Wilkinson JM, Cooper JR, et al. Accuracy of joint aspiration for the preoperative diagnosis of infection in total hip arthroplasty. J Arthroplast. 2006;21(2):221–226. doi: 10.1016/j.arth.2005.05.027. [DOI] [PubMed] [Google Scholar]
- 12.Bounameaux H, de Moerloose P, Perrier A, et al. Plasma measurement of D-dimer as diagnostic aid in suspected venous thromboembolism: an overview. Thromb Haemost. 1994;71(1):1–6. [PubMed] [Google Scholar]
- 13.Gris JC, Bouvier S, Cochery-Nouvellon E, et al. Fibrin-related markers in patients with septic shock: individual comparison of D-dimers and fibrin monomers impacts on prognosis. Thromb Haemost. 2011;106(6):1228–1230. doi: 10.1160/TH11-07-0489. [DOI] [PubMed] [Google Scholar]
- 14.Hansrani V, Khanbhai M, McCollum C. The diagnosis and management of early deep vein thrombosis. Adv Exp Med Biol. 2017;906:23–31. doi: 10.1007/5584_2016_103. [DOI] [PubMed] [Google Scholar]
- 15.Ribera T, Monreal L, Armengou L, et al. Synovial fluid D-dimer concentration in foals with septic joint disease. J Vet Intern Med. 2011;25(5):1113–1117. doi: 10.1111/j.1939-1676.2011.0758.x. [DOI] [PubMed] [Google Scholar]
- 16.Schwameis M, Steiner MM, Schoergenhofer C, et al. D-dimer and histamine in early stage bacteremia: a prospective controlled cohort study. Eur J Intern Med. 2015;26(10):782–786. doi: 10.1016/j.ejim.2015.10.024. [DOI] [PubMed] [Google Scholar]
- 17.Libby P, Simon DI. Inflammation and thrombosis: the clot thickens. Circulation. 2001;103(13):1718–1720. doi: 10.1161/01.CIR.103.13.1718. [DOI] [PubMed] [Google Scholar]
- 18.Chung S, Kim JE, Park S, et al. Neutrophil and monocyte activation markers have prognostic impact in disseminated intravascular coagulation: in vitro effect of thrombin on monocyte CD163 shedding. Thromb Res. 2011;127(5):450–456. doi: 10.1016/j.thromres.2010.12.021. [DOI] [PubMed] [Google Scholar]
- 19.Turk SM, Cansu DU, Teke HU, et al. Can we predict thrombotic tendency in rheumatoid arthritis? A thromboelastographic analysis (with ROTEM) Clin Rheumatol. 2018;37(9):2341–2349. doi: 10.1007/s10067-018-4134-y. [DOI] [PubMed] [Google Scholar]
- 20.Tan L, Wang Q, Zeng T, et al. Clinical significance of detecting HLA-DR, 14-3-3eta protein and d-dimer in the diagnosis of rheumatoid arthritis. Biomark Med. 2018;12(7):697–705. doi: 10.2217/bmm-2017-0371. [DOI] [PubMed] [Google Scholar]
- 21.Kwiecinski J, Josefsson E, Jin T. Fibrinolysis is down-regulated in mouse collagen-induced arthritis, but its normalization does not alleviate the course of disease. Inflamm Res. 2011;60(11):1021–1029. doi: 10.1007/s00011-011-0363-0. [DOI] [PubMed] [Google Scholar]
- 22.So AK, Varisco PA, Kemkes-Matthes B, et al. Arthritis is linked to local and systemic activation of coagulation and fibrinolysis pathways. J Thromb Haemost. 2003;1(12):2510–2515. doi: 10.1111/j.1538-7836.2003.00462.x. [DOI] [PubMed] [Google Scholar]
- 23.Stewart LK, Nordenholz KE, Courtney M, et al. Comparison of acute and convalescent biomarkers of inflammation in patients with acute pulmonary embolism treated with systemic fibrinolysis vs. placebo. Blood Coagul Fibrinolysis. 2017;28(8):675–680. doi: 10.1097/MBC.0000000000000669. [DOI] [PubMed] [Google Scholar]
- 24.Hofman Z, de Maat S, Hack CE, et al. Bradykinin: inflammatory product of the coagulation system. Clin Rev Allergy Immunol. 2016;51(2):152–161. doi: 10.1007/s12016-016-8540-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Di Minno MN, Iervolino S, Peluso R, et al. Hemostatic and fibrinolytic changes are related to inflammatory conditions in patients with psoriatic arthritis--effect of different treatments. J Rheumatol. 2014;41(4):714–722. doi: 10.3899/jrheum.130850. [DOI] [PubMed] [Google Scholar]
- 26.Shahi A, Kheir MM, Tarabichi M, et al. Serum D-dimer test is promising for the diagnosis of periprosthetic joint infection and timing of reimplantation. J Bone Joint Surg Am. 2017;99(17):1419–1427. doi: 10.2106/JBJS.16.01395. [DOI] [PubMed] [Google Scholar]
- 27.Lee YS, Lee YK, Han SB, et al. Natural progress of D-dimer following total joint arthroplasty: a baseline for the diagnosis of the early postoperative infection. J Orthop Surg Res. 2018;13(1):36. doi: 10.1186/s13018-018-0730-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.